Environmental Risk of Pesticides for Fish in Small- and Medium-Sized Streams of Switzerland
Abstract
1. Introduction
2. Materials and Methods
2.1. Analytical Data and Sampling Design
2.2. Risk Assessment—Single Substances
2.3. Risk Assessment—Mixtures
2.4. Identification of Fish-Relevant Pesticides
2.5. Review of Non-Apical Sublethal Effect Data
2.6. Vulnerability of Indigenous Fish Species
3. Results and Discussion
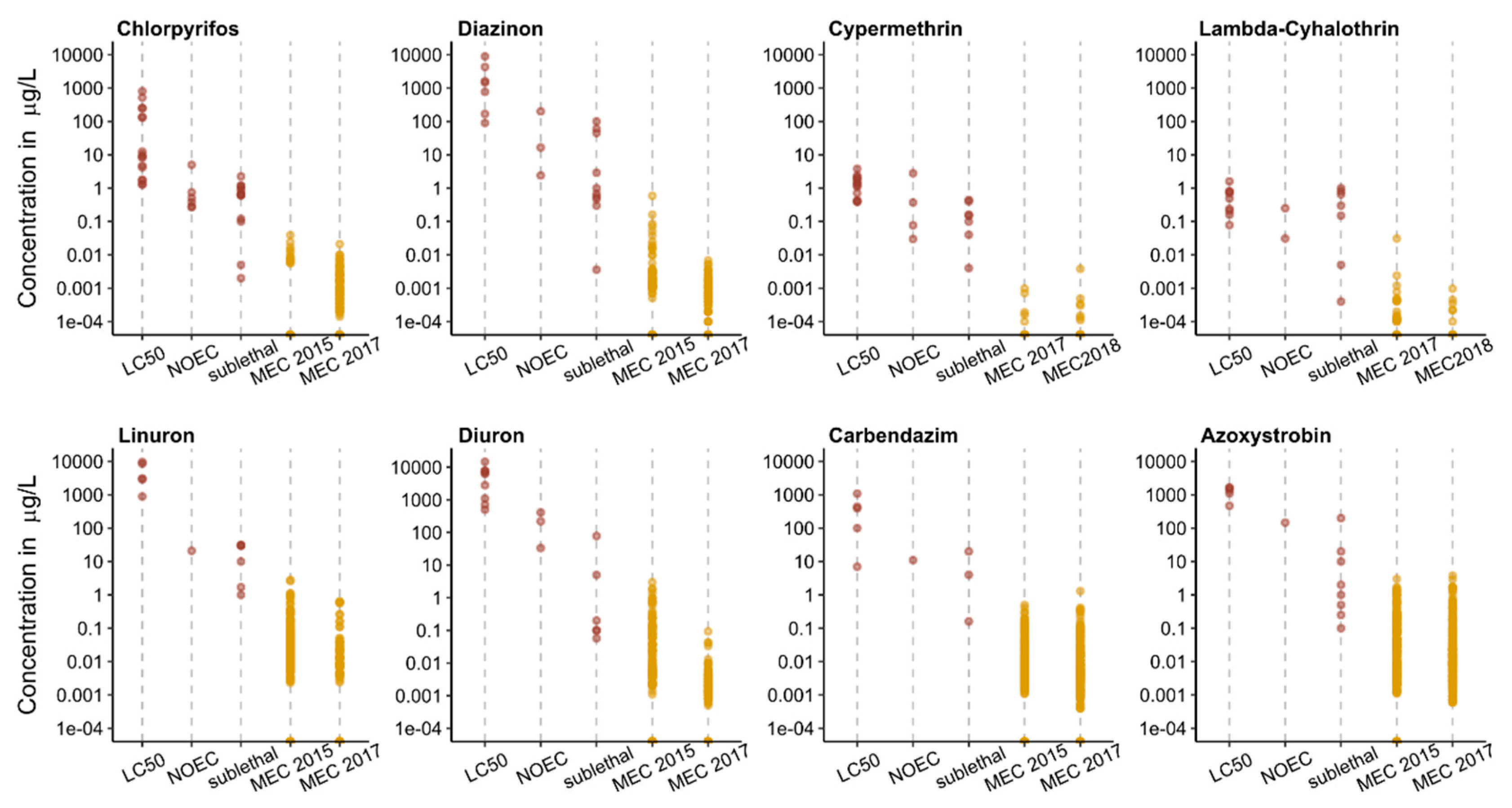
3.1. Single Substance Risk
3.2. Mixture Risk
3.3. Sublethal Effects
3.4. Vulnerability of Resident Fish Species
3.5. Uncertainties and Limitations of Our Risk Assessment
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Norman, J.E.; Mahler, B.J.; Nowell, L.H.; Van Metre, P.C.; Sandstrom, M.W.; Corbin, M.A.; Qian, Y.; Pankow, J.F.; Luo, W.; Fitzgerald, N.B.; et al. Daily stream samples reveal highly complex pesticide occurrence and potential toxicity to aquatic life. Sci. Total Environ. 2020, 715, 136795. [Google Scholar] [CrossRef]
- Stehle, S.; Buba, S.; Schulz, R. Compilation and analysis of global surface water concentrations for individual insecticide compounds. Sci. Total Environ. 2018, 639, 516–525. [Google Scholar] [CrossRef] [PubMed]
- Budd, R.; Wang, D.; Ensminger, M.; Phillips, B. An evaluation of temporal and spatial trends of pyrethroid concentrations in California surface waters. Sci. Total Environ. 2020, 718, 137402. [Google Scholar] [CrossRef] [PubMed]
- Moschet, C.; Wittmer, I.; Simovic, J.; Junghans, M.; Piazzoli, A.; Singer, H.; Stamm, C.; Leu, C.; Hollender, J. How a Complete Pesticide Screening Changes the Assessment of Surface Water Quality. Environ. Sci. Technol. 2014, 48, 5423–5432. [Google Scholar] [CrossRef]
- Spycher, S.; Mangold, S.; Doppler, T.; Junghans, M.; Wittmer, I.; Stamm, C.; Singer, H. Pesticide risks in small streams: How to get as close as possible to the stress imposed on aquatic organisms. Environ. Sci. Technol. 2018, 52, 4526–4535. [Google Scholar] [CrossRef]
- Spycher, S.; Teichler, R.; Vonwyl, E.; Longrée, P.; Stamm, C.; Singer, H.; Daouk, S.; Doppler, T.; Junghans, M.; Kunz, M. Anhaltend hohe PSM-Belastung in Bächen. Aqua Gas 2019, 4, 14–25. [Google Scholar]
- Rösch, A.; Beck, B.; Hollender, J.; Singer, H. Picogram per liter quantification of pyrethroid and organophosphate insecticides in surface waters: A result of large enrichment with liquid–liquid extraction and gas chromatography coupled to mass spectromtry using atmospheric pressure chemical Ionization. Anal. Bioanal. Chem. 2019, 411, 3151–3164. [Google Scholar] [CrossRef] [PubMed]
- Rösch, A.; Beck, B.; Hollender, J.; Stamm, C.; Singer, H.; Doppler, T.; Junghans, M. Geringe Konzentrationen mit grosser Wirkung: Nachweis von Pyrethroid-und Organophosphatinsektiziden in Schweizer Bächen im pg/l Bereich. Aqua Gas 2019, 11, 54–66. [Google Scholar]
- Langer, M.; Junghans, M.; Spycher, S.; Koster, M.; Baumgartner, C.; Vermeirssen, E.; Werner, I. Hohe ökotoxikologische Risiken in Bächen—NAWA SPEZ untersucht Bäche in Gebieten landwirtschaftlicher Nutzung. Aqua Gas 2017, 4, 58–68. [Google Scholar]
- Junghans, M.; Langer, M.; Baumgartner, C.; Vermeirssen, E.; Werner, I. Ökotoxikologische Risiken in Bächen durch Effekte aus Organismen bestätigt. Aqua Gas 2019, 4, 26–34. [Google Scholar]
- Moermond, C.T.; Kase, R.; Korkaric, M.; Ågerstrand, M. CRED: Criteria for reporting and evaluating ecotoxicity data. Environ. Toxicol. Chem. 2016, 35, 1297–1309. [Google Scholar] [CrossRef]
- EFSA. Conclusion on the peer review of epoxiconazole. EFSA Sci. Rep. 2008, 138, 1–80. [Google Scholar]
- EFSA. Conclusion on the peer review of the pesticide risk assessment of the active substance tebuconazole. EFSA J. 2014, 12, 3485. [Google Scholar]
- Sabra, F.S.; Mehana, E.S.E.D. Pesticides toxicity in fish with particular reference to insecticides. Asian J. Agric. Food Sci. 2015, 3, 40–60. [Google Scholar]
- Ullah, S.; Zorriehzahra, M.J. Ecotoxicology: A review of pesticides induced toxicity in fish. Adv. Anim. Vet. Sci. 2015, 3, 40–57. [Google Scholar] [CrossRef]
- Perez-Parada, A.; Goyenola, G.; Texeira de Mello, F.; Heinzen, H. Recent advances and open questions around pesticide dynamics and effects on freshwater fishes. Curr. Opin. Environ. Sci. Health 2018, 4, 38–44. [Google Scholar] [CrossRef]
- Bojarski, B.; Witeska, M. Blood biomarkers of herbicide, insecticide, and fungicide toxicity to fish—A review. Environ. Sci. Pollut. Res. 2020, 27, 19236–19250. [Google Scholar] [CrossRef] [PubMed]
- Werner, I.; Young, T. Pyrethroid insecticides—Exposure and impacts in the aquatic environment. In The Encyclopedia of the Anthropocene; DellaSala, D.A., Goldstein, M.I., Eds.; Elsevier: Oxford, UK, 2018; Volume 5, pp. 119–126. [Google Scholar] [CrossRef]
- Baldwin, D.H.; Spromberg, J.A.; Collier, T.K.; Scholz, N.L. A fish of many scales: Extrapolating sublethal pesticide exposures to the productivity of wild salmon populations. Ecol. Appl. 2009, 19, 2004–2015. [Google Scholar] [CrossRef] [PubMed]
- Sih, A.; Bell, A.V.; Kerby, J.L. Two stressors are deadlier than one. Trends Ecol. Evol. 2004, 19, 274–276. [Google Scholar] [CrossRef]
- Laetz, C.A.; Baldwin, D.H.; Collier, T.K.; Hebert, V.; Stark, J.D.; Scholz, N.L. The synergistic toxicity of pesticide mixtures: Implications for risk assessment and the conservation of endangered Pacific salmon. Environ. Health Persp. 2009, 117, 348–353. [Google Scholar] [CrossRef]
- Bacchetta, C.; Rossi, A.; Ale, A.; Campana, M.; Parma, M.J.; Cazenave, J. Combined toxicological effects of pesticides: A fish multi-biomarlker approach. Ecol. Indic. 2014, 36, 532–538. [Google Scholar] [CrossRef]
- Relyea, R.; Hoverman, J. Assessing the ecology in ecotoxicology: A review and synthesis in freshwater systems. Ecol. Lett. 2006, 9, 1157–1171. [Google Scholar] [CrossRef] [PubMed]
- Yamamuro, M.; Komuro, T.; Kamiya, H.; Kato, T.; Hasegawa, H.; Kameda, Y. Neonicotinoids disrupt aquatic food webs and decrease fishery yields. Science 2019, 366, 620–623. [Google Scholar] [CrossRef]
- Blackwell, B.R.; Ankley, G.T.; Bradley, P.M.; Houck, K.A.; Makarov, S.S.; Medvedev, A.V.; Swintek, J.; Villeneuve, D.L. Potential toxicity of complex mixtures in surface waters from a nationwide survey of US streams: Identifying in Vitro Bioactivities and Causative Chemicals. Environ. Sci. Technol. 2019, 53, 973–983. [Google Scholar] [CrossRef]
- Burkhardt-Holm, P.; Giger, W.; Güttinger, H.; Ochsenbein, U.; Peter, A.; Scheurer, K.; Segner, H.; Staub, E.; Suter, M.J.-F. Where have all the fish gone? Environ. Sci. Technol. 2005, 39, 441A–447A. [Google Scholar] [CrossRef] [PubMed]
- Brooks, M.L.; Fleishman, E.; Brown, L.; Lehman, P.; Werner, I.; Johnson, M.L.; Scholz, N.; Mitchelmore, C.; Parker, A.; Stoms, D.; et al. Life histories, salinity zones, and sublethal contributions of contaminants to pelagic fish declines illustrated with a case study of San Francisco Estuary, California, USA. Estuaries Coasts 2011, 35, 603–621. [Google Scholar] [CrossRef]
- Kirchhofer, A.; Breitenstein, M.; Zaugg, B. Rote Liste der Fische und Rundmauler der Schweiz; Umwelt-Vollzug Nr. 0734; Bundesamt für Umwelt, Bern, und Schweizer Zentrum für die Kartographie der Fauna: Neuenburg, Switzerland, 2007; p. 64. [Google Scholar]
- EU Commission. European Union Guideline for the Derivation of Environmental Quality Standards; Guidance Document No. 27; European Commission: Brussels, Belgium, 2011. [Google Scholar] [CrossRef]
- Ashauer, R.; Kuhl, R.; Zimmer, E.; Junghans, M. Effect modeling quantifies the difference between the toxicity of average pesticide concentrations and time-variable exposures from water quality monitoring. Environ. Toxicol. Chem. 2020, 39, 2158–2168. [Google Scholar] [CrossRef]
- Price, P.; Dhein, E.; Hamer, M.; Han, X.; Heneweer, M.; Junghans, M.; Kunz, P.; Magyar, C.; Penning, H.; Rodriguez, C. A decision tree for assessing effects from exposures to multiple substances. Environ. Sci. Eur. 2012, 24, 26. [Google Scholar] [CrossRef]
- Price, P.; Han, X.; Junghans, M.; Kunz, P.; Watts, C.; Leveret, D. An application of a decision tree for assessing effects from exposures to multiple substances to the assessment of human and ecological effects from combined exposures to chemicals observed in surface waters and wastewater effluents. Environ. Sci. Eur. 2012, 24, 34–47. [Google Scholar] [CrossRef][Green Version]
- Van Straalen, N.M. Biodiversity of ecotoxicological responses in animals. Neth. J. Zool. 1994, 44, 112–129. [Google Scholar] [CrossRef]
- Ibrahim, L.; Preuss, T.G.; Ratte, H.T.; Hommen, U. A list of fish species that are potentially exposed to pesticides in edge-of-field water bodies in the European Union—A first step towards identifying vulnerable representatives for risk assessment. Environ. Sci. Poll. Res. 2013, 20, 2679–2687. [Google Scholar] [CrossRef]
- Spalinger, L.; Dönni, W.; Guthruf, J. NAWA Trend Biologie: 2. Kampagne (2015) Fachbericht Fische Report; Federal Office for Environment: Bern, Switzerland, 2017; p. 75. [Google Scholar]
- EFSA. Guidance on tiered risk assessment for plant protection products for aquatic organisms in edge-of-field surface waters. EFSA J. 2013, 11, 3290. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020; Available online: https://R-project.org/ (accessed on 3 February 2021).
- Brady, J.A.; Wallender, W.W.; Werner, I.; Fard, B.M.; Zalom, F.G.; Oliver, M.N.; Wilson, B.W.; Mata, M.M.; Henderson, J.D.; Deanovic, L.A.; et al. Pesticide runoff from orchard floors: A comparative analysis of diazinon and esfenvalerate. Agric. Ecosyst. Environ. 2006, 115, 56–68. [Google Scholar] [CrossRef]
- Jorgenson, B.C.; Wissel-Tyson, C.; Young, T.M. Factors contributing to the off-target transport of pyrethroid insecticides from urban surfaces. J. Agric. Food Chem. 2012, 60, 7333–7340. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jorgenson, B.; Brown, L.; Macneale, K.; Schlenk, D.; Scholz, N.L.; Spromberg, J.; Werner, I.; Weston, D.; Young, T.; Zhang, M.; et al. Predicted transport of pyrethroid insecticides from an urban landscape to surface water. Environ. Toxicol. Chem. 2013, 32, 2469–2477. [Google Scholar] [CrossRef]
- Dax, A.; Stravs, M.; Stamm, C.; Ort, C.; La Cecilia, D.; Singer, H. MS2Field: Mikroverunreinigungen mobil messen. Zeitlich hochaufgelöste Messungen zeigen realistisches Ausmass akuter Gewässerbelastungen. Aqua Gas 2020, 12, 14. [Google Scholar]
- Federal Office for Agriculture. Zurückgezogene Wirkstoffe Aus Anhang 1 der PSMV. 2017. Available online: https://www.blw.admin.ch/blw/de/home/nachhaltige-produktion/pflanzenschutz/pflanzenschutzmittel/zugelassene-pflanzenschutzmittel.html (accessed on 28 January 2021).
- Jeffries, K.M.; Hinch, S.G.; Gale, M.K.; Clark, T.D.; Lotto, A.G.; Casselman, M.T.; Li, S.; Rechisky, E.L.; Porter, A.D.; Welch, D.W.; et al. Immune response genes and pathogen presence predict migration survival in wild salmon smolts. Mol. Ecol. 2014, 23, 5803–5815. [Google Scholar] [CrossRef]
- Hayes, T.B.; Hansen, M. From silent spring to silent night: Agrochemicals and the anthropocene. Elem. Sci. Anthr. 2017, 5, 57. [Google Scholar] [CrossRef]
- Scholz, N.L.; Fleishman, E.; Brown, L.; Brooks, M.L.; Mitchelmore, C.; Werner, I.; Johnson, M.L.; Schlenk, D. Pesticides and the decline of pelagic fishes in western North America’s largest estuarine ecosystem. BioScience 2012, 62, 428–434. [Google Scholar] [CrossRef]
- Fong, S.; Louie, S.; Werner, I.; Davis, J.; Connon, R.E. Contaminant Effects on California Bay–Delta Species and Human Health. San Fr. Estuary Watershed Sci. 2016, 14. Available online: http://escholarship.org/uc/item/52m780xj (accessed on 28 January 2021).
- Brander, S.M.; Gabler, M.K.; Fowler, N.L.; Connon, R.E.; Schlenk, D. Pyrethroid Pesticides as endocrine disruptors: Molecular mechanisms in vertebrates with a focus on fishes. Environ. Sci. Technol. 2016, 50, 8977–8992. [Google Scholar] [CrossRef] [PubMed]
- Vieira, C.E.D.; Dos Reis Martinez, C.B. The pyrethroid λ-cyhalothrin induces biochemical, genotoxic, and physiological alterations in the teleost Prochilodus lineatus. Chemosphere 2018, 210, 958–967. [Google Scholar] [CrossRef]
- Clifford, M.A.; Eder, K.J.; Werner, I.; Hedrick, R.P. Synergistic effects of esfenvalerate and infectious hematopoietic necrosis virus on juvenile Chinook salmon mortality. Environ. Toxicol. Chem. 2005, 24, 1766–1772. [Google Scholar] [CrossRef]
- Muranli, F.D.G.; Güner, U. Induction of micronuclei and nuclear abnormalities in erythrocytes of mosquito fish (Gambusia affinis) following exposure to the pyrethroid insecticide lambda-cyhalothrin. Mutat. Res. Genet. Toxicol. Environ. Mutagenesis 2011, 726, 104–108. [Google Scholar] [CrossRef]
- Tierney, K.B.; Ross, P.S.; Kennedy, C.J. Linuron and carbaryl differentially impair baseline amino acid and bile salt olfactory responses in three salmonids. Toxicology 2007, 231, 175–187. [Google Scholar] [CrossRef] [PubMed]
- Sandahl, J.F.; Baldwin, D.H.; Jenkins, J.J.; Scholz, N.L. Odor-evoked field potentials as indicators of sublethal neurotoxicity in juvenile coho salmon (Oncorhynchus kisutch) exposed to copper, chlorpyrifos, or esfenvalerate. Can. J. Fish. Aquat. Sci. 2004, 61, 404–413. [Google Scholar] [CrossRef]
- Sandahl, J.F.; Baldwin, D.H.; Jenkins, J.J.; Scholz, N.L. Comparative thresholds for acetylcholinesterase inhibition and behavioral impairment in coho salmon exposed to chlorpyrifos. Environ. Toxicol. Chem. 2005, 24, 136–145. [Google Scholar] [CrossRef]
- Moore, A.; Waring, C.P. Sublethal effects of the pesticide diazinon on olfactory function in mature male Atlantic salmon parr. J. Fish Biol. 1996, 48, 758–775. [Google Scholar] [CrossRef]
- Uren Webster, T.M.; Perry, M.H.; Santos, E.M. The herbicide linuron inhibits cholesterol biosynthesis and induces cellular stress responses in brown trout. Environ. Sci. Technol. 2015, 49, 3110–3118. [Google Scholar] [CrossRef]
- Saglio, P.; Trijasse, S. Behavioral responses to atrazine and diuron in goldfish. Arch. Environ. Contam. Toxicol. 1998, 35, 484–491. [Google Scholar] [CrossRef]
- Jiang, J.; Shi, Y.; Yu, R.; Chen, L.; Zhao, X. Biological response of zebrafish after short-term exposure to azoxystrobin. Chemosphere 2018, 202, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Andrade, T.S.; Henriques, J.; Almeida, A.; Machado, A.L.; Koba, O.; Giang, P.T.; Soares, A.M.V.M.; Domingues, I. Carbendazim exposure induces developmental, biochemical and behavioural disturbance in zebrafish embryos. Aquat. Toxicol. 2016, 170, 390–399. [Google Scholar] [CrossRef] [PubMed]
- Lange, K.; Bruder, A.; Matthaei, C.D.; Brodersen, J.; Paterson, R.A. Multiple-stressor effects on freshwater fish: Importance of taxonomy and life stage. Fish Fish. 2018, 19, 974–983. [Google Scholar] [CrossRef]
- Baird, D.J.; Van den Brink, P.J. Using biological traits to predict species sensitivity to toxic substances. Ecotox. Environ. Saf. 2007, 67, 296–302. [Google Scholar] [CrossRef]
- Segner, H. Moving beyond a descriptive aquatic toxicology: The value of biological process and trait information. Aquat. Toxicol. 2011, 105, 50–55. [Google Scholar] [CrossRef]
- Spurgeon, D.; Lahve, E.; Robinson, L.; Short, S.; Kille, P. Species sensitivity to toxic substances: Evolution, ecology and applications. Front. Environ. Sci. 2020, 8, 588380. [Google Scholar] [CrossRef]
- Schäfers, C.; Nagel, R. Fish toxicity and population dynamics: Effects of 3, 4-dichloroaniline and the problems of extrapolation. In Sublethal and Chronic Effects of Pollutants on Frehswater Fish; Müller, R., Lloyd, R., Eds.; Fishing News Books for FAOP; Blackwell Scientific Publisher: London, UK, 1994; Chapter 20; pp. 229–238. [Google Scholar]
- Spromberg, J.A.; Birge, W.S. Modelling the effect of chronic toxicity on fish populations: The influence of life history strategy. Enviro. Toxicol. Chem. 2000, 24, 1532–1540. [Google Scholar] [CrossRef]
- Segner, H. Reproductive and developmental toxicity in fishes. In Reproductive and Developmental Toxicology; Gupta, R.C., Ed.; Elsevier: Amsterdam, The Netherlands, 2011; pp. 1145–1166. [Google Scholar]
- Hutchinson, T.H.; Solbe, J.; Kloepper-Sams, P.J. Analysis of the ecetoc aquatic toxicity (EAT) database III—Comparative toxicity of chemical substances to different life stages of aquatic organisms. Chemosphere 1998, 36, 129–142. [Google Scholar] [CrossRef]
- Mayer, C.M.; Wahl, D.H. The relationship between prey selectivity and growth and survival in a larval fish. Can. J. Fish. Aquat. Sci. 1997, 54. [Google Scholar] [CrossRef]
- Sullivan, L.J.; Ignoffo, T.R.; Baskerville-Bridges, B.; Ostrach, D.J.; Kimmerer, W.J. Prey selection of larval and juvenile planktivorous fish: Impacts of introduced prey. Environ. Biol. Fishes 2016, 99, 633–646. [Google Scholar] [CrossRef]
- Boyle, T.P.; Fairchild, J.F.; Haverland, P.S.; Lebo, J.A.; Robinson-Wilson, E. Ecological restructuring in experimental aquatic mesocosms due to the application of diflubenzuron. Environ. Toxicol. Chem. 1996, 15, 1806–1814. [Google Scholar] [CrossRef]
- Kottelat, M.; Freyhof, J. Handbook of European Freshwater Fishes; Kottelat, Cornol and Freyhof: Berlin, Germany, 2007; 646p, ISBN 978-2-8399-0298-4. [Google Scholar]
- Riedl, C.; Peter, A. Timing of brown trout spawning in Alpine rivers with special consideration of egg burial depth. Ecol. Freshw. Fish 2013, 22, 384–397. [Google Scholar] [CrossRef]
- Birk, S.; Chapman, D.; Carvalho, L.; Spears, B.M.; Andersen, H.E.; Argillier, C.; Auer, S.; Baattrup-Pedersen, A.; Banin, L.; Beklioğlu, M.; et al. Impacts of multiple stressors on freshwater biota across spatial scales and ecosystems. Nat. Ecol. Evol. 2020, 4, 1060–1068. [Google Scholar] [CrossRef] [PubMed]
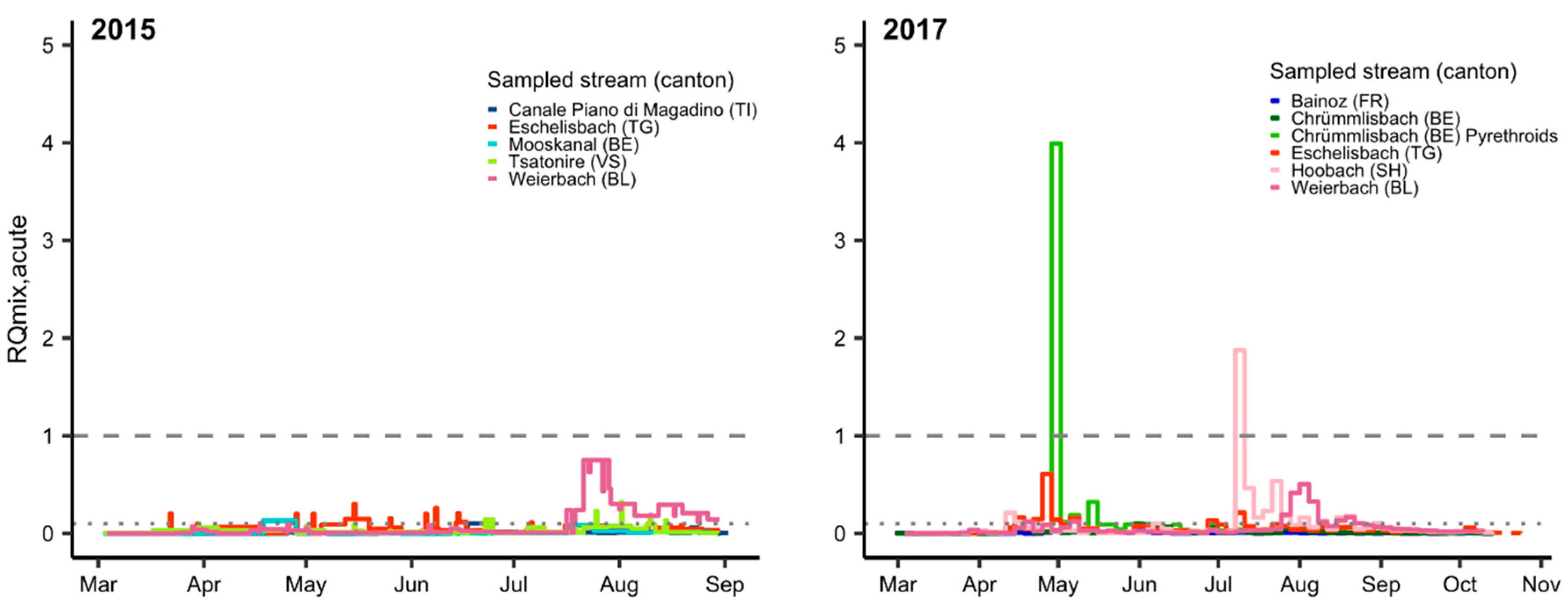
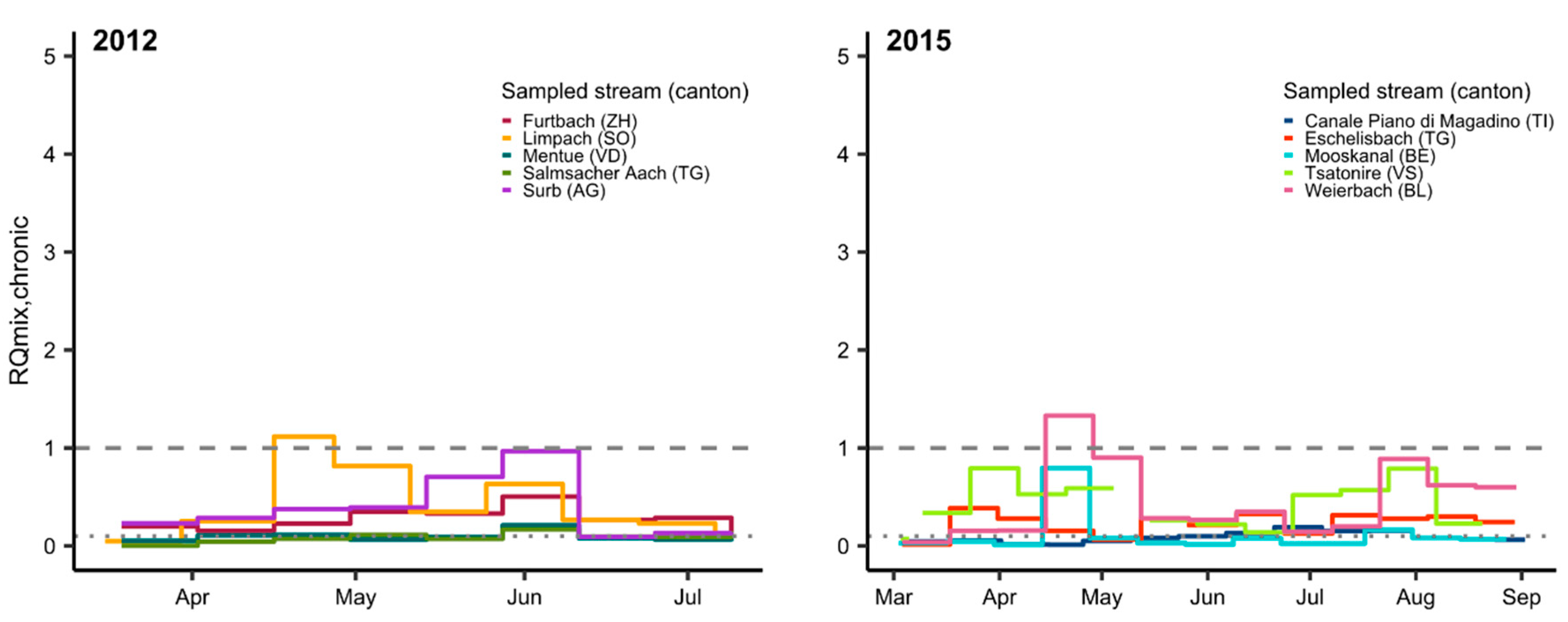
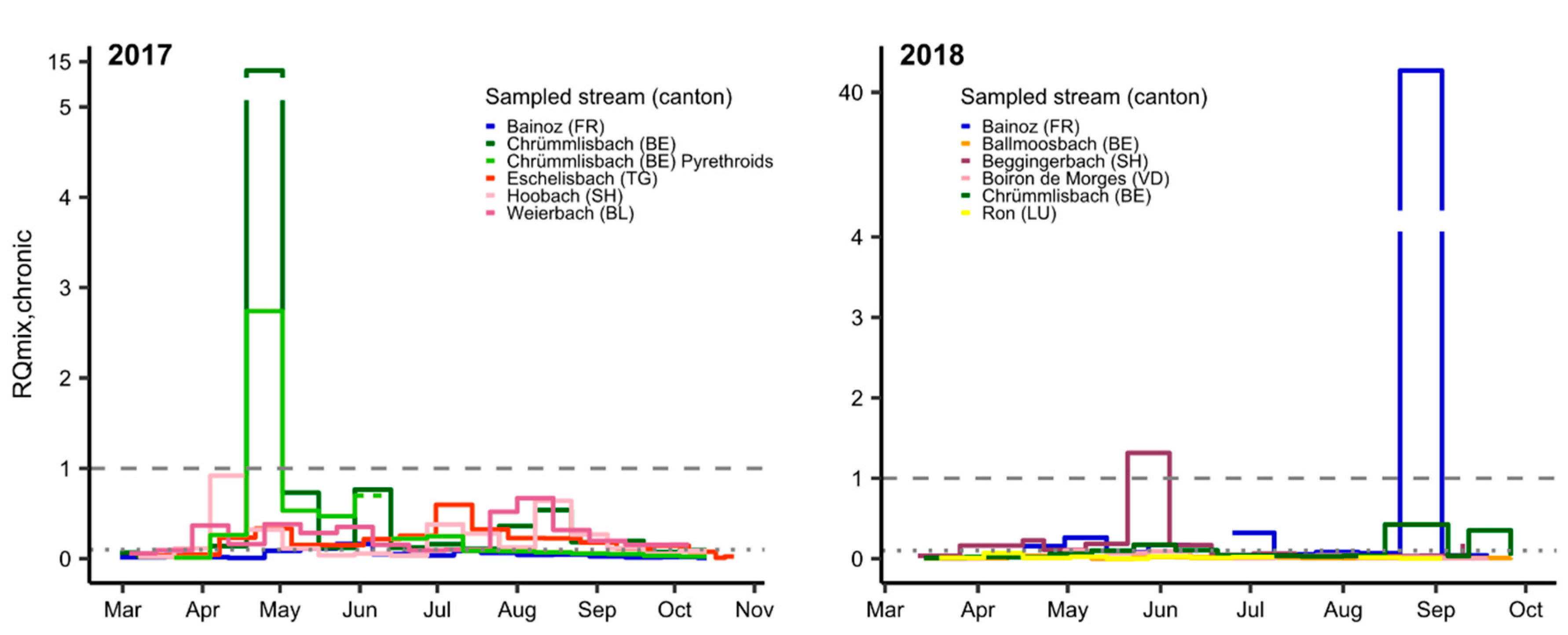
| (A) Substances with RQi ≥ 1 | Total Days of Exceedance (Number of Sites Affected) | ||||||
|---|---|---|---|---|---|---|---|
| 2012 | 2015 | 2017 a | 2018 b | ||||
| RQi | Chronic 1 | Acute 2 | Chronic | Acute | Chronic | Chronic | |
| λ-Cyhalothrin * (IN) | ≥0.1 ≥1.0 | NA | NA | NA | 10.5 (1) 3.5 (1) | 28 (1) 14 (1) | 42 (2) |
| Cypermethrin * (IN) | ≥0.1 ≥1.0 | NA | NA | NA | - | 14 (1) | 42 (1) 14 (1) |
| Deltamethrin * (IN) | ≥0.1 ≥1.0 | NA | NA | NA | - | - | 28 (1) 14 (1) |
| Carbendazim * (FU) | ≥0.1 ≥1.0 | - | 62(4) | 40.5 (1) | 77 (3) 3.5 (1) | 66.5 (3) | NA |
| Fenpropimorph * (FU) | ≥0.1 ≥1.0 | 53 (2) | - | - | - | 28 (1) 14 (1) | NA |
| (B) Substances with RQi ≥ 0.1 and <1 | |||||||
| Chlorpyrifos * (IN) | ≥0.1 | NA | 11 (3) | 70 (3) | 3.5 (1) | 63 (3) c | 84 (3) |
| Chlorpyrifos-methyl * (IN) | ≥0.1 | NA | 0.5 (1) | - | - | - | - |
| Diazinon * (IN) | ≥0.1 | 70 (2) | - | 28 (1) | - | - | NA |
| Permethrin * (IN) | ≥0.1 | NA | NA | NA | - | - | 14 (1) |
| S-Metolachlor * (HE) | ≥0.1 | 126 (3) | - | 56 (1) | - | - | NA |
| Linuron * (HE) | ≥0.1 | 14 (1) | - | 79.5(3) | - | 24.5 (1) | NA |
| Diuron * (HE) | ≥0.1 | - | - | 69.5(1) | - | - | NA |
| Aclonifen (HE) | ≥0.1 | - | - | 14 (1) | NA | NA | NA |
| Pendimethalin * (HE) | ≥0.1 | NA | NA | NA | - | 14 (1) | NA |
| Epoxiconazole * (FU) | ≥0.1 | 42 (2) | - | 54.5 (1) | - | 52.5 (2) | NA |
| Tebuconazole (FU) | ≥0.1 | - | - | - | - | 14 (1) | NA |
| Fluopyram * (FU) | ≥0.1 | NA | - | 40 (1) | - | 70 (1) | NA |
| Flusilazole (FU) | ≥0.1 | - | - | 26.5 (1) | - | 14 (1) | NA |
| Pyraclostrobin * (FU) | ≥0.1 | 28 (1) | - | - | - | - | NA |
| Spiroxamine * (FU) | ≥0.1 | - | - | 28 (1) | - | - | NA |
| Azoxystrobin (FU) | ≥0.1 | - | - | - | - | 14 (1) | NA |
| Fluazinam * (FU) | ≥0.1 | - | - | - | - | 14 (1) | NA |
| Fludioxonil (FU) | ≥0.1 | - | - | - | - | 14 (1) | NA |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Werner, I.; Schneeweiss, A.; Segner, H.; Junghans, M. Environmental Risk of Pesticides for Fish in Small- and Medium-Sized Streams of Switzerland. Toxics 2021, 9, 79. https://doi.org/10.3390/toxics9040079
Werner I, Schneeweiss A, Segner H, Junghans M. Environmental Risk of Pesticides for Fish in Small- and Medium-Sized Streams of Switzerland. Toxics. 2021; 9(4):79. https://doi.org/10.3390/toxics9040079
Chicago/Turabian StyleWerner, Inge, Anke Schneeweiss, Helmut Segner, and Marion Junghans. 2021. "Environmental Risk of Pesticides for Fish in Small- and Medium-Sized Streams of Switzerland" Toxics 9, no. 4: 79. https://doi.org/10.3390/toxics9040079
APA StyleWerner, I., Schneeweiss, A., Segner, H., & Junghans, M. (2021). Environmental Risk of Pesticides for Fish in Small- and Medium-Sized Streams of Switzerland. Toxics, 9(4), 79. https://doi.org/10.3390/toxics9040079








