In Vitro Fish Models for the Analysis of Ecotoxins and Temperature Increase in the Context of Global Warming
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Ecotoxicological Studies Regarding Climate Change
2.3. Image Analysis and Statistic
3. Results
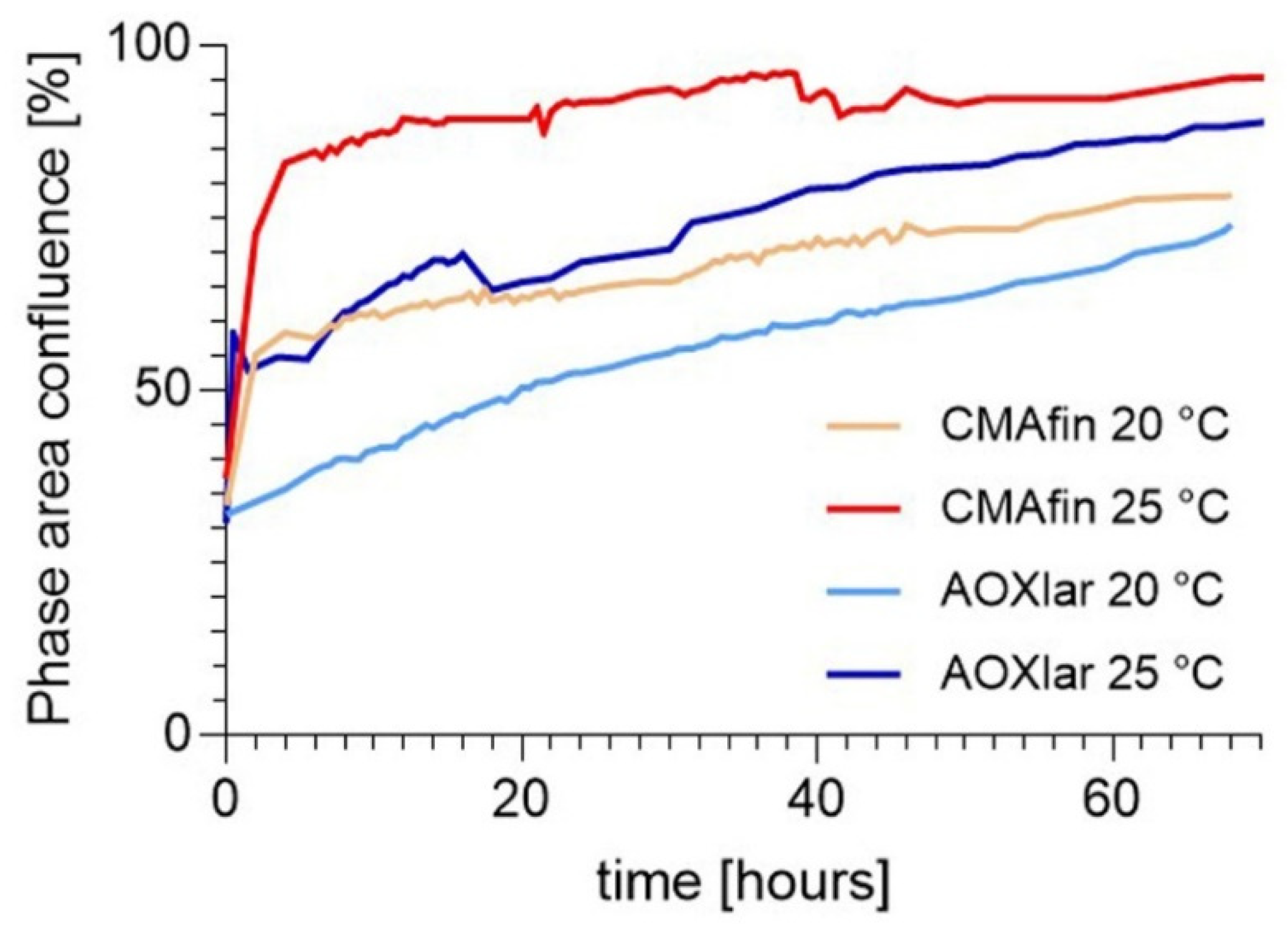
3.1. Faster Cell Proliferation at Higher Temperatures
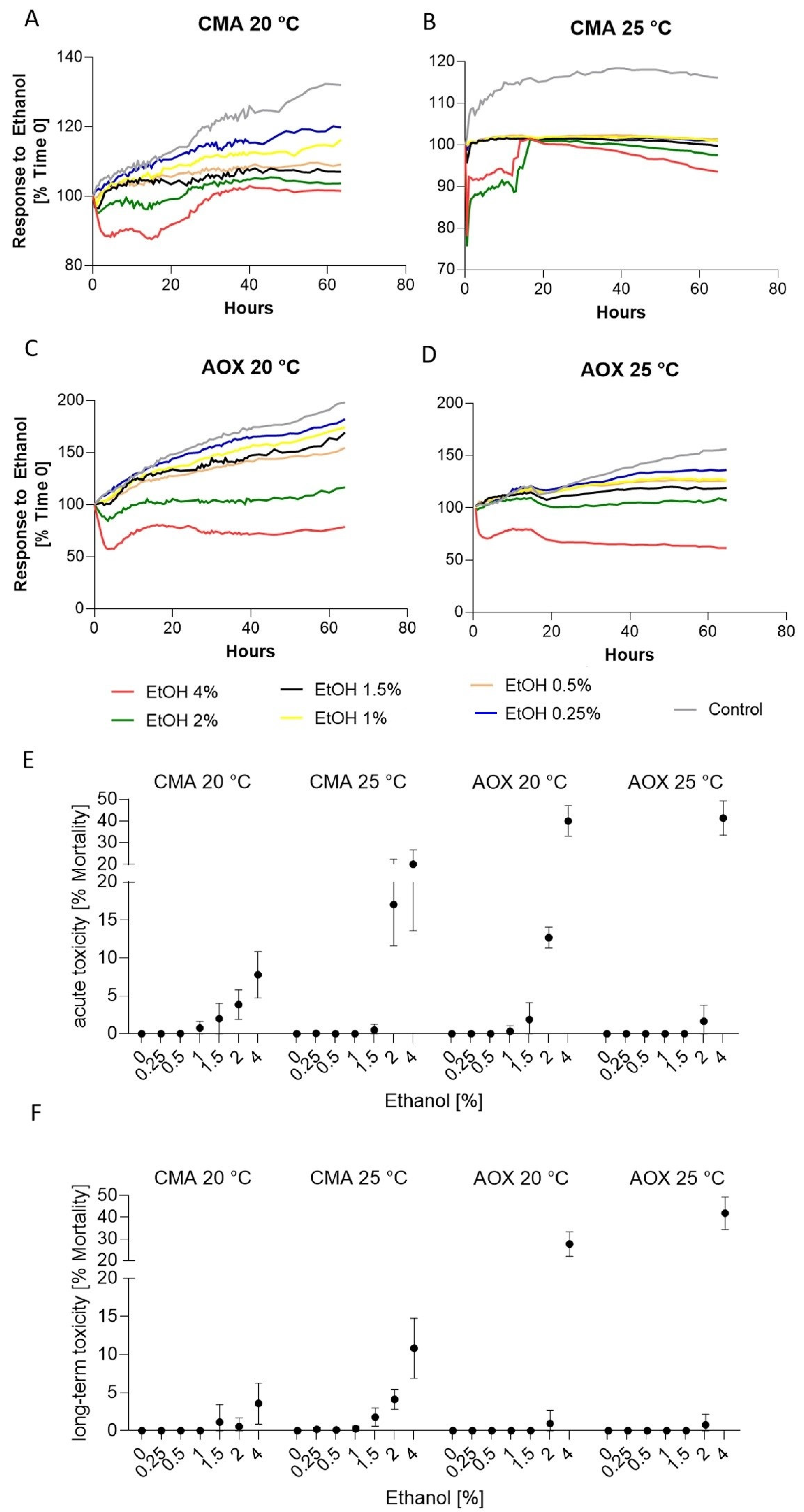
3.2. Response to Ethanol
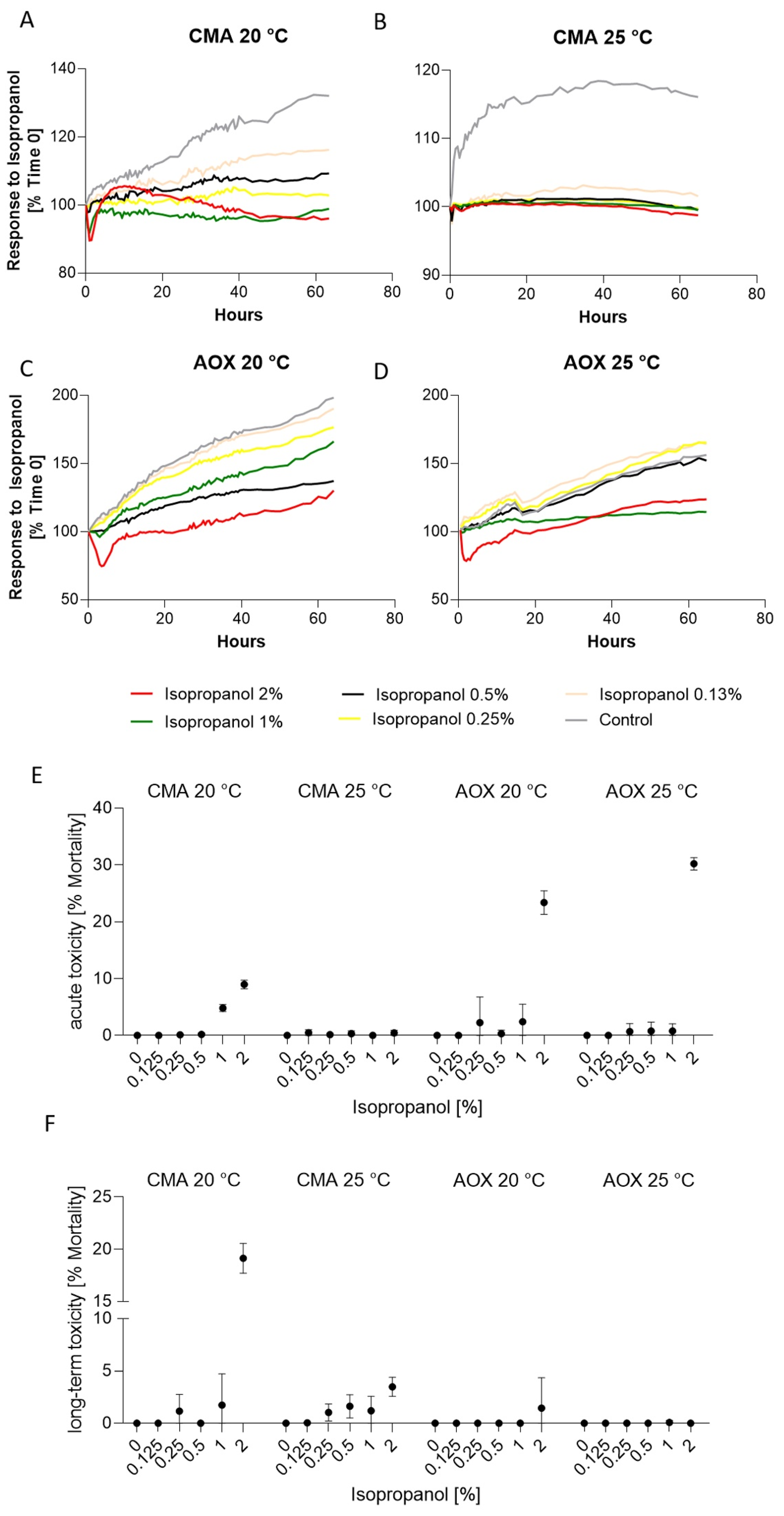
3.3. Response to Isopropanol
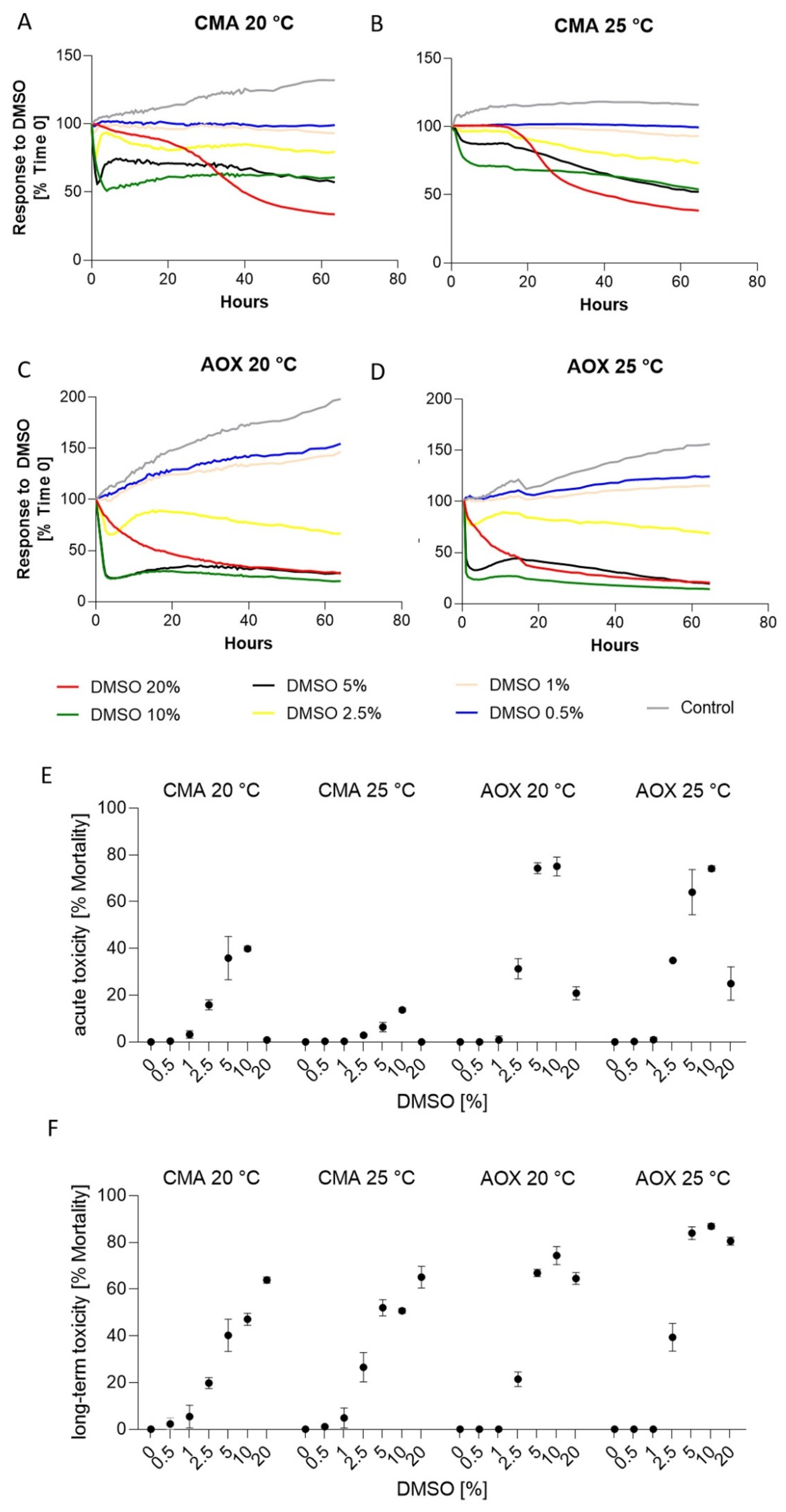
3.4. Response to Dimethyl Sulfoxide (DMSO)
4. Discussion
4.1. Effects of Temperature Increase
4.2. Effect of Ethanol and Isopropanol
4.3. Effects of DMSO
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lefevre, S.; Wang, T.; McKenzie, D.J. The role of mechanistic physiology in investigating impacts of global warming on fishes. J. Exp. Biol. 2021, 224 (Suppl. 1), jeb238840. [Google Scholar] [CrossRef]
- Farrell, A.P. Pragmatic perspective on aerobic scope: Peaking, plummeting, pejus and apportioning. J. Fish Biol. 2016, 88, 322–343. [Google Scholar] [CrossRef] [Green Version]
- Farrell, A.P.; Richards, J.G. Defining hypoxia: An integrative synthesis of the responses of fish to hypoxia. In Fish Physiology; Richards, J.G., Farrell, A.P., Brauner, C.J., Eds.; Academic Press: Cambridge, MA, USA, 2009; Volume 27, Chapter 11; pp. 487–503. [Google Scholar]
- Pörtner, H.O.; Knust, R. Climate Change Affects Marine Fishes through the Oxygen Limitation of Thermal Tolerance. Science 2007, 315, 95–97. [Google Scholar] [CrossRef] [Green Version]
- Sandström, A.; Karås, P. Effects of Eutrophication on Young-of-the-year Freshwater Fish Communities in Coastal Areas of the Baltic. Environ. Biol. Fishes 2002, 63, 89–101. [Google Scholar] [CrossRef]
- Bennett, E.M.; Carpenter, S.R.; Caraco, N.F. Human Impact on Erodable Phosphorus and Eutrophication: A Global Perspective: Increasing accumulation of phosphorus in soil threatens rivers, lakes, and coastal oceans with eutrophication. BioScience 2001, 51, 227–234. [Google Scholar] [CrossRef]
- Doney, S.C. The Growing Human Footprint on Coastal and Open-Ocean Biogeochemistry. Science 2010, 328, 1512–1516. [Google Scholar] [CrossRef] [Green Version]
- Brown, P.T.; Caldeira, K. Greater future global warming inferred from Earth’s recent energy budget. Nature 2017, 552, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Meehl, G.A.; Arblaster, J.M.; Bates, S.; Richter, J.H.; Tebaldi, C.; Gettelman, A.; Medeiros, B.; Bacmeister, J.; DeRepentigny, P.; Rosenbloom, N.; et al. Characteristics of Future Warmer Base States in CESM2. Earth Space Sci. 2020, 7, e2020EA001296. [Google Scholar] [CrossRef]
- Diaz, R.J.; Rosenberg, R. Sprading Dead Zones and Consequences for Marine Ecosystems. Science 2008, 321, 926–929. [Google Scholar] [CrossRef] [PubMed]
- Pörtner, H.O.; Farrell, A.P. Physiology and Climate Change. Science 2008, 322, 690–692. [Google Scholar] [CrossRef] [PubMed]
- Stuart-Smith, R.D.; Edgar, G.J.; Bates, A.E. Thermal limits to the geographic distributions of shallow-water marine species. Nat. Ecol. Evol. 2017, 1, 1846–1852. [Google Scholar] [CrossRef]
- Geraldi, N.R.; Kellison, G.T.; Bacheler, N.M. Climate Indices, Water Temperature, and Fishing Predict Broad Scale Variation in Fishes on Temperate Reefs. Front. Mar. Sci. 2019, 6, 30. [Google Scholar] [CrossRef]
- Dahlke, F.T.; Wohlrab, S.; Butzin, M.; Pörtner, H.-O. Thermal bottlenecks in the life cycle define climate vulnerability of fish. Science 2020, 369, 65–70. [Google Scholar] [CrossRef]
- Pankhurst, N.W.; Munday, P.L. Effects of climate change on fish reproduction and early life history stages. Mar. Freshw. Res. 2011, 62, 1015–1026. [Google Scholar] [CrossRef] [Green Version]
- Rehberger, K.; Kropf, C.; Segner, H. In vitro or not in vitro: A short journey through a long history. Environ. Sci. Eur. 2018, 30, 23. [Google Scholar] [CrossRef] [PubMed]
- Calow, P.; Forbes, V. Does ecotoxicology inform ecological risk assessment. Environ. Sci. Technol. 2003, 37, 146A–151A. [Google Scholar] [CrossRef] [Green Version]
- Segner, H.; Wenger, M.; Möller, A.M.; Köllner, B.; Casanova-Nakayama, A. Immunotoxic effects of environmental toxicants in fish—How to assess them? Environ. Sci. Pollut. Res. Int. 2011, 19, 2465–2476. [Google Scholar] [CrossRef] [PubMed]
- van Straalen, N.M. Ecotoxicology becomes stress ecology. Environ. Sci. Technol. 2003, 37, 324a–330a. [Google Scholar] [CrossRef] [Green Version]
- Fischer, M.; Belanger, S.E.; Berckmans, P.; Bernhard, M.J.; Bláha, L.; Coman Schmid, D.E.; Dyer, S.D.; Haupt, T.; Hermens, J.L.M.; Hultman, M.T.; et al. Repeatability and Reproducibility of the RTgill-W1 Cell Line Assay for Predicting Fish Acute Toxicity. Toxicol. Sci. 2019, 169, 353–364. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- ECHA. European Chemicals Agency. Available online: https://echa.europa.eu (accessed on 13 July 2021).
- Ceresana. 2021. Available online: https://www.ceresana.com/en/market-studies/chemicals/solvents/ (accessed on 24 September 2021).
- MassDep. 2011. Available online: https://www.mass.gov/doc/large-volume-ethanol-spills-environmental-impacts-response-options/download (accessed on 24 September 2021).
- NaBu. 2015. Available online: https://mecklenburg-vorpommern.nabu.de/natur-und-landschaft/naturschutz/mecklenburg-vorpommern/19480.html (accessed on 24 September 2021).
- Lee, P.A.; De Mora, S.J. Intracellular dimethylsulfoxide (DMSO) in unicellular marine algae: Speculations on its origin and possible biological role. J. Phycol. 1999, 35, 8–18. [Google Scholar] [CrossRef]
- Lee, P.A.; Mora, S.J.D.; Levasseur, M. A review of dimethylsulfoxide in aquatic environments. Atmosphere-Ocean 1999, 37, 439–456. [Google Scholar] [CrossRef]
- Zhao, Y.; Schlundt, C.; Booge, D.; Bange, H.W. A decade of dimethyl sulfide (DMS), dimethylsulfoniopropionate (DMSP) and dimethyl sulfoxide (DMSO) measurements in the southwestern Baltic Sea. Biogeosciences 2021, 18, 2161–2179. [Google Scholar] [CrossRef]
- Tanneberger, K.; Knöbel, M.; Busser, F.J.; Sinnige, T.L.; Hermens, J.L.; Schirmer, K. Predicting fish acute toxicity using a fish gill cell line-based toxicity assay. Environ. Sci. Technol. 2013, 47, 1110–1119. [Google Scholar] [CrossRef] [PubMed]
- OECD. Test No. 203: Fish, Acute Toxicity Test; OECD Publishing: Paris, France, 2005. [Google Scholar]
- Nelson, J.S.; Grande, T.C.; Wilson, M.V.H. Fishes of the World, 5th ed.; 2016; p. 752. Available online: https://www.wiley.com/en-ua/Fishes+of+the+World,+5th+Edition-p-9781118342336 (accessed on 13 July 2021).
- Bochert, R.; Luft, P. Combined effect of temperature and live feed period on growth and survival of Coregonus maraena (Bloch, 1779) larvae. Aquac. Res. 2019, 50, 2972–2977. [Google Scholar] [CrossRef]
- Jennerich, H.-J.; Schulz, N. Zur Situation des Ostseeschnäpels (Coregonus lavaretus Balticus, Thienemann, 1922). In Mecklenburg-Vorpommern; Landesforschungsanstalt für Landwirtschaft und Fischerei: Gülzow, Germany, 2009; pp. 12–20. [Google Scholar]
- Schulz, N. Das Wiedereinbürgerungs- und Besatzprogramm des Ostseeschnäpels Coregonus lavaretus Balticus (Thienemann) in der Vorpommerschen Boddenlandschaft, Rückblick und Ausblick; Fisch und Umwelt Mecklenburg-Vorpommern eV: Rostock, Germany, 2000; pp. 45–59. [Google Scholar]
- Bochert, R.; Luft, P.; Gebhard, R. Temperature preferences of three maraena whitefish (Coregonus maraena) size classes under aquaculture conditions. Aquac. Res. 2020, 51, 2160–2163. [Google Scholar] [CrossRef]
- Rebl, A.; Verleih, M.; Nipkow, M.; Altmann, S.; Bochert, R.; Goldammer, T. Gradual and Acute Temperature Rise Induces Crossing Endocrine, Metabolic, and Immunological Pathways in Maraena Whitefish (Coregonus maraena). Front. Genet. 2018, 9, 241. [Google Scholar] [CrossRef]
- Ficker, H.; Mazzucco, R.; Gassner, H.; Wanzenböck, J.; Dieckmann, U. Stocking strategies for a pre-alpine whitefish population under temperature stress. Ecol. Model. 2016, 320, 170–176. [Google Scholar] [CrossRef] [Green Version]
- Kottelat, M.; Freyhof, J. Handbook of European Fresh water Fishes; Publications Kottelat: Cornol, Switzerland, 2007. [Google Scholar]
- Chebanov, M.S.; Galich, E.V. Sturgeon Hatchery Manual; FAO: Ankara, Turkey, 2013; p. 297. [Google Scholar]
- Smith, C.L. National Audubon Society Field Guide to Tropical Marine Fishes of the Caribbean, the Gulf of Mexico, Florida, the Bahamas, and Bermuda; Alfred A. Knopf: New York, NY, USA, 1997; p. 720. [Google Scholar]
- Niklitschek, E.J.; Secor, D.H. Modeling spatial and temporal variation of suitable nursery habitats for Atlantic sturgeon in the Chesapeake Bay. Estuar. Coast. Shelf Sci. 2005, 64, 135–148. [Google Scholar] [CrossRef]
- Niklitschek, E.J.; Secor, D.H. Dissolved oxygen, temperature and salinity effects on the ecophysiology and survival of juvenile Atlantic sturgeon in estuarine waters: II. Model development and testing. J. Exp. Mar. Biol. Ecol. 2009, 381, S161–S172. [Google Scholar] [CrossRef]
- Niklitschek, E.J.; Secor, D.H. Experimental and field evidence of behavioural habitat selection by juvenile Atlantic Acipenser oxyrinchus oxyrinchus and shortnose Acipenser brevirostrum sturgeons. J. Fish Biol. 2010, 77, 1293–1308. [Google Scholar] [CrossRef]
- Larsson, S. Thermal preference of Arctic charr, Salvelinus alpinus, and brown trout, Salmo trutta—Implications for their niche segregation. Environ. Biol. Fishes 2005, 73, 89–96. [Google Scholar] [CrossRef]
- ASMFC. Habitat addendum IV to amendment 1 to the interstate fishery managment plan for Atlantic sturgeon. Manag. Plans FMP Rev. 2012, 16, 1–15. [Google Scholar]
- Mohler, J.W. Culture Manual for the Atlantic Sturgeon. Acipenser Oxyrinchus Oxyrinchus; U.S. Fish & Wildlife Service, Region 5: Washington, DC, USA, 2003; p. 68.
- Smith, T.I.J. The fishery, biology, and management of Atlantic sturgeon, Acipenser oxyrhynchus, in North America. Environ. Biol. Fishes 1985, 14, 61–72. [Google Scholar] [CrossRef]
- Kelly, J.L.; Arnold, D.E. Effects of Ration and Temperature on Growth of Age-0 Atlantic Sturgeon. N. Am. J. Aquac. 1999, 61, 51–57. [Google Scholar] [CrossRef]
- Smith, T.I.J.; Dingley, E.K.; Marchette, D.E. Induced Spawning and Culture of Atlantic Sturgeon. Progress. Fish-Cult. 1980, 42, 147–151. [Google Scholar]
- Grunow, B.; Noglick, S.; Kruse, C.; Gebert, M. Isolation of cells from Atlantic sturgeon Acipenser oxyrinchus oxyrinchus and optimization of culture conditions. Aquat. Biol. 2011, 14, 67–75. [Google Scholar] [CrossRef] [Green Version]
- Chapman, P.J.; James, D.I.; Watson, A.J.; Hopkins, G.V.; Waddell, I.D.; Ogilvie, D.J. Incucyte DRC: An R package for the dose response analysis of live cell imaging data. F1000Research 2016, 5, 962. [Google Scholar] [CrossRef] [Green Version]
- Tian, X.L.; Jiang, S.Y.; Zhang, X.L.; Yang, J.; Cui, J.H.; Liu, X.L.; Gong, K.R.; Yan, S.C.; Zhang, C.Y.; Shao, G. Potassium bisperoxo (1,10-phenanthroline) oxovanadate suppresses proliferation of hippocampal neuronal cell lines by increasing DNA methyltransferases. Neural Regen. Res. 2019, 14, 826–833. [Google Scholar]
- Lanigan, T.M.; Rasmussen, S.M.; Weber, D.P.; Athukorala, K.S.; Campbell, P.L.; Fox, D.A.; Ruth, J.H. Real time visualization of cancer cell death, survival and proliferation using fluorochrome-transfected cells in an IncuCyte® imaging system. J. Biol. Methods 2020, 7, e133. [Google Scholar] [CrossRef]
- Servili, A.; Canario, A.V.M.; Mouchel, O.; Muñoz-Cueto, J.A. Climate change impacts on fish reproduction are mediated at multiple levels of the brain-pituitary-gonad axis. Gen. Comp. Endocrinol. 2020, 291, 113439. [Google Scholar] [CrossRef]
- Domenici, P.; Allan, B.J.M.; Lefrançois, C.; McCormick, M.I. The effect of climate change on the escape kinematics and performance of fishes: Implications for future predator-prey interactions. Conserv. Physiol. 2019, 7, coz078. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alfonso, S.; Gesto, M.; Sadoul, B. Temperature increase and its effects on fish stress physiology in the context of global warming. J. Fish Biol. 2020, 98, 1496–1508. [Google Scholar] [CrossRef]
- Clarke, A.; Johnston, N.M. Scaling of metabolic rate with body mass and temperature in teleost fish. J. Anim. Ecol. 1999, 68, 893–905. [Google Scholar] [CrossRef]
- Arrhenius, S. Quantitative Laws in Biological Chemistry; Bell, G. and Sons, Ltd.: London, UK, 1915; p. 164. [Google Scholar]
- Boltzmann, L. Weitere Studien über das Wärmegleichgewicht unter Gasmolekülen; Akademie der Wissenschaften: Wien, Austria, 1872; pp. 275–370. [Google Scholar]
- Samaras, A.; Papandroulakis, N.; Lika, K.; Pavlidis, M. Water temperature modifies the acute stress response of European sea bass, Dicentrarchus labrax L. (1758). J. Therm. Biol. 2018, 78, 84–91. [Google Scholar] [CrossRef]
- Wendelaar Bonga, S.E. The stress response in fish. Physiol. Rev. 1997, 77, 591–625. [Google Scholar] [CrossRef]
- Sylvester, J.R. Possible effects of thermal effluents on fish: A review. Environ. Pollut. 1972, 3, 205–215. [Google Scholar] [CrossRef]
- van Denderen, D.; Gislason, H.; van den Heuvel, J.; Andersen, K.H. Global analysis of fish growth rates shows weaker responses to temperature than metabolic predictions. Glob. Ecol. Biogeogr. 2020, 29, 2203–2213. [Google Scholar] [CrossRef]
- Viadero, R.C. Factors Affecting Fish Growth and Production. In Water Encyclopedia; Lehr, J.H., Keeley, J., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2005; pp. 129–133. [Google Scholar]
- Brooke, L.T.; Call, D.J.; Geiger, D.L.; Northcott, C.E. Acute Toxicities of Organic Chemicals to Fathead Minnows (Pimephales promelas); Lake Superior Research Institute: Superior, WI, USA, 1984; Volume I, p. 436. [Google Scholar]
- ECHA. Ethanol. Short-Term Toxicity to Fish. Available online: https://echa.europa.eu/de/registration-dossier/-/registered-dossier/16105/6/2/2 (accessed on 13 July 2021).
- ECHA. Propan-2-ol. Short-Term Toxicity to Fish. Available online: https://echa.europa.eu/de/registration-dossier/-/registered-dossier/15339/6/2/2 (accessed on 13 July 2021).
- Vera, L.M.; Bello, C.; Paredes, J.F.; Carmona-Antoñanzas, G.; Sánchez-Vázquez, F.J. Ethanol toxicity differs depending on the time of day. PLoS ONE 2018, 13, e0190406. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- ECHA. Dimethyl Sulfoxide. Endpoint Summary. Available online: https://echa.europa.eu/de/registration-dossier/-/registered-dossier/15007/6/2/1 (accessed on 13 July 2021).
- Elmoazzen, H.Y.; Poovadan, A.; Law, G.K.; Elliott, J.A.; McGann, L.E.; Jomha, N.M. Dimethyl sulfoxide toxicity kinetics in intact articular cartilage. Cell Tissue Bank. 2007, 8, 125–133. [Google Scholar] [CrossRef]
- Jamalzadeh, L.; Ghafoori, H.; Sariri, R.; Rabuti, H.; Nasirzade, J.; Hasani, H.; Aghamaali, M.R. Cytotoxic Effects of Some Common Organic Solvents on MCF-7, RAW-264.7 and Human Umbilical Vein Endothelial Cells. Avicenna J. Med. Biochem. 2016, 4, 10–33453. [Google Scholar] [CrossRef] [Green Version]
- Willford, W.A. Toxicity of Dimethyl Sulfoxide (DMSO) to Fish; Service 1967, Report 20; U.S. Fish Wildlife: Washington, DC, USA, 1967; p. 8.
- Benville, P.E.; Smith, C.E.; Shanks, W.E. Some toxic effects of dimethyl sulfoxide in salmon and trout. Toxicol. Appl. Pharmacol. 1968, 12, 156–178. [Google Scholar] [CrossRef]
- Bols, N.C.; Boliska, S.A.; Dixon, D.G.; Hodson, P.V.; Kaiser, K.L.E. The use of fish cell cultures as an indication of contaminant toxicity to fish. Aquat. Toxicol. 1985, 6, 147–155. [Google Scholar] [CrossRef]
- Vinken, M.; Blaauboer, B.J. In vitro testing of basal cytotoxicity: Establishment of an adverse outcome pathway from chemical insult to cell death. Toxicol Vitr. 2017, 39, 104–110. [Google Scholar] [CrossRef] [Green Version]

| Ethanol [%] | CMA 20 °C (Compared to the Control) | CMA 25 °C (Compared to the Control) | CMA 20 °C vs. CMA 25 °C | AOX 20 °C (Compared to the Control) | AOX 25 °C (Compared to the Control) | AOX 20 °C vs. AOX 25 °C | CMA vs. AOX at 20 °C | CMA vs. AOX at 25 °C | |
|---|---|---|---|---|---|---|---|---|---|
| acute toxicity | 0.25 | >0.999 | >0.999 | 0.251 | >0.999 | >0.999 | >0.999 | >0.999 | 0.251 |
| 0.5 | >0.999 | >0.999 | 0.419 | >0.999 | >0.999 | >0.999 | 0.252 | 0.356 | |
| 1.0 | 0.936 | >0.999 | 0.141 | >0.999 | >0.999 | 0.356 | 0.511 | >0.999 | |
| 1.5 | 0.16 | 0.514 | 0.217 | >0.999 | >0.999 | 0.135 | 0.951 | 0.256 | |
| 2.0 | 0.036 | 0.022 | 0.006 | 0.051 | 0.464 | <0.0001 | <0.0001 | 0.003 | |
| 4.0 | 0.002 | 0.026 | 0.028 | 0.004 | 0.005 | 0.845 | <0.0001 | 0.100 | |
| long-term toxicity | 0.25 | >0.999 | >0.999 | 0.172 | >0.999 | >0.999 | >0.999 | >0.999 | 0.172 |
| 0.5 | >0.999 | >0.999 | 0.153 | >0.999 | >0.999 | >0.999 | >0.999 | 0.153 | |
| 1.0 | >0.999 | >0.999 | 0.192 | >0.999 | >0.999 | >0.999 | >0.999 | 0.192 | |
| 1.5 | >0.999 | 0.11 | 0.623 | >0.999 | >0.999 | >0.999 | 0.356 | 0.024 | |
| 2.0 | >0.999 | 0.015 | 0.006 | 0.904 | >0.999 | 0.890 | 0.707 | 0.022 | |
| 4.0 | 0.028 | 0.001 | 0.023 | 0.002 | 0.001 | 0.023 | <0.0001 | <0.0001 | |
| Isopropanol [%] | CMA 20 °C (Compared to the Control) | CMA 25 °C (Compared to the Control) | CMA 20 °C vs. CMA 25 °C | AOX 20 °C (Compared to the Control) | AOX 25 °C (Compared to the Control) | AOX 20 °C vs. AOX 25 °C | CMA vs. AOX at 20 °C | CMA vs. AOX at 25 °C | |
|---|---|---|---|---|---|---|---|---|---|
| acute toxicity | 0.125 | >0.999 | 0.207 | 0.099 | >0.999 | >0.999 | >0.999 | >0.999 | 0.099 |
| 0.25 | >0.999 | >0.999 | 0.725 | >0.999 | >0.999 | 0.534 | 0.377 | 0.469 | |
| 0.5 | 0.904 | 0.639 | 0.672 | >0.999 | >0.999 | 0.600 | 0.728 | 0.593 | |
| 1.0 | 0.018 | >0.999 | <0.0001 | >0.999 | >0.999 | 0.364 | 0.176 | 0.271 | |
| 2.0 | 0.016 | 0.146 | 0.001 | 0.007 | 0.016 | 0.025 | 0.003 | <0.0001 | |
| long-term toxicity | 0.125 | >0.999 | >0.999 | 0.198 | >0.999 | >0.999 | >0.999 | >0.999 | 0.198 |
| 0.25 | 0.874 | 0.361 | 0.888 | >0.999 | >0.999 | >0.999 | 0.195 | 0.044 | |
| 0.5 | >0.999 | 0.065 | 0.026 | >0.999 | >0.999 | >0.999 | 0.356 | 0.026 | |
| 1.0 | >0.999 | 0.172 | 0.326 | >0.999 | 0.416 | 0.356 | 0.203 | 0.165 | |
| 2.0 | 0.018 | 0.004 | 0.001 | 0.416 | >0.999 | 0.356 | 0.001 | 0.001 | |
| DMSO [%] | CMA 20 °C (Compared to the Control) | CMA 25 °C (Compared to the Control) | CMA 20 °C vs. CMA 25 °C | AOX 20 °C (Compared to the Control) | AOX 25 °C (Compared to the Control) | AOX 20 °C vs. AOX 25 °C | CMA vs. AOX at 20 °C | CMA vs. AOX at 25 °C | |
|---|---|---|---|---|---|---|---|---|---|
| acute toxicology | 0.5 | >0.999 | >0.999 | 0.922 | >0.999 | >0.999 | 0.356 | 0.280 | 0.658 |
| 1.0 | 0.398 | >0.999 | 0.012 | >0.999 | >0.999 | 0.904 | 0.086 | 0.271 | |
| 2.5 | 0.043 | 0.206 | 0.000 | 0.179 | 0.334 | 0.333 | 0.001 | <0.0001 | |
| 5.0 | 0.001 | 0.021 | 0.001 | 0.006 | 0.015 | 0.085 | <0.0001 | <0.0001 | |
| 10.0 | 0.004 | 0.002 | <0.0001 | 0.006 | 0.002 | 0.669 | <0.0001 | <0.0001 | |
| 20.0 | >0.999 | >0.999 | 0.016 | 0.724 | 0.515 | 0.329 | <0.0001 | 0.001 | |
| long term toxicology | 0.5 | >0.999 | >0.999 | 0.438 | >0.999 | >0.999 | >0.999 | 0.134 | 0.003 |
| 1.0 | >0.999 | >0.999 | 0.858 | >0.999 | >0.999 | >0.999 | 0.064 | 0.064 | |
| 2.5 | 0.293 | 0.233 | 0.092 | 0.912 | >0.999 | 0.003 | 0.436 | 0.041 | |
| 5.0 | 0.033 | 0.008 | 0.023 | 0.038 | 0.027 | <0.0001 | <0.0001 | <0.0001 | |
| 10.0 | 0.019 | 0.018 | 0.043 | 0.002 | 0.003 | 0.001 | <0.0001 | <0.0001 | |
| 20.0 | 0.001 | 0.000 | 0.629 | 0.135 | 0.215 | <0.0001 | 0.632 | 0.001 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grunow, B.; Franz, G.P.; Tönißen, K. In Vitro Fish Models for the Analysis of Ecotoxins and Temperature Increase in the Context of Global Warming. Toxics 2021, 9, 286. https://doi.org/10.3390/toxics9110286
Grunow B, Franz GP, Tönißen K. In Vitro Fish Models for the Analysis of Ecotoxins and Temperature Increase in the Context of Global Warming. Toxics. 2021; 9(11):286. https://doi.org/10.3390/toxics9110286
Chicago/Turabian StyleGrunow, Bianka, George Philipp Franz, and Katrin Tönißen. 2021. "In Vitro Fish Models for the Analysis of Ecotoxins and Temperature Increase in the Context of Global Warming" Toxics 9, no. 11: 286. https://doi.org/10.3390/toxics9110286
APA StyleGrunow, B., Franz, G. P., & Tönißen, K. (2021). In Vitro Fish Models for the Analysis of Ecotoxins and Temperature Increase in the Context of Global Warming. Toxics, 9(11), 286. https://doi.org/10.3390/toxics9110286







