Health Impacts and Biomarkers of Prenatal Exposure to Methylmercury: Lessons from Minamata, Japan
Abstract
1. Methylmercury Toxicity
2. History and Background of Minamata Disease
3. Methylmercury Pollution in the Minamata Area According to Analyses of Preserved Umbilical Cords
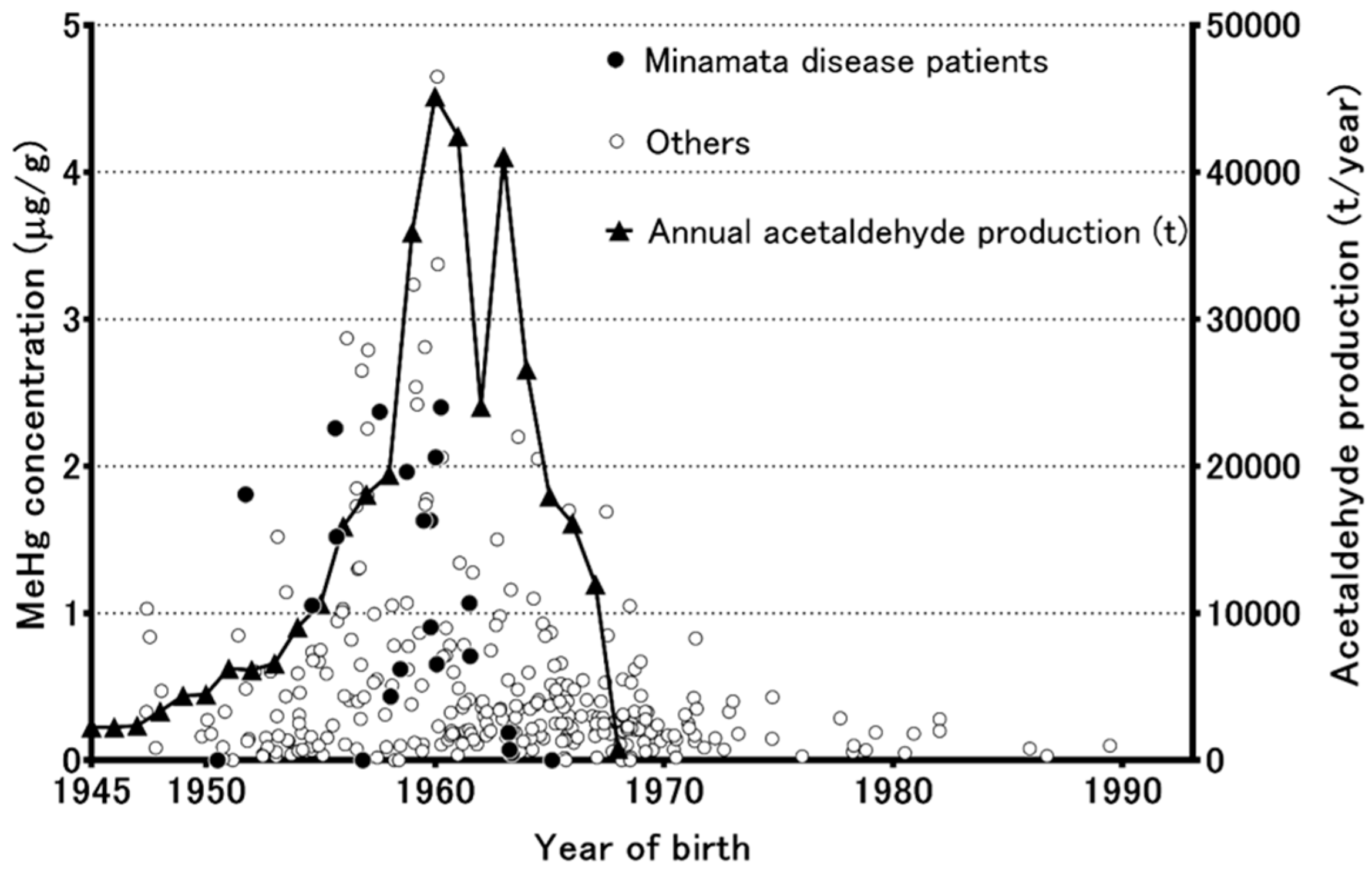
4. Changes in the Sex Ratio in Minamata City
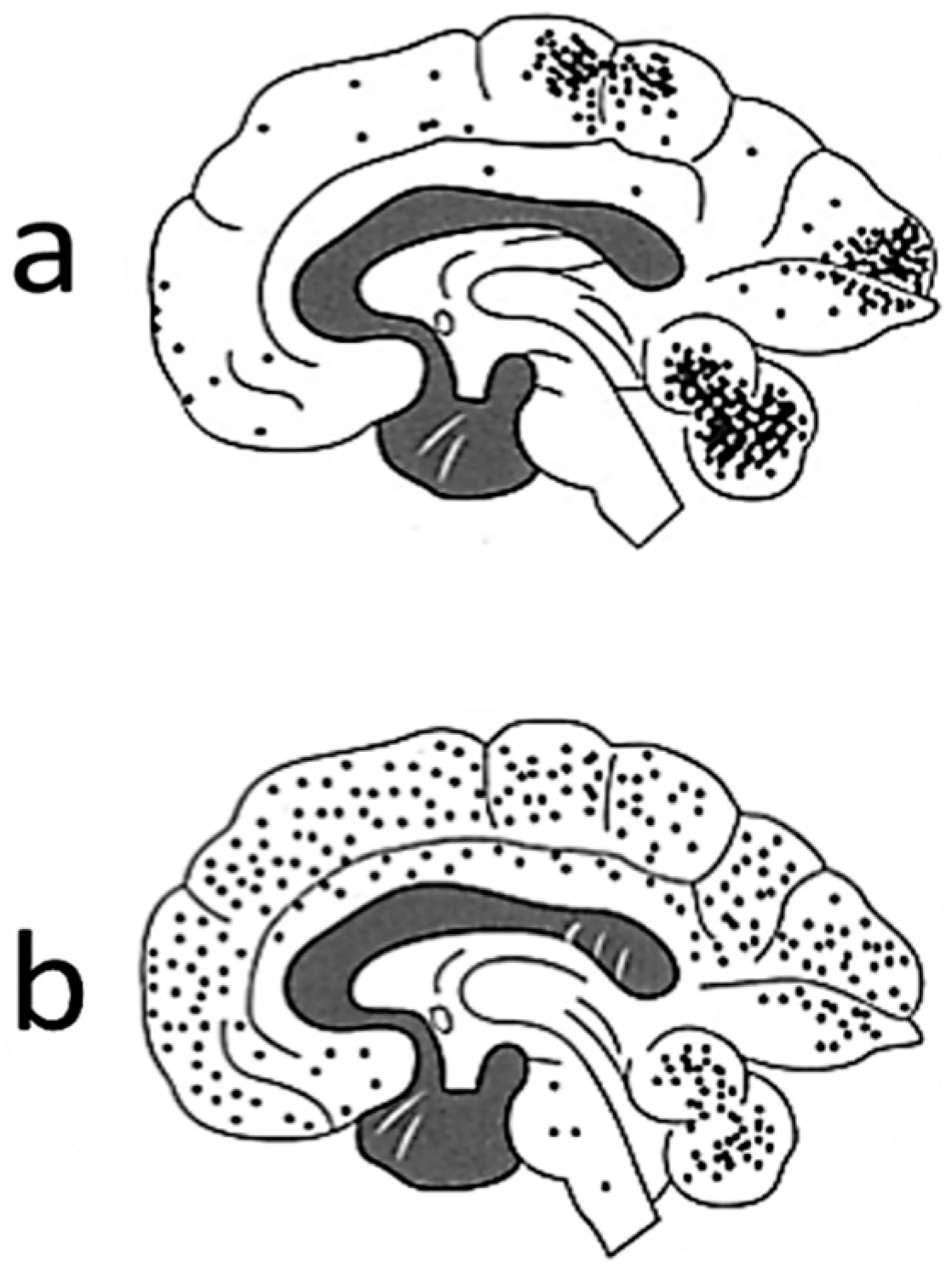
5. Neuropathology in Fetuses
6. Kinetics of Methylmercury in Fetuses
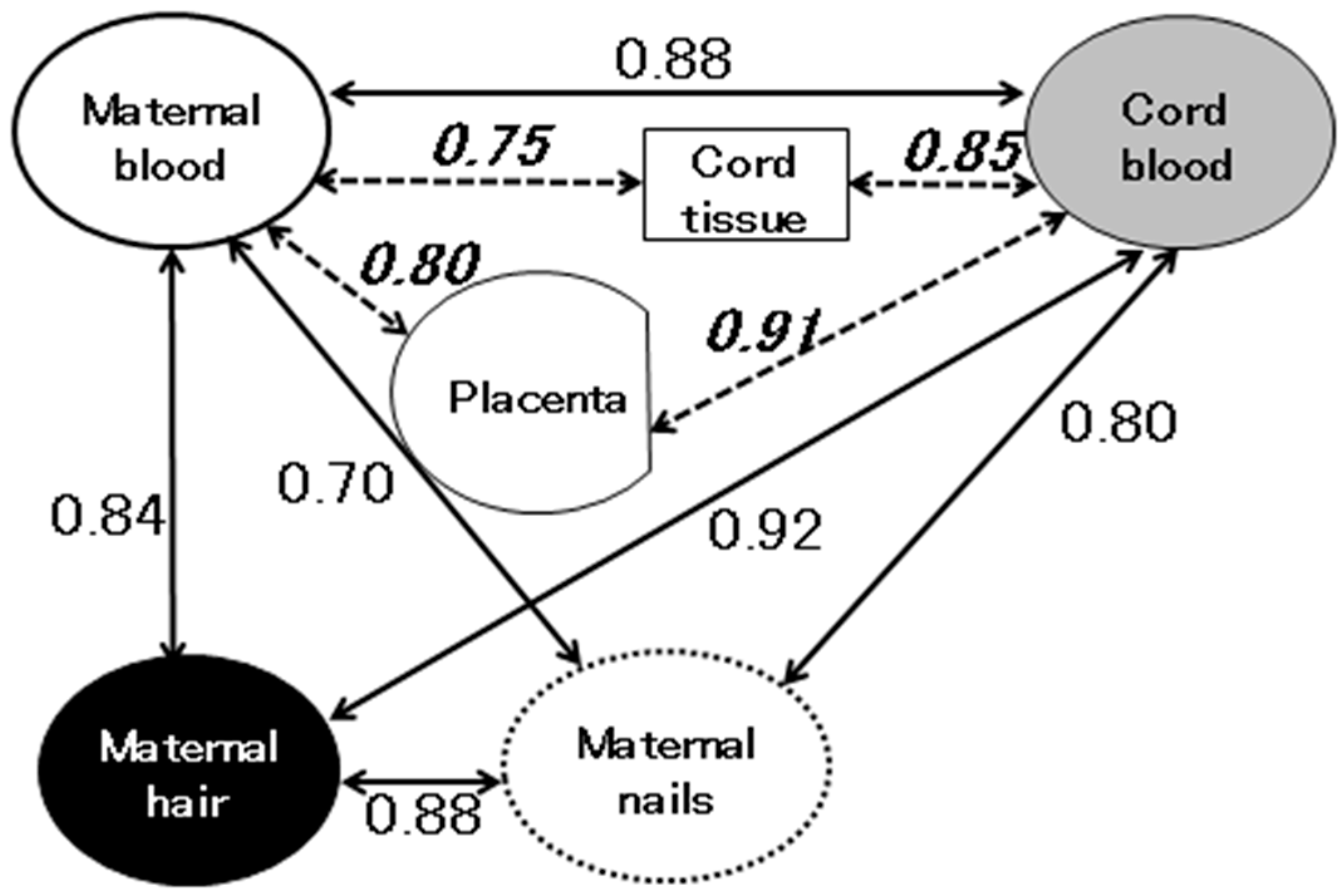
7. Exposure Assessment in Fetuses
8. Summary
Author Contributions
Funding
Conflicts of Interest
References
- NRC (National Research Council). Toxicological Effects of Methylmercury; Academic Press: Washington, DC, USA, 2000. [Google Scholar]
- UNIDO (United Nations Industrial Development Organization). Protocols for Environmental and Health Assessment of Mercury Released by Artisanal and Small-Scale Gold Miners; United Nations Industrial Development Organization: Vienna, Austria, 2008. [Google Scholar]
- WHO (World Health Organization). Inorganic Mercury. Environmental Health Criteria 118; World Health Organization: Geneva, Switzerland, 1991. [Google Scholar]
- UNEP (United Nations Environment Programme). Global Mercury Assessment 2013: Sources, Emissions, Releases and Environmental Transport; UNEP Chemicals Branch: Geneva, Switzerland, 2013. [Google Scholar]
- WHO (World Health Organization). Methylmercury. Environmental Health Criteria 101; World Health Organization: Geneva, Switzerland, 1990. [Google Scholar]
- Yin, Z.; Jiang, H.; Syversen, T.; Rocha, J.B.; Farina, M.; Aschner, M. The methylmercury-l-cysteine conjugate is a substrate for the l-type large neutral amino acid transporter. J. Neurochem. 2008, 107, 1083–1090. [Google Scholar] [CrossRef] [PubMed]
- Hirayama, K. Effect of amino acids on brain uptake of methyl mercury. Toxicol. Appl. Pharmacol. 1980, 55, 318–323. [Google Scholar] [CrossRef]
- Aschner, M.; Clarkson, T.W. Methyl mercury uptake across bovine brain capillary endothelial cells in vitro: The role of amino acids. Pharmacol. Toxicol. 1989, 64, 293–297. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, T.; Eto, K. The Pathology of Minamata Disease; Kyushu University Press: Fukuoka, Japan, 1999. [Google Scholar]
- Study Group. Minamata Disease; Kumamoto University: Kumamoto, Japan, 1966. [Google Scholar]
- Bakir, F.; Damluji, S.F.; Amin-Zaki, L.; Murtadha, M.; Khalidi, A.; al-Rawi, N.Y.; Tikriti, S.; Dahahir, H.I.; Clarkson, T.W.; Smith, J.C.; et al. Methylmercury poisoning in Iraq. Science 1973, 181, 230–241. [Google Scholar] [CrossRef] [PubMed]
- Amin-Zaki, L.; Elhassani, S.; Majeed, M.A.; Clarkson, T.W.; Doherty, R.A.; Greenwood, M.R.; Giovanoli-Jakubczak, T. Perinatal methylmercury poisoning in Iraq. Am. J. Dis. Child. 1976, 130, 1070–1076. [Google Scholar] [CrossRef] [PubMed]
- Harada, M. Congenital minamata disease: Intrauterine methylmercury poisoning. Teratology 1978, 18, 285–288. [Google Scholar] [CrossRef] [PubMed]
- Nishigaki, S.; Harada, M. Methylmercury and selenium in umbilical cords of inhabitants of the minamata area. Nature 1975, 258, 324–325. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, M.; Murata, K.; Tsuruta, K.; Miyamoto, K.; Akagi, H. Retrospective study on temporal and regional variations of methylmercury concentrations in preserved umbilical cords collected from inhabitants of the minamata area, Japan. Ecotoxicol. Environ. Saf. 2010, 73, 1144–1149. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, M.; Nakano, A.; Akagi, H. Declining minamata male birth ratio associated with increased male fetal death due to heavy methylmercury pollution. Environ. Res. 2001, 87, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Harada, M.; Akagi, H.; Tsuda, T.; Kizaki, T.; Ohno, H. Methylmercury level in umbilical cords from patients with congenital minamata disease. Sci. Total Environ. 1999, 234, 59–62. [Google Scholar] [CrossRef]
- Akagi, H.; Grandjean, P.; Takizawa, Y.; Weihe, P. Methylmercury dose estimation from umbilical cord concentrations in patients with minamata disease. Environ. Res. 1998, 77, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, M.; Murata, K.; Domingo, J.L.; Yamamoto, M.; Oliveira, R.B.; Kawakami, S.; Nakamura, M. Implications of mercury concentrations in umbilical cord tissue in relation to maternal hair segments as biomarkers for prenatal exposure to methylmercury. Environ. Res. 2016, 149, 282–287. [Google Scholar] [CrossRef] [PubMed]
- Mocarelli, P.; Gerthoux, P.M.; Ferrari, E.; Patterson, D.G., Jr.; Kieszak, S.M.; Brambilla, P.; Vincoli, N.; Signorini, S.; Tramacere, P.; Carreri, V.; et al. Paternal concentrations of dioxin and sex ratio of offspring. Lancet 2000, 355, 1858–1863. [Google Scholar] [CrossRef]
- Tokuomi, H.; Okajima, T.; Kanai, J.; Tsunoda, M.; Ichiyasu, Y.; Misumi, H.; Shimomura, K.; Takaba, M. Minamata disease. World Neurol. 1961, 2, 536–545. [Google Scholar] [PubMed]
- Wakabayashi, K.; Kakita, A.; Sakamoto, M.; Su, M.; Iwanaga, K.; Ikuta, F. Variability of brain lesions in rats administered methylmercury at various postnatal development phases. Brain Res. 1995, 705, 267–272. [Google Scholar] [CrossRef]
- Sakamoto, M.; Wakabayashi, K.; Kakita, A.; Hitoshi, T.; Adachi, T.; Nakano, A. Widespread neuronal degeneration in rats following oral administration of methylmercury during the postnatal developing phase: A model of fetal-type minamata disease. Brain Res. 1998, 784, 351–354. [Google Scholar] [CrossRef]
- Aschner, M.; Clarkson, T.W. Mercury 203 distribution in pregnant and nonpregnant rats following systemic infusions with thiol-containing amino acids. Teratology 1987, 36, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Kajiwara, Y.; Yasutake, A.; Adachi, T.; Hirayama, K. Methylmercury transport across the placenta via neutral amino acid carrier. Arch. Toxicol. 1996, 70, 310–314. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, M.; Kakita, A.; Wakabayashi, K.; Takahashi, H.; Nakano, A.; Akagi, H. Evaluation of changes in methylmercury accumulation in the developing rat brain and its effects: A study with consecutive and moderate dose exposure throughout gestation and lactation periods. Brain Res. 2002, 949, 51–59. [Google Scholar] [CrossRef]
- Honnda, S.; Hylandaer, L.; Sakamoto, M. Recent advances in evaluation of health effects on mercury with special reference to methylmercury―A minireview. Environ. Health Prev. Med. 2006, 11, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, M.; Kubota, M.; Liu, X.J.; Murata, K.; Nakai, K.; Satoh, H. Maternal and fetal mercury and n-3 polyunsaturated fatty acids as a risk and benefit of fish consumption to fetus. Environ. Sci. Technol. 2004, 38, 3860–3863. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, M.; Kubota, M.; Matsumoto, S.; Nakano, A.; Akagi, H. Declining risk of methylmercury exposure to infants during lactation. Environ. Res. 2002, 90, 185–189. [Google Scholar] [CrossRef]
- Sakamoto, M.; Yasutake, A.; Domingo, J.L.; Chan, H.M.; Kubota, M.; Murata, K. Relationships between trace element concentrations in chorionic tissue of placenta and umbilical cord tissue: Potential use as indicators for prenatal exposure. Environ. Int. 2013, 60, 106–111. [Google Scholar] [CrossRef] [PubMed]
- Stern, A.H.; Smith, A.E. An assessment of the cord blood: Maternal blood methylmercury ratio: Implications for risk assessment. Environ. Health Perspect. 2003, 111, 1465–1470. [Google Scholar] [CrossRef] [PubMed]
- Arbuckle, T.E.; Liang, C.L.; Morisset, A.S.; Fisher, M.; Weiler, H.; Cirtiu, C.M.; Legrand, M.; Davis, K.; Ettinger, A.S.; Fraser, W.D.; et al. Maternal and fetal exposure to cadmium, lead, manganese and mercury: The mirec study. Chemosphere 2016, 163, 270–282. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Chen, L.; Wang, C.; Zhou, Y.; Zhang, Y.; Wang, Y.; Shi, R.; Gao, Y.; Tian, Y. Prenatal low-level mercury exposure and infant neurodevelopment at 12 months in rural northern china. Environ. Sci. Pollut. Res. Int. 2016, 23, 12050–12059. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Lee, C.K.; Kim, K.H.; Lee, J.T.; Suh, C.; Kim, S.Y.; Kim, J.H.; Son, B.C.; Kim, D.H.; Lee, S. Factors associated with total mercury concentrations in maternal blood, cord blood, and breast milk among pregnant women in busan, korea. Asia Pac. J. Clin. Nutr. 2016, 25, 340–349. [Google Scholar] [PubMed]
- Huang, S.H.; Weng, K.P.; Lin, C.C.; Wang, C.C.; Lee, C.T.; Ger, L.P.; Wu, M.T. Maternal and umbilical cord blood levels of mercury, manganese, iron, and copper in southern Taiwan: A cross-sectional study. J. Chin. Med. Assoc. 2017, 80, 442–451. [Google Scholar] [CrossRef] [PubMed]
- Vigeh, M.; Nishioka, E.; Ohtani, K.; Omori, Y.; Matsukawa, T.; Koda, S.; Yokoyama, K. Prenatal mercury exposure and birth weight. Reprod. Toxicol. 2018, 76, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, M.; Chan, H.M.; Domingo, J.L.; Koriyama, C.; Murata, K. Placental transfer and levels of mercury, selenium, vitamin e, and docosahexaenoic acid in maternal and umbilical cord blood. Environ. Int. 2018, 111, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Rice, D.; Barone, S., Jr. Critical periods of vulnerability for the developing nervous system: Evidence from humans and animal models. Environ. Health Perspect. 2000, 108, 511–533. [Google Scholar] [CrossRef] [PubMed]
- WHO (World Health Organization). Children’s Exposure to Mercury Compounds; World Health Organization: Geneva, Switzerland, 2010. [Google Scholar]
- Boischio, A.A.; Cernichiari, E.; Henshel, D. Segmental hair mercury evaluation of a single family along the upper madeira basin, Brazilian Amazon. Cad. Saude Publica 2000, 16, 681–686. [Google Scholar] [CrossRef] [PubMed]
- Cernichiari, E.; Brewer, R.; Myers, G.J.; Marsh, D.O.; Lapham, L.W.; Cox, C.; Shamlaye, C.F.; Berlin, M.; Davidson, P.W.; Clarkson, T.W. Monitoring methylmercury during pregnancy: Maternal hair predicts fetal brain exposure. Neurotoxicology 1995, 16, 705–710. [Google Scholar] [PubMed]
- Alfthan, G.V. Toenail mercury concentration as a biomarker of methylmercury exposure. Biomarkers 1997, 2, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Hinners, T.; Tsuchiya, A.; Stern, A.H.; Burbacher, T.M.; Faustman, E.M.; Marien, K. Chronologically matched toenail-hg to hair-hg ratio: Temporal analysis within the Japanese community (U.S.). Environ. Health 2012, 11, 81. [Google Scholar] [CrossRef] [PubMed]
- Ohno, T.; Sakamoto, M.; Kurosawa, T.; Dakeishi, M.; Iwata, T.; Murata, K. Total mercury levels in hair, toenail, and urine among women free from occupational exposure and their relations to renal tubular function. Environ. Res. 2007, 103, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, M.; Chan, H.M.; Domingo, J.L.; Oliveira, R.B.; Kawakami, S.; Murata, K. Significance of fingernail and toenail mercury concentrations as biomarkers for prenatal methylmercury exposure in relation to segmental hair mercury concentrations. Environ. Res. 2015, 136, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Grandjean, P.; Budtz-Jørgensen, E.; Jørgensen, P.J.; Weihe, P. Umbilical cord mercury concentration as biomarker of prenatal exposure to methylmercury. Environ. Health Perspect. 2005, 113, 905–908. [Google Scholar] [CrossRef] [PubMed]
| Study Site | Measure | Maternal Blood Hg | Cord Blood Hg | Cord Blood/Maternal Blood Ratio | Sampling Years and (Published Year) References | |
|---|---|---|---|---|---|---|
| Ten cites, Canada | T-Hg | μg/L | 0.562 (n = 1673) | 0.802 (n = 1419) | 1.43 | 2008–2011 (2016) [32] |
| Laizhou By, China | T-Hg | μg/L | 0.72 (n = 410) | 1.20 (n = 410) | 1.67 | 2010–2012 (2016) [33] |
| Busan, Korea | T-Hg | μg/L | 3.12 (n = 127) | 5.46 (n = 127) | 1.75 | 2009–2010 (2016) [34] |
| Tong Gang, Taiwan | T-Hg | μg/L | 2.24 (n = 145) | 2.30 (n = 145) | 1.03 | 2010–2011 (2017) [35] |
| Tokyo, Japan | T-Hg | μg/L | 4.97 (n = 334) | 10.15 (n = 334) | 2.04 | 2010–2012 (2018) [36] |
| Kumamoto, Japan | T-Hg | ng/g | 3.79 (n = 54) | 7.26 (n = 54) | 1.92 | 2006–2007 (2018) [37] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sakamoto, M.; Tatsuta, N.; Izumo, K.; Phan, P.T.; Vu, L.D.; Yamamoto, M.; Nakamura, M.; Nakai, K.; Murata, K. Health Impacts and Biomarkers of Prenatal Exposure to Methylmercury: Lessons from Minamata, Japan. Toxics 2018, 6, 45. https://doi.org/10.3390/toxics6030045
Sakamoto M, Tatsuta N, Izumo K, Phan PT, Vu LD, Yamamoto M, Nakamura M, Nakai K, Murata K. Health Impacts and Biomarkers of Prenatal Exposure to Methylmercury: Lessons from Minamata, Japan. Toxics. 2018; 6(3):45. https://doi.org/10.3390/toxics6030045
Chicago/Turabian StyleSakamoto, Mineshi, Nozomi Tatsuta, Kimiko Izumo, Phuong Thanh Phan, Loi Duc Vu, Megumi Yamamoto, Masaaki Nakamura, Kunihiko Nakai, and Katsuyuki Murata. 2018. "Health Impacts and Biomarkers of Prenatal Exposure to Methylmercury: Lessons from Minamata, Japan" Toxics 6, no. 3: 45. https://doi.org/10.3390/toxics6030045
APA StyleSakamoto, M., Tatsuta, N., Izumo, K., Phan, P. T., Vu, L. D., Yamamoto, M., Nakamura, M., Nakai, K., & Murata, K. (2018). Health Impacts and Biomarkers of Prenatal Exposure to Methylmercury: Lessons from Minamata, Japan. Toxics, 6(3), 45. https://doi.org/10.3390/toxics6030045




