A Retrospective Analysis of Agricultural Herbicides in Surface Water Reveals Risk Plausibility for Declines in Submerged Aquatic Vegetation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Collection and Analysis
2.2. Sources for Toxicity Thresholds to Aquatic Life, Including SAV, and Pesticide and Land Use
3. Results
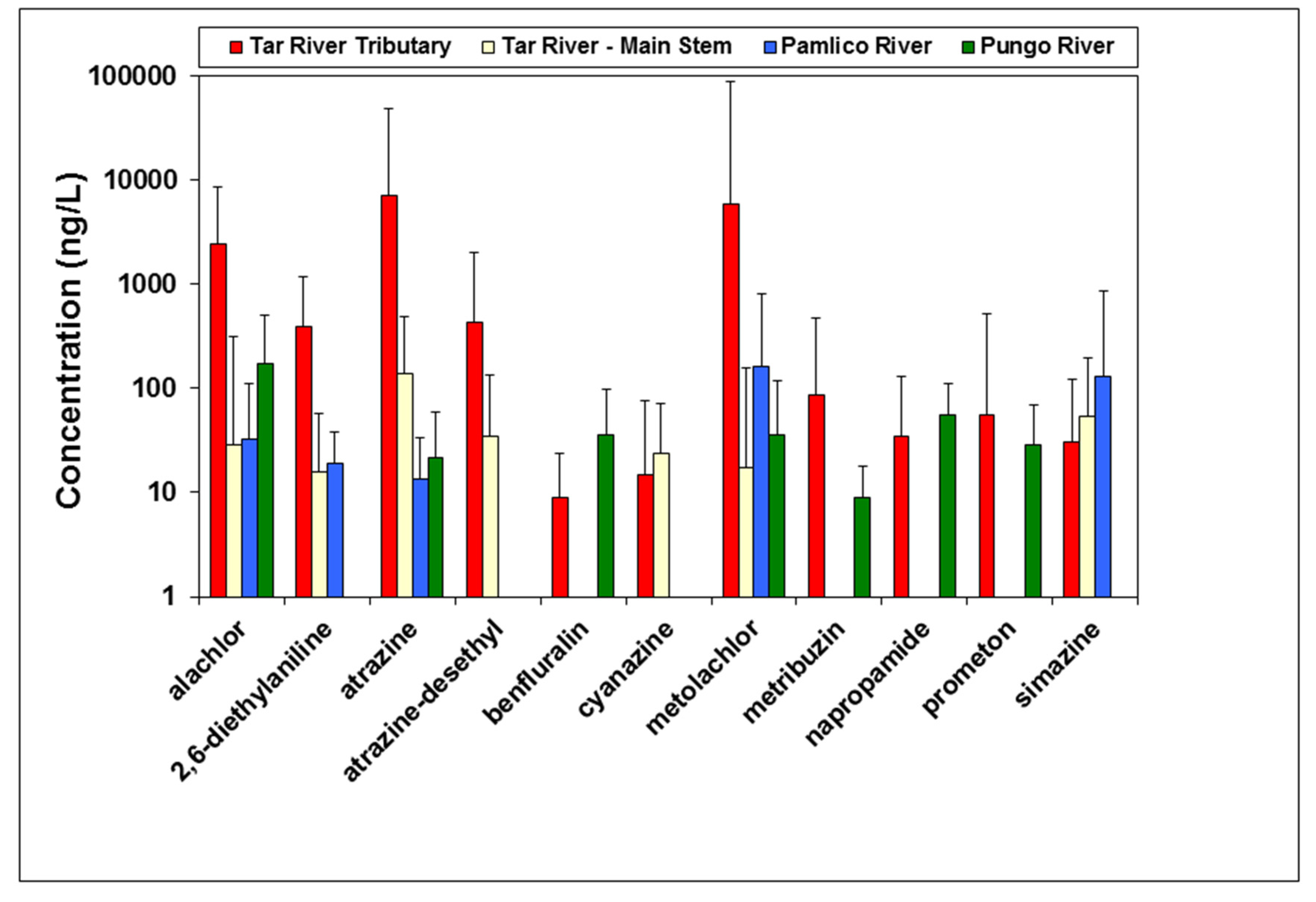
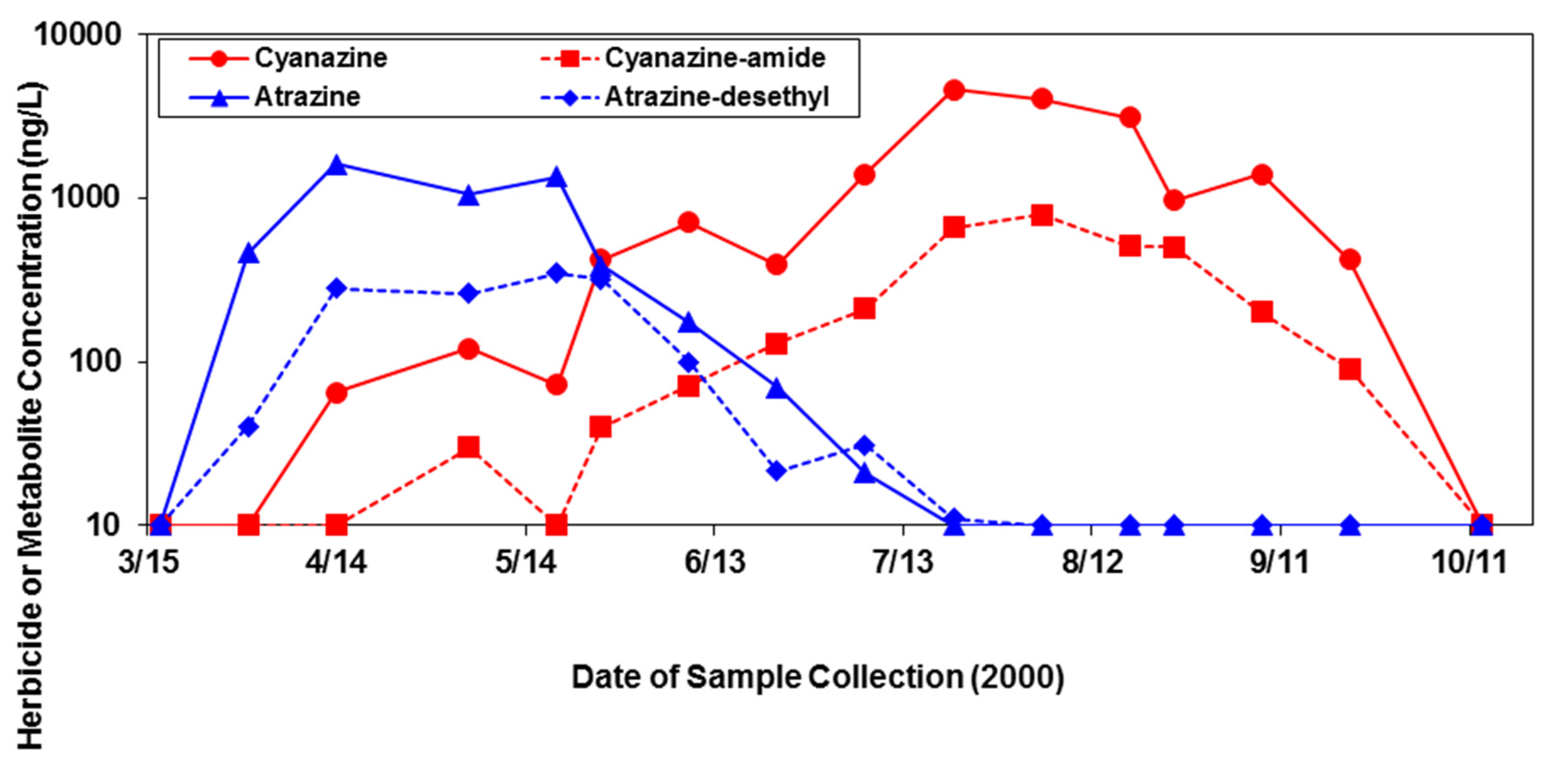
3.1. Herbicides in Surface Water
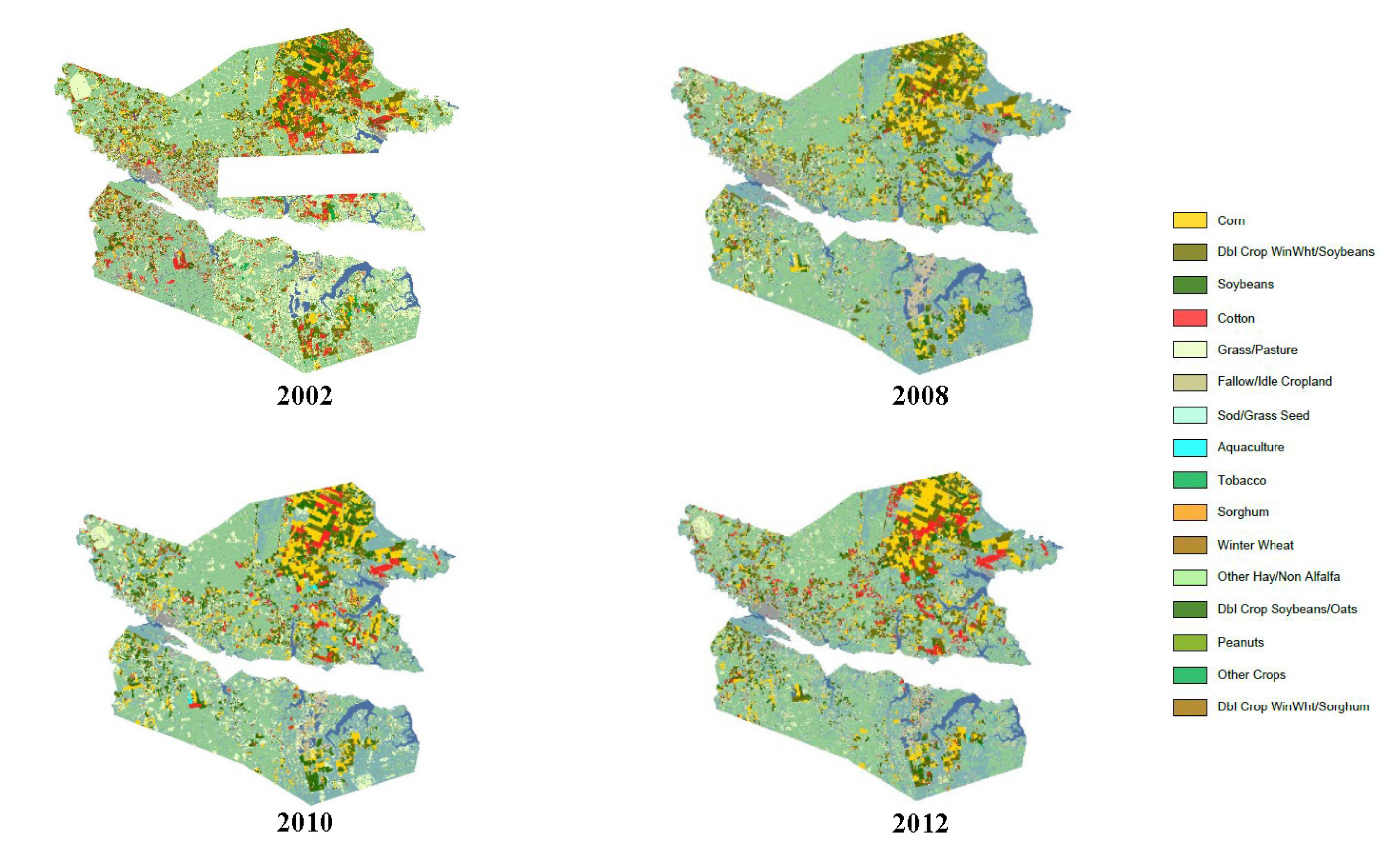
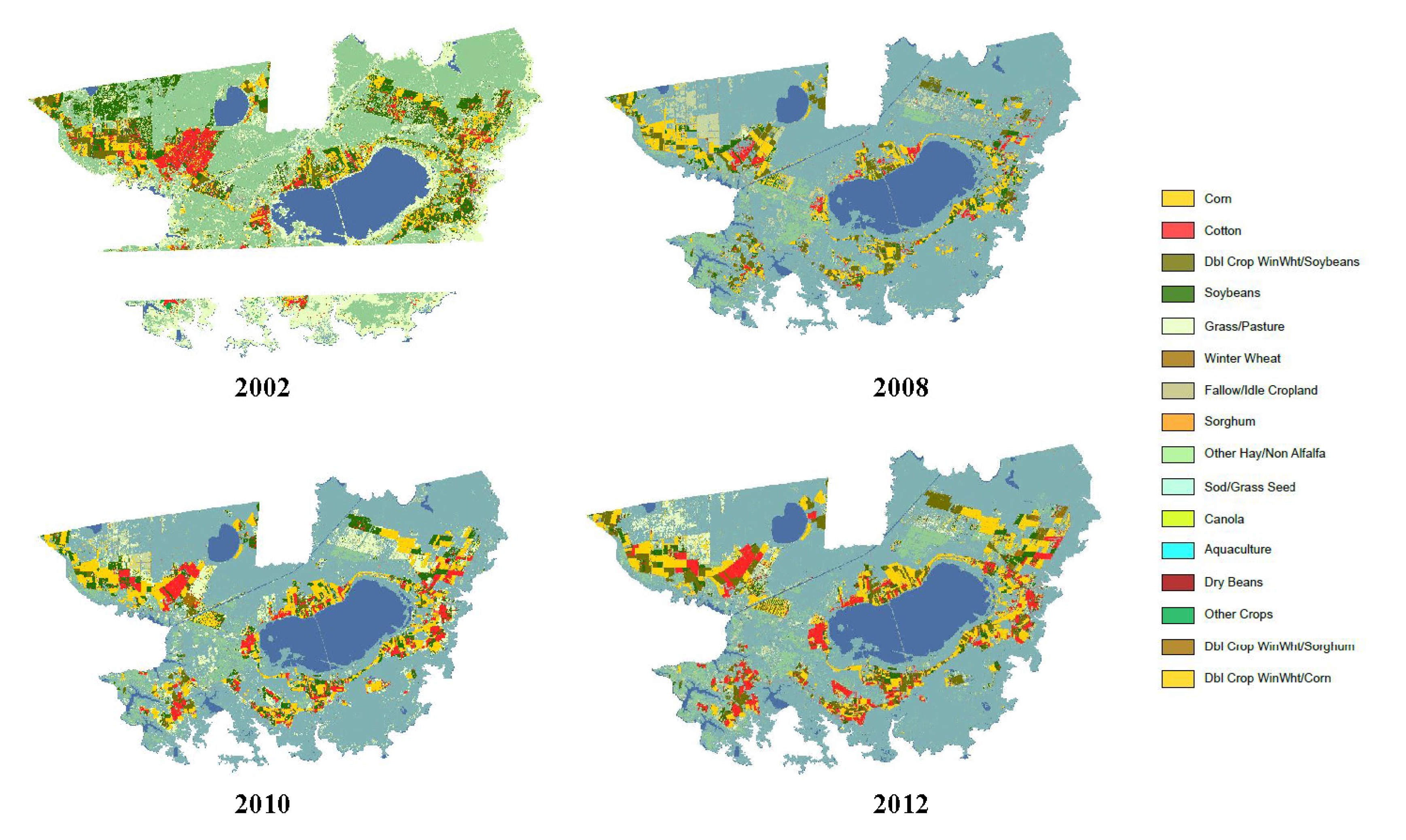
3.2. Toxicity Thresholds for Aquatic Life, Including SAV, and Pesticide and Land Use
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Albemarle-Pamlico National Estuary Partnership. The Albemarle-Pamlico Estuary. 2015. Available online: http://portal.ncdenr.org/web/apnep/our-estuaries (accessed on 7 September 2015).
- Steel, J. Albemarle-Pamlico Estuarine System Technical Analysis of Status and Trends; Albemarle-Pamlico Estuarine Study Report 91-01; NC Department of Environmental Quality: Raleigh, NC, USA, 1991.
- Carpenter, D.E.; Dubbs, L. 2012 Albemarle-Pamlico Ecosystem Assessment; Albemarle-Pamlico National Estuary Partnership: Columbia NC, USA, 2012.
- U.S. Department of Agriculture. North Carolina Census Publications. 2012. Available online: http://www.agcensus.usda.gov/Publications/2012/Online_Resources/County_Profiles/North_Carolina (accessed on 16 October 2015).
- U.S. Department of Agriculture. Virginia Census Publications. 2012. Available online: http://www.agcensus.usda.gov/Publications/2012/Online_Resources/County_Profiles/Virginia (accessed on 16 October 2015).
- Albemarle-Pamlico National Estuary Partnership. North Carolina Submerged Aquatic Vegetation. 2015. Available online: http://portal.ncdenr.org/web/apnep/sav-map (accessed on 16 October 2015).
- North Carolina Department of Environmental Quality. Division of Marine Fisheries, N.C. Coastal Habitats, Submerged Aquatic Vegetation. Available online: http://portal.ncdenr.org/web/mf/habitat/SAV (accessed on 16 October 2015).
- Moorman, M.; Kolb, K.R.; Supak, S. Estuarine Monitoring Programs in the Albemarle Sound Study Area, North Carolina; Open-File Report 2014-1110; U.S. Geological Survey: Reston, VA, USA, 2014.
- North Carolina Sea Grant. Coastal Water Quality Handbook; Report no. UNC–SG–97–04; North Carolina State University: Raleigh NC, USA, 1997.
- Deaton, A.S.; Chappell, W.S.; Hart, K.; O’Neal, J.; Boutin, B. North Carolina Coastal Habitat Protection Plan; North Carolina Department of Environment and Natural Resources, Division of Marine Fisheries: Morehead City, NC, USA, 2010.
- Moorman, M.C.; Augspurger, T.; Stanton, J.D.; Smith, A. Where’s the grass? Disappearing submerged aquatic vegetation and declining water quality in Lake Mattamuskeet. J. Fish Wildl. Manag. 2017. [Google Scholar] [CrossRef]
- Pait, A.S.; DeSouza, A.E.; Farrow, D.R.G. Agricultural Pesticide Use in Coastal Areas: A National Summary; Strategic Environmental Assessments Division, Office of Ocean Resources Conservation and Assessment National Ocean Service and National Oceanic and Atmospheric Administration: Rockville, MD, USA, 1992.
- North Carolina Department of Agriculture. Agricultural Statistics Division. 1999. Available online: http://www.agr.state.nc.us/stats (accessed on 20 October 2014).
- Gianessi, L.P.; Silvers, C.S. Trends in Crop Pesticide Use: Comparing 1992 and 1997; National Center for Food and Agricultural Policy: Washington, DC, USA, 2000.
- McCarthy, A.M. Fate and Distribution of Current-Use Pesticides in the Albemarle-Pamlico Estuarine System of North Carolina. Ph.D. Thesis, North Carolina State University, Raleigh, NC, USA, 2002. [Google Scholar]
- Mueller, T.C.; Senseman, S.A.; Wauchope, R.D.; Clegg, C.; Young, R.W.; Southwick, L.M.; Riley, M.B.; Moye, H.A.; Dumas, J.A.; Mersie, W.; et al. An interlaboratory comparison of empore solid-phase extraction disk recovery of atrazine, bromacil, chlorpyrifos and metolachlor from water samples. J. AOAC Intern. 2000, 83, 1327–1333. [Google Scholar]
- Chaves, A.; Shea, D.; Danehower, D. Analysis of chlorothalonil and degradation products in soil and water by GC/MS and LC/MS. Chemosphere 2008, 71, 629–638. [Google Scholar] [CrossRef] [PubMed]
- Zuagg, S.D.; Sandstrom, M.W.; Smith, S.G.; Fehlberg, K.M. Methods of Analysis by the U.S. Geological Survey National Water Quality Laboratory-Determination of Pesticides in Water by C-18 Solid-Phase Extraction and Capillary-Column Gas Chromatography/Mass Spectrometry with Selected-Ion Monitoring; Open-File Report 95-181; U.S. Geological Survey: Denver, CO, USA, 1995.
- U.S. Environmental Protection Agency. Office of Pesticide Programs’ Aquatic Life Benchmarks. 2014. Available online: http://www.epa.gov/oppefed1/ecorisk_ders/aquatic_life_benchmark.htm (accessed on 25 November 2014).
- North Carolina Department of Environment and Natural Resources. Division of Water Resources. 2013. Available online: http://portal.ncdenr.org/web/wq/ps/csu/swstandards (accessed on 25 November 2014).
- Canadian Council of Ministers of the Environment. Canadian Environmental Quality Guidelines. 2014. Available online: http://www.ccme.ca/en/resources/canadian_environmental_quality_guidelines/index.html (accessed on 25 November 2014).
- GfK. Crop Health Agriculture. 2014. Available online: http://www.gfk.com/en-us/about-gfk/gfk-in-your-country/ (accessed on 1 February 2016).
- U.S. Department of Agriculture. National Agricultural Statistics Service. 2014. Available online: http://www.nass.usda.gov/ (accessed on 17 November 2014).
- McConnell, L.L.; Rice, C.P.; Hapeman, C.J.; Drakeford, L.; Harman-Fetcho, J.A.; Bialek, K.; Fulton, M.H.; Leight, A.K.; Allen, G. Agricultural pesticides and selected degradation products in five tidal regions and the main stem of Chesapeake Bay, USA. Environ. Toxicol. Chem. 2007, 26, 2567–2578. [Google Scholar] [CrossRef] [PubMed]
- McConnell, L.L.; Harman-Fetcho, J.A.; Hagy III, J.D. Measured concentrations of herbicides and model predictions of atrazine fate in the Patuxent River Estuary. J. Environ. Qual. 2004, 33, 594–604. [Google Scholar] [CrossRef] [PubMed]
- Oros, D.R.; Jarman, W.M.; Lowe, T.; David, N.; Lowe, S.; Davis, J.A. Surveillance for previously unmonitored organic contaminants in the San Francisco Estuary. Mar. Pollut. Bull. 2003, 46, 1102–1110. [Google Scholar] [CrossRef]
- Woodside, M.D.; Ruhl, K.E. Pesticides in Streams in the Tar-Pamlico Drainage Basin, North Carolina, 1992–1994; Water Resources Investigations Report 01-4067; U.S. Geological Survey: Raleigh, NC, USA, 2001.
- Skrobialowski, S.C. Water Quality Assessment of the Albemarle-Pamlico Drainage Basin, North Carolina and Virginia; Water Resources Investigations Report 96-4104; U.S. Geological Survey: Raleigh, NC, USA, 1996.
- Spruill, T.B.; Harned, D.A.; Ruhl, P.M.; Eimers, J.L.; McMahon, G.; Smith, K.E.; Galeone, D.R.; Woodside, M.D. Water Quality in the Albemarle-Pamlico Drainage Basin; North Carolina and Virginia, 1992–1995; Circular 1157; U.S. Geological Survey: Denver, CO, USA, 1998.
- Ryberg, K.R.; Vecchia, A.V.; Gilliom, R.J.; Martin, J.D. Pesticide Trends in Major Rivers of the United States, 1992–2010; Scientific Investigations Report 2014-5135; National Water-Quality Assessment Program; U.S. Geological Survey: Reston, VA, USA, 2014.
- Pan, H.; Li, X.; Xu, X.; Gao, S. Phytotoxicity of four herbicides on Ceratophyllum demersum, Vallisneria natans and Elodea nuttallii. J. Environ. Sci. 2009, 21, 307–312. [Google Scholar] [CrossRef]
- Christopher, S.V.; Bird, T.V. The effects of herbicides on development of Myriophyllum spicatum L. cultured in vitro. J. Environ. Qual. 1992, 21, 203–207. [Google Scholar] [CrossRef]
- Brain, R.A.; Hoberg, J.; Hosmer, A.J.; Wall, S.B. Influence of light intensity on the toxicity of atrazine to the submerged freshwater aquatic macrophyte Elodea canadensis. Ecotoxicol. Environ. Saf. 2012, 79, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Cedergreen, N.; Spliid, N.H.; Streibig, J.C. Species-specific sensitivity of aquatic macrophytes towards two herbicides. Ecotoxicol. Environ. Saf. 2004, 58, 314–323. [Google Scholar] [CrossRef] [PubMed]
- Funderburk, S.L.; Mihursky, J.A.; Jordan, S.J.; Riley, D. Habitat Requirements for Chesapeake Bay Living Resources; Habitat Objectives Workgroup, Living Resources Subcommittee and Chesapeake Research Consortium, with Assistance from Maryland Department of Natural Resources: Solomons, MD, USA, 1991.
- Cedergreen, N.; Streibig, J.C. The toxicity of herbicides to non-target aquatic plants and algae: Assessment of predictive factors and hazard. Pest Manag. Sci. 2005, 61, 1152–1160. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, A.M.; Bales, J.D.; Cope, W.G.; Shea, D. Modeling pesticide fate in a small tidal estuary. Ecol. Model. 2007, 200, 149–159. [Google Scholar] [CrossRef]
- Hively, W.D.; Hapeman, C.J.; McConnell, L.L.; Fisher, T.R.; Rice, C.P.; McCarty, G.W.; Sadeghi, A.M.; Whitall, D.R.; Downey, P.M.; de Guzmán, G.T.N.; et al. Relating nutrient and herbicide fate with landscape features and characteristics of 15 subwatersheds in the Choptank River watershed. Sci. Total Environ. 2011, 409, 3866–3878. [Google Scholar] [CrossRef] [PubMed]
- Dantin, D.D.; Boustany, R.G.; Lewis, M.A.; Jordan, S.J.; Moss, R.F.; Michot, T.C. Effects of nutrient pre-exposure on atrazine toxicity to Vallisneria americana Michx. (wild celery). Arch. Environ. Contam. Toxicol. 2010, 58, 622–630. [Google Scholar] [CrossRef] [PubMed]
- Burkholder, J.M.; Dickey, D.A.; Kinder, C.A.; Reed, R.E.; Mallin, M.A.; McIver, M.R.; Cahoon, L.B.; Melia, G.; Brownie, C.; Smith, J.; et al. Comprehensive trend analysis of nutrients and related variables in a large eutrophic estuary: A decadal study of anthropogenic and climatic influences. Limnol. Oceanogr. 2006, 51, 463–487. [Google Scholar] [CrossRef]
- Kettle, N.P. Exposing compounding uncertainties in sea level rise assessments. J. Coast. Res. 2012, 28, 161–173. [Google Scholar] [CrossRef]
- Findlay, S.E.G.; Strayer, D.L.; Smith, S.D.; Curri, N. Magnitude and patterns of change in submerged aquatic vegetation of the tidal freshwater Hudson River. Estuar. Coasts 2014, 37, 1233–1242. [Google Scholar] [CrossRef]

| Herbicide | Measured Geometric Mean Concentration | Measured Maximum Concentration | US EPA OPP Aquatic Life Benchmarks | NC Surface Water Aquatic Life Standards | CCME Water Quality Guidelines a | ||
|---|---|---|---|---|---|---|---|
| Nonvascular Plants | Vascular Plants | Freshwater | Saltwater | Freshwater | |||
| Alachlor | 674 | 6100 | 1640 | 2300 | NA | NA | NA |
| Atrazine | 1836 | 41,000 | 1000 | 37,000 | 150,000 | 76,000 | 1800 |
| Metolachlor | 1521 | 82,000 | NA | NA | 240,000 | NA | 7800 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Powell, K.W.; Cope, W.G.; LePrevost, C.E.; Augspurger, T.; McCarthy, A.M.; Shea, D. A Retrospective Analysis of Agricultural Herbicides in Surface Water Reveals Risk Plausibility for Declines in Submerged Aquatic Vegetation. Toxics 2017, 5, 21. https://doi.org/10.3390/toxics5030021
Powell KW, Cope WG, LePrevost CE, Augspurger T, McCarthy AM, Shea D. A Retrospective Analysis of Agricultural Herbicides in Surface Water Reveals Risk Plausibility for Declines in Submerged Aquatic Vegetation. Toxics. 2017; 5(3):21. https://doi.org/10.3390/toxics5030021
Chicago/Turabian StylePowell, Kelly W., W. Gregory Cope, Catherine E. LePrevost, Tom Augspurger, Annette M. McCarthy, and Damian Shea. 2017. "A Retrospective Analysis of Agricultural Herbicides in Surface Water Reveals Risk Plausibility for Declines in Submerged Aquatic Vegetation" Toxics 5, no. 3: 21. https://doi.org/10.3390/toxics5030021
APA StylePowell, K. W., Cope, W. G., LePrevost, C. E., Augspurger, T., McCarthy, A. M., & Shea, D. (2017). A Retrospective Analysis of Agricultural Herbicides in Surface Water Reveals Risk Plausibility for Declines in Submerged Aquatic Vegetation. Toxics, 5(3), 21. https://doi.org/10.3390/toxics5030021






