Hair Microelement Profile as a Prognostic Tool in Parkinson’s Disease
Abstract
:1. Introduction
2. Materials and Methods
2.1. Recruitment of Participants
2.2. Hair Collection
2.3. Hair Analysis
2.4. Statistical Analysis
2.5. Principal Component Analysis and Hierarchical Cluster Analysis
- At the beginning, we have a collection of N atomic cluster, and each cluster contains one data point.
- According to a linkage method, the closest clusters are found.
- The closest clusters are melted into new clusters.
- The melted clusters are removed from the collection of clusters.
3. Results
3.1. Hair Analysis
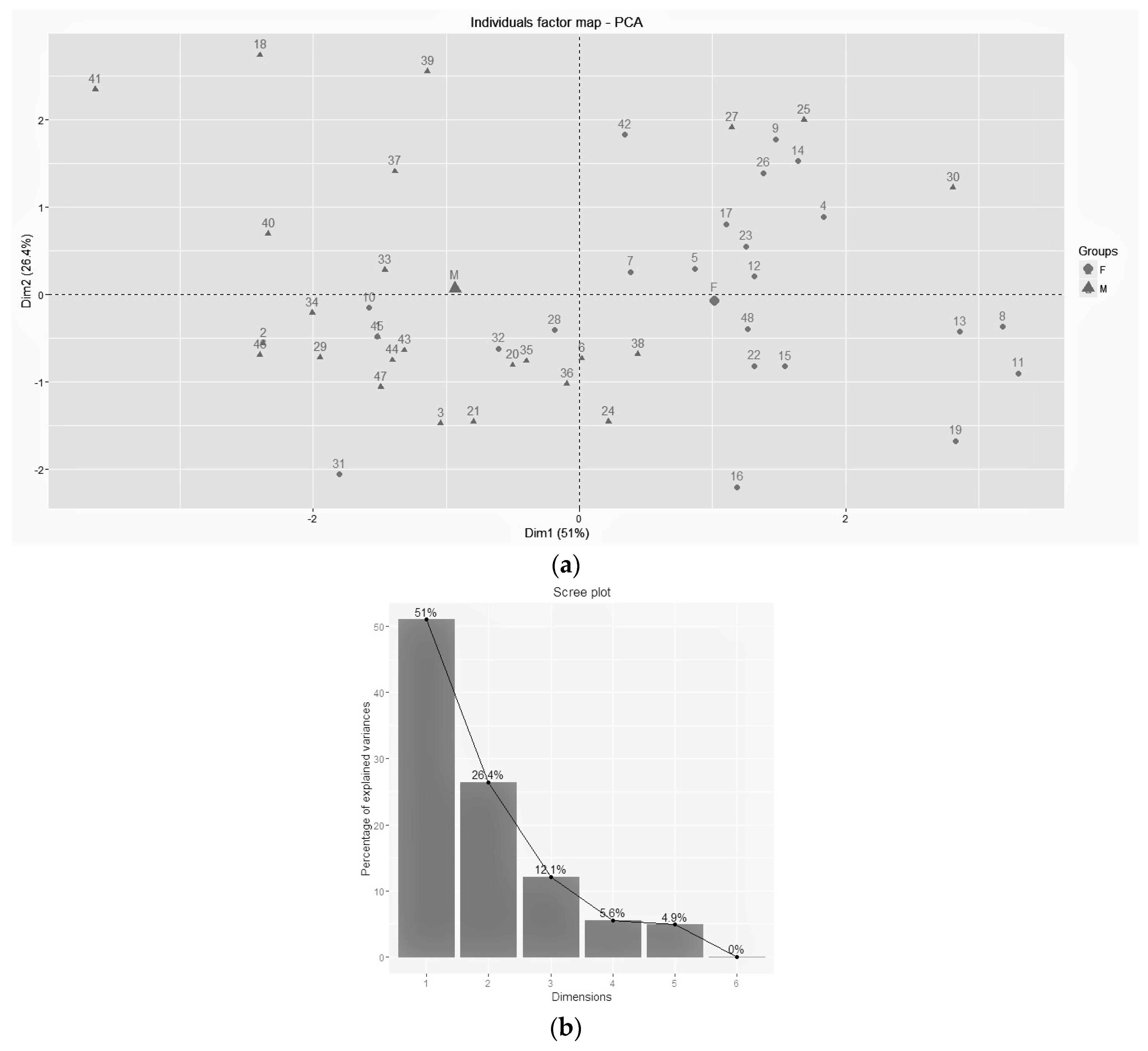
3.2. PCA
3.3. Hierarchical Clustering Analysis
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cannon, J.R.; Greenamyre, J.T. Gene-environment interactions in Parkinson’s disease: Specific evidence in humans and mammalian models. Neurobiol. Dis. 2013, 57, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Ratner, M.H.; Farb, D.H.; Ozer, J.; Feldman, R.G.; Durso, R. Younger age at onset of sporadic Parkinson’s disease among subjects occupationally exposed to metals and pesticides. Interdiscip. Toxicol. 2014, 7, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.; Costello, S.; Cockburn, M.; Zhang, X.; Bronstein, J.; Ritz, B. Parkinson’s disease risk from ambient exposure to pesticides. Eur. J. Epidemiol. 2011, 26, 547–555. [Google Scholar] [CrossRef] [PubMed]
- McMillan, G. Is electric arc welding linked to manganism or Parkinson’s disease? Toxicol. Rev. 2005, 24, 237–257. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Chakraborty, S.; Peres, T.V.; Bowman, A.B.; Aschner, M. Manganese-induced neurotoxicity: From C. elegans to humans. Toxicol. Res. 2015, 4, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Di Lorenzo, F. Iron and Parkinson’s disease. Neuro Endocrinol. Lett. 2015, 36, 24–27. [Google Scholar] [PubMed]
- Carloni, M.; Nasuti, C.; Fedeli, D.; Montani, M.; Amici, A.; Vadhana, M.S.; Gabbianelli, R. The impact of early life permethrin exposure on development of neurodegeneration in adulthood. Exp. Gerontol. 2012, 47, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Carloni, M.; Nasuti, C.; Fedeli, D.; Montani, M.; Amici, A.; Vadhana, M.S.D.; Gabbianelli, R. Early life permethrin exposure induces long-term brain changes in Nurr1, NF-kB and Nrf-2. Brain Res. 2013, 1515, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Fedeli, D.; Carloni, M.; Nasuti, C.; Gambini, A.; Scocco, V.; Gabbianelli, R. Early life permethrin exposure leads to hypervitaminosis D, nitric oxide and catecholamines impairment. Pestic. Biochem. Physiol. 2013, 107, 93–97. [Google Scholar] [CrossRef] [PubMed]
- Fedeli, D.; Montani, M.; Carloni, M.; Nasuti, C.; Amici, A.; Gabbianelli, R. Leukocyte Nurr1 as peripheral biomarker of early-life environmental exposure to permethrin insecticide. Biomarkers 2012, 17, 604–609. [Google Scholar] [CrossRef] [PubMed]
- Nasuti, C.; Gabbianelli, R.; Falcioni, M.L.; Di Stefano, A.; Sozio, P.; Cantalamessa, F. Dopaminergic system modulation, behavioural changes, and oxidative stress after neonatal administration of pyrethroids. Toxicology 2007, 229, 194–205. [Google Scholar] [CrossRef] [PubMed]
- Nasuti, C.; Carloni, M.; Fedeli, D.; Gabbianelli, R.; Di Stefano, A.; Cerasa, L.S.; Isabel, S.; Domingues, V.; Ciccocioppo, R. Effects of early life permethrin exposure on spatial working memory and on monoamine levels in different brain areas of pre-senescent rats. Toxicology 2013, 303, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Dusek, P.; Roos, P.M.; Litwin, T.; Schneider, S.A.; Flaten, T.P.; Aaseth, J. The neurotoxicity of iron, copper and manganese in Parkinson's and Wilson’s diseases. J. Trace Elem. Med. Biol. 2015, 31, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Barnham, K.J.; Bush, A.I. Biological metals and metal-targeting compounds in major neurodegenerative diseases. Chem. Soc. Rev. 2014, 43, 6727–6749. [Google Scholar] [CrossRef] [PubMed]
- Martín-Camean, A.; Molina-Villalba, I.; Jos, A.; Iglesias-Linares, A.; Solano, E.; Camean, A.M.; Gil, F. Biomonitorization of chromium, copper, iron, manganese and nickel in scalp hair from orthodontic patients by atomic absorption spectrometry. Environ. Toxicol. Pharmacol. 2014, 37, 759–771. [Google Scholar] [CrossRef] [PubMed]
- Bader, M.; Dietz, M.C.; Ihrig, A.; Triebig, G. Biomonitoring of manganese in blood, urine and caxillary hair following low-dose exposure during the manufacture of dry cell batteries. Int. Arch. Occup. Environ. Health 1999, 72, 521–527. [Google Scholar] [CrossRef] [PubMed]
- Aparecida da França, S.; Ferrera Dario, M.; Brigatto Esteves, V.; Rolim Baby, V.; Robles Velasco, M.V. Types of Hair Dye and Their Mechanisms of Action. Cosmetics 2015, 2, 110–126. [Google Scholar] [CrossRef]
- EPA. Progress Report: Metal Mixtures and Children’s Health. Available online: http://cfpub.epa.gov/ncer_abstracts/INDEX.cfm/fuseaction/display.abstractDetail/abstract/7978/report/2004 (accessed on 1 June 2004).
- Nasuti, C.; Ferraro, S.; Giovannetti, R.; Piangerelli, M.; Gabbianelli, R. Metal and Microelement Biomarkers of Neurodegeneration in Early Life Permethrin-Treated Rats. Toxics 2016, 4, 3. [Google Scholar] [CrossRef]
- Ambeskovic, M.; Fuchs, E.; Beaumier, P.; Gerken, M.; Metz, G.A. Hair trace elementary profiles in aging rodents and primates: Links to altered cell homeodynamics and disease. Biogerontology 2013, 14, 557–567. [Google Scholar] [CrossRef] [PubMed]
- Geier, D.A.; Kern, J.K.; King, P.G.; Sykes, L.K.; Geier, M.R. Hair toxic metal concentrations and autism spectrum disorder severity in young children. Int. J. Environ. Res. Public Health 2012, 9, 4486–4497. [Google Scholar] [CrossRef] [PubMed]
- Domingues, V.F.; Nasuti, C.; Piangerelli, M.; Correia-Sá, L.; Ghezzo, A.; Marini, M.; Abruzzo, P.M.; Visconti, P.; Giustozzi, M.; Rossi, G.; et al. Pyrethroid Pesticide Metabolite in Urine and Microelements in Hair of Children Affected by Autism Spectrum Disorders: A Preliminary Investigation. Int. J. Environ. Res. Public Health 2016, 13, 388. [Google Scholar] [CrossRef] [PubMed]
- Andersen, A.D.; Binzer, M.; Stenager, E.; Gramsbergen, J.B. Cerebrospinal fluid biomarkers for Parkinson’s disease—A systematic review. Acta Neurol. Scand. 2016. [Google Scholar] [CrossRef] [PubMed]
- Bohnen, N.I.; Gedela, S.; Kuwabara, H.; Constantine, G.M.; Mathis, C.A.; Studenski, S.A.; Moore, R.Y. Selective hyposmia and nigrostriatal dopaminergic denervation in Parkinson’s disease. J. Neurol. 2007, 254, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Postuma, R.B.; Berg, D.; Stern, M.; Poewe, W.; Olanow, C.W.; Oertel, W.; Obeso, J.; Marek, K.; Litvan, I.; Lang, A.E.; et al. MDS clinical diagnostic criteria for Parkinson’s disease. Mov. Disord. 2015, 30, 1591–1601. [Google Scholar] [CrossRef] [PubMed]
- Goetz, C.G.; Poewe, W.; Rascol, O.; Sampaio, C.; Stebbins, G.T.; Counsell, C.; Giladi, N.; Holloway, R.G.; Moore, C.G.; Wenning, G.K.; et al. Movement Disorder Society Task Force report on the Hoehn and Yahr staging scale: Status and recommendations. Mov. Disord. 2004, 19, 1020–1028. [Google Scholar] [CrossRef] [PubMed]
- Ha, Y.; Tsay, O.G.; Churchill, D.G. A tutorial and mini-review of the ICP-MS technique for determinations of transition metal ion and main group element concentration in the neurodegenerative and brain sciences. Monatshefte Chem. Chem. Mon. 2011, 142, 385–398. [Google Scholar] [CrossRef]
- Gellein, K.; Lierhagen, S.; Brevik, P.S.; Teigen, M.; Kaur, P.; Singh, T.; Flaten, T.P.; Syversen, T. Trace Element Profiles in Single Strands of Human Hair Determined by HR-ICP-MS. Biol. Trace Elem. Res. 2008, 123, 250–260. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, T. Determination of Trace Elements in Hair by ICP-MS; Agilent Technologies Application Note AN-0812–0133EN; Agilent Technologies, Inc.: Santa Clara, CA, USA, 2000. [Google Scholar]
- Goullé, J.P.; Mahieu, L.; Castermant, J.; Neveu, N.; Bonneau, L.; Lainé, G.; Bouige, D.; Lacroix, C. Metal and metalloid multi-elementary ICP-MS validation in whole blood, plasma, urine and hair. Reference values. Forensic. Sci. Int. 2005, 153, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Murtagh, F.; Legendre, P. Ward’s hierarchical clustering method: Clustering criterion and agglomerative algorithm. J. Classif. 2011, 31, 274–295. [Google Scholar] [CrossRef]
- Nagarajan, A.; Ning, Y.; Reisner, K.; Buraei, Z.; Larsen, J.P.; Hobert, O.; Doitsidou, M. Progressive Degeneration of Dopaminergic Neurons through TRP Channel-Induced Cell Death. J. Neurosci. 2014, 34, 5738–5746. [Google Scholar] [CrossRef] [PubMed]
- Ådén, E.; Carlsson, M.; Poortvliet, E.; Stenlund, H.; Linder, J.; Edström, M.; Forsgren, L.; Håglin, L. Dietary intake and olfactory function in patients with newly diagnosed Parkinson’s disease: A case-control study. Nutr. Neurosci. 2011, 14, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Willis, A.W.; Evanoff, B.A.; Lian, M.; Galarza, A.; Wegrzyn, A.; Schootman, M.; Racette, B.A. Metal Emissions and Urban Incident Parkinson Disease: A Community Health Study of Medicare Beneficiaries by Using Geographic Information Systems. Am. J. Epidemiol. 2010, 172, 1357–1363. [Google Scholar] [CrossRef] [PubMed]
- Bush, A.I. Metals and neuroscience. Curr. Opin. Chem. Biol. 2000, 4, 184–191. [Google Scholar] [CrossRef]
- Caudle, M.W.; Guillot, T.S.; Lazo, C.R.; Miller, G.R. Industrial toxicants and Parkinson’s disease. Neurotoxicology 2012, 33, 178–188. [Google Scholar] [CrossRef] [PubMed]
- Goldman, S.M. Environmental Toxins and Parkinson’s Disease. Annu. Rev. Pharmacol. Toxicol. 2014, 54, 141–164. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Kim, J. Degeneration of Dopaminergic Neurons Due to Metabolic Alterations and Parkinson’s Disease. Front. Aging Neurosci. 2016, 8, 65–76. [Google Scholar] [CrossRef] [PubMed]

| Metals and Microelements (ppm) | Control (n = 24) # Mean ± SEM | PD (n = 24) # Mean ± SEM | PD (n = 46) Mean ± SEM |
|---|---|---|---|
| Na | 727.018 ± 125.760 | 620.395 ± 92.464 | 628.611 ± 92.464 |
| Mg | 207.201 ± 30.075 | 153.617 * ± 24.717 | 174.294 ± 27.013 |
| Al | 111.452 ± 22.754 | 262.138 ± 64.209 | 283.230 ± 66.506 |
| Si | 303.830 ± 62.176 | 308.630 ±32.806 | 335.799 ± 41.869 |
| P | 367.064 ± 42.159 | 538.565 ± 59.665 | 503.966 ± 53.817 |
| S | 42372.277 ± 1205.134 | 41186.553 ± 954.733 | 40144.768 ± 1037.435 |
| K | 855.179 ± 142.877 | 1008.183 ± 127.685 | 1014.116 ± 125.460 |
| Ca | 2309.983 ± 350.837 | 1361.330 * ± 237.619 | 1674.490 * ± 266.350 |
| Cr | 2.633 ± 1.745 | 0.937 ± 0.113 | 1.341 ± 0.211 |
| Mn | 0.255± 0.032 | 0.195± 0.032 | 1.775 ± 0.591 |
| Fe | 15.174 ± 2.119 | 11.643 ± 1.156 | 24.812 ± 3.064 |
| Ni | 2.018 ± 0.388 | 3.162 ± 0.890 | 2.846 ± 0.713 |
| Cu | 25.276± 9.854 | 16.900± 3.011 | 16.467 ± 2.925 |
| Zn | 126.588± 9.575 | 103.297± 9.310 | 177.398 ± 12.844 |
| As | 0.024 ± 0.004 | 0.027 ± 0.002 | 0.024 ± 0.002 |
| Se | 0.522 ± 0.093 | 0.462 ± 0.022 | 0.428 ± 0.023 |
| Rb | 0.932 ± 0.162 | 1.495 ± 0.217 | 1.350 ± 0.197 |
| Sr | 14.034 ± 2.700 | 7.297 * ± 1.289 | 8.367 * ± 1.844 |
| Ag | 0.102 ± 0.025 | 0.093 ± 0.016 | 0.097 ± 0.016 |
| Cd | 0.024 ± 0.006 | 0.009 §± 0.001 | 0.009 * ± 0.001 |
| Sn | 0.670 ± 0.188 | 0.446 ± 0.146 | 0.417 ± 0.112 |
| Sb | 0.075 ± 0.015 | 0.103 ± 0.045 | 0.196 ± 0.113 |
| Hg | 9.665 ± 3.113 | 6.552 ± 1.026 | 4.588 ± 0.844 |
| Pb | 1.038 ± 0.241 | 0.735 ± 0.083 | 0.627 ± 0.069 |
| Ca/Mg | 12.210 ± 2.006 | 10.721 ± 1.503 | 11.834 ± 1.823 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stefano, F.; Cinzia, N.; Marco, P.; Marco, G.; Rita, G.; Augusto, F.; Rosita, G. Hair Microelement Profile as a Prognostic Tool in Parkinson’s Disease. Toxics 2016, 4, 27. https://doi.org/10.3390/toxics4040027
Stefano F, Cinzia N, Marco P, Marco G, Rita G, Augusto F, Rosita G. Hair Microelement Profile as a Prognostic Tool in Parkinson’s Disease. Toxics. 2016; 4(4):27. https://doi.org/10.3390/toxics4040027
Chicago/Turabian StyleStefano, Ferraro, Nasuti Cinzia, Piangerelli Marco, Guidi Marco, Giovannetti Rita, Ferri Augusto, and Gabbianelli Rosita. 2016. "Hair Microelement Profile as a Prognostic Tool in Parkinson’s Disease" Toxics 4, no. 4: 27. https://doi.org/10.3390/toxics4040027
APA StyleStefano, F., Cinzia, N., Marco, P., Marco, G., Rita, G., Augusto, F., & Rosita, G. (2016). Hair Microelement Profile as a Prognostic Tool in Parkinson’s Disease. Toxics, 4(4), 27. https://doi.org/10.3390/toxics4040027








