Evaluation of Glycol Ether as an Alternative to Perchloroethylene in Dry Cleaning
Abstract
:1. Introduction
2. Methodology
3. Results and Discussion
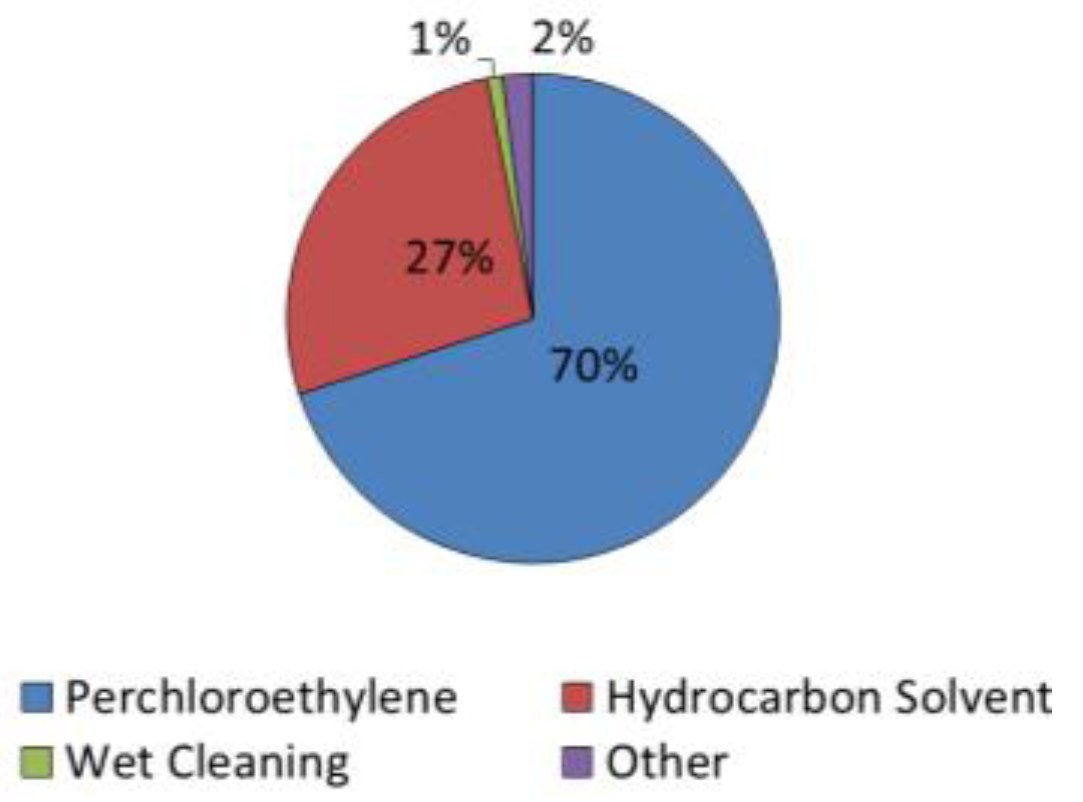
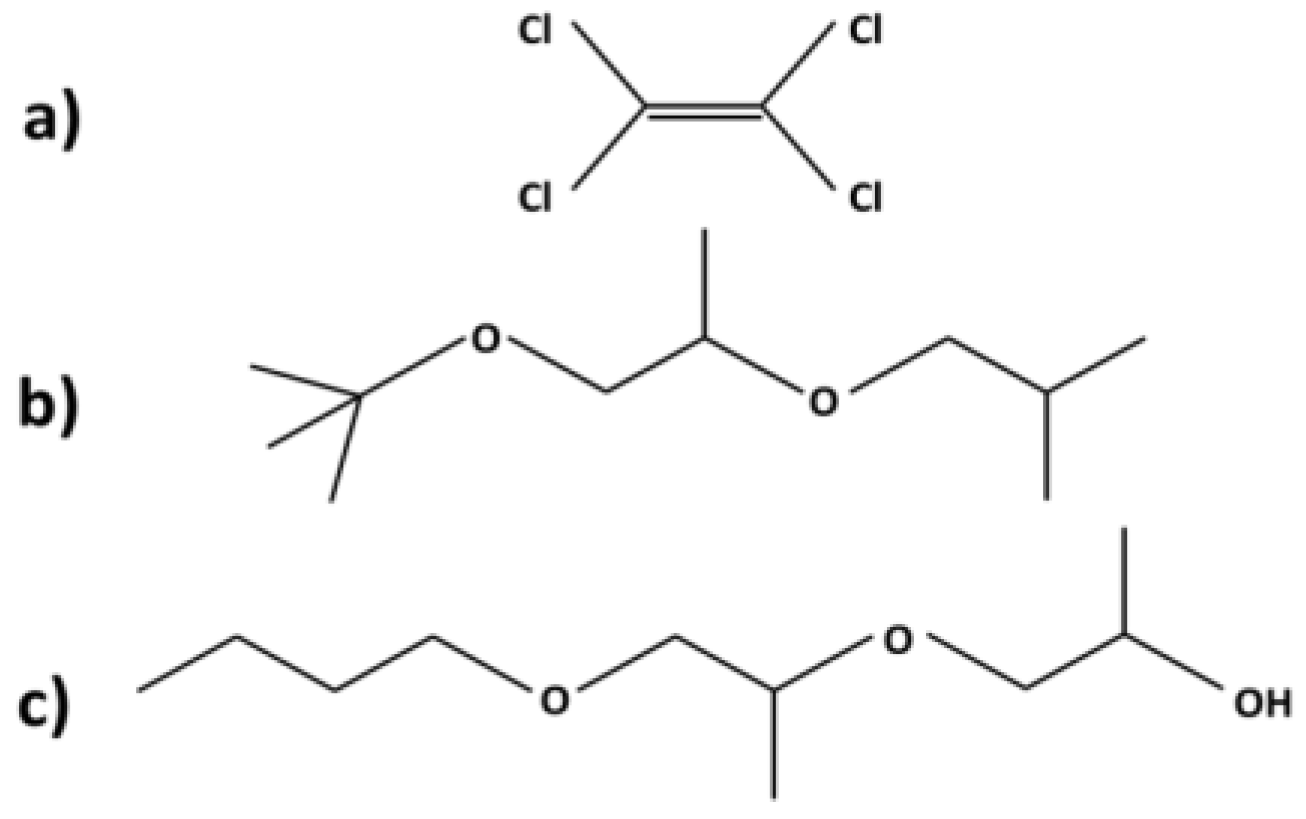
3.1. Chemical Toxicity
3.2. Employee Exposure Levels
| Exposure pathway | Exposure level (ppm) | 8-h TWA (ppm) | ||
|---|---|---|---|---|
| De-soiling a | 0.01 | 39 | N/A | N/A |
| Transfer-based machine operated a | 13 | 153 | N/A | N/A |
| Dry-to-Dry machine operated b,c | 0.3 | 83 | 4.1 | 5 |
| Pressing a,b | 0.1 | 6.5 | 0.5 | 1.1 |
| Customer Service a,b | 0 | 15 | N/A | 0.1 |
| Maintenance a | N/A | 334 | N/A | N/A |
3.3. Customer Exposure Levels
| Concentration (nmol/cm2) | Average | Standard deviation |
|---|---|---|
| Cotton | 17.0 | 5.96 |
| Polyester | 45.5 | 11.7 |
| Silk | ND | - |
| Wool | 31.5 | 11.8 |
3.4. Impacts to General Population
| POCP | PCE a,b,c | GE a,b |
|---|---|---|
| Average | 0.9 | 37.2 |
| Standard Deviation | 0.7 | 21.6 |
| Min | 0.0 | 17.0 |
| Max | 2.0 | 80.0 |
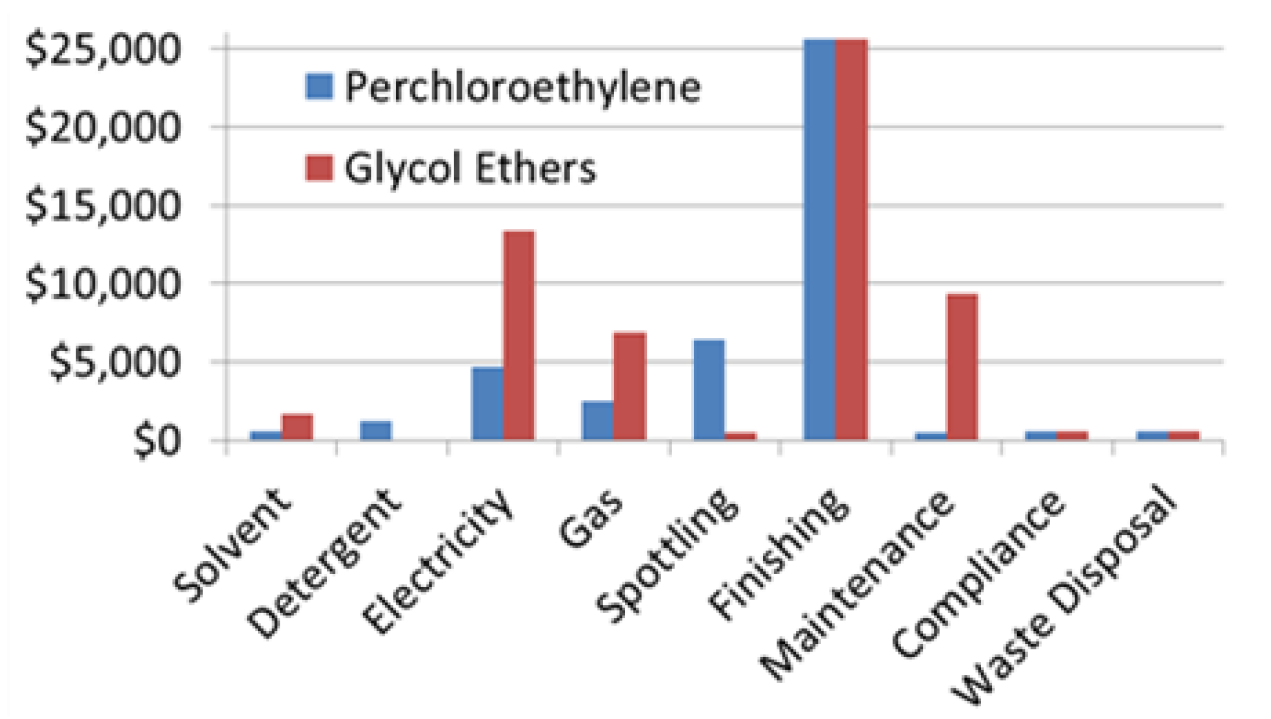
3.5. Costs
- Processing 40,000 pounds of clothing per year and 27 loads per week (1380 loads per year);
- 35-pound dry-to-dry closed-loop machine including secondary control;
- 60 gallons of PCE per year at a cost of $10 per gallon;
- 50 gallons of detergent per year at a cost of $25 per gallon;
- Annual electricity costs were based on case studies and brought to current 2014 dollars using CPI conversion factor for electricity prices;
- Annual gas costs were based on case studies and brought to current 2014 dollars using CPI conversion factor for utility gas prices;
- Spotting labor was found based on case studies to be 2.46 h per week at $10 per h labor cost;
- Finishing labor was found based on case studies to be 9.85 h per week at $10 per hour labor cost;
- Maintenance labor of one hour per week was found based on case studies at $10 per hour cost;
- Due to use of spin disk filters in case studies, the maintenance equipment cost was assumed to be zero;
- Compliance labor estimates were based on case studies and suggested one hour per week at $10 per hour labor cost;
- Case studies showed that two drums of hazardous waste were produced per year and a disposal cost of $275 per drum;
- Processing of 40,000 pounds of clothing per year and 27 loads per week (1380 loads per year);
- 35-pound machine used;
- Annual solvent use was found from the case study and lower volatility than PCE to be 50 gallons per year at a cost of $33 per gallon from Caled and Rynex prices;
- No detergent was used based on case study;
- Annual electricity was normalized to 40,000 pounds of clothing cleaned per year;
- Annual gas was normalized to 40,000 pounds of clothing cleaned per year;
- Spotting labor was estimated based on a case study to be 1 h per week at $10 per hour labor cost;
- Finishing labor was estimated based on a case study to be 9.85 h per week at $10 per hour labor cost;
- Maintenance labor of 18 h per week was determined based on a case study with $10-per-hour associated costs;
- Maintenance equipment cost was assumed to be zero based on the case study;
- Compliance labor was based on case studies and found to be one hour per week at $10 per hour labor cost;
- The case study shows that two drums of hazardous waste were produced per year and a disposal cost of $275 per drum.
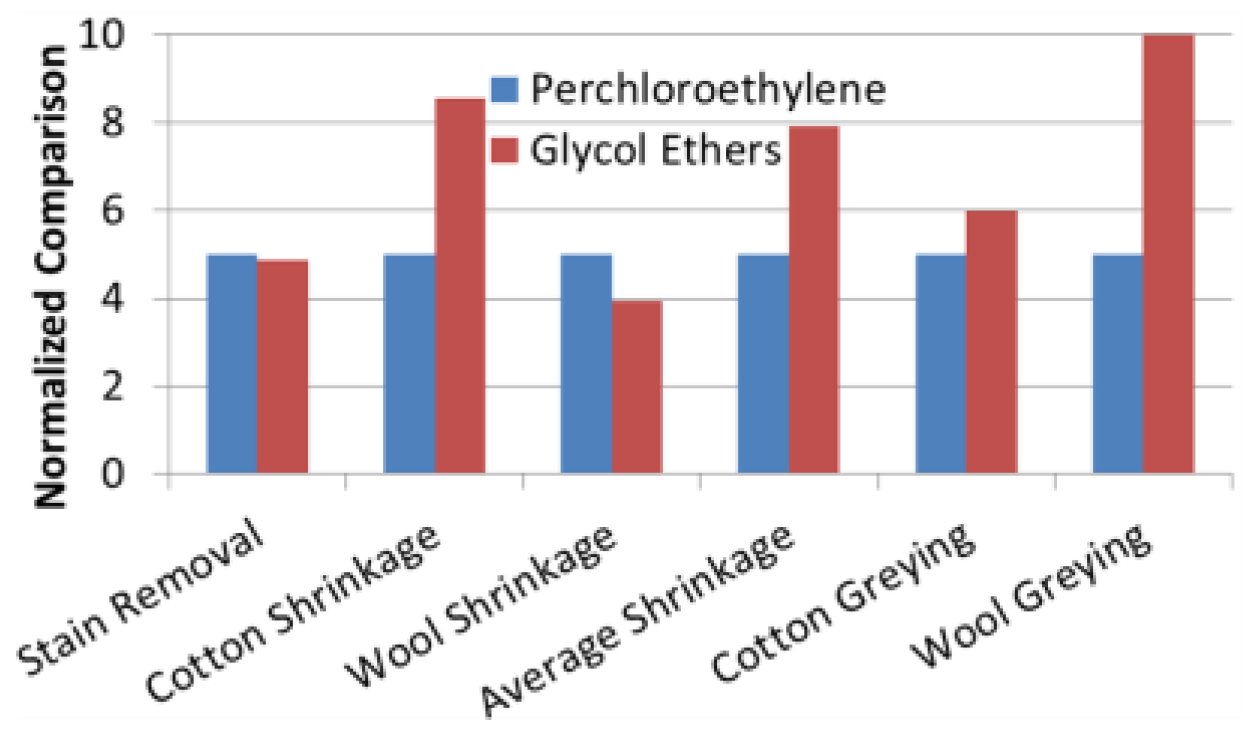
3.6. Cleaning Effectiveness
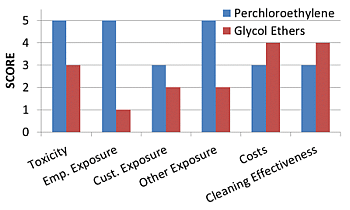
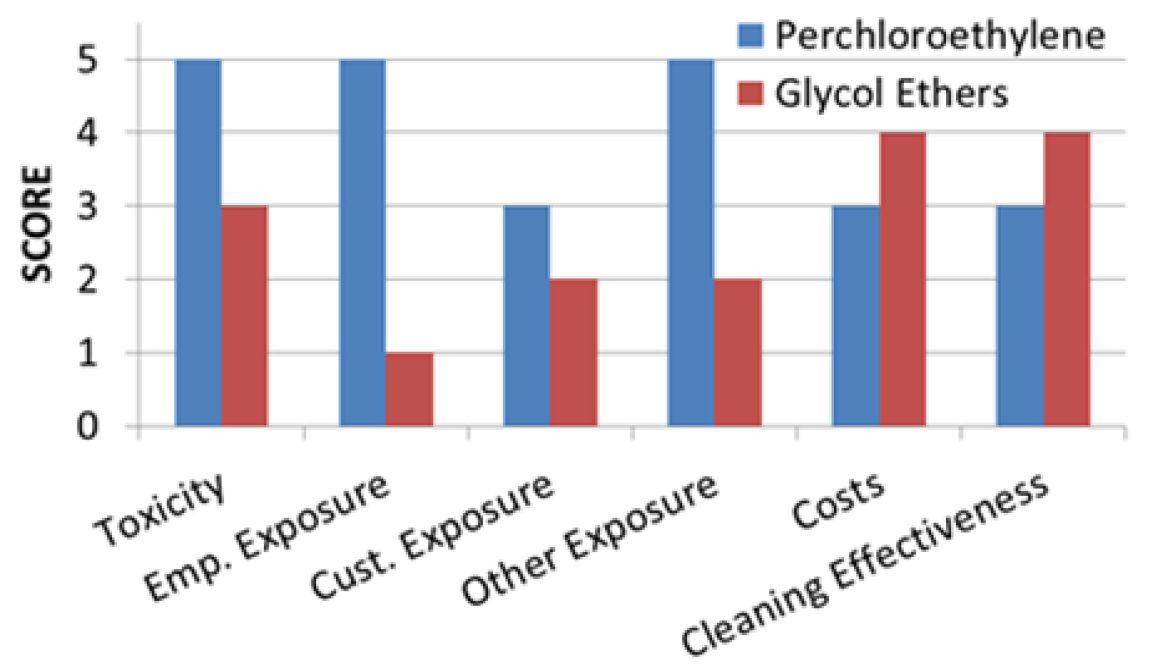
3.7. Overall Comparison
| Criteria | PCE | GE |
|---|---|---|
| Chemical Toxicity | The EPA reclassified PCE as a likely human carcinogen: The average LD50 is 10,150 ± 8277 mg/kg | Fewer adverse effects have been detected; and among having been detected, effects generally were mild: No HAP Lower fire-hazard cost, the average LD50 is 3122 ± 1334 mg/kg |
| Employee Exposure Levels | Susceptible to several adverse reproductive effects PCE with an average of 181 ± 294 ppm in air at dry cleaning establishments PCE degradation corresponds with a half-life in air ranging from 40 to 70 days with an average of 52.3 ± 12.8 days | “All propylene GEs are currently believed to be relatively safe,” and “most ethylene GEs with ‘methyl’ in their names are relatively toxic.” A half-life in air was found to be 7.6 h |
| Customer Exposure Levels | Principally occurs through exposure to residual PCE present on the garments at high levels, ranging anywhere from 10 to 56 (nmol/cm2) | No inhalation exposure assessments for consumers appears to have been conducted with regard to dry cleaning exposure risks |
| Impacts to General Population | PCE’s persistence in groundwater and in residences co-located with dry cleaners The average of Henry’s Law constant for PCE is 1.68 × 10−2 ± 2.07 × 10−3 atm-m3/mol at 25 °C | DPnB and DPtB are readily biodegradable: The average of Henry’s Law constant for GEs is 9.95 × 10−7 ± 1.48 × 10−6 atm-m3/mol at 25 °C |
| Costs | $1,000/lb machine cost with $1,000 install fee totalled a $40,000 capital investment (assuming 35-pound machine) approximately $43,000/year operating cost | With a comparable capital investment (assuming 35-pound machine) the operating cost for GE is 37% higher than that of PCE at approximatly $59,000/year; Shorter wash cycle corresponds to less chemical usage |
| Cleaning Effectiveness | Less cotton shrinkage, less average shrinkage, less wool greying when compared to GE | Effective on water and oil-based stains Safer for most fabrics; impared zipper functions |
4. Conclusions
Acknowledgments
Conflicts of Interest
Author Contributions
References
- Anonymous. Solvetex ii Quality Performents of Solvents; CINET Professional Textile Care: Ophemert, Gelderland, The Netherlands, 2011. [Google Scholar]
- Rastkari, N.; Yunesian, M.; Ahmadkhaniha, R. Exposure assessment to trichloroethylene and perchloroethylene for workers in the dry cleaning industry. Bull. Environ. Contam. Toxicol. 2011, 86, 363–367. [Google Scholar] [CrossRef]
- Raaschou-Nielsen, O.; Hansen, J.; Christensen, J.M.; Blot, W.J.; McLaughlin, J.K.; Olsen, J.H. Urinary concentrations of trichloroacetic acid in danish workers exposed to trichloroethylene. 1947–1985. Am. J. Ind. Med. 2001, 39, 320–327. [Google Scholar] [CrossRef]
- Mundt, K.A.; Birk, T.; Burch, M.T. Critical review of the epidemiological literature on occupational exposure to perchloroethylene and cancer. Int. Arch. Occup. Environ. Health 2003, 76, 473–491. [Google Scholar] [CrossRef]
- Aschengrau, A.; Weinberg, J.M.; Janulewicz, P.A.; Romano, M.E.; Gallagher, L.G.; Winter, M.R.; Martin, B.R.; Vieira, V.M.; Webster, T.F.; White, R.F. Affinity for risky behaviors following prenatal and early childhood exposure to tetrachloroethylene (pce)-contaminated drinking water: A retrospective cohort study. Environ. Health 2011, 10, 1–13. [Google Scholar] [CrossRef]
- USEPA. Fact Sheet on Perchloroethylene, also Known as Tetrachloroethylene; EPA: Washington, DC, USA, 2012. [Google Scholar]
- USEPA. Dry Cleaning Alternative Solvents: Health and Environmental Impacts; California Environmental Protection Agency Air Resource Board: Sacramento, CA, USA, 2012. [Google Scholar]
- USEPA. Case Study: Liquid Carbon Dioxide (co2) Surfactant System for Garment Care; EPA: Washington, DC, USA, 2012. [Google Scholar]
- Lee, M.K.C. 10.5 Synthetic Solvent Drycleaning, Permit Handbook; Bay Area Air Quality Management District: Washington, DC, US, 2007. [Google Scholar]
- Hoover, S. Comments on Human Health and Environmental Hazards for Dry Cleaning Solvents Included in sf Environment’s Alternatives Assessment Fact Sheet; Office of Environmental Health Hazard Assessment: Sacramento, CA, USA, 2012. [Google Scholar]
- NTP. Toxicology and carcinogenesis studies of propylene glycol mono-t-butyl ether (cas no. 57018-52-7) in f344/n rats and b6c3f1 mice and a toxicology study of propylene glycol mono-t-butyl ether in male nbr rats (inhalation studies). Natl. Toxicol. Progr. Tech. Rep. 2004, 515, 1–306. [Google Scholar]
- Cordier, S.; Multigner, L. Occupational exposure to glycol ethers and ovarian function. Occup. Environ. Med. 2005, 62, 507–508. [Google Scholar] [CrossRef]
- Cordier, S.; Garnier, R.; Gazin, V.; Multigner, L.; Vasseur, P.; Beausoleil, C.; Laudet-Hesbert, A.; Bonnin, F.; Bonvallot, N.; Chenu, C. Éthers de glycol: Nouvelles données toxicologiques; Inserm: Paris, France, 2006. [Google Scholar]
- Hsieh, G.; Wang, J.; Cheng, T.; Chen, P. Prolonged menstrual cycles in female workers exposed to ethylene glycol ethers in the semiconductor manufacturing industry. Occup. Environ. Med. 2005, 62, 510–516. [Google Scholar] [CrossRef]
- Staples, C.A.; Davis, J.W. An examination of the physical properties, fate, ecotoxicity and potential environmental risks for a series of propylene glycol ethers. Chemosphere 2002, 49, 61–73. [Google Scholar] [CrossRef]
- OECD Screening Information Data Set. Initial Assessment Report for Siam 17—Propylene Glycol Ethers; SIAM: Arona, Italy, 2003. [Google Scholar]
- Brown Dzubow, R.; Makris, S.; Siegel Scott, C.; Barone, S. Early lifestage exposure and potential developmental susceptibility to tetrachloroethylene. Birth Defects Res. Part B 2010, 89, 50–65. [Google Scholar]
- Murphy, I.P. Rather Switch than Fight? Available online: ttps://americandrycleaner.com/articles/rather-switch-fight-part-2-2 (accessed on 13 June 2012).
- Office of Pollution Prevention and Toxics U.S. Environmental Protection Agency. Chemical Summary for Perchloroethylene; EPA: Washington, DC, USA, 1994. [Google Scholar]
- Anhui Lixing Chemical Co., Ltd. Chemical Book, Di(Propylene Glycol) Tert-Butyl Ether Basic Information. Available online: http://www.chemicalbook.com/ProductIndex_EN.aspx (accessed on 1 June 2008).
- MacWilliam, L. The Dry Cleaning Dilemma. Available online: http://www.myhealthyhome.com/wp-content/uploads/2011/02/TheDryCleaningDilemmawReferences.pdf (accessed on 5 June 2010).
- Company, M.A. Material Safety Data Sheet-Pce. Available online: http://www.MartinAsphalt.com (accessed on 17 December 2006).
- Environmental Health Dept.-Grace Construction Products. Material Safety Data Sheet. Available online: http://www.na.graceconstruction.com/concrete/download/DARACMMR.PDF (accessed on 27 January 2000).
- EMD Millipore. Dipropylene Glycol Monomethyl Ether. Available online: http://www.emdmillipore.com/is-bin/INTERSHOP.enfinity/WFS/Merck-US-Site/en_CA/-/USD/ViewPDF-Print.pdf?RenderPageType=ProductDetail&CatalogCategoryID=CDSb.s1LnJwAAAEWgeEfVhTl&ProductUUID=FUCsHfETqKQAAAE2scxwtCII&PortalCatalogUUID=t02b.s1LX0MAAAEWc9UfVhTl (accessed on 25 March 2014).
- U.S. Department of Labor Occupational Safety and Health Administration. Reducing Worker Exposure to Perchloroethylene (perc) in Dry Cleaning. Available online: http://www.osha.gov/Publications/osha3253.html (accessed on 24 April 2005).
- USDHH. Centers for Disease Control and Prvention, National Institute for Occupational Safety and Health, Niosh Pocket Guide to Chemical Hazards; National Institute for Occupational Safety and Health: Cincinnati, OH, USA, 2010. [Google Scholar]
- USEPA. Cleaner Technologies Substitutes Assessment: Professional Fabricare Processes (Appendix c); EPA: Washington, DC, USA, 1998. [Google Scholar]
- Gold, L.S.; De Roos, A.J.; Waters, M.; Stewart, P. Systematic literature review of uses and levels of occupational exposure to tetrachloroethylene. J. Occup. Environ. Hyg. 2008, 5, 807–839. [Google Scholar]
- Materna, B.L. Occupational exposure to perchloroethylene in the dry cleaning industry. Am. Ind. Hyg. Assoc. J. 1985, 46, 268–273. [Google Scholar] [CrossRef]
- Räisänen, J.; Niemelä, R.; Rosenberg, C. Tetrachloroethylene emissions and exposure in dry cleaning. J. Air Waste Manag. Assoc. 2001, 51, 1671–1675. [Google Scholar] [CrossRef]
- McKernan, L.T.; Ruder, A.M.; Petersen, M.R.; Hein, M.J.; Forrester, C.L.; Sanderson, W.T.; Ashley, D.L.; Butler, M.A. Biological exposure assessment to tetrachloroethylene for workers in the dry cleaning industry. Environ. Health 2008, 7, 1–10. [Google Scholar]
- California Department of Public Health Division of Environmental and Occupational Disease Control Occupational Health Branch. Glycol ethers. Hazard Evaluation System and Information System, Fact Sheet; California Department of Public Health Division of Environmental and Occupational Disease Control Occupational Health Branch: Sacramento, CA, USA, 2009. [Google Scholar]
- Permissible Exposure Limits for Chemical Contaminants. Available online: https://www.dir.ca.gov/title8/ac1.pdf (accessed on 15 August 1994).
- Edney, E.; Kleindienst, T.; Corse, E. Room temperature rate constants for the reaction of oh with selected chlorinated and oxygenated hydrocarbons. Int. J. Chem. Kinet. 1986, 18, 1355–1371. [Google Scholar]
- Tuazon, E.C.; Atkinson, R.; Aschmann, S.M.; Goodman, M.A.; Winer, A.M. Atmospheric reactions of chloroethenes with the oh radical. Int.J. Chem. Kinet. 1988, 20, 241–265. [Google Scholar] [CrossRef]
- Chiao, F.; Currie, R.; McKone, T. Intermedia Transfer Factors for Contaminants Found at Hazardous Waste Sites: Trichloroethylene (tce). Final Draft Report, Risk Science Program; Department of Environmental Toxicology, University of California: Davis, CA, USA, 1994. [Google Scholar]
- Sherlach, K.S.; Gorka, A.P.; Dantzler, A.; Roepe, P.D. Quantification of perchloroethylene residues in dry-cleaned fabrics. Environ. Toxicol. Chem. 2011, 30, 2481–2487. [Google Scholar] [CrossRef]
- USEPA. Toxicological Review of Tetrachloroethylene (Perchloroethylene); EPA: Washington, DC, USA, 2012. [Google Scholar]
- Agency for Toxic Substances and Registry (ASTDR). Toxicological Profile for Tetrachloroethylene; ASTDR: Atlanta, GA, USA, 2012. [Google Scholar]
- Tansel, B.; Eyma, R.R. Volatile organic contaminant emissions from wastewater treatment plants during secondary treatment. Water Air Soil Pollut. 1999, 112, 315–325. [Google Scholar] [CrossRef]
- Science in the Courtroom. TCE and PCE Toxicity, Uses, and Properties. Available online: http://serc.carleton.edu/woburn/issues/tce_toxicity.html (accessed on 18 June 2009).
- Kido, K.; Shiratori, T.; Watanabe, T.; Nakatsuka, H.; Ohashi, M.; Ikeda, M. Correlation of tetrachloroethylene in blood and in drinking water: A case of well water pollution. Bull. Environ. Contam. Toxicol. 1989, 43, 444–453. [Google Scholar]
- Canter, L.W.; Sabatini, D.A. Contamination of public ground water supplies by superfund sites. Int. J. Environ. Stud. 1994, 46, 35–57. [Google Scholar] [CrossRef]
- USEPA. Basic Information about Tetrachloroethylene in Drinking Water; EPA: Washington, DC, USA, 2012. [Google Scholar]
- Schreiber, J.S.; Hudnell, H.K.; Geller, A.M.; House, D.E.; Aldous, K.M.; Force, M.S.; Langguth, K.; Prohonic, E.J.; Parker, J.C. Apartment residents’ and day care workers’ exposures to tetrachloroethylene and deficits in visual contrast sensitivity. Environ. Health Perspect. 2002, 110, 655. [Google Scholar]
- McDermott, M.J.; Mazor, K.A.; Shost, S.J.; Narang, R.S.; Aldous, K.M.; Storm, J.E. Tetrachloroethylene (pce, perc) levels in residential dry cleaner buildings in diverse communities in New York city. Environ. Health Perspect. 2005, 113, 1336. [Google Scholar] [CrossRef]
- Atkinson, R. Gas-phase tropospheric chemistry of organic compounds: A review. Atmos. Environ. Part A 1990, 24, 1–41. [Google Scholar] [CrossRef]
- Carter, W.P. Development of ozone reactivity scales for volatile organic compounds. Air Waste 1994, 44, 881–899. [Google Scholar] [CrossRef]
- Derwent, R.; Jenkin, M.; Saunders, S. Photochemical ozone creation potentials for a large number of reactive hydrocarbons under european conditions. Atmos. Environ. 1996, 30, 181–199. [Google Scholar] [CrossRef]
- Chang, C.-C.; Chen, T.-Y.; Lin, C.-Y.; Yuan, C.-S.; Liu, S.-C. Effects of reactive hydrocarbons on ozone formation in southern taiwan. Atmos. Environ. 2005, 39, 2867–2878. [Google Scholar] [CrossRef]
- Cheng, H.; Guo, H.; Saunders, S.; Lam, S.; Jiang, F.; Wang, X.; Simpson, I.; Blake, D.; Louie, P.; Wang, T. Assessing photochemical ozone formation in the pearl river delta with a photochemical trajectory model. Atmos. Environ. 2010, 44, 4199–4208. [Google Scholar] [CrossRef]
- Derwent, R.; Jenkin, M.; Passant, N.; Pilling, M. Reactivity-based strategies for photochemical ozone control in Europe. Environ. Sci. Policy 2007, 10, 445–453. [Google Scholar] [CrossRef]
- Evtyugina, M.; Pio, C.; Nunes, T.; Pinho, P.; Costa, C. Photochemical ozone formation at Portugal west coast under sea breeze conditions as assessed by master chemical mechanism model. Atmos. Environ. 2007, 41, 2171–2182. [Google Scholar] [CrossRef]
- Pinho, P.; Lemos, L.; Pio, C.; Evtyugina, M.; Nunes, T.; Jenkin, M. Detailed chemical analysis of regional-scale air pollution in western portugal using an adapted version of mcm v3. 1. Sci. Total Environ. 2009, 407, 2024–2038. [Google Scholar] [CrossRef]
- Saunders, S.; Jenkin, M.; Derwent, R.; Pilling, M. Protocol for the development of the master chemical mechanism, mcm v3 (part a): Tropospheric degradation of non-aromatic volatile organic compounds. Atmos. Chem. Phys. 2003, 3, 161–180. [Google Scholar] [CrossRef]
- Derwent, D. Reactivity Scales as Comparative Tools for Chemical Mechanisms: SAPRC vs. MCM. Available online: http://www.arb.ca.gov/research/reactivity/3-25-2009/derwent-present.pdf (accessed on 28 March 2009).
- Altenstedt, J.; Karin, P. POCP for Individual VOC under European Conditions; Swedish Environmental Research Institute: Stockholm, Sweden, 1998. [Google Scholar]
- Koppmann, R. Volatile Organic Compounds in the Atmosphere; John Wiley & Sons: Hoboken, NJ, USA, 2008; pp. 1–512. [Google Scholar]
- Morris, M.; Wolf, K. Evaluation of New and Emerging Technologies for Textile Cleaning. Available online: https://dtsc.ca.gov/PollutionPrevention/upload/P2_REP_Emerging_Technology_Textile_Cleaning.pdf (accessed on 1 August 2005).
- The Dow Chemical Company. Product Safety Assessment Dipropylene Glycol n-Butyl Ether; The Dow Chemical Company: Midland, MI, USA, 2008. [Google Scholar]
- CALED. Caled better Cleaning with Chemistry. Available online: http://www.caledclean.com/index.cfm/category/37/solvents.cfm (accessed on 1 August 2014).
- Rynex. Rynex-3 Dry Cleaning Solvent Equinox Chemicals. Available online: http://rynex.com/PDF/Rynex%20MSDS%20WEBSITE.pdf (accessed on 10 November 2010).
- UNION. Drycleaning Products, Perchloreoethylene machines L & P 800 series. Available online: http://www.uniondc.com/products/union-products-perc-lp800.html (accessed on 17 August 2012).
- Index, C.P. Archived Consumer Price Index Detailed Report Information. Available online: http://www.bls.gov/cpi/cpi_dr.htm (accessed on 1 May 2007).
- Anonymous. Assessment of Alternatives to Perchloroethylene for the Dry Cleaning Industry; Toxic Use Reduction Institute: University of Massachusetts, Lowell, 2007. [Google Scholar]
- Wolf, K.; Morris, M. Spotting Chemicals: Alternatives to Perchloroethylene and Trichloroethylene in the Textile Cleaning Industry; Institute for Research and Technical Assistance: Los Angeles, CA, USA, 2007. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hesari, N.; Francis, C.M.; Halden, R.U. Evaluation of Glycol Ether as an Alternative to Perchloroethylene in Dry Cleaning. Toxics 2014, 2, 115-133. https://doi.org/10.3390/toxics2020115
Hesari N, Francis CM, Halden RU. Evaluation of Glycol Ether as an Alternative to Perchloroethylene in Dry Cleaning. Toxics. 2014; 2(2):115-133. https://doi.org/10.3390/toxics2020115
Chicago/Turabian StyleHesari, Nikou, Chelsea M. Francis, and Rolf U. Halden. 2014. "Evaluation of Glycol Ether as an Alternative to Perchloroethylene in Dry Cleaning" Toxics 2, no. 2: 115-133. https://doi.org/10.3390/toxics2020115
APA StyleHesari, N., Francis, C. M., & Halden, R. U. (2014). Evaluation of Glycol Ether as an Alternative to Perchloroethylene in Dry Cleaning. Toxics, 2(2), 115-133. https://doi.org/10.3390/toxics2020115