Ipconazole Induces Oxidative Stress, Cell Death, and Proinflammation in SH-SY5Y Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Cell Culture
2.3. Cell Viability Evaluation (MTT)
2.4. ROS Production
2.5. Apoptotic Assay with Caspase3/7 Activity
2.6. Molecular Assay by Real-Time PCR
- Bax (Bcl-2-associated X protein): ‘CCCCCGAGAGGTCTTTTTCC’ ‘CCTTGAGCACCAGTTTGCTG’
- Casp3 (Caspase 3): ‘GTGGAGGCCGACTTCTTGTA’ ‘TTTCAGCATGGCACAAAGCG’
- APAF1 (Apoptotic protease-activating factor 1): ‘TCTTCCAGTGGTAAAGATTCAGTT’ ‘CGGAGACGGTCTTTAGCCTC’
- BNIP3 (BCL2-interacting protein 3): ‘CCTCAGCATGAGGAACACGA’ ‘GCCACCCCAGGATCTAACAG’
- Bcl2 (B-cell lymphoma 2): ‘TCTCATGCCAAGGGGGAAAC’ ‘TCCCGGTTATCGTACCCTGT’
- NLRP3 (NLR family pyrin domain containing 3): ‘CCCCGTAATCAACGGGACAA’ ‘AGCCAAATGCTTACCAGAAAGT’
- Casp1 (Caspase 1): ‘GAAAAGCCATGGCCGACAAG’ ‘GCCCCTTTCGGAATAACGGA’
- IL1β (Interleukin-1 beta): ‘CCAGCTACGAATCTCCGACC’ ‘TATCCTGTCCCTGGAGGTGG’
- NFκB (Nuclear factor kappa B): ‘TTTTCGACTACGCGGTGACA’ ‘GTTACCCAAGCGGTCCAGAA’
- TNFα (Tumor necrosis factor alpha): ‘CTGGAAAGGACACCATGAGCA’‘TCTCTCAGCTCCACGCCATT’
- IL6 (Interleukin 6): ‘CCAGTACCCCCAGGAGAAGA’ ‘CAGCTCTGGCTTGTTCCTCA’
- NRF2 (Nuclear factor erythroid 2-related factor 2): ‘CTGGTCATCGGAAAACCCCA’ ‘TCTGCAATTCTGAGCAGCCA’
- SOD (Superoxide dismutase): ‘CCACTGCTGGGGATTGATGT’ ‘CGTGGTTTACTTTTTGCAAGCC’
- GPx (Glutathione peroxidase): ’TTCGAGCCCAACTTCATGCT’ ‘CGATGTCAGGCTCGATGTCA’.
- GAPDH (glyceraldehyde-3-phosphate dehydrogenase): (‘GAGAAGGCTGGGGCTCATTT‘ ‘AGTGATGGCATGGACTGTGG’) was used as a housekeeping gene. We extracted the efficiencies from the raw data using LinRegPCR software 20210614 [16].
2.7. Statistics
3. Results
3.1. Effect of Ipconazole on SH-SY5Y Cell Viability
3.2. Effect of Ipconazole on ROS Production in SH-SY5Y Cells
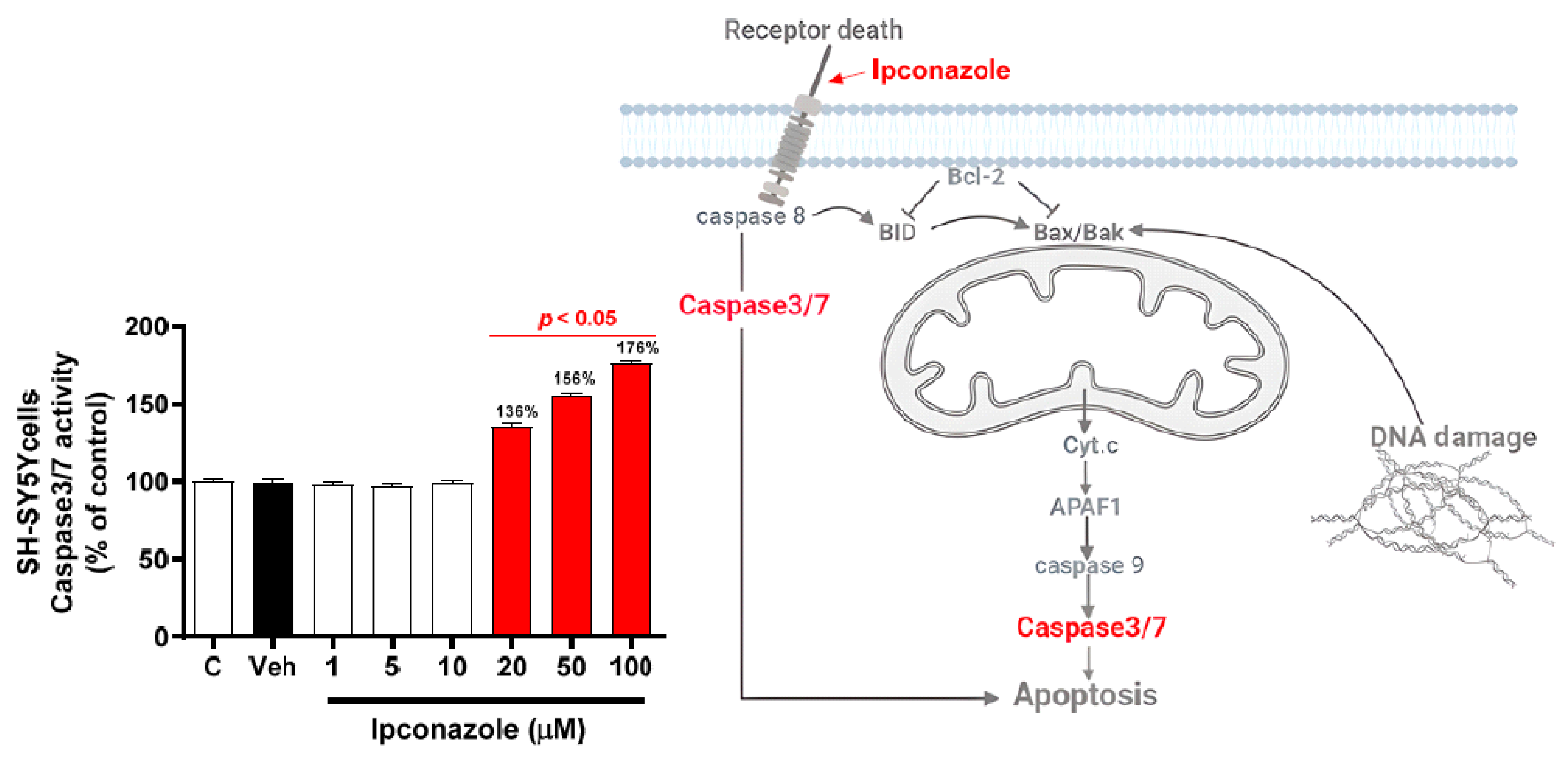
3.3. Effect of Ipconazole on Caspase3/7 Activity in SH-SY5Y Cells
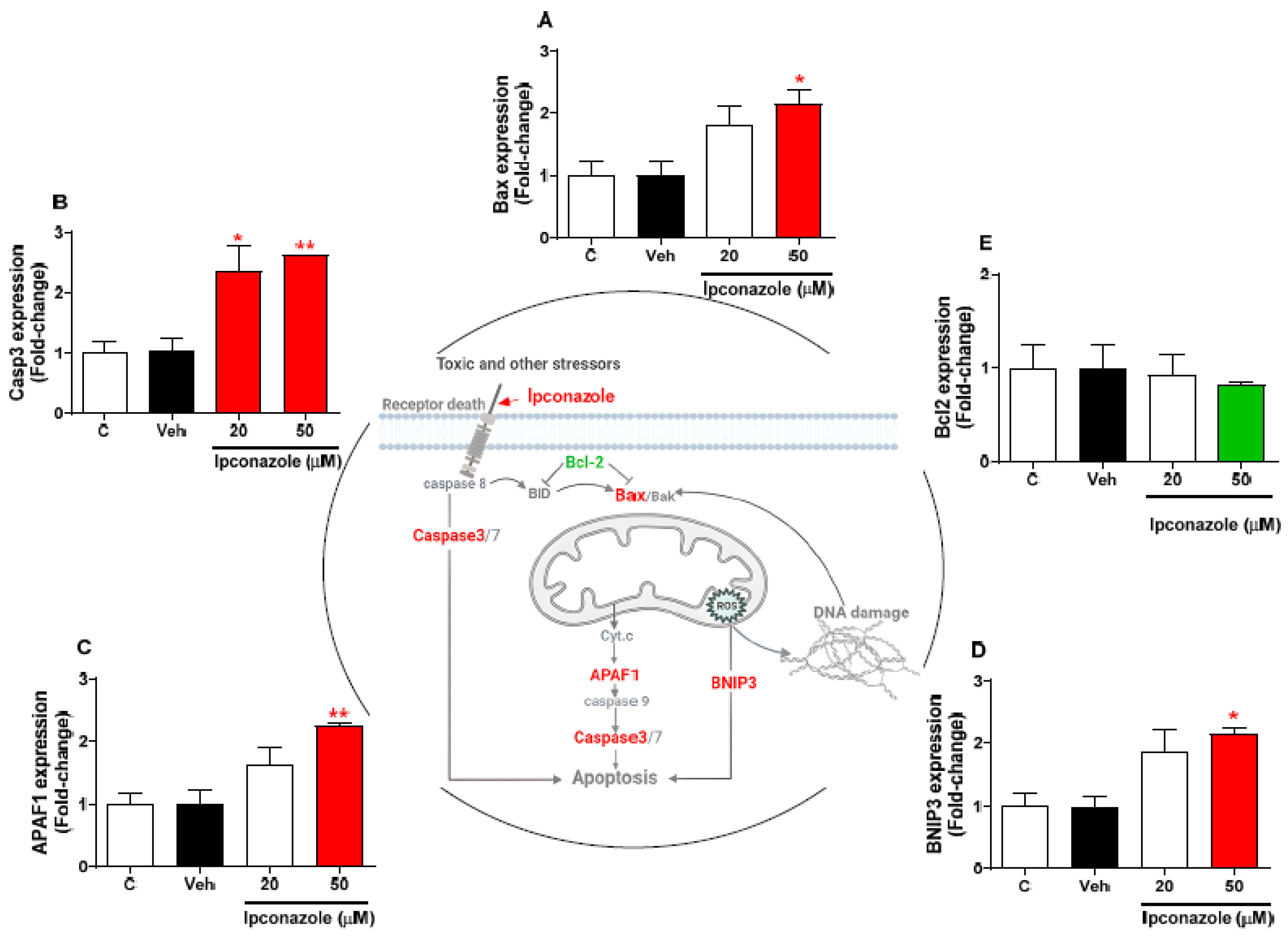
3.4. Effect of Ipconazole on Cell-Death-Related Gene Expression in SH-SY5Y Cells
3.5. Effect of Ipconazole on the Expression of Inflammasome-Related Genes in SH-SY5Y Cells
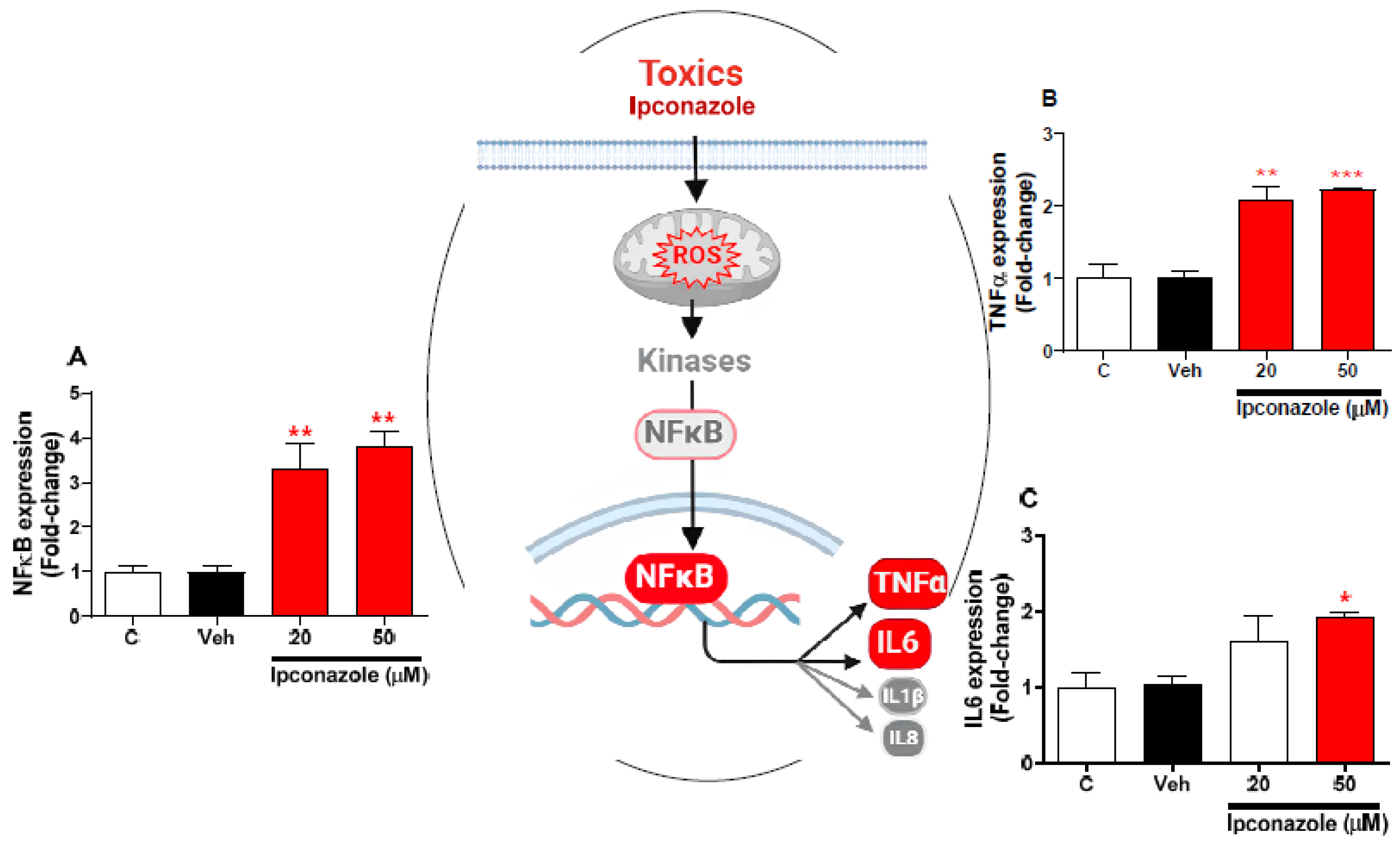
3.6. Effect of Ipconazole on the Expression of Inflammation-Related Genes in SH-SY5Y Cells
3.7. Effect of Ipconazole on the Expression of Antioxidant-Related Genes in SH-SY5Y Cells
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bernardes, M.F.F.; Pazin, M.; Pereira, L.C.; Dorta, D.J. Toxicology Studies-Cells, Drugs and Environment. In Impact of Pesticides on Environmental and Human Health; IntechOpen: London, UK, 2015; pp. 195–233. [Google Scholar]
- Hayes, T.B.; Hansen, M.; Kapuscinski, A.R.; Locke, K.A.; Barnosky, A. From silent spring to silent night: Agrochemicals and the anthropocene. Elem. Sci. Anth. 2017, 5, 57. [Google Scholar] [CrossRef]
- Hernández, A.F.; Gil, F.; Lacasaña, M.; Rodríguez-Barranco, M.; Tsatsakis, A.M.; Requena, M.; Alarcón, R. Pesticide exposure and genetic variation in xenobiotic-metabolizing enzymes interact to induce biochemical liver damage. Food Chem. Toxicol. 2013, 61, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Petit, A.N.; Fontaine, F.; Vatsa, P.; Clément, C.; Vaillant-Gaveau, N. Fungicide impacts on photosynthesis in crop plants. Photosynth. Res. 2012, 111, 315–326. [Google Scholar] [CrossRef] [PubMed]
- Mosiichuk, N.; Husak, V.; Storey, K.B.; Lushchak, V. Acute Exposure to the Penconazole-Containing Fungicide Topas Induces Metabolic Stress in Goldfish. Chem. Res. Toxicol. 2021, 34, 2441–2449. [Google Scholar] [CrossRef]
- Chaâbane, M.; Tir, M.; Hamdi, S.; Boudawara, O.; Jamoussi, K.; Boudawara, T.; Ghorbel, R.E.; Zeghal, N.; Soudani, N. Improvement of heart redox states contributes to the beneficial effects of selenium against penconazole-induced cardiotoxicity in adult rats. Biol. Trace Elem. Res. 2016, 169, 261–270. [Google Scholar] [CrossRef]
- Kouzayha, A.; Rahman Rabaa, A.; Al Iskandarani, M.; Beh, D.; Budzinski, H.; Jaber, F. Multiresidue method for determination of 67 pesticides in water samples using solid-phase extraction with centrifugation and gas chromatography-mass spectrometry. Am. J. Anal. Chem. 2012, 3, 257–265. [Google Scholar] [CrossRef]
- Thrikawala, S.; Mesmar, F.; Bhattacharya, B.; Muhsen, M.; Mukhopadhyay, S.; Flores, S.; Upadhyay, S.; Vergara, L.; Gustafsson, J.Å.; Williams, C.; et al. Triazole fungicides induce adipogenesis and repress osteoblastogenesis in zebrafish. Toxicol. Sci. 2023, 132, 119–130. [Google Scholar] [CrossRef]
- Santos-Miranda, A.; Joviano-Santos, J.V.; Cruz-Nascimento, T.; Neri, E.A.; Souza, D.S.; Marques, L.P.; Krieger, J.E.; Roman-Campos, D. The fungicide Tebuconazole induces electromechanical cardiotoxicity in murine heart and human cardiomyocytes derived from induced pluripotent stem cells. Toxicol. Lett. 2022, 359, 96–105. [Google Scholar] [CrossRef]
- Saishoji, T.; Ito, A.; Kumazawa, S.; Chuman, H. Structure-activity relationships of enantiomers of the azole fungicide ipconazole and its related compounds: Fungicidal and plant growth inhibitory activities. J. Pestic. Sci. 1998, 23, 129–136. [Google Scholar] [CrossRef]
- Authority, E.F.S. Statement concerning the review of the approval of the active substance ipconazole. EFSA J. 2022, 20, e07133. [Google Scholar]
- Penagos-Tabares, F.; Sulyok, M.; Faas, J.; Krska, R.; Khiaosa-Ard, R.; Zebeli, Q. Residues of pesticides and veterinary drugs in diets of dairy cattle from conventional and organic farms in Austria. Environ. Pollut. 2023, 316 Pt 2, 120626. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.; Banik, A.; Eum, J.; Hwang, B.J.; Kwon, S.H.; Kee, Y. Ipconazole Disrupts Mitochondrial Homeostasis and Alters GABAergic Neuronal Development in Zebrafish. Int. J. Mol. Sci. 2022, 24, 496. [Google Scholar] [CrossRef] [PubMed]
- Barrios-Arpi, L.; Arias, Y.; Lopez-Torres, B.; Ramos-Gonzalez, M.; Ticli, G.; Prosperi, E.; Rodríguez, J.-L. In Vitro Neurotoxicity of Flumethrin Pyrethroid on SH-SY5Y Neuroblastoma Cells: Apoptosis Associated with Oxidative Stress. Toxics 2022, 10, 131. [Google Scholar] [CrossRef] [PubMed]
- Castillo, G.; Barrios-Arpi, L.; Ramos-Gonzalez, M.; Vidal, P.; Gonzales-Irribarren, A.; Ramos-Cevallos, N.; Rodríguez, J.L. Neurotoxicity associated with oxidative stress and inflammasome gene expression induced by allethrin in SH-SY5Y cells. Toxicol. Ind. Health 2022, 38, 777–788. [Google Scholar] [CrossRef] [PubMed]
- Ramakers, C.; Ruijter, J.M.; Deprez, R.H.L.; Moorman, A.F.M. Assumption-free analysis of quantitative real-time polymerase chain reaction (PCR) data. Neurosci. Lett. 2003, 339, 62–66. [Google Scholar] [CrossRef]
- Holmström, K.M.; Finkel, T. Cellular mechanisms and physiological consequences of redox-dependent signalling. Nat. Rev. Mol. Cell Biol. 2014, 15, 411–421. [Google Scholar] [CrossRef]
- Bock, F.J.; Tait, S.W.G. Mitochondria as multifaceted regulators of cell death. Nat. Rev. Mol. Cell Biol. 2020, 21, 85–100. [Google Scholar] [CrossRef]
- Kelley, N.; Jeltema, D.; Duan, Y.; He, Y. The NLRP3 Inflammasome: An Overview of Mechanisms of Activation and Regulation. Int. J. Mol. Sci. 2019, 20, 3328. [Google Scholar] [CrossRef]
- Minatel, I.O.; Francisqueti, F.V.; Corrêa, C.R.; Lima, G.P.P. Antioxidant Activity of γ-Oryzanol: A Complex Network of Interactions. Int. J. Mol. Sci. 2016, 17, 1107. [Google Scholar] [CrossRef]
- Vasconcelos, A.R.; Dos Santos, N.B.; Scavone, C.; Munhoz, C.D. Nrf2/ARE Pathway Modulation by Dietary Energy Regulation in Neurological Disorders. Front. Pharmacol. 2019, 10, 33. [Google Scholar] [CrossRef]
- Hamdi, H.; Rjiba-Touati, K.; Ayed-Boussema, I.; M’nassri, A.; Chaabani, H.; Rich, S.; Abid-Essefi, S. Epoxiconazole caused oxidative stress related DNA damage and apoptosis in PC12 rat Pheochromocytoma. Neuro Toxicol. 2022, 89, 184–190. [Google Scholar] [CrossRef]
- Ray, P.D.; Bo-Wen, H.; Yoshiaki, T. Reactive oxygen species (ROS) homeostasis and redox regulation in cellular signaling. Cell. Signal. 2012, 24, 981–990. [Google Scholar] [CrossRef]
- Hamdi, H.; Graiet, I.; Abid-Essefi, S.; Eyer, J. Epoxiconazole profoundly alters rat brain and properties of neural stem cells. Chemosphere 2022, 288 Pt 3, 132640. [Google Scholar] [CrossRef]
- Nieradko-Iwanicka, B.; Borzecki, A. Subacute poisoning of mice with deltamethrin produces memory impairment reduced locomotor activity, liver damage and changes in blood morphology in the mechanism of oxidative stress. Pharmacol. Rep. 2015, 67, 535–541. [Google Scholar] [CrossRef] [PubMed]
- Loh, K.P.; Huang, S.H.; De Silva, R.R.; Tan, B.K.H.; Zhu, Y.Z. Oxidative stress: Apoptosis in neuronal injury. Curr. Alzheimer Res. 2006, 3, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Alam, R.T.; Imam, T.S.; Abo-Elmaaty, A.M.A.; Arisha, A.H. Amelioration of fenitrothion induced oxidative DNA damage and inactivation of caspase-3 in the brain and spleen tissues of male rats by N-acetylcysteine. Life Sci. 2019, 231, 116534. [Google Scholar] [CrossRef] [PubMed]
- Othmène, Y.B.; Hamdi, H.; Salem, I.B.; Annabi, E.; Amara, I.; Neffati, F.; Najjar, M.F.; Abid-Essefi, S. Oxidative stress, DNA damage and apoptosis induced by tebuconazole in the kidney of male Wistar rat. Chem. Biol. Interact. 2020, 330, 109114. [Google Scholar] [CrossRef]
- Brentnall, M.; Rodríguez-Menocal, L.; De Guevara, R.L.; Cepero, E.; Boise, L.H. Caspasa-9, caspasa-3 y caspasa-7 tienen funciones distintas durante la apoptosis intrínseca. BMC Cell Biol. 2013, 14, 32. [Google Scholar] [CrossRef]
- Kannan, K.; Jain, S.K. Oxidative stress and apoptosis. Pathophysiology 2000, 73, 153–163. [Google Scholar] [CrossRef]
- Salakou, S.; Kardamakis, D.; Tsamandas, A.; Zolota, V.; Apostolakis, E.; Tzelepi, V.; Papathanasopoulos, P.; Bonikos, D.; Papapetropoulos, T.; Petsas, T.; et al. Increased Bax/Bcl-2 ratio up-regulates caspase-3 and increases apoptosis in the thymus of patients with myasthenia gravis. In Vivo 2007, 21, 123–132. [Google Scholar]
- Ola, M.S.; Nawaz, W.; Ahsan, H. Role of Bcl-2 family proteins and caspases in the regulation of apoptosis. Mol. Cell. Biochem. 2011, 351, 41–58. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, S.; Narita, M.; Tsujimoto, Y. Bcl-2 family proteins regulate the release of apoptogenic cytochrome c by the mitochondrial channel VDAC. Nature 1999, 399, 483–487. [Google Scholar] [CrossRef] [PubMed]
- Green, D.R.; Reed, J.C. Mitochondria and apoptosis. Science 1998, 281, 1309–1312. [Google Scholar] [CrossRef]
- Lei, Q.; Yi, T.; Chen, C. NF-kappaB-Gasdermin D (GSDMD) axis couples oxidative stress and NACHT, LRR and PYD Domains-Containing Protein 3 (NLRP3) inflammasome-mediated cardiomyocyte pyroptosis following myocardial infarction. Med. Sci. Monit. 2018, 24, 6044–6052. [Google Scholar] [CrossRef]
- Shi, J.; Zhao, Y.; Wang, K.; Shi, X.; Wang, Y.; Huang, H.; Zhuan, Y.; Cai, T.; Wang, F.; Shao, F. Cleavage of GSDMD by inflammatory caspases determines pyroptotic cell death. Nature 2015, 526, 660–665. [Google Scholar] [CrossRef]
- He, W.; Wan, H.; Hu, L.; Chen, P.; Wang, X.; Huang, Z.; Yang, Z.; Zhong, C.; Han, J. Gasdermin D is an executor of pyroptosis and required for interleukin-1beta secretion. Cell Res. 2015, 25, 1285–1298. [Google Scholar] [CrossRef] [PubMed]
- Dostert, C.; Pétrilli, V.; Bruggen, R.V.; Steele, C.; Mossman, B.T.; Tschopp, J. Innate Immune Activation through Nalp3 Inflammasome Sensing of Asbestos and Silica. Science 2008, 320, 674–677. [Google Scholar] [CrossRef]
- Cruz, C.M.; Rinna, A.; Forman, H.J.; Ventura, A.L.M.; Persechini, P.M.; Ojcius, D.M. ATP activates a reactive oxygen species-dependent oxidative stress response and secretion of proinflammatory cytokines in macrophages. J. Biol. Chem. 2007, 282, 2871–2879. [Google Scholar] [CrossRef]
- Zhou, R.; Yazdi, A.S.; Menu, P.; Tschopp, J. A role for mitochondria in NLRP3 inflammasome activation. Nature 2011, 469, 221–225. [Google Scholar] [CrossRef]
- Nakahira, K.; Haspel, J.A.; Rathinam, V.A.K.; Lee, S.; Dolinay, T.; Lam, H.C.; Englert, J.A.; Rabinovitch, M.; Cernadas, M.; Kim, H.P.; et al. Autophagy proteins regulate innate immune responses by inhibiting the release of mitochondrial DNA mediated by the NALP3 inflammasome. Nat. Immunol. 2011, 12, 222–230. [Google Scholar] [CrossRef]
- Parajuli, B.; Sonobe, Y.; Horiuchi, H.; Takeuchi, H.; Mizuno, T.; Suzumura, A. Oligomeric amyloid β induces IL-1β processing via production of ROS: Implication in Alzheimer’s disease. Cell Death Dis. 2013, 4, e975. [Google Scholar] [CrossRef]
- Salminen, A.; Ojala, J.; Suuronen, T.; Kaarniranta, K.; Kauppinen, A. Amyloid-beta oligomers set fire to inflammasomes and induce Alzheimer’s pathology. J. Cell Mol. Med. 2008, 12, 2255–2262. [Google Scholar] [CrossRef]
- Saresella, M.; La Rosa, F.; Piancone, F.; Zoppis, M.; Marventano, I.; Calabrese, E.; Rainone, V.; Nemni, R.; Mancuso, R.; Clerici, M. The NLRP3 and NLRP1 inflammasomes are activated in Alzheimer’s disease. Mol. Neurod. 2016, 11, 23. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.S.; Tan, L.; Jiang, T.; Zhu, X.C.; Wang, H.F.; Jia, C.D.; Yu, J.T. Amyloid-β induces NLRP1-dependent neuronal pyroptosis in models of Alzheimer’s disease. Cell Death Dis. 2014, 5, e1382. [Google Scholar] [CrossRef] [PubMed]
- Masters, S.L.; O’Neill, L.A. Disease-associated amyloid and misfolded protein aggregates activate the inflammasome. Trends Mol. Med. 2011, 17, 276–282. [Google Scholar] [CrossRef]
- Bu, X.L.; Wang, X.; Xiang, Y.; Shen, L.L.; Wang, Q.H.; Liu, Y.H.; Jiao, S.S.; Wang, Y.R.; Cao, H.Y.; Yi, X.; et al. The association between infec-tious burden and Parkinson’s disease: A case-control study. Park. Relat. Disord. 2015, 21, 877–881. [Google Scholar] [CrossRef]
- Zhou, Y.; Lu, M.; Du, R.H.; Qiao, C.; Jiang, C.Y.; Zhang, K.Z.; Ding, J.H.; Hu, G. MicroRNA-7 tar-gets Nod-like receptor protein 3 inflammasome to modulate neuroinflammation in the pathogenesis of Parkinson’s disease. Mol. Neurodegener. 2016, 11, 28. [Google Scholar] [CrossRef] [PubMed]
- Simpson, D.S.A.; Oliver, P.L. ROS Generation in Microglia: Understanding Oxidative Stress and Inflammation in Neurodegenerative Disease. Antioxidants 2020, 9, 743. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.M.; Toltin, A.C.; Gamba, L.M.; Molina, M.A. Deltamethrin-evoked ER stress promotes neuroinflammation in the adult mouse hippocampus. Cells 2022, 11, 1961. [Google Scholar] [CrossRef]
- Chen, L.; Na, R.; Boldt, E.; Ran, Q. NLRP3 inflammasome activation by mitochondrial reactive oxygen species plays a key role in long-term cognitive impairment induced by paraquat exposure. Neurobiol. Aging 2015, 36, 2533–2543. [Google Scholar] [CrossRef]
- Gargouri, B.; Bhatia, H.S.; Bouchard, M.; Fiebich, B.L.; Fetoui, H. Inflammatory and oxidative mechanisms potentiate bifenthrin-induced neurological alterations and anxiety-like behavior in adult rats. Toxicol. Lett. 2018, 294, 73–86. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Wang, F.; Jia, L.; Chai, R.; Wang, H.; Wang, X. 2,4-dichlorophenoxyacetic acid induces ROS activation in NLRP3 inflammatory body-induced autophagy disorder in microglia and the protective effect of Lycium barbarum polysaccharide. Environ. Toxicol. 2022, 37, 1136,1151. [Google Scholar] [CrossRef] [PubMed]
- Ćupić Miladinović, D.; Prevendar Crnić, A.; Peković, S.; Dacić, S.; Ivanović, S.; Santibanez, J.F. Recovery of brain cholinesterases and effect on parameters of oxidative stres and apoptosis in quails (Coturnix japonica) after chlorpyrifos and vitamin B1 administration. Chem. Biol. Interact. 2021, 333, 109312. [Google Scholar] [CrossRef] [PubMed]
- Nieradko-Iwanicka, B.; Konopelko, M. Effect of lambdacyhalothrin on locomotor activity, memory, selected biochemical parameters, tumor necrosis factor α, and interleukin 1β in a mouse model. Int. J. Environ. Res. Public Health 2020, 17, 9240. [Google Scholar] [CrossRef]
- Tebay, L.E.; Robertson, H.; Durant, S.T.; Vitale, S.R.; Penning, T.M.; Dinkova-Kostova, A.T. Mechanisms of activation of the transcription factor Nrf2 by redox stressors, nutrient cues, and energy status and the pathways through which it attenuates degenerative disease. Free Radic. Biol. Med. 2015, 88 Pt B, 108–146. [Google Scholar] [CrossRef]
- Nguyen, T.; Nioi, P.; Pickett, C.B. The Nrf2-antioxidant response element signaling pathway and its activation by oxidative stress. J. Biol. Chem. 2009, 284, 13291–13295. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Villaorduña, C.; Mendoza-Carlos, M.; Chuyma, M.; Avilés, J.; Avalos-Diaz, A.; Lozano-Reategui, R.; Garcia-Ruiz, J.; Panduro-Tenazoa, N.; Vargas, J.; Moran-Quintanilla, Y.; et al. Ipconazole Induces Oxidative Stress, Cell Death, and Proinflammation in SH-SY5Y Cells. Toxics 2023, 11, 566. https://doi.org/10.3390/toxics11070566
Villaorduña C, Mendoza-Carlos M, Chuyma M, Avilés J, Avalos-Diaz A, Lozano-Reategui R, Garcia-Ruiz J, Panduro-Tenazoa N, Vargas J, Moran-Quintanilla Y, et al. Ipconazole Induces Oxidative Stress, Cell Death, and Proinflammation in SH-SY5Y Cells. Toxics. 2023; 11(7):566. https://doi.org/10.3390/toxics11070566
Chicago/Turabian StyleVillaorduña, Carlos, Mariano Mendoza-Carlos, Manuel Chuyma, Jhon Avilés, Ayda Avalos-Diaz, Ronald Lozano-Reategui, Juan Garcia-Ruiz, Nadia Panduro-Tenazoa, Jessy Vargas, Ysabel Moran-Quintanilla, and et al. 2023. "Ipconazole Induces Oxidative Stress, Cell Death, and Proinflammation in SH-SY5Y Cells" Toxics 11, no. 7: 566. https://doi.org/10.3390/toxics11070566
APA StyleVillaorduña, C., Mendoza-Carlos, M., Chuyma, M., Avilés, J., Avalos-Diaz, A., Lozano-Reategui, R., Garcia-Ruiz, J., Panduro-Tenazoa, N., Vargas, J., Moran-Quintanilla, Y., & Rodríguez, J.-L. (2023). Ipconazole Induces Oxidative Stress, Cell Death, and Proinflammation in SH-SY5Y Cells. Toxics, 11(7), 566. https://doi.org/10.3390/toxics11070566








