Freshwater Fish Siberian Dace Ingest Microplastics in the Remote Yenisei Tributary
Abstract
1. Introduction
2. Materials and Methods
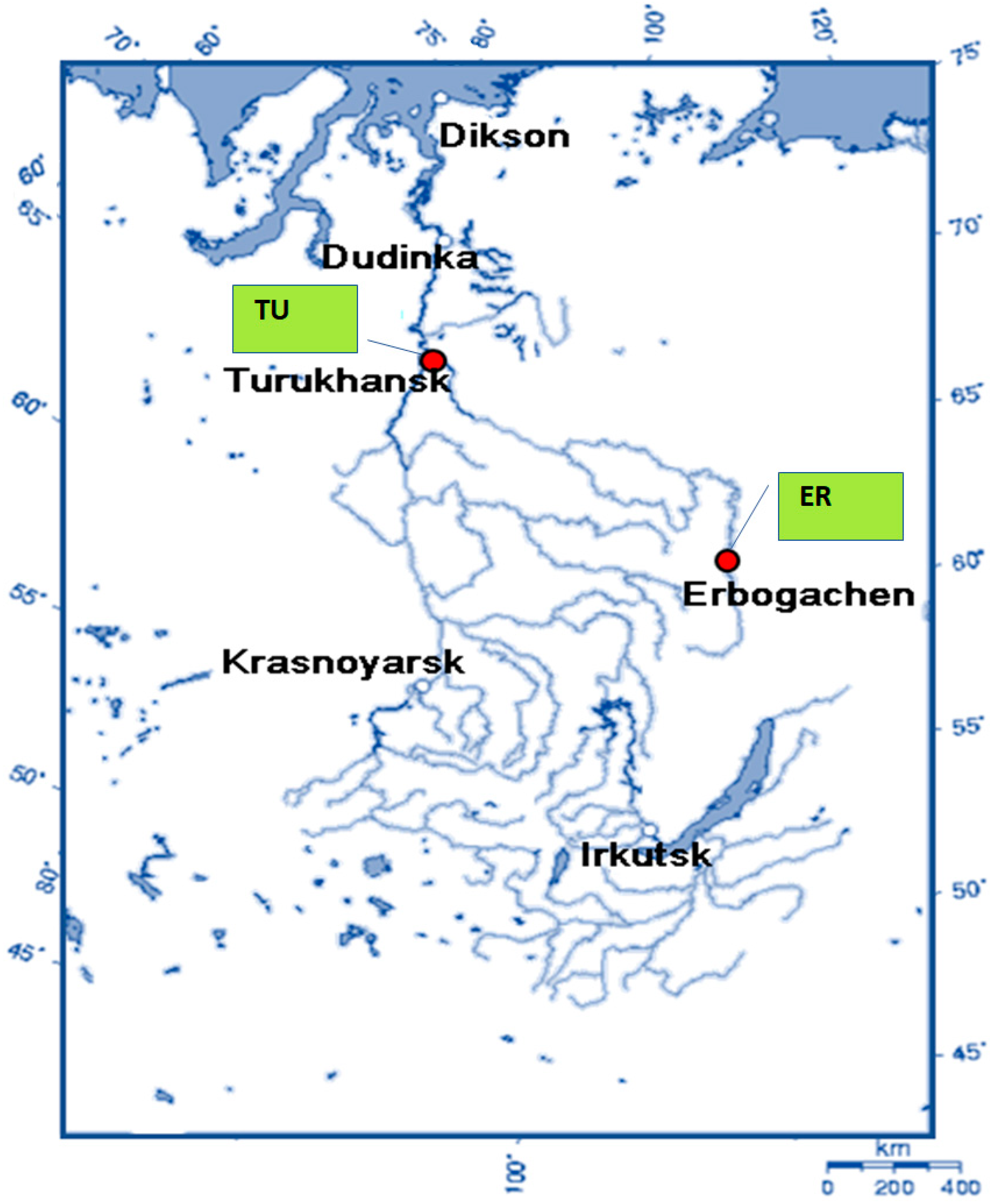
2.1. Study Object and Area
2.2. Biological Analysis of the Fish and Sample Preparation
2.3. Microplastic Extraction, Detection, and Quantification
2.4. Contamination Prevention and Control Procedures
3. Results
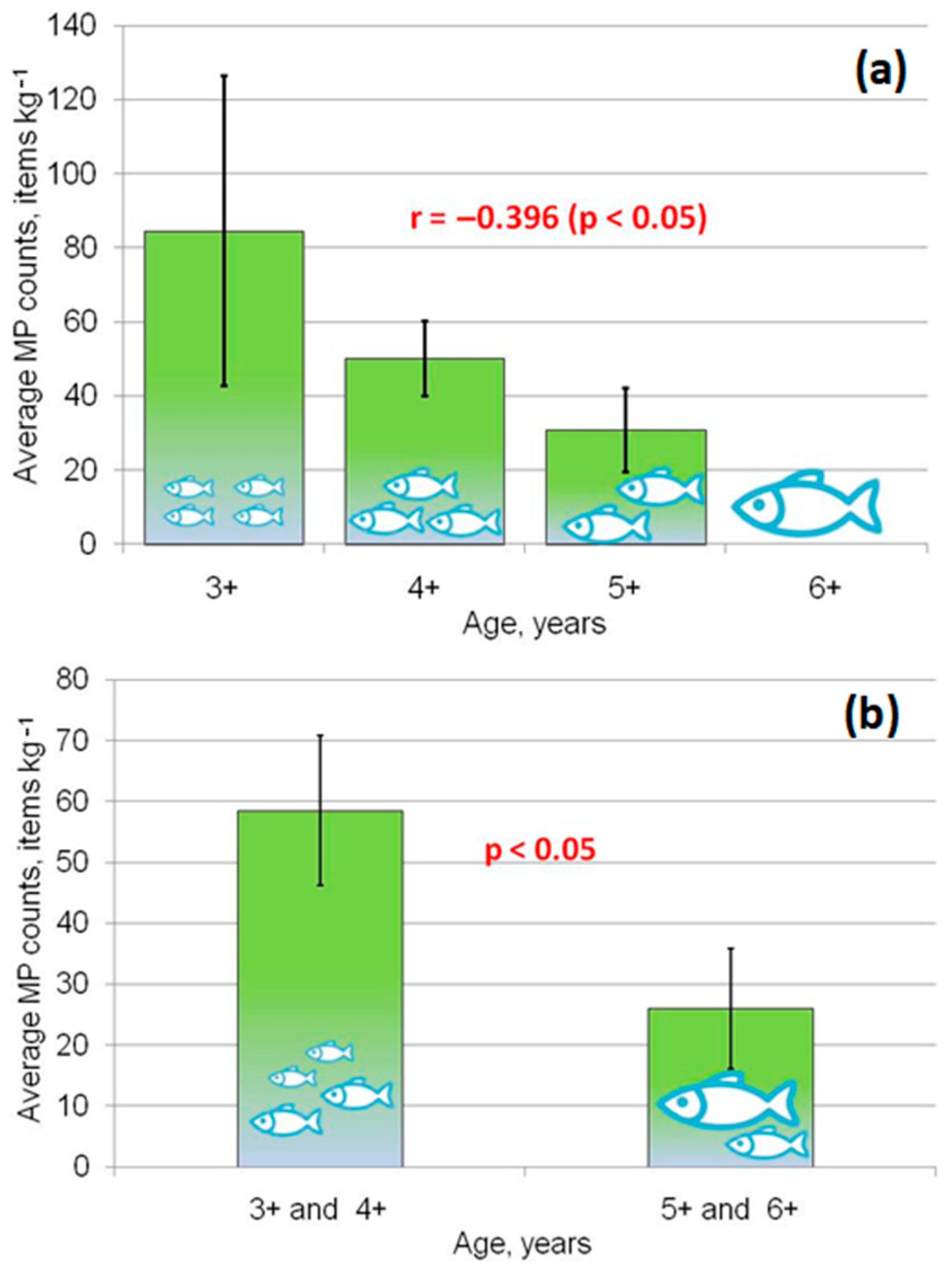
3.1. Biological Characteristics of Siberian dace from the N. Tunguska River
3.2. Microplastic Abundance in Gastrointestinal Tracts of the Fish
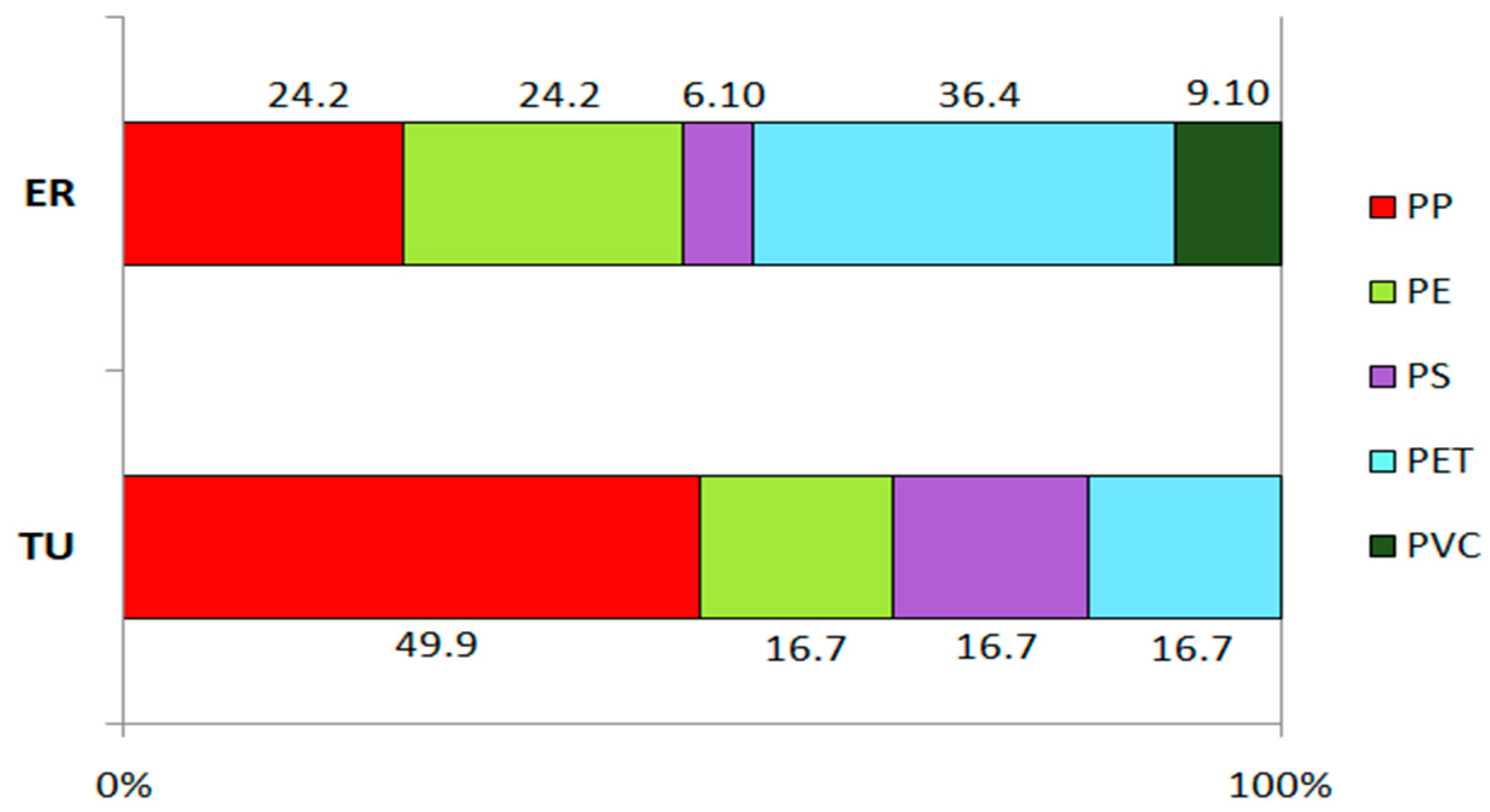
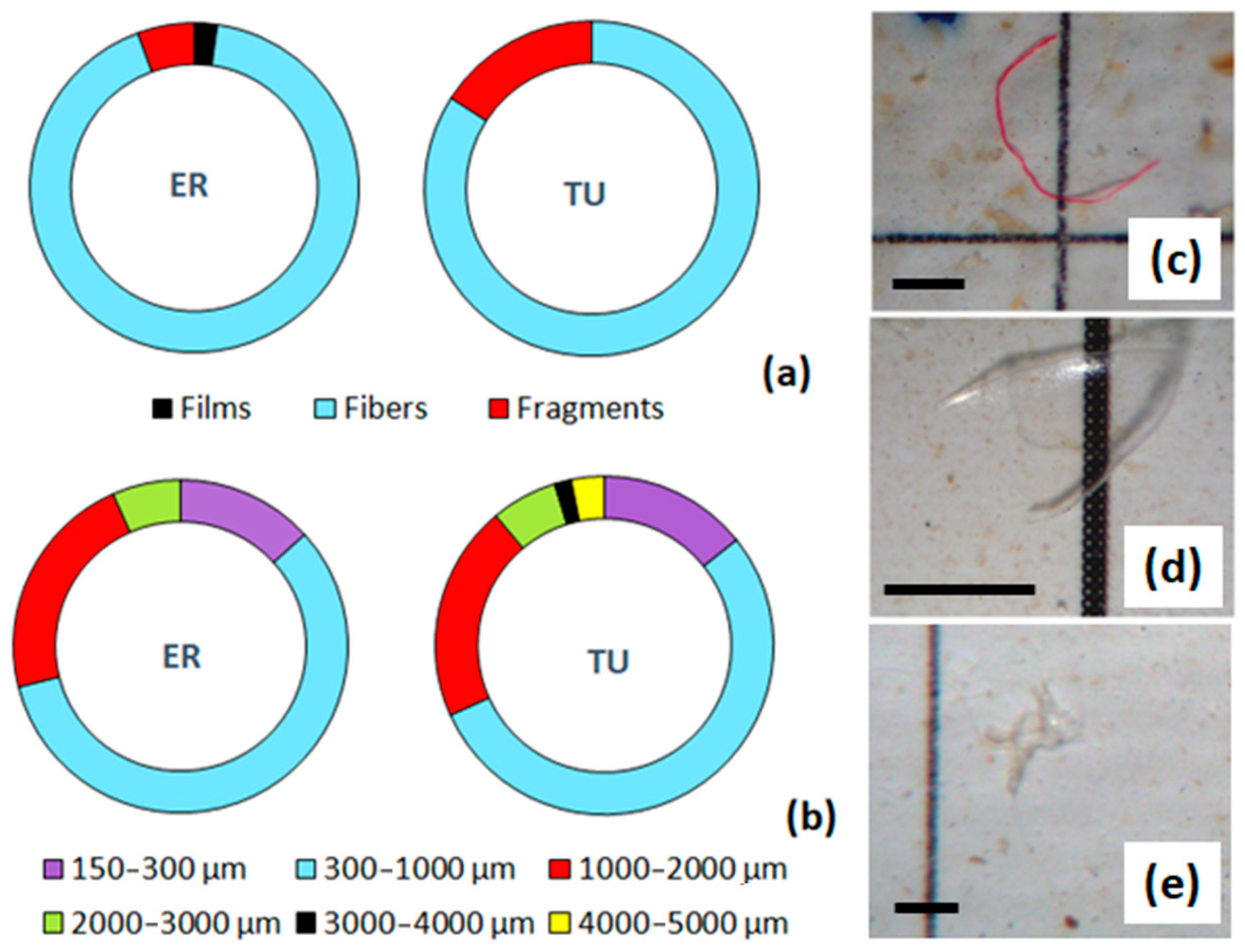
3.3. Polymer Composition and Morphology of Microplastics
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Horton, A.A. Plastic pollution: When do we know enough? J. Hazard. Mater. 2022, 422, 126885. [Google Scholar] [CrossRef] [PubMed]
- Arthur, C.; Baker, J.; Bamford, H. (Eds.) Proceedings of the International Research Workshop on the Occurrence, Effects, and Fate of Microplastic Marine Debris, Tacoma, WA, USA, 9–11 September 2008; NOAA Technical Memorandum NOS-OR&R-30; NOAA Marine Debris Division: Silver Spring, MD, USA, 2009. [Google Scholar]
- Boucher, J.; Friot, D. Primary Microplastics in the Oceans: A Global Evaluation of Sources; IUCN: Gland, Switzerland, 2017. [Google Scholar]
- Koutnik, V.S.; Leonard, J.; Alkidim, S.; DePrima, F.J.; Ravi, S.; Hoek, E.M.; Mohanty, S.K. Distribution of microplastics in soil and freshwater environments: Global analysis and framework for transport modeling. Environ. Pollut. 2021, 274, 116552. [Google Scholar] [CrossRef] [PubMed]
- Bank, M.S.; Hansson, S.V. The microplastic cycle: An introduction to a complex issue. In Microplastic in the Environment: Pattern and Process, Environmental Contamination Remediation and Management; Bank, M.S., Ed.; Springer: Cham, Switzerland, 2022; pp. 1–16. [Google Scholar] [CrossRef]
- Prata, J.C.; da Costa, J.P.; Lopes, I.; Duarte, A.C.; Rocha-Santos, T. Environmental exposure to microplastics: An overview on possible human health effects. Sci. Total Environ. 2020, 702, 134455. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Song, B.; Liang, J.; Niu, Q.; Zeng, G.; Shen, M.; Deng, J.; Luo, Y.; Wen, X.; Zhang, Y. Microplastics and associated contaminants in the aquatic environment: A review on their ecotoxicological effects, trophic transfer, and potential impacts to human health. J. Hazard. Mater. 2021, 405, 124187. [Google Scholar] [CrossRef] [PubMed]
- Chae, Y.; Kim, D.; Kim, S.W.; An, Y. Trophic transfer and individual impact of nano-sized polystyrene in a four-species freshwater food chain. Sci. Rep. 2018, 8, 284. [Google Scholar] [CrossRef]
- Lehel, J.; Murphy, S. Microplastics in the food chain: Food safety and environmental aspects. Rev. Environ. Contam. Toxicol. 2021, 259, 1–49. [Google Scholar] [CrossRef]
- Cássio, F.; Batista, D.; Pradhan, A. Plastic interactions with pollutants and consequences to aquatic ecosystems: What we know and what we do not know. Biomolecules 2022, 12, 798. [Google Scholar] [CrossRef]
- Abdolahpur Monikh, F.; Chupani, L.; Smerkova, K.; Bosker, T.; Cizar, P.; Krzyzanek, V.; Richtera, L.; Franek, R.; Zuskova, E.; Skoupy, R.; et al. Engineered nanoselenium supplemented fish diet: Toxicity comparison with ionic selenium and stability against particle dissolution, aggregation and release. Environ. Sci. Nano 2020, 7, 2325–2336. [Google Scholar] [CrossRef]
- Singh, N.; Bhagat, J.; Tiwari, E.; Khandelwal, N.; Darbha, G.K.; Shyama, S.K. Metal oxide nanoparticles and polycyclic aromatic hydrocarbons alter nanoplastic’s stability and toxicity to zebrafish. J. Hazard. Mater. 2021, 407, 124382. [Google Scholar] [CrossRef]
- Yuan, Z.; Nag, R.; Cummins, E. Human health concerns regarding microplastics in the aquatic environment—From marine to food systems. Sci. Total Environ. 2022, 823, 153730. [Google Scholar] [CrossRef]
- Jin, M.; Wang, X.; Ren, T.; Wang, J.; Shan, J. Microplastics contamination in food and beverages: Direct exposure to humans. J. Food Sci. 2021, 86, 2816–2837. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Armendáriz, C.; Alejandro-Vega, S.; Paz-Montelongo, S.; Gutiérrez-Fernández, Á.J.; Carrascosa-Iruzubieta, C.J.; Hardisson-de la Torre, A. Microplastics as Emerging Food Contaminants: A Challenge for Food Safety. Int. J. Environ. Res. Public Health 2022, 19, 1174. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Presence of microplastics and nanoplastics in food, with particular focus on seafood. EFSA J. 2016, 14, 4501. [Google Scholar]
- Galafassi, S.; Campanale, C.; Massarelli, C.; Uricchio, V.F.; Volta, P. Do freshwater fish eat microplastics? A review with a focus on effects on fish health and predictive traits of MPs ingestion. Water 2021, 13, 2214. [Google Scholar] [CrossRef]
- Tekman, M.B.; Gutow, L.; Macario, A.; Haas, A.; Walter, A.; Bergmann, M. Alfred Wegener Institute Helmholtz Centre for Polar and Marine Research. Literbase. Available online: https://litterbase.awi.de/litter (accessed on 3 November 2022).
- Sirina, A.A. The folks next door. Russian settlers and Evenki of the upper flow of the Lower Tunguska (19th-early 21st century). Études Mong. Sib. Cent. Tibét. 2018, 49. [Google Scholar] [CrossRef]
- Kottelat, M.; Freyhof, J. Handbook of European Freshwater Fishes; Publications Kottelat, Cornol and Freyhof: Berlin, Germany, 2007; 646p. [Google Scholar]
- Lobón-Cerviá, J.; Dgebuadze, Y.; Utrilla, C.G.; Rincón, P.A.; Granado-Lorencio, C. The reproductive tactics of dace in central Siberia: Evidence for temperature regulation of the spatio-temporal variability of its life history. J. Fish Biol. 1996, 48, 1074–1087. [Google Scholar] [CrossRef]
- Frank, Y.A.; Vorobiev, D.S.; Kayler, O.A.; Vorobiev, E.D.; Kulinicheva, K.S.; Trifonov, A.A.; Soliman Hunter, T. Evidence for microplastics contamination of the remote tributary of the Yenisei River, Siberia—The pilot study results. Water 2021, 13, 3248. [Google Scholar] [CrossRef]
- Hile, R. Age determination of fish from scales; Method and application to fish cultural problems. Progress. Fish-Cult. 1936, 3, 1–5. [Google Scholar] [CrossRef]
- Pravdin, I.F. Guide for the Study of Fish; Pishhevaja Promyshlennost’: Moscow, Russia, 1966; 376p. (In Russian) [Google Scholar]
- Khan, F.R.; Shashoua, Y.; Crawford, A.; Drury, A.; Sheppard, K.; Stewart, K.; Sculthorp, T. ‘The Plastic Nile’: First Evidence of Microplastic Contamination in Fish from the Nile River (Cairo, Egypt). Toxics 2020, 8, 22. [Google Scholar] [CrossRef]
- Capone, A.; Petrillo, M.; Misic, C. Ingestion and elimination of anthropogenic fibres and microplastic fragments by the European anchovy (Engraulis encrasicolus) of the NW Mediterranean Sea. Mar. Biol. 2020, 167, 166. [Google Scholar] [CrossRef]
- Jahan, S.; Strezov, V.; Weldekidan, H.; Kumar, R.; Kan, T.; Sarkodie, A.S.; He, J.; Dastjerdi, B.; Wilson, S.P. Interrelationship of microplastic pollution in sediments and oysters in a seaport environment of the eastern coast of Australia. Sci. Total Environ. 2019, 695, 133924. [Google Scholar] [CrossRef]
- Dawson, A.L.; Motti, C.A.; Kroon, F.J. Solving a sticky situation: Microplastic analysis of lipid-rich tissue. Front. Environ. Sci. 2020, 8, 563565. [Google Scholar] [CrossRef]
- Frias, J.P.G.L.; Nash, R. Microplastics: Finding a consensus on the definition. Mar. Pollut. Bull. 2019, 138, 145–147. [Google Scholar] [CrossRef]
- Student. The probable error of a mean. Biometrika 1908, 6, 1–25. [Google Scholar] [CrossRef]
- Mann, H.B.; Whitney, D.R. On a test of whether one of two random variables is stochastically larger than the other. Ann. Math. Stat. 1947, 18, 50–60. [Google Scholar] [CrossRef]
- Kirch, W. Pearson’s Correlation Coefficient. In Encyclopedia of Public Health; Kirch, W., Ed.; Springer: Dordrecht, Germany, 2008; pp. 1090–1091. [Google Scholar] [CrossRef]
- Koelmans, A.A.; Nor, N.H.M.; Hermsen, E.; Kooi, M.; Mintenig, S.M.; De France, J. Microplastics in freshwaters and drinking water: Critical review and assessment of data quality. Water Res. 2019, 155, 410–422. [Google Scholar] [CrossRef]
- Frank, Y.A.; Vorobiev, E.D.; Vorobiev, D.S.; Trifonov, A.A.; Antsiferov, D.V.; Soliman Hunter, T.; Wilson, S.P.; Strezov, V. Preliminary screening for microplastic concentrations in the surface water of the Ob and Tom Rivers in Siberia, Russia. Sustainability 2021, 13, 80. [Google Scholar] [CrossRef]
- van der Wal, M.; van der Meulen, M.; Tweehuijsen, G.; Peterlin, M.; Palatinus, A.; Kovač Viršek, M.; Coscia, L.; Kržan, A. Identification and Assessment of Riverine Input of (Marine) Litter; European Commission: Bruxelles, Belgium, 2015; 186p. [Google Scholar]
- Free, C.M.; Jensen, O.P.; Mason, S.A.; Eriksen, M.; Williamson, N.; Boldgiv, B. High-levels of microplastic pollution in a large, remote, mountain lake. Mar. Pollut. Bull. 2014, 85, 156–163. [Google Scholar] [CrossRef]
- Jiang, C.; Yin, L.; Li, Z.; Wen, X.; Luo, X.; Hu, S.; Yang, H.; Long, Y.; Deng, B.; Huang, L.; et al. Microplastic pollution in the rivers of the Tibet Plateau. Environ. Pollut. 2019, 249, 91–98. [Google Scholar] [CrossRef]
- Collard, F.; Gasperi, J.; Gabrielsen, G.W.; Tassin, B. Plastic particle ingestion by wild freshwater fish: A critical review. Environ. Sci. Technol. 2019, 53, 12974–12988. [Google Scholar] [CrossRef]
- Parvin, F.; Jannat, S.; Tareq, S.M. Abundance, characteristics and variation of microplastics in different freshwater fish species from Bangladesh. Sci. Total Environ. 2021, 784, 147137. [Google Scholar] [CrossRef] [PubMed]
- Horton, A.A.; Jürgens, M.D.; Lahive, E.; van Bodegom, P.M.; Vijver, M.G. The influence of exposure and physiology on microplastic ingestion by the freshwater fish Rutilus rutilus (roach) in the River Thames, UK. Environ. Pollut. 2018, 236, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Su, L.; Nan, B.; Hassell, K.L.; Craig, N.J.; Pettigrove, V. Microplastics biomonitoring in Australian urban wetlands using a common noxious fish (Gambusia holbrooki). Chemosphere 2019, 228, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Pegado, T.S.E.S.; Schmid, K.; Winemiller, K.O.; Chelazzi, D.; Cincinelli, A.; Dei, L.; Giarrizzo, T. First evidence of microplastic ingestion by fishes from the Amazon River estuary. Mar. Pollut. Bull. 2018, 133, 814–821. [Google Scholar] [CrossRef] [PubMed]
- Zheng, K.; Fan, Y.; Zhu, Z.; Chen, G.; Tang, C.; Penga, X. Occurrence and species-specific distribution of plastic debris in wild freshwater fish from the Pearl River catchment, China. Environ. Toxicol. Chem. 2019, 38, 1504–1513. [Google Scholar] [CrossRef]
- Jabeen, K.; Su, L.; Li, J.N.; Yang, D.Q.; Tong, C.F.; Mu, J.L.; Shi, H.H. Microplastics and mesoplastics in fish from coastal and fresh waters of China. Environ. Pollut. 2017, 221, 141–149. [Google Scholar] [CrossRef]
- McNeish, R.E.; Kim, L.H.; Barrett, H.A.; Mason, S.A.; Kelly, J.J.; Hoellein, T.J. Microplastic in riverine fish is connected to species traits. Sci. Rep. 2018, 8, 11639. [Google Scholar] [CrossRef]
- Arias, A.H.; Ronda, A.C.; Oliva, A.L.; Marcovecchio, J.E. Evidence of microplastic ingestion by fish from the Bahía Blanca Estuary in Argentina, South America. Bull. Environ. Contam. Toxicol. 2019, 102, 750–756. [Google Scholar] [CrossRef]
- Frank, Y.A.; Vorobiev, E.D.; Babkina, I.B.; Antsiferov, D.V.; Vorobiev, D.S. Microplastics in fish gut, first records from the Tom River in West Siberia, Russia. Vestn. Tomsk. Gos. Univ. Biol. 2020, 52, 130–139. [Google Scholar] [CrossRef]
- Park, T.-J.; Lee, S.-H.; Lee, M.-S.; Lee, J.-K.; Lee, S.-H.; Zoh, K.-D. Occurrence of microplastics in the Han River and riverine fish in South Korea. Sci. Total Environ. 2020, 708, 134535. [Google Scholar] [CrossRef]
- Tien, C.J.; Wang, Z.X.; Chen, C.S. Microplastics in water, sediment and fish from the Fengshan River system: Relationship to aquatic factors and accumulation of polycyclic aromatic hydrocarbons by fish. Environ. Pollut. 2020, 265, 114962. [Google Scholar] [CrossRef]
- McGoran, A.R.; Clark, P.F.; Morritt, D. Presence of microplastic in the digestive tracts of European flounder, Platichthys flesus, and European smelt, Osmerus eperlanus, from the River Thames. Environ. Pollut. 2017, 220, 744–751. [Google Scholar] [CrossRef]
- Wesch, C.; Bredimus, K.; Paulus, M.; Klein, R. Towards the suitable monitoring of ingestion of microplastics by marine biota: A review. Environ. Pollut. 2016, 218, 1200–1208. [Google Scholar] [CrossRef]
- Wang, S.; Zhang, C.; Pan, Z.; Sun, D.; Zhou, A.; Xie, S.; Wang, J.; Zou, J. Microplastics in wild freshwater fish of different feeding habits from Beijiang and Pearl River Delta regions, south China. Chemosphere 2020, 258, 127345. [Google Scholar] [CrossRef]
- Lim, K.P.; Lim, P.E.; Yusoff, S.; Sun, C.; Ding, J.; Loh, K.H. A Meta-Analysis of the characterisations of plastic ingested by fish globally. Toxics 2022, 10, 186. [Google Scholar] [CrossRef]
- Elias, S. Impacts of chemical pollution on marine ecosystems. In Threats to the Arctic; Elias, S.A., Ed.; Elsevier: Cambridge, MA, USA, 2021; pp. 93–121. [Google Scholar] [CrossRef]
- Murphy, F.; Ewins, C.; Carbonnier, F.; Quinn, B. Wastewater treatment works (WwTW) as a source of microplastics in the aquatic environment. Environ. Sci. Technol. 2016, 50, 5800–5808. [Google Scholar] [CrossRef]
- Ziajahromi, S.; Neale, P.A.; Leusch, F.D.L. Wastewater treatment plant effluent as a source of microplastics: Review of the fate, chemical interactions and potential risks to aquatic organisms. Water Sci. Technol. 2016, 74, 2253–2269. [Google Scholar] [CrossRef]
- Qiao, R.; Deng, Y.; Zhang, S.; Wolosker, M.B.; Zhu, Q.; Ren, H.; Zhang, Y. Accumulation of different shapes of microplastics initiates intestinal injury and gut microbiota dysbiosis in the gut of zebrafish. Chemosphere 2019, 236, 124334. [Google Scholar] [CrossRef]
- Jabeen, K.; Li, B.; Chen, Q.; Su, L.; Wu, C.; Hollert, H.; Shi, H. Effects of virgin microplastics on goldfish (Carassius auratus). Chemosphere 2018, 213, 323–332. [Google Scholar] [CrossRef]
- Lu, Y.; Zhang, Y.; Deng, Y.; Jiang, W.; Zhao, Y.; Geng, J.; Ding, L.; Ren, H. Uptake and accumulation of polystyrene microplastics in Zebrafish (Danio rerio) and toxic effects in liver. Environ. Sci. Technol. 2016, 50, 4054–4060. [Google Scholar] [CrossRef]
- Ding, J.; Zhang, S.; Razanajatovo, R.M.; Zou, H.; Zhu, W. Accumulation, tissue distribution, and biochemical effects of polystyrene microplastics in the freshwater fish red tilapia (Oreochromis niloticus). Environ. Pollut. 2018, 238, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Aryani, D.; Khalifa, M.A.; Herjayanto, M.; Solahudin, E.A.; Rizki, E.M.; Halwatiyah, W.; Istiqomah, H.; Maharani, S.H.; Wahyudin, H.; Pratama, G. Penetration of microplastics (polyethylene) to several organs of Nile Tilapia (Oreochromis niloticus). IOP Conf. Ser. Earth Environ. Sci. 2021, 715, 012061. [Google Scholar] [CrossRef]
| Site | N | L (mm) | l (mm) | Q (g) | q (g) | m(GIT) (g) |
|---|---|---|---|---|---|---|
| ER | 20 | 179.9 ± 17.6 | 158.9 ± 12.4 | 48.33 ± 11.50 | 42.75 ± 10.10 | 2.99 ± 1.25 |
| TU | 20 | 152.2 ± 15.1 | 136.8 ± 12.9 | 30.21 ± 9.51 | 26.34 ± 8.25 | 1.73 ± 0.65 |
| Site | N | % of Individuals with MPs | Av. 1 MP Content, Items ind−1 | Av.1 MP Content, Items kg−1 |
|---|---|---|---|---|
| ER | 20 | 75.0 | 2.10 ± 2.29 | 48.0 ± 56.4 |
| TU | 20 | 45.0 | 1.00 ± 1.38 | 38.1 ± 49.3 |
| Both sites | 40 | 60.0 | 1.55 ± 1.95 | 43.1 ± 52.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Frank, Y.; Vorobiev, D.; Mandal, A.; Lemeshko, Y.; Rakhmatullina, S.; Darbha, G.K. Freshwater Fish Siberian Dace Ingest Microplastics in the Remote Yenisei Tributary. Toxics 2023, 11, 38. https://doi.org/10.3390/toxics11010038
Frank Y, Vorobiev D, Mandal A, Lemeshko Y, Rakhmatullina S, Darbha GK. Freshwater Fish Siberian Dace Ingest Microplastics in the Remote Yenisei Tributary. Toxics. 2023; 11(1):38. https://doi.org/10.3390/toxics11010038
Chicago/Turabian StyleFrank, Yulia, Danil Vorobiev, Abhishek Mandal, Yana Lemeshko, Svetlana Rakhmatullina, and Gopala Krishna Darbha. 2023. "Freshwater Fish Siberian Dace Ingest Microplastics in the Remote Yenisei Tributary" Toxics 11, no. 1: 38. https://doi.org/10.3390/toxics11010038
APA StyleFrank, Y., Vorobiev, D., Mandal, A., Lemeshko, Y., Rakhmatullina, S., & Darbha, G. K. (2023). Freshwater Fish Siberian Dace Ingest Microplastics in the Remote Yenisei Tributary. Toxics, 11(1), 38. https://doi.org/10.3390/toxics11010038






