Occurrence of Natural and Synthetic Micro-Fibers in the Mediterranean Sea: A Review
Abstract
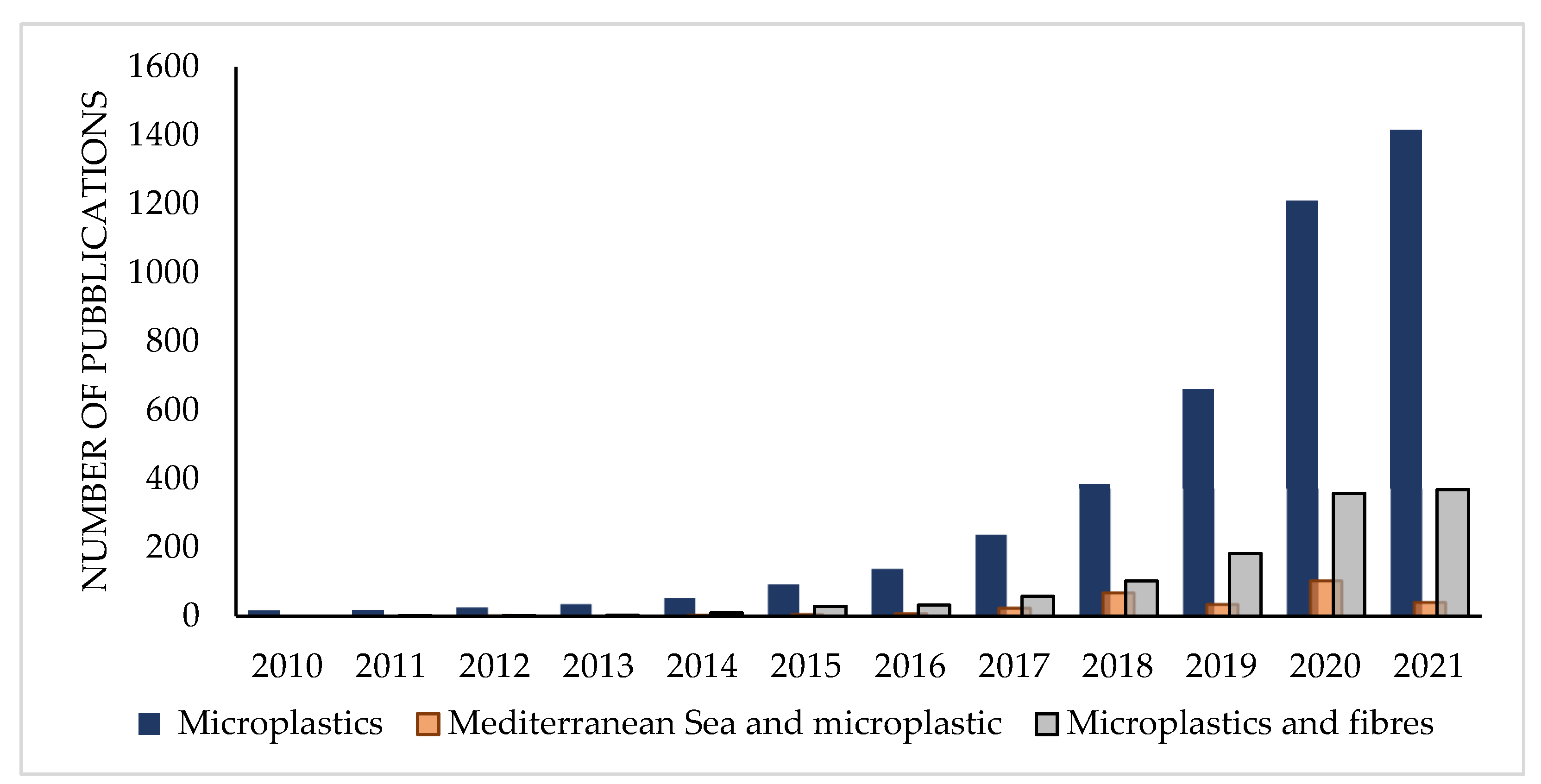
:1. Introduction
Non-Synthetic MFs Toxicity
2. Discussion
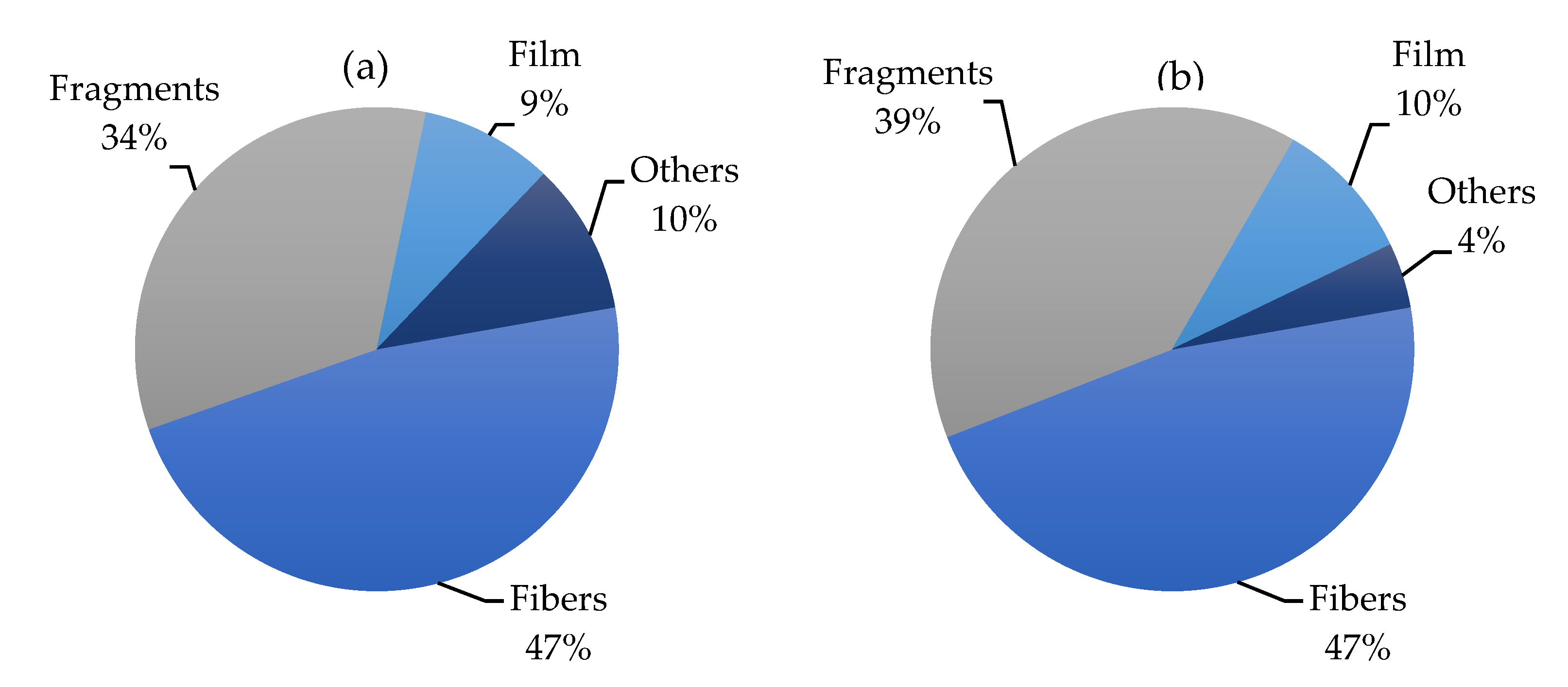
2.1. Most Abundance Shapes
2.2. Non-Synthetic Composition of MFs in the Mediterranean Sea
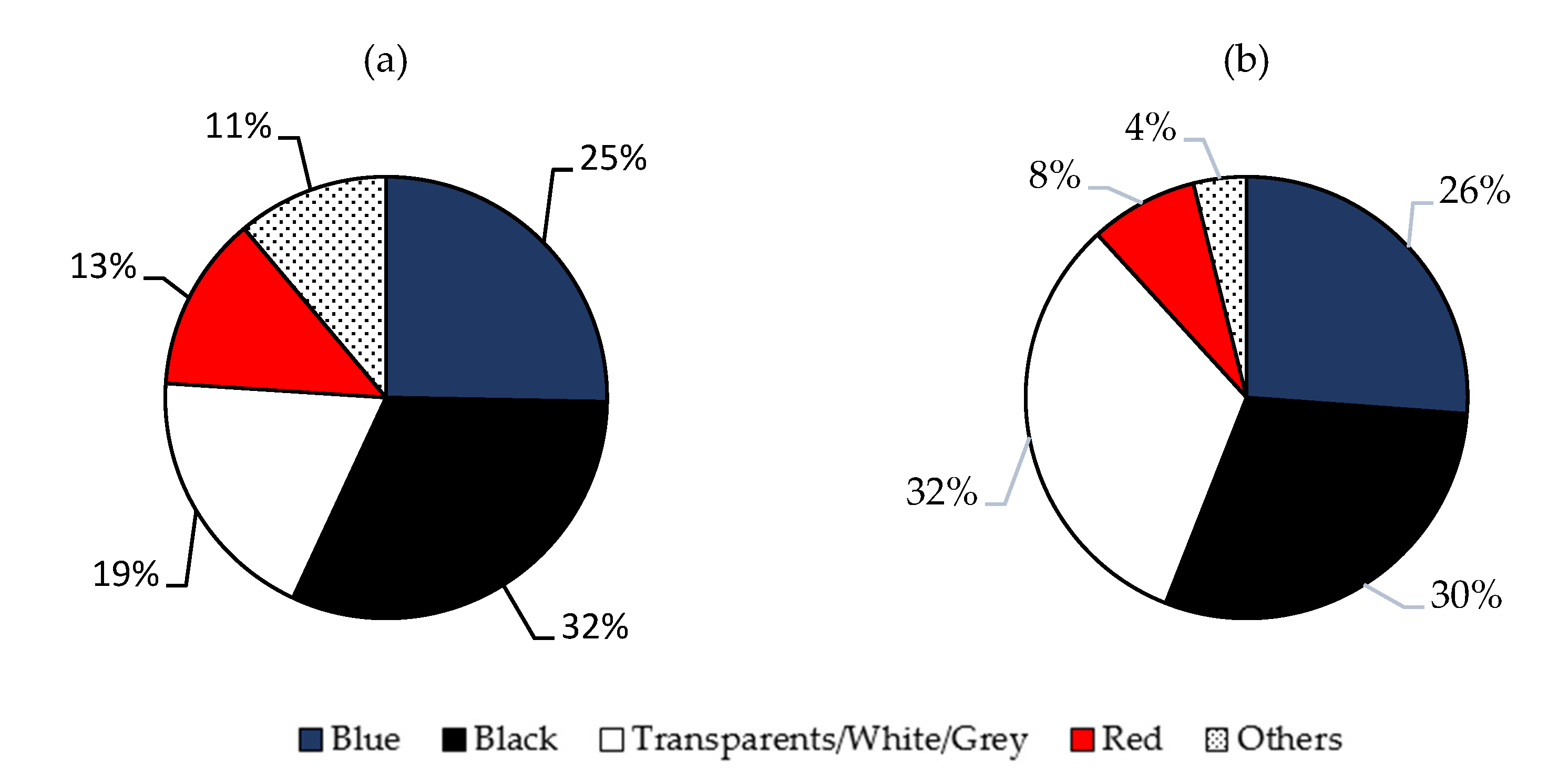
2.3. Color of MFs
2.4. Self-Contamination
2.5. Size
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Abeynayaka, A.; Kojima, F.; Miwa, Y.; Ito, N.; Nihei, Y.; Fukunaga, Y.; Yashima, Y.; Itsubo, N. Rapid Sampling of Suspended and Floating Microplastics in Challenging Riverine and Coastal Water Environments in Japan. Water 2020, 12, 1903. [Google Scholar] [CrossRef]
- Cozzolino, L.; Nicastro, K.R.; Zardi, G.I.; de los Santos, C.B. Species-specific plastic accumulation in the sediment and canopy of coastal vegetated habitats. Sci. Total Environ. 2020, 723, 138018. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, N.B.; Hüffer, T.; Thompson, R.C.; Hassellöv, M.; Verschoor, A.; Daugaard, A.E.; Rist, S.; Karlsson, T.; Brennholt, N.; Cole, M.; et al. Are We Speaking the Same Language? Recommendations for a Definition and Categorization Framework for Plastic Debris. Environ. Sci. Technol. 2019, 53, 1039–1047. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Antunes, J.; Frias, J.; Sobral, P. Microplastics on the Portuguese coast. Mar. Pollut. Bull. 2018, 131, 294–302. [Google Scholar] [CrossRef] [PubMed]
- Cincinelli, A.; Martellini, T.; Guerranti, C.; Scopetani, C.; Chelazzi, D.; Giarrizzo, T. A potpourri of microplastics in the sea surface and water column of the Mediterranean Sea. TrAC Trends Anal. Chem. 2019, 110, 321–326. [Google Scholar] [CrossRef]
- Cincinelli, A.; Scopetani, C.; Chelazzi, D.; Lombardini, E.; Martellini, T.; Katsoyiannis, A.; Fossi, M.C.; Corsolini, S. Microplastic in the surface waters of the Ross Sea (Antarctica): Occurrence, distribution and characterization by FTIR. Chemosphere 2017, 175, 391–400. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.; Huang, J.; Zheng, Y.; Yang, Y.; Zhang, Y.; He, F.; Chen, H.; Quan, G.; Yan, J.; Li, T.; et al. Environmental occurrences, fate, and impacts of microplastics. Ecotoxicol. Environ. Saf. 2019, 184, 109612. [Google Scholar] [CrossRef]
- Hurley, R.; Woodward, J.; Rothwell, J.J. Microplastic contamination of river beds significantly reduced by catchment-wide flooding. Nat. Geosci. 2018, 11, 251–257. [Google Scholar] [CrossRef]
- Klein, S.; Worch, E.; Knepper, T.P. Occurrence and spatial distribution of microplastics in river shore sediments of the rhine-main area in Germany. Environ. Sci. Technol. 2015, 49, 6070–6076. [Google Scholar] [CrossRef]
- Nizzetto, L.; Bussi, G.; Futter, M.N.; Butterfield, D.; Whitehead, P.G. A theoretical assessment of microplastic transport in river catchments and their retention by soils and river sediments. Environ. Sci. Process. Impacts 2016, 18, 1050–1059. [Google Scholar] [CrossRef]
- Dris, R.; Gasperi, J.; Rocher, V.; Tassin, B. Synthetic and non-synthetic anthropogenic fibers in a river under the impact of Paris Megacity: Sampling methodological aspects and flux estimations. Sci. Total Environ. 2018, 618, 157–164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gasperi, J.; Wright, S.L.; Dris, R.; Collard, F.; Mandin, C.; Guerrouache, M.; Langlois, V.; Kelly, F.J.; Tassin, B. Microplastics in air: Are we breathing it in? Curr. Opin. Environ. Sci. Health 2018, 1, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Koelmans, A.A.; Bakir, A.; Burton, G.A.; Janssen, C.R. Microplastic as a Vector for Chemicals in the Aquatic Environment: Critical Review and Model-Supported Reinterpretation of Empirical Studies. Environ. Sci. Technol. 2016, 50, 3315–3326. [Google Scholar] [CrossRef] [PubMed]
- Hahladakis, J.N.; Velis, C.A.; Weber, R.; Iacovidou, E.; Purnell, P. An overview of chemical additives present in plastics: Migration, release, fate and environmental impact during their use, disposal and recycling. J. Hazard. Mater. 2018, 344, 179–199. [Google Scholar] [CrossRef] [PubMed]
- Wagner, M.; Scherer, C.; Alvarez-Muñoz, D.; Brennholt, N.; Bourrain, X.; Buchinger, S.; Fries, E.; Grosbois, C.; Klasmeier, J.; Marti, T.; et al. Microplastics in freshwater ecosystems: What we know and what we need to know. Environ. Sci. Eur. 2014, 26, 12. [Google Scholar] [CrossRef] [Green Version]
- Kärrman, A.; Schönlau, C.; Engwall, M. Exposure and Effects of Microplastics on Wildlife. A review of existing data. DiVA. 2016, p. 39. Available online: https://www.diva-portal.org/smash/get/diva2:921211/FULLTEXT01.pdf (accessed on 14 March 2022).
- Anbumani, S.; Kakkar, P. Ecotoxicological effects of microplastics on biota: A review. Environ. Sci. Pollut. Res. 1999, 25, 14373–14396. [Google Scholar] [CrossRef] [PubMed]
- Fossi, M.C.; Pedà, C.; Compa, M.; Tsangaris, C.; Alomar, C.; Claro, F.; Ioakeimidis, C.; Galgani, F.; Hema, T.; Deudero, S.; et al. Bioindicators for monitoring marine litter ingestion and its impacts on Mediterranean biodiversity. Environ. Pollut. 2018, 237, 1023–1040. [Google Scholar] [CrossRef]
- Filgueiras, A.V.; Gago, J.; Campillo, J.A.; León, V.M. Microplastic distribution in surface sediments along the Spanish Mediterranean continental shelf. Environ. Sci. Pollut. Res. 2019, 26, 21264–21273. [Google Scholar] [CrossRef]
- Lusher, A.L.; McHugh, M.; Thompson, R.C. Occurrence of microplastics in the gastrointestinal tract of pelagic and demersal fish from the English Channel. Mar. Pollut. Bull. 2013, 67, 94–99. [Google Scholar] [CrossRef]
- Lusher, A.L.; Tirelli, V.; O’Connor, I.; Officer, R. Microplastics in Arctic polar waters: The first reported values of particles in surface and sub-surface samples. Sci. Rep. 2015, 5, 14947. [Google Scholar] [CrossRef] [Green Version]
- Woodall, L.C.; Sanchez-Vidal, A.; Canals, M.; Paterson, G.L.J.; Coppock, R.; Sleight, V.; Calafat, A.; Rogers, A.D.; Narayanaswamy, B.E.; Thompson, R.C. The deep sea is a major sink for microplastic debris. R. Soc. Open Sci. 2014, 1, 140317. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Obbard, R.W.; Sadri, S.; Wong, Y.Q.; Khitun, A.A.; Baker, I.; Richard, C. Who Where Why—Wordpress blog—Community mapping examples. Earth’s Future 2014, 2, 315–320. [Google Scholar] [CrossRef]
- Neves, D.; Sobral, P.; Ferreira, J.L.; Pereira, T. Ingestion of microplastics by commercial fish off the Portuguese coast. Mar. Pollut. Bull. 2015, 101, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Remy, F.; Collard, F.; Gilbert, B.; Compère, P.; Eppe, G.; Lepoint, G. When Microplastic Is Not Plastic: The Ingestion of Artificial Cellulose Fibers by Macrofauna Living in Seagrass Macrophytodetritus. Environ. Sci. Technol. 2015, 49, 11158–11166. [Google Scholar] [CrossRef] [PubMed]
- Bellas, J.; Martínez-Armental, J.; Martínez-Cámara, A.; Besada, V.; Martínez-Gómez, C. Ingestion of microplastics by demersal fish from the Spanish Atlantic and Mediterranean coasts. Mar. Pollut. Bull. 2016, 109, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Güven, O.; Gökdağ, K.; Jovanović, B.; Kıdeyş, A.E. Microplastic litter composition of the Turkish territorial waters of the Mediterranean Sea, and its occurrence in the gastrointestinal tract of fish. Environ. Pollut. 2017, 223, 286–294. [Google Scholar] [CrossRef] [PubMed]
- Barrows, A.P.W.; Cathey, S.E.; Petersen, C.W. Marine environment microfiber contamination: Global patterns and the diversity of microparticle origins. Environ. Pollut. 2018, 237, 275–284. [Google Scholar] [CrossRef] [Green Version]
- Bessa, F.; Barría, P.; Neto, J.M.; Frias, J.P.G.L.; Otero, V.; Sobral, P.; Marques, J.C. Occurrence of microplastics in commercial fish from a natural estuarine environment. Mar. Pollut. Bull. 2018, 128, 575–584. [Google Scholar] [CrossRef]
- Rios-Fuster, B.; Alomar, C.; Compa, M.; Guijarro, B.; Deudero, S. Anthropogenic particles ingestion in fish species from two areas of the western Mediterranean Sea. Mar. Pollut. Bull. 2019, 144, 325–333. [Google Scholar] [CrossRef]
- Hossain, M.S.; Rahman, M.S.; Uddin, M.N.; Sharifuzzaman, S.M.; Chowdhury, S.R.; Sarker, S.; Nawaz Chowdhury, M.S. Microplastic contamination in Penaeid shrimp from the Northern Bay of Bengal. Chemosphere 2020, 238, 124688. [Google Scholar] [CrossRef]
- Parton, K.J.; Godley, B.J.; Santillo, D.; Tausif, M.; Omeyer, L.C.M.; Galloway, T.S. Investigating the presence of microplastics in demersal sharks of the North-East Atlantic. Sci. Rep. 2020, 10, 12204. [Google Scholar] [CrossRef] [PubMed]
- Iliff, S.M.; Wilczek, E.R.; Harris, R.J.; Bouldin, R.; Stoner, E.W. Evidence of microplastics from benthic jellyfish (Cassiopea xamachana) in Florida estuaries. Mar. Pollut. Bull. 2020, 159, 111521. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Yang, Y.; Ding, J.; Zhu, B.; Gao, W. Microfibers: A preliminary discussion on their definition and sources. Environ. Sci. Pollut. Res. 2019, 26, 29497–29501. [Google Scholar] [CrossRef] [PubMed]
- Bal, B.; Ghosh, S.; Das, A.P. Microbial recovery and recycling of manganese waste and their future application: A review. Geomicrobiol. J. 2018, 36, 85–96. [Google Scholar] [CrossRef]
- Mohanty, S.; Ghosh, S.; Bal, B.; Prasad, A. A review of biotechnology processes applied for manganese recovery from wastes. Rev. Environ. Sci. Bio/Technol. 2018, 17, 791–811. [Google Scholar] [CrossRef]
- Suaria, G.; Achtypi, A.; Perold, V.; Lee, J.R.; Pierucci, A.; Bornman, T.G.; Aliani, S.; Ryan, P.G. Microfibers in oceanic surface waters: A global characterization. Sci. Adv. 2020, 6, 1–9. [Google Scholar] [CrossRef]
- Avio, C.G.; Pittura, L.; d’Errico, G.; Abel, S.; Amorello, S.; Marino, G.; Gorbi, S.; Regoli, F. Distribution and characterization of microplastic particles and textile microfibers in Adriatic food webs: General insights for biomonitoring strategies. Environ. Pollut. 2020, 258, 113766. [Google Scholar] [CrossRef]
- Pedrotti, M.L.; Petit, S.; Eyheraguibel, B.; Kerros, M.E.; Elineau, A.; Ghiglione, J.F.; Loret, J.F.; Rostan, A.; Gorsky, G. Pollution by anthropogenic microfibers in North-West Mediterranean Sea and efficiency of microfiber removal by a wastewater treatment plant. Sci. Total Environ. 2021, 758, 144195. [Google Scholar] [CrossRef]
- Karimah, A.; Ridho, M.R.; Munawar, S.S.; Adi, D.S.; Ismadi; Damayanti, R.; Subiyanto, B.; Fatriasari, W.; Fudholi, A. A review on natural fibers for development of eco-friendly bio-composite: Characteristics, and utilizations. J. Mater. Res. Technol. 2021, 13, 2442–2458. [Google Scholar] [CrossRef]
- Cai, H.; Du, F.; Li, L.; Li, B.; Li, J.; Shi, H. A practical approach based on FT-IR spectroscopy for identi fi cation of semi-synthetic and natural celluloses in microplastic investigation. Sci. Total Environ. 2019, 669, 692–701. [Google Scholar] [CrossRef]
- Conley, K.; Clum, A.; Deepe, J.; Lane, H.; Beckingham, B. Wastewater treatment plants as a source of microplastics to an urban estuary: Removal efficiencies and loading per capita over one year. Water Res. X 2019, 3, 100030. [Google Scholar] [CrossRef] [PubMed]
- Helcoski, R.; Yonkos, L.T.; Sanchez, A.; Baldwin, A.H. Wetland soil microplastics are negatively related to vegetation cover and stem density. Environ. Pollut. 2019, 256, 113391. [Google Scholar] [CrossRef] [PubMed]
- Athey, S.N.; Erdle, L.M. Are We Underestimating Anthropogenic Micro fi ber Pollution? A Critical Review of Occurrence, Methods, and Reporting. Environ. Toxicol. Chem. 2021, 41, 822–837. [Google Scholar] [CrossRef] [PubMed]
- O’neill, C.; Hawkes, F.R.; Hawkes, D.L.; Lourenço, N.D.; Pinheiro, H.M.; Delée, W. Colour in textile effluents-sources, measurement, discharge consents and simulation: A review. J. Chem. Technol. Biotechnol. 1999, 74, 1009–1018. [Google Scholar] [CrossRef]
- Kwak, J.I.; Liu, H.; Wang, D.; Lee, Y.H.; Lee, J.S.; An, Y.J. Critical review of environmental impacts of microfibers in different environmental matrices. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2022, 251, 109196. [Google Scholar] [CrossRef]
- Verma, Y. Acute toxicity assessment of textile dyes and textile and dye industrial effluents using Daphnia magna bioassay. Toxic Ind. Health 2008, 24, 491–500. [Google Scholar] [CrossRef]
- Ferraz, E.R.; Li, Z.; Boubriak, O.; de Oliveira, D.P. De Current Issues Hepatotoxicity Assessment of the Azo Dyes Disperse Orange 1 (DO1), Disperse Red 1 (DR1,) and Disperse Red 13 (DR13) in HEPG2 Cells. J. Toxicol. Environ. Health Part A 2012, 75, 991–999. [Google Scholar] [CrossRef] [Green Version]
- OEKO-TEX OEKO-TEX. Available online: https://www.oeko-tex.com/en/ (accessed on 14 March 2022).
- No, C.A.S. Agents Classified by the IARC Monographs. Lancet Oncol. 2016, 1–123, 1–37. [Google Scholar]
- Shen, B.; Liu, H.; Ou, W.; Eilers, G.; Zhou, S. Toxicity induced by Basic Violet 14, Direct Red 28 and Acid Red 26 in zebrafish larvae. J. Appl. Toxicol. 2015, 35, 1473–1480. [Google Scholar] [CrossRef]
- IARC. Chemical agents and related occupations. In IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; IARC: Lyon, France, 2012; Volume 100, pp. 9–562. [Google Scholar]
- McCormick, A.R.; Hoellein, T.J.; London, M.G.; Hittie, J.; Scott, J.W.; Kelly, J.J. Microplastic in surface waters of urban rivers: Concentration, sources, and associated bacterial assemblages. Ecosphere 2016, 7, e01556. [Google Scholar] [CrossRef]
- Espinosa, C.; Esteban, M.Á.; Cuesta, A.; Microplastics in Aquatic Environments and and Their Toxicological Implications for Fish. Licens. InTech 2016, 113–145. Available online: https://www.intechopen.com/chapters/52031 (accessed on 14 March 2022).
- Zhao, Y.; Wang, C.; Xia, S.; Jiang, J.; Hu, R.; Yuan, G.; Hu, J. Biosensor medaka for monitoring intersex caused by estrogenic chemicals. Environ. Sci. Technol. 2014, 48, 2413–2420. [Google Scholar] [CrossRef] [PubMed]
- Rochman, C.M.; Lewison, R.L.; Eriksen, M.; Allen, H.; Cook, A.M.; Teh, S.J. Polybrominated diphenyl ethers (PBDEs) in fish tissue may be an indicator of plastic contamination in marine habitats. Sci. Total Environ. 2014, 476–477, 622–633. [Google Scholar] [CrossRef] [PubMed]
- Meeker, J.D.; Sathyanarayana, S.; Swan, S.H. Phthalates and other additives in plastics: Human exposure and associated health outcomes. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 2097–2113. [Google Scholar] [CrossRef] [Green Version]
- Das, A.; Mishra, S. Biodegradation of the metallic carcinogen hexavalent chromium Cr(VI) by an indigenously isolated bacterial strain. J. Carcinog. 2010, 9, 6. [Google Scholar] [CrossRef]
- Taylor, M.L.; Gwinnett, C.; Robinson, L.F.; Woodall, L.C. Plastic microfibre ingestion by deep-sea organisms. Sci. Rep. 2016, 6, 33997. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bergmann, M.; Gutow, L.; Klages, M. Marine Anthropogenic Litter; Springer Nature: Berlin, Germany, 2015; ISBN 9783319165103. [Google Scholar]
- Suaria, G.; Avio, C.G.; Lattin, G.; Regoli, F.; Aliani, S.; Marche, A.I. Neustonic microplastics in the Southern Adriatic Sea. Prelim. Results Micro 2015, 28. [Google Scholar] [CrossRef]
- Avio, C.G.; Gorbi, S.; Regoli, F. Experimental development of a new protocol for extraction and characterization of microplastics in fish tissues: First observations in commercial species from Adriatic Sea. Mar. Environ. Res. 2015, 111, 18–26. [Google Scholar] [CrossRef]
- Collard, F.; Gilbert, B.; Eppe, G.; Parmentier, E.; Das, K. Detection of Anthropogenic Particles in Fish Stomachs: An Isolation Method Adapted to Identification by Raman Spectroscopy. Arch. Environ. Contam. Toxicol. 2015, 69, 331–339. [Google Scholar] [CrossRef]
- Alomar, C.; Sureda, A.; Capó, X.; Guijarro, B.; Tejada, S.; Deudero, S. Microplastic ingestion by Mullus surmuletus Linnaeus, 1758 fish and its potential for causing oxidative stress. Environ. Res. 2017, 159, 135–142. [Google Scholar] [CrossRef]
- Alomar, C.; Deudero, S. Evidence of microplastic ingestion in the shark Galeus melastomus Rafinesque, 1810 in the continental shelf off the western Mediterranean Sea. Environ. Pollut. 2017, 223, 223–229. [Google Scholar] [CrossRef]
- Avio, C.G.; Cardelli, L.R.; Gorbi, S.; Pellegrini, D.; Regoli, F. Microplastics pollution after the removal of the Costa Concordia wreck: First evidences from a biomonitoring case study. Environ. Pollut. 2017, 227, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Compa, M.; Ventero, A.; Iglesias, M.; Deudero, S. Ingestion of microplastics and natural fibres in Sardina pilchardus (Walbaum, 1792) and Engraulis encrasicolus (Linnaeus, 1758) along the Spanish Mediterranean coast. Mar. Pollut. Bull. 2018, 128, 89–96. [Google Scholar] [CrossRef]
- Digka, N.; Tsangaris, C.; Torre, M.; Anastasopoulou, A.; Zeri, C. Microplastics in mussels and fish from the Northern Ionian Sea. Mar. Pollut. Bull. 2018, 135, 30–40. [Google Scholar] [CrossRef]
- Duncan, E.M.; Broderick, A.C.; Fuller, W.J.; Galloway, T.S.; Godfrey, M.H.; Hamann, M.; Limpus, C.J.; Lindeque, P.K.; Mayes, A.G.; Omeyer, L.C.M.; et al. Microplastic ingestion ubiquitous in marine turtles. Glob. Chang. Biol. 2019, 25, 744–752. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anastasopoulou, A.; Kovač Viršek, M.; Bojanić Varezić, D.; Digka, N.; Fortibuoni, T.; Koren, Š.; Mandić, M.; Mytilineou, C.; Pešić, A.; Ronchi, F.; et al. Assessment on marine litter ingested by fish in the Adriatic and NE Ionian Sea macro-region (Mediterranean). Mar. Pollut. Bull. 2018, 133, 841–851. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Garin, O.; Vighi, M.; Aguilar, A.; Tsangaris, C.; Digka, N.; Kaberi, H.; Borrell, A. Boops boops as a bioindicator of microplastic pollution along the Spanish Catalan coast. Mar. Pollut. Bull. 2019, 149, 110648. [Google Scholar] [CrossRef]
- Savoca, S.; Capillo, G.; Mancuso, M.; Faggio, C.; Panarello, G.; Crupi, R.; Bonsignore, M.; D’Urso, L.; Compagnini, G.; Neri, F.; et al. Detection of artificial cellulose microfibers in Boops boops from the northern coasts of Sicily (Central Mediterranean). Sci. Total Environ. 2019, 691, 455–465. [Google Scholar] [CrossRef]
- Giani, D.; Baini, M.; Galli, M.; Casini, S.; Fossi, M.C. Microplastics occurrence in edible fish species (Mullus barbatus and Merluccius merluccius) collected in three different geographical sub-areas of the Mediterranean Sea. Mar. Pollut. Bull. 2019, 140, 129–137. [Google Scholar] [CrossRef]
- Valente, T.; Sbrana, A.; Scacco, U.; Jacomini, C.; Bianchi, J.; Palazzo, L.; de Lucia, G.A.; Silvestri, C.; Matiddi, M. Exploring microplastic ingestion by three deep-water elasmobranch species: A case study from the Tyrrhenian Sea. Environ. Pollut. 2019, 253, 342–350. [Google Scholar] [CrossRef]
- Capillo, G.; Savoca, S.; Panarello, G.; Mancuso, M.; Branca, C.; Romano, V.; D’Angelo, G.; Bottari, T.; Spanò, N. Quali-quantitative analysis of plastics and synthetic microfibers found in demersal species from Southern Tyrrhenian Sea (Central Mediterranean). Mar. Pollut. Bull. 2020, 150, 110596. [Google Scholar] [CrossRef]
- Capone, A.; Petrillo, M.; Misic, C. Ingestion and elimination of anthropogenic fibres and microplastic fragments by the European anchovy (Engraulis encrasicolus) of the NW Mediterranean Sea. Mar. Biol. 2020, 167, 166. [Google Scholar] [CrossRef]
- Renzi, M.; Blašković, A. Chemical fingerprint of plastic litter in sediments and holothurians from Croatia: Assessment & relation to different environmental factors. Mar. Pollut. Bull. 2020, 153, 110994. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Romeu, O.; Constenla, M.; Carrassón, M.; Campoy-Quiles, M.; Soler-Membrives, A. Are anthropogenic fibres a real problem for red mullets (Mullus barbatus) from the NW Mediterranean? Sci. Total Environ. 2020, 733, 139336. [Google Scholar] [CrossRef] [PubMed]
- Savoca, S.; Bottari, T.; Fazio, E.; Bonsignore, M.; Mancuso, M.; Luna, G.M.; Romeo, T.; D’Urso, L.; Capillo, G.; Panarello, G.; et al. Plastics occurrence in juveniles of Engraulis encrasicolus and Sardina pilchardus in the Southern Tyrrhenian Sea. Sci. Total Environ. 2020, 718, 137457. [Google Scholar] [CrossRef] [PubMed]
- Carreras-Colom, E.; Constenla, M.; Soler-Membrives, A.; Cartes, J.E.; Baeza, M.; Carrassón, M. A closer look at anthropogenic fiber ingestion in Aristeus antennatus in the NW Mediterranean Sea: Differences among years and locations and impact on health condition. Environ. Pollut. 2020, 263, 114567. [Google Scholar] [CrossRef] [PubMed]
- Bayo, J.; Rojo, D.; Martínez-Baños, P.; López-Castellanos, J.; Olmos, S. Commercial Gilthead Seabream (Sparus aurata L.) from the Mar Menor Coastal Lagoon as Hotspots of Microplastic Accumulation in the Digestive System. Public Health 2021, 18, 6844. [Google Scholar] [CrossRef] [PubMed]
- Yozukmaz, A. Investigation of microplastics in edible wild mussels from İzmir Bay (Aegean Sea, Western Turkey): A risk assessment for the consumers. Mar. Pollut. Bull. 2021, 171, 112733. [Google Scholar] [CrossRef]
- Sayed, A.E.D.H.; Hamed, M.; Badrey, A.E.A.; Ismail, R.F.; Osman, Y.A.A.; Osman, A.G.M.; Soliman, H.A.M. Microplastic distribution, abundance, and composition in the sediments, water, and fishes of the Red and Mediterranean seas, Egypt. Mar. Pollut. Bull. 2021, 173, 112966. [Google Scholar] [CrossRef]
- Faure, F.; Saini, C.; Potter, G.; Galgani, F.; de Alencastro, L.F.; Hagmann, P. An evaluation of surface micro- and mesoplastic pollution in pelagic ecosystems of the Western Mediterranean Sea. Environ. Sci. Pollut. Res. 2015, 22, 12190–12197. [Google Scholar] [CrossRef]
- Suaria, G.; Avio, C.G.; Mineo, A.; Lattin, G.L.; Magaldi, M.G.; Belmonte, G.; Moore, C.J.; Regoli, F.; Aliani, S. The Mediterranean Plastic Soup: Synthetic polymers in Mediterranean surface waters. Sci. Rep. 2016, 6, 37551. [Google Scholar] [CrossRef] [Green Version]
- Fastelli, P.; Blašković, A.; Bernardi, G.; Romeo, T.; Čižmek, H.; Andaloro, F.; Russo, G.F.; Guerranti, C.; Renzi, M. Plastic litter in sediments from a marine area likely to become protected (Aeolian Archipelago’s islands, Tyrrhenian sea). Mar. Pollut. Bull. 2016, 113, 526–529. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Orejón, L.F.; Sardá, R.; Ramis-Pujol, J. Floating plastic debris in the Central and Western Mediterranean Sea. Mar. Environ. Res. 2016, 120, 136–144. [Google Scholar] [CrossRef] [PubMed]
- Van der Hal, N.; Ariel, A.; Angel, D.L. Exceptionally high abundances of microplastics in the oligotrophic Israeli Mediterranean coastal waters. Mar. Pollut. Bull. 2017, 116, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Cannas, S.; Fastelli, P.; Guerranti, C.; Renzi, M. Plastic litter in sediments from the coasts of south Tuscany (Tyrrhenian Sea). Mar. Pollut. Bull. 2017, 119, 372–375. [Google Scholar] [CrossRef]
- Mistri, M.; Infantini, V.; Scoponi, M.; Granata, T.; Moruzzi, L.; Massara, F.; De Donati, M.; Munari, C. Small plastic debris in sediments from the Central Adriatic Sea: Types, occurrence and distribution. Mar. Pollut. Bull. 2017, 124, 435–440. [Google Scholar] [CrossRef]
- Abidli, S.; Antunes, J.C.; Ferreira, J.L.; Lahbib, Y.; Sobral, P.; Trigui El Menif, N. Microplastics in sediments from the littoral zone of the north Tunisian coast (Mediterranean Sea). Estuar. Coast. Shelf Sci. 2018, 205, 1–9. [Google Scholar] [CrossRef]
- Sanchez-Vidal, A.; Thompson, R.C.; Canals, M.; De Haan, W.P. The imprint of microfibres in Southern European deep seas. PLoS ONE 2018, 13, e0207033. [Google Scholar] [CrossRef]
- Simon-Sánchez, L.; Grelaud, M.; Garcia-Orellana, J.; Ziveri, P. River Deltas as hotspots of microplastic accumulation: The case study of the Ebro River (NW Mediterranean). Sci. Total Environ. 2019, 687, 1186–1196. [Google Scholar] [CrossRef]
- Suaria, G.; Musso, M.; Achtypi, A.; Bassotto, D.; Aliani, S. Textile Fibres in Mediterranean Surface Waters: Abundance and Composition; Springer International Publishing: Cham, Switzerland, 2020; ISBN 9783030459093. [Google Scholar]
- Pojar, I.; Kochleus, C.; Dierkes, G.; Ehlers, S.M.; Reifferscheid, G.; Stock, F. Quantitative and qualitative evaluation of plastic particles in surface waters of the Western Black Sea. Environ. Pollut. 2021, 268, 115724. [Google Scholar] [CrossRef]
- Bošković, N.; Joksimović, D.; Peković, M.; Perošević-Bajčeta, A.; Bajt, O. Marine Science and Engineering Microplastics in Surface Sediments along the Montenegrin Coast, Adriatic Sea: Types, Occurrence, and Distribution. J. Mar. Sci. Eng. 2021, 9, 841. [Google Scholar] [CrossRef]
- Angiolillo, M.; Gérigny, O.; Valente, T.; Fabri, M.C.; Tambute, E.; Rouanet, E.; Claro, F.; Tunesi, L.; Vissio, A.; Daniel, B.; et al. Distribution of seafloor litter and its interaction with benthic organisms in deep waters of the Ligurian Sea (Northwestern Mediterranean). Sci. Total Environ. 2021, 788, 147745. [Google Scholar] [CrossRef] [PubMed]
- Miller, R.Z.; Watts, A.J.R.; Winslow, B.O.; Galloway, T.S.; Barrows, A.P.W. Mountains to the sea: River study of plastic and non-plastic microfiber pollution in the northeast USA. Mar. Pollut. Bull. 2017, 124, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Rochman, C.M.; Tahir, A.; Williams, S.L.; Baxa, D.V.; Lam, R.; Miller, J.T.; Teh, F.C.; Werorilangi, S.; Teh, S.J. Anthropogenic debris in seafood: Plastic debris and fibers from textiles in fish and bivalves sold for human consumption. Sci. Rep. 2015, 5, 14340. [Google Scholar] [CrossRef] [PubMed]
- Nadal, M.A.; Alomar, C.; Deudero, S. High levels of microplastic ingestion by the semipelagic fish bogue Boops boops (L.) around the Balearic Islands. Environ. Pollut. 2016, 214, 517–523. [Google Scholar] [CrossRef] [PubMed]
- Naidoo, T.; Smit, A.J.; Glassom, D. Plastic ingestion by estuarine mullet Mugil cephalus (Mugilidae) in an urban harbour, KwaZulu-Natal, South Africa. Afr. J. Mar. Sci. 2016, 38, 145–149. [Google Scholar] [CrossRef]
- Herrera, A.; Ŝtindlová, A.; Martínez, I.; Rapp, J.; Romero-Kutzner, V.; Samper, M.D.; Montoto, T.; Aguiar-González, B.; Packard, T.; Gómez, M. Microplastic ingestion by Atlantic chub mackerel (Scomber colias) in the Canary Islands coast. Mar. Pollut. Bull. 2019, 139, 127–135. [Google Scholar] [CrossRef]
- Le Guen, C.; Suaria, G.; Sherley, R.B.; Ryan, P.G.; Aliani, S.; Boehme, L.; Brierley, A.S. Microplastic study reveals the presence of natural and synthetic fibres in the diet of King Penguins (Aptenodytes patagonicus) foraging from South Georgia. Environ. Int. 2020, 134, 105303. [Google Scholar] [CrossRef]
- Wesch, C.; Barthel, A.K.; Braun, U.; Klein, R.; Paulus, M. No microplastics in benthic eelpout (Zoarces viviparus): An urgent need for spectroscopic analyses in microplastic detection. Environ. Res. 2016, 148, 36–38. [Google Scholar] [CrossRef]
- Comnea-Stancu, I.R.; Wieland, K.; Ramer, G.; Schwaighofer, A.; Lendl, B. On the Identification of Rayon/Viscose as a Major Fraction of Microplastics in the Marine Environment: Discrimination between Natural and Manmade Cellulosic Fibers Using Fourier Transform Infrared Spectroscopy. Appl. Spectrosc. 2017, 71, 939–950. [Google Scholar] [CrossRef]
- Faruk, O.; Bledzki, A.K.; Fink, H.P.; Sain, M. Progress report on natural fiber reinforced composites. Macromol. Mater. Eng. 2014, 299, 9–26. [Google Scholar] [CrossRef]
- Röder, T.; Moosbauer, J.; Wöss, K.; Schlader, S.; Kraft, G. Man-Made Cellulose Fibres-a Comparison Based on Morphology and Mechanical Properties. Lenzinger Berichte 2013, 91, 7–12. [Google Scholar]
- Bredereck, K.; Hermanutz, F. Man-made cellulosics. Color. Technol. 2008, 35, 59–75. [Google Scholar] [CrossRef]
- Morseletto, P. A new framework for policy evaluation: Targets, marine litter, Italy and the Marine Strategy Framework Directive. Mar. Policy 2020, 117, 103956. [Google Scholar] [CrossRef]
- Henry, B.; Laitala, K.; Klepp, I.G. Microfibres from apparel and home textiles: Prospects for including microplastics in environmental sustainability assessment. Sci. Total Environ. 2019, 652, 483–494. [Google Scholar] [CrossRef] [PubMed]
- Stagioni, M.; Montanini, S.; Vallisneri, M. Feeding habits of European hake, Merluccius merluccius (Actinopterygii: Gadiformes: Merlucciidae), from the northeastern Mediterranean sea. Acta Ichthyol. Piscat. 2011, 41, 277–284. [Google Scholar] [CrossRef]
- Matić-Skoko, S.; Šegvić-Bubić, T.; Mandić, I.; Izquierdo-Gomez, D.; Arneri, E.; Carbonara, P.; Grati, F.; Ikica, Z.; Kolitari, J.; Milone, N.; et al. Evidence of subtle genetic structure in the sympatric species Mullus barbatus and Mullus surmuletus (Linnaeus, 1758) in the Mediterranean Sea. Sci. Rep. 2018, 8, 676. [Google Scholar] [CrossRef]
- Anastasopoulou, A.; Mytilineou, C.; Smith, C.J.; Papadopoulou, K.N. Plastic debris ingested by deep-water fish of the Ionian Sea (Eastern Mediterranean). Deep. Res. Part I Oceanogr. Res. Pap. 2013, 74, 11–13. [Google Scholar] [CrossRef]
- Matiddi, M.; Hochsheid, S.; Camedda, A.; Baini, M.; Cocumelli, C.; Serena, F.; Tomassetti, P.; Travaglini, A.; Marra, S.; Campani, T.; et al. Loggerhead sea turtles (Caretta caretta): A target species for monitoring litter ingested by marine organisms in the Mediterranean Sea. Environ. Pollut. 2017, 230, 199–209. [Google Scholar] [CrossRef]
- Boerger, C.M.; Lattin, G.L.; Moore, S.L.; Moore, C.J. Plastic ingestion by planktivorous fishes in the North Pacific Central Gyre. Mar. Pollut. Bull. 2010, 60, 2275–2278. [Google Scholar] [CrossRef]
- Cole, M.; Webb, H.; Lindeque, P.K.; Fileman, E.S.; Halsband, C.; Galloway, T.S. Isolation of microplastics in biota-rich seawater samples and marine organisms. Sci. Rep. 2014, 4, 4528. [Google Scholar] [CrossRef] [Green Version]
- Gwinnett, C.; Miller, R.Z. Are we contaminating our samples? A preliminary study to investigate procedural contamination during field sampling and processing for microplastic and anthropogenic microparticles. Mar. Pollut. Bull. 2021, 173, 113095. [Google Scholar] [CrossRef] [PubMed]
- Ng, K.L.; Obbard, J.P. Prevalence of microplastics in Singapore’s coastal marine environment. Mar. Pollut. Bull. 2006, 52, 761–767. [Google Scholar] [CrossRef] [PubMed]
- Prata, J.C.; Reis, V.; da Costa, J.P.; Mouneyrac, C.; Duarte, A.C.; Rocha-Santos, T. Contamination issues as a challenge in quality control and quality assurance in microplastics analytics. J. Hazard. Mater. 2021, 403, 123660. [Google Scholar] [CrossRef]
- Cowger, W.; Booth, A.M.; Hamilton, B.M.; Thaysen, C.; Primpke, S.; Munno, K.; Lusher, A.L.; Dehaut, A.; Vaz, V.P.; Liboiron, M.; et al. Special Issue: Microplastics Reporting Guidelines to Increase the Reproducibility and Comparability of Research on Microplastics. Appl. Spectrosc. 2020, 74, 1066–1077. [Google Scholar] [CrossRef]
- Miller, E.; Sedlak, M.; Lin, D.; Box, C.; Holleman, C.; Rochman, C.M.; Sutton, R. Recommended best practices for collecting, analyzing, and reporting microplastics in environmental media: Lessons learned from comprehensive monitoring of San Francisco Bay. J. Hazard. Mater. 2021, 409, 124770. [Google Scholar] [CrossRef] [PubMed]
- Gaylarde, C.; Baptista-Neto, J.A.; da Fonseca, E.M. Plastic microfibre pollution: How important is clothes’ laundering? Heliyon 2021, 7, e07105. [Google Scholar] [CrossRef]
- Zayen, A.; Sayadi, S.; Chevalier, C.; Boukthir, M.; Ben Ismail, S.; Tedetti, M. Microplastics in surface waters of the Gulf of Gabes, southern Mediterranean Sea: Distribution, composition and influence of hydrodynamics. Estuar. Coast. Shelf Sci. 2020, 242, 106832. [Google Scholar] [CrossRef]
- Schönlau, C.; Karlsson, T.M.; Rotander, A.; Nilsson, H.; Engwall, M.; van Bavel, B.; Kärrman, A. Microplastics in sea-surface waters surrounding Sweden sampled by manta trawl and in-situ pump. Mar. Pollut. Bull. 2020, 153, 111019. [Google Scholar] [CrossRef]
- Kuklinski, P.; Wicikowski, L.; Koper, M.; Grala, T.; Leniec-Koper, H.; Barasiński, M.; Talar, M.; Kamiński, I.; Kibart, R.; Małecki, W. Offshore surface waters of Antarctica are free of microplastics, as revealed by a circum-Antarctic study. Mar. Pollut. Bull. 2019, 149, 110573. [Google Scholar] [CrossRef]
- Scopetani, C.; Esterhuizen-Londt, M.; Chelazzi, D.; Cincinelli, A.; Setälä, H.; Pflugmacher, S. Self-contamination from clothing in microplastics research. Ecotoxicol. Environ. Saf. 2020, 189, 110036. [Google Scholar] [CrossRef]
- Grigorakis, S.; Mason, S.A.; Drouillard, K.G. Determination of the gut retention of plastic microbeads and microfibers in goldfish (Carassius auratus). Chemosphere 2017, 169, 233–238. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Area | Year of Sampling | Predominant Type (%) | Instrumental Method | References |
|---|---|---|---|---|
| Calvi Bay (Corsica) | 2011–2012 | All fibrous in shape | Raman | [25] |
| Southern Adriatic Sea | 2013 | 78.5% fragments | FTIR | [61] |
| Central and North Adriatic Sea | 2014 | 57% fragments | FTIR | [62] |
| Gulf of Lions (France) | 2013 | 37.1%. fibers | Raman | [63] |
| Spanish Mediterranean coast | 2014 | 71% fibers | n.a. | [26] |
| Mediterranean coast of Turkey | 2015 | 70% fibers | FTIR | [27] |
| Mallorca Island (Balearic Islands, Western Mediterranean) | 2014–2015 | 97% fibers | FTIR | [64] |
| Mallorca Island (Balearic Islands, Western Mediterranean) | n.a. | 86.4% fibers | FTIR | [65] |
| Giglio Island | 2014 | 60% fragments | FTIR | [66] |
| Western Spanish Mediterranean coast | 2015 | 83% fibers | FTIR | [67] |
| Northern Ionian Sea(M. galloprovincialis; S. pilchardus, P. erithrinus, M. barbatus) | 2015 | 77.8% fragments | FTIR | [68] |
| 80% fragments | ||||
| 73.3% fragments | ||||
| 83.3% fragments | ||||
| Northern Cyprus | n.a. | 85.3% fibers | FTIR | [69] |
| Adriatic and NE Ionian Sea (Croatian Sea; Slovenian Sea, NE Ionian sea) | 2014–2015 | 75.6% fibers | n.a. | [70] |
| 97.7% fibers | ||||
| 79% fragments | ||||
| Spanish Catalan coast | 2018 | ~60% fragments | FTIR | [71] |
| Tyrrhenian Sea (Northern coasts of Sicily, Gulf of Patti) | 2019 | 93.3% fibers | µ-Raman, XPS and SEM-EDX | [72] |
| Mediterranean Sea (European hake, Red mullet) | n.a. | 81% fibers | n.a. | [73] |
| 44% fibers | ||||
| Anzio coast (south of Rome, Tyrrhenian Sea) | 2018 | 85.7% fibers | FTIR | [74] |
| Tyrrhenian Sea (northern coasts of Sicily, Gulf of Patti | 2017 | 97.1% fibers | ATR-FTIR and µ-Raman | [75] |
| Iberian Peninsula coast and Balearic Islands (Western Mediterranean Sea) | 2015 | 92.9% fibers | n.a. | [30] |
| Ligurian Sea | 2011–2014 | n.a. fibers | FTIR | [76] |
| Silba Island and Telašćica (Croatia, Adriatic Sea) | 2007 and 2018 | 39.4–43.3% fibers | µ-FTIR | [77] |
| 35.7–57.5% fibers | ||||
| NW Mediterranean (Catalan coast) | n.a. | 97% fibers | Raman | [78] |
| Northern, Central and Southern Adriatic Sea (Pelagic, benthopelagic, demerdal and benthic organism) | 2016 | 38% fragments | µ-FTIR | [38] |
| 50% fragments | ||||
| 53% fragments | ||||
| 61% fragments | ||||
| Gulf of Patti (Southern Tyrrhenian Sea) | 2019 | 93.3% fibers | FTIR and Raman | [79] |
| Catalan coast (NW Mediterranean Sea) | 2007, 2017 and 2018 | 84.6% fibers | FTIR | [80] |
| Southeast Spain | 2018, 2019 | 71.7% fibers | FTIR | [81] |
| Turkey, Izmir bay | 2020 | 87.2% fibers | n.a. | [82] |
| Egypt cost (Mars Mtruh, Port Said, Alexandria, Damietta) | 2020 | 100% fibers | ATR-FTIR | [83] |
| 50% fragments | ||||
| 96.2% fragments | ||||
| 85.2% fragments |
| Area | Year of Sampling | Predominant Type (%) | Instrumental Method | References |
|---|---|---|---|---|
| Mediterranean Sea | 2001–2012 | All fibrous in shape | FTIR | [22] |
| Gulf of Lion, the BalearicIslands, Sardinia and Corsica | 2012 | 72% fragments | n.a. | [84] |
| Southern Adriatic Sea | 2013 | 78.5% fragments | FTIR | [61] |
| Mediterranean Sea | 2013 | n.a. | ATR-FTIR | [85] |
| Aeolian Archipelago (central Mediterranean and Tyrrhenian sea) | n.a. | >85% fibers | n.a. | [86] |
| Mediterranean cost of Turkey | 2015 | 70% fibers | FTIR | [27] |
| Balearic Islands, Adriatic and Ionian Sea | 2011 and 2013 | 87.3% fragments | n.a. | [87] |
| Israeli Mediterranean coast | 2013–2015 | 96.2% fragments | n.a. | [88] |
| Tyrrhenian Sea | 2012 | >88% fibers | n.a. | [89] |
| Central Adriatic Sea | 2015 | 69.3% fibers | FTIR | [90] |
| Northern Tunisian coast(South Lake of Tunis, North Lake of Tunis, Carthage, Goulette) | 2017 | 66.8% fibers | FTIR | [91] |
| 87.3% fibers | ||||
| 71% fibers | ||||
| 98.8% fibers | ||||
| Alboran, Catalan, Cretan and Levantine Sea | 2009–2015 | All fibrous in shape | FTIR | [92] |
| Ebro River Delta (Catalonia, Spain, Northwestern Mediterranean) (Sand, benthic sediment, surface water) | 2017 | 89.5% fibers | µ-FTIR | [93] |
| 75.1% fibers | ||||
| 46.1% fibers | ||||
| Spanish Mediterranean Coast | 2014–2015 | 82.9% fibers | n.a. | [19] |
| Silba Island and Telašćica (Croatia, Adriatic Sea) | 2007 and 2018 | 33.1–76.9% fibers | µ-FTIR | [77] |
| 82.7–97.3% fibers | ||||
| Central-western Mediterranean Sea | 2017 | All fibrous in shape | FTIR | [94] |
| Mediterranean Sea | 2017 | All fibrous in shape | µ-FTIR | [37] |
| Northwestern Mediterranean Sea (Naples, Corsica north and south-east cost of France) | 2014 | All fibrous in shape | FTIR | [39] |
| Danube delta | 2018 | 74.6% fibers | ATR-FTIR | [95] |
| Montenegrin cost | 2019 | 55.5% fibers | ATR-FTIR | [96] |
| Ligurian Sea coastal | 2018 | 80% fibers | n.a. | [97] |
| Species | No of Individuals | The total Amount of MFs | No of Identified MFs | Plastic Materials (%) | Non-Synthetic Materials (%) | Reference | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PVC | PET | PP | PE | PA | Nylon | PEST | PS | Others | Cellulose | Cellulose-Based | Others | |||||
| Macroinvertebrates * | 235 | 91 | 11 | 100 | [25] | |||||||||||
| Holothuria tubulosa (Gmelin, 1788) (Telaščica, Silba) | 170 | n.a. | n.a. | 13.2 | 21.6 | 27.5 | 1.2 | 9.8 | 13.9 | 12.7 | [77] ** | |||||
| 17.6 | 12.9 | 12.8 | 12.7 | 11.6 | 5.3 | 25.8 | 1.4 | |||||||||
| Invertebrates and fishes * | <500 | 2079 | 100 | 1 | 10 | 74 | 8 | 7 | [38] | |||||||
| Boobs boops (Linnaeus, 1758) | 30 | 80 | 16 | n.a. | [72] | |||||||||||
| Teleosts * | 125 | 18 | n.a. | 31.2 | 6.2 | 12.5 | 31.2 | 18.7 | [75] ** | |||||||
| Elasmobranchs * | ||||||||||||||||
| Mullus barbatus (Linnaeus, 1758) | 118 | 167 | 39 | 31.1 | 56.8 | [78] | ||||||||||
| Mullus surmuletus (Linnaeus, 1758) | 417 | n.a. | n.a. | 36.3 | 33.3 | 30.3 | [64] | |||||||||
| Galeus melastomus (Rafinesque, 1810) | 125 | n.a. | n.a. | 27.3 | 12.1 | 4.5 | 3 | 19.7 | 33.3 | [65] | ||||||
| Caretta caretta (Linnaeus, 1758) | 102 | 811 | 169 | 20.7 | 4.9 | 61.2 | 5.8 | 7.4 | [69] | |||||||
| Chelonia mydas (Linnaeus, 1758) | ||||||||||||||||
| Engraulis encrasicolus (Linnaeus, 1758) | 9 | 35 | 19 | 45.7 | 54.3 | [63] ** | ||||||||||
| Plankton | 29 | 1140 | n.a. | 10 | 41 | 3 | 12 | 5 | 22 | 7 | [61] ** | |||||
| Sardina pilchardus (Walbaum, 1792) | 105 | 41 | 24 | 12.5 | 8.3 | 4.2 | 8.3 | 54.1 | 8.3 | 4.2 | [67] ** | |||||
| Engraulis encrasicolus (Linnaeus, 1758) | ||||||||||||||||
| Sardina pilchardus (Walbaum, 1758) | 264 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | [79] ** | ||||||||
| Engraulis encrasicolus (Linnaeus, 1758) | ||||||||||||||||
| Sparus aurata (Linnaeus, 1758) | 17 | 279 | n.a. | 2.2 | 2.9 | 21.5 | 2.2 | 4.4 | 2.2 | 71.3 | [81] | |||||
| Caranx crysos (Mitchill, 1815), Liza aurata (Risso, 1810), Siganus rivulatus (Rüppell, 1828), and Epinephelus caninus (Valenciennes, 1843) | 3 | 480 | n.a. | 35.0 | 6.7 | 5 | 16.6 | 36.7 | [83] | |||||||
| 3 | 383 | n.a. | 23.3 | 6.7 | 15.7 | 53.3 | ||||||||||
| 3 | 526 | n.a. | 18.8 | 8.4 | 8.4 | 6.7 | 56.7 | |||||||||
| 3 | 648 | n.a. | 18.8 | 8.4 | 8.4 | 6.7 | 56.7 | |||||||||
| Sample | No of Samples | The Total Amount of MFs | Subset of MFs for Analysis | Plastic Materials (%) | Non-Synthetic Materials (%) | Reference | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PVC | PET | PP | PE | PA | Nylon | PEST | PAN | PS | Others | Cellulose | Cellulose-Based | Others | ||||||
| Sediment | 12 | n.a. | n.a. | 23 | 14.7 | 5.3 | 56.9 | [22] | ||||||||||
| 29 | 202 | all | 12.9 | 1 | 1 | 1 | 4.5 | 79.7 | [92] | |||||||||
| Sediment (Telaščica) | 51 | n.a. | n.a. | 23 | 26.9 | 22.6 | 9.7 | 3.6 | [77] * | |||||||||
| Sediment(Silba) | 18 | 16.2 | 14 | 12.2 | 17.2 | 1.2 | 6.8 | 13.7 | 0.8 | |||||||||
| Beaches | 5 | 197 | 25 | 8 | 16 | 24 | 12 | 16 | 12 | [93] * | ||||||||
| Sediment | n.a. | 229 | ||||||||||||||||
| Surface water | n.a. | 293 | ||||||||||||||||
| Seawater | 29 | 1140 | n.a. | 10 | 41 | 3 | 12 | 5 | 22 | 7 | [61] * | |||||||
| 916 | 23,593 | 2134 | 0.4 | 0.3 | 0.7 | 6.2 | 0.7 | 79.5 | 12.3 | [37] ** | ||||||||
| 108 | 5466 | 336 | 0.9 | 0.6 | 0.9 | 4.2 | 0.3 | 47.3 | 39.6 | 5.4 | [94] | |||||||
| Seawater (Haliotis outfall) | 9 | 65 | 27 | 9 | 30 | 22 | 13 | 17 | 35 | [39] *** | ||||||||
| Seawater (Point B) | 9 | 23 | 15 | 17 | 33 | 17 | 33 | 72 | ||||||||||
| Seawater (Bastia) | 9 | 32 | 24 | 60 | 40 | 58 | ||||||||||||
| Seawater (Dyfamed) | 9 | 178 | 38 | 38 | 62 | 47 | ||||||||||||
| Surface water | 12 | 3289 | 93 | 33.3 | 30.1 | 1.1 | 1.1 | 4.3 | 4.3 | 3.3 | [95] | |||||||
| Sediment | 10 | 688 | 103 | 54.5 | 9.7 | 2.0 | 22.2 | 5.1 | 6.4 | [96] | ||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santini, S.; De Beni, E.; Martellini, T.; Sarti, C.; Randazzo, D.; Ciraolo, R.; Scopetani, C.; Cincinelli, A. Occurrence of Natural and Synthetic Micro-Fibers in the Mediterranean Sea: A Review. Toxics 2022, 10, 391. https://doi.org/10.3390/toxics10070391
Santini S, De Beni E, Martellini T, Sarti C, Randazzo D, Ciraolo R, Scopetani C, Cincinelli A. Occurrence of Natural and Synthetic Micro-Fibers in the Mediterranean Sea: A Review. Toxics. 2022; 10(7):391. https://doi.org/10.3390/toxics10070391
Chicago/Turabian StyleSantini, Saul, Eleonora De Beni, Tania Martellini, Chiara Sarti, Demetrio Randazzo, Roberto Ciraolo, Costanza Scopetani, and Alessandra Cincinelli. 2022. "Occurrence of Natural and Synthetic Micro-Fibers in the Mediterranean Sea: A Review" Toxics 10, no. 7: 391. https://doi.org/10.3390/toxics10070391
APA StyleSantini, S., De Beni, E., Martellini, T., Sarti, C., Randazzo, D., Ciraolo, R., Scopetani, C., & Cincinelli, A. (2022). Occurrence of Natural and Synthetic Micro-Fibers in the Mediterranean Sea: A Review. Toxics, 10(7), 391. https://doi.org/10.3390/toxics10070391









