Fasting Enhances the Acute Toxicity of Acrylonitrile in Mice via Induction of CYP2E1
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Animals and Treatment
2.3. Behavioral Observations
2.4. Sample Preparation
2.5. Preparation of Liver Microsomes
2.6. CYP2E1 Activity, CN- Content, and GSH Assay
2.7. Statistical and Data Analysis
3. Results
3.1. Effect of Fasting on Acute Toxicity in Mice Exposed to Different Doses of AN
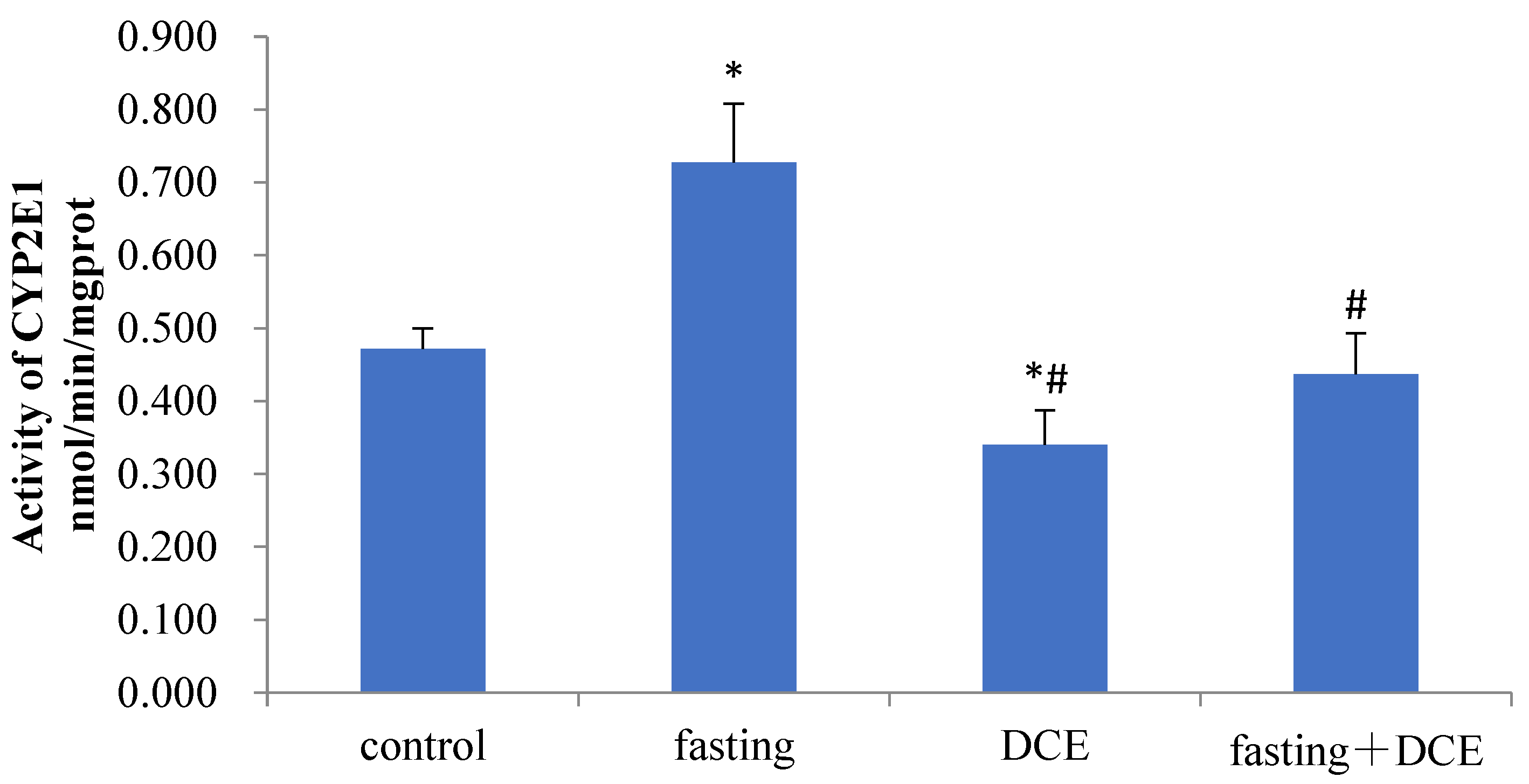
3.2. Effects of Fasting and DCE on Hepatic CYP2E1 Activity
3.3. Effects of Fasting and DCE on AN-Induced Acute Toxicity
3.4. Effects of Fasting and DCE on Tissue CN- in Mice Treated with AN
3.5. Effects of Fasting and DCE on Tissue GSH Level in Mice Treated with AN
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AN | Acrylonitrile |
| CYP2E1 | Cytochrome P450 2E1 |
| CN- | Cyanide ions |
| DCE | trans-1,2-dichloroethylene |
| GSH | Glutathione |
| HSC | Hematopoietic stem cells |
| i.p. | Intraperitoneally |
References
- Golbidi, S.; Daiber, A.; Korac, B.; Li, H.; Essop, M.F.; Laher, I. Health benefits of fasting and caloric restriction. Curr. Diabetes Rep. 2017, 17, 123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schroder, J.D.; Falqueto, H.; Mânica, A.; Zanini, D.; de Oliveira, T.; de Sá, C.A.; Cardoso, A.M.; Manfredi, L.H. Effects of time-restricted feeding in weight loss, metabolic syndrome and cardiovascular risk in obese women. J. Transl. Med. 2021, 19, 3. [Google Scholar] [CrossRef] [PubMed]
- De Cabo, R.; Mattson, M.P. Effects of Intermittent Fasting on Health, Aging, and Disease. N. Engl. J. Med. 2019, 381, 2541–2551. [Google Scholar] [CrossRef] [PubMed]
- Uehara, T.; Kosyk, O.; Jeannot, E.; Bradford, B.U.; Tech, K.; Macdonald, J.M.; Boorman, G.A.; Chatterjee, S.; Mason, R.P.; Melnyk, S.B.; et al. Acetaminophen-induced acute liver injury in HCV transgenic mice. Toxicol. Appl. Pharmacol. 2013, 266, 224–232. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lammers, L.A.; Achterbergh, R.; Mathôt, R.A.A.; Romijn, J.A. The effects of fasting on drug metabolism. Expert Opin. Drug Metab. Toxicol. 2020, 16, 79–85. [Google Scholar] [CrossRef]
- Yang, C.S.; Brady, J.F.; Hong, J.Y. Dietary effects on cytochromes P450, xenobiotic metabolism, and toxicity. FASEB J. 1992, 6, 737–744. [Google Scholar] [CrossRef]
- Spencer, P.S.; Palmer, V.S. Interrelationships of undernutrition and neurotoxicity: Food for thought and research attention. Neurotoxicology 2012, 33, 605–616. [Google Scholar] [CrossRef] [Green Version]
- De Smedt, T.; De Cremer, K.; Vleminckx, C.; Fierens, S.; Mertens, B.; Van Overmeire, I.; Bader, M.; De Paepe, P.; Göen, T.; Nemery, B.; et al. Acrylonitrile exposure in the general population following a major train accident in Belgium: A human biomonitoring study. Toxicol. Lett. 2014, 231, 344–351. [Google Scholar] [CrossRef] [Green Version]
- Scott, A. Acrylonitrile fumes kill one in Belgium. Chemical & Engineering News. 13 May 2013. Available online: http://cen.acs.org/articles/91/i19/Acrylonitrile-Fumes-Kill-One-Belgium.html?type=paidArticleContent: (accessed on 10 December 2020).
- Specia, M. Thousands Evacuated after Train Carrying Toxic Chemicals Derails in Tennessee. 2015. Available online: https://mashable.com/2015/07/02/train-derailment-tennessee/ (accessed on 27 January 2021).
- The Agency for Toxic Substances and Disease Registry (ATSDR). Toxicological Profile for Acrylonitrile. Available online: https://www.atsdr.cdc.gov/ToxProfiles/tp125.pdf (accessed on 16 June 2022).
- Wang, H.; Chanas, B.; Ghanayem, B.I. Cytochrome P450 2E1 (CYP2E1) is essential for acrylonitrile metabolism to cyanide: Comparative studies using CYP2E1-null and wild-type mice. Drug Metab. Dispos. 2002, 30, 911–917. [Google Scholar] [CrossRef] [Green Version]
- Caito, S.W.; Yu, Y.; Aschner, M. Differential inflammatory response to acrylonitrile in rat primary astrocytes and microglia. Neurotoxicology 2014, 42, 1–7. [Google Scholar] [CrossRef]
- Benz, F.W.; Nerland, D.E.; Corbett, D.; Li, J. Biological markers of acute acrylonitrile intoxication in rats as a function of dose and time. Fundam. Appl. Toxicol. 1997, 36, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Miller, K.W.; Yang, C.S. Studies on the mechanisms of induction of N-nitrosodimethylamine demethylase by fasting, acetone, and ethanol. Arch. Biochem. Biophys. 1984, 229, 483–491. [Google Scholar] [CrossRef]
- Brown, B.L.; Allis, J.W.; Simmons, J.E.; House, D.E. Fasting for less than 24 h induces cytochrome P450 2E1 and 2B1/2 activities in rats. Toxicol. Lett. 1995, 81, 39–44. [Google Scholar] [CrossRef]
- Kopecký, J.; Gut, I.; Nerudová, J.; Zachardová, D.; Holecek, V. Two routes of acrylonitrile metabolism. J. Hyg. Epidemiol. Microbiol. Immunol. 1980, 24, 356–362. [Google Scholar] [PubMed]
- Kopecký, J.; Gut, I.; Nerudová, J.; Zachardová, D.; Holeĉek, V.; Filip, J. Acrylonitrile metabolism in the rat. Arch. Toxicol. Suppl. 1980, 4, 322–324. [Google Scholar]
- Abreu, M.E.; Ahmed, A.E. Metabolism of acrylonitrile to cyanide. In vitro studies. Drug Metab. Dispos. 1980, 8, 376–379. [Google Scholar]
- Reinke, L.A.; Moyer, M.J. p-Nitrophenol hydroxylation. A microsomal oxidation which is highly inducible by ethanol. Drug Metab. Dispos. 1985, 13, 548–552. [Google Scholar]
- Koop, D.R. Hydroxylation of p-nitrophenol by rabbit ethanol-inducible cytochrome P-450 isozyme 3a. Mol. Pharmacol. 1986, 29, 399–404. [Google Scholar]
- Ballantyne, B.; Bright, J.E.; Williams, P. The post-mortem rate of transformation of cyanide. Forensic Sci. 1974, 3, 71–76. [Google Scholar] [CrossRef]
- Suhua, W.; Rongzhu, L.; Wenrong, X.; Guangwei, X.; Xiaowu, Z.; Shizhong, W.; Ye, Z.; Fangan, H.; Aschner, M. Induction or inhibition of cytochrome P450 2E1 modifies the acute toxicity of acrylonitrile in rats: Biochemical evidence. Arch. Toxicol. 2010, 84, 461–469. [Google Scholar] [CrossRef]
- Mitchell, J.R.; Verweij, M.; Brand, K.; van de Ven, M.; Goemaere, N.; van den Engel, S.; Chu, T.; Forrer, F.; Müller, C.; de Jong, M.; et al. Short-term dietary restriction and fasting precondition against ischemia reperfusion injury in mice. Aging Cell 2010, 9, 40–53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tinkum, K.L.; Stemler, K.M.; White, L.S.; Loza, A.J.; Jeter-Jones, S.; Michalski, B.M.; Kuzmicki, C.; Pless, R.; Stappenbeck, T.S.; Piwnica-Worms, D.; et al. Fasting protects mice from lethal DNA damage by promoting small intestinal epithelial stem cell survival. Proc. Natl. Acad. Sci. USA 2015, 112, E7148–E7154. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, C.W.; Adams, G.B.; Perin, L.; Wei, M.; Zhou, X.; Lam, B.S.; Da Sacco, S.; Mirisola, M.; Quinn, D.I.; Dorff, T.B.; et al. Prolonged fasting reduces IGF-1/PKA to promote hematopoietic-stem-cell-based regeneration and reverse immunosuppression. Cell Stem Cell 2014, 14, 810–823. [Google Scholar] [CrossRef] [Green Version]
- Dirks-Naylor, A.J.; Kouzi, S.A.; Yang, S.; Tran, N.T.; Bero, J.D.; Mabolo, R.; Phan, D.T.; Whitt, S.D.; Taylor, H.N. Can short-term fasting protect against doxorubicin-induced cardiotoxicity? World J. Biol. Chem. 2014, 5, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Dorff, T.B.; Groshen, S.; Garcia, A.; Shah, M.; Tsao-Wei, D.; Pham, H.; Cheng, C.W.; Brandhorst, S.; Cohen, P.; Wei, M.; et al. Safety and feasibility of fasting in combination with platinum-based chemotherapy. BMC Cancer 2016, 16, 360. [Google Scholar] [CrossRef] [Green Version]
- Templeman, I.; Smith, H.A.; Chowdhury, E.; Chen, Y.C.; Carroll, H.; Johnson-Bonson, D.; Hengist, A.; Smith, R.; Creighton, J.; Clayton, D.; et al. A randomized controlled trial to isolate the effects of fasting and energy restriction on weight loss and metabolic health in lean adults. Sci. Transl. Med. 2021, 13, eabd8034. [Google Scholar] [CrossRef] [PubMed]
- Baillie, T.A.; Rettie, A.E. Role of biotransformation in drug-induced toxicity: Influence of intra- and inter-species differences in drug metabolism. Drug Metab. Pharmacokinet. 2011, 26, 15–29. [Google Scholar] [CrossRef] [Green Version]
- Zhou, S.; Chan, E.; Duan, W.; Huang, M.; Chen, Y.Z. Drug bioactivation, covalent binding to target proteins and toxicity relevance. Drug Metab. Rev. 2005, 37, 41–213. [Google Scholar] [CrossRef]
- Szkudelski, T.; Okulicz, M.; Bialik, I.; Szkudelska, K. The influence of fasting on liver sulfhydryl groups, glutathione peroxidase and glutathione-S-transferase activities in the rat. J. Physiol. Biochem. 2004, 60, 1–6. [Google Scholar] [CrossRef]
- Tsuchiya, Y.; Sakai, H.; Hirata, A.; Yanai, T. Effects of food restriction on the expression of genes related to acetaminophen-induced liver toxicity in rats. J. Toxicol. Pathol. 2018, 31, 267–274. [Google Scholar] [CrossRef] [Green Version]
- Ramaiah, S.K.; Apte, U.; Mehendale, H.M. Cytochrome P4502E1 induction increases thioacetamide liver injury in diet-restricted rats. Drug Metab. Dispos. 2001, 29, 1088–1095. [Google Scholar] [PubMed]
- Guengerich, F.P. Cytochrome P450 2E1 and its roles in disease. Chem. Biol. Interact. 2020, 322, 109056. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.Y.; Pan, J.M.; Gonzalez, F.J.; Gelboin, H.V.; Yang, C.S. The induction of a specific form of cytochrome P-450 (P-450j) by fasting. Biochem. Biophys. Res. Commun. 1987, 142, 1077–1083. [Google Scholar] [CrossRef]
- Ma, J.; Zhang, C.; He, Y.; Chen, X.; Lin, G. Fasting augments pyrrolizidine alkaloid-induced hepatotoxicity. Arch. Toxicol. 2022, 96, 639–651. [Google Scholar] [CrossRef] [PubMed]
- O’Shea, D.; Davis, S.N.; Kim, R.B.; Wilkinson, G.R. Effect of fasting and obesity in humans on the 6-hydroxylation of chlorzoxazone: A putative probe of CYP2E1 activity. Clin. Pharmacol. Ther. 1994, 56, 359–367. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Dong, Y.; Lu, R.; Yang, B.; Wang, S.; Xing, G.; Jiang, Y. Susceptibility to the acute toxicity of acrylonitrile in streptozotocin-induced diabetic rats: Protective effect of phenethyl isothiocyanate, a phytochemical CYP2E1 inhibitor. Drug Chem. Toxicol. 2021, 44, 130–139. [Google Scholar] [CrossRef]
- Li, F.; Lu, R.; Zhao, T.; Zhang, X.; Wang, S.; Xing, G. Comparing the protective effects of three sulfur compounds against acrylonitrile-induced acute toxicity in CYP2E1-induced rats. Toxicol. Ind. Health 2019, 35, 387–397. [Google Scholar] [CrossRef]
- Thier, R.; Lewalter, J.; Bolt, H.M. Species differences in acrylonitrile metabolism and toxicity between experimental animals and humans based on observations in human accidental poisonings. Arch. Toxicol. 2000, 74, 184–189. [Google Scholar] [CrossRef]
- Bolt, H.M.; Roos, P.H.; Thier, R. The cytochrome P-450 isoenzyme CYP2E1 in the biological processing of industrial chemicals: Consequences for occupational and environmental medicine. Int. Arch. Occup. Environ. Health 2003, 76, 174–185. [Google Scholar] [CrossRef]
- Pessayre, D.; Dolder, A.; Artigou, J.Y.; Wandscheer, J.C.; Descatoire, V.; Degott, C.; Benhamou, J.P. Effect of fasting on metabolite-mediated hepatotoxicity in the rat. Gastroenterology 1979, 77, 264–271. [Google Scholar] [CrossRef]
- Walker, R.M.; Massey, T.E.; McElligott, T.F.; Racz, W.J. Acetaminophen toxicity in fed and fasted mice. Can. J. Physiol. Pharmacol. 1982, 60, 399–404. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, T.; Wang, R.S.; Elovaara, E.; Park, S.S.; Gelboin, H.V.; Vainio, H. Cytochrome P450-related differences between rats and mice in the metabolism of benzene, toluene and trichloroethylene in liver microsomes. Biochem. Pharmacol. 1993, 45, 1079–1085. [Google Scholar] [PubMed]
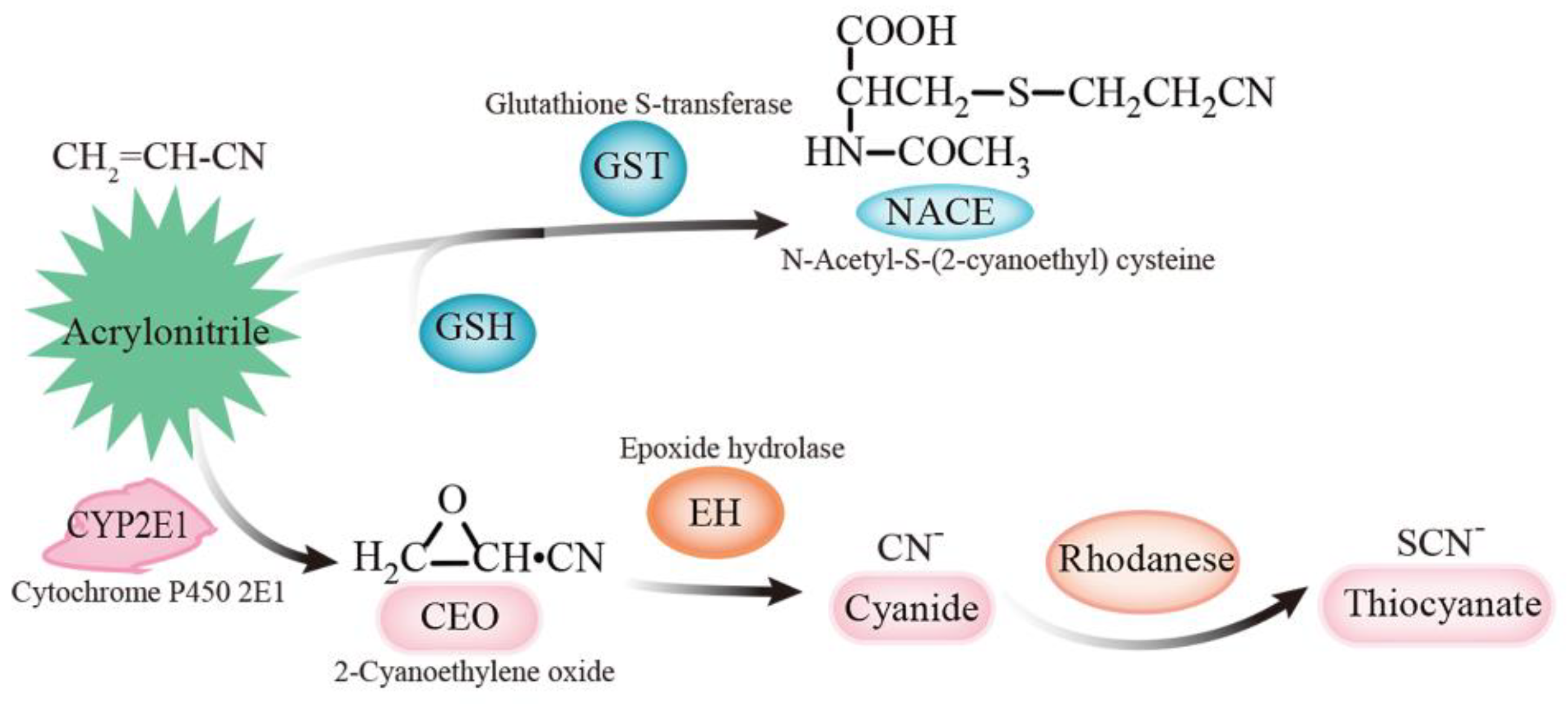
| Group | Weight (g) | Convulsions | Loss of Righting Reflex | Time to Loss of Righting Reflex (min) | Mortality | Time to Mortality (min) |
|---|---|---|---|---|---|---|
| 10 mg/kg AN | 26.6 ± 1.1 | 2 | 0 | - | 0 | - |
| fasting + 10 mg/kg AN | 18.4 ± 0.5 a | 3 | 0 | - | 0 | - |
| 20 mg/kg AN | 26.7 ± 2.0 | 1 | 2 | 12.0 ± 4.2 | 0 | - |
| fasting + 20 mg/kg AN | 19.3 ± 1.5 a | 7 a | 4 | 13.0 ± 5.7 | 1 | 65 |
| 40 mg/kg AN | 26.9 ± 1.2 | 8 | 6 | 13.5 ± 5.5 | 1 | 129 |
| fasting + 40 mg/kg AN | 18.3 ± 1.2 a | 8 | 8 | 13.4 ± 4.6 | 8 a | 56 ± 11.7 |
| Group | Weight (g) | Convulsion(n) | Loss of Righting Reflex (n) | Time to Loss of Righting Reflex (min) | Mortality (n) | Time to Death (min) |
|---|---|---|---|---|---|---|
| 40 mg/kg AN | 28.5 ± 0.9 | 6 | 4 | 30.0 ± 9.8 | 0 | - |
| DCE + 40 mg/kg AN | 29.4 ± 1.4 | 0 a | 0 a | - | 0 | - |
| Fasting + 40 mg/kg AN | 19.5 ±0.9 a | 4 | 8 a,b | 15.8 ± 5.1 | 5 a | 62.6 ± 18.3 |
| Fasting + DCE + 40 mg/kg AN | 19.9 ± 0.7 a,b | 0 ac | 1 c | 54 | 2 | 146.5 ± 82.7 |
| Group | Brain CN- μg/g Tissue | Liver CN- μg/g Tissue | Liver GSH nmol/mg Prot | Brain GSH nmol/mg Prot |
|---|---|---|---|---|
| Normal | - | - | 15.25 ± 1.99 | 6.22 ± 0.63 |
| Fasting | - | - | 6.83 ± 1.21 * | 7.38 ± 0.06 |
| DCE | - | - | 17.48 ± 1.01 | 7.22 ± 0.31 |
| Fasting + DCE | - | - | 5.20 ± 1.02 * | 7.27 ± 0.72 |
| 40 mg/kg AN | 0.159 ± 0.038 | 0.607 ± 0.047 | 7.13 ± 2.75 * | 8.01 ± 0.54 |
| DCE + 40 mg/kg AN | - | 0.024 ± 0.010 a | 3.33 ± 1.02 *,#,a | 7.03 ± 0.86 |
| Fasting + 40 mg/kg AN | 0.415 ± 0.084 a | 0.725 ± 0.096 a,b | 2.51 ± 0.52 *,#,a | 7.02 ± 0.92 |
| Fasting + DCE + 40 mg/kg AN | 0.115 ± 0.020 b | 0.265 ± 0.069 a,b,c | 2.18 ± 0.71 *,#,a | 6.19 ± 0.51 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, S.; Xing, G.; Li, F.; Yang, B.; Zhang, Y.; Aschner, M.; Lu, R. Fasting Enhances the Acute Toxicity of Acrylonitrile in Mice via Induction of CYP2E1. Toxics 2022, 10, 337. https://doi.org/10.3390/toxics10060337
Wang S, Xing G, Li F, Yang B, Zhang Y, Aschner M, Lu R. Fasting Enhances the Acute Toxicity of Acrylonitrile in Mice via Induction of CYP2E1. Toxics. 2022; 10(6):337. https://doi.org/10.3390/toxics10060337
Chicago/Turabian StyleWang, Suhua, Guangwei Xing, Fang Li, Bobo Yang, Yu Zhang, Michael Aschner, and Rongzhu Lu. 2022. "Fasting Enhances the Acute Toxicity of Acrylonitrile in Mice via Induction of CYP2E1" Toxics 10, no. 6: 337. https://doi.org/10.3390/toxics10060337
APA StyleWang, S., Xing, G., Li, F., Yang, B., Zhang, Y., Aschner, M., & Lu, R. (2022). Fasting Enhances the Acute Toxicity of Acrylonitrile in Mice via Induction of CYP2E1. Toxics, 10(6), 337. https://doi.org/10.3390/toxics10060337







