Association between Environmental Tobacco Smoke Exposure and Adaptive Behavior in Individuals with Autism Spectrum Disorder
Abstract
:1. Introduction
- We assumed that children with ASD showed a significant lag in adaptive behavior compared with children suffering from nonspecified developmental difficulties and the neurotypical population.
- We hypothesized a significantly negative relationship between ETS exposure and adaptive behavior in children with ASD.
- What is the profile of adaptive behavior in children with ASD in Slovakia compared with the standardized norms?
- What is the agreement between objective and subjective indicators of exposure to environmental tobacco smoke?
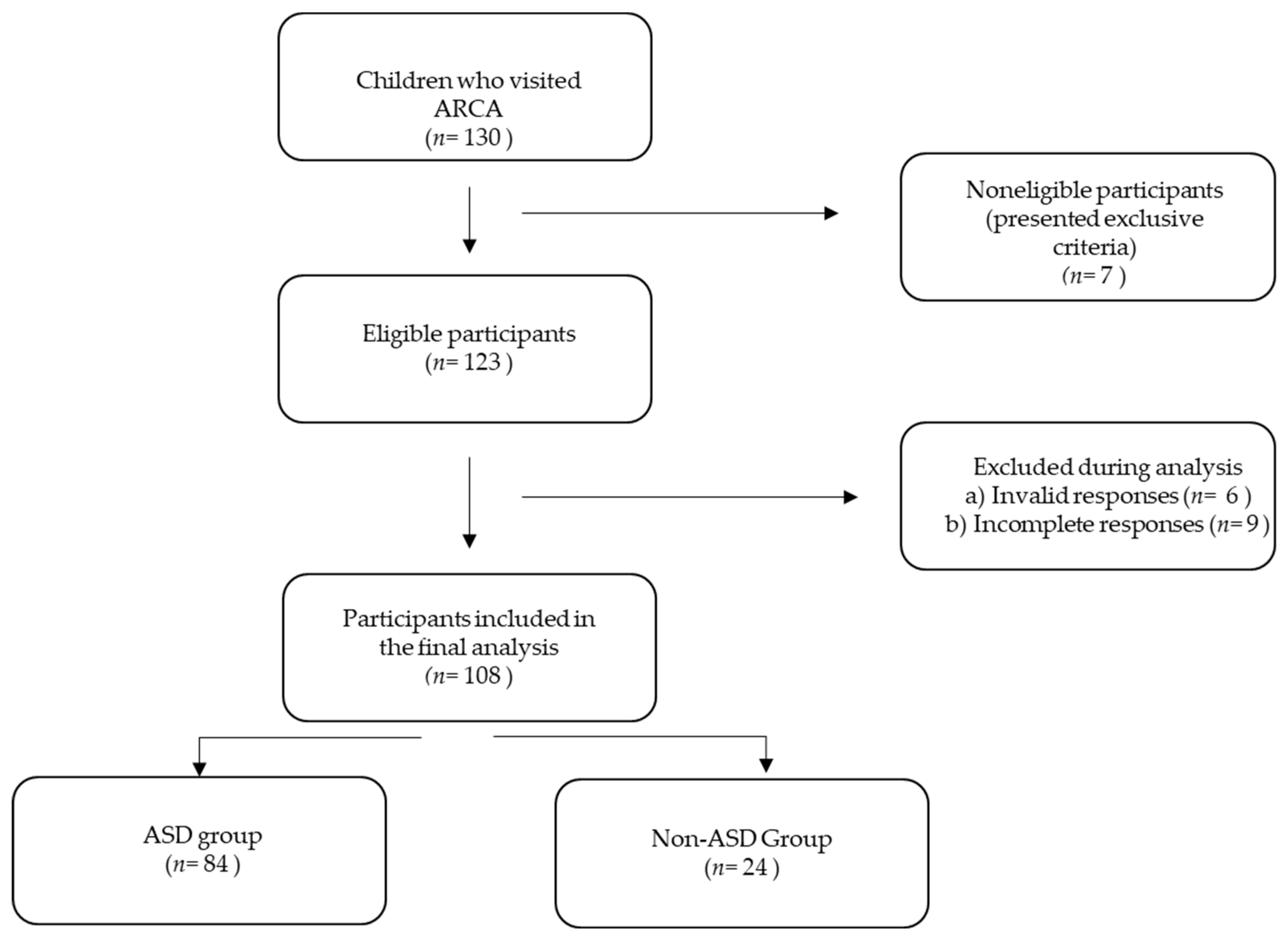
2. Materials and Methods
2.1. Sample
2.2. Diagnostic Evaluation of ASD
2.3. Adaptive Behavior Assessment: Vineland Adaptive Behavior Scale (VABS-3)
2.4. Assessment of ETS Exposure
2.5. Urine Cotinine Concentration Level Measurement
2.6. Statistical Analysis
2.7. Ethical Approval
3. Results
4. Discussion
Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kanne, S.M.; Gerber, A.J.; Quirmbach, L.M.; Sparrow, S.S.; Cicchetti, D.V.; Saulnier, C.A. The Role of Adaptive Behavior in Autism Spectrum Disorders: Implications for Functional Outcome. J. Autism Dev. Disord. 2011, 41, 1007–1018. [Google Scholar] [CrossRef]
- Klin, A.; Saulnier, C.A.; Sparrow, S.S.; Cicchetti, D.V.; Volkmar, F.R.; Lord, C. Social and Communication Abilities and Disabilities in Higher Functioning Individuals with Autism Spectrum Disorders: The Vineland and the ADOS. J. Autism Dev. Disord. 2007, 37, 748–759. [Google Scholar] [CrossRef]
- Yang, S.; Paynter, J.M.; Gilmore, L. Vineland Adaptive Behavior Scales: II Profile of Young Children with Autism Spectrum Disorder. J. Autism Dev. Disord. 2016, 46, 64–73. [Google Scholar] [CrossRef] [Green Version]
- Paul, R.; Loomis, R.; Chawarska, K. Adaptive Behavior in Toddlers Under Two with Autism Spectrum Disorders. J. Autism Dev. Disord. 2014, 44, 264–270. [Google Scholar] [CrossRef]
- WHO. WHO Releases New International Classification of Diseases (ICD 11). Available online: https://www.who.int/news/item/18-06-2018-who-releases-new-international-classification-of-diseases-(icd-11) (accessed on 4 April 2021).
- Sevcikova, L.; Babjakova, J.; Jurkovicova, J.; Samohyl, M.; Stefanikova, Z.; Machacova, E.; Vondrova, D.; Janekova, E.; Hirosova, K.; Filova, A.; et al. Exposure to Environmental Tobacco Smoke in Relation to Behavioral, Emotional, Social and Health Indicators of Slovak School Children. Int. J. Environ. Res. Public Health 2018, 15, 1374. [Google Scholar] [CrossRef] [Green Version]
- Bandiera, F.C.; Richardson, A.K.; Lee, D.J.; He, J.P.; Merikangas, K.R. Secondhand Smoke Exposure and Mental Health Among Children and Adolescents. Arch. Pediatr. Adolesc. Med. 2011, 165, 332–338. [Google Scholar] [CrossRef] [Green Version]
- Berman, T.; Barnett-Itzhaki, Z.; Axelrod, R.; Keinan-Boker, L.; Shimony, T.; Goldsmith, R.; Göen, T.; Geva, H.; Rosen, L. Socioeconomic Inequalities in Exposure to Environmental Tobacco Smoke in Children in Israel. Environ. Int. 2018, 121, 643–648. [Google Scholar] [CrossRef]
- Roberts, C.; Wagler, G.; Carr, M.M. Environmental Tobacco Smoke: Public Perception of Risks of Exposing Children to Second- and Third-Hand Tobacco Smoke. J. Pediatr. Health Care 2017, 31, e7–e13. [Google Scholar] [CrossRef]
- Moran, V.E. Cotinine: Beyond That Expected, More than a Biomarker of Tobacco Consumption. Front. Pharmacol. 2012, 3, 173. [Google Scholar] [CrossRef] [Green Version]
- Kim, S. Overview of Cotinine Cutoff Values for Smoking Status Classification. Int. J. Environ. Res. Public Health 2016, 13, 1236. [Google Scholar] [CrossRef]
- Raja, M.; Garg, A.; Yadav, P.; Jha, K.; Handa, S. Diagnostic Methods for Detection of Cotinine Level in Tobacco Users: A Review. J. Clin. Diagn. Res. 2016, 10, ZE04–ZE06. [Google Scholar] [CrossRef]
- Florescu, A.; Ferrence, R.; Einarson, T.; Selby, P.; Soldin, O.; Koren, G. Methods for Quantification of Exposure to Cigarette Smoking and Environmental Tobacco Smoke: Focus on Developmental Toxicology. Ther. Drug Monit. 2009, 31, 14–30. [Google Scholar] [CrossRef] [Green Version]
- Al-Sayed, E.M.; Ibrahim, K.S. Second-Hand Tobacco Smoke and Children. Toxicol. Ind. Health 2014, 30, 635–644. [Google Scholar] [CrossRef]
- Protano, C.; Vitali, M. The New Danger of Thirdhand Smoke: Why Passive Smoking Does Not Stop at Secondhand Smoke. Environ. Health Perspect. 2011, 119, a422. [Google Scholar] [CrossRef]
- Jung, Y.; Lee, A.M.; McKee, S.A.; Picciotto, M.R. Maternal Smoking and Autism Spectrum Disorder: Meta-Analysis with Population Smoking Metrics as Moderators. Sci. Rep. 2017, 7, 4315. [Google Scholar] [CrossRef]
- Kaur, B. The Association between Autism Spectrum Disorders and Secondhand Tobacco Exposure. Master’s Thesis, Wright State University, Dayton, OH, USA, 2014; p. 40. [Google Scholar]
- Kalkbrenner, A.E.; Schmidt, R.J.; Penlesky, A.C. Environmental Chemical Exposures and Autism Spectrum Disorders: A Review of the Epidemiological Evidence. Curr. Probl. Pediatr. Adolesc. Health Care 2014, 44, 277–318. [Google Scholar] [CrossRef] [Green Version]
- Tang, S.; Wang, Y.; Gong, X.; Wang, G.; Tang, S.; Wang, Y.; Gong, X.; Wang, G. A Meta-Analysis of Maternal Smoking during Pregnancy and Autism Spectrum Disorder Risk in Offspring. Int. J. Environ. Res. Public Health 2015, 12, 10418–10431. [Google Scholar] [CrossRef] [Green Version]
- Cheslack-Postava, K.; Sourander, A.; Hinkka-Yli-Salomäki, S.; McKeague, I.W.; Surcel, H.M.; Brown, A.S. A Biomarker-Based Study of Prenatal Smoking Exposure and Autism in a Finnish National Birth Cohort. Autism Res. 2021, 14, 2444–2453. [Google Scholar] [CrossRef]
- Berger, K.; Pearl, M.; Kharrazi, M.; Li, Y.; DeGuzman, J.; She, J.; Behniwal, P.; Lyall, K.; Windham, G. The Association of in Utero Tobacco Smoke Exposure, Quantified by Serum Cotinine, and Autism Spectrum Disorder. Autism Res. 2021, 14, 2017–2026. [Google Scholar] [CrossRef]
- Tiesler, C.M.T.; Chen, C.-M.; Sausenthaler, S.; Herbarth, O.; Lehmann, I.; Schaaf, B.; Krämer, U.; von Berg, A.; von Kries, R.; Wichmann, H.E.; et al. Passive Smoking and Behavioural Problems in Children: Results from the LISAplus Prospective Birth Cohort Study. Environ. Res. 2011, 111, 1173–1179. [Google Scholar] [CrossRef]
- Padrón, A.; Galán, I.; Rodríguez-Artalejo, F. Second-Hand Smoke Exposure and Psychological Distress in Adolescents. A Population-Based Study. Tob. Control 2014, 23, 302–307. [Google Scholar] [CrossRef]
- Padrón, A.; Galán, I.; García-Esquinas, E.; Fernández, E.; Ballbè, M.; Rodríguez-Artalejo, F. Exposure to Secondhand Smoke in the Home and Mental Health in Children: A Population-Based Study. Tob. Control 2016, 25, 307–312. [Google Scholar] [CrossRef] [Green Version]
- Gatzke-Kopp, L.M.; Willoughby, M.T.; Warkentien, S.M.; O’Connor, T.; Granger, D.A.; Blair, C. Magnitude and Chronicity of Environmental Smoke Exposure Across Infancy and Early Childhood in a Sample of Low-Income Children. Nicotine Tob. Res. 2019, 21, 1665–1672. [Google Scholar] [CrossRef]
- Chen, R.; Clifford, A.; Lang, L.; Anstey, K.J. Is Exposure to Secondhand Smoke Associated with Cognitive Parameters of Children and Adolescents? A Systematic Literature Review. Ann. Epidemiol. 2013, 23, 652–661. [Google Scholar] [CrossRef]
- Wang, H.; Li, F.; Zhang, Y.; Jiang, F.; Zhang, J. The Association between Exposure to Secondhand Smoke and Psychological Symptoms among Chinese Children. BMC Public Health 2019, 19, 923. [Google Scholar] [CrossRef] [Green Version]
- Khorasanchi, Z.; Bahrami, A.; Avan, A.; Jaberi, N.; Rezaey, M.; Bahrami-Taghanaki, H.; Ferns, G.A.; Ghayour-Mobarhan, M. Passive Smoking Is Associated with Cognitive and Emotional Impairment in Adolescent Girls. J. Gen. Psychol. 2019, 146, 68–78. [Google Scholar] [CrossRef]
- Salem, E.A.-A.; Saheen, H.M.; Allam, H.K. Secondhand Tobacco Smoke, Does It Have an Impact on the Neurobehavioral Performance of the Exposed Children? Environ. Sci. Pollut. Res. 2020, 27, 44701–44708. [Google Scholar] [CrossRef]
- Jung, Y.; Hsieh, L.S.; Lee, A.M.; Zhou, Z.; Coman, D.; Heath, C.J.; Hyder, F.; Mineur, Y.S.; Yuan, Q.; Goldman, D.; et al. An Epigenetic Mechanism Mediates Developmental Nicotine Effects on Neuronal Structure and Behavior. Nat. Neurosci. 2016, 19, 905–914. [Google Scholar] [CrossRef]
- Bandiera, F.C. What Are Candidate Biobehavioral Mechanisms Underlying the Association between Secondhand Smoke Exposure and Mental Health? Med. Hypotheses 2011, 77, 1009–1010. [Google Scholar] [CrossRef] [Green Version]
- Kim, K.M.; Lim, M.H.; Kwon, H.J.; Yoo, S.J.; Kim, E.; Kim, J.W.; Ha, M.; Paik, K.C. Associations between Urinary Cotinine and Symptoms of Attention Deficit/Hyperactivity Disorder and Autism Spectrum Disorder. Environ. Res. 2018, 166, 481–486. [Google Scholar] [CrossRef]
- Yang, J.H.; Strodl, E.; Wu, C.A.; Yin, X.N.; Wen, G.M.; Sun, D.L.; Xian, D.X.; Chen, J.Y.; Chen, Y.J.; Chen, J.; et al. Association between Environmental Tobacco Smoke Exposure in Early Life and Autistic-like Behaviors in Chinese Preschoolers. J. Psychosom. Res. 2022, 152, 110680. [Google Scholar] [CrossRef] [PubMed]
- Lord, C.; Risi, S.; Lambrecht, L.; Cook, E.H.; Leventhal, B.L.; DiLavore, P.C.; Pickles, A.; Rutter, M. The Autism Diagnostic Observation Schedule—Generic: A Standard Measure of Social and Communication Deficits Associated with the Spectrum of Autism. J. Autism Dev. Disord. 2000, 30, 205–223. [Google Scholar] [CrossRef] [PubMed]
- Lord, C.; Rutter, M.; Le Couteur, A. Autism Diagnostic Interview-Revised: A Revised Version of a Diagnostic Interview for Caregivers of Individuals with Possible Pervasive Developmental Disorders. J. Autism Dev. Disord. 1994, 24, 659–685. [Google Scholar] [CrossRef] [PubMed]
- Sparrow, S.S.; Cicchetti, D.V.; Saulnier, C.A. Vineland Adaptive Behavior Scales: Vineland-III: Manual; PsychCorp.: Bloomington, MN, USA, 2016. [Google Scholar]
- Pepperdine, C.R.; McCrimmon, A.W. Test Review: Vineland Adaptive Behavior Scales, (Vineland-3) by Sparrow, S.S., Cicchetti, D.V., & Saulnier, C.A. Can. J. Sch. Psychol. 2018, 33, 157–163. [Google Scholar] [CrossRef]
- Sobotova, L.; Liu, Y.H.; Burakoff, A.; Sevcikova, L.; Weitzman, M. Household Exposure to Secondhand Smoke Is Associated with Decreased Physical and Mental Health of Mothers in the USA. Matern. Child Health J. 2011, 15, 128–137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rosner, B.A. Fundamentals of Biostatistics; Duxbury. 2000. Available online: http://balis.bibalex.org/EN/OPAC/Home/SearchOPAC?keyword=Rosner%2C%20Bernard (accessed on 14 February 2022).
- Nevill, R.E.; Hedley, D.; Uljarević, M.; Butter, E.; Mulick, J.A. Adaptive Behavior Profiles in Young Children with Autism Spectrum Disorder Diagnosed under DSM-5 Criteria. Res. Autism Spectr. Disord. 2017, 43–44, 53–66. [Google Scholar] [CrossRef]
- Mouga, S.; Almeida, J.; Café, C.; Duque, F.; Oliveira, G. Adaptive Profiles in Autism and Other Neurodevelopmental Disorders. J. Autism Dev. Disord. 2015, 45, 1001–1012. [Google Scholar] [CrossRef]
- Paul, R.; Miles, S.; Cicchetti, D.; Sparrow, S.; Klin, A.; Volkmar, F.; Coflin, M.; Booker, S. Adaptive Behavior in Autism and Pervasive Developmental Disorder-Not Otherwise Specified: Microanalysis of Scores on the Vineland Adaptive Behavior Scales. J. Autism Dev. Disord. 2004, 34, 223–228. [Google Scholar] [CrossRef]
- Tomanik, S.S.; Pearson, D.A.; Loveland, K.A.; Lane, D.M.; Bryant Shaw, J. Improving the Reliability of Autism Diagnoses: Examining the Utility of Adaptive Behavior. J. Autism Dev. Disord. 2007, 37, 921–928. [Google Scholar] [CrossRef]
- Golya, N.; McIntyre, L.L. Variability in Adaptive Behaviour in Young Children with Autism Spectrum Disorder. J. Intellect. Dev. Disabil. 2018, 43, 102–111. [Google Scholar] [CrossRef]
- Lord, C.; Brugha, T.S.; Charman, T.; Cusack, J.; Dumas, G.; Frazier, T.; Jones, E.J.H.; Jones, R.M.; Pickles, A.; State, M.W.; et al. Autism Spectrum Disorder. Nat. Rev. Dis. Primers 2020, 6, 5. [Google Scholar] [CrossRef]
- Dwyer, J.B.; McQuown, S.C.; Leslie, F.M. The Dynamic Effects of Nicotine on the Developing Brain. Pharmacol. Ther. 2009, 122, 125–139. [Google Scholar] [CrossRef] [Green Version]
- Yolton, K.; Dietrich, K.; Auinger, P.; Lanphear, B.P.; Hornung, R. Exposure to Environmental Tobacco Smoke and Cognitive Abilities among U.S. Children and Adolescents. Environ. Health Perspect. 2005, 113, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, N.N.; Loy, S.L.; Lim, P.Y.; Al Mamun, A.; Jan Mohamed, H.J. Early Life Secondhand Smoke Exposure Assessed by Hair Nicotine Biomarker May Reduce Children’s Neurodevelopment at 2 years of Age. Sci. Total Environ. 2018, 610–611, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Chastang, J.; Baïz, N.; Cadwalladder, J.S.; Robert, S.; Dywer, J.; Charpin, D.A.; Caillaud, D.; de Blay, F.; Raherison, C.; Lavaud, F.; et al. Postnatal Environmental Tobacco Smoke Exposure Related to Behavioral Problems in Children. PLoS ONE 2015, 10, e0133604. [Google Scholar] [CrossRef] [Green Version]
- Khalil, N.; Kaur, B.; Lawson, A.; Ebert, J.; Nahhas, R. Secondhand Smoke Exposure Is Associated with Autism Spectrum Disorder in US Males but Not in Females: Results from the National Survey on Children’s Health. Available online: https://www.environmentmed.org/article.asp?issn=2468-5690;year=2018;volume=3;issue=1;spage=8;epage=17;aulast=Khalil (accessed on 30 March 2021).
- Larsson, M.; Weiss, B.; Janson, S.; Sundell, J.; Bornehag, C.-G. Associations between Indoor Environmental Factors and Parental-Reported Autistic Spectrum Disorders in Children 6–8 Years of Age. Neurotoxicology 2009, 30, 822–831. [Google Scholar] [CrossRef] [Green Version]
- Abd Majid, M.S.; Johani, F.; Mohamed, N.E.; Diyana, S.; Isa, Z. Secondhand Smoke Exposure and Autism Spectrum Disorder: A Meta-Analysis. Sains Malays. 2020, 49, 1615–1625. [Google Scholar] [CrossRef]
- Argalasova, L.; Zitnanova, I.; Vondrova, D.; Dvorakova, M.; Laubertova, L.; Jurkovicova, J.; Stofko, J.; Weitzman, M.; Waczulikova, I.; Simko, M. Self-Reported Exposure to ETS (Environmental Tobacco Smoke), Urinary Cotinine, and Oxidative Stress Parameters in Pregnant Women-The Pilot Study. Int. J. Environ. Res. Public Health 2019, 16, 1656. [Google Scholar] [CrossRef] [Green Version]
- Protano, C.; Andreoli, R.; Manini, P.; Vitali, M. How Home-Smoking Habits Affect Children: A Cross-Sectional Study Using Urinary Cotinine Measurement in Italy. Int. J. Public Health 2012, 57, 885–892. [Google Scholar] [CrossRef]
| Characteristic | ASD Group (n = 84) | Non-ASD Group (n = 24) | p-Value ^ | |
|---|---|---|---|---|
| Age, mean (SD) | 5.4 (3.0) | 8.1 (3.6) | <0.001 *** | |
| Sex, N (%) | female | 13 (15.5) | 4 (16.7) | n.s. |
| male | 71 (84.5) | 20 (83.3) | n.s. | |
| Order of birth, N (%) | 1 | 40 (47.6) | 13 (54.2) | <0.05 * |
| 2 | 33 (39.3) | 7 (29.2) | n.s. | |
| 3 | 6 (7.1) | 3 (12.5) | n.s. | |
| 4 | 5 (6.0) | 1 (4.2) | n.s. | |
| Preterm birth, N (%) | no | 77 (91.7) | 22(91.7) | n.s. |
| yes | 7(8.3) | 2(8.3) | n.s. | |
| LBW, N (%) | no | 80 (95.2) | 22 (91.7) | n.s. |
| yes | 4 (4.8) | 2 (8.3) | n.s. | |
| Diagnostic Tool | Total N | Minimum | Maximum | Mean | Standard Deviation |
|---|---|---|---|---|---|
| ADIR_A | 84 | 0.1 | 0.9 | 0.5 | 0.2 |
| ADIR_B | 84 | 0.1 | 0.1 | 0.5 | 0.2 |
| ADIR_C | 84 | 0.0 | 0.8 | 0.3 | 0.2 |
| ADOS_SA | 84 | 3.0 | 10.0 | 7.3 | 1.6 |
| ADOS_RRB | 84 | 2.0 | 10.0 | 8.6 | 1.4 |
| ADOS_CSS | 84 | 4.0 | 10.0 | 8.0 | 1.6 |
| Main Domains | Non-ASD Group Mean (SD) | ASD Group Mean (SD) | p-Value ^ |
|---|---|---|---|
| Adaptive Behavior Composite | 78.6 (15.6) | 69.6 (10.8) | <0.05 * |
| Communication | 80.9 (16.6) | 65.8 (19.2) | <0.01 ** |
| Daily Living Skills | 85.1 (15.7) | 75.4 (11.9) | <0.01 ** |
| Socialization | 72.4 (18.4) | 66.4 (14.6) | n.s. |
| Motor skills | 90.2 (14.5) | 81.8 (10.8) | <0.01 ** |
| Subdomains | Non-ASD Group Mean (SD) | ASD Group Mean (SD) | p-Value |
| Receptive | 12.0 (3.4) | 8.0 (4.4) | <0.001 *** |
| Expressive | 12.2 (3.7) | 8.4 (4.6) | <0.001 *** |
| Written | 11.0 (3.3) | 10.4 (3.3) | n.s. |
| Personal | 12.9 (2.9) | 10.6 (2.5) | <0.001 *** |
| Domestic | 12.3 (3.2) | 11.4 (2.2) | n.s. |
| Community | 11.8 (3.3) | 9.9 (2.9) | <0.05 * |
| Interpersonal relationships | 9.2 (3.4) | 8.3 (2.8) | n.s. |
| Play and leisure time | 10.7 (3.9) | 9.0 (3.4) | <0.05 * |
| Coping skills | 9.8 (3.4) | 9.3 (2.4) | n.s. |
| Gross motor | 14.0 (2.9) | 13.1(2.0) | <0.05 * |
| Fine motor | 13.0 (2.8) | 11.0 (2.9) | <0.01 ** |
| Indicators of Subjective ETS Exposure | ASD | p-Value ^ | ||||
|---|---|---|---|---|---|---|
| No | Yes | |||||
| N | % | N | % | |||
| Subjective ETS exposure of the child | No | 12 | 85.7 | 23 | 57.5 | 0.05 * |
| Yes | 2 | 14.3 | 17 | 42.5 | ||
| Smoking of mother before pregnancy | No | 13 | 92.9 | 33 | 82.5 | n.s. |
| Yes | 1 | 7.1 | 7 | 17.5 | ||
| Smoking of father during mother’s pregnancy | No | 12 | 85.7 | 30 | 75.0 | n.s. |
| Yes | 2 | 14.3 | 10 | 25.0 | ||
| ETS exposure of mother during pregnancy | Not at all | 12 | 85.7 | 22 | 56.4 | n.s. |
| Once a week | 2 | 14.3 | 10 | 25.6 | ||
| More times a week/every day | 0 | 0.0 | 7 | 17.9 | ||
| Current smoking of the mother | No | 13 | 92.9 | 31 | 77.5 | n.s. |
| Yes | 1 | 7.1 | 9 | 22.5 | ||
| Current smoking of the father | No | 12 | 85.7 | 27 | 69.2 | n.s. |
| Yes | 2 | 14.3 | 12 | 30.8 | ||
| Smoking in the household | No | 13 | 92.9 | 25 | 62.5 | <0.05 * |
| Yes | 1 | 7.1 | 15 | 37.5 | ||
| Number of cigarettes smoked at home | Not at all | 13 | 92.9 | 24 | 60.0 | n.s. |
| ≤10 | 1 | 7.1 | 10 | 25.0 | ||
| >10 | 0 | 0.0 | 6 | 15.0 | ||
| Subjective vs. Objective ETS Exposure | |
|---|---|
| Measures of Agreement | Total |
| Kappa | 0.613 |
| Spearman correlation | 0.62 |
| Sensitivity | 70.59 |
| Specificity | 89.29 |
| Positive predictive value | 87.05 |
| Negative predictive value | 74.85 |
| Diagnostic accuracy | 82.22 |
| Indicators of Subjective ETS Exposure | Concentration of Urinary Cotinine | p-Value ^ | ||||
|---|---|---|---|---|---|---|
| Below the Median | Above the Median | |||||
| N | % | N | % | |||
| ETS exposure of the child | No | 25 | 89.3 | 5 | 29.4 | <0.001 *** |
| Yes | 3 | 10.7 | 12 | 70.6 | ||
| Quantification of ETS exposure of the child | Not at all | 18 | 64.3 | 5 | 29.4 | <0.01 ** |
| Once a week | 9 | 32.1 | 5 | 29.4 | ||
| More times a week/every day | 1 | 3.6 | 7 | 41.2 | ||
| Smoking of mother before pregnancy | No | 26 | 92.9 | 13 | 76.5 | n.s. |
| Yes | 2 | 7.1 | 4 | 23.5 | ||
| Smoking of father during mother’s pregnancy | No | 25 | 89.3 | 10 | 58.8 | <0.05 * |
| Yes | 3 | 10.7 | 7 | 41.2 | ||
| ETS exposure of mother during pregnancy | Not at all | 22 | 78.6 | 7 | 43.8 | <0.05 * |
| Once a week | 5 | 17.9 | 6 | 37.5 | ||
| More times a week/every day | 1 | 3.6 | 3 | 18.8 | ||
| Current smoking of the mother | No | 27 | 96.4 | 10 | 58.8 | <0.01 ** |
| Yes | 1 | 3.6 | 7 | 41.2 | ||
| Current smoking of the father | No | 25 | 89.3 | 8 | 50.0 | <0.01** |
| Yes | 3 | 10.7 | 8 | 50.0 | ||
| Number of cigarettes smoked at home | Not at all | 26 | 92.9 | 5 | 29.4 | <0.001 *** |
| ≤10 | 1 | 3.6 | 8 | 47.1 | ||
| >10 | 1 | 3.6 | 4 | 23.5 | ||
| Smoking in the household | No | 26 | 92.9 | 6 | 35.3 | <0.001 *** |
| Yes | 2 | 7.1 | 11 | 64.7 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szapuova, Z.J.; Argalasova, L.; Vondrova, D.; Jansakova, K.; Belica, I.; Kopcikova, M.; Babinska, K.; Ostatnikova, D. Association between Environmental Tobacco Smoke Exposure and Adaptive Behavior in Individuals with Autism Spectrum Disorder. Toxics 2022, 10, 189. https://doi.org/10.3390/toxics10040189
Szapuova ZJ, Argalasova L, Vondrova D, Jansakova K, Belica I, Kopcikova M, Babinska K, Ostatnikova D. Association between Environmental Tobacco Smoke Exposure and Adaptive Behavior in Individuals with Autism Spectrum Disorder. Toxics. 2022; 10(4):189. https://doi.org/10.3390/toxics10040189
Chicago/Turabian StyleSzapuova, Zofia Janik, Lubica Argalasova, Diana Vondrova, Katarina Jansakova, Ivan Belica, Maria Kopcikova, Katarina Babinska, and Daniela Ostatnikova. 2022. "Association between Environmental Tobacco Smoke Exposure and Adaptive Behavior in Individuals with Autism Spectrum Disorder" Toxics 10, no. 4: 189. https://doi.org/10.3390/toxics10040189
APA StyleSzapuova, Z. J., Argalasova, L., Vondrova, D., Jansakova, K., Belica, I., Kopcikova, M., Babinska, K., & Ostatnikova, D. (2022). Association between Environmental Tobacco Smoke Exposure and Adaptive Behavior in Individuals with Autism Spectrum Disorder. Toxics, 10(4), 189. https://doi.org/10.3390/toxics10040189








