Ecotoxicology: The Challenges for the 21st Century
Abstract
:1. Introduction: A Short History of Ecological Risk Assessment and Environmental Protection
2. The Development of Ecotoxicological Approaches
- the standard conditions of laboratory tests may not reproduce the variability of physical, chemical and biological parameters of aquatic and terrestrial ecosystems;
- the sensitivity of a few selected test species may not be representative of the distribution of sensitivity among the species of complex biological communities;
- the possible interactions among various stress factors of different origin that may affect the ecosystem are not accounted for;
- single species tests cannot account for the interactions and indirect effects that regulate the functioning of biological communities.
- How is the fraction of affected species distributed within trophic levels?
- Are all the levels of the food chain adequately protected?
- Is the biodiversity within the different trophic levels adequately protected?
- Are “keystone” species included within PAF?
- Does the loss of the species included within PAF substantially damage the structure and function of the ecosystem?
- Can the levels of primary and secondary productivity be adequately maintained?
- improving the methodological standardization for a better comparison and reproducibility of the results;
- improving the capability to extrapolate from specific test conditions to the variability of the characteristics of natural ecosystems;
- improving the transparency of the procedures to estimate a safe concentration from mesocosm results.
3. Assessing Exposure
4. Characterizing Risk
- Is the natural homeostatic capability of the ecosystem sufficient to counteract the effect of stress factors?
- How relevant are indirect ecological effects (competition, predation, etc.) for enhancing or reducing the effect of a stressor?
- How much time is required for a complete recovery of the community (resilience) if the stress factor is not continuous?
5. The Need for More Ecologically-Based Approaches
- What is the probability of the loss of a population?
- How much will the biodiversity change?
- Is the structure of the food-web protected?
- How much will productivity decrease?
- The complexity of biological communities: the characteristics of a community are not just the sum of the characteristics of individual populations; the structure and function of the community is regulated by emergent properties that are not easy to be described and predicted at lower hierarchical levels;
- The complexity of stress factors: toxic agents are only a component of the potential stress factors that may alter the behavior of natural populations and communities; the combination of multiple stress factors (anthropogenic and natural, such as temperature, oxygen depletion in water, water shortage in soil, etc.) needs to be taken into account for explaining environmental changes.
- the reproductive strategy and the potential growth rate ® that allows for, for “r” strategist populations, a more rapid re-colonization if the stress pressure ends or an easier genetic adaptation if the stress pressure continues;
- the presence of less sensitive resting larval stages;
- the dispersal capability.
6. The Activity of the European Commission
- to develop tools that go beyond the classical concept of PEC and that are capable of describing more realistic exposure patterns for single chemicals and mixtures;
- to increase the ecological realism of effects assessment by improving the use of higher tier and field or semi-field data and of methods that can predict or assess ecological processes;
- to define good practices for ecological modeling and agree on scenarios and ecological endpoints, so that models can be used to assess ecological risks for a wide range of species, regions and environmental conditions;
- to improve the development of site-specific assessments, taking into account the characteristics of exposed ecosystems;
- to account for the complexity of real ecosystems, considering the effects of biotic and abiotic interactions at community and ecosystem levels.
6.1. Challenges in Exposure Assessment
6.1.1. Better Use of Monitoring Data and Databases
6.1.2. Improvement of Modeling Approaches
- modeling the fate of polar and ionized chemicals, as well as nanomaterials;
- developing models capable of predicting time and space variability for a realistic exposure assessment;
- developing ecologically realistic scenarios accounting for the variability of environmental characteristics;
- developing models capable of describing the food web path of chemicals and also determining specific organism parameters to extend the applicability of bioaccumulation models;
- assessing the role of vegetation uptake in regulating the input to the food chain and the organic carbon cycle.
6.1.3. Sorption and Bioavailability
6.2. Challenges in Effect Assessment
6.2.1. Issues to Be Accounted for
6.2.1.1. Effect Assessment for Complex Exposure Patterns
6.2.1.2. Effect Assessment at Low Hierarchical Level
6.2.1.3. Ecosystem Vulnerability
6.2.1.4. Endocrine Disrupting Effects
6.2.1.5. Indirect Ecological Effects
6.2.1.6. Assessing Effects of Mixtures of Chemicals and Stressors
6.2.2. Tools to Be Better Developed or Improved
6.2.2.1. Higher Tier Effect Assessment
6.2.2.2. Trait-Based Risk Assessment
6.2.2.3. Ecological Modeling
6.3. Ecologically-Based Risk Characterization
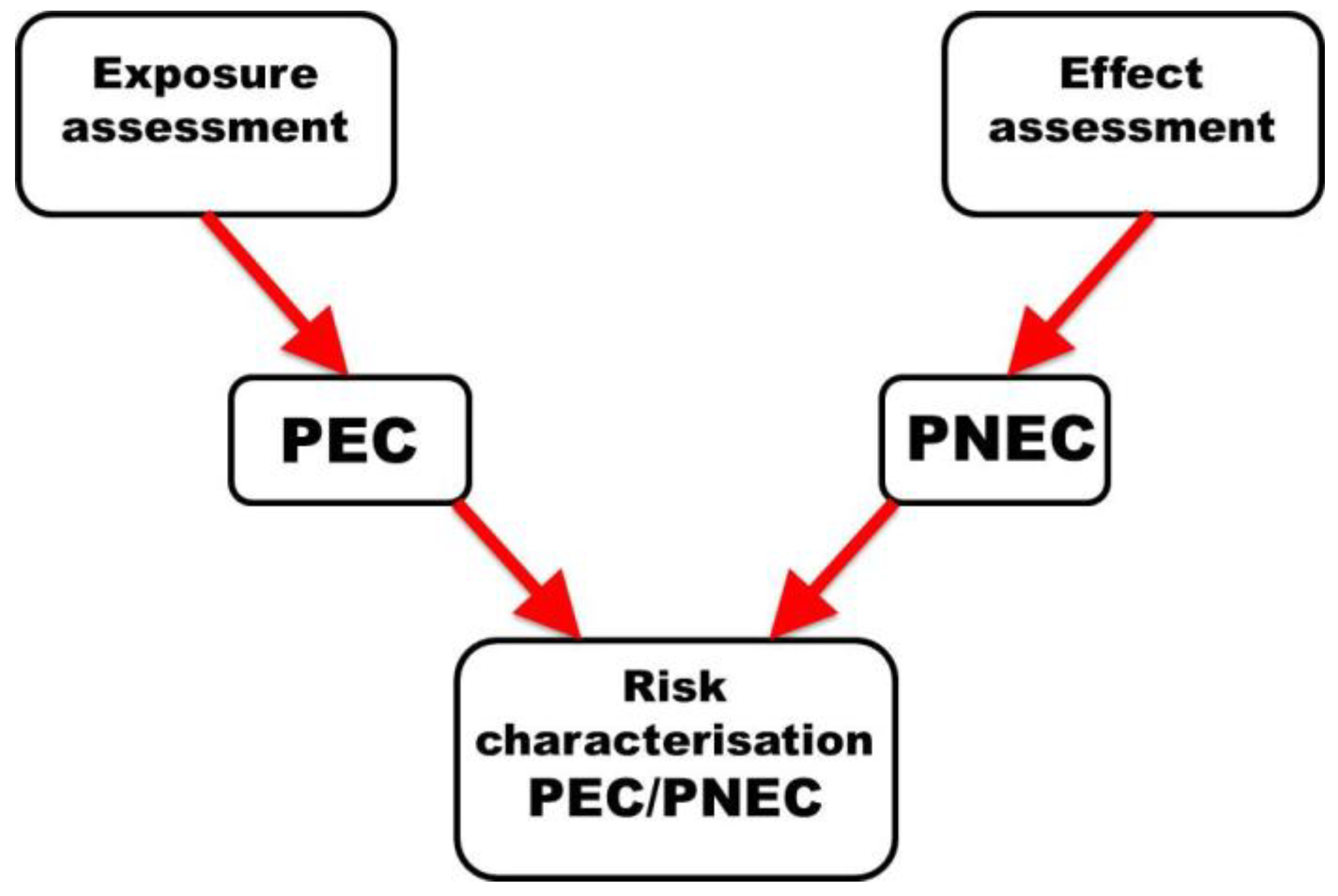
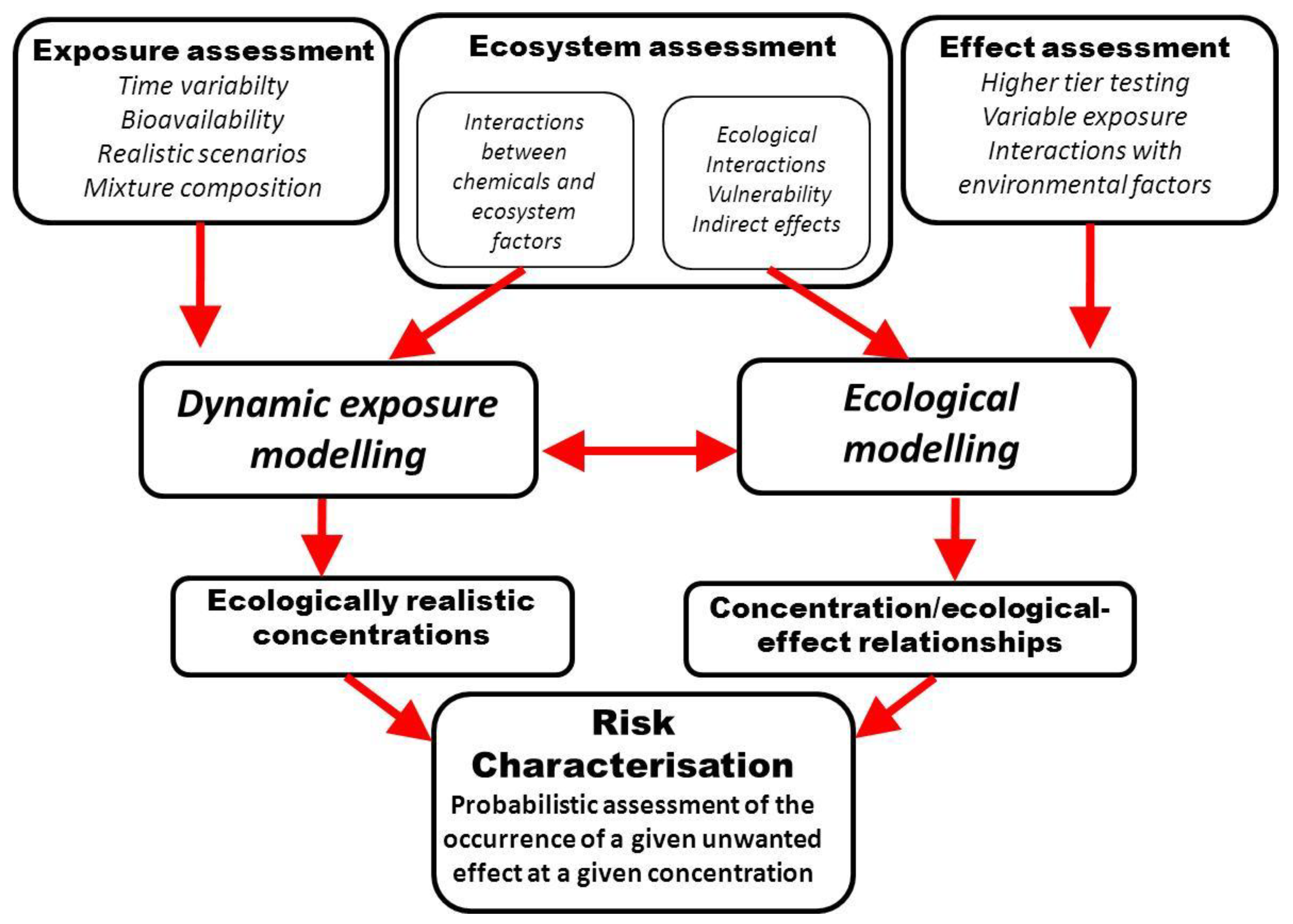
7. Conclusions: Toward a New Paradigm in ERA
Acknowledgments
Conflicts of Interest
References
- Carson, R. Silent Spring; Houghton Mifflin Co.: Boston, MA, USA, 1962; p. 279. [Google Scholar]
- The Environmental Agency UK. The Water Quality. Rotherham (UK). Available online: http://www.environment-agency.gov.uk/research/planning/34383.aspx (accessed on 12 November 2013).
- U.S. Environmental Protection Agency (US EPA). Quality Criteria for Water; Office of Water and Hazardous Materials: Washington, DC, USA, 1974. [Google Scholar]
- CSTE/EEC. EEC Water Quality Objectives for chemicals dangerous to aquatic environments. Rev. Environ. Contam. Toxicol. 1994, 137, 83–110. [Google Scholar] [CrossRef]
- EC (European Commission). Directive 2000/60/EC of the European Parliament and of the Council of 23 October 2000 establishing a framework for Community action in the field of water policy. Off. J. Eur. Communities 2000, L327, 1–72. [Google Scholar]
- Vighi, M.; Finizio, A.; Villa, S. The evolution of the Environmental Quality Concept: From the US EPA Red Book to the European Water Framework Directive. Environ. Sci. Pollut. Res. 2006, 13, 9–14. [Google Scholar]
- Van Straalen, N.M. Ecotoxicology becomes stress ecology. Environ. Sci. Technol. 2003, 37, 325–330. [Google Scholar]
- EC (European Commission). Technical Guidance Document (TGD) on Risk Assessment of Chemical Substances, 2nd ed; European Chemical Bureau, Joint Research Centre: Luxembourg, Luxembourg, 2003. [Google Scholar]
- Chapman, P.M.; Fairbrother, A.; Brown, D. A critical evaluation of safety (uncertainty) factors for ecological risk assessment. Environ. Toxicol. Chem. 1998, 17, 99–108. [Google Scholar] [CrossRef]
- Hampel, M.; González-Mazo, E.; Vale, C.; Blasco, J. Derivation of predicted no effect concentrations (PNEC) for marine environmental risk assessment: Application of different approaches to the model contaminant Linear AlkylbenzeneSulphonates (LAS) in a site-specific environment. Environ. Int. 2007, 33, 486–491. [Google Scholar] [CrossRef]
- Kooijman, S.A.L.M. A safety factor for LC50 values allowing for differences in sensitivity among species. Water Res. 1987, 21, 269–276. [Google Scholar] [CrossRef]
- Van Straalen, N.M.; Denneman, C.A.J. Ecotoxicological evaluation of soil quality criteria. Ecotoxicol. Environ. Saf. 1989, 17, 190–204. [Google Scholar] [CrossRef]
- Species Sensitivity Distribution in Ecotoxicology; Posthuma, L.; Suter, G.W.; Traas, T.P. (Eds.) Lewis Publishers: Boca Raton, FL, USA, 2002; p. 587.
- Sala, S.; Migliorati, S.; Monti, G.S.; Vighi, M. SSD-based rating system for the classification of pesticide risk on biodiversity. Ecotoxicology 2012, 21, 1050–1062. [Google Scholar] [CrossRef]
- Klepper, O.; van de Meent, D. Mapping the Potentially Affected Fraction (PAF) of Species as an Indicator of Generic Toxic Stress; National Institute of Public Health and the Environment: Bilthoven, The Netherlands, 1997; p. 93. [Google Scholar]
- Van Leeuwen, K. Ecotoxicological effect assessment in the Netherlands: recent developments. Environ. Manag. 1990, 14, 779–792. [Google Scholar] [CrossRef]
- EC (European Commission). Common Implementation Strategy for the Water Framework Directive2000/60/EC; (Technical Report—20103991); European Commission: Luxembourg, Luxembourg, 2010. [Google Scholar]
- ECHA. Chapter R.7c: Endpoint Specific Guidance Guidance for the Implementation of REACH. In Guidance on Information Requirements and Chemical Safety Assessment; European Chemicals Agency: Helsinki, Finland, 2012; p. 239. [Google Scholar]
- EFSA. DRAFT Guidance Document on tiered risk assessment for plant protection products for aquatic organisms in edge-of-field surface waters. EFSA J. 2013, 11, 3290. [Google Scholar]
- Bayne, B.L.; Livingstone, D.R.; Moore, M.N.; Widdows, J. A citochemical and biochemical index of stress in Mytilus edulis. Mar. Pollut. Bull. 1976, 7, 221–229. [Google Scholar]
- Peakall, D.B. Effects of toxaphene in epatic enzyme induction and circulating steroid levels in the rat. Environ. Health Perspect. 1976, 13, 117–125. [Google Scholar] [CrossRef]
- Payne, J.F. Mixed function oxidases in marine organisms in relation to petroleum hydrocarbons metabolism and detection. Mar. Pollut. Bull. 1977, 8, 112–121. [Google Scholar] [CrossRef]
- Van Aggelen, G.; Ankley, G.T.; Baldwin, W.S.; Bearden, D.W.; Benson, W.H.; Chipman, J.K.; Collette, T.W.; Craft, J.A.; Denslow, N.D.; Embry, M.R.; et al. Integrating omic technologies into aquatic ecological risk assessment and environmental monitoring: hurdles, achievements, and future outlook. Environ. Health Perspect. 2010, 118, 1–5. [Google Scholar]
- Martyniuk, C.J.; Griffitt, R.J.; Denslow, N.D. Omics in aquatic toxicology: Not just another microarray. Environ. Toxicol. Chem. 2011, 30, 263–264. [Google Scholar] [CrossRef]
- Ankley, G.T.; Daston, G.P.; Degitz, S.J.; Denslow, N.D.; Hoke, R.A.; Kennedy, S.W.; Miracle, A.L.; Perkins, E.J.; Snape, J.; Tillit, D.E.; et al. Toxicogenomics in Regulatory Ecotoxicology. Environ. Sci. Technol. 2006, 40, 4055–4065. [Google Scholar] [CrossRef]
- Mackay, D. Multimedia Environmental Models. The Fugacity Approach; Lewis Publisher: Boca Raton, FL, USA, 2001; p. 261. [Google Scholar]
- EC. FOCUS Groundwater Scenarios in the EU Review of Active Substances. European Commission: Luxembourg, Luxembourg, 2000. [Google Scholar]
- Kammenga, J.; Laskowski, R. Demography in Ecotoxicology; John Wiley & Sons: Chichester, UK, 2000; p. 297. [Google Scholar]
- Hommen, U.; Baveco, J.M.; Galic, J.N.; van den Brink, P.J. Potential application of ecological models in the European environmental risk assessment of chemicals. I: Review of protection goals in EU directives and regulations. Integr. Environ. Assess. Manag. 2010, 6, 325–337. [Google Scholar] [CrossRef]
- Kersting, K. Normalized ecosystem strain: A system parameter for the analysis of toxic stress in microecosystems. Ecol. Bull. 1984, 36, 150–153. [Google Scholar]
- Boivin, M.E.Y.; Breure, A.M.; Posthuma, L.; Rutgers, M. Determination of field effects of contaminants: Significance of pollution-induced community tolerance. Hum. Ecol. Risk Assess. 2002, 8, 1035–1055. [Google Scholar] [CrossRef]
- EC (European Commission). Regulation (EC) No 1907/2006 of the European Parliament and of the Council of 18 December 2006 concerning the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH). Off. J. Eur. Union 2006, L396, 1–849. [Google Scholar]
- EC (European Commission). Council Directive 91/414/EEC of 15 July 1991 concerning the placing of plant protection products on the market. Off. J. Eur. Communities 1991, L230, 1–32. [Google Scholar]
- EC (European Commission). Directive 98/8/EC of the European Parliament and of the Council of 16 February 1998 concerning the placing of biocidal products on the market. Off. J. Eur. Communities 1998, L123, 1–63. [Google Scholar]
- EC (European Commission). Directive 2008/56/EC of the European Parliament and of the Council of 17 June 2008 establishing a framework for Community action in the field of marine environmental policy (Marine Strategy Framework Directive). Off. J. Eur. Union 2008, L164, 19–40. [Google Scholar]
- EC (European Commission). Directive 2009/147/EC of the European Parliament and of the Council of 30 November 2009 on the conservation of wild birds. Off. J. Eur. Union 2010, L20, 7–25. [Google Scholar]
- EC (European Commission). Council Directive 92/43/EEC of 21 May 1992 on the conservation of natural habitats and of wild fauna and flora. Off. J. Eur. Communities 1992, L206, 7–50. [Google Scholar]
- EC (European Commission). SCHER, SCENIHR, SCCS Opinion on: Addressing the New Challenges for Risk Assessment.; European Commission: Brussels, Belgium, 2013; p. 154. [Google Scholar]
- Di Guardo, A.; Hermens, J. Challenges for exposure prediction in ecological risk assessment. Integr. Environ. Assess. Manag. 2013, 9, e4–e14. [Google Scholar] [CrossRef]
- Liess, M.; Brown, C.; Dohmen, P.; Duquesne, S.; Heimbach, F.; Kreuger, J.; Lagadic, L.; Reinert, W.; Maund, S.; Streloke, M.; et al. Effects of Pesticides in the Field—EPIF; SETAC Press: Brussels, Belgium, 2005; p. 136. [Google Scholar]
- Clements, W.H.; Newman, M.C. Community Ecotoxicology; John Wiley and Sons: Chichester, UK, 2002; p. 336. [Google Scholar]
- Reinert, K.H.; Giddings, J.M.; Judd, L. Effects analysis of time varying or repeated exposures in aquatic ecological risk assessment of agrochemicals. Environ. Toxicol. Chem. 2002, 21, 1977–1992. [Google Scholar] [CrossRef]
- Boxall, A.B.; Brown, C.D.; Barrett, K.L. Higher-tier laboratory methods for assessing the aquatic toxicity of pesticides. Pest Manag. Sci. 2002, 58, 637–648. [Google Scholar] [CrossRef]
- Ashauer, R.; Boxall, A.B.; Brown, C.D. Predicting effects on aquatic organisms from fluctuating or pulsed exposure to pesticides. Environ. Toxicol. Chem. 2006, 25, 1899–1912. [Google Scholar] [CrossRef]
- Brock, T.; Alix, A.; Brown, C.; Capri, E.; Gottesbueren, B.; Heimbach, F.; Lythgo, C.; Schulz, R.; Streloke, M. Linking Aquatic Exposure and Effects in the Registration Procedure of Plant Protection Products; SETAC Press: Pensacola, FL, USA, 2009; p. 440. [Google Scholar]
- Ashauer, R.; Brown, C.D. Highly time-variable exposure to chemicals—Toward an assessment strategy. Integr. Environ. Assess. Manag. 2013, 9, e27–e33. [Google Scholar] [CrossRef]
- De Lange, H.J.; Lahr, J.; van der Pol, J.J.C.; Wessels, Y.; Faber, J.H. Ecological vulnerability in wildlife. An expert judgment and multi-criteria analysis tool using ecological traits to assess relative impact of pollutants. Environ. Toxicol. Chem. 2009, 28, 2233–2240. [Google Scholar] [CrossRef]
- Liess, M.; von der Ohe, P.C. Analyzing effects of pesticides on invertebrate communities in streams. Environ. Toxicol. Chem. 2005, 24, 954–965. [Google Scholar] [CrossRef]
- Ippolito, A.; Sala, S.; Faber, J.H.; Vighi, M. Ecological vulnerability analysis: A river basin case study. Sci. Total Environ. 2010, 408, 3880–3890. [Google Scholar] [CrossRef]
- De Laender, F.; Janssen, C. Brief communication: The ecosystem perspective in ecotoxicology as a way forward for the ecological risk assessment of chemicals. Integr. Environ. Assess. Manag. 2013, 9, e34–e38. [Google Scholar] [CrossRef]
- Villa, S.; Migliorati, S.; Monti, G.S.; Vighi, M. Toxicity on the luminescent bacterium Vibrio fischeri (Beijerinck). II: Response to complex mixtures of heterogeneous chemicals at low levels of individual components. Ecotoxicol. Environ. Saf. 2012, 86, 93–100. [Google Scholar] [CrossRef]
- EC (European Commission). SCHER, SCCS, SCENIHR, Opinion on: Toxicity and Assessment of Chemical Mixtures. European Commission: Brussels, Belgium, 2012. [Google Scholar]
- Backhaus, T.; Faust, M. Predictive environmental risk assessment of chemical mixtures: A conceptual framework. Environ. Sci. Technol. 2012, 46, 2564–2573. [Google Scholar] [CrossRef]
- Porsbring, T.; Backhaus, T.; Johansson, P.; Kuylenstierna, M.; Blanck, H. Mixture toxicity from photosystem II inhibitors on microalgal community succession is predictable by concentration addition. Environ. Toxicol. Chem. 2010, 29, 2806–2813. [Google Scholar] [CrossRef]
- Backhaus, T.; Porsbring, T.; Arrhenius, A.; Brosche, S.; Johansson, P.; Blanck, H. Single substance and mixture toxicity of five pharmaceuticals and personal care products to marine periphyton communities. Environ. Toxicol. Chem. 2011, 30, 2030–2040. [Google Scholar] [CrossRef]
- Di Toro, D.M.; Allen, H.E.; Bergman, H.L.; Meyer, J.S.; Paquin, P.R.; Santore, R.C. A biotic ligand model of the acute toxicity of metals I. Technical basis. Environ. Toxicol. Chem. 2001, 20, 2383–2396. [Google Scholar] [CrossRef]
- De Schamphelaere, K.A.C.; Janssen, C.R. Development and field validation of a biotic ligand model predicting chronic copper toxicity to Daphnia magna. Environ. Toxicol. Chem. 2004, 23, 1365–1375. [Google Scholar] [CrossRef]
- Baird, D.J.; Baker, C.J.O.; Brua, R.; Hajibabaei, M.; McNicol, K.; Pascoe, T.J.; de Zwart, D. Toward a knowledge infrastructure for traits-based ecological risk assessment. Integr. Environ. Assess. Manag. 2011, 7, 209–215. [Google Scholar] [CrossRef]
- Van den Brink, P.J.; Baird, D.J.; Baveco, H.; Focks, A. The use of traits-based approaches and eco(toxico)logical models to advance the ecological risk assessment framework for chemicals. Integr. Environ. Assess. Manag. 2013, 9, e47–e57. [Google Scholar] [CrossRef]
- Ecological Models for Regulatory Risk Assessments of Pesticides: Developing a Strategy for the Future; Thorbek, P.; Forbes, V.; Heimbach, F.; Hommen, U.; Thulke, H.H.; van den Brink, P.J.; Wogram, J.; Grimm, V. (Eds.) Society of Environmental Toxicology and Chemistry (SETAC) and CRC Press: Pensacola/Boca Raton, FL, USA, 2010; p. 127.
- Grimm, V.; Martin, B.T. Mechanistic effect modeling for ecological risk assessment: Where to go from here. Integr. Environ. Assess. Manag. 2013, 9, e58–e63. [Google Scholar] [CrossRef]
- CREAM. Mechanistic Effect Models for Ecological Risk Assessment of Chemicals. 7th Framework Programme. Available online: http://cream-itn.eu/ (accessed on 12 November 2013).
- Forbes, V.E.; Calow, P. Developing predictive systems models to address complexity and relevance for ecological risk assessment. Integr. Environ. Assess. Manag. 2013, 9, e75–e80. [Google Scholar] [CrossRef]
- EC (European Commission). SCHER, SCENIHR, SCCS. Opinion on: Making Risk Assessment More Relevant for Risk Management. European Commission: Brussels, Belgium, 2013. [Google Scholar]
- Cairns, J., Jr. Putting the eco in ecotoxicology. Regul. Toxicol. Pharm. 1988, 8, 226–238. [Google Scholar] [CrossRef]
- EC. Guidance Document on Estimating Persistence and Degradation Kinetics from Environmental Fate Studies on Pesticides in EU Registration. European Commission: Luxembourg, Luxembourg, 2006. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Vighi, M.; Villa, S. Ecotoxicology: The Challenges for the 21st Century. Toxics 2013, 1, 18-35. https://doi.org/10.3390/toxics1010018
Vighi M, Villa S. Ecotoxicology: The Challenges for the 21st Century. Toxics. 2013; 1(1):18-35. https://doi.org/10.3390/toxics1010018
Chicago/Turabian StyleVighi, Marco, and Sara Villa. 2013. "Ecotoxicology: The Challenges for the 21st Century" Toxics 1, no. 1: 18-35. https://doi.org/10.3390/toxics1010018
APA StyleVighi, M., & Villa, S. (2013). Ecotoxicology: The Challenges for the 21st Century. Toxics, 1(1), 18-35. https://doi.org/10.3390/toxics1010018





