1. Introduction
Logistics management in the clinical trials industry is a very challenging undertaking. The reasons for this are varied: there are multiple stakeholders, the processes are complex, many different software applications are involved, the tasks are labour intensive, and the quality standards are stringent. Current business practices are inefficient due to the lack of structure in the flow of information for coordination and collaboration, the presence of disparate software applications, and difficulties in implementing automation technologies. On the other hand, the clinical trials industry is vital for society since its output results in new treatment opportunities for an increasingly larger number of afflictions. At the same time, from an economic point of view, it is a high-value industry employing a highly skilled workforce. The motivation for this paper is to address the inefficiencies in current logistics management practices in the industry by reviewing the state-of-the-art and developing a framework for logistics management in the industry that uses contemporary tools and technologies.
A clinical trial is usually for a new drug that is developed by a pharmaceutical company or other sponsors such as a university research centre. Due to regulatory requirements, testing for the drug needs to be at arms-length and is performed by an independent laboratory. Patients are enrolled at sites (for example, hospitals) for participating in the drug trial. A Contract Research Organization (CRO) is usually involved in managing the logistics of the clinical trial. This paper takes the perspective of a CRO. The CRO works with the pharmaceutical company on developing a testing plan that involves multiple sites with multiple patients. The testing plan includes administration of the drug to the patients and collecting samples of analytes such as blood, urine, tumors, etc. The CRO, in consultation with the sponsor develops manuals for the testing plan, labels, and kits. A kit includes items such as collection tubes, gel packs, and air waybills for subsequent shipment of samples to laboratories. The CRO may also provide additional logistical services such as tracking shipments from the collection sites to the testing laboratories.
In such a setting, logistics management involves complex coordination of tasks. The CRO is in touch with the sponsor both during the initial design phase of the trial as well as during implementation. Sometimes, amendments are made to the collection plans such as the addition of new sites, new patients, new assessment types, and the frequency of testing or dosage. The CRO is also in constant touch with the sites as well as the laboratories. Based on the treatment plan, there are preset collection time points which must be managed. For example, the supplies (unexpired labels and kits) required for each enrolled patient at a site should be available at the right time (the collection tubes may have expiry dates). The CRO must ensure that the samples are sent to the laboratories within a window of integrity and track such shipments, resolving discrepancies in a timely manner.
The clinical trials industry has stringent quality requirements. Collection and shipment must happen exactly as specified in the collection plan. Patient and analyte labels must be error free. Kits are expensive and have limited shelf life. Therefore, the right number of kits need to be sent to each site at the right time. Shipments in this industry are global, requiring greater attention to shipment lead times. The shortage of kits may result in samples not being collected and analyzed, while overproduction or expiration of kits increases costs. In addition, the collected samples are valuable, expensive to collect, and non-renewable. They must be maintained carefully to be analyzed by the assay labs. Therefore, a large cross-functional team needs to work together to have the right supplies at the sites and to maintain the stability of samples from sites to the assay labs.
This paper makes three contributions. The literature on logistics management in clinical trials is limited; in fact, most of the literature is at a very conceptual level or focuses on distinct aspects of clinical trials such as patient recruitment. The second contribution of this paper is to propose an integrative software framework for logistics management in the clinical trials context. The third contribution of this paper is the design of a resource management and project planning tool, which facilitates and optimizes operational planning.
The rest of the paper is organized as follows:
Section 2 presents a literature review of the theories and methods used in logistics management in this and other industries.
Section 3 details the complex operations involved in clinical trials and analyzes the challenges in current practice.
Section 4 details a conceptual software framework solution for clinical trials logistics.
Section 5 proposes a resource and project planning tool for clinical trial logistics management. Finally,
Section 6 summarizes this work and
Section 7 lists areas for future research.
2. Literature Review
We classified the literature relating to logistics management for clinical trials into five categories: (1) clinical trials industry requirements and challenges, (2) service industry and servitization concepts, (3) project management, (4) contemporary software tools and architectures, and (5) visualization. These five aspects need to be understood together to understand the challenges addressed in this paper.
2.1. Clinical Trial Industry Background
The US Food and Drug Administration (FDA) approved 45 novel drugs in 2015; 16 of these drugs were First-in-Class, meaning they used an innovative method to treat disease. In total, 21 out of 45 drugs were for rare or orphan diseases that affect 200,000 people or less in the United States [
1]. On average 28 novel drugs per year were approved from 2006 to 2014. According to [
2], redrawing samples from a patient causes dissatisfaction and may lead to a withdrawal of the patient from the study. Replacing a patient is also challenging due to narrower patient population sizes that recent studies are focusing on and can cost up to USD 25,000. According to [
3], the number of developmental drug compounds increased from 2040 compounds in 2001 to 3240 in 2011. This number reached 7000 in 2016 and the average time to develop a drug is 10 to 15 years; less than 12% of drugs obtain approval after the clinical trial stage; the average cost of drug development including the failure cost was
$ 2.6 billion from 2000 to 2010 [
4].
The median of the total number of procedures in each clinical trial was 105.9 in 2000–2003 and increased to 166.6 in 2008–2011 [
5]. The median total work burden for each protocol was 55 in 2000–2003 and increased to 180 in 2004–2007. In total, 54% of the sites and 78% of the subjects involved in clinical trials were located outside of US in the year 2008 [
6]. There are four phases in a clinical trial (refer to
Table 1) [
7].
2.2. Challenges and Requirements in Managing Clinical Trials
According to a study by [
8] on 68 new drugs developed by 10 pharmaceutical companies, the total capitalized cost is about 802 million dollars to develop a new drug. The authors state that most of the failures in clinical trials are due to management problems rather than issues with study design. Therefore, project management approaches should be used to manage each trial’s resources efficiently. Communication and collaboration amongst team members, streamlined and flexible processes with computerized data capturing systems are key elements in successful trial management, particularly as pharmaceutical companies are outsourcing their clinical trials [
9]. In commercial trials when the time to market is important, time has a higher priority than budget. In [
10], an approach based on the concept of Product-Based Planning is presented as an alternative to fixed-price contracts for the outsourcing for clinical logistics tasks. While our paper does not explicitly consider contract pricing, it can be extended to do so based on the ideas presented. A survey on clinical research outsourcing is discussed in [
11].
According to [
12], based on a survey of 28 professionals involved in outsourcing, the most common outsourced services are clinical monitoring (96% of respondents), data management (92% of respondents), and project management (81% of respondents) (as cited in [
11], pp. 364–365). This study further states that although outsourcing of clinical trial activities has been increasing, the level of satisfaction is low.
According to [
12], as the number of treatments, procedures and foreign sites increases, the complexity of the Clinical Trial Material (CTM) supply chain rises. The main challenges of the global supply chain are different customs rules in each country, the cost of transportation (especially for cold-chain CTM), and longer delivery times. They suggest that having real-time and accurate information on deliveries and inventory at each site improves this system.
Ref. [
13] evaluated outsourcing companies in the pharmaceutical industry based on the theory of dynamic capability: the ability to use resources to respond to market changes in a timely manner. The finding shows that pharmaceutical companies tend to outsource to companies that have higher dynamic capability rather than higher capacity [
14].
The gap in the literature that outsourcing brings up is the challenge of complexity and coordination when the logistics are no longer conducted in-house.
2.3. Service Industry and Servitization
The service industry is an industry that provides non-tangible value, as opposed to tangible products [
15].
In clinical trials logistics management, there is a need for collaborative service management and the need to focus on Service Dominant (S-D) logic as opposed to traditional Goods Dominant (G-D) logic [
16,
17]. Modular architecture is also a good example of S-D logic [
18]. However, a significant barrier to collaboration is the lack of trust amongst partners in a supply chain [
19].
Many manufacturers have moved from only goods production to supplying supplemental services beside their products. This phenomenon is called servitization in much of the literature. Examples of the servitization model can be seen in companies such as Xerox, which provides document management through their photocopy machines, automotive companies that provide warranty and maintenance services to their customers, VLT (Video Lottery Terminals) producers that install and maintain the VLT machines in casinos, and Rolls Royce provides fixed cost per hour maintenance services on its aircraft engines to its airline clients. Frameworks for the transition to servitization have been discussed in the literature. Examples are: Ref. [
20] with the use of big data, Ref. [
21] for a specific framework in healthcare, and [
22] for a generic servitization framework for manufacturers. Ref. [
23] conclude that service extension and service skill improvements benefit business performance significantly compared to product design modifications.
While these are not examples from the clinical logistics industry, these concepts are important for clinical logistics because the concept of servitization for clinical trials would apply to label production, kit assembly, and shipping box assemblies for samples requiring refrigeration. Although these items could be thought of as products, they have to be delivered to sites or labs in a timely manner and need customization. This is a gap in the literature that was identified.
2.4. Project Management
Project management in the service industries including software development should have flexible goals and methods. Examples of new project management techniques, such as agile and dynamic project management, are used to design open-source software such as the Linux operating system [
16] and modular architectures [
17].
Refs. [
9,
24] provide key guidelines for project management in clinical trials such as having a project manager, a project plan, communication and collaboration, and computerized data capture systems. Ref. [
10] suggests that a deliverable-based project management, such as the PRINCE2 method, is a good technique to manage clinical trials. Ref. [
25] identifies the requirements for an integrated clinical trial supply chain solution. Ref. [
26], identifies five aspects of a good project management practice, which are scientific management, humanistic management, risk management, quality management, change management, and project cost management. However, none of the articles explain the detailed framework of their concepts.
Ref. [
27] indicated that industries who incorporated a project management model earlier, such as petrochemical and defense industries, perform better than the younger industries such as pharmaceutical Research and Development (R&D) and financial services.
Surveys have shown that project managers in the service sector excel in cost and procurement planning [
28]. However, they perform poorly in quality planning and management, which has a high impact on project success.
Ref. [
29] stated that the distinct success factors for R&D projects are “following plan, interpretation, minimal startup problems, meet technical goals, and negotiation”, and the distinct success factors for software development projects are “shared vision, risk management, project management, and adaptability”.
Ref. [
30] surveyed existing algorithms and techniques for solving project scheduling problems in the construction industry. Ref. [
31] prepared a taxonomy for planning and scheduling for construction project management and concluded that the concepts of the Gantt chart, Critical Path Method (CPM), Project Evaluation and Review Technique (PERT), Line of Balance (LOB), Critical Chain Project Management (CCPM), and Last Planner System (LPS) are widely prevalent.
Ref. [
32] summarized and categorized existing Resource-Constrained Project Scheduling (RCPS) concepts. The popular models were found to have multiple modes, generalized time lags, and net present value-based objectives. Specific examples from different industries have been discussed in this article, such as [
33], which applies RCPS in the early stages of drug discovery in the pharmaceutical research industry.
Ref. [
34] surveyed solution models for project scheduling under uncertainty. Their survey includes “reactive scheduling, stochastic project scheduling, fuzzy project scheduling, robust (proactive) scheduling and sensitivity analysis” (p. 289).
We discovered a gap in the literature in which there are no publications on project and resource planning for clinical trial logistics.
2.5. Contemporary Software Tools
Semantic web was founded by the World Wide Web Consortium (W3C). “The Semantic Web provides a common framework that allows data to be shared and reused across application, enterprise, and community boundaries” [
35]. The World Wide Web is the web of documents, while semantic web is the web of data. Semantic web integrates all the data from different sources using RDF (Resource Descriptive Framework) that can be easily read by machines. Semantic web can be seen as a larger and more robust tool than relational databases [
36]. Ref. [
37] identifies five key elements to improve supply chain operations in aerospace, of which one is a unified system of record that can be achieved by semantic web technology.
Ref. [
38] designed a high-level semantic web system for patient and specimen tracking in clinical trials. Ref. [
39] used semantic web to recruit patients for the purpose of clinical trials. Ref. [
40] designed semantic software to unify the health record information of patients to improve the health care system.
Ref. [
41] propose a semantic web service-oriented model for project management. Furthermore, Ref. [
42] uses a more detailed approach for integrating and sharing information in construction projects from heterogeneous information formats such as text, relational and XML. Other than [
38], no other paper specifically applied semantic web to the logistics management of clinical trials. Semantic web can be applied as a robust query system for the entire duration of a clinical trial such as Study Setup, Study Management, Sample Management, and Tracking.
Ref. [
43] defines a software agent as an entity that acts autonomously on behalf of others. Applications of software agents can be seen in marketing, assistance systems, search engines, and chatter bots. The agents work best in an open system, such as the internet, which has the characteristics of “continuous availability, extensibility, modularity, arms-length relationships, concurrency, asynchronous work, decentralized control, and inconsistent information” [
44]. Ref. [
45] describes the main characteristics of software agents as autonomy, social ability, reactivity, and proactiveness. Ref. [
43] presents a survey of the programming languages and platforms for creating software agents. Potential platforms for building the software agents are ZEUS, JADE, JAFMAS, RETSINA, and Agent Building Shell.
Ref. [
46] concludes that project management guidelines, such as the Project Management Body of Knowledge (PMBOK), fail most of the time due to poor communication, lack of monitoring and non-performance measurement. They use agent technology to improve implementation of PMBOK and reduce failures.
Ref. [
47] designed a multi-agent platform for order promising and production planning in a multi-enterprise supply chain network. This model brings an integrated, proactive, and efficient approach to supply chain management.
Ref. [
48] designed a framework that includes the Radio Frequency Identification (RFID) technology into a multi-agent system to maximize the efficiency of logistics management.
Ref. [
49] designed the framework for a software agent system that integrates disparate business processes in a hospital for the purpose of clinical trials. This paper does not cover the non-hospital processes of clinical trials.
There remains a gap in the integration of software agents, supply chain and logistics management, hospital processes for clinical trials, and project management.
Ref. [
50] designed an electronic upload and reporting system for image transfer from clinical sites to Imaging Core Laboratories (ICLs). In this system, images are uploaded into a secure web portal and are automatically quality checked. Other articles focus more on the improvement of eCRF (electronic Case Report Form), which is used to collect all of the information about a patient, i.e., the visits, medication dose, adverse events, etc.
The literature is lacking in coverage of Sample Inventory Logs (SILs) or Sample Requisition Forms (SRFs), which are used to record the samples collected at the sites. A similar approach to [
50] can be applied in this area.
Business Process Management Systems (BPMS) are information systems that orchestrate and control business processes [
51]. Ref. [
52] defines BPMS as a system with many flexible processes that consume real-time data from different sources. The role of BPMS in organizations is to streamline administrative tasks and interoperate with other applications.
Ref. [
52] used the semantic web approach to integrate heterogonous information to support BPMS. Ref. [
53] designed an architecture to integrate environmental information such as unforeseen events captured by the Internet of Things (IoT) with BPMS. A case study performed by [
54] in a justice organization in Brazil showed that BPMS significantly improved project management.
Ref. [
51] identified the common problems in BPMS are collaboration, interoperability, security, and dynamicity. Ref. [
55] demonstrated a longitudinal study on the effects of workflow management systems, which are part of BPMS, on the performance of organizations. According to this study, only half the companies successfully implement the workflow management systems in at least one of their processes. They found that the workflow management system supported only 28% of the overall processes.
Ref. [
56] states that current BPMS disregard collaborative work and also assume constant availability of resources per period, which is not linked to the calendar. They propose a scheduling approach that considers resource dependencies and conflicts.
To summarize, BPMS are effective in streamlining execution and monitoring of business activities. However, their compatibility and integration with other applications should be improved. Also, BPMS are execution oriented and lack the planning component. The gap identified is that a comprehensive framework for clinical trial logistics is absent in the literature.
2.6. Visualization and Collaboration Tools
According to [
31], the Gantt chart is a suitable tool for visualization of activity schedules. In this paper, the authors studied the effect of 3D visualization of project tasks and resources compared to Gantt chart and spreadsheet. This study showed a 40% increase in accuracy and a 39% decrease in monitoring time. Ref. [
57] created a framework to visualize the interactions and cooperation among human and non-human resources in construction projects. Ref. [
58] discusses the research on geo-visualization techniques. It is shown the literature is moving from representation to analysis, which is closer to the real meaning of visualization. Ref. [
59] proposed a visual tracking system for compound management delivery in laboratories, but it can be applied to any workflow system to reduce variability.
Ref. [
60] performed an exploratory study on a group of users that used Upwave, an online collaboration tool. The motivation to use this tool was found to engage with other team members remotely, access all information about the project in one place, learn a new tool, and intrinsic interest in sharing information with teams. It was found that making checklists, templates, and other artifacts are best practices that can be supported by collaboration tools.
Ref. [
61] built a knowledge conversion model based on Information and Communication Technology (ICT) for virtual teams. They mention that collaboration in a virtual team requires motivation, supportive leadership, and suitable technology.
Ref. [
62] designed an integrated collaborative tool for a precast supply chain. An architecture was developed that is based on Context-Aware Cloud Computing Building Information Modelling (CAABIM).
In summary, the techniques reviewed, especially [
59,
60] can be applied in clinical trials logistics management. The Gantt chart or network chart is useful for delivering Study Setup deliverables, but do not work for Sample Management where the CRO has no control over time and is only responsible for tracking samples. Ref. [
61] can be a future solution that can be built after a framework is created for clinical trial logistics management. This again represents a gap or opportunity for the themes covered in this paper. In our overall assessment of the literature, we identified the following gaps/opportunities in clinical trial logistics:
There is no reported conceptual framework based on contemporary software tools and architectures, all interacting with specialized software applications for accounting, inventory, and label or kit design software.
There is no reported project planning tool or methodology for resource management. In particular, this paper will look at different managerial strategies to plan and execute logistics tasks.
3. Project Management Challenges in Clinical Trials
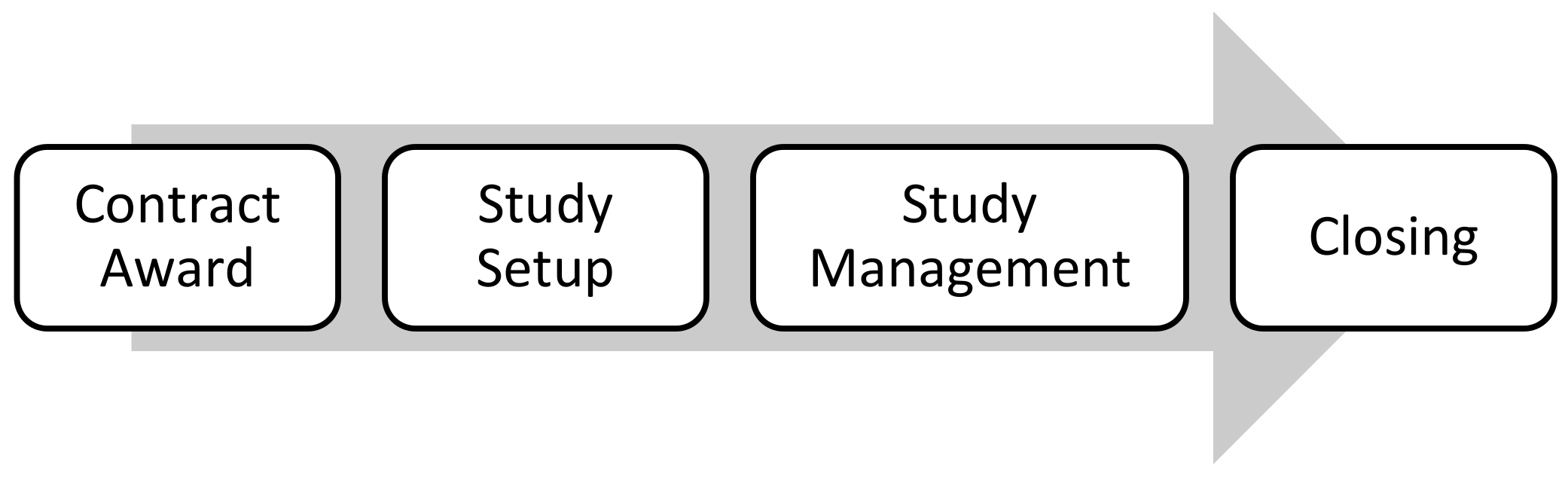
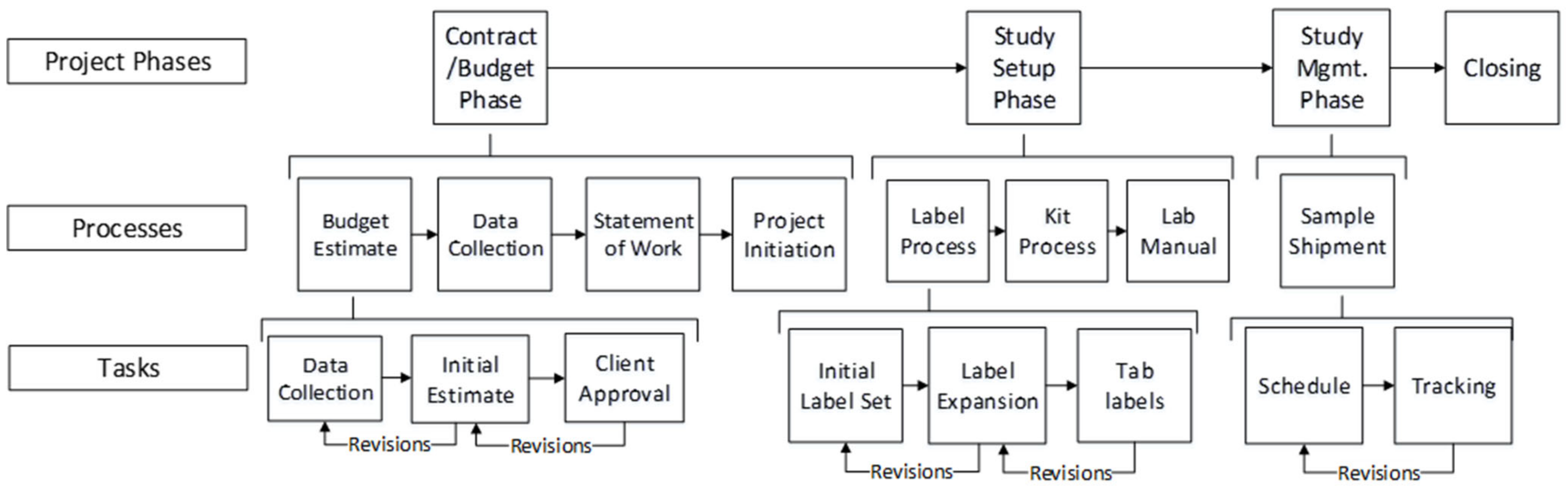
A clinical trial project is called a protocol. It is a unique endeavor and involves many components and resources to be executed. Project management in a clinical trial involves complex coordination activities to ensure the protocol is executed correctly and within the timeframe and budget. A clinical trial project is divided into four steps: contract award, Study Setup, Study Management, and closing (
Figure 1).
In the contract award step, the Sponsors of clinical trials outsource the management of these trials to CROs (Contract Research Organizations). In the Study Setup phase, the CRO designs the required supplies and coordinates the activities with the site and lab staff before starting the clinical trial. Study Management is a sequence of actions that starts when a patient is entered into the clinical trial.
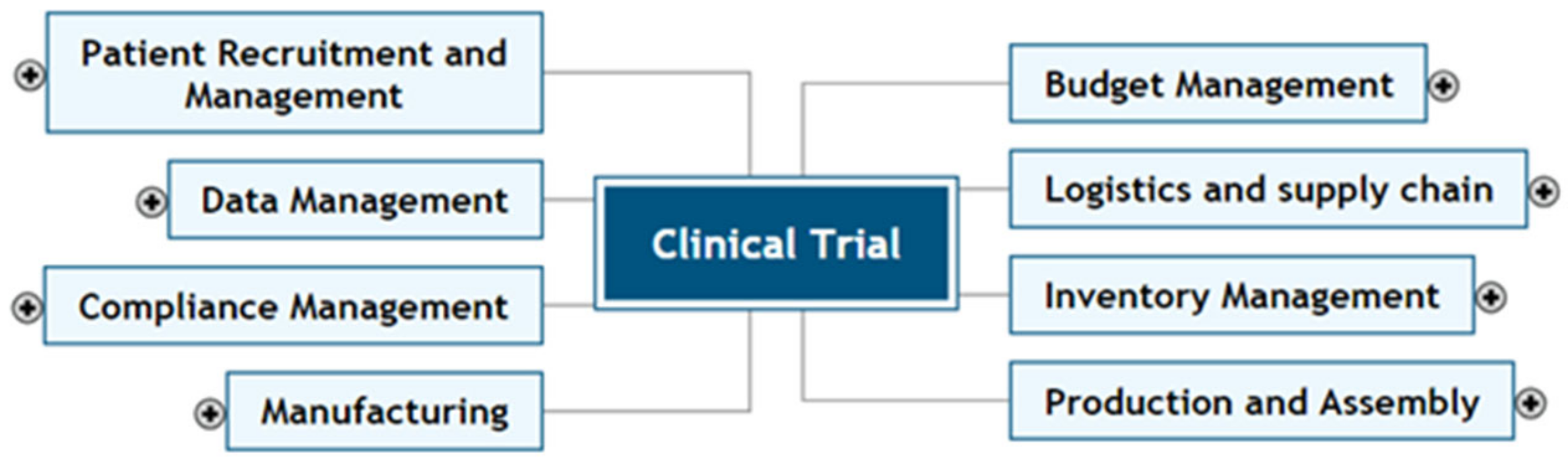
Figure 2 shows the main components of Study Management.
Participant recruitment and management include recruiting eligible patients and managing their health and safety.
Budget management includes managing the allocated money for each activity of the protocol and amendments.
Logistics and supply chain include coordination of shipping supplies and treatments from the manufacturer to the sites and collected samples from the sites to the assay laboratories.
Inventory management includes maintaining the appropriate level of unexpired supplies and treatments at each site.
Production and Assembly includes designing and producing the desired supplies (sample collection kits) and treatments for clinical trials.
Data management includes tracking the logistical data such as data about patients, supplies, samples, sample shipments, and clinical data such as laboratory results.
Compliance management includes meeting regulatory compliance practices while conducting clinical trials. Examples of regulators are the Institutional Review Board (IRB), and Ethical Review Board (ERB) or Research Ethics Board (REB) regulations.
In the closing stage, the CRO ensures that all the requirements of the clinical trial have been properly addressed as planned in the contract.
3.1. Protocol Amendments
According to a study by [
63], about 60% of protocols had one or more amendments, and each amendment had an average of 6.9 changes. The most changes were identified to be changes to eligibility criteria of the patients, number and types of safety assessments, and revisions to general information such as protocol title and information about staff. An amendment to a protocol cost
$453,932 on average. The major costs are related to the increase in investigational sites and contract change orders with CROs and other third-party providers. It must be noted that the sample size for clinical trials can be computed statistically [
64].
According to [
65], every trial has nearly three to five protocol amendments after the trial commencement. Protocol amendments especially change in eligibility criteria and the target patient’s population to statistically test the differential effects of treatment on the old and new population.
3.2. Demand for Patients
It is possible to estimate the required number of patients at every phase or stage of clinical trials to find statistically meaningful results. For example, Ref. [
65] presents some sample calculation modelling for clinical trials. However, it is difficult to recruit the desired level of patients with specific criteria and therefore makes the demand uncertain. Integrated information systems [
40] and marketing strategies [
66] can improve the recruiting process.
3.3. Data Management
Figure 3 shows the flow of logistical data in a clinical trial. The main data elements are:
Enrollment log that includes the list of enrolled patients in a site.
Supplies or kits that are produced by the CRO for the sites.
Shipments schedule to ship collected samples from the site to the central laboratory for analysis.
Sample shipments that the site ships to the central laboratory.
Sample Inventory Log (SIL) is the manifest of samples being shipped in a shipment. Electronic SIL (eSIL) is the electronic format of the SIL.
Notifications that are in the form of emails that are sent to inform the laboratory of incoming samples shipments.
Figure 3.
Existing Practice for Study and Sample Management.
Figure 3.
Existing Practice for Study and Sample Management.
The main issue in this area is that SILs are hand-written because of confidentiality, accountability, traceability, and security reasons. Going fully electronic is not secure if the data is manipulated or stolen. The CRO is responsible for checking the quality of the data collected at the site and transcribing the data into digital format for the sponsors and labs. When the number of samples increases, paper SILs become voluminous, and this task becomes time-consuming.
3.4. Diverse Software Applications and Data Sources
Lack of integration among computer applications or sources of data causes duplicate entries of data, inefficiency, and errors in the system. For example, in the Study Setup phase, where products such as labels and manuals are designed, the CRO transcribes the data from the source documents, such as protocol or email, into the label or manual design software.
Sample Management becomes challenging when the number of studies, sites, patients, sample types and time points increase. Some of the risks in clinical trials are losing collected blood samples, losing sample integrity due to storage at an incorrect temperature, and losing a sample collection schedule due to not having supplies at the site. The CRO reacts to reduce the impacts of this risk; however, proactive planning needs to replace reaction. For example, if a sample is being shipped from a site to the wrong laboratory, the CRO quickly contacts the courier company to re-route the package. Proactive planning would be to ensure that the waybills used by the site have the correct lab address pre-printed on them.
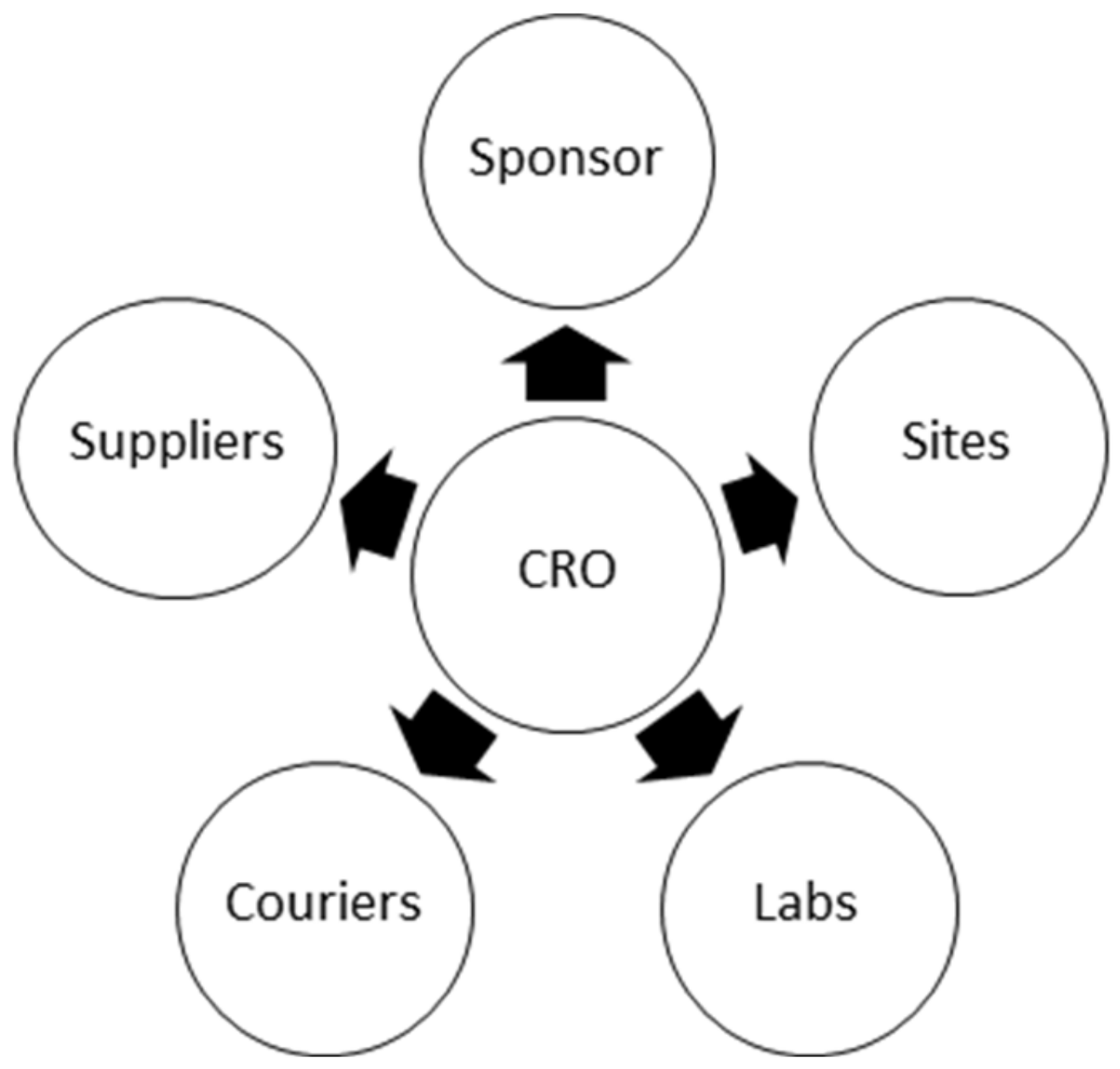
Clinical trials require cooperation of many participant organizations such as the sponsor, sites, labs, and CROs (
Figure 4). Therefore, an optimum way of collaboration is essential to meet the objectives.
Almost all the collaboration among the CROs, assay labs, sponsor and sites are conducted via email. As the number of organizations increases, the number of emails increases as well. Processing all workflows via email is inefficient and error prone.
4. Development of a Streamlined Integrative Conceptual Framework
A conceptual framework is now proposed to improve project management in clinical trials. The high-level objectives of the framework have been retrieved from [
38], which are electronic traceability, manufacturing visibility, compliance with quality management standards, integration of workflows for greater visibility, and a unified system of record.
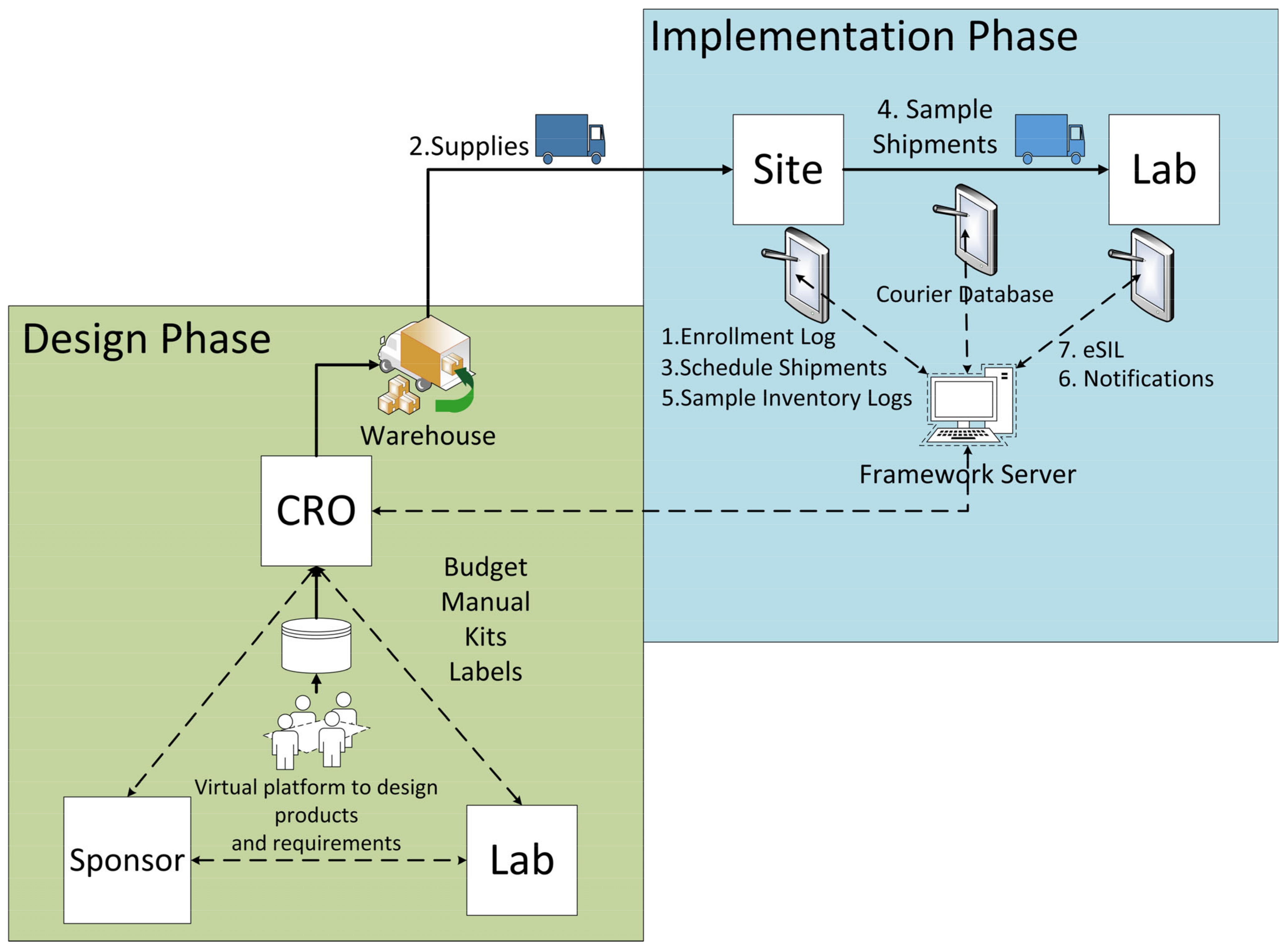
Figure 5 shows the high-level architecture of the framework. The lower left box shows the design phase of a clinical trial, and the upper right box shows the implementation phase of a clinical trial.
In the design phase, the CRO works with the sponsor and the analytical laboratories to design the testing plan, laboratory manuals, products, and services.
The implementation phase starts when a new site is opened, and patients are enrolled (1). Then, the required product is shipped to the sites (2). After patient enrollment sample collection and shipment are scheduled (3), then sample inventory logs are prepared (5), delivery notifications are sent to the labs (6), eSIL is created (7). In this framework, a central database system tracks data in real time and each stakeholder (site, lab, CRO, courier company) has a designated web portal to view relevant information and to input necessary data or communicate with other team members.
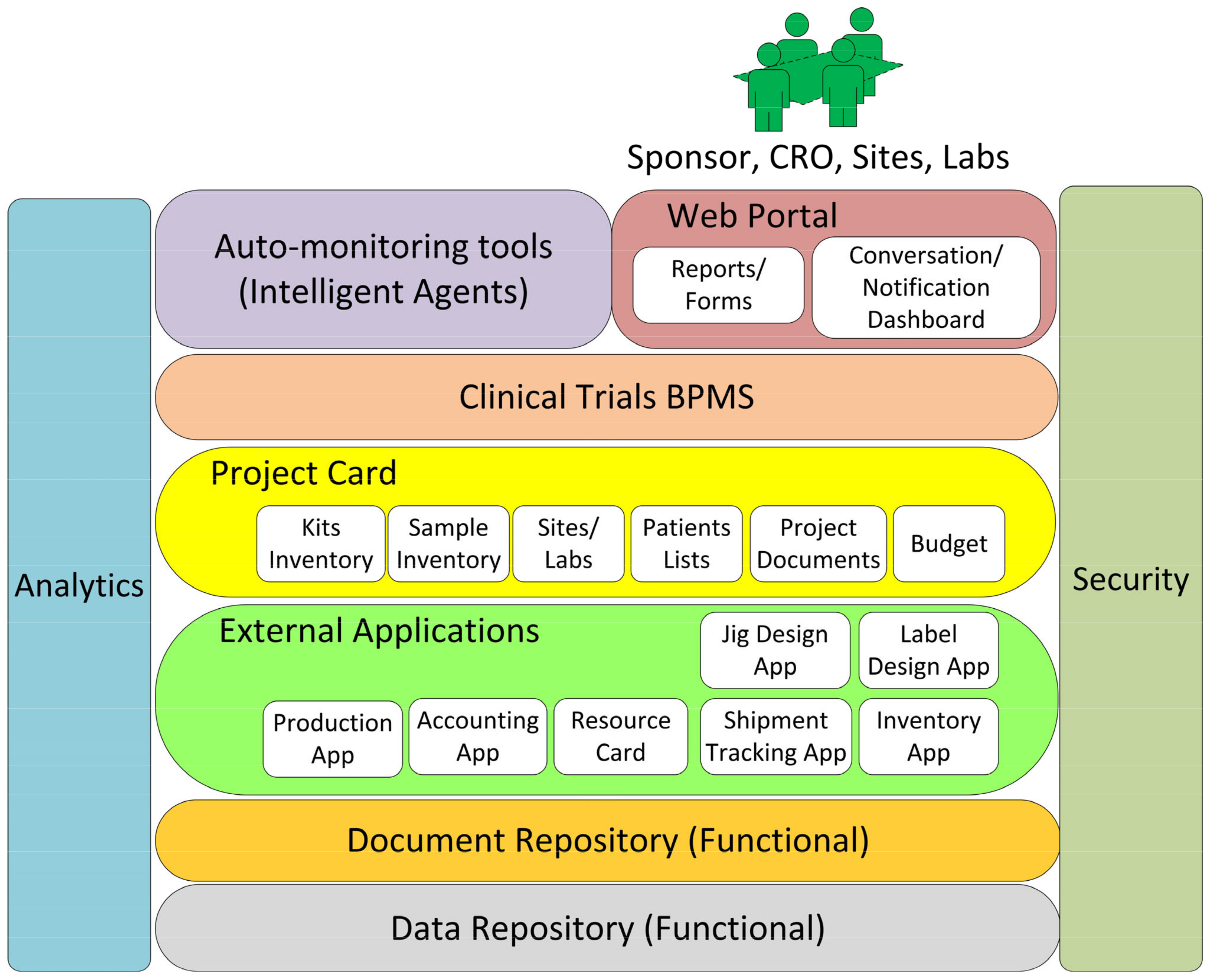
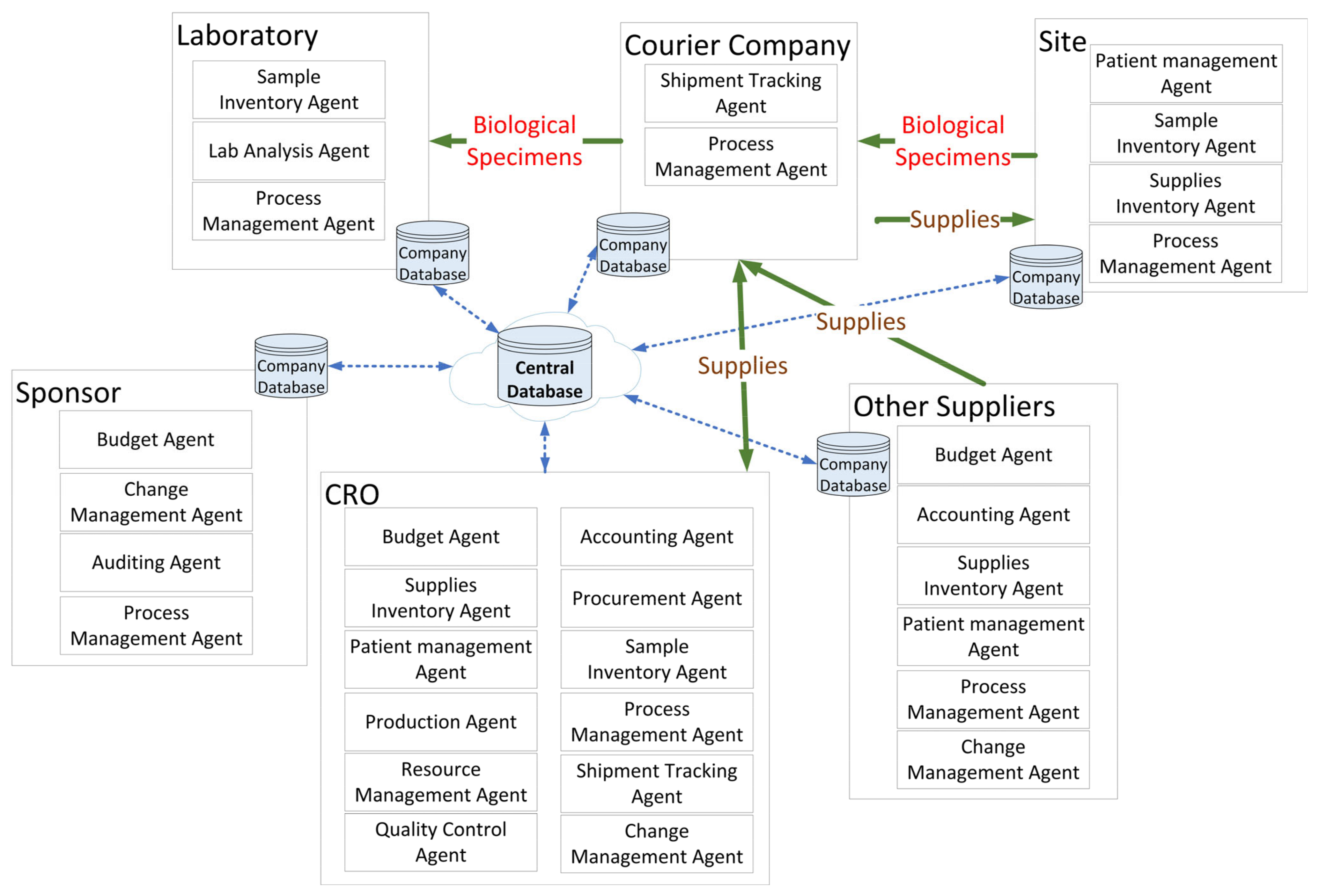
Figure 6 shows the detailed architecture (core) of the framework. This framework from top to bottom consists of:
Web portal and its interactions with human users.
Auto-monitoring tools implemented using intelligent software agents.
Clinical trials BPMS to execute and orchestrate the tasks amongst resources and stakeholders.
Project card to host data regarding all project activities in one place.
External applications (for specialized tasks).
Document repository.
Data repository.
Analytics module to capture key performance indicators through the layers.
Security.
4.1. Web Portal and Its Interactions with Human Users
A web portal is incorporated into this framework to improve communication and collaboration among the stakeholders in clinical trials logistics management. The web portal will have forms to input the data, reports to show the data in real time to the teams, a file system to store and share the documents, and a dashboard for notifications and conversations with other team members. A conversation and notification dashboard is needed amongst team members for better collaborations. For example, the dashboard will send reminders to the site, e.g., when to collect and ship samples to the labs. This framework also integrates collaboration tools, such as email systems, to make communication more organized and efficient.
Figure 6.
High-Level Architecture of the Framework.
Figure 6.
High-Level Architecture of the Framework.
4.2. Intelligent Agents
Figure 7 shows the proposed architecture of the multi-intelligent agent system for clinical trial logistics management. The agent breakdown is shown in
Table 2,
Table 3,
Table 4,
Table 5,
Table 6 and
Table 7. The objective of designing intelligent software agents for this industry is automated data collection, data monitoring, and decision making.
4.3. Clinical Trials BPMS Requirements
The aim of implementing BPMS in clinical trial logistics management is to integrate processes and automate tasks.
Figure 8 shows the proposed workflow diagram for clinical trials BPMS, which are divided into three areas: Study Setup, Study Management, and Sample Management. The detailed requirement of BPMS for each area will be explained in the following.
Study Setup involves setting up the budget, designing laboratory manual, kits, and services specific to a protocol. The main requirements for BPMS in this phase are:
A platform for documenting the budget requirements such as type and number of supplies, sample shipment frequencies, and contacts.
A platform to design labels, kits, manuals and share the drafts with sponsors for comments and approvals. Templates for designing labels, jigs, and manuals should be populated automatically by the available data in the budget.
A platform to train site staff about the testing plans.
Change the management functionality to update the affected data.
Study Management involves the production of the kits that have been designed in the Study Setup. The main requirements for BPMS in this phase are:
Patient’s enrollment and kit inventory databases should be shared between the sites and CRO.
The CRO should be notified by the system to produce and ship the required kits.
If the kits need to be modified, the sponsor should identify in the system in which time point data should be changed and an automatic notification should be sent to the stakeholders.
There should be a collaborative platform for changing the design of the kits between CRO and sponsor due to frequent changes in the protocols. Conversation systems can be incorporated in the platform.
Shipments from CRO to sites can be tracked by the system; shipment notification should be sent to the sites automatically.
The approved site orders should be sent to the production department to plan inventory and production.
Sites can track their requests on the platform.
The Sample Management process involves managing the collected samples from patients at the sites. The samples should be stored at a certain temperature and transported to the labs to be analyzed. The main requirements for BPMS in this phase are:
SILs should be filled out electronically at sites either manually or using barcodes with data integrity checks on forms. A similar approach to [
51] can be applied here.
A conversation platform is available as a hyperlink on the form to contact other team members for questions.
The user can view the list of samples collected for each patient and schedule sample shipment either manually or automatically. The site staff will be notified when they should ship the samples.
This system can be linked to the courier database to track the shipments.
Automatic shipment notification along with the list of samples in the shipment and tracking number should be sent to the assay laboratory.
Labs need to confirm receipt of samples on the platform. Follow-ups will be sent in case of no response.
The platform should be able to handle sample discrepancies by contacting responsible parties.
4.4. Project Card
The project card concept is central to the framework. A project card can be incorporated into the system that can act as a data and document management system for a project. The following data can be queried from a project card:
The progress in the entire project or a specific area.
The resources that have been involved in any tasks.
The link to documents that have been collected during the project.
The history of tasks and changes.
The list of supplies used in the clinical trials.
The list of contacts.
The list of patients and their collected samples.
The link to conversations and notifications.
4.5. External Applications
External applications can be any applications that need to be connected to the BPMS and Project Card to produce products or services. Examples of external applications are the ERP system, label design system, inventory software, and accounting software. Any data or document that is produced for a specific project by an application will be stored in the project card. Conversely, any data that external applications require for a project will come from the project card.
We propose an external application called Resource Card that will work with the Project Card and BPMS. A resource card will serve as a bridge between resource management and project management. It will log employee’s projects, training, interest and skills. Resource cards can be useful for planning and managing human resources in the long and short term.
4.6. Document Repository
The document repository is a place to store any company’s business-related documents such as accounting and operational documents.
4.7. Data Repository
The data repository is a place to store all the company’s business-related data such as accounting and operational data.
4.8. Analytics (As Shown Spanning the Entire Architecture in Figure 7)
Analytics can be used to analyze the collected data from the framework and find meaningful data patterns or answer questions that can improve the company’s performance. Examples of the questions or data that can be investigated using analytics are as follows:
Which sites are making errors that require training? Which suppliers and partner companies are making errors and reasons?
What are the common errors?
What are the bottlenecks?
Identify the common trends for supplies and kits in all clinical trials. SKU wise, size wise, design wise, quantity wise.
Identify the historical demand for supplies and services and forecast future demand.
4.9. Security (As Shown Spanning the Entire Architecture in Figure 7)
The role of security is to control access to all components of the main framework. For example, operations managers and employees need access to the web portal, but they do not need access to the structure of the data repository or BPMS. The IT manager will be responsible for managing the security and controlling the access of different users or stakeholders to different components of the framework.
4.10. Implementation
The framework was implemented for an industry partner company specialized in clinical trial logistics management. The components that have been implemented are as follows:
4.10.1. Database System (Project Card)
A relational database system was built in Microsoft SQL Server 2017. The number of tables has been used to hold important data regarding protocol information, specimens’ shipments, assessment types, cost models, etc. Important and confidential data have been encrypted. Database users have limited access to data depending on their role in the company. The database has been backed up continuously to be able to recover any damage to the data.
4.10.2. Application Design in BPM Software
The manual business processes have been transformed into an electronic workflow system using Agile Point BPM software NX v7.0. This includes the design of logic and user forms and reports. The applications have been connected to the back-end database to retrieve and write data. This automation has increased the efficiency of the specimen shipment logistics by about 50%, quality control process by 40%, and kit production process by 45%.
4.10.3. Integration of BPM Software with External Software
Applications have been connected to external applications, such as the inventory management software’s database, to communicate data.
4.10.4. Intelligent Agents
An intelligent agent has been created to automatically track the status of shipments and generate required notifications. Another agent has been designed to read the emails from other stakeholders, such as analytical laboratories, and extract the required information for each protocol and shipment. An agent has been created to track the kit inventory consumption and generate notifications as soon as the inventory level hits a critical threshold. Other agents are yet to be implemented.
4.10.5. Reports and Dashboards for Every Protocol
Customized reports have been created for the users in every department to be able to see their respective data in real-time. Every protocol has a dashboard that shows high-level information about all components of the protocol such as kits inventory, collected samples inventory, discrepancy lists, the overall status of the protocol, etc.
5. Resource and Project Planning in the Clinical Trial Logistics Management
In the designed framework for clinical trials logistics management, the Project Card holds data about a clinical trial project, the Resource Card holds information about resources available in the organization to perform the tasks, and BPMS distributes the project tasks to the resources.
5.1. Description of the Project Tasks and Resources
The resources that are needed for a clinical trial logistics project are broken down to the Contract Team (CT), Project Manager (PM), Operation Associate (OA), and Quality Control (QC). PMs attend meetings with external study team members, find out the requirements, and coordinate deliverables and changes with internal team members (Oas). Oas performs simpler and more structured tasks, while PMs and CTs perform more complex tasks with less or sometimes no structure. QC checks the quality of the work through the entire processes of a project.
Table 8 shows the resources that are involved in each phase of a clinical trial logistics management project.
In the contract and phase, the CT meets with the sponsor to identify requirements and the Statement of Work (SOW) for the protocol. This phase typically takes 1–2 weeks to complete. In Study Setup, all the required materials, such as labels, collection manual, and kits, are designed and approved by the sponsor. Study Setup takes a month typically and starts right after the contract phase. Study Setup triggers most processes such as label design and production, manuals design and print, site initiation visit presentation, Jig design, and kit design and production.
In Study Management, the resources should be available when a new patient is recruited onto a site or when each patient progresses from initial cycles to the others. Examples of the tasks in this phase are replenishing sample collection supplies, handling protocol amendments, sample shipment tracking, and Sample Management.
Replenishing sample collection supplies is based on the patients enrolled in each site. For the duration of the study, each site continues to accept new patients to reach or maintain their desired number of patients. New patients require kits for initial time points, and therefore, the site will ask CROs for kits. In the meantime, existing patients may choose to continue through the study or withdraw from the study. Each period requires its own supplies or collection kits, which need to be prepared and sent by the CROs. Sites will send re-order forms to ask the CRO to replenish the supplies or kits required for new patients or existing patients progressing through collection time points. Some sponsors might ask for bulk supplies instead of kitted supplies, for example, bulk labels or collection tubes. The re-order forms are used to control or monitor the number of kits used by each site. The due date to replenish re-order forms varies from 2 to 5 business days.
The patient enrolment log helps to order the right number of items listed on a re-order form. The arrival rate of re-order forms depends on patient recruitment rate, and number and duration of cycles in a study. The higher recruitment rate leads to ordering screening kits more frequently. The longer the study duration, the more frequent orders will be because of the expiry date of some kits.
Amendments or change orders are very common in a clinical trial. The sponsor might decide to change the collection manual, labels, and kits anytime during the study. These changes should be reflected in the budget and any other subsequent supplies and materials (kits, labels, manual) that will be affected by the change. New patients enrolled in the study after any amendments must be treated according to the new amendment and receive amended collection materials.
Sample shipments should be scheduled to ship the collected samples from the site to the laboratory. The frequency of the shipments varies from the day of collection to monthly and quarterly. The frequency of the shipment depends on the window of integrity of the sample, and the capacity of the site and lab. The content of each shipment can be determined from the patient’s enrolment tracker and sample collection tracker. Special shipping kits suitable for maintaining the desired temperature are used to transfer samples from the site to the laboratory. Shipments should be tracked, and labs should be notified of upcoming shipments to prepare the analysis. Any delays should be resolved and documented.
Sample Management is the function of keeping track of the collected samples and ensuring they have been shipped from the site and received by the laboratory. Information about collected and shipped samples is documented in a Sample Inventory Log (SIL) for the sponsor and laboratories. Any discrepancy should be resolved and documented.
At the closing stage, the project manager ensures all the requirements for the clinical trial have been addressed. The discrepancies should be resolved, outstanding fees be paid, and all documents should be archived for future references.
5.2. Task Flow
The outputs of clinical trial logistics management are products and services such as labels, kits, inventory management, and Sample Management. The workflow consists of three levels, which are project phases, processes for each project phase, and tasks for each process.
Figure 9 shows the main project phases of clinical trial logistics management, and some examples of processes and the tasks within them are budget creation, label creation, and Sample Management.
Table 9 shows an example of how due dates will be assigned to each process within each project phase. The due date for each process is the required finish time of the last task of a process. For example, for the Budget phase, if we want to initiate the Budget Estimate Process, we need to assign a due date for Client Approval task, which is the last task of the Budget Estimate Process. Many of the processes can be re-initiated for a project because of many reasons, such as revisions, amendments, setting up a new site, and recruiting new patients. To reinitiate, a new due date should be assigned for that process of any phase.
5.3. Conceptual Solution for Planning the Tasks and Resources
This solution consists of two parts. The first part is a planning and scheduling tool that plans the tasks and resources. The second part is a task execution tool, BPMS, that executes the planned tasks.
The planning and scheduling tool will have input data that needs to be entered by the management and output data that the system will generate. The main processes of each phase of a project should be identified in a Gantt chart inside the project card. Note that the processes are listed in the Gantt chart, while the tasks of that process are listed in the BPMS. The due date for each process should be identified by the Project Manager of that project. The duration for each task that is in the BPMS can be estimated by a time study of the current practice or expert opinion. The tasks dependencies are also known and are reflected in the business processes that are located on the BPMS; for example, “confirm lab addresses” should be performed before “order preprinted airwaybill”.
The planning tool will assign human resources, such as the PMs, QC, and Oas, to the project according to the resource card. The resource card will have the list of resources with their availability, qualifications, and type of tasks and projects that the resources prefer to work with. The planning tool shall also identify the start and finish date for each task automatically according to the due date, the duration of each task, and task dependencies (processors and successors), and resource availability.
The management choices and objectives can vary from one organization to the other and from time to time depending on the priorities of the company. The planning tool should be flexible enough that allows the management to combine different choices or objectives at any time and for a given planning horizon. Company wide objectives could be to finish all of the processes of all projects by their due dates, optimize the company’s capacity, or determine the appropriate level of resources for a given planning horizon to meet the company’s objectives.
The tool can accommodate different task assignment objectives such as:
Assign tasks of any project (protocol) to people in a group according to their qualifications. For example, in the QC group, some people can only use QC labels, while some are qualified to QC budgets as well as labels.
Include familiarity factors for resources.
Assign similar projects to one project manager for more efficiency.
Assign urgent or high-priority tasks to more experienced people to process them faster.
Assign tasks according to the resource’s workload.
Transfer resources from one resource group to the other for cross training and when work demand moves from one department to the other.
Project tasks stay with the same person or group who started the first task of that job within that process to increase the familiarity factor.
Assign simpler tasks to new employees.
To design a planning tool that will address the management choices, two main planning models are demonstrated. The first model is a task scheduling LP, and the second model is a resource planning LP. The task scheduling LP will be used when the operations manager has a fixed number of resources and wants to know how to schedule tasks accordingly. However, the resource planning LP will help the manager not only schedule the tasks, but to find out the required number of resources needed for every resource group in order to finish all tasks without any tardiness.
5.4. Model 1—Task Scheduling LP
Schedule sub-tasks by building a scheduling LP (Basic Scenario). Three scenarios will be tested within this model to see the effect of each scenario on the objective function. The scenarios are fixed shared resources among all projects, fixed dedicated resources for each project, and fixed dedicated resources in project work groups.
5.4.1. Scenario I—Fixed Shared Resources among All Projects
In a shared or non-dedicated resource model, resources are shared amongst projects (one resource should work on multiple projects). This model is feasible and economical when each project does not demand dedicated resources and resources have time to process tasks for multiple parallel projects. In most projects, there is slack between tasks and processes, such that the resources dedicated to them become idle during those situations. Therefore, it is economical to share the resources amongst projects. In this model, shared resources will be fixed to see their effects on the objective function. The fixed resources represent the resources on hand.
5.4.2. Scenario II—Fixed Dedicated Resources for Each Project
In the dedicated model, the resource will focus on one or a set of similar projects and cannot be shared with all other projects. This makes him or her work more efficient and accurate and, as a result, will benefit clinical trial logistics more. Clinical trials are performed under stringent quality standards. Therefore, quality is as important as efficiency. However, the drawback of this model is that the dedicated resources are not always utilized, because of the slacks involved in one or more projects that they are assigned to. Another drawback is that in the dedicated model, resources are not flexible enough (require longer learning curve) to be moved from task to the other. In this scenario, the number of dedicated resources for each project shall be determined by the management.
5.4.3. Scenario III—Fixed Dedicated Resources in Project Work Groups
In this model, resources will work within work groups. Each work group will have its own dedicated resources. Similar projects or projects from the same sponsor will be assigned to a work group. This model takes the characteristics of both shared and dedicated resource models since it dedicates a group of resources to several projects and resources can be shared amongst those projects if needed. In this scenario, the number of resources in each work group will be fixed by the management.
5.5. Model 2—Resource Planning LP
This model determines the optimal number of resources to meet the objective function within the planning horizon.
5.5.1. Scenario IV—Variable Shared Resources
Same as Scenario I, but with a variable number of resources.
5.5.2. Scenario V—Variable Dedicated Resources
Same as Scenario II, but with a variable number of resources.
5.5.3. Scenario VI—Variable Dedicated Work Groups
Same as Scenario III, but with a variable number of resources.
5.6. Background Research on RCMPSP
Some of the research articles in the area of project planning and scheduling have already been mentioned in the Literature Review section [
31,
32,
33,
35]. In addition to the above articles, Ref. [
67] formulated a mixed integer linear programming model to the RCPSP (Resource Constrained Project Scheduling Problem) using the multi-mode approach, which means that a task can be performed in different modes and, in each mode, the number of resources and the duration for each task is different. The model in [
67] has been a central reference in many other articles. Ref. [
68] extended the formulation in [
67] to solve the RCMPSP (Resource Constrained Multi-Project Scheduling Problem), which facilitates the scheduling of multi projects. In addition, they dedicate resources to each project while [
67] shares resources.
Our model uses both [
67,
68], with extensions to use processes in addition to projects, having a process–precedence relationship in addition to task–precedence relationship, using only one resource type for each mode, having deadlines for processes in addition to projects, and running several scenarios with fixed and shared number of resources. Our approach to solving the RCMPSP for the clinical trial logistics management is shown in
Table 10. In this table, there are two main linear programming models that are used for task planning and resource planning.
Three scenarios (non-dedicated resources, dedicated resources, resources in work groups) are incorporated in each model to be used by the managers in the clinical trials industry. In the task planning model, we fix the number of resources in each scenario. However, for the resource planning model, we let the number of resources be variables and let the LP model determine the optimal level of resources. is a variable that represents the total number of shared resources from each resource group to complete all projects. is a variable that represents the total number of dedicated resources to each project (v) to complete all projects. is used in Scenarios I, III, IV, and VI, because resources are shared amongst the projects in one or more work groups. is used in Scenario II and IV, because resources are dedicated to each project.
In Scenarios III and IV (resources in work groups), a multiple number of work groups can be used, while no work group (or in other words, all resource groups are part of one work group) is used in non-dedicated and dedicated resource configurations models. In all resource configuration, each mode represents one or more resource groups (g). In other words, a mode represents a unique combination of resources and the rate at which that task will be processed in that mode. In all scenarios, one or multiple projects and processes can be used.
The formulation for the task planning model in Scenarios I and II and the task planning model in Scenario III will be shown in
Section 5.7 and
Section 5.8 respectively. The formulation for the resource planning model in Scenarios IV and V and the resource planning model in Scenario VI will be shown in
Section 5.9 and
Section 5.10, respectively.
5.7. Task Planning Model (Scenarios I and II)
5.7.1. Sets and Indices
5.7.2. Parameters
or
5.7.3. Decision Variables
5.7.4. Mathematical Model
The Objective Function (1) minimizes the total weighted tardiness over all projects and processes. Constraint Set (2) ensures that all tasks are scheduled only once for all projects and processes. Constraint Set (3) enforces the precedence relationships amongst tasks within each process and project. Constraint Set (4) enforces the precedence relationship amongst processes of each project. Constraint Set (5) determines the maximum number of renewable shared resources per period across all projects during the planning horizon. Constraint Set (6) determines the maximum number of renewable dedicated resources per period to each project during the planning horizon. Constraint 5 is used in Scenario I and Constraint 6 is used in Scenario II. Constraint Sets (7) and (8) calculate the weighted tardiness for projects and processes respectively. Constraint Sets (9) and (10) specify the feasible range for decision variables.
5.8. Task Planning (Scenario III)
5.8.1. Sets and Indices
Assumption: k belongs to one g only. In other words, one person belongs only to one group and cannot be in more than one group.
5.8.2. Parameters
5.8.3. Decision Variables
5.8.4. Mathematical Model
The Objective Function (11) minimizes the total weighted tardiness over all projects and processes. Constraint Set (12) ensures that all tasks are scheduled only once for all projects and processes. Constraint Set (13) enforces the precedence relationships amongst tasks within each process and project. Constraint Set (14) enforces the precedence relationship amongst processes of each project. Constraint Set (15) determines the maximum number of renewable resources per period during the planning horizon. Constraint Sets (16) and (17) calculate the weighted tardiness for projects and processes, respectively. Constraint Set (18) ensures each project is only in one work group. Constraint Set (19) ensures that each task of a project is assigned a mode that belongs to the work group of that project. Constraint Sets (20)–(22) specify the feasible range for decision variables.
5.9. Resource Planning Model (Scenario IV and V)
5.9.1. Sets and Indices
5.9.2. Parameters
5.9.3. Decision Variables
Or
5.9.4. Mathematical Model
The Objective Function (23) minimizes the total weighted tardiness over all projects and processes. Constraint Set (24) ensures that all tasks are scheduled only once for all projects and processes. Constraint Set (25) enforces the precedence relationships amongst tasks within each process and project. Constraint Set (26) enforces the precedence relationship amongst processes of each project. Constraint Set (27) determines the maximum number of renewable shared resources per period across all projects during the planning horizon. Constraint Set (28) determines the maximum number of renewable dedicated resources per period to each project during the planning horizon. Constraint Sets (29) and (30) calculate the weighted tardiness for projects and processes, respectively. Constraint Sets (31)–(33) specify the feasible range for decision variables.
5.10. Resource Planning Model (Scenario VI)
5.10.1. Sets and Indices
Assumption: k belongs to one g only. In other words, one person belongs only to one group and cannot be in more than one group.
5.10.2. Parameters
5.10.3. Decision Variables
5.10.4. Mathematical Model
The Objective Function (33) minimizes the total weighted tardiness over all projects and processes. Constraint Set (34) ensures that all tasks are scheduled only once for all projects and processes. Constraint Set (35) enforces the precedence relationships amongst tasks within each process and project. Constraint Set (36) enforces the precedence relationship amongst processes of each project. Constraint Set (37) determines the maximum number of renewable resources per period during the planning horizon. Constraint Sets (38) and (39) calculate the weighted tardiness for projects and processes, respectively. Constraint Set (40) ensures each project is only in one work group. Constraint Set (41) ensures that each task of a project is assigned a mode that belongs to the work group of that project. Constraint Sets (42)–(44) specify the feasible range for decision variables.
5.11. A Case Study for Resource and Project Planning
We chose to study a real case in a CRO to create a resource and project planning tool. There are many tasks and processes in clinical trial logistics management. But for simplicity, the data for one of the processes (Label Production for Clinical Trials Sample Collection) was retrieved and studied. Labels have information such as patient ID, sample ID, date and time of sample collection, type of assessment, and customized barcodes. The labels are affixed on the sample collection tubes and have important information about the samples. Incorrect information on the labels might cause samples to be mishandled. Therefore, design and production of labels must meet stringent quality standards.
The workflow for label production process is shown in
Figure 10. For a new protocol, the label process starts with Create Label Data (Task 1). However, for the existing protocol, the process does not always start from the beginning because the label has already been designed and it only needs to be replicated if more labels are needed. For replicating exiting design, the process only recurs from Replicate Label Data (Task 6). Often, information on the labels needs to be changed because of protocol amendments and the label process should start from the beginning (Task 1) to create new label designs or revise the existing ones. The label process can be broken down into three sub-processes: Label Design and Production (Task 1 to Task 10), Label Design Modification (Task 1 to Task 5), and Label Reproduction (Task 6 to Task 10).
The full set of data consists of 70 projects that have been active in a 3-month period. A sample row of data regarding label process is shown in
Table 11. The data includes 10 clinical trial projects (protocols), and within each project, there are multiple processes. Each process represents either Label Design and Production (Process 1) or Label Design Modification (Process 2) or label reproduction (Process 3). The same kind of process might be repeated within the planning horizon. Each process has an arrival date that represents when the request is made, and a due date that represents when that process needs to be performed by. Having arrival date and due date for each process makes this example different than the common RCMPSP examples that have multiple projects with one due date at the end. The objective function in this example is to minimize the total tardiness of all processes.
In this example, within each project, processes are assumed to be in sequence. Each process is given a time window (early finish date and late finish date) to be finished. The earliest finish time is calculated using the CPM (Critical Path) forward path. The latest finish time is calculated using the backward path starting from the end of the planning horizon. The task within each process is also sequential in this example and each task also has an early finish date and late finish date.
For simplicity, processing times are assumed constant regardless of the size of the job and are shown in
Table 12. The resources needed to perform each job have also been identified.
5.11.1. Inter-Scenarios Comparisons
The LP formulation for each scenario has been solved using GLPK 4.65 and Gurobi 8.1 optimization solvers. Each scenario is compared with its adjacent scenario in
Table 13. To be able to compare scenarios, the number of projects has been fixed to 10 (17 processes total) for all scenarios, and number of resources has been fixed to 6 for Scenarios I, II, and III. The weight of tardiness is set to 1 for all the scenarios.
The number of modes used in Scenarios I and IV is 1, while the number of modes used in Scenarios II, III, V, and VI is 2. The mode in all scenarios represents one or more resource groups (g) with a defined number of resources needed and the time it takes to process the task. In other words, a mode represents a unique combination of resources and the rate at which that task will be processed in that mode.
In Scenarios I and IV, only one mode (resource group) is allocated to every task, because it is assumed that all resources within each resource group work at the same speed.
In Scenario II, two modes were used that each consists of three resource types. For example, m1 consists of PM1, QC1, OA2, and m2 consists of PM2, QC2, and OA2. We have set the BR (number of resources) equal to 1 to make each resource type a unique resource from that mode. For example, PM1 represents an individual project manager in Scenario II. Scenario V is the same as Scenario II, except that BR is a variable. In this scenario, BR can represent one or many people depending on the work demand. For example, the results might end up having two resources for PM1, which implies that one project manager is not enough for that role. The number of modes depends on the number of people in each department. For example, if we have three project managers, then we need three modes.
However, in Scenario III and VI, two modes are allocated to each task, because we have two work groups that consist of different resources. Mode 1 and 2 belong to work group 1 and 2, respectively. Each resource group is assigned a subscript that shows the work group and mode that it belongs to. For example, PM1 means the project managers group from work group 1 or mode 1. Each inter-scenario comparison will be explained in the following sub-sections.
5.11.2. Scenario I vs. IV
In Scenario I, six shared resources have been assigned (two PM, two QC, two OA), i.e., the system output task schedule with 66 days tardiness. In Scenario IV, 10 projects were given to the LP to minimize the total tardiness of the projects by finding the optimal number of resources for each department. The LP outputs a total of nine resources (two PM, three QC, four OA).
5.11.3. Scenario I vs. II
In Scenario I, six shared resources were used; however, in Scenario II, six dedicated resources were used. For example, in Scenario II, resource type PM1 is assigned to projects 1, 2, 3, 4, and 5, while PM2 is assigned to projects 6, 7, 8, 9, and10. The output of Scenario II shows that the total tardiness increased to 96 days compared to Scenario I, which has a tardiness of 66 days. This is because in Scenario I, resources are shared across all projects, so each project has access to two resource types. However, in Scenario II, each project has access to only one resource type because of the dedication policy.
5.11.4. Scenario II vs. V
In Scenarios II and V, resource types are dedicated to projects. The only difference is that Scenario V has a variable number of resources for each resource type, while Scenario II has a fixed number of resources for each resource type. In Scenario II, six resources were used and the output came out to be 96 days of tardiness. The same tasks were given to Scenario V to minimize the tardiness; the output showed an increase of resources to a total of 15 resources (PM1 = 3, QC1 = 2, OA1 = 3, PM2 = 2, QC2 = 3, OA2 = 2).
5.11.5. Scenario II vs. III
In Scenario III, two work groups have been created that each includes three resource types, each resource type has given only one resource (only one person in each resource’s type, a total of six resources). In Scenario II, there are six resources that have been dedicated to one-to-many projects. In Scenario III, resources are being assigned to a work group by the LP. The results show less tardiness for Scenario III (68 days) than Scenario II (96 days). One reason for the better performance of Scenario III is that the assignment of projects to the work groups is conducted optimally by the LP rather than manual assignments of projects by management.
5.11.6. Scenario III vs. VI
In Scenario III, the number of resources for each work group has been fixed (three resources for work group 1 and three resources for work group 2), while in Scenario VI, the number of resources are variables. Scenario III has an output tardiness of 68 days with six resources, while Scenario IV has an output tardiness of 0 days with a total of 15 resources.
The second difference is in the assignment of projects to each work group in each scenario. In Scenario III, five projects have been assigned to each work group, while in Scenario VI, work group 1 received six projects and work group 2 received four projects.
5.11.7. Scenario IV vs. V
Scenario IV determines the optimal number of resources for a shared resource structure, while Scenario V determines the optimal number of resources for a dedicated resource structure. The results show that Scenario IV performs better (9 resources) than Scenario V (15 resources) with respect to the total number of resources. The reason for that is that resources are being shared in Scenario IV and fewer overall resources are needed as opposed to the dedicated resource model.
5.11.8. Scenario V vs. VI
Scenario V and VI both showed 0 tardiness and 15 total number of resources. However, the breakdown of the number of resources for each resource type is different in each scenario. For example, in Scenario V, PM1 has three and PM2 has two resources, while in Scenario VI, PM1 has two and PM2 has one resource.
5.11.9. Intra-Scenario Comparisons
In this subsection, each scenario will be given two datasets, baseline and second example, and results will be compared within that scenario. For the second example, one of the parameters will be changed to see the behavior of the scenario; for example, the number of resources or projects will be changed.
5.11.10. Scenario I (6 Resources vs. 9 Resources)
The number of resources has been increased from six in the baseline to nine in the second example scenario. Nine resources have been evenly distributed to each resource type (three PMs, three QCs, three OAs). The output tardiness decreased to 8 days from 66 days. This validates the behavior of this scenario, because as the number of resources increases, tardiness must decrease if all other parameters such as the number of projects stay constant. The details of this comparison are in
Table 14.
5.11.11. Scenario II (6 Resources vs. 30 Resources)
The number of resources has been increased from 6 in the baseline to 30 in the second example scenario. Each project has been given only one dedicated resource of each type (for example for project 1, BR[PM1]= BR[QC1] = BR[OA1] = 1) that cannot be shared with other projects. The tardiness has decreased from 96 days in the baseline to 0 in the second example. The results validate the behavior of this scenario, because in our dataset, processes are in sequence in each project. Therefore, each project requires only one dedicated resource of each resource type at a time to finish its processes by their due dates. The details of this comparison are in
Table 15.
5.11.12. Scenario III (6 Resources vs. 18 Resources)
The number of resources has been increased from 6 in the baseline to 18 in the second example scenario. Eighteen resources have been distributed evenly to each work group in the second example. The results show that the second example has a lower tardiness (10 days) than the baseline (68 days). Projects have been distributed to all three work groups in the second example. This implies that as the number of resources increases, tardiness decreases. The details of this comparison are in
Table 16.
5.11.13. Scenario IV (10 Projects vs. 20 Projects)
The number of projects has been changed from 10 in the baseline to 20 in the second example. The output shows the number of resources has been increased from 9 to 20 resources. This implies that as the number of projects increases, the number of resources increases. The details of this comparison are in
Table 17.
5.11.14. Scenario V (6 Type of Resources vs. 3 Type of Resources)
This scenario is very similar to Scenario II with the only difference being that the number of resources is variable in this scenario. In the baseline model, one resource is assigned to more than one project, while in the second example, each resource is assigned to one and only one project. The number of resource types has decreased from six in the baseline to three in the second example, because in the second example each project will be assigned only one dedicated resource of each type and there is no need to differentiate resources. The output of the second example shows an assignment of 1 resource to every resource type and project (total of 30 resources), while the baseline has a total of 15 resources. This implies that more resource dedication to projects requires more resources. The details of this comparison are in
Table 18.
5.11.15. Scenario VI (2 Work Groups vs. 3 Work Groups)
The number of work groups has been increased from two in the baseline to three in the second example scenario. The output of the second example shows 17 resources while the baseline has 15 resources. This implies that more work groups require more resources to finish all the projects with 0 tardiness. The details of this comparison are in
Table 19.
5.12. Computational Effort
The full set of data for the label process consists of 70 projects.
Table 20 shows the solution time for the case with 70 projects for Scenarios IV, V, and VI. Since Scenarios I, II, and III take the number of resources as input, a similar comparison for these scenarios is not practical.
The LP model was solved using Gurobi optimizer version 8.1.0. The computer that was used has the following specifications: Windows 10 Home, a 64-bit operating system, an x64-based processor, an Intel Core i7-6700 (3.4 GHz) processor with installed 16.0 GB RAM.
5.13. Cost Analysis
Scenarios IV, V, and VI were chosen to perform an analysis on the effects of resource cost and tardiness costs on the number of resources and total tardiness of projects. These scenarios minimize the tardiness costs by changing the number of resources and tardiness of each project or process. Scenarios I, II, and III were not considered for this analysis, because the number of resources is fixed in these scenarios.
For this analysis, resource costs were also added to the objective function of each scenario to minimize the total resource cost as well as the total tardiness of projects. The new objective function is as follows:
where
= unit cost of tardiness for projects,
= unit cost of tardiness for processes, and
= unit cost of each resource
k that belongs to group
g.
In practice, costs of tardiness in logistics management of clinical trials can be found by identifying the consequences of delays in the logistics processes. For example, delays in the shipping of the collection kits to the site will cause missing a sample collection, or delays in coordinating of a sample shipment with a site will delay the sample shipment to the lab, and in a worst-case scenario, unsatisfactory services will cause a patient to withdraw from a clinical trial.
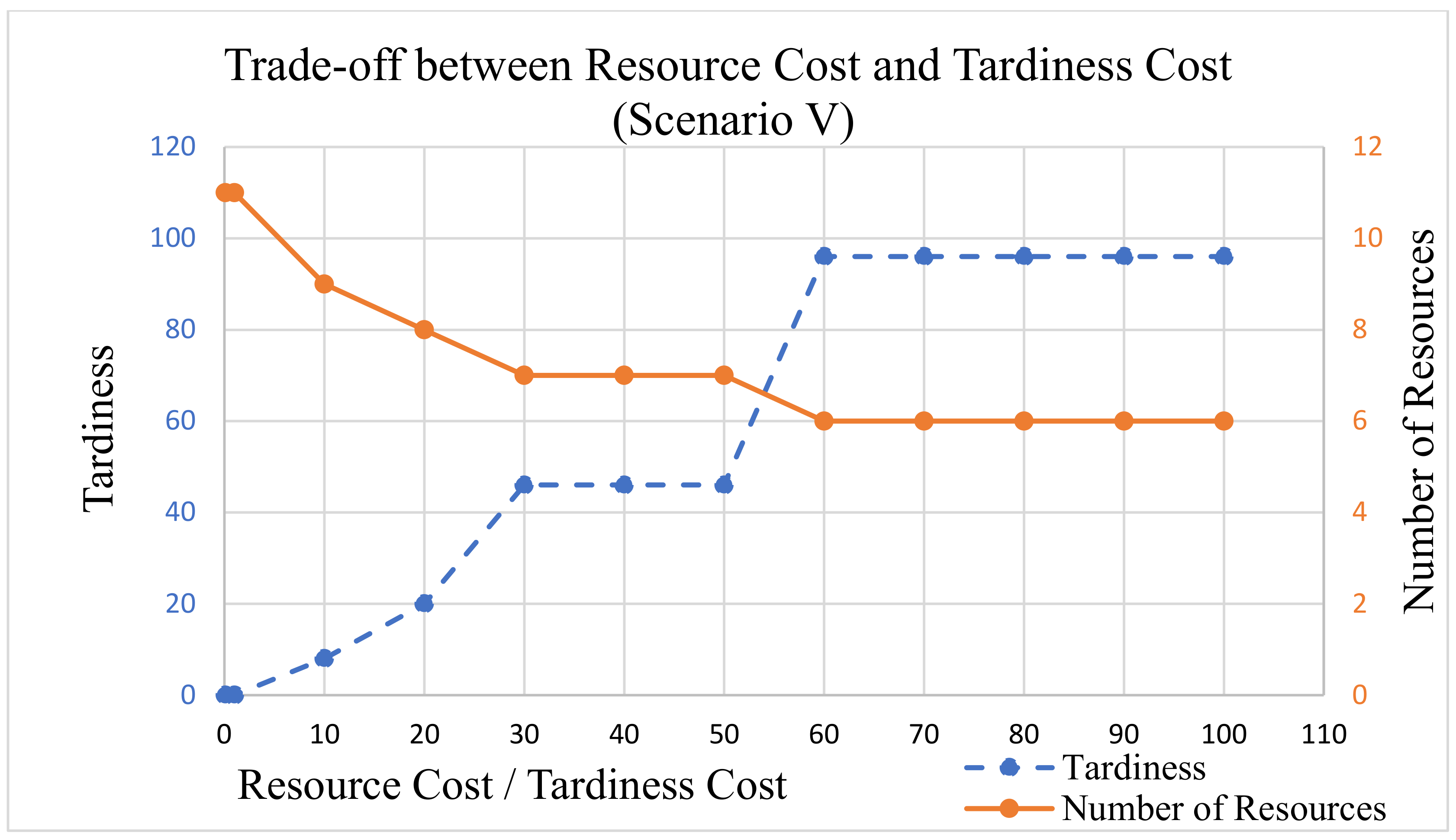
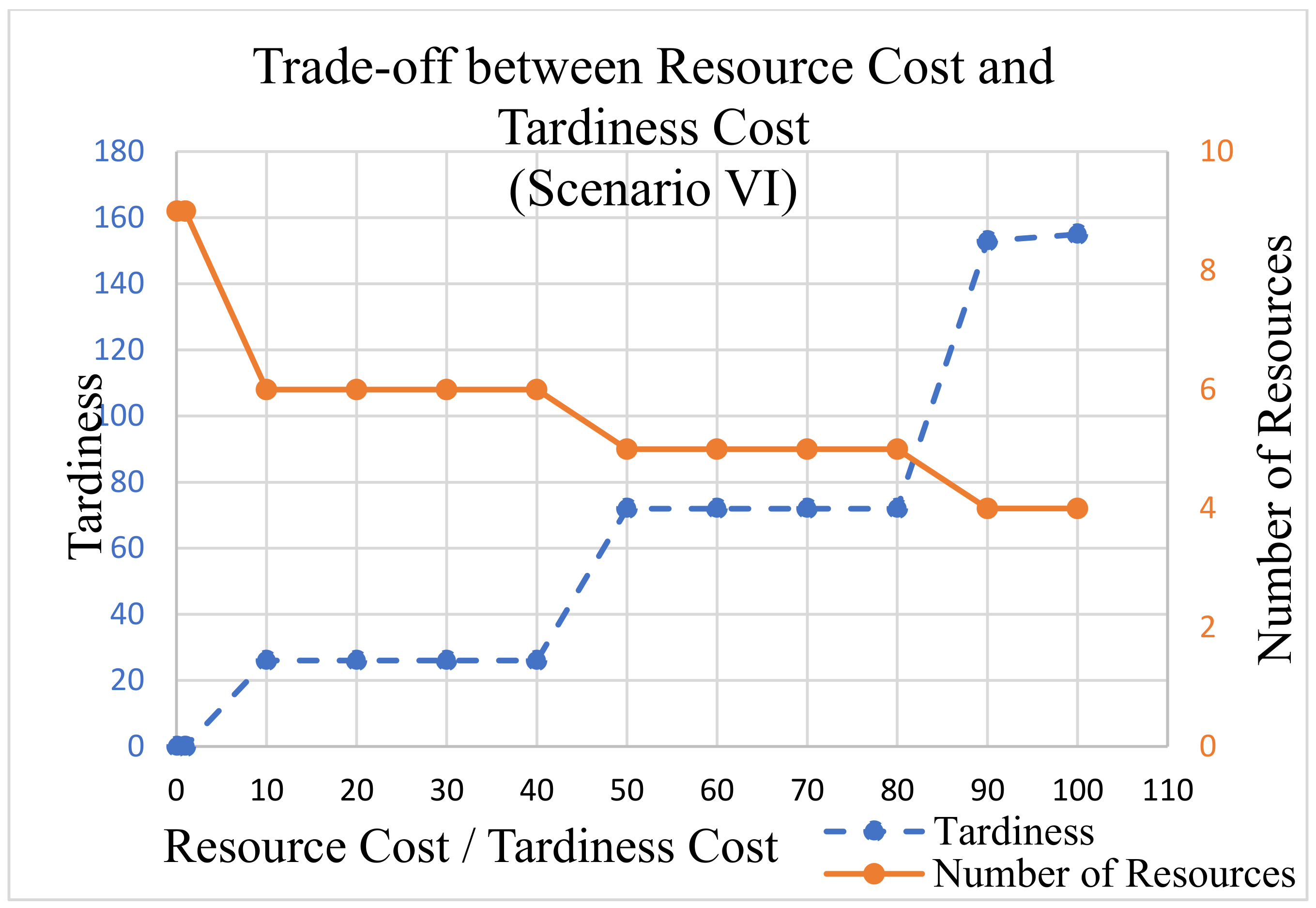
In this analysis,
is not used, because our clinical trials example data do not include due dates on projects. The
value was fixed to 1 as the tardiness cost for processes. The
value is set at 1.5 for PM, 1 for QC, and 1 for OA.
was multiplied by {1, 10, 20, 30, 40, 50, 60, 70, 80, 90, 100} during each LP run. The output tardiness and the number of resources were recorded for each run and are displayed in
Figure 11,
Figure 12 and
Figure 13 for Scenarios IV, V, and VI, respectively. The X-axis in these figures represents the ratio of resource cost to tardiness cost, which starts from 1 and is increased by 10. The Y-axis represents tardiness, and the right y-axis represents the number of resources.
For example, for Scenario IV at ratios between 0 and less than or equal to 1, shown in
Figure 11, the number of resources stays constant at 9 and the total tardiness stays at 0. At the ratio of 10, the number of resources decreases to 6 and tardiness increases to 26. The number of resources and tardiness stays unchanged at ratios of 20, 30, and 40. At the ratio of 50, the number of resources decreases to 5 and tardiness increases to 72. The number of resources and tardiness stays unchanged at ratios of 60, 70, and 80. At the ratio of 90, the number of resources decreases to 4 and tardiness increases to 153. At the ratio of 100, the number of resources decreases to its lowest level, which is 3, and tardiness increases to 253.
Also, in
Figure 12 and
Figure 13, as the ratio of resource cost to tardiness cost increases, the number of resources decreases and tardiness increases. This is because the resources become so costly that the LP tends to delay the tasks instead of using more resources. Inversely, as the ratio of resource cost to tardiness cost decreases, the number of resources increases and tardiness decreases. In this case, the LP uses more human resources than time resources (tardiness).
6. Discussion and Conclusions
Clinical trials require complex coordination of tasks and involve many resources that need to be planned and managed carefully. Currently, due to disparate data sources among large number of stakeholders in clinical trials, logistics management is inefficient.
While the literature includes conceptual solutions or pathways for managing logistics in areas other than clinical trials, project management in general being an example, the area of clinical logistics is quite challenging in practice because of the diverse sources of data, complex interactions, timeliness and accuracy requirements, and the need for tedious manual coordination. Towards that end, this paper focused on issues in the logistics management of clinical trials and proposed a framework using contemporary software tools and technology.
The main components of this framework from top to bottom are web portal, intelligent agents, BPMS, project card, external applications, document repository, data repository, and analytics. The framework proposed here brings together an integrative platform to exchange data among computers faster and remove many redundant work and possible errors. Most tasks can be automated using intelligent software agents, as was the case in our implementation. It also facilitates easy and efficient collaboration across organizations. Data can be analyzed using the analytics component to obtain meaningful data and eventually improve workflows to better manage the projects. The widespread global revolution in Artificial Intelligence (AI) and Machine Learning (ML) makes a compelling case for a data driven intelligent agent approach.
The natural question that comes up is how such an architecture can improve operational excellence. To this end, we demonstrate the use of a quantitative tool (which is data dependent) for resource and project planning, a critical element for operational excellence. When resources are well allocated and managed, a CRO has both satisfied customers and employees.
The planning tool also contributes to the RCMPSP literature by contrasting several different resource scenarios such as the shared pool, dedicated resources for each project, and the creation of several work groups with dedicated resources. These are Pareto trade-offs. While the shared pool is the most efficient, there are reasons why an organization may wish to use dedicated resources to reduce complexity or create several work groups to improve team morale. Additional features of this tool that are tailored to the logistics management of clinical trials are as follows: multiple due dates for processes, resource familiarity factors with tasks and projects, and an objective function to minimize the total tardiness of processes and projects.
7. Future Research
This framework can be further improved by enhancing its existing components for clinical trials and similar industries. A longitudinal study can be performed to test the applicability of this framework in industries and measure what its impact is on office productivity. Another area of future research is to come up with a detailed quality tracker within this framework so that the CRO can assess how well it is performing. In the age of big data and data analytics, it is also important to track other key performance indicators. Finally, machine learning techniques could be used to improve the project planning tool. For example, it would be interesting to see how the task estimates evolve, how they compare across the three management scenarios, and how realistic the project and resource allocation tasks themselves are and, if not, how they should be adjusted.