How Fermentation Affects the Antioxidant Properties of Cereals and Legumes
Abstract
1. Antioxidant Compounds in Food Matrices
1.1. Phenolic Compounds
1.2. Antioxidant Peptides and Protein Derivatives
1.3. Synthetic Antioxidants
2. Bioaccessibility and Bioavailability of Antioxidant Compounds
3. Estimation of the Antioxidant Activity in Foods
3.1. In Vitro Assays
3.2. Ex Vivo Assays
3.3. In Vivo Assays
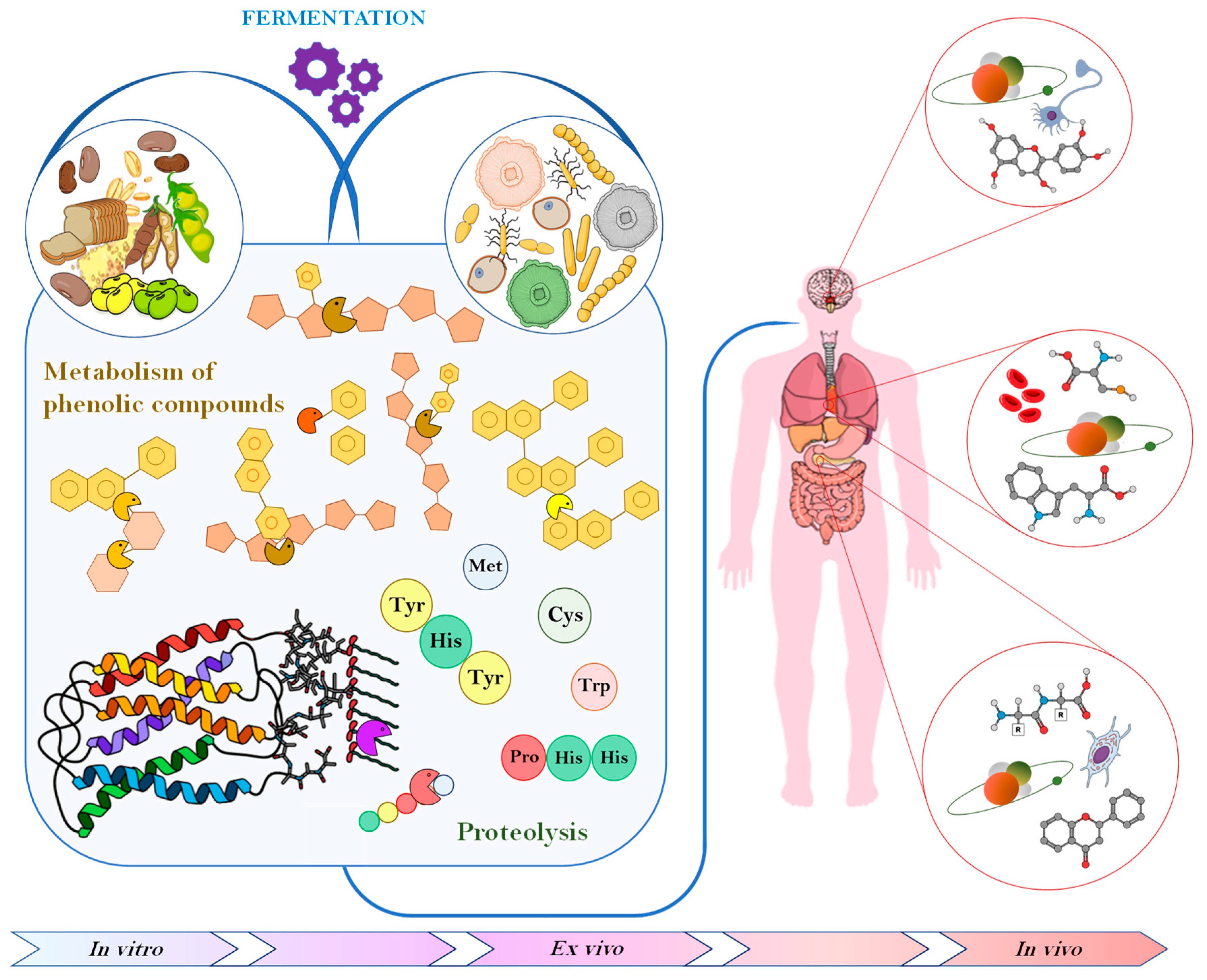
4. Effect of Microbial Fermentation on the Antioxidant Activity
4.1. Metabolic Activities Affecting Phenolics
4.1.1. Metabolic Activities Affecting Phenolic Acids
4.1.2. Metabolic Activities Affecting Flavonoids
4.1.3. Metabolic Activities Affecting Tannins
4.2. Release of Antioxidant Peptides
4.3. Secondary Effects of Fermentation
4.3.1. Vitamins
4.3.2. Production of Exopolysaccharides
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Carocho, M.; Ferreira, I.C. A review on antioxidants, prooxidants and related controversy: Natural and synthetic compounds, screening and analysis methodologies and future perspectives. Food Chem. Toxicol. 2013, 51, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Lobo, V.; Patil, A.; Phatak, A.; Chandra, N. Free radicals, antioxidants and functional foods: Impact on human health. Pharmacogn. Rev. 2010, 4, 118. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, K.; Aggarwal, B.; Singh, R.; Buttar, H.; Wilson, D.; De Meester, F. Food antioxidants and their anti-inflammatory properties: A potential role in cardiovascular diseases and cancer prevention. Diseases 2016, 4, 28. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.N.; Bristi, N.J.; Rafiquzzaman, M. Review on in vivo and in vitro methods evaluation of antioxidant activity. Saudi Pharm. J. 2013, 21, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Brewer, M.S. Natural antioxidants: Sources, compounds, mechanisms of action, and potential applications. Compr. Rev. Food Sci. Food Saf. 2011, 10, 221–247. [Google Scholar] [CrossRef]
- Atmaca, G. Antioxidant effects of sulfur-containing amino acids. Yonsei Med. J. 2004, 45, 776–788. [Google Scholar] [CrossRef] [PubMed]
- Rizzello, C.G.; Tagliazucchi, D.; Babini, E.; Rutella, G.S.; Saa, D.L.T.; Gianotti, A. Bioactive peptides from vegetable food matrices: Research trends and novel biotechnologies for synthesis and recovery. J. Funct. Foods 2016, 27, 549–569. [Google Scholar] [CrossRef]
- Sarmadi, B.H.; Ismail, A. Antioxidative peptides from food proteins: A review. Peptides 2010, 31, 1949–1956. [Google Scholar] [CrossRef]
- Pokorný, J. Are natural antioxidants better–and safer–than synthetic antioxidants? Eur. J. Lipid Sci. Technol. 2007, 109, 629–642. [Google Scholar] [CrossRef]
- Box, J.D. Investigation of the Folin-Ciocalteau phenol reagent for the determination of polyphenolic substances in natural waters. Water Res. 1983, 17, 511–525. [Google Scholar] [CrossRef]
- Shahidi, F.; Zhong, Y. Measurement of antioxidant activity. J. Funct. Foods 2015, 18, 757–781. [Google Scholar] [CrossRef]
- Singh, B.; Singh, J.P.; Kaur, A.; Singh, N. Phenolic composition and antioxidant potential of grain legume seeds: A review. Food Res. Int. 2017, 101, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Gobbetti, M.; De Angelis, M.; Di Cagno, R.; Calasso, M.; Archetti, G.; Rizzello, C.G. Novel insights on the functional/nutritional features of the sourdough fermentation. Int. J. Food Microbiol. 2019, 302, 103–113. [Google Scholar] [CrossRef] [PubMed]
- Verni, M.; Rizzello, C.G.; Coda, R. Fermentation biotechnology applied to cereal industry by-products: Nutritional and functional insights. Front. Nutr. 2019, 6, 42. [Google Scholar] [CrossRef] [PubMed]
- Dey, T.B.; Chakraborty, S.; Jain, K.K.; Sharma, A.; Kuhad, R.C. Antioxidant phenolics and their microbial production by submerged and solid state fermentation process: A review. Trends Food Sci. Technol. 2016, 53, 60–74. [Google Scholar]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [CrossRef] [PubMed]
- Lupea, A.X.; Pop, M.; Cacig, S. Structure-radical scavenging activity relationships of flavonoids from Ziziphus and Hydrangea extracts. Rev. Chim. 2008, 59, 309–313. [Google Scholar]
- Shahidi, F.; Naczk, M. Phenolics in Food and Nutraceuticals; CRC press: Boca Raton, FL, USA, 2003; pp. 1–558. [Google Scholar]
- Shahidi, F.; Ambigaipalan, P. Phenolics and polyphenolics in foods, beverages and spices: Antioxidant activity and health effects−A review. J. Funct. Foods 2015, 18, 820–897. [Google Scholar] [CrossRef]
- Shahidi, F.; Zhong, Y. Bioactive peptides. J. AOAC Int. 2008, 91, 914–931. [Google Scholar]
- Chen, H.M.; Muramoto, K.; Yamauchi, F.; Fujimoto, K.; Nokihara, K. Antioxidative properties of histidine-containing peptides designed from peptide fragments found in the digests of a soybean protein. J. Agric. Food Chem. 1998, 46, 49–53. [Google Scholar] [CrossRef]
- Sánchez, A.; Vázquez, A. Bioactive peptides: A review. Food Qual. Saf. 2017, 1, 29–46. [Google Scholar] [CrossRef]
- Nanditha, B.; Prabhasankar, P. Antioxidants in bakery products: A review. Crit. Rev. Food Sci. Nutr. 2008, 49, 1–27. [Google Scholar] [CrossRef]
- EFSA. Panel on food additives and nutrient sources added to food (ANS); Scientific opinion on the reevaluation of butylated hydroxyanisole—BHA (E 320) as a food additive. EFSA J. 2011, 9, 2392. [Google Scholar] [CrossRef]
- EFSA. Panel on food additives and nutrient sources added to food (ANS); Scientific opinion on the reevaluation of butylated hydroxytoluene BHT (E 321) as a food additive. EFSA J. 2012, 10, 2588. [Google Scholar] [CrossRef]
- Carbonell-Capella, J.M.; Buniowska, M.; Barba, F.J.; Esteve, M.J.; Frígola, A. Analytical methods for determining bioavailability and bioaccessibility of bioactive compounds from fruits and vegetables: A review. Compr. Rev. Food Sci. Food Saf. 2014, 13, 155–171. [Google Scholar] [CrossRef]
- Martin, K.R.; Appel, C.L. Polyphenols as dietary supplements: A double-edged sword. Nutr. Diet. Suppl. 2010, 2, 1–12. [Google Scholar] [CrossRef]
- Hackman, R.M.; Polagruto, J.A.; Yan Zhu, Q.; Sun, B.; Fujii, H.; Keen, C.L. Flavanols: Digestion, absorption and bioactivity. Phytochem. Rev. 2008, 7, 195–208. [Google Scholar] [CrossRef]
- D’Archivio, M.; Filesi, C.; Di Benedetto, R.; Gargiulo, R.; Giovannini, C.; Masella, R. Polyphenols, dietary sources and bioavailability. Ann. Ist. Super. Sanita 2007, 43, 348. [Google Scholar]
- Cermak, R.; Durazzo, A.; Maiani, G.; Böhm, V.; Kammerer, D.R.; Carle, R.; Wiczkowski, W.; Piskula, M.K.; Galensa, R. The influence of postharvest processing and storage of foodstuffs on the bioavailability of flavonoids and phenolic acids. Mol. Nutr. Food Res. 2009, 53, S184–S193. [Google Scholar] [CrossRef]
- García-Mora, P.; Martín-Martínez, M.; Bonache, M.A.; González-Múniz, R.; Peñas, E.; Frias, J.; Martinez-Villaluenga, C. Identification, functional gastrointestinal stability and molecular docking studies of lentil peptides with dual antioxidant and angiotensin I converting enzyme inhibitory activities. Food Chem. 2017, 221, 464–472. [Google Scholar] [CrossRef]
- Coda, R.; Rizzello, C.G.; Pinto, D.; Gobbetti, M. Selected lactic acid bacteria synthesize antioxidant peptides during sourdough fermentation of cereal flours. Appl. Environ. Microbiol. 2012, 78, 1087–1096. [Google Scholar] [CrossRef]
- Rizzello, C.G.; Lorusso, A.; Russo, V.; Pinto, D.; Marzani, B.; Gobbetti, M. Improving the antioxidant properties of quinoa flour through fermentation with selected autochthonous lactic acid bacteria. Int. J. Food Microbiol. 2017, 241, 252–261. [Google Scholar] [CrossRef]
- Filannino, P.; Di Cagno, R.; Gobbetti, M. Metabolic and functional paths of lactic acid bacteria in plant foods: Get out of the labyrinth. Curr. Opin. Biotechnol. 2018, 49, 64–72. [Google Scholar] [CrossRef]
- Jiménez, N.; Esteban-Torres, M.; Mancheño, J.M.; de las Rivas, B.; Muñoz, R. Tannin degradation by a novel tannase enzyme present in some Lactobacillus plantarum strains. Appl. Environ. Microbiol. 2014, 80, 2991–2997. [Google Scholar] [CrossRef]
- Raccach, M. The antimicrobial activity of phenolic antioxidant in food: A review. J. Food Saf. 1984, 6, 141–170. [Google Scholar] [CrossRef]
- Sánchez-Maldonado, A.F.; Schieber, A.; Gänzle, M.G. Structure—function relationships of the antibacterial activity of phenolic acids and their metabolism by lactic acid bacteria. J. Appl. Microbiol. 2011, 111, 1176–1184. [Google Scholar] [CrossRef]
- Anson, N.M.; Selinheimo, E.; Havenaar, R.; Aura, A.M.; Mattila, I.; Lehtinen, P.; Bast, A.; Poutanen, K.; Haenen, G.R. Bioprocessing of wheat bran improves in vitro bioaccessibility and colonic metabolism of phenolic compounds. J. Agric. Food Chem. 2009, 57, 6148–6155. [Google Scholar] [CrossRef]
- Manini, F.; Brasca, M.; Plumed-Ferrer, C.; Morandi, S.; Erba, D.; Casiraghi, M.C. Study of the chemical changes and evolution of microbiota during sourdoughlike fermentation of wheat bran. Cereal Chem. 2014, 91, 342–349. [Google Scholar] [CrossRef]
- Savolainen, O.I.; Coda, R.; Suomi, K.; Katina, K.; Juvonen, R.; Hanhineva, K.; Poutanen, K. The role of oxygen in the liquid fermentation of wheat bran. Food Chem. 2014, 153, 424–431. [Google Scholar] [CrossRef]
- Xie, C.Y.; Gu, Z.X.; You, X.; Liu, G.; Tan, Y.; Zhang, H. Screening of edible mushrooms for release of ferulic acid from wheat bran by fermentation. Enz. Microb. Technol. 2010, 46, 125–128. [Google Scholar] [CrossRef]
- Benedetti, S.; Primiterra, M.; Tagliamonte, M.C.; Carnevali, A.; Gianotti, A.; Bordoni, A.; Canestrari, F. Counteraction of oxidative damage in the rat liver by an ancient grain (Kamut brand khorasan wheat). Nutrition 2012, 28, 436–441. [Google Scholar] [CrossRef]
- Schmidt, C.G.; Gonçalves, L.M.; Prietto, L.; Hackbart, H.S.; Furlong, E.B. Antioxidant activity and enzyme inhibition of phenolic acids from fermented rice bran with fungus Rizhopus oryzae. Food Chem. 2014, 146, 371–377. [Google Scholar] [CrossRef]
- Razak, D.L.A.; Rashid, N.Y.A.; Jamaluddin, A.; Sharifudin, S.A.; Long, K. Enhancement of phenolic acid content and antioxidant activity of rice bran fermented with Rhizopus oligosporus and Monascus purpureus. Biocatal. Agric. Biotechnol. 2015, 4, 33–38. [Google Scholar] [CrossRef]
- Webber, D.M.; Hettiarachchy, N.S.; Li, R.; Horax, R.; Theivendran, S. Phenolic profile and antioxidant activity of extracts prepared from fermented heat-stabilized defatted rice bran. J. Food Sci. 2014, 79, H2383–H2391. [Google Scholar] [CrossRef]
- Katina, K.; Laitila, A.; Juvonen, R.; Liukkonen, K.H.; Kariluoto, S.; Piironen, V.; Landberg, R.; Aman, P.; Poutanen, K. Bran fermentation as a means to enhance technological properties and bioactivity of rye. Food Microbiol. 2007, 24, 175–186. [Google Scholar] [CrossRef]
- Koistinen, V.M.; Katina, K.; Nordlund, E.; Poutanen, K.; Hanhineva, K. Changes in the phytochemical profile of rye bran induced by enzymatic bioprocessing and sourdough fermentation. Food Res. Intern. 2016, 89, 1106–1115. [Google Scholar] [CrossRef]
- Nordlund, E.; Katina, K.; Aura, A.M.; Poutanen, K. Changes in bran structure by bioprocessing with enzymes and yeast modifies the in vitro digestibility and fermentability of bran protein and dietary fibre complex. J. Cereal Sci. 2013, 58, 200–208. [Google Scholar] [CrossRef]
- Svensson, L.; Sekwati-Monang, B.; Lutz, D.L.; Schieber, A.; Ganzle, M.G. Phenolic acids and flavonoids in nonfermented and fermented red sorghum (Sorghum bicolor (L.) Moench). J. Agric. Food Chem. 2010, 58, 9214–9220. [Google Scholar] [CrossRef]
- Shumoy, H.; Gabaza, M.; Vandevelde, J.; Raes, K. Soluble and bound phenolic contents and antioxidant capacity of tef injera as affected by traditional fermentation. J. Food Compos. Anal. 2017, 58, 52–59. [Google Scholar] [CrossRef]
- Dongmo, S.N.; Sacher, B.; Kollmannsberger, H.; Becker, T. Key volatile aroma compounds of lactic acid fermented malt based beverages–impact of lactic acid bacteria strains. Food Chem. 2017, 229, 565–573. [Google Scholar] [CrossRef]
- Yu, B.; Lu, Z.X.; Bie, X.M.; Lu, F.X.; Huang, X.Q. Scavenging and anti-fatigue activity of fermented defatted soybean peptides. Eur. Food Res. Technol. 2008, 226, 415–421. [Google Scholar] [CrossRef]
- Sanjukta, S.; Rai, A.K.; Muhammed, A.; Jeyaram, K.; Talukdar, N.C. Enhancement of antioxidant properties of two soybean varieties of Sikkim Himalayan region by proteolytic Bacillus subtilis fermentation. J. Funct. Foods 2015, 14, 650–658. [Google Scholar] [CrossRef]
- Dueñas, M.; Hernández, T.; Lamparski, G.; Estrella, I.; Muñoz, R. Bioactive phenolic compounds of soybean (Glycine max cv. Merit): Modifications by different microbiological fermentations. Pol. J. Food Nutr. Sci. 2012, 62, 241–250. [Google Scholar]
- Cho, K.M.; Hong, S.Y.; Math, R.K.; Lee, J.H.; Kambiranda, D.M.; Kim, J.M.; Islam, S.A.; Yun, M.G.; Cho, J.J.; Lim, W.J.; et al. Biotransformation of phenolics (isoflavones, flavanols and phenolic acids) during the fermentation of cheonggukjang by Bacillus pumilus HY1. Food Chem. 2009, 114, 413–419. [Google Scholar] [CrossRef]
- Georgetti, S.R.; Vicentini, F.T.M.C.; Yokoyama, C.Y.; Borin, M.D.F.; Spadaro, A.C.C.; Fonseca, M.J.V. Enhanced in vitro and in vivo antioxidant activity and mobilization of free phenolic compounds of soybean flour fermented with different β-glucosidase-producing fungi. J. Appl. Microbiol. 2009, 106, 459–466. [Google Scholar] [CrossRef]
- Avila, M.; Jaquet, M.; Moine, D.; Requena, T.; Pelaez, C.; Arigoni, F.; Jankovic, I. Physiological and biochemical characterization of the two α-L-rhamnosidases of Lactobacillus plantarum NCC245. Microbiology 2009, 155, 2739–2749. [Google Scholar] [CrossRef]
- McCue, P.; Horii, A.; Shetty, K. Mobilization of phenolic antioxidants from defatted soybean powders by Lentinus edodes during solid-state bioprocessing is associated with enhanced production of laccase. Innov. Food Sci. Emerg. Technol. 2004, 5, 385–392. [Google Scholar] [CrossRef]
- Hu, Y.; Ge, C.; Yuan, W.; Zhu, R.; Zhang, W.; Du, L.; Xue, J. Characterization of fermented black soybean natto inoculated with Bacillus natto during fermentation. J. Sci. Food Agric. 2010, 90, 1194–1202. [Google Scholar] [CrossRef]
- Landete, J.M.; Hernández, T.; Robredo, S.; Duenas, M.; de las Rivas, B.; Estrella, I.; Munoz, R. Effect of soaking and fermentation on content of phenolic compounds of soybean (Glycine max cv. Merit) and mung beans (Vigna radiata L. Wilczek). Int. J. Food Sci. Nutr. 2015, 66, 203–209. [Google Scholar] [CrossRef]
- Hubert, J.; Berger, M.; Nepveu, F.; Paul, F.; Daydé, J. Effects of fermentation on the phytochemical composition and antioxidant properties of soy germ. Food Chem. 2008, 109, 709–721. [Google Scholar] [CrossRef]
- Riciputi, Y.; Serrazanetti, D.I.; Verardo, V.; Vannini, L.; Caboni, M.F.; Lanciotti, R. Effect of fermentation on the content of bioactive compounds in tofu-type products. J. Funct. Foods 2016, 27, 131–139. [Google Scholar] [CrossRef]
- Budryn, G.; Klewicka, E.; Grzelczyk, J.; Gałązka-Czarnecka, I.; Mostowski, R. Lactic acid fermentation of legume seed sprouts as a method of increasing the content of isoflavones and reducing microbial contamination. Food Chem. 2019, 285, 478–484. [Google Scholar] [CrossRef]
- Fiorda, F.A.; de Melo Pereira, G.V.; Thomaz-Soccol, V.; Medeiros, A.P.; Rakshit, S.K.; Soccol, C.R. Development of kefir-based probiotic beverages with DNA protection and antioxidant activities using soybean hydrolyzed extract, colostrum and honey. LWT-Food Sci. Technol. 2016, 68, 690–697. [Google Scholar] [CrossRef]
- Di Cagno, R.; Mazzacane, F.; Rizzello, C.G.; Vincentini, O.; Silano, M.; Giuliani, G.; De Angelis, M.; Gobbetti, M. Synthesis of isoflavone aglycones and equol in soy milks fermented by food-related lactic acid bacteria and their effect on human intestinal Caco-2 cells. J. Agric. Food Chem. 2010, 58, 10338–10346. [Google Scholar] [CrossRef]
- Dueñas, M.; Fernández, D.; Hernández, T.; Estrella, I.; Muñoz, R. Bioactive phenolic compounds of cowpeas (Vigna sinensis L). Modifications by fermentation with natural microflora and with Lactobacillus plantarum ATCC 14917. J. Sci. Food Agric. 2005, 85, 297–304. [Google Scholar] [CrossRef]
- Bautista-Expósito, S.; Peñas, E.; Dueñas, M.; Silván, J.M.; Frias, J.; Martínez-Villaluenga, C. Individual contributions of Savinase and Lactobacillus plantarum to lentil functionalization during alkaline pH-controlled fermentation. Food Chem. 2018, 257, 341–349. [Google Scholar] [CrossRef]
- Bartolomé, B.; Estrella, I.; Hernandez, T. Changes in phenolic compounds in lentils (Lens culinaris) during germination and fermentation. Z. Lebensm Unters Forsch. A 1997, 205, 290–294. [Google Scholar] [CrossRef]
- Gänzle, M.G. Enzymatic and bacterial conversions during sourdough fermentation. Food Microbiol. 2014, 37, 2–10. [Google Scholar] [CrossRef]
- Rodríguez, H.; Curiel, J.A.; Landete, J.M.; de las Rivas, B.; de Felipe, F.L.; Gómez-Cordovés, C.; Mancheño, J.M.; Muñoz, R. Food phenolics and lactic acid bacteria. Int. J. Food Microbiol. 2009, 132, 79–90. [Google Scholar] [CrossRef]
- Filannino, P.; Gobbetti, M.; De Angelis, M.; Di Cagno, R. Hydroxycinnamic acids used as external acceptors of electrons: An energetic advantage for strictly heterofermentative lactic acid bacteria. Appl. Environ. Microbiol. 2014, 80, 7574–7582. [Google Scholar] [CrossRef]
- Filannino, P.; Bai, Y.; Di Cagno, R.; Gobbetti, M.; Gänzle, M.G. Metabolism of phenolic compounds by Lactobacillus spp. during fermentation of cherry juice and broccoli puree. Food Microbiol. 2015, 46, 272–279. [Google Scholar] [CrossRef]
- Senger, D.R.; Li, D.; Jaminet, S.C.; Cao, S. Activation of the Nrf2 cell defense pathway by ancient foods: Disease prevention by important molecules and microbes lost from the modern western diet. PLoS ONE 2016, 11, e0148042. [Google Scholar] [CrossRef]
- Chatonnet, P.; Dubourdieu, D.; Boidron, J.N.; Lavigne, V. Synthesis of volatile phenols by Saccharomyces cerevisiae in wines. J. Sci. Food Agric. 1993, 62, 191–202. [Google Scholar] [CrossRef]
- Gasson, M.J.; Kitamura, Y.; McLauchlan, W.R.; Narbad, A.; Parr, A.J.; Parsons, E.L.H.; Payne, J.; Rhodes, M.J.C.; Walton, N.J. Metabolism of ferulic acid to vanillin. A bacterial gene of the enoyl-SCoA hydratase/isomerase superfamily encodes an enzyme for the hydration and cleavage of a hydroxycinnamic acid SCoA thioester. J. Biol. Chem. 1998, 273, 4163–4170. [Google Scholar] [CrossRef]
- Longo, M.A.; Sanromán, M.A. Production of food aroma compounds: Microbial and enzymatic methodologies. Food Technol. Biotechnol. 2006, 44, 335–353. [Google Scholar]
- Donaghy, J.; Kelly, P.F.; McKay, A.M. Detection of ferulic acid esterase production by Bacillus spp. and lactobacilli. Appl. Microbiol. Biotechnol. 1998, 50, 257–260. [Google Scholar] [CrossRef]
- Benoit, I.; Danchin, E.G.; Bleichrodt, R.J.; de Vries, R.P. Biotechnological applications and potential of fungal feruloyl esterases based on prevalence, classification and biochemical diversity. Biotechnol. Lett. 2008, 30, 387–396. [Google Scholar] [CrossRef]
- Mathew, S.; Abraham, T.E. Ferulic acid: An antioxidant found naturally in plant cell walls and feruloyl esterases involved in its release and their applications. Crit. Rev. Biotechnol. 2004, 24, 59–83. [Google Scholar] [CrossRef]
- Aurilia, V.; Parracino, A.; Saviano, M.; D’Auria, S. The psychrophilic bacterium Pseudoalteromonas halosplanktis TAC125 possesses a gene coding for a cold-adapted feruloyl esterase activity that shares homology with esterase enzymes from γ-proteobacteria and yeast. Gene 2007, 397, 51–57. [Google Scholar] [CrossRef]
- Coghe, S.; Benoot, K.; Delvaux, F.; Vanderhaegen, B.; Delvaux, F.R. Ferulic acid release and 4-vinylguaiacol formation during brewing and fermentation: Indications for feruloyl esterase activity in Saccharomyces cerevisiae. J. Agric. Food Chem. 2004, 52, 602–608. [Google Scholar] [CrossRef]
- Anson, N.M.; Hemery, Y.M.; Bast, A.; Haenen, G.R. Optimizing the bioactive potential of wheat bran by processing. Food. Funct. 2012, 3, 362–375. [Google Scholar] [CrossRef]
- Koistinen, V.M.; Nordlund, E.; Katina, K.; Mattila, I.; Poutanen, K.; Hanhineva, K.; Aura, A.M. Effect of bioprocessing on the in vitro colonic microbial metabolism of phenolic acids from rye bran fortified breads. J. Agric. Food Chem. 2017, 65, 1854–1864. [Google Scholar] [CrossRef]
- Xiao, Y.; Xing, G.; Rui, X.; Li, W.; Chen, X.; Jiang, M.; Dong, M. Enhancement of the antioxidant capacity of chickpeas by solid state fermentation with Cordyceps militaris SN-18. J. Funct. Foods 2014, 10, 210–222. [Google Scholar] [CrossRef]
- McCue, P.; Shetty, K. Health benefits of soy isoflavonoids and strategies for enhancement: A review. Crit. Rev. Food Sci. Nutr. 2004, 44, 361–367. [Google Scholar] [CrossRef]
- Randhir, R.; Vattem, D.; Shetty, K. Solid-state bioconversion of fava bean by Rhizopus oligosporus for enrichment of phenolic antioxidants and L-DOPA. Inn. Food Sci. Emerg. Technol. 2004, 5, 235–244. [Google Scholar] [CrossRef]
- Bhanja, T.; Kumari, A.; Banerjee, R. Enrichment of phenolics and free radical scavenging property of wheat koji prepared with two filamentous fungi. Bioresour. Technol. 2009, 100, 2861–2866. [Google Scholar] [CrossRef]
- Iwamoto, K.; Tsuruta, H.; Nishitaini, Y.; Osawa, R. Identification and cloning of a gene encoding tannase (tannin acylhydrolase) from Lactobacillus plantarum ATCC 14917T. Syst. Appl. Microbiol. 2008, 31, 269–277. [Google Scholar] [CrossRef]
- Ramírez-Coronel, M.A.; Viniegra-Gonzalez, G.; Darvill, A.; Augur, C. A novel tannase from Aspergillus niger with β-glucosidase activity. Microbiology 2003, 149, 2941–2946. [Google Scholar] [CrossRef]
- Sánchez-Patán, F.; Tabasco, R.; Monagas, M.; Requena, T.; Peláez, C.; Moreno-Arribas, M.V.; Bartolomé, B. Capability of Lactobacillus plantarum IFPL935 to catabolize flavan-3-ol compounds and complex phenolic extracts. J. Agric. Food Chem. 2012, 60, 7142–7151. [Google Scholar] [CrossRef]
- Gleńsk, M.; Hurst, W.J.; Glinski, V.B.; Bednarski, M.; Gliński, G.A. Isolation of 1-(30,40-Dihydroxyphenyl)-3-(2″,4″,6″-trihydroxyphenyl)-propan-2-ol from grape seed extract and evaluation of its antioxidant and antispasmodic potential. Molecules 2019, 24, 2466. [Google Scholar] [CrossRef]
- Gobbetti, M.; De Angelis, M.; Corsetti, A.; Di Cagno, R. Biochemistry and physiology of sourdough lactic acid bacteria. Trends Food Sci. Technol. 2005, 16, 57–69. [Google Scholar] [CrossRef]
- Galli, V.; Mazzoli, L.; Luti, S.; Venturi, M.; Guerrini, S.; Paoli, P.; Vincenzini, M.; Granchi, L.; Pazzagli, L. Effect of selected strains of lactobacilli on the antioxidant and anti-inflammatory properties of sourdough. Int. J. Food Microbiol. 2018, 286, 55–65. [Google Scholar] [CrossRef]
- Matsuo, M. In vivo antioxidant activity of methanol extract from quinoa fermented with Rhizopus oligosporus. J. Nutr. Sci. Vitaminol. 2005, 51, 449–452. [Google Scholar] [CrossRef]
- Apostolidis, E.; Kwon, Y.I.; Ghaedian, R.; Shetty, K. Fermentation of milk and soymilk by Lactobacillus bulgaricus and Lactobacillus acidophilus enhances functionality for potential dietary management of hyperglycemia and hypertension. Food Biotechnol. 2007, 21, 217–236. [Google Scholar] [CrossRef]
- Niu, L.Y.; Jiang, S.T.; Pan, L.J. Preparation and evaluation of antioxidant activities of peptides obtained from defatted wheat germ by fermentation. J. Food Sci. Technol. 2013, 50, 53–61. [Google Scholar] [CrossRef]
- Watanabe, N.; Fujimoto, K.; Aoki, H. Antioxidant activities of the water-soluble fraction in tempeh-like fermented soybean (GABA-tempeh). Int. J. Food Sci. Nutr. 2007, 58, 577–587. [Google Scholar] [CrossRef]
- Zhu, Y.P.; Fan, J.F.; Cheng, Y.Q.; Li, L.T. Improvement of the antioxidant activity of Chinese traditional fermented okara (Meitauza) using Bacillus subtilis B2. Food Control 2008, 19, 654–661. [Google Scholar] [CrossRef]
- Dei Più, L.; Tassoni, A.; Serrazanetti, D.I.; Ferri, M.; Babini, E.; Tagliazucchi, D.; Gianotti, A. Exploitation of starch industry liquid by-product to produce bioactive peptides from rice hydrolyzed proteins. Food Chem. 2014, 155, 199–206. [Google Scholar] [CrossRef]
- Yike, I. Fungal proteases and their pathophysiological effects. Mycopathologia 2011, 171, 299–323. [Google Scholar] [CrossRef]
- Lee, J.S.; Rho, S.J.; Kim, Y.W.; Lee, K.W.; Lee, H.G. Evaluation of biological activities of the short-term fermented soybean extract. Food Sci. Biotechnol. 2013, 22, 973–978. [Google Scholar] [CrossRef]
- Amadou, I.; Gbadamosi, O.S.; Shi, Y.; Kamara, M.T.; Jin, S. Identification of antioxidative peptides from Lactobacillus plantarum Lp6 fermented soybean protein meal. Res. J. Microbiol. 2010, 5, 372–380. [Google Scholar] [CrossRef]
- Gibbs, B.F.; Zougman, A.; Masse, R.; Mulligan, C. Production and characterization of bioactive peptides from soy hydrolysate and soy-fermented food. Food Res. Int. 2004, 37, 123–131. [Google Scholar] [CrossRef]
- Wang, D.; Wang, L.J.; Zhu, F.X.; Zhu, J.Y.; Chen, X.D.; Zou, L.; Saito, M. In vitro and in vivo studies on the antioxidant activities of the aqueous extracts of Douchi (a traditional Chinese salt-fermented soybean food). Food Chem. 2008, 107, 1421–1428. [Google Scholar] [CrossRef]
- Tamam, B.; Syah, D.; Suhartono, M.T.; Kusuma, W.A.; Tachibana, S.; Lioe, H.N. Proteomic study of bioactive peptides from tempeh. J. Biosci. Bioeng. 2019, in press. [Google Scholar] [CrossRef]
- Starzyńska-Janiszewska, A.; Stodolak, B.; Gómez-Caravaca, A.M.; Mickowska, B.; Martin-Garcia, B.; Byczyński, Ł. Mould starter selection for extended solid-state fermentation of quinoa. LWT-Food Sci. Technol. 2019, 99, 231–237. [Google Scholar] [CrossRef]
- Limón, R.I.; Peñas, E.; Torino, M.I.; Martínez-Villaluenga, C.; Dueñas, M.; Frias, J. Fermentation enhances the content of bioactive compounds in kidney bean extracts. Food Chem. 2015, 172, 343–352. [Google Scholar] [CrossRef]
- Zhang, Z.; Lei, Z.; Yun, L.; Zhongzhi, L.; Chen, Y. Chemical composition and bioactivity changes in stale rice after fermentation with Cordyceps sinensis. J. Biosci. Bioeng. 2008, 106, 188–193. [Google Scholar] [CrossRef]
- Ogbonna, J.C. Microbiological production of tocopherols: Current state and prospects. Appl. Microbiol. Biotechnol. 2009, 84, 217–225. [Google Scholar] [CrossRef]
- Wang, Y.C.; Yu, R.C.; Chou, C.C. Antioxidative activities of soymilk fermented with lactic acid bacteria and bifidobacteria. Food Microbiol. 2006, 23, 128–135. [Google Scholar] [CrossRef]
- Frias, J.; Miranda, M.L.; Doblado, R.; Vidal-Valverde, C. Effect of germination and fermentation on the antioxidant vitamin content and antioxidant capacity of Lupinus albus L. var. Multolupa. Food Chem. 2005, 92, 211–220. [Google Scholar] [CrossRef]
- Małgorzata, W.; Joanna, H.; Konrad, P.M. Effect of solid-state fermentation with Rhizopus oligosporus on bioactive compounds and antioxidant capacity of raw and roasted buckwheat groats. It. J. Food Sci. 2015, 27, 424–431. [Google Scholar]
- Chou, S.T.; Chung, Y.C.; Peng, H.Y.; Hsu, C.K. Improving antioxidant status in aged mice by 50% ethanol extract from red bean fermented by Bacillus subtilis. J. Sci. Food Agric. 2013, 93, 2562–2567. [Google Scholar] [CrossRef]
- Gianotti, A.; Danesi, F.; Verardo, V.; Serrazanetti, D.I.; Valli, V.; Russo, A.; Riciputi, Y.; Tossani, N.; Caboni, M.F.; Guerzoni, M.E.; et al. Role of cereal type and processing in whole grain in vivo protection from oxidative stress. Front. Biosci. 2011, 16, 1609–1618. [Google Scholar] [CrossRef]
- Uchida, M.; Ishii, I.; Inoue, C.; Akisato, Y.; Watanabe, K.; Hosoyama, S.; Toida, T.; Ariyoshi, N.; Kitada, M. Kefiran reduces atherosclerosis in rabbits fed a high cholesterol diet. J. Atheroscler. Throm. 2010, 1006100247. [Google Scholar] [CrossRef]
- Li, Y.T.; Meng, S.L.; Wang, L.B.; Wang, Y.P.; Zu, X.Y.; Yang, Y.N.; Zhang, Z.Y. Antioxidative activity of exopolysaccharide extract from fermented wheat distillers. Adv. J. Food Sci. Technol. 2014, 6, 1067–1075. [Google Scholar] [CrossRef]
- Pozzo, L.; Vizzarri, F.; Ciardi, M.; Nardoia, M.; Palazzo, M.; Casamassima, D.; Longo, V. The effects of fermented wheat powder (Lisosan G) on the blood lipids and oxidative status of healthy rabbits. Food Chem. Toxicol. 2015, 84, 1–7. [Google Scholar] [CrossRef]
- Chung, Y.C.; Chang, C.T.; Chao, W.W.; Lin, C.F.; Chou, S.T. Antioxidative activity and safety of the 50 ethanolic extract from red bean fermented by Bacillus subtilis IMR-NK1. J. Agric. Food Chem. 2002, 50, 2454–2458. [Google Scholar] [CrossRef]
- La Marca, M.; Beffy, P.; Pugliese, A.; Longo, V. Fermented wheat powder induces the antioxidant and detoxifying system in primary rat hepatocytes. PLoS ONE 2013, 8, e83538. [Google Scholar] [CrossRef]
- Donot, F.; Fontana, A.; Baccou, J.C.; Schorr-Galindo, S. Microbial exopolysaccharides: Main examples of synthesis, excretion, genetics and extraction. Carbohydr. Polym. 2012, 87, 951–962. [Google Scholar] [CrossRef]
- Welman, A.D.; Maddox, I.S. Exopolysaccharides from lactic acid bacteria: Perspectives and challenges. Trends Biotechnol. 2003, 21, 269–274. [Google Scholar] [CrossRef]
- Wang, J.; Hu, S.; Nie, S.; Yu, Q.; Xie, M. Reviews on mechanisms of in vitro antioxidant activity of polysaccharides. Oxid. Med. Cell. Longev. 2016. [Google Scholar] [CrossRef]
- Adesulu-Dahunsi, A.T.; Jeyaram, K.; Sanni, A.I.; Banwo, K. Production of exopolysaccharide by strains of Lactobacillus plantarum YO175 and OF101 isolated from traditional fermented cereal beverage. PeerJ 2018, 6, e5326. [Google Scholar] [CrossRef]
- Liu, C.F.; Tseng, K.C.; Chiang, S.S.; Lee, B.H.; Hsu, W.H.; Pan, T.M. Immunomodulatory and antioxidant potential of Lactobacillus exopolysaccharides. J. Sci. Food Agric. 2011, 91, 2284–2291. [Google Scholar] [CrossRef]
- Patten, D.A.; Laws, A.P. Lactobacillus-produced exopolysaccharides and their potential health benefits: A review. Benef. Microbes 2015, 6, 457–471. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, C.; Li, D.; Zhao, Y.; Zhang, X.; Zeng, X.; Yang, Z.; Li, S. Antioxidant activity of an exopolysaccharide isolated from Lactobacillus plantarum C88. Int. J. Biol. Macromol. 2013, 54, 270–275. [Google Scholar] [CrossRef]
- Guo, Y.; Pan, D.; Li, H.; Sun, Y.; Zeng, X.; Yan, B. Antioxidant and immunomodulatory activity of selenium exopolysaccharide produced by Lactococcus lactis subsp. lactis. Food Chem. 2013, 138, 84–89. [Google Scholar] [CrossRef]
- Pan, D.; Mei, X. Antioxidant activity of an exopolysaccharide purified from Lactococcus lactis subsp. lactis 12. Carbohydr. Polym. 2010, 80, 908–914. [Google Scholar] [CrossRef]
- Leung, P.H.; Zhao, S.; Ho, K.P.; Wu, J.Y. Chemical properties and antioxidant activity of exopolysaccharides from mycelial culture of Cordyceps sinensis fungus Cs-HK1. Food Chem. 2009, 114, 1251–1256. [Google Scholar] [CrossRef]
- Yan, J.K.; Li, L.; Wang, Z.M.; Leung, P.H.; Wang, W.Q.; Wu, J.Y. Acidic degradation and enhanced antioxidant activities of exopolysaccharides from Cordyceps sinensis mycelial culture. Food Chem. 2009, 117, 641–646. [Google Scholar] [CrossRef]
- Chen, Y.; Mao, W.; Tao, H.; Zhu, W.; Qi, X.; Chen, Y.; Li, H.; Zhao, C.; Yang, Y.; Hou, Y.; et al. Structural characterization and antioxidant properties of an exopolysaccharide produced by the mangrove endophytic fungus Aspergillus sp. Y16. Biores. Technol. 2011, 102, 8179–8184. [Google Scholar] [CrossRef]
| Matrix | Microorganisms Employed | Process Parameters | Effect | Reference |
|---|---|---|---|---|
| Wheat bran | Baker’s yeast | 20 °C for 20 h | Release of phenolic acids and improved bioaccessibility and colonic metabolism of phenolic acids | [38] |
| Spontaneous fermentation conducted mainly by Lactobacillus, Leuconostoc and Pediococcus spp. | Backslopping for 13 days at 18 °C | Release of ferulic acid | [39] | |
| Baker’s yeast and LAB | 20 °C for 24 h in anaerobic condition | Conversion of ferulic and caffeic acids into their derivatives and increase in sinapic acid. | [40] | |
| Hericium erinaceus and enzymes | 25 °C for 7 days | Release of ferulic acid | [41] | |
| Kamut bread | Baker’s yeast and spontaneously fermented sourdough | 30 °C for 1.5 h | Response to oxidative stress in in vivo studies with rats. | [42] |
| Rice bran | Rhizopus oryzae CTT 1217 | 30 °C for 120 h | Increase ferulic acid and DPPH scavenging activity. Inhibition of peroxidase and polyphenol oxidase. | [43] |
| Rhizopus oligosporus F0020 Monascus purpureus F0061 | 32 °C for 12 days | Release of phenolic acids and increase of FRAP | [44] | |
| Bacillus subtilis subsp. subtilis NRRL NRS-744 | 37 °C for 8 days | Release of phenolic acids | [45] | |
| Rye bran | Baker’s yeast | 25 °C for 14 h | Increase of phenolic acids | [46] |
| Baker’s yeast | 20 °C for 24 h | Release of phenolic acids but no improved bioaccessibility in in vitro digestive systems | [47,48] | |
| Sorghum flour | Lactobacillus plantarum FUA3171, Lactobacillus casei FUA3166, Lactobacillus fermentum FUA3165 Lactobacillus reuteri FUA3168 | 34 °C for 24 h | Release of phenolic acids and flavonoids | [49] |
| Tef pancake | Spontaneously fermented sourdough | 25 °C for 24-120 h | Solubilization of bound phenolics and improved antioxidant potentials on FRAP and ABTS | [50] |
| Malt based beverage | Lb. plantarum Lp758, Lp765, Lp725, Lactobacillus brevis Lb986, Lactobacillus amylolyticus LaTL3, LaT15 | 30 °C for 72 h | Decarboxylation of phenolic acids. | [51] |
| Soy | Bacillus subtilis SHZ | 30 °C for 36 h | Superoxide radical scavenging activity and reducing power potential | [52] |
| B. subtilis MTCC5480, MTCC1747 | 42 °C for 24 h | Increase in the antioxidant activity due to both phenolic compounds and peptides | [53] | |
| Aspergillus oryzae ATCC 1011 Rhizopus oryzae ATCC 24563 Bacillus subtilis ATCC 6051 | 30 °C for 48 h | Increase in phenolic acids and flavonoids | [54] | |
| Bacillus pumilus HY1 | 37 °C for 60 h | Increase in phenolic acids, flavonoids and tannins monomeric forms | [55] | |
| Aspergillus awamori Aspergillus niger Aspergillus niveus | 30 °C for 5 days | Increase in phenolic acids and flavonoids as consequence of β-glucosidase activity | [56] | |
| R. oligosporus | Room temperature for 20 days | Increase in phenolic acids and flavonoids | [57] | |
| Lentinus edodes CY-35 | Room temperature for 50 days | Increase in phenolic acids and flavonoids as consequence of laccase and β-glucosidase activities | [58] | |
| Bacillus natto | 37 °C for 48 h | Increase in phenolic acids and flavonoids | [59] | |
| Lactobacillus plantarum CECT 748 T | 30 °C for 48 h | Increase in phenolic acids and flavonoids | [60] | |
| Pool of selected LAB | 37 °C for 48 h | Increase in phenolic acids, flavonoids, saponins, phytosterols, and tocopherols | [61] | |
| Lactobacillus casei Lactobacillus acidophilus | 32 °C for 15 h | Increased of the aglycones/glycosylated isoflavones ratio and bound phenolics | [62] | |
| Lactobacillus casei 0979 after germination | 30 °C for 24 h | Increase in isoflavones glycosides and aglycones | [63] | |
| Kefir grains containing LAB and yeasts | 30 °C for 24 h | Increase of isoflavones and improved antioxidant activities on DPPH and ABTS | [64] | |
| Lb. plantarum DPPMA24W, DPPMASL33 Lb. fermentum DPPMA114 Lactobacillus rhamnosus DPPMAAZ1 | 30 °C for 96 h | Increase of isoflavone aglycones especially equol | [65] | |
| Cowpeas | Spontaneously fermented Lb. plantarum ATCC 14917 | 37 °C for 48 h | Increase of phenolic acids derivatives and flavonoids. Improved antioxidant activity on DPPH | [66] |
| Lentils | Lb. plantarum CECT 748 and commercial protease | 37 °C for 15 h | Reduction of ROS on RAW 264.7 cells | [67] |
| Spontaneously fermented | 35 °C for 4 days | Decrease of condensed tannin and increase of monomers. | [68] |
| Matrix | Microorganisms Employed | Process Parameters | Effect | Reference |
|---|---|---|---|---|
| Wheat flour | Lactobacillus farciminis A11, A19, H3, Lactobacillus rossiae A20, Gd40, Lactobacillus sanfranciscensis B3, I4, Lactobacillus plantarum O4, Lactobacillus brevis A7 | 30 °C for 24 h | Reduction of ROS on RAW 264.7, H-end and Caco-2 cells | [93] |
| Defatted wheat germ | Bacillus subtilis B1 | 37 °C for 24 h | Unidentified peptides | [96] |
| Rice protein | Bacillus pumilus AG1 | 37 °C for 72 h | Identified peptide sequences with high antioxidant activity | [99] |
| Wheat, spelt, rye, and kamut flours | Pool of selected LAB | 37 °C for 24 h | Identified peptide sequences with high antioxidant activity | [32] |
| Quinoa | Rhizopus oligosporus NRRL2710 | 36 °C for 24 h | Increased ex vivo and in vivo activities of superoxide dismutase, GSHPx, and TBARS | [94] |
| L. plantarum T0A10 | 37 °C for 24 h | Identified peptides with antioxidant activity on human keratinocytes NCTC 2544 | [33] | |
| R. oligosporus ATCC 64063 Aspergillus oryzae DSM 1861 Neurospora intermedia DSM 1965 | 31 °C for 6 days 25 °C for 6 days 30 °C for 5 days | Increase of OH and ABTS radical scavenging activity due to potentially bioactive peptides | [107] | |
| Soy | Spontaneously sourdough containing Rhizopus spp., LAB and yeasts | Room temperature for 2 days | Identified peptide sequences with potential antioxidant activity | [106] |
| B. subtilis SHZ | 30 °C for 36 h | Superoxide radical scavenging activity and reducing power potential | [52] | |
| B. subtilis MTCC5480, MTCC1747 | 42 °C for 24 h | Increase in the antioxidant activity due to both phenolic compounds and peptides | [53] | |
| Lb, plantarum Lp6 | 30 °C for 24 h | Identified peptide sequences with high antioxidant activity | [102] | |
| Aspergillus orizae | 30 °C for 3 days 45 °C for 4 days | Unidentified low molecular weight peptides | [100] | |
| Rhizopus microsporus | 36 °C for 25 h | Improvement in the antioxidant activity attributed to amino acids and peptides | [97] | |
| Bifidobacterium sp. | 37 °C for 48 h | Inhibition of ascorbate autoxidation, superoxide radical scavenging activity and reducing power potential peroxide | [105] | |
| Aspergillus oryzae | 30 °C for 60 h | Superoxide dismutase and glutathione peroxidase activities in vivo | [104] | |
| Kidney beans | B. subtilis CECT 39T Lb. plantarum CECT 748T | 30 °C for 96 h 37 °C for 96 h | Improved antioxidant activity on ORAC-FL | [108] |
| Lentils | Lb. plantarum CECT 748 and commercial protease | 37 °C for 15 h | Reduction of ROS on RAW 264.7 cells | [67] |
| Matrix | Microorganisms Employed | Process Parameters | Effect | Reference |
|---|---|---|---|---|
| Stale rice | Cordyceps sinensis | 25 °C for 7 days | Increase of tocopherol and superoxide dismutase activity in mice | [109] |
| Rice medium | Lactobacillus kefiranofaciens | EPS responsible of atherosclerosis prevention | [115] | |
| Wheat distillers’ grains | Preussia aemulans | Room temperature for 7 days | EPS formation with high radical scavenging activity and metal ions chelating ability | [116] |
| Wheat germ and bran | Yeasts and lactobacilli | High vitamin content responsible for in vivo antioxidant activity in liver and kidney | [117] | |
| Kidney beans | Bacillus subtilis IMR-NK1 | 30 °C for 48 h | Improved antioxidant activity on DPPH, reducing power potential and Fe2+ chelating ability | [118] |
| Bacillus subtilis BCRC 14716 | 30 °C for 48 h | Increased vitamin E levels in liver and brain of rabbit, and superoxide dismutase activity in the brain | [113] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Verni, M.; Verardo, V.; Rizzello, C.G. How Fermentation Affects the Antioxidant Properties of Cereals and Legumes. Foods 2019, 8, 362. https://doi.org/10.3390/foods8090362
Verni M, Verardo V, Rizzello CG. How Fermentation Affects the Antioxidant Properties of Cereals and Legumes. Foods. 2019; 8(9):362. https://doi.org/10.3390/foods8090362
Chicago/Turabian StyleVerni, Michela, Vito Verardo, and Carlo Giuseppe Rizzello. 2019. "How Fermentation Affects the Antioxidant Properties of Cereals and Legumes" Foods 8, no. 9: 362. https://doi.org/10.3390/foods8090362
APA StyleVerni, M., Verardo, V., & Rizzello, C. G. (2019). How Fermentation Affects the Antioxidant Properties of Cereals and Legumes. Foods, 8(9), 362. https://doi.org/10.3390/foods8090362








