Inter-Varietal Diversity of Typical Volatile and Phenolic Profiles of Croatian Extra Virgin Olive Oils as Revealed by GC-IT-MS and UPLC-DAD Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. EVOO Samples
2.2. Sensory Analysis
2.3. Chemical Standards and Standard Solutions
2.4. Analysis of Volatile Compounds by GC-IT-MS
2.5. Analysis of Phenols by UPLC-DAD
2.6. Statistical Data Elaboration
3. Results
3.1. Volatile Aroma Compound Profiles
3.1.1. Hydrocarbons
3.1.2. Monoterpenes and Sesquiterpenes
3.1.3. Aldehydes
3.1.4. Ketones
3.1.5. Alcohols
3.1.6. Acids
3.1.7. Esters
3.1.8. Benzenoids
3.1.9. Furanoids
3.1.10. Miscellaneous Compounds
3.1.11. Odor Activity Values (OAV)
3.1.12. Multivariate Statistical Analysis
3.2. Phenols
3.3. Sensory Attributes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- European Economic Community. Commission Regulation (EEC) No 2568/91 of 11 July 1991 (and later modifications) on the characteristics of olive oil and olive-residue oil and the relevant methods of analysis. Off. J. Eur. Communities 1991, L248, 1–83. [Google Scholar]
- European Union. Regulation (EU) No 1151/2012 of the European parliament and of the Council of 21 November 2012 (and later modifications) on quality schemes for agricultural products and foodstuffs. Off. J. Eur. Union 2012, L343, 1–29. [Google Scholar]
- International Olive Council. COI/T.20/Doc. No. 22 Method for the Organoleptic Assessment of Extra Virgin Olive Oil Applying to Use a Designation of Origin; International Olive Oil Council: Madrid, Spain, 2005. [Google Scholar]
- Daisa, P.; Hatzakis, E. Quality assessment and authentication of virgin olive oil by NMR spectroscopy: A critical review. Anal. Chim. Acta 2013, 765, 1–27. [Google Scholar] [CrossRef] [PubMed]
- Aparicio, R.; Morales, M.T.; Aparicio-Ruiz, R.; Tena, N.; García-González, D.L. Authenticity of olive oil: Mapping and comparing official methods and promising alternatives. Food Res. Int. 2013, 54, 2025–2038. [Google Scholar] [CrossRef]
- López-Cortés, I.; Salazar-García, D.C.; Velázquez-Martí, B.; Salazar, D.M. Chemical characterization of traditional varietal olive oils in East of Spain. Food Res. Int. 2013, 54, 1934–1940. [Google Scholar] [CrossRef][Green Version]
- Olmo-García, L.; Polari, J.J.; Li, X.; Bajoub, A.; Fernández-Gutiérrez, A.; Wang, S.C.; Carrasco-Pancorbo, A. Study of the minor fraction of virgin olive oil by a multi-class GC–MS approach: Comprehensive quantitative characterization and varietal discrimination potential. Food Res. Int. 2019, 125, 108649. [Google Scholar] [CrossRef]
- Kritioti, A.; Menexes, G.; Drouza, C. Chemometric characterization of virgin olive oils of the two major Cypriot cultivars based on their fatty acid composition. Food Res. Int. 2018, 103, 426–437. [Google Scholar] [CrossRef]
- Angerosa, F. Virgin olive oil odour notes: Their relationships with volatile compounds from the lipoxygenase pathway and secoiridoid compounds. Food Chem. 2000, 68, 283–287. [Google Scholar] [CrossRef]
- Angerosa, F. Influence of volatile compounds on virgin olive oil quality evaluated by analytical approaches and sensor panels. Eur. J. Lipid Sci. Technol. 2002, 104, 639–660. [Google Scholar] [CrossRef]
- Angerosa, F.; Campestre, C. Sensory quality: Methodologies and applications. In Handbook of Olive Oil—Analysis and Properties; Aparicio, R., Harwood, J., Eds.; Springer: New York, NY, USA, 2013; pp. 523–560. [Google Scholar]
- Campestre, C.; Angelini, G.; Gasbarri, C.; Angerosa, F. The compounds responsible for the sensory profile in monovarietal virgin olive oils. Molecules 2017, 22, 1833. [Google Scholar] [CrossRef]
- Servili, M.; Montedoro, G. Contribution of phenolic compounds to virgin olive oil quality. Eur. J. Lipid Sci. Technol. 2002, 104, 602–613. [Google Scholar] [CrossRef]
- Taticchi, A.; Esposto, S.; Servili, M. The basis of the sensory properties of virgin olive oil. In Olive Oil Sensory Science; Monteleone, E., Langstaff, S., Eds.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2014; pp. 33–54. [Google Scholar]
- Blasi, F.; Pollini, L.; Cossignani, L. Varietal authentication of extra virgin olive oils by triacylglycerols and volatiles analysis. Foods 2019, 8, 58. [Google Scholar] [CrossRef] [PubMed]
- Cecchi, T.; Alfei, B. Volatile profiles of Italian monovarietal extra virgin olive oils via HS-SPME–GC–MS: Newly identified compounds, flavors molecular markers, and terpenic profile. Food Chem. 2013, 141, 2025–2035. [Google Scholar] [CrossRef] [PubMed]
- García-González, D.L.; Romero, N.; Aparicio, R. Comparative study of virgin olive oil quality from single varieties cultivated in Chile and Spain. J. Agric. Food Chem. 2010, 58, 12899–12905. [Google Scholar] [CrossRef] [PubMed]
- Ocakoglu, D.; Tokatli, F.; Ozen, B.; Korel, F. Distribution of simple phenols, phenolic acids and flavonoids in Turkish monovarietal extra virgin olive oils for two harvest years. Food Chem. 2009, 113, 401–410. [Google Scholar] [CrossRef]
- Piscopo, A.; De Bruno, A.; Zappia, A.; Ventre, C.; Poiana, M. Characterization of monovarietal olive oils obtained from mills of Calabria region (Southern Italy). Food Chem. 2016, 213, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Sagratini, G.; Maggi, F.; Caprioli, G.; Cristalli, G.; Ricciutelli, M.; Torregiani, E.; Vittori, S. Comparative study of aroma profile and phenolic content of Montepulciano monovarietal red wines from the Marches and Abruzzo regions of Italy using HS-SPME–GC–MS and HPLC–MS. Food Chem. 2012, 132, 1592–1599. [Google Scholar] [CrossRef] [PubMed]
- Angerosa, F.; Servili, M.; Selvaggini, R.; Taticchi, A.; Esposto, S.; Montedoro, G. Volatile compounds in virgin olive oil: Occurrence and their relationship with the quality. J. Chromatogr. A 2004, 1054, 17–31. [Google Scholar] [CrossRef]
- Kalua, C.M.; Allen, M.S.; Bedgood, D.R.; Bishop, A.G.; Prenzler, P.D.; Robards, K. Olive oil volatile compounds, flavour development and quality: A critical review. Food Chem. 2007, 100, 273–286. [Google Scholar] [CrossRef]
- Vichi, S.; Pizzale, L.; Conte, L.S.; Buxaderas, S.; Lόpez-Tamames, E. Solid-Phase microextraction in the analysis of virgin olive oil volatile fraction: Characterization of virgin olive oils from two distinct geographical areas of Northern Italy. J. Agric. Food Chem. 2003, 51, 6572–6577. [Google Scholar] [CrossRef]
- Andrewes, P.; Busch, J.L.H.C.; De Joode, T.; Groenewegen, A.; Alexandre, H. Sensory properties of virgin olive oil polyphenols: Identification of deacetoxy-ligstroside aglycon as a key contributor to pungency. J. Agric. Food Chem. 2003, 51, 1415–1420. [Google Scholar] [CrossRef] [PubMed]
- Bendini, A.; Cerretani, L.; Carrasco-Pancorbo, A.; Gómez-Caravaca, A.M.; Segura-Carretero, A.; Fernández-Gutiérrez, A.; Lercker, G. Phenolic molecules in virgin olive oils: A survey of their sensory properties, health effects, antioxidant activity and analytical methods. An overview of the last decade. Molecules 2007, 12, 1679–1719. [Google Scholar] [CrossRef] [PubMed]
- Amirante, P.; Clodoveo, M.L.; Tamborrino, A.; Leone, A.; Dugo, G. Oxygen concentration control during olive oil extraction process: A new system to emphasize the organoleptic and healthy properties of virgin olive oil. Acta Hortic. 2012, 949, 473–480. [Google Scholar] [CrossRef]
- Taticchi, A.; Esposto, S.; Veneziani, G.; Urbani, S.; Selvaggini, R.; Servili, M. The influence of the malaxation temperature on the activity of polyphenoloxidase and peroxidase and on the phenolic composition of virgin olive oil. Food Chem. 2013, 136, 975–983. [Google Scholar] [CrossRef] [PubMed]
- Angerosa, F.; Basti, C.; Vito, R. Virgin olive oil volatile compounds from lipoxygenase pathway and characterization of some Italian cultivars. J. Agric. Food Chem. 1999, 47, 836–839. [Google Scholar] [CrossRef] [PubMed]
- Cirilli, M.; Caruso, G.; Gennai, C.; Urbani, S.; Frioni, E.; Ruzzi, M.; Servili, M.; Gucci, R.; Poerio, E.; Muleo, R. The role of polyphenoloxidase, peroxidase, and β-glucosidase in phenolics accumulation in Olea europaea L. fruits under different water regimes. Front. Plant Sci. 2017, 8, 717. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Agriculture, Republic of Croatia. Available online: https://poljoprivreda.gov.hr/istaknute-teme/hrana-111/oznake-kvalitete/zoi-zozp-zts-poljoprivrednih-i-prehrambenih-proizvoda/zasticena-oznaka-izvornosti-zoi/1206 (accessed on 9 October 2019).
- International Olive Council, Madrid, Spain. Available online: http://www.internationaloliveoil.org/estaticos/view/131-world-olive-oil-figures (accessed on 9 October 2019).
- Poljuha, D.; Sladonja, B.; Šetić, E.; Milotić, A.; Bandelj, D.; Jakše, J.; Javornik, B. DNA fingerprinting of olive varieties in Istria (Croatia) by microsatellite markers. Sci. Hortic. 2008, 115, 223–230. [Google Scholar] [CrossRef]
- Brkić Bubola, K.; Koprivnjak, O.; Sladonja, B.; Lukić, I. Volatile compounds and sensory profiles of monovarietal virgin olive oils from Buža, Črna and Rosinjola cultivars in Istria (Croatia). Food Technol. Biotechnol. 2012, 50, 192–198. [Google Scholar]
- Giacometti, J.; Milin, Č.; Giacometti, F.; Ciganj, Z. Characterisation of monovarietal olive oils obtained from Croatian cvs. Drobnica and Buža during the ripening period. Foods 2018, 7, 188. [Google Scholar] [CrossRef]
- Kulišić Bilušić, T.; Melliou, E.; Giacometti, J.; Čaušević, A.; Čorbo, S.; Landeka, M.; Magiatis, P. Phenolics, fatty acids, and biological potential of selected Croatian EVOOs. Eur. J. Lipid Sci. Technol. 2017, 119, 1700108. [Google Scholar] [CrossRef]
- Lukić, I.; Carlin, S.; Horvat, I.; Vrhovsek, U. Combined targeted and untargeted profiling of volatile aroma compounds with comprehensive two-dimensional gas chromatography for differentiation of virgin olive oils according to variety and geographical origin. Food Chem. 2019, 270, 403–414. [Google Scholar] [CrossRef] [PubMed]
- Lukić, I.; Krapac, M.; Lukić, M.; Vrhovsek, U.; Godena, S.; Brkić Bubola, K. Towards understanding the varietal typicity of virgin olive oil by correlating sensory and compositional analysis data: A case study. Food Res. Int. 2018, 112, 78–89. [Google Scholar] [CrossRef] [PubMed]
- Šarolić, M.; Gugić, M.; Friganović, E.; Tuberoso, C.I.G.; Jerković, I. Phytochemicals and other characteristics of Croatian monovarietal extra virgin olive oils from Oblica, Lastovka and Levantinka varieties. Molecules 2015, 20, 4395–4409. [Google Scholar] [CrossRef] [PubMed]
- Žanetić, M.; Cerretani, L.; Škevin, D.; Politeo, O.; Vitanović, E.; Jukić Špika, M.; Perica, S.; Ožić, M. Influence of polyphenolic compounds on the oxidative stability of virgin olive oils from selected autochthonous varieties. J. Food Agric. Environ. 2013, 11, 126–131. [Google Scholar]
- Žanetić, M.; Škevin, D.; Vitanović, E.; Jukić Špika, M.; Perica, S. Survey of phenolic compounds and sensorial profile of Dalmatian virgin olive oils. Pomol. Croat. 2011, 17, 19–30. [Google Scholar]
- Brkić Bubola, K.; Koprivnjak, O.; Sladonja, B.; Škevin, D.; Belobrajić, I. Chemical and sensorial changes of Croatian monovarietal olive oils during ripening. Eur. J. Lipid Sci. Technol. 2012, 114, 1400–1408. [Google Scholar] [CrossRef]
- Jerman Klen, T.; Golc Wondra, A.; Vrhovšek, U.; Mozetič Vodopivec, B. Phenolic profiling of olives and olive oil process-derived matrices using UPLC-DAD-ESI-QTOF-HRMS analysis. J. Agric. Food Chem. 2015, 63, 3859–3872. [Google Scholar] [CrossRef]
- Xia, J.; Sinelnikov, I.; Han, B.; Wishart, D.S. MetaboAnalyst 3.0—Making metabolomics more meaningful. Nucleic Acids Res. 2015, 43, W251–W257. [Google Scholar] [CrossRef]
- Aparicio, R.; Luna, G. Characterization of monovarietal virgin olive oils. Eur. J. Lipid Sci. Technol. 2002, 104, 614–627. [Google Scholar] [CrossRef]
- Morales, M.T.; Luna, G.; Aparicio, R. Comparative study of virgin olive oil sensory defects. Food Chem. 2005, 91, 293–301. [Google Scholar] [CrossRef]
- Reiners, J.; Grosch, W. Odorants of virgin olive oils with different flavor profiles. J. Agric. Food Chem. 1998, 46, 2754–2763. [Google Scholar] [CrossRef]
- Reboredo-Rodríguez, P.; González-Barreiro, C.; Cancho-Grande, B.; Simal-Gándara, J. Improvements in the malaxation process to enhance the aroma quality of extra virgin olive oils. Food Chem. 2014, 158, 534–545. [Google Scholar] [CrossRef] [PubMed]
- Inglese, P.; Famiani, F.; Galvano, F.; Servili, M.; Esposto, S.; Urbani, S. Factors affecting extra-virgin olive oil composition. In Horticultural Reviews; Janick, J., Ed.; John Wiley & Sons Inc.: Hoboken, NJ, USA, 2010; pp. 83–147. [Google Scholar]
- Romero, N.; Saavedra, J.; Tapia, F.; Sepúlveda, B.; Aparicio, R. Influence of agroclimatic parameters on phenolic and volatile compounds of Chilean virgin olive oils and characterization based on geographical origin, cultivar and ripening stage. J. Sci. Food Agric. 2016, 96, 583–592. [Google Scholar] [CrossRef] [PubMed]
- Beck, J.J.; Higbee, B.S.; Light, D.M.; Gee, W.S.; Merrill, G.B.; Hayashi, J.M. Hull split and damaged almond volatiles attract male and female navel orangeworm moths. J. Agric. Food Chem. 2012, 60, 8090–8096. [Google Scholar] [CrossRef] [PubMed]
- Lukić, I.; Krapac, M.; Horvat, I.; Godena, S.; Kosić, U.; Brkić Bubola, K. Three-factor approach for balancing the concentrations of phenols and volatiles in virgin olive oil from a late-ripening olive cultivar. LWT Food Sci. Technol. 2018, 87, 194–202. [Google Scholar] [CrossRef]
- Majetić Germek, V.; Koprivnjak, O.; Butinar, B.; Pizzale, L.; Bučar-Miklavčič, M.; Conte, L.S. Influence of phenols mass fraction in olive (Olea europaea L.) paste on volatile compounds in Buža cultivar virgin olive oil. J. Agric. Food Chem. 2013, 61, 5921–5927. [Google Scholar]
- Angerosa, F.; Mostallino, R.; Basti, C.; Vito, R. Influence of malaxation temperature and time on the quality of virgin olive oils. Food Chem. 2001, 72, 19–28. [Google Scholar] [CrossRef]
- Aparicio, R.; Morales, M.T. Characterization of olive ripeness by green aroma compounds of virgin olive oil. J. Agric. Food Chem. 1998, 46, 1116–1122. [Google Scholar] [CrossRef]
- Hongsoongnern, P.; Chambers, E. A lexicon for green odor or flavor and characteristics of chemicals associated with green. J. Sens. Stud. 2008, 23, 205–221. [Google Scholar] [CrossRef]
- Vichi, S.; Guadayol, J.M.; Caixach, J.; Lopez-Tamames, E.; Buxaderas, S. Monoterpene and sesquiterpene hydrocarbons of virgin olive oil by headspace solidphase microextraction coupled to gas chromatography/mass spectrometry. J. Chromatogr. A 2006, 1125, 117–123. [Google Scholar] [CrossRef]
- Williams, M.; Salas, J.J.; Sanchez, J.; Harwood, J.L. Lipoxygenase pathway in olive callus cultures (Olea europaea). Phytochemistry 2000, 53, 13–19. [Google Scholar] [CrossRef]
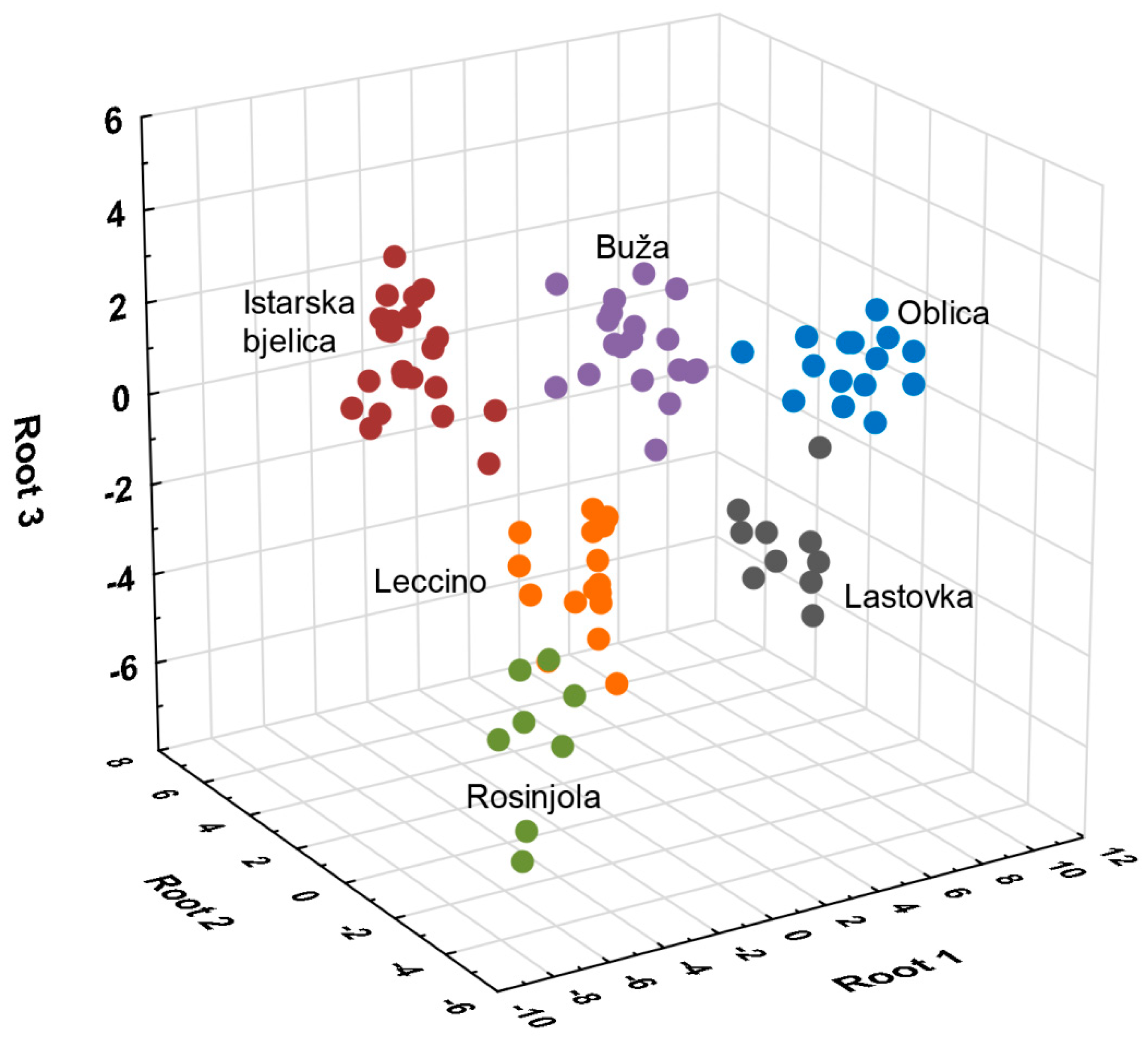
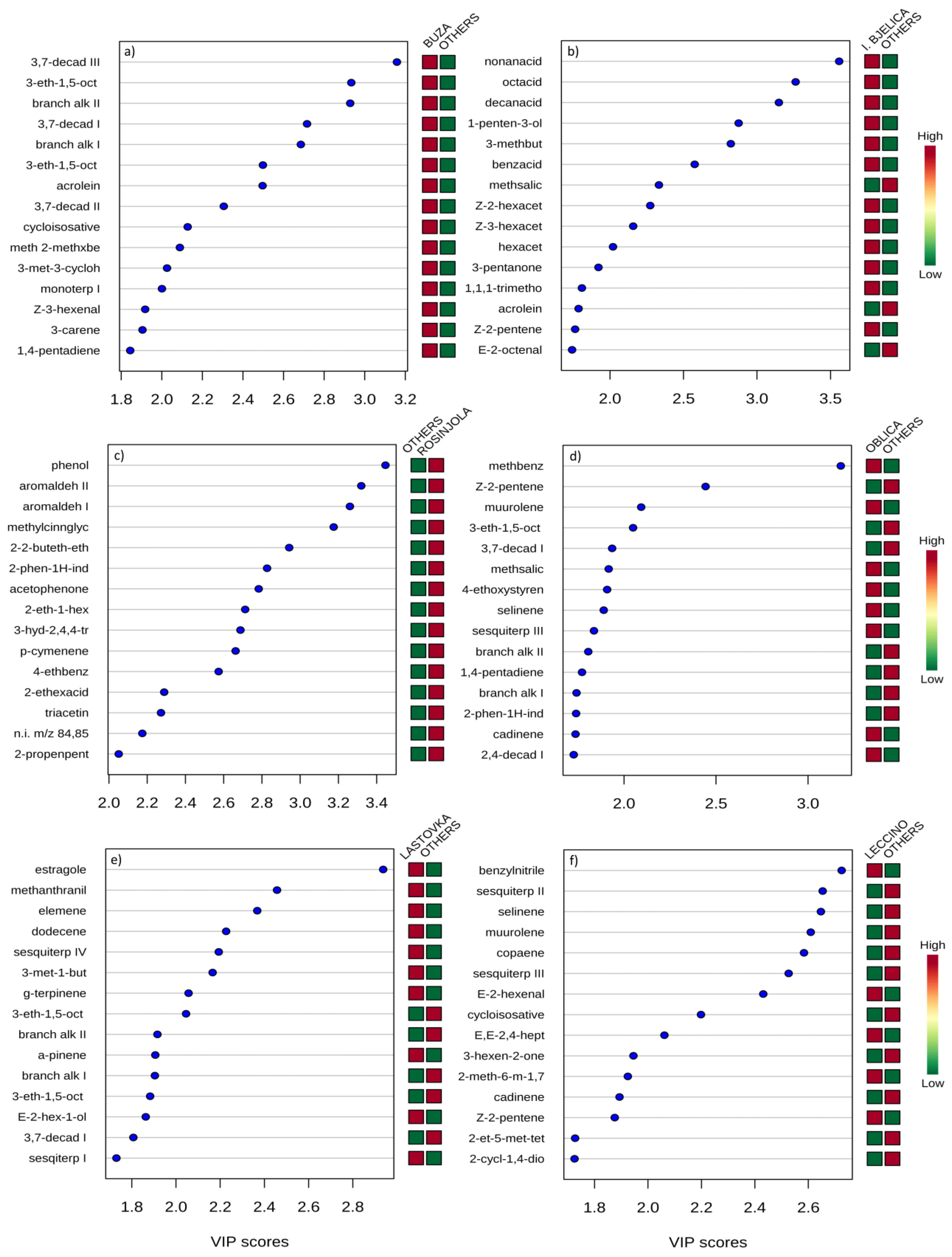
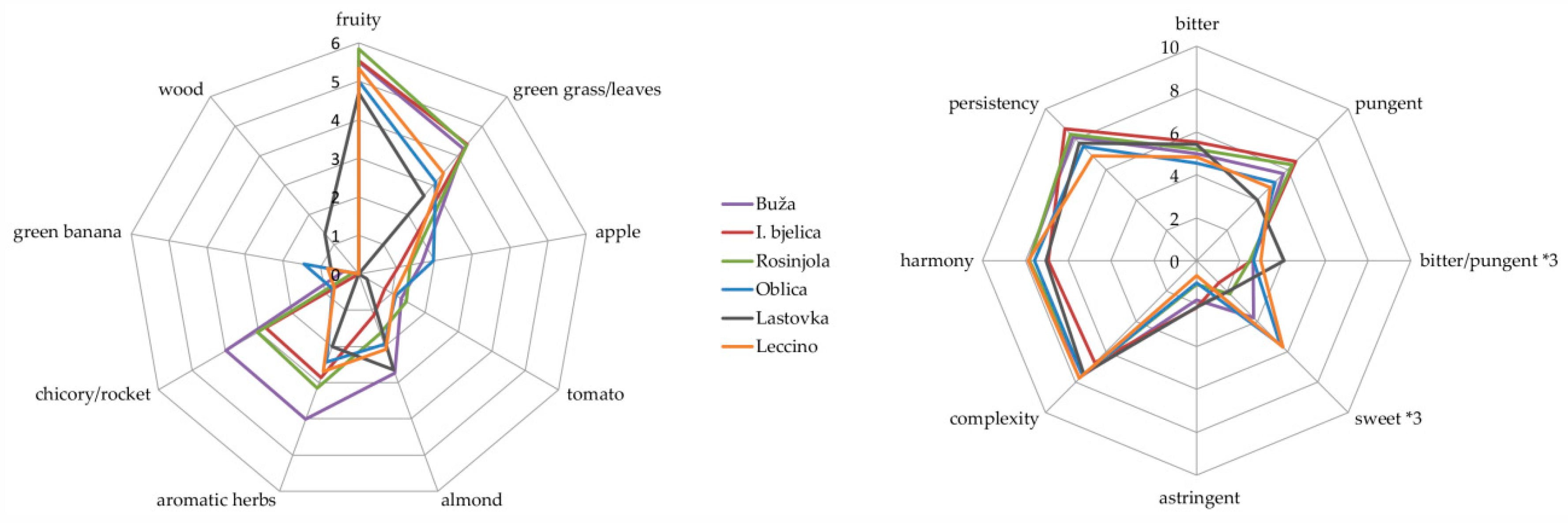
| Compound | ID | LRI | Variety | |||||
|---|---|---|---|---|---|---|---|---|
| Buža | I. bjelica | Rosinjola | Oblica | Lastovka | Leccino | |||
| hydrocarbons | ||||||||
| propane | MS | <700 | 6.64 bc | 14.91 a | 1.12 c | 11.49 ab | 12.51 a | 5.43 c |
| (Z)-2-pentene | MS | <700 | 61.19 b | 80.27 a | 60.31 b | 33.98 c | 43.63 c | 83.09 a |
| 1,4-pentadiene (mg/kg) | LRI,MS | <700 | 0.17 a | 0.16 a | 0.15 ab | 0.10 c | 0.09 c | 0.12 b |
| 1,3-pentadiene | LRI,MS | <700 | 81.73 a | 75.36 ab | 71.98 ab | 53.80 c | 48.98 c | 62.92 bc |
| 1-heptene | LRI,MS | 731 | 5.42 | 16.64 | 3.25 | 9.76 | 5.72 | 6.13 |
| 3,5-dimethylheptane | MS | 753 | 1.61 | 3.59 | 3.63 | 2.38 | 0.33 | 2.86 |
| propanal | LRI,MS | 778 | 7.00 | 5.23 | 7.36 | 5.10 | 5.68 | 7.04 |
| octane | S,LRI,MS | 800 | 31.03 | 35.33 | 27.29 | 30.78 | 40.92 | 27.11 |
| 2,6-dimethyl-3-heptene | MS | 821 | 0.20 b | 0.58 b | 6.17 a | 0.00 b | 0.00 b | 0.19 b |
| (E)-2-octene | LRI,MS | 832 | 3.50 | 4.60 | 3.81 | 3.36 | 5.62 | 3.60 |
| branched-chain alkene I (n.i.) | MS | 953 | 94.32 a | 63.73 c | 85.98 ab | 47.29 d | 38.96 d | 74.97 bc |
| branched-chain alkene II (n.i.) | MS | 960 | 84.13 a | 56.70 c | 71.63 b | 41.67 d | 35.22 d | 65.24 bc |
| decane | S,LRI,MS | 997 | 31.53 ab | 19.98 bc | 39.92 a | 9.49 c | 7.39 c | 14.67 c |
| 3-ethyl-1,5-octadiene I (mg/kg) | LRI,MS | 1006 | 0.36 a | 0.26 c | 0.34 ab | 0.18 d | 0.15 d | 0.31 b |
| 3-ethyl-1,5-octadiene II (mg/kg) | LRI,MS | 1020 | 0.40 a | 0.25 c | 0.35 ab | 0.20 d | 0.16 d | 0.31 b |
| 3-ethyl-1,5-octadiene III | LRI,MS | 1033 | 2.99 | 12.91 | 3.19 | 1.24 | 0.79 | 2.60 |
| branched-chain alkene III (n.i.) | MS | 1067 | 0.37 | 10.08 | 0.45 | 0.06 | 0.01 | 0.48 |
| 3,7-decadiene I | LRI,MS | 1074 | 76.81 a | 44.98 c | 71.69 ab | 33.06 d | 29.07 d | 65.14 b |
| branched-chain alkene IV (n.i.) | MS | 1077 | 3.59 | 6.51 | 1.87 | 1.62 | 0.72 | 2.92 |
| 3,7-decadiene II (mg/kg) | LRI,MS | 1080 | 0.29 a | 0.17 b | 0.28 a | 0.15 bc | 0.13 c | 0.26 a |
| 3,7-decadiene III (mg/kg) | LRI,MS | 1084 | 0.29 a | 0.15 c | 0.25 ab | 0.15 c | 0.11 c | 0.21 b |
| branched-chain alkene V (n.i.) | MS | 1087 | 7.98 ab | 11.87 a | 6.30 ab | 4.05 b | 3.00 b | 5.80 b |
| undecane | S,LRI,MS | 1095 | 18.56 ab | 9.75 b | 39.12 a | 3.29 b | 2.09 b | 9.83 b |
| dodecane | S,LRI,MS | 1203 | 23.4 ab | 14.16 bc | 35.51 a | 4.64 c | 2.64 c | 13.66 bc |
| dodecene | LRI,MS | 1241 | 27.26 bc | 33.95 b | 17.25 cd | 14.74 cd | 58.52 a | 8.43 d |
| 3-propylcyclohexene | MS | 1247 | 10.97 bc | 7.93 c | 14.15 ab | 13.41 ab | 9.90 bc | 14.85 a |
| 1,5,5,6-tetramethyl-1,3-cyclohexadiene | LRI,MS | 1365 | 2.46 bc | 1.55 c | 4.38 abc | 5.73 a | 5.52 ab | 1.82 c |
| 2,2-dimethyl-(Z)-3-hexene I | MS | 1499 | 28.41 a | 16.09 b | 33.60 a | 26.67 a | 14.60 b | 10.76 b |
| 2,2-dimethyl-(Z)-3-hexene II | MS | 1560 | 4.79 | 2.12 | 6.05 | 4.78 | 2.39 | 2.88 |
| monoterpenes | ||||||||
| α-pinene | S,LRI,MS | 1015 | 6.28 b | 4.91 b | 12.25 b | 8.90 b | 41.71 a | 2.79 b |
| camphene | S,LRI,MS | 1053 | 0.23 b | 0.10 b | 0.25 b | 0.31 b | 1.35 a | 0.04 b |
| β-pinene | S,LRI,MS | 1099 | 0.41 | 0.77 | 1.98 | 1.10 | 83.74 | 0.46 |
| sabinene | S,LRI,MS | 1112 | 2.07 | 0.79 | 2.09 | 2.94 | 49.44 | 17.20 |
| 3-carene | S,LRI,MS | 1139 | 4.72 a | 3.06 b | 4.20 ab | 0.97 c | 2.29 bc | 0.83 c |
| monoterpene I (n.i.) | MS | 1140 | 2.77 a | 1.84 b | 2.54 ab | 0.40 c | 1.35 bc | 0.27 c |
| myrcene | S,LRI,MS | 1157 | 8.60 b | 4.36 b | 19.76 ab | 11.48 b | 41.67 a | 11.85 b |
| α-terpinene | S,LRI,MS | 1171 | 0.37 | 0.23 | 1.12 | 0.72 | 10.84 | 9.91 |
| limonene (mg/kg) | S,LRI,MS | 1191 | 0.02 | 0.08 | 0.02 | 0.02 | 0.64 | 0.01 |
| β-phellandrene | LRI,MS | 1201 | 0.00 b | 0.00 b | 0.00 b | 0.03 b | 4.25 a | 0.02 b |
| (Z)-ocimene | S,LRI,MS | 1230 | 15.14 b | 8.00 b | 25.27 ab | 34.84 a | 37.26 a | 10.72 b |
| γ-terpinene | S,LRI,MS | 1238 | 30.50 b | 4.96 b | 4.17 b | 3.74 b | 436.30 a | 40.80 b |
| (E)-ocimene (mg/kg) | S,LRI,MS | 1245 | 0.21 bc | 0.09 c | 0.27 abc | 0.52 a | 0.43 ab | 0.12 c |
| monoterpene II (n.i.) | MS | 1260 | 0.69 bc | 0.40 c | 1.28 abc | 1.82 a | 1.49 ab | 0.53 bc |
| terpinolene | S,LRI,MS | 1275 | 0.44 | 0.28 | 0.70 | 0.38 | 3.83 | 0.84 |
| (Z)-alloocimene | LRI,MS | 1349 | 11.38 | 9.68 | 10.20 | 23.02 | 8.22 | 7.20 |
| (E)-alloocimene | S,LRI,MS | 1357 | 16.79 | 12.63 | 13.25 | 37.22 | 10.66 | 9.26 |
| linalool | S,LRI,MS | 1536 | 15.44 bc | 8.94 c | 19.00 abc | 31.43 ab | 40.98 a | 22.04 abc |
| sesquiterpenes | ||||||||
| (+)-cycloisosativene (mg/kg) | LRI,MS | 1477 | 1.15 a | 0.87 b | 0.80 b | 0.89 ab | 0.24 c | 0.26 c |
| α-copaene (mg/kg) | LRI,MS | 1487 | 8.44 a | 7.02 ab | 5.81 b | 8.77 a | 2.14 c | 1.64 c |
| sesquiterpene I (n.i.) | MS | 1536 | 0.00 b | 0.00 b | 0.00 b | 0.00 b | 3.44 a | 0.00 b |
| sesquiterpene II (n.i.) (mg/kg) | MS | 1583 | 0.29 a | 0.27 a | 0.23 a | 0.28 a | 0.08 b | 0.05 b |
| sesquiterpene III (n.i.) (mg/kg) | MS | 1683 | 0.11 ab | 0.09 b | 0.09 b | 0.13 a | 0.03 c | 0.02 c |
| δ-selinene | LRI,MS | 1698 | 24.52 b | 25.84 ab | 10.32 c | 32.29 a | 6.82 c | 2.07 c |
| γ-elemene (mg/kg) | MS | 1704 | 0.04 b | 0.05 b | 0.05 b | 0.06 b | 0.20 a | 0.05 b |
| α-muurolene (mg/kg) | LRI,MS | 1719 | 1.65 b | 1.42 b | 1.26 b | 2.10 a | 0.49 c | 0.34 c |
| sesquiterpene IV (n.i.) | MS | 1736 | 0.63 c | 3.02 c | 26.21 b | 2.33 c | 51.54 a | 29.75 b |
| α-farnesene (mg/kg) | LRI,MS | 1745 | 0.53 a | 0.29 b | 0.84 a | 0.12 b | 0.25 b | 0.17 b |
| δ-cadinene (mg/kg) | LRI,MS | 1750 | 0.14 ab | 0.11 b | 0.17 a | 0.17 a | 0.06 c | 0.07 c |
| aldehydes | ||||||||
| acrolein | LRI,MS | 829 | 6.07 a | 2.34 d | 4.66 ab | 4.23 bc | 2.60 cd | 3.75 bc |
| 2-methylbutanal | LRI,MS | 903 | 10.74 | 13.39 | 11.93 | 7.86 | 18.70 | 13.76 |
| 3-methylbutanal | S,LRI,MS | 906 | 10.51 b | 22.29 a | 10.65 b | 8.49 b | 12.31 b | 12.26 b |
| 2-methyl-2-pentenal | MS | 930 | 1.08 c | 0.26 cd | 3.34 a | 2.15 b | 0.85 cd | 0.00 d |
| (E)-2-butenal | S,LRI,MS | 1024 | 11.44 | 9.98 | 12.87 | 9.10 | 18.39 | 25.45 |
| unsaturated aliphatic aldehyde I (n.i.) | MS | 1036 | 3.27 a | 2.25 b | 2.63 ab | 2.48 ab | 1.56 b | 1.68 b |
| unsaturated aliphatic aldehyde II (n.i.) | MS | 1051 | 0.25 | 5.31 | 0.55 | 0.03 | 0.04 | 0.25 |
| hexanal (mg/kg) | S,LRI,MS | 1070 | 0.24 b | 0.29 b | 0.27 b | 0.44 a | 0.53 a | 0.20 b |
| (Z)-2-pentenal | LRI,MS | 1093 | 11.73 ab | 7.43 c | 16.92 a | 8.09 bc | 5.29 c | 5.70 c |
| (E)-2-pentenal | S,LRI,MS | 1115 | 52.30 a | 48.87 a | 43.67 ab | 39.40 ab | 24.74 b | 42.38 ab |
| (E)-3-hexenal | LRI,MS | 1125 | 57.10 a | 42.81 b | 67.22 a | 43.82 ab | 23.48 c | 35.30 bc |
| (Z)-3-hexenal (mg/kg) | LRI,MS | 1130 | 1.36 a | 0.40 cd | 0.96 abc | 1.09 ab | 0.47 bcd | 0.14 d |
| heptanal | LRI,MS | 1175 | 4.37 | 3.60 | 4.33 | 3.19 | 3.03 | 3.90 |
| (Z)-2-hexenal | LRI,MS | 1189 | 44.49 | 31.63 | 48.50 | 33.38 | 39.16 | 32.68 |
| (E)-2-hexenal (mg/kg) | S,LRI,MS | 1205 | 19.38 bc | 22.54 b | 21.96 b | 11.90 cd | 5.92 d | 34.95 a |
| octanal (mg/kg) | S,LRI,MS | 1280 | 0.10 a | 0.11 a | 0.09 a | 0.07 b | 0.07 b | 0.09 a |
| (Z)-2-heptenal | LRI,MS | 1312 | 10.60 | 7.65 | 4.80 | 11.30 | 7.66 | 7.12 |
| (E,E)-2,4-hexadienal (mg/kg) | LRI,MS | 1381 | 0.27 ab | 0.16 cd | 0.35 a | 0.20 bc | 0.07 d | 0.13 cd |
| (E,Z)-2,4-hexadienal (mg/kg) | LRI,MS | 1385 | 1.56 ab | 1.06 bc | 2.19 a | 0.87 cd | 0.38 d | 0.62 cd |
| (E)-2-octenal | S,LRI,MS | 1417 | 11.75 a | 5.53 c | 8.12 bc | 10.47 ab | 7.17 bc | 9.15 b |
| (E,E)-2,4-heptadienal | LRI,MS | 1448 | 33.23 b | 19.61 d | 30.68 bcd | 32.10 bc | 21.83 c | 44.72 a |
| 2-isopropylidene-3-methylhexa-3,5-dienal | MS | 1460 | 0.36 bc | 0.33 c | 0.65 ab | 0.11 c | 0.09 c | 0.70 a |
| (E,E)-2,4-decadienal | LRI,MS | 1750 | 0.65 bc | 0.49 c | 1.06 ab | 1.52 a | 0.85 b | 0.85 b |
| (E,Z)-2,4-decadienal | LRI,MS | 1794 | 0.67 b | 0.26 c | 0.47 bc | 1.13 a | 0.93 ab | 0.61 bc |
| tetradecanal | LRI,MS | 1909 | 1.05 | 0.97 | 1.04 | 1.41 | 0.99 | 0.62 |
| ketones | ||||||||
| 3-pentanone (mg/kg) | LRI,MS | 962 | 0.09 c | 0.23 a | 0.09 c | 0.13 bc | 0.19 ab | 0.08 c |
| 1-penten-3-one (mg/kg) | S,LRI,MS | 1008 | 0.29 a | 0.29 a | 0.26 ab | 0.18 bc | 0.12 c | 0.22 ab |
| 3-hexen-2-one | MS | 1121 | 0.57 b | 0.67 a | 0.58 b | 0.44 c | 0.55 b | 0.45 c |
| 2-octanone | LRI,MS | 1275 | 6.12 | 0.62 | 0.54 | 1.55 | 1.51 | 0.89 |
| 2-methyl-6-methylene-1,7-octadien-3-one | LRI,MS | 1303 | 44.85 a | 21.50 b | 51.45 a | 24.25 b | 16.32 b | 62.59 a |
| 6-methyl-5-hepten-2-one | LRI,MS | 1326 | 2.15 bc | 1.94 bc | 2.63 abc | 3.02 ab | 4.40 a | 1.40 c |
| 2-cyclohexene-1,4-dione I | MS | 1712 | 3.62 ab | 2.27 cd | 5.11 a | 3.20 bc | 2.05 bcd | 1.04 d |
| 2-cyclohexene-1,4-dione II | MS | 1803 | 1.15 ab | 0.68 cd | 1.57 a | 1.06 abc | 0.64 cd | 0.29 d |
| (Z)-cinerolone | MS | 2002 | 0.20 b | 0.13 c | 0.19 b | 0.04 d | 0.03 d | 0.28 a |
| 1-(2,6,6-trimethyl-1-cyclohexen-1-yl)-1-penten-3-one | MS | 2056 | 0.18 c | 0.25 bc | 0.14 c | 0.51 a | 0.47 ab | 0.12 c |
| 4′-ethoxy-2′-hydroxyoctadecanophenone | MS | 2081 | 0.26 bc | 0.12 c | 0.90 a | 0.24 bc | 0.26 bc | 0.41 b |
| alcohols | ||||||||
| 2-methyl-2-propanol | LRI,MS | 886 | 4.98 | 4.93 | 8.45 | 0.03 | 0.00 | 4.33 |
| 1-methoxy-2-propanol (mg/kg) | MS | 917 | 0.15 b | 0.12 b | 0.18 ab | 0.15 b | 0.23 a | 0.14 b |
| 3-pentanol | LRI,MS | 1097 | 11.84 | 16.51 | 6.53 | 6.20 | 15.09 | 5.92 |
| 2-pentanol | LRI,MS | 1109 | 1.04 | 2.75 | 0.67 | 2.76 | 1.78 | 1.59 |
| 1-penten-3-ol (mg/kg) | LRI,MS | 1148 | 0.18 b | 0.28 a | 0.19 b | 0.14 b | 0.19 b | 0.18 b |
| 3-methyl-1-butanol | S,LRI,MS | 1197 | 19.96 b | 26.70 b | 22.74 b | 27.21 b | 57.71 a | 23.80 b |
| 1-pentanol | S,LRI,MS | 1239 | 10.37 bc | 16.24 ab | 7.86 bc | 20.10 a | 22.07 a | 6.96 c |
| (E)-2-penten-1-ol | S,LRI,MS | 1300 | 62.07 | 65.28 | 64.27 | 43.81 | 67.92 | 61.24 |
| (Z)-2-penten-1-ol (mg/kg) | S,LRI,MS | 1308 | 0.26 a | 0.31 a | 0.27 a | 0.21 b | 0.28 a | 0.25 ab |
| 1-hexanol (mg/kg) | S,LRI,MS | 1342 | 0.70 | 1.21 | 1.20 | 1.17 | 1.58 | 0.76 |
| (E)-3-hexen-1-ol | LRI,MS | 1352 | 27.42 ab | 27.45 ab | 27.49 ab | 37.90 a | 46.03 a | 14.76 b |
| (Z)-3-hexen-1-ol (mg/kg) | S,LRI,MS | 1372 | 1.63 b | 1.08 bc | 1.50 bc | 2.77 a | 1.92 ab | 0.61 c |
| (E)-2-hexen-1-ol (mg/kg) | S,LRI,MS | 1394 | 1.00 b | 1.68 b | 1.37 b | 1.05 b | 3.42 a | 1.42 b |
| (Z)-2-hexen-1-ol | S,LRI,MS | 1402 | 7.53 | 122.27 | 11.84 | 11.20 | 15.53 | 10.63 |
| 1-heptanol | S,LRI,MS | 1445 | 4.05 | 2.16 | 2.23 | 3.38 | 3.39 | 2.71 |
| 2-ethyl-1-hexanol | S,LRI,MS | 1480 | 4.58 d | 5.90 c | 10.39 a | 4.32 d | 4.01 d | 6.93 b |
| 1-octanol | S,LRI,MS | 1546 | 7.65 | 5.29 | 5.41 | 5.56 | 6.01 | 5.98 |
| 2,4-hexadien-1-ol | LRI,MS | 1568 | 0.11 | 0.10 | 0.00 | 0.65 | 0.64 | 0.00 |
| 1-nonanol | S,LRI,MS | 1650 | 3.73 | 2.56 | 3.14 | 2.53 | 3.32 | 3.10 |
| 3-methyl-3-cyclohexen-1-ol | MS | 1744 | 4.66 a | 2.34 b | 4.39 a | 2.25 b | 0.65 b | 0.88 b |
| 2-(2-butoxyethoxy)-ethanol | LRI,MS | 1779 | 3.83 b | 2.43 b | 42.91 a | 7.86 b | 7.62 b | 9.91 b |
| 2,2′-oxybis-1-propanol | LRI,MS | 1865 | 4.04 | 3.46 | 4.51 | 2.27 | 2.59 | 2.44 |
| tetradecanol | LRI,MS | 2158 | 25.23 a | 26.72 a | 12.11 ab | 31.35 a | 13.64 ab | 6.70 b |
| acids | ||||||||
| acetic acid (mg/kg) | S,LRI,MS | 1427 | 1.82 | 2.55 | 2.46 | 2.49 | 4.53 | 1.70 |
| acid (n.i.) (mg/kg) | MS | 1476 | 0.91 ab | 0.82 b | 1.17 a | 1.10 a | 1.18 a | 1.07 a |
| butanoic acid | S,LRI,MS | 1602 | 27.65 c | 41.57 bc | 40.46 bc | 52.13 ab | 72.47 a | 30.43 bc |
| hexanoic acid (mg/kg) | S,LRI,MS | 1823 | 0.76 c | 1.23 bc | 1.32 bc | 1.74 ab | 2.21 a | 1.00 c |
| 2-ethylhexanoic acid | LRI,MS | 1925 | 3.41 c | 8.59 c | 52.77 a | 10.47 bc | 12.29 bc | 23.46 b |
| (E)-3-hexenoic acid | LRI,MS | 1926 | 12.93 b | 9.33 bc | 16.87 ab | 24.46 a | 5.08 bc | 1.38 c |
| octanoic acid (mg/kg) | S,LRI,MS | 2033 | 0.48 b | 7.82 a | 1.29 b | 1.65 b | 1.47 b | 0.59 b |
| sorbic acid | LRI,MS | 2048 | 7.83 b | 4.94 bc | 14.78 a | 8.44 b | 4.58 bc | 1.04 c |
| nonanoic acid (mg/kg) | S,LRI,MS | 2139 | 41.76 b | 434.34 a | 54.30 b | 41.10 b | 55.83 b | 29.51 b |
| decanoic acid (mg/kg) | S,LRI,MS | 2244 | 0.07 b | 0.43 a | 0.14 b | 0.02 b | 0.06 b | 0.06 b |
| esters | ||||||||
| allyl acetate | LRI,MS | 805 | 53.61 | 58.55 | 69.96 | 43.21 | 30.69 | 37.81 |
| methyl acetate | LRI,MS | 816 | 91.00 b | 166.56 a | 101.38 ab | 59.88 b | 115.21 ab | 81.99 b |
| 1,1,1-trimethoxyethane | MS | 871 | 61.25 b | 328.53 a | 117.33 ab | 50.70 b | 85.45 b | 24.70 b |
| ethyl 2-methylbutanoate | LRI,MS | 1042 | 0.90 | 0.64 | 0.55 | 0.70 | 0.70 | 0.51 |
| isoamyl acetate | S,LRI,MS | 1114 | 4.91 a | 4.92 a | 2.26 b | 2.88 b | 3.19 b | 1.69 b |
| methyl 3-methyl-2-butenoate | LRI,MS | 1157 | 3.79 | 0.85 | 2.84 | 2.61 | 2.86 | 1.75 |
| methyl hexanoate | LRI,MS | 1178 | 1.29 | 1.01 | 2.05 | 1.51 | 1.02 | 1.29 |
| ethyl hexanoate | S,LRI,MS | 1228 | 0.06 | 0.23 | 0.31 | 0.50 | 0.35 | 0.12 |
| hexyl acetate | S,LRI,MS | 1265 | 21.75 b | 89.41 a | 25.62 b | 11.18 b | 91.10 a | 13.11 b |
| (Z)-3-hexen-1-yl acetate (mg/kg) | S,LRI,MS | 1309 | 0.27 b | 0.62 a | 0.20 b | 0.05 b | 0.26 b | 0.09 b |
| (Z)-2-hexen-1-yl acetate | LRI,MS | 1326 | 1.52 b | 4.02 a | 1.44 b | 0.91 b | 1.75 b | 0.91 b |
| (E)-3-hexenyl butanoate | LRI,MS | 1454 | 2.23 abc | 0.88 c | 2.34 ab | 1.47 bc | 2.94 a | 1.71 abc |
| 3-hydroxy-2,4,4-trimethylpentyl 2-methylpropanoate | MS | 1854 | 0.37 c | 0.16 c | 4.94 a | 1.06 bc | 0.95 bc | 1.54 b |
| methyl cinnamoylglycinate | MS | 1960 | 3.44 b | 3.34 b | 6.22 a | 2.62 c | 2.25 c | 3.33 b |
| triacetin | LRI,MS | 2049 | 1.09 bc | 0.41 c | 5.19 a | 1.44 bc | 1.02 bc | 1.77 b |
| 2-propenyl pentanoate | MS | 2074 | 3.07 bc | 1.38 cd | 7.81 a | 3.79 b | 2.39 bcd | 0.80 d |
| methyl 3-oxo-2-pentyl-cyclopentaneacetate | MS | 2257 | 7.59 ab | 3.69 c | 12.29 a | 8.44 ab | 8.06 ab | 6.03 bc |
| benzenoids | ||||||||
| benzene | LRI,MS | 926 | 9.83 | 14.67 | 14.44 | 12.05 | 11.62 | 10.10 |
| toluene (mg/kg) | LRI,MS | 1027 | 0.10 | 0.17 | 0.07 | 0.40 | 0.17 | 0.10 |
| m-xylene | LRI,MS | 1122 | 22.55 | 19.69 | 15.30 | 105.52 | 26.14 | 17.65 |
| p-xylene (mg/kg) | LRI,MS | 1128 | 0.06 | 0.06 | 0.05 | 0.22 | 0.07 | 0.05 |
| o-xylene (mg/kg) | LRI,MS | 1172 | 0.03 | 0.03 | 0.02 | 0.11 | 0.04 | 0.03 |
| p-ethyltoluene | LRI,MS | 1213 | 16.73 | 17.37 | 11.75 | 56.70 | 14.44 | 11.29 |
| 1,3,5-trimethylbenzene (mesitylene) | LRI,MS | 1235 | 11.77 | 11.52 | 12.45 | 32.07 | 9.69 | 8.84 |
| 2-ethyltoluene | LRI,MS | 1251 | 7.00 | 6.28 | 5.39 | 18.11 | 5.10 | 4.07 |
| p-cymene | S,LRI,MS | 1262 | 1.51 | 1.57 | 2.15 | 2.87 | 31.68 | 4.46 |
| m-cymene | LRI,MS | 1270 | 19.46 | 16.99 | 16.47 | 40.75 | 10.97 | 10.45 |
| 1,3-diethylbenzene | LRI,MS | 1293 | 1.54 | 0.75 | 0.84 | 2.14 | 0.59 | 0.71 |
| o-cymene | LRI,MS | 1296 | 1.64 | 1.38 | 1.37 | 3.62 | 0.94 | 1.09 |
| 1-ethyl-3,5-dimethylbenzene | LRI,MS | 1316 | 4.25 | 4.17 | 3.80 | 8.75 | 2.79 | 3.11 |
| 1,2,3-trimethylbenzene (hemellitol) | LRI,MS | 1324 | 8.25 | 6.56 | 7.35 | 14.88 | 4.70 | 4.04 |
| anisole | LRI,MS | 1329 | 3.77 | 2.45 | 2.91 | 1.27 | 3.42 | 1.85 |
| 1,2,4,5-tetramethylbenzene | LRI,MS | 1413 | 1.89 | 1.36 | 1.44 | 3.99 | 1.13 | 1.09 |
| isodurene | LRI,MS | 1422 | 2.78 | 2.21 | 2.22 | 5.44 | 2.06 | 1.80 |
| 1,2,3,4-tetramethylbenzene | MS | 1423 | 2.78 | 2.20 | 2.20 | 5.39 | 2.01 | 1.81 |
| p-cymenene | LRI,MS | 1436 | 9.05 b | 7.21 c | 13.45 a | 7.49 c | 6.48 c | 6.87 c |
| methyl benzoate | LRI,MS | 1601 | 14.74 b | 3.88 c | 22.08 b | 41.94 a | 13.97 b | 4.01 c |
| acetophenone | S,LRI,MS | 1626 | 14.76 b | 13.46 b | 20.73 a | 13.08 b | 12.68 b | 13.60 b |
| estragole | LRI,MS | 1655 | 0.06 c | 0.03 c | 0.02 c | 0.90 b | 1.40 a | 0.01 c |
| 4-ethylbenzaldehyde | LRI,MS | 1689 | 6.13 b | 4.73 c | 9.68 a | 4.66 c | 3.77 c | 4.66 c |
| methyl salicylate | LRI,MS | 1755 | 47.45 ab | 18.17 d | 62.13 a | 61.68 a | 37.24 bc | 30.00 c |
| aromatic aldehyde I (n.i.) | MS | 1810 | 9.56 b | 9.57 b | 18.54 a | 6.75 c | 5.56 c | 9.18 c |
| aromatic aldehyde II (n.i.) | MS | 1839 | 9.12 b | 9.29 b | 18.39 a | 6.46 c | 5.50 c | 8.81 c |
| benzyl alcohol | S,LRI,MS | 1851 | 20.21 c | 22.03 c | 24.09 bc | 23.65 bc | 35.74 ab | 39.17 a |
| 2-phenylethyl alcohol (mg/kg) | S,LRI,MS | 1885 | 0.33 | 0.30 | 0.39 | 0.35 | 0.32 | 0.38 |
| benzyl nitrile | LRI,MS | 1896 | 0.04 b | 0.00 b | 0.16 b | 0.00 b | 0.00 b | 1.17 a |
| lilial | LRI,MS | 2020 | 1.55 bc | 1.58 bc | 1.04 bc | 2.42 ab | 3.74 a | 1.01 c |
| methyl 2-methoxybenzoate | LRI,MS | 2037 | 5.21 a | 0.43 c | 4.21 ab | 3.49 a b | 2.45 bc | 1.19 c |
| complex benzenoid (n.i.) | MS | 2041 | 3.94 b | 5.40 b | 3.69 b | 12.24 a | 11.25 a | 3.07 b |
| 2-phenoxyethanol | LRI,MS | 2106 | 3.30 bc | 2.07 c | 4.99 bc | 10.29 a | 7.11 ab | 5.49 bc |
| methyl anthranilate | LRI,MS | 2200 | 0.73 b | 0.05 c | 0.76 b | 1.15 b | 2.04 a | 0.01 c |
| menthyl salicylate | MS | 2273 | 1.25 b | 2.57 a | 2.16 ab | 2.74 a | 2.27 a | 1.39 b |
| 4-ethoxystyrene | MS | 2348 | 1.97 c | 1.96 c | 2.20 bc | 3.07 a | 2.74 ab | 1.95 c |
| benzoic acid | LRI,MS | 2381 | 11.32 b | 44.78 a | 38.56 a | 5.62 b | 7.75 b | 14.51 b |
| furanoids | ||||||||
| 2-ethylfuran (mg/kg) | LRI,MS | 938 | 0.14 b | 0.08 cd | 0.22 a | 0.09 bc | 0.05 cd | 0.03 d |
| 2-vinylfuran | LRI,MS | 1059 | 10.26 b | 5.89 bc | 17.85 a | 9.15 b | 5.32 bc | 1.94 c |
| 4-methyl-2,3-dihydrofuran | LRI,MS | 1184 | 4.98 ab | 4.59 b | 3.83 b | 6.13 a | 4.90 ab | 3.86 b |
| 2-pentylfuran | LRI,MS | 1225 | 7.81 | 7.60 | 7.33 | 8.82 | 10.79 | 62.68 |
| 5-methyl-2-furancarboxaldehyde | LRI,MS | 1551 | 4.99 b | 3.33 c | 7.19 a | 3.58 bc | 1.90 cd | 1.55 d |
| 2(5H)-furanone | LRI,MS | 1722 | 5.90 | 3.67 | 5.23 | 2.23 | 1.42 | 2.94 |
| 5-ethyl-2(5H)-furanone (mg/kg) | LRI,MS | 1733 | 0.19 a | 0.08 bc | 0.19 a | 0.15 ab | 0.06 bc | 0.03 c |
| 2-ethyl-5-methyl-tetrahydrofuran (mg/kg) | MS | 1933 | 0.44 ab | 0.21 cd | 0.58 a | 0.34 bc | 0.15 cd | 0.07 d |
| miscellaneous | ||||||||
| dimethyl sulfide | LRI,MS | 739 | 1.21 ab | 1.26 ab | 2.49 a | 1.18 ab | 0.74 b | 1.81 ab |
| 2,4-dihydro-5-methyl-3H-pyrazol-3-one I | MS | 978 | 0.90 | 2.14 | 1.10 | 10.00 | 4.62 | 0.38 |
| 1,2-dihydro-5-methyl-3H-pyrazol-3-one | MS | 1109 | 0.85 | 1.17 | 0.52 | 4.51 | 2.12 | 0.09 |
| 2,4-dihydro-5-methyl-3H-pyrazol-3-one II | MS | 1150 | 0.08 | 0.35 | 0.09 | 1.82 | 0.60 | 0.01 |
| 2-phenyl-1H-indole | MS | 1502 | 78.56 c | 93.8 b | 139.59 a | 67.51 c | 74.11 c | 95.38 b |
| dimethyl sulfoxide | LRI,MS | 1540 | 26.39 abc | 15.52 c | 27.62 ab | 32.02 a | 23.96 abc | 16.65 bc |
| n.i. (m/z 189,207,131) | MS | 1688 | 0.51 a | 0.21 b | 0.45 ab | 0.47 a | 0.25 ab | 0.19 b |
| n.i. (m/z 84,85,41,42,39,133,147,175) | MS | 1990 | 1.91 b | 1.20 b | 3.72 a | 1.07 b | 1.32 b | 1.21 b |
| phenol | S,LRI,MS | 1972 | 5.27 c | 6.01 b | 9.88 a | 4.38 d | 4.16 d | 5.95 b |
| totals (mg/kg) | ||||||||
| total hydrocarbons | 2.12 a | 1.54 b | 1.99 a | 1.13 c | 1.01 c | 1.70 b | ||
| total monoterpenes | 0.35 b | 0.23 b | 0.41 b | 0.71 b | 1.85 a | 0.26 b | ||
| total sesquiterpenes | 12.36 a | 10.15 a | 9.29 a | 12.54 a | 3.54 b | 2.66 b | ||
| total aldehydes | 23.19 b | 24.78 b | 26.10 b | 14.79 c | 7.64 c | 36.37 a | ||
| total ketones | 0.44 b | 0.56 a | 0.41 bc | 0.34 c | 0.33 c | 0.37 c | ||
| total alcohols | 4.12 bc | 5.02 bc | 4.95 bc | 5.69 ab | 7.89 a | 3.52 c | ||
| total acids | 45.85 b | 447.25 a | 60.81 b | 48.19 b | 65.37 b | 33.99 b | ||
| total esters | 0.52 b | 1.29 a | 0.56 b | 0.25 b | 0.61 b | 0.27 b | ||
| total benzenoids | 0.80 | 0.81 | 0.87 | 1.60 | 0.88 | 0.79 | ||
| total furanoids | 0.80 | 0.40 | 1.04 | 0.61 | 0.28 | 0.76 | ||
| total miscellaneous | 0.12 b | 0.12 b | 0.19 a | 0.12 b | 0.11 b | 0.12 b | ||
| Volatile Compound | Sensory Descriptor (Aroma) * | Threshold * | Odor Activity Value (OAV) | |||||
|---|---|---|---|---|---|---|---|---|
| Buža | I. bjelica | Rosinjola | Oblica | Lastovka | Leccino | |||
| OAV > 1 | ||||||||
| (Z)-3-hexenal | leaf-like, green, apple-like | 1.7 | 800.00 a | 235.29 cd | 564.71 abc | 641.18 ab | 276.47 bcd | 82.35 d |
| 1-penten-3-one | leaf, green, pungent, sweet | 0.73 | 397.26 a | 397.26 a | 356.16 ab | 246.58 bc | 164.38 c | 301.37 ab |
| (E)-2-hexenal | green, apple-like, bitter almond | 420 | 46.14 bc | 53.67 b | 52.29 b | 28.33 cd | 14.10 d | 83.21 a |
| hexanal | green, sweet, green apple, grassy | 75 | 3.20 b | 3.87 b | 3.60 b | 5.87 a | 7.07 a | 2.67 b |
| 3-methylbutanal | malty | 5.2 | 2.02 b | 4.29 a | 2.05 b | 1.63 b | 2.37 b | 2.36 b |
| 1-hexanol | fruit, banana, soft, grass | 400 | 1.75 | 3.03 | 3.00 | 2.93 | 3.95 | 1.90 |
| 2-methylbutanal | malty | 5.4 | 1.99 | 2.48 | 2.21 | 1.46 | 3.46 | 2.55 |
| hexanoic acid | pungent, rancid, sweaty | 700 | 1.09 c | 1.76 bc | 1.89 bc | 2.49 ab | 3.16 a | 1.43 c |
| (Z)-3-hexen-1-yl acetate | green, banana-like, olive fruity | 200 | 1.35 b | 3.10 a | 1.00 b | 0.25 b | 1.30 b | 0.45 b |
| (E)-2-octenal | herbaceous, spicy | 4 | 2.94 a | 1.38 c | 2.03 bc | 2.62 ab | 1.79 bc | 2.29 b |
| octanoic acid | oily, fatty | 3000 | 0.16 b | 2.61 a | 0.43 b | 0.55 b | 0.49 b | 0.20 b |
| (Z)-3-hexen-1-ol | green, apple, leaf-like, banana | 1100 | 1.48 b | 0.98 bc | 1.36 bc | 2.52 a | 1.75 ab | 0.55 c |
| ethyl 2-methylbutanoate | fruity | 0.72 | 1.25 | 0.89 | 0.76 | 0.97 | 0.97 | 0.71 |
| OAV < 1 | ||||||||
| 1-penten-3-ol | lawn, olive, leaf, pungent | 400 | 0.45 b | 0.70 a | 0.48 b | 0.35 b | 0.48 b | 0.45 b |
| (E)-2-hexen-1-ol | green, grass, leaves, sweet | 5000 | 0.20 b | 0.34 b | 0.27 b | 0.21 b | 0.68 a | 0.28 b |
| 3-methyl-1-butanol | woody, whiskey, sweet | 100 | 0.20 b | 0.27 b | 0.23 b | 0.27 b | 0.58 a | 0.24 b |
| octanal | fatty, sharp, citrus-like, soapy | 320 | 0.31 a | 0.34 a | 0.28 a | 0.22 b | 0.22 b | 0.28 a |
| (E)-2-penten-1-ol | green fruity, fresh olive fruits | 250 | 0.25 | 0.26 | 0.26 | 0.18 | 0.27 | 0.24 |
| (E)-2-pentenal | green, apple, bitter almond | 300 | 0.17 a | 0.16 a | 0.15 ab | 0.13 ab | 0.08 b | 0.14 ab |
| butanoic acid | rancid, cheese | 650 | 0.04 c | 0.06 bc | 0.06 bc | 0.08 ab | 0.11 a | 0.05 bc |
| (E,Z)-2,4-decadienal | deep-fried | 10 | 0.07 b | 0.03 c | 0.05 bc | 0.11 a | 0.09 ab | 0.06 bc |
| hexyl acetate | green, fruity, sweet, apple | 1040 | 0.02 b | 0.09 a | 0.02 b | 0.01 b | 0.09 a | 0.01 b |
| 1-pentanol | fruity, strong, sticky, balsamic | 470 | 0.02 bc | 0.03 ab | 0.02 bc | 0.04 a | 0.05 a | 0.01 c |
| octane | sweety, alcane | 940 | 0.03 | 0.04 | 0.03 | 0.03 | 0.04 | 0.03 |
| 2-octanone | mould, green | 510 | 0.01 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| (E)-3-hexen-1-ol | green, bitter | 1500 | 0.02 ab | 0.02 ab | 0.02 ab | 0.03 a | 0.03 a | 0.01 b |
| 1-nonanol | fatty, rancid | 280 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 |
| heptanal | oily, fatty, woody | 500 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 |
| (E,E)-2,4-heptadienal | fatty, rancid | 3620 | 0.01 b | 0.01 d | 0.01 bcd | 0.01 bc | 0.01 c | 0.01 a |
| (E,E)-2,4-decadienal | deep-fried | 180 | 0.00 bc | 0.00 c | 0.01 ab | 0.01 a | 0.00 b | 0.00 b |
| 6-methyl-5-hepten-2-one | pungent, green | 1000 | 0.00 bc | 0.00 bc | 0.00 abc | 0.00 ab | 0.00 a | 0.00 c |
| 3-pentanone | fruity, green, sweet | 70,000 | 0.00 c | 0.00 a | 0.00 c | 0.00 bc | 0.00 ab | 0.00 c |
| Phenol | Variety | |||||
|---|---|---|---|---|---|---|
| Buža | I. bjelica | Rosinjola | Oblica | Lastovka | Leccino | |
| simple phenols | ||||||
| tyrosol | 4.87 b | 11.29 a | 3.69 b | 9.10 ab | 12.28 a | 5.60 b |
| hydroxytyrosol | 5.40 c | 10.21 b | 5.59 bc | 6.33 bc | 20.17 a | 6.47 bc |
| hydroxytyrosol acetate * | 0.35 c | 0.67 b | 0.37 bc | 0.42 bc | 1.44 a | 0.50 bc |
| vanillin | 0.21 b | 0.16 bc | 0.20 bc | 0.12 c | 0.11 c | 0.31 a |
| phenolic acids | ||||||
| vanillic acid | 0.31 | 0.32 | 0.33 | 0.18 | 0.37 | 0.25 |
| p-coumaric acid | 1.26 bc | 0.90 c | 0.82 cd | 1.69 b | 2.80 a | 0.34 d |
| flavonoids | ||||||
| luteolin | 2.02 bc | 2.95 a | 2.86 ab | 2.93 a | 3.35 a | 1.89 c |
| apigenin | 0.55 bc | 0.87 a | 0.66 b | 0.33 d | 0.39 d | 0.46 cd |
| lignans | ||||||
| pinoresinol | 9.97 a | 4.02 c | 6.98 b | 3.21 c | 3.68 c | 4.14 c |
| Acetoxypinoresinol * | 6.72 c | 14.11 a | 11.39 ab | 8.69 bc | 11.94 ab | 7.49 c |
| secoiridoids | ||||||
| Secologanoside * | 0.03 b | 0.04 b | 0.03 b | 0.04 b | 0.06 a | 0.04 b |
| elenolic acid glucoside * | 0.04 bc | 0.04 c | 0.05 abc | 0.05 ab | 0.06 a | 0.04 c |
| 3,4-DHPEA-EDA * | 95.50 b | 115.68 b | 104.93 b | 98.46 b | 121.33 b | 175.06 a |
| oleuropein aglycone I * | 72.56 bc | 94.57 ab | 109.85 a | 49.09 cd | 115.05 a | 41.17 d |
| p-HPEA-EDA * | 49.15 b | 82.70 a | 47.35 b | 76.79 a | 49.21 b | 87.49 a |
| oleuropein + ligstroside aglycones I & II * | 43.38 b | 97.82 a | 49.19 b | 38.63 b | 49.33 b | 30.11 b |
| oleuropein aglycone II * | 64.44 c | 79.72 abc | 100.61 a | 42.38 d | 94.14 ab | 71.77 bc |
| ligstroside aglycon III * | 1.66 c | 4.60 a | 1.82 c | 2.79 bc | 1.99 c | 4.04 ab |
| oleuropein aglycone III * | 9.06 c | 15.84 a | 11.66 bc | 11.40 bc | 13.75 ab | 9.85 c |
| total phenols | 367.25 c | 536.49 a | 458.38 abc | 352.63 c | 501.45 ab | 447.00 bc |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lukić, I.; Lukić, M.; Žanetić, M.; Krapac, M.; Godena, S.; Brkić Bubola, K. Inter-Varietal Diversity of Typical Volatile and Phenolic Profiles of Croatian Extra Virgin Olive Oils as Revealed by GC-IT-MS and UPLC-DAD Analysis. Foods 2019, 8, 565. https://doi.org/10.3390/foods8110565
Lukić I, Lukić M, Žanetić M, Krapac M, Godena S, Brkić Bubola K. Inter-Varietal Diversity of Typical Volatile and Phenolic Profiles of Croatian Extra Virgin Olive Oils as Revealed by GC-IT-MS and UPLC-DAD Analysis. Foods. 2019; 8(11):565. https://doi.org/10.3390/foods8110565
Chicago/Turabian StyleLukić, Igor, Marina Lukić, Mirella Žanetić, Marin Krapac, Sara Godena, and Karolina Brkić Bubola. 2019. "Inter-Varietal Diversity of Typical Volatile and Phenolic Profiles of Croatian Extra Virgin Olive Oils as Revealed by GC-IT-MS and UPLC-DAD Analysis" Foods 8, no. 11: 565. https://doi.org/10.3390/foods8110565
APA StyleLukić, I., Lukić, M., Žanetić, M., Krapac, M., Godena, S., & Brkić Bubola, K. (2019). Inter-Varietal Diversity of Typical Volatile and Phenolic Profiles of Croatian Extra Virgin Olive Oils as Revealed by GC-IT-MS and UPLC-DAD Analysis. Foods, 8(11), 565. https://doi.org/10.3390/foods8110565










