Characterization of Aroma-Active Compounds in Seed Extract of Black Cumin (Nigella sativa L.) by Aroma Extract Dilution Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. N. sativa (Black Cumin) Seed Extract
2.3. Extraction of Volatile Compounds
2.4. GC/FID and GC/MS/O Analysis of Aroma Compounds
2.5. Aroma Extract Dilution Analysis
2.6. Sensory Descriptive Analysis
2.6.1. Panel
2.6.2. Preparation and Presentation of the Samples
2.6.3. Intensity and Similarity Tests
2.6.4. Descriptive Analysis
2.6.5. Statistical Analysis
3. Results and Discussion
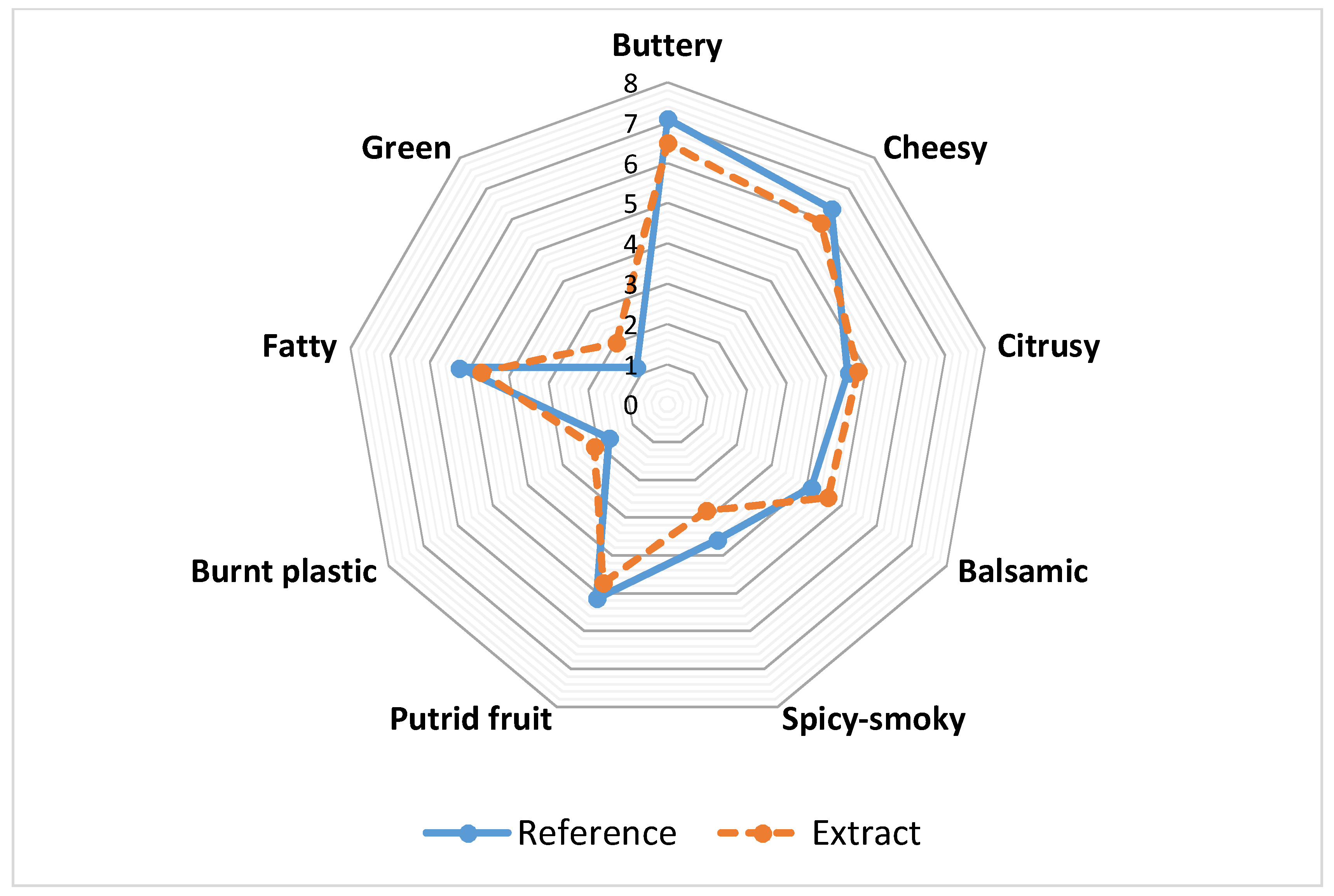
3.1. Sensory Analysis
3.1.1. Odor Sensory Profiles
3.1.2. Similarity and Intensity Evaluation of Aromatic Extract
3.2. Aroma Compounds of Black Cumin Seed Extract
3.3. GC-MS-O Results
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ali, G.H.; Blunden, G. Pharmacological and toxicological properties of Nigella sativa. Phytother. Res. 2003, 17, 299–303. [Google Scholar] [CrossRef] [PubMed]
- Ramadan, M.F. Nutritional value, functional properties and nutraceutical applications of black cumin (Nigella sativa L.): An overview. Int. J. Food Sci. Technol. 2007, 42, 1208–1218. [Google Scholar] [CrossRef]
- Kiralan, M.; Kiralan, S.S. Changes in volatile compounds of black cumin oil and hazelnut oil by microwave heating process. J. Am. Oil Chem. Soc. 2015, 92, 1445–1450. [Google Scholar] [CrossRef]
- Kiralan, M.; Ulas, M.; Özaydın, A.; Özdemir, N.; Özkan, G.; Bayrak, A.; Ramadan, M.F. Blends of cold pressed black cumin oil and sunflower oil with improved stability: A study based on changes in the levels of volatiles, tocopherols and thymoquinone during accelerated oxidation conditions. J. Food Biochem. 2017, 41, 1–10. [Google Scholar] [CrossRef]
- Salarzaei, M.; Anbari, M.; Parooei, F. Physiological effects of Nigella sativa on body: A review. Indo Am. J. Pharm. Sci. 2017, 4, 2890–2892. [Google Scholar]
- Garcia-Gonzalez, D.L.; Tena, N.; Aparicio, R. Characterization of olive paste volatiles to predict the sensory quality of virgin olive oil. Eur. J. Lipid Sci. Technol. 2007, 109, 663–672. [Google Scholar] [CrossRef]
- Kesen, S.; Kelebek, H.; Sen, K.; Ulas, M.; Selli, S. GC–MS–olfactometric characterization of the key aroma compounds in Turkish olive oils by application of the aroma extract dilution analysis. Food Res. Int. 2013, 54, 1987–1994. [Google Scholar] [CrossRef]
- Kiralan, M.; Özkan, G.; Bayrak, A.; Ramadan, M.F. Physicochemical properties and stability of black cumin (Nigella sativa) seed oil as affected by different extraction methods. Ind. Crops Prod. 2014, 57, 52–58. [Google Scholar] [CrossRef]
- Liu, X.; Abd El-Aty, A.M.; Cho, S.K.; Yang, A.; Park, J.H.; Shim, J.H. Characterization of secondary volatile profiles in Nigella sativa seeds from two different origins using accelerated solvent extraction and gas chromatography-mass spectrometry. Biomed. Chromatogr. 2012, 26, 1157–1162. [Google Scholar] [CrossRef] [PubMed]
- Amanpour, A.; Kelebek, H.; Kesen, S.; Selli, S. Characterization of aroma-active compounds in Iranian cv. Mari Olive Oil by aroma extract dilution analysis and GC–MS-Olfactometry. J. Am. Oil Chem. Soc. 2016, 93, 1595–1603. [Google Scholar] [CrossRef]
- Selli, S.; Kelebek, H. Aromatic profile and odour-activity value of blood orange juices obtained from Moro and Sanguinello (Citrus sinensis L. Osbeck). Ind. Crops Prod. 2011, 33, 727–733. [Google Scholar] [CrossRef]
- Schieberle, P.; Grosch, W. Evaluation of the flavour of wheat and rye crust by aroma extract dilution analysis. Z. Lebensm. Unters. Forsch. 1987, 185, 111–113. [Google Scholar] [CrossRef]
- Fickert, B.; Schieberle, P. Identification of the key odorants in barly malt (caramalt) using GC/MS techniques and odour dilution analyses. Nahrung 1998, 42, 371–375. [Google Scholar] [CrossRef]
- Tairu, A.O.; Hofmann, T.; Schieberle, P. Studies on the key odorants formed by roasting of wild mango seeds (Irvingia gabonensis). J. Agric. Food Chem. 2000, 48, 2391–2394. [Google Scholar] [CrossRef] [PubMed]
- Kesen, S.; Kelebek, H.; Selli, S. Characterization of the volatile, phenolic and antioxidant properties of monovarietal olive oil obtained from cv. Halhali. J. Am. Oil Chem. Soc. 2013, 90, 1685–1696. [Google Scholar] [CrossRef]
- Amanpour, A.; Kelebek, H.; Selli, S. Aroma constituents of shade-dried aerial parts of Iranian dill (Anethum graveolens L.) and savory (Satureja sahendica Bornm.) by solvent-assisted flavor evaporation technique. Food Meas. 2017, 11, 1430–1439. [Google Scholar] [CrossRef]
- Amanpour, A.; Sonmezdag, A.S.; Kelebek, H.; Selli, S. GC-MS-olfactometric characterization of the most aroma-active components in a representative aromatic extract from Iranian saffron (Crocus sativus L.). Food Chem. 2015, 182, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Z.; Lu, J.R. Strategies for enhancing fermentative production of acetoin: A review. Biotechnol. Adv. 2014, 32, 492–503. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H. Volatile flavor compounds in yogurt: A review. Crit. Rev. Food Sci. Nutr. 2010, 50, 938–950. [Google Scholar] [CrossRef] [PubMed]
- Buttery, R.G.; Orts, W.J.; Takeoka, G.R.; Nam, Y. Volatile flavor components of rice cakes. J. Agric. Food Chem. 1999, 47, 4353–4356. [Google Scholar] [CrossRef] [PubMed]
- Selli, S.; Kelebek, H.; Kesen, S.; Sonmezdag, A.S. GC-MS olfactometric and LC-DAD-ESI-MS/MS characterization of key odorants and phenolic compounds in black dry-salted olives. J. Sci. Food Agric. 2018. [Google Scholar] [CrossRef] [PubMed]
- Sabatini, N.; Perri, E.; Marsilio, V. An investigation on molecular partition of aroma compounds in fruit matrix and brine medium of fermented table olives. Innov. Food Sci. Emerg. Technol. 2009, 10, 621–626. [Google Scholar] [CrossRef]
- Pico, J.; Martinez, M.M.; Bernal, J.; Gomez, M. Evolution of volatile compounds in gluten-free bread: From dough to crumb. Food Chem. 2017, 227, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Kesen, S.; Kelebek, H.; Selli, S. Characterization of the key aroma compounds in turkish olive oils from different geographic origins by application of aroma extract dilution analysis (AEDA). J. Agric. Food Chem. 2014, 62, 391–401. [Google Scholar] [CrossRef] [PubMed]
- Morales, M.T.; Luna, G.; Aparicio, R. Comparative study of virgin olive oil sensory defects. Food Chem. 2005, 91, 293–301. [Google Scholar] [CrossRef]
- Buttery, R.G.; Seifert, R.M.; Guadagni, D.G.; Ling, L.C. Characterization of additional volatile components of tomato. J. Agric. Food Chem. 1971, 19, 524–529. [Google Scholar] [CrossRef]
- Alexander, H.C.; McCarty, W.M.; Bartlett, E.A.; Syverud, A.N. Aqueous odor and taste threshold values of industrial chemicals. J. Am. Water Works Assoc. 1982, 74, 595–599. [Google Scholar] [CrossRef]
- Pyysalo, T.; Suihko, M.; Honkanen, E. Odour thresholds of the major volatiles identified in cloudberry (Rubus chamaemorus L.) and arctic bramble (Rubus arcticus L.). LWT Lebensm. Wiss. Technol. 1977, 10, 36–39. [Google Scholar]
- Larsen, M.; Poll, L. Odour thresholds of some important aroma compounds in strawberries Geruchsschwellen einiger wichtiger Aromas toffe der Erdbeeren. Z. Lebensm. Unters. Forsch. 1992, 195, 120–123. [Google Scholar] [CrossRef]
- Pino, J.A.; Mesa, J. Contribution of volatile compounds to mango (Mangifera indica L.) aroma. Flavour Fragr. J. 2006, 21, 207–213. [Google Scholar] [CrossRef]
- Art, S.; Position, A. Untersuchungen zur Struktur-Aktivit its-Beziehung bei Geruchsstoffen. Z. Lebensm. Unters. Forsch. 1988, 187, 215–223. [Google Scholar]
- Buttery, R.G.; Turnbaugh, J.G.; Ling, L.C. Contribution of volatiles to rice aroma. J. Agric. Food Chem. 1988, 36, 1006–1009. [Google Scholar] [CrossRef]

| No. | LRI a | Compounds | Concentration b | Identification c |
|---|---|---|---|---|
| Carboxylic acids | ||||
| 1 | 1450 | Acetic acid | 3088 ± 28.2 | LRI, MS, Std |
| 2 | 1538 | Propanoic acid | 1187 ± 40.8 | LRI, MS, Std |
| 3 | 1562 | Isobutanoic acid | 218 ± 2.0 | LRI, MS, Std |
| 4 | 1628 | Butanoic acid | 138 ± 4.6 | LRI, MS, Std |
| 5 | 1638 | Propenoic acid | 147 ± 6.6 | LRI, MS, tent |
| 6 | 1731 | Pentanoic acid | 176 ± 4.5 | LRI, MS, Std |
| 7 | 1854 | Hexanoic acid | 309 ± 6.1 | LRI, MS, Std |
| 8 | 1954 | (E)-3-Hexenoic acid | 516 ± 13.8 | LRI, MS, Std |
| 9 | 1960 | Heptanoic acid | 126 ± 4.1 | LRI, MS, Std |
| 10 | 2047 | Octanoic acid | 112 ± 3.8 | LRI, MS, Std |
| 11 | 2178 | Nonanoic acid | 95 ± 4.5 | LRI, MS, Std |
| 12 | 2930 | Hexadecanoic acid | 346 ± 2.5 | LRI, MS, Std |
| 13 | 3181 | Octadecanoic acid | 116 ± 2.1 | LRI, MS, Std |
| Total | 6574 | |||
| Alcohols | ||||
| 14 | 1121 | 2-Methyl-3-butanol | 2627 ± 7.8 | LRI, MS, Std |
| 15 | 1182 | 3-Penten-2-ol | 394 ± 2.7 | LRI, MS, Std |
| 16 | 1332 | (Z)-2-Methyl-2-buten-1-ol | 110 ± 2.2 | LRI, MS, Std |
| 17 | 1651 | Furfuryl alcohol | 227 ± 4.7 | LRI, MS, Std |
| 18 | 1796 | 2-(2-Ethoxyethoxy) ethanol | 9 ± 0.1 | LRI, MS, tent |
| 19 | 1881 | Benzyl alcohol | 123 ± 1.9 | LRI, MS, Std |
| 20 | 1916 | Phenethyl alcohol | 131 ± 0.0 | LRI, MS, Std |
| Total | 3621 | |||
| Phenols | ||||
| 21 | 1873 | Guaiacol | 12 ± 0.1 | LRI, MS, Std |
| 22 | 1989 | Phenol | 125 ± 1.8 | LRI, MS, Std |
| 23 | 2158 | Eugenol | 89 ± 1.1 | LRI, MS, Std |
| Total | 226 | |||
| Terpene | ||||
| 30 | 1205 | Limonene | 176 ± 0.3 | LRI, MS, Std |
| Total | 176 | |||
| Esters | ||||
| 24 | 1740 | Benzyl acetate | 46 ± 3.6 | LRI, MS, Std |
| 25 | 2593 | Ethyl 4-ethoxybenzoate | 85 ± 2.0 | LRI, MS, tent |
| Total | 131 | |||
| Ketones | ||||
| 26 | 1320 | Acetoin | 9394 ± 87 | LRI, MS, Std |
| 27 | 1326 | Hydroxyacetone | 808 ± 24.7 | LRI, MS, Std |
| Total | 10,202 | |||
| Aldehyde | ||||
| 28 | 1470 | Furfural | 144 ± 3.1 | LRI, MS, Std |
| Total | 144 | |||
| Lactone | ||||
| 29 | 1614 | Butyrolactone | 48 ± 0.5 | LRI, MS, Std |
| Total | 48 | |||
| Hydrocarbons | ||||
| 31 | 1132 | M-xylene | 357 ± 17.6 | LRI, MS, tent |
| 32 | 1270 | Styrene | 65 ± 1.4 | LRI, MS, tent |
| Total | 422 | |||
| General Total | 21,544 |
| No. | Aroma-Active Compounds | LRI a | Odor Descriptions b | FD c | OT (ppm) d | OAV e |
|---|---|---|---|---|---|---|
| 1 | Unknown I | 1158 | Burnt plastic | 16 | - | - |
| 2 | 3-Penten-2-ol | 1182 | Green | 4 | 0.4 [25] | 1.0 |
| 3 | Limonene | 1205 | Citrusy | 256 | 0.01 [26] | 17.6 |
| 4 | Styrene | 1270 | Balsamic | 256 | 0.0036 [27] | 18.2 |
| 5 | Acetoin | 1320 | Buttery | 1024 | 0.055 [28] | 170.8 |
| 6 | Unknown II | 1394 | Putrid fruit | 32 | - | - |
| 7 | Propanoic acid | 1538 | Fatty | 128 | 0.1 [29] | 11.9 |
| 8 | Isobutanoic acid | 1562 | Cheesy | 512 | 0.01 [29] | 22.0 |
| 9 | Pentanoic acid | 1731 | Cheesy | 32 | 0.07 [30] | 2.5 |
| 10 | Hexanoic acid | 1854 | Cheesy | 64 | 0.093 [31] | 3.3 |
| 11 | Guaiacol | 1873 | Smoky | 64 | 0.003 [32] | 3.8 |
| 12 | Phenethyl alcohol | 1916 | Honey, floral | 16 | 0.086 [28] | 1.5 |
| 13 | Eugenol | 2158 | Spicy-smoky | 128 | 0.006 [30] | 14.8 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kesen, S.; Amanpour, A.; Tsouli Sarhir, S.; Sevindik, O.; Guclu, G.; Kelebek, H.; Selli, S. Characterization of Aroma-Active Compounds in Seed Extract of Black Cumin (Nigella sativa L.) by Aroma Extract Dilution Analysis. Foods 2018, 7, 98. https://doi.org/10.3390/foods7070098
Kesen S, Amanpour A, Tsouli Sarhir S, Sevindik O, Guclu G, Kelebek H, Selli S. Characterization of Aroma-Active Compounds in Seed Extract of Black Cumin (Nigella sativa L.) by Aroma Extract Dilution Analysis. Foods. 2018; 7(7):98. https://doi.org/10.3390/foods7070098
Chicago/Turabian StyleKesen, Songul, Armin Amanpour, Salwa Tsouli Sarhir, Onur Sevindik, Gamze Guclu, Hasim Kelebek, and Serkan Selli. 2018. "Characterization of Aroma-Active Compounds in Seed Extract of Black Cumin (Nigella sativa L.) by Aroma Extract Dilution Analysis" Foods 7, no. 7: 98. https://doi.org/10.3390/foods7070098
APA StyleKesen, S., Amanpour, A., Tsouli Sarhir, S., Sevindik, O., Guclu, G., Kelebek, H., & Selli, S. (2018). Characterization of Aroma-Active Compounds in Seed Extract of Black Cumin (Nigella sativa L.) by Aroma Extract Dilution Analysis. Foods, 7(7), 98. https://doi.org/10.3390/foods7070098






