Microbiological Parameters in the Primary Production of Berries: A Pilot Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Collection of Data and Sampling Strategy
2.2. Microbiological Analysis and RNA Extraction
2.3. Two Step Reverse Transcription Seminested-PCR (RT-PCR) and Quantitative Real-Time Reverse Transcription-PCR (Real-Time RT-PCR)
2.4. Statistical Analysis
3. Results
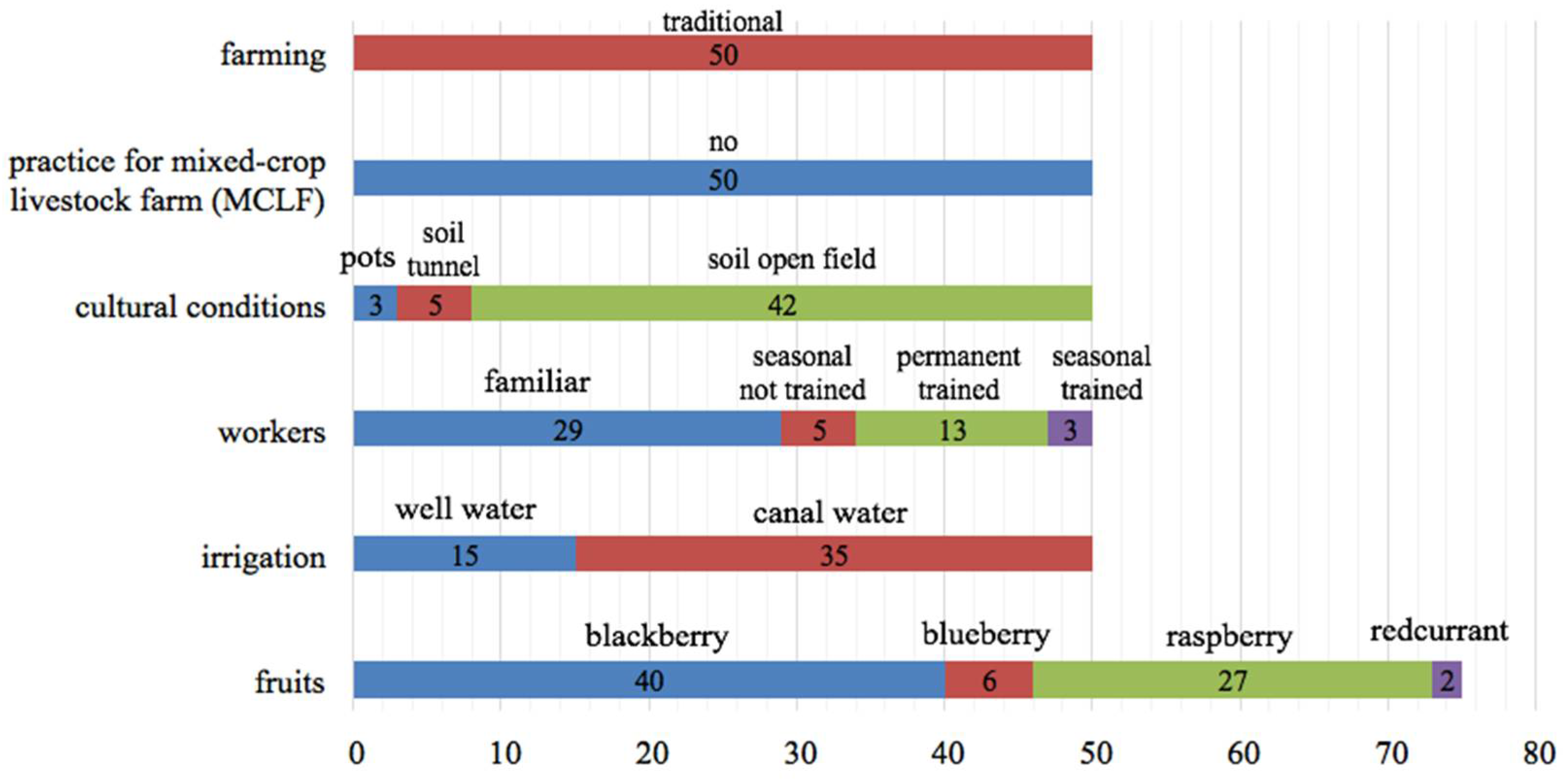
3.1. Collection of Data and Sampling Strategy
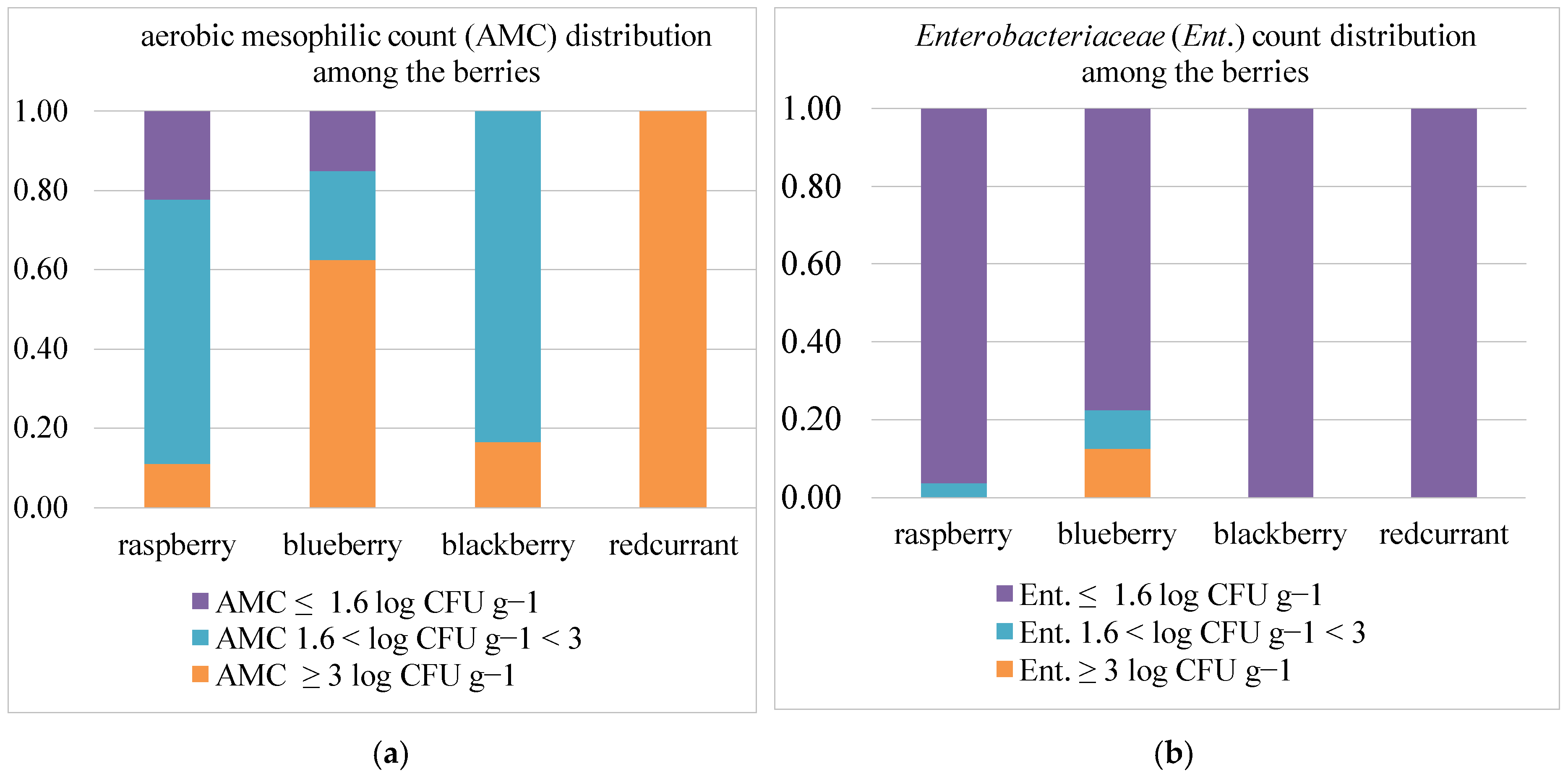
3.2. Microbiological Measurements and Risks Related to Crop Management
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fitzgerald, M.; Thornton, L.; O’Gorman, J.; O’Connor, L.; Garvey, P.; Boland, M.; Part, A.M.; Rogalska, J.; Coughlan, H.; MacDiarmada, J.; et al. Hepatitis A Outbreak Control Team. Outbreak of hepatitis A infection associated with the consumption of frozen berries, Ireland, 2013—Linked to an international outbreak. Eurosurveillance 2014, 19, 20942. [Google Scholar] [CrossRef] [PubMed]
- Brazelton, C. World Blueberry Acreage & Production. North American Blueberry Council. Available online: http://www.chilealimentos.com/2013/phocadownload/Aprocesados_congelados/nabc_2012-world-blueberry-acreage-production.pdf (accessed on 3 June 2018).
- Milivojevic, J.; Slatnar, A.; Mikulic-Petkovsek, M.; Stampar, F.; Nikolic, M.; Veberic, R. The influence of early yield on the accumulation of major taste and health-related compounds in black and red currant cultivars (Ribes spp.). J. Agric. Food. Chem. 2012, 14, 2682–2691. [Google Scholar] [CrossRef] [PubMed]
- FAO. Faostat Statistics Database. Available online: http://www.fao.org/faostat/en/ (accessed on 3 June 2018).
- Knudsen, D.M.; Yamamoto, S.A.; Harris, L.J. Survival of Salmonella spp. and Escherichia coli O157:H7 on fresh and frozen strawberries. J. Food. Prot. 2001, 64, 1483–1488. [Google Scholar] [CrossRef] [PubMed]
- Holden, N.; Pritchard, L.; Toth, I. Colonization outwith the colon: Plants as an alternative environmental reservoir for human pathogenic enterobacteria. FEMS Microbiol. Rev. 2009, 33, 689–703. [Google Scholar] [CrossRef] [PubMed]
- Lynch, M.F.; Tauxe, R.V.; Hedberg, C.W. The growing burden of foodborne outbreaks due to contaminated fresh produce: Risks and opportunities. Epidemiol. Infect. 2009, 137, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Berger, C.N.; Sodha, S.V.; Shaw, R.K.; Griffin, P.M.; Pink, D.; Hand, P.; Frankel, G. Fresh fruit and vegetables as vehicles for the transmission of human pathogens. Environ. Microbiol. 2010, 12, 2385–2397. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maunula, L.; Roivainen, M.; Keranen, M.; Makela, S.; Soderberg, K.; Summa, M.; von Bonsdorff, C.H.; Lappalainen, M.; Korhonen, T.; Kuusi, M.; et al. Detection of human norovirus from frozen raspberries in a cluster of gastroenteritis outbreaks. Eurosurveillance 2009, 14, 19435. [Google Scholar] [PubMed]
- Einoder-Moreno, M.; Lange, H.; Grepp, M.; Osborg, E.; Vainio, K.; Vold, L. Non-heat-treated frozen raspberries the most likely vehicle of a norovirus outbreak in Oslo, Norway, November 2013. Epidemiol. Infect. 2016, 144, 2765–2772. [Google Scholar] [CrossRef] [PubMed]
- Collier, M.G.; Khudyakov, Y.E.; Selvage, D.; Adams-Cameron, M.; Epson, E.; Cronquist, A.; Jervis, R.H.; Lamba, K.; Kimura, A.C.; Sowadsky, R.; et al. Outbreak of hepatitis a in the USA associated with frozen pomegranate arils imported from Turkey: An epidemiological case study. Lancet Infect. Dis. 2014, 14, 976–981. [Google Scholar] [CrossRef]
- Falkenhorst, G.; Krusell, L.; Lisby, M.; Madsen, S.B.; Bottiger, B.; Molbak, K. Imported frozen raspberries cause a series of norovirus outbreaks in Denmark, 2005. Eurosurveillance 2005, 10, 2795. [Google Scholar] [CrossRef]
- Hjertqvist, M.; Johansson, A.; Svensson, N.; Abom, P.E.; Magnusson, C.; Olsson, M.; Hedlund, K.O.; Andersson, Y. Four outbreaks of norovirus gastroenteritis after consuming raspberries, Sweden, June-August 2006. Eurosurveillance 2006, 11, 3038. [Google Scholar] [CrossRef]
- Ponka, A.; Maunula, L.; Von Bonsdorff, C.H.; Lyytikainen, O. Outbreak of calicivirus gastroenteritis associated with eating frozen raspberries. Eurosurveillance 1999, 4, 66–69. [Google Scholar] [CrossRef] [PubMed]
- Bruni, R.; Taffon, S.; Equestre, M.; Chionne, P.; Madonna, E.; Rizzo, C.; Tosti, M.E.; Alfonsi, V.; Ricotta, L.; De Medici, D.; et al. Key role of sequencing to trace hepatitis a viruses circulating in Italy during a large multi-country European foodborne outbreak in 2013. PLoS ONE 2016, 11, e0149642. [Google Scholar] [CrossRef] [PubMed]
- Gossner, C.M.; Severi, E. Three simultaneous, food-borne, multi-country outbreaks of hepatitis a virus infection reported in epis-fwd in 2013: What does it mean for the European Union? Eurosurveillance 2014, 19, 20941. [Google Scholar] [CrossRef] [PubMed]
- Scavia, G.; Alfonsi, V.; Taffon, S.; Escher, M.; Bruni, R.; Medici, D.; Pasquale, S.D.; Guizzardi, S.; Cappelletti, B.; Iannazzo, S.; et al. A large prolonged outbreak of hepatitis A associated with consumption of frozen berries, Italy, 2013–14. J. Med. Microbiol. 2017, 66, 342–349. [Google Scholar] [CrossRef] [PubMed]
- Alegbeleye, O.O.; Singleton, I.; Sant’Ana, A.S. Sources and contamination routes of microbial pathogens to fresh produce during field cultivation: A review. Food Microbiol. 2018, 73, 177–208. [Google Scholar] [CrossRef] [PubMed]
- Beuchat, L.R.; Ryu, J.H. Produce handling and processing practices. Emerg. Infect. Dis. 1997, 4, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Franz, E.; van Diepeningen, A.D.; de Vos, O.J.; van Bruggen, A.H. Effects of cattle feeding regimen and soil management type on the fate of Escherichia coli O157:H7 and Salmonella enterica serovar typhimurium in manure, manure-amended soil, and lettuce. Appl. Environ. Microbiol. 2005, 71, 6165–6174. [Google Scholar] [CrossRef] [PubMed]
- Laidler, M.R.; Tourdjman, M.; Buser, G.L.; Hostetler, T.; Repp, K.K.; Leman, R.; Samadpour, M.; Keene, W.E. Escherichia coli O157:H7 infections associated with consumption of locally grown strawberries contaminated by deer. Clin. Infect. Dis. 2013, 57, 1129–1134. [Google Scholar] [CrossRef] [PubMed]
- Ferens, W.A.; Hovde, C.J. Escherichia coli O157:H7: Animal reservoir and sources of human infection. Foodborne Pathog. Dis. 2011, 8, 465–487. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, C.; de Roda Husman, A.M.; Altavilla, N.; Deere, D.; Ashbolt, N. Fate and transport of surface water pathogens in watersheds. Crit. Rev. Environ. Sci. Technol. 2003, 33, 299–361. [Google Scholar] [CrossRef]
- Fonseca, J.M.; Fallon, S.D.; Sanchez, C.A.; Nolte, K.D. Escherichia coli survival in lettuce fields following its introduction through different irrigation systems. J. Appl. Microbiol. 2011, 110, 893–902. [Google Scholar] [CrossRef] [PubMed]
- Delbeke, S.; Ceuppens, S.; Hessel, C.T.; Castro, I.; Jacxsens, L.; De Zutter, L.; Uyttendaele, M. Microbial safety and sanitary quality of strawberry primary production in Belgium: Risk factors for Salmonella and Shiga toxin-producing Escherichia coli contamination. Appl. Environ. Microbiol. 2015, 81, 2562–2570. [Google Scholar] [CrossRef] [PubMed]
- Chitarra, W.; Decastelli, L.; Garibaldi, A.; Gullino, M.L. Potential uptake of Escherichia coli O157:H7 and Listeria monocytogenes from growth substrate into leaves of salad plants and basil grown in soil irrigated with contaminated water. Int. J. Food. Microbiol. 2014, 189, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Maunula, L.; Kaupke, A.; Vasickova, P.; Soderberg, K.; Kozyra, I.; Lazic, S.; van der Poel, W.H.; Bouwknegt, M.; Rutjes, S.; Willems, K.A.; et al. Tracing enteric viruses in the European berry fruit supply chain. Int. J. Food. Microbiol. 2013, 167, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Salaheen, S.; Chowdhury, N.; Hanning, I.; Biswas, D. Zoonotic bacterial pathogens and mixed crop-livestock farming. Poult. Sci. 2015, 94, 1398–1410. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bengtsson, J.; Ahnström, J.; Weibull, A.-C. The effects of organic agriculture on biodiversity and abundance: A meta-analysis. J. Appl. Ecol. 2005, 42, 261–269. [Google Scholar] [CrossRef]
- EFSA Panel on Biological Hazards (BIOHAZ). Scientific Opinion on the risk posed by pathogens in food of non-animal origin. Part 2 (Salmonella and Norovirus in berries). EFSA J. 2014, 12, 3706. [Google Scholar]
- Hamilton, K.E.; Umber, J.; Hultberg, A.; Tong, C.; Schermann, M.; Diez-Gonzalez, F.; Bender, J.B. Validation of Good Agricultural Practices (GAP) on Minnesota vegetable farms. Foodborne Pathog. Dis. 2015, 12, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Lauer, W.F.; Sidi, C.D.; Tourniaire, J.P.; Hammack, T. IQ-Check Salmonella II: Real-time polymerase chain reaction test kit. J. AOAC Int. 2009, 92, 1865–1870. [Google Scholar] [PubMed]
- Afnor Certification for IQ-Check: Real-Time Polymerase Chain Reaction Test Kit for Salmonella II. Available online: https://nf-validation.afnor.org/wp-content/uploads/sites/2/2014/03/BRD-07-06-07-04_en.pdf (accessed on 4 July 2018).
- International Organization for Standardization. Microbiology of Food and Animal Feeding Stuffs—Horizontal Method for the Detection of Salmonella spp.; ISO 6579-1:2017; ISO: Geneva, Switzerland, 2017. [Google Scholar]
- Afnor Certification forI-Check: Real-Time Polymerase Chain Reaction Test Kit for Listeria monocytogenes in All Human Food Products. Available online: https://nf-validation.afnor.org/wp-content/uploads/sites/2/2014/03/BRD-07-10-04-05_en.pdf (accessed on 4 July 2018).
- International Organization for Standardization. Microbiology of the Food Chain—Horizontal Method for the Detection and Enumeration of Listeria monocytogenes and of Listeria spp.—Part 1: Detection Method; ISO 11290-1:2017; ISO: Geneva, Switzerland, 2017. [Google Scholar]
- International Organization for Standardization. Microbiology of Food and Animal Feed—Real-Time Polymerase Chain Reaction (PCR)-Based Method for the Detection of Food-Borne Pathogens—Horizontal Method for the Detection of Shiga Toxin-Producing Escherichia coli (STEC) and the Determination of O157, O111, O26, O103 and O145 Serogroups; ISO/TS 13136:2012; ISO: Geneva, Switzerland, 2012. [Google Scholar]
- International Organization for Standardization. Microbiology of the Food Chain—Horizontal Method for the Enumeration of Microorganisms—Part 1: Colony Count at 30 Degrees C by the Pour Plate Technique; ISO 4833-1:2013; ISO: Geneva, Switzerland, 2013. [Google Scholar]
- International Organization for Standardization. Microbiology of Food and Animal Feeding Stuffs—Horizontal Method for the Detection and Enumeration of Coliforms—Most Probable Number Technique; ISO 4831:2006; ISO: Geneva, Switzerland, 2006. [Google Scholar]
- International Organization for Standardization. Microbiology of Food and Animal Feeding Stuffs—Horizontal Method for the Detection and Enumeration of Presumptive Escherichia coli—Most Probable Number Technique; ISO 7251:2005; ISO: Geneva, Switzerland, 2005. [Google Scholar]
- International Organization for Standardization. Microbiology of Food and Animal Feed—Horizontal Method for Determination of Hepatitis A Virus and Norovirus in Food Using Real-Time RT-PCR—Part 2: Method for Qualitative Detection; ISO/TS 15216-2:2013; ISO: Geneva, Switzerland, 2013. [Google Scholar]
- Terio, V.; Bottaro, M.; Di Pinto, A.; Catella, C.; Chironna, M.; Bozzo, G.; Kingsley, D.H.; Bonerba, E.; Morea, A.; Martella, V. Outbreak of hepatitis a in Italy associated with frozen redcurrants imported from poland: A case study. Food. Environ. Virol. 2015, 7, 305–308. [Google Scholar] [CrossRef] [PubMed]
- Pinto, R.M.; Costafreda, M.I.; Bosch, A. Risk assessment in shellfish-borne outbreaks of hepatitis a. Appl. Environ. Microbiol. 2009, 75, 7350–7355. [Google Scholar] [CrossRef] [PubMed]
- Ragaert, P.; Devlieghere, F.; Loos, S.; Dewulf, J.; Van Langenhove, H.; Foubert, I.; Vanrolleghem, P.; Debevere, J. Microbiological and physiological processes affecting odor quality of strawberries during storage. Commun. Agric. Appl. Biol. Sci. 2004, 69, 227–230. [Google Scholar] [PubMed]
- Gómez-López, V.M. Decontamination of Fresh and Minimally Processed Produce; John Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar]
- EFSA Panel on Biological Hazards (BIOHAZ); Ricci, A.; Allende, A.; Bolton, D.; Chemaly, M.; Davies, R.; Fernández Escámez, P.S.; Girones, R.; Herman, L.; Koutsoumanis, K.; et al. Guidance on the requirements for the development of microbiological criteria. EFSA J. 2017, 11, e05052. [Google Scholar]
- Xu, W.; Wu, C. The impact of pulsed light on decontamination, quality, and bacterial attachment of fresh raspberries. Food Microbiol. 2016, 57, 135–143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Parameter | Method | Reference |
|---|---|---|
| Salmonella spp. | Real-time polymerase chain reaction (Real-time PCR) | [32,33] |
| Culture technique | [34] | |
| Listeria monocytogenes | Real-time PCR | [35] |
| Culture technique | [36] | |
| Shiga Toxin-producing E. coli (STEC) | Real-time PCR | [37] |
| Culture technique | [37] | |
| Aerobic Mesophilic Count | Culture technique | [38] |
| Enterobacteriaceae | Culture technique | [39] |
| Escherichia coli | Culture technique | [40] |
| hepatitis A virus (HAV) and norovirus genogroup I and II (Nov GGI/GGII) | Real time-PCR | [41,42] |
| Farm Characteristics and Results of the Questionnaires | Results Microbiology Analysis (CFU/g) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Id | Prod | Fruit | Farm Size | Irrigation | Workers | Cultural Conditions | AMC | E. coli | Ent. |
| 1 | 1 | blackberry | B | well water | ST | soil open field | 14,000 | <10 | 1500 |
| 2 | 1 | blackberry | B | well water | ST | soil open field | 1500 | <10 | <10 |
| 3 | 2 | blackberry | B | well water | ST | soil open field | 130 | <10 | <10 |
| 4 | 2 | blackberry | B | well water | ST | soil open field | 1200 | <10 | <40 |
| 5 | 3 | raspberry | B | well water | ST | soil open field | 2900 | <10 | <10 |
| 6 | 3 | raspberry | B | well water | ST | soil open field | 760 | <10 | <10 |
| 7 | 4 | blackberry | B | canal water | fam | soil open field | 9100 | <10 | <40 |
| 8 | 4 | blackberry | B | canal water | fam | soil open field | 1000 | <10 | <10 |
| 9 | 5 | blackberry | S | canal water | fam | soil open field | 290 | <10 | <10 |
| 10 | 6 | raspberry | S | canal water | fam | soil open field | 320 | <10 | <10 |
| 11 | 7 | blueberry | S | canal water | fam | soil open field | 580 | <10 | <10 |
| 12 | 8 | red currant | S | canal water | SNT | soil open field | 2900 | <10 | <10 |
| 13 | 9 | raspberry | B | canal water | SNT | soil open field | 160 | <10 | <10 |
| 14 | 9 | raspberry | B | canal water | SNT | soil open field | 250 | <10 | <10 |
| 15 | 10 | raspberry | B | canal water | fam | Pots | 310 | <10 | <10 |
| 16 | 10 | raspberry | B | canal water | fam | Pots | <10 | <10 | <10 |
| 17 | 11 | blackberry | B | canal water | SNT | soil tunnel | 150 | <10 | <10 |
| 18 | 11 | blackberry | B | canal water | SNT | soil tunnel | <40 | <10 | <10 |
| 19 | 12 | blackberry | S | canal water | fam | soil open field | <10 | <10 | <10 |
| 20 | 13 | blackberry | B | canal water | fam | soil open field | <40 | <10 | <10 |
| 21 | 13 | blackberry | B | canal water | fam | soil open field | 2200 | <10 | <10 |
| 22 | 14 | blackberry | S | canal water | fam | soil open field | 1500 | <10 | <40 |
| 23 | 15 | blackberry | S | canal water | fam | soil open field | 1000 | <10 | <10 |
| 24 | 16 | blueberry | B | canal water | fam | soil open field | 98,000 | <10 | <10 |
| 25 | 16 | blueberry | B | canal water | fam | soil open field | <400 | <10 | <10 |
| 26 | 17 | blackberry | B | canal water | PT | soil open field | 600 | <10 | <10 |
| 27 | 17 | blackberry | B | canal water | PT | soil open field | 35,000 | 60 | 1500 |
| 28 | 18 | raspberry | S | canal water | fam | soil open field | 700 | <10 | <10 |
| 29 | 19 | raspberry | S | canal water | fam | Pots | 800 | <10 | <10 |
| 30 | 20 | blackberry | S | well water | fam | soil open field | 700 | <10 | <10 |
| 31 | 21 | red currant | S | canal water | fam | soil open field | 8800 | <10 | <10 |
| 32 | 22 | blackberry | S | canal water | fam | soil open field | 5200 | <10 | <10 |
| 33 | 23 | raspberry | S | canal water | fam | soil open field | 50 | <10 | <10 |
| 34 | 24 | blackberry | B | canal water | PT | soil tunnel | 260 | <10 | <10 |
| 35 | 24 | blackberry | B | canal water | PT | soil tunnel | 18,000 | <10 | <40 |
| 36 | 25 | raspberry | S | canal water | SNT | soil open field | 50 | <10 | <10 |
| 37 | 26 | blackberry | B | canal water | fam | soil open field | 22,000 | <10 | <10 |
| 38 | 26 | blackberry | B | canal water | fam | soil open field | 1400 | <10 | <40 |
| 39 | 27 | blackberry | S | canal water | fam | soil open field | 17,000 | <10 | <10 |
| 40 | 28 | blackberry | S | well water | fam | soil open field | 1500 | <10 | <40 |
| 41 | 29 | raspberry | S | canal water | fam | soil open field | 660 | <10 | <10 |
| 42 | 30 | raspberry | B | canal water | fam | soil tunnel | 500 | <10 | <10 |
| 43 | 30 | raspberry | B | canal water | fam | soil tunnel | 100 | <10 | <10 |
| 44 | 31 | raspberry | B | canal water | fam | soil open field | 280 | <10 | <10 |
| 45 | 31 | raspberry | B | canal water | fam | soil open field | <10 | <10 | <10 |
| 55 | 32 | raspberry | B | well water | fam | soil open field | 100 | <10 | <10 |
| 46 | 32 | raspberry | B | canal water | fam | soil open field | <10 | <10 | <10 |
| 47 | 33 | raspberry | S | canal water | fam | soil open field | <10 | <10 | <10 |
| 48 | 34 | blackberry | S | canal water | SNT | soil open field | <10 | <10 | <10 |
| 49 | 35 | raspberry | S | canal water | fam | soil open field | 600 | <10 | <10 |
| 50 | 36 | blackberry | S | canal water | fam | soil open field | 1000 | <10 | <10 |
| 51 | 37 | raspberry | S | canal water | fam | soil open field | 1900 | <10 | <400 |
| 52 | 38 | blackberry | S | canal water | fam | Pots | 250 | <10 | <10 |
| 53 | 39 | blackberry | B | well water | PT | soil open field | 43,000 | <10 | 9900 |
| 54 | 39 | blackberry | B | well water | fam | soil open field | 2400 | <10 | <10 |
| 56 | 40 | blackberry | B | canal water | PT | soil open field | 9600 | <10 | <10 |
| 57 | 40 | blackberry | B | canal water | PT | soil open field | 8,100,000 | 45,000 | 50,000 |
| 58 | 41 | blackberry | B | canal water | PT | soil open field | 1500 | <10 | <40 |
| 59 | 41 | blackberry | B | canal water | PT | soil open field | 43,000 | <10 | 150 |
| 60 | 42 | blackberry | S | well water | fam | soil tunnel | 8600 | <10 | <40 |
| 61 | 43 | raspberry | B | canal water | PT | soil open field | 4600 | <10 | <10 |
| 62 | 43 | raspberry | B | canal water | PT | soil open field | 900 | <10 | <10 |
| 63 | 44 | blackberry | S | well water | PT | soil open field | 120 | <10 | 210 |
| 64 | 45 | raspberry | B | well water | PT | soil open field | <10 | <10 | <10 |
| 65 | 45 | raspberry | B | well water | PT | soil open field | <10 | <10 | <10 |
| 66 | 46 | blackberry | B | well water | PT | soil open field | 100 | <10 | <10 |
| 67 | 46 | blueberry | B | well water | PT | soil open field | 180 | <10 | <10 |
| 68 | 47 | blackberry | B | well water | PT | soil tunnel | <10 | <10 | <10 |
| 69 | 47 | blackberry | B | well water | PT | soil tunnel | 20,000 | <10 | 700 |
| 70 | 48 | blackberry | B | well water | PT | soil open field | <10 | <10 | <10 |
| 71 | 48 | blackberry | B | well water | PT | soil open field | 12,000 | <10 | 3800 |
| 72 | 49 | raspberry | B | well water | PT | soil open field | <400 | <10 | <10 |
| 73 | 49 | raspberry | B | well water | PT | soil open field | 180 | <10 | <10 |
| 74 | 50 | blueberry | B | well water | PT | soil open field | 110 | <10 | <10 |
| 75 | 50 | blueberry | B | well water | PT | soil open field | 410 | <10 | <10 |
| Bacteria | Median | Mean | SD | Range | Max |
|---|---|---|---|---|---|
| Log Aerobic Mesophilic Count | 2.778 | 2.801 | 1.140 | 5.910 | 6.908 |
| Log Enterobacteriaceae | 1.000 | 1.059 | 0.430 | 3.650 | 4.653 |
| Log Escherichia coli | 1.000 | 1.325 | 0.760 | 3.700 | 4.699 |
| Factor | Level | N | Ent. | % | χ2 | Df | p-Value |
|---|---|---|---|---|---|---|---|
| Berry | Blackberry | 56 | 16 | 28.6% | 12.649 | 1 | 0.000 |
| Others | 19 | 1 | 5.3% | ||||
| Workers | Trained | 22 | 11 | 50.0% | 3.278 | 1 | 0.070 |
| Not Trained | 36 | 6 | 16.7% | ||||
| Farm Size | Small | 25 | 5 | 20.0% | 0.010 | 1 | 0.922 |
| Big | 50 | 12 | 24.0% | ||||
| Water Type and Irrigation | Canal water | 50 | 9 | 18.0% | 1.150 | 1 | 0.283 |
| Well water | 25 | 8 | 32.0% | ||||
| Cultural Conditions | Pots | 4 | 0 | 0.0% | 1.757 | 2 | 0.415 |
| Soil Open Field | 62 | 14 | 22.6% | ||||
| Soil Tunnel | 9 | 3 | 33.3% |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Macori, G.; Gilardi, G.; Bellio, A.; Bianchi, D.M.; Gallina, S.; Vitale, N.; Gullino, M.L.; Decastelli, L. Microbiological Parameters in the Primary Production of Berries: A Pilot Study. Foods 2018, 7, 105. https://doi.org/10.3390/foods7070105
Macori G, Gilardi G, Bellio A, Bianchi DM, Gallina S, Vitale N, Gullino ML, Decastelli L. Microbiological Parameters in the Primary Production of Berries: A Pilot Study. Foods. 2018; 7(7):105. https://doi.org/10.3390/foods7070105
Chicago/Turabian StyleMacori, Guerrino, Giovanna Gilardi, Alberto Bellio, Daniela Manila Bianchi, Silvia Gallina, Nicoletta Vitale, Maria Lodovica Gullino, and Lucia Decastelli. 2018. "Microbiological Parameters in the Primary Production of Berries: A Pilot Study" Foods 7, no. 7: 105. https://doi.org/10.3390/foods7070105
APA StyleMacori, G., Gilardi, G., Bellio, A., Bianchi, D. M., Gallina, S., Vitale, N., Gullino, M. L., & Decastelli, L. (2018). Microbiological Parameters in the Primary Production of Berries: A Pilot Study. Foods, 7(7), 105. https://doi.org/10.3390/foods7070105






