Lactic Acid Bacteria from Kefir Increase Cytotoxicity of Natural Killer Cells to Tumor Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Cell Culture
2.3. Preparation of Lactic Acid Bacteria Mixture
2.4. Measurement of NK Cell Activity and Tumor Cytotoxicity
2.5. RT-PCR (Reverse Transcription Polymerase Chain Reaction)
2.6. IFN-γ Measurement
2.7. Statistical Analysis
3. Results and Discussion
3.1. Induction of NK Activity by the Six Lactic Acid Bacteria from Homemade-Kefir
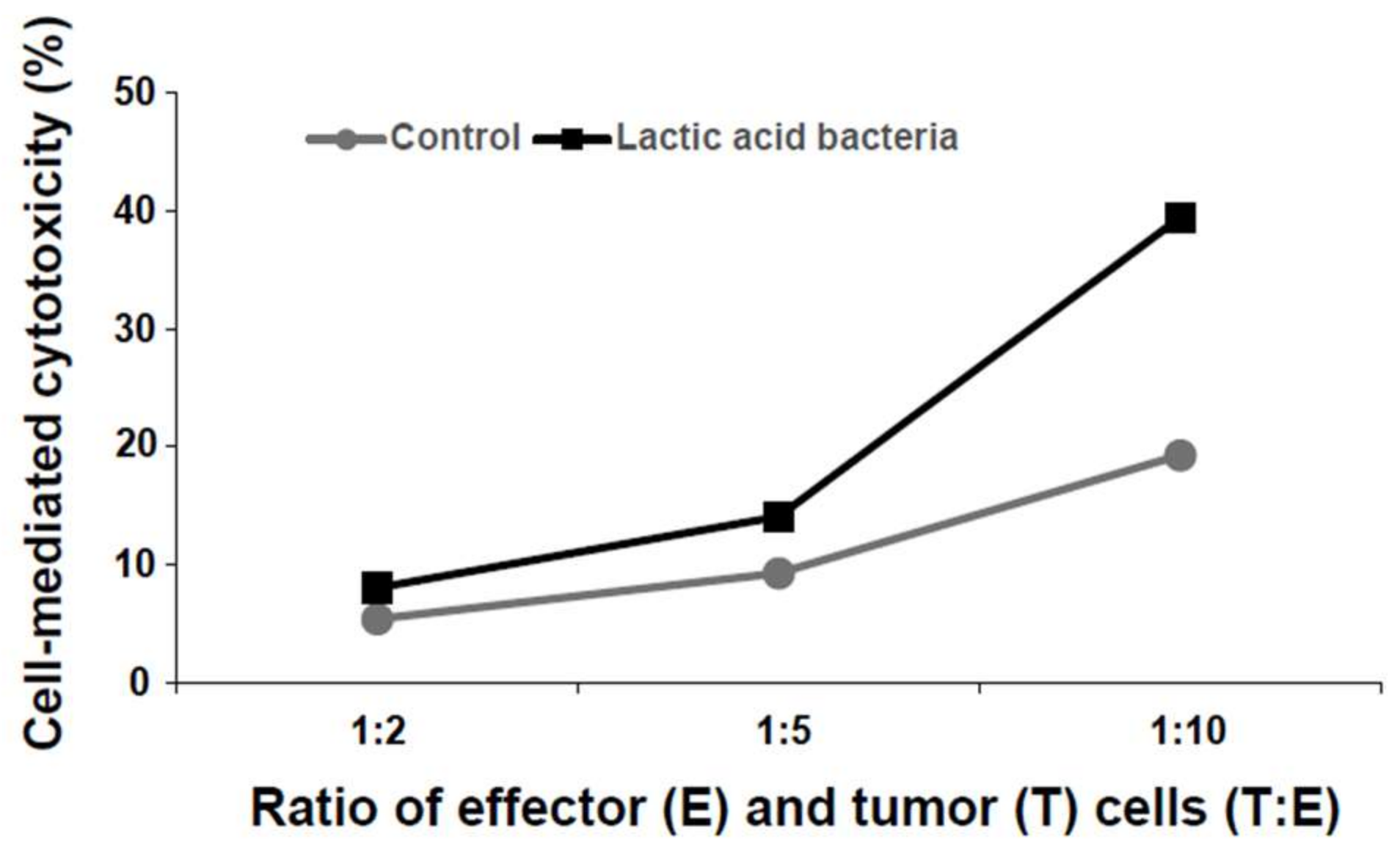
3.2. Induction of Cytotoxicity by the Six Lactic Acid Bacteria Against Human Colorectal Tumor Cells
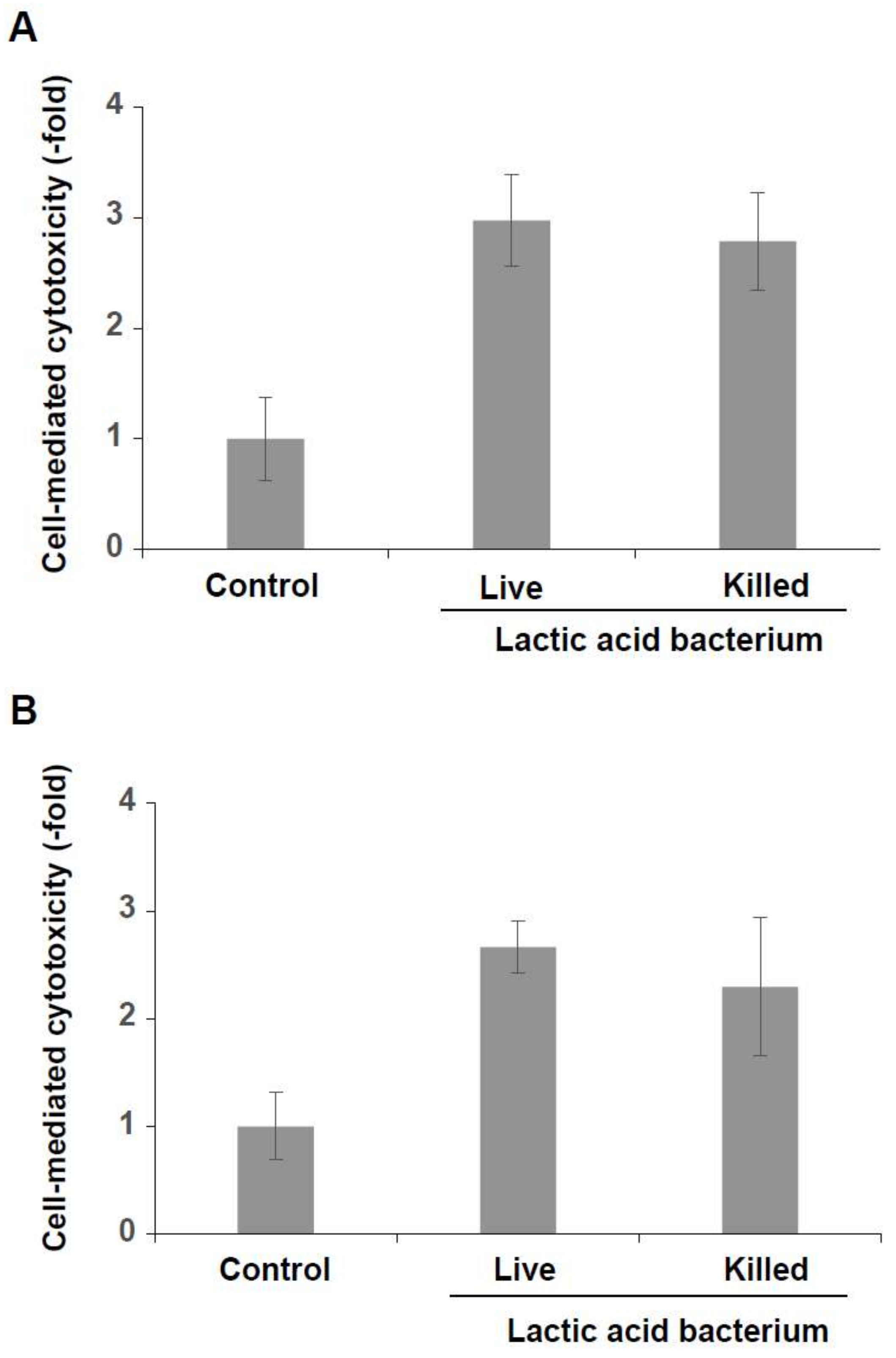
3.3. Induction of Cytotoxicity of KHYG-1 Cells by the Killed Lactic Acid Bacteria Mixture
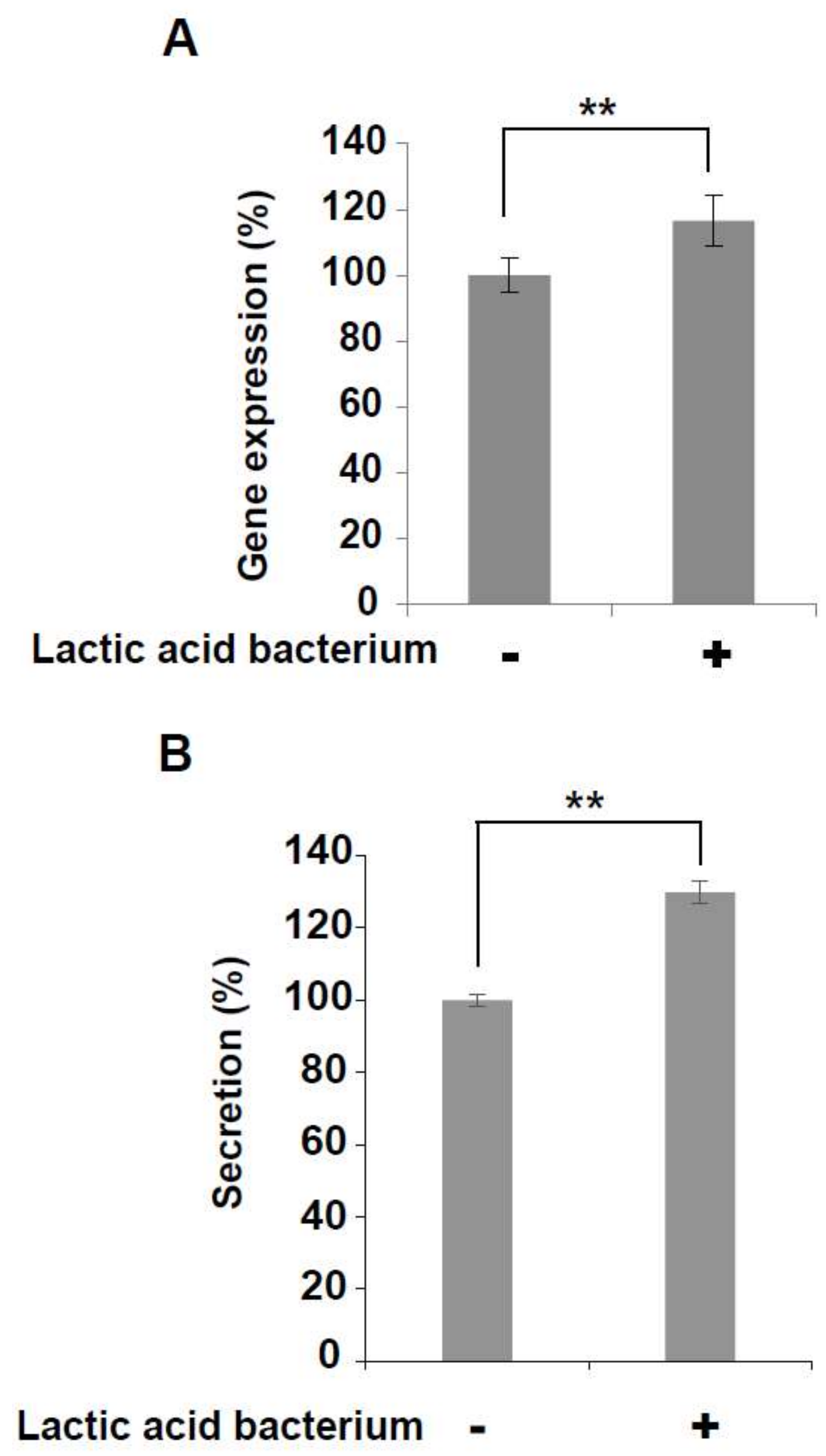
3.4. Increased Expression and Secretion Levels of IFN-γ in KHYG-1 Cells
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Bourrie, B.C.; Willing, B.P.; Cotter, P.D. The Microbiota and Health Promoting Characteristics of the Fermented Beverage Kefir. Front. Microbiol. 2016, 7, 647. [Google Scholar] [CrossRef] [PubMed]
- Angulo, L.; Lopez, E.; Lema, C. Microflora present in kefir grains of the Galician region (North-West of Spain). J. Dairy Res. 1993, 60, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Pintado, M.E.; Da Silva, J.A.L.; Fernandes, P.B.; Malcata, F.X.; Hogg, T.A. Microbiological and rheological studies on Portuguese kefir grains. Int. J. Food Sci. Technol. 1996, 31, 15–26. [Google Scholar] [CrossRef]
- Garrote, G.L.; Abraham, A.G.; De Antoni, G.L. Chemical and microbiological characterisation of kefir grains. J. Dairy Res. 2001, 68, 639–652. [Google Scholar] [CrossRef] [PubMed]
- Simova, E.; Beshkova, D.; Angelov, A.; Hristozova, T.; Frengova, G.; Spasov, Z. Lactic acid bacteria and yeasts in kefir grains and kefir made from them. J. Ind. Microbiol. Biotechnol. 2002, 28, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Witthuhn, R.C.; Schoeman, T.; Britz, T.J. Isolation and characterization of the microbial population of different South African kefir grains. Int. J. Dairy Technol. 2004, 57, 33–37. [Google Scholar] [CrossRef]
- Witthuhn, R.; Schoeman, T.; Britz, T. Characterisation of the microbial population at different stages of Kefir production and Kefir grain mass cultivation. Int. Dairy J. 2005, 15, 383–389. [Google Scholar] [CrossRef]
- Yüksekdağ, Z.; Beyatli, Y.; Aslim, B. Determination of some characteristics coccoid forms of lactic acid bacteria isolated from Turkish kefirs with natural probiotic. LWT Food Sci. Technol. 2004, 37, 663–667. [Google Scholar] [CrossRef]
- Mainville, I.; Robert, N.; Lee, B.; Farnworth, E.R. Polyphasic characterization of the lactic acid bacteria in kefir. Syst. Appl. Microbiol. 2006, 29, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.-C.; Wang, S.-Y.; Chen, M.-J. Microbiological study of lactic acid bacteria in kefir grains by culture-dependent and culture-independent methods. Food Microbiol. 2008, 25, 492–501. [Google Scholar] [CrossRef] [PubMed]
- Garofalo, C.; Osimani, A.; Milanović, V.; Aquilanti, L.; De Filippis, F.; Stellato, G.; Di Mauro, S.; Turchetti, B.; Buzzini, P.; Ercolini, D.; et al. Bacteria and yeast microbiota in milk kefir grains from different Italian regions. Food Microbiol. 2015, 49, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Zanirati, D.F.; Abatemarco, M.; de Cicco Sandes, S.H.; Nicoli, J.R.; Nunes, Á.C.; Neumann, E. Selection of lactic acid bacteria from Brazilian kefir grains for potential use as starter or probiotic cultures. Anaerobe 2015, 32, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Korsak, N.; Taminiau, B.; Leclercq, M.; Nezer, C.; Crevecoeur, S.; Ferauche, C.; Detry, E.; Delcenserie, V.; Daube, G. Short communication: Evaluation of the microbiota of kefir samples using metagenetic analysis targeting the 16S and 26S ribosomal DNA fragments. J. Dairy Sci. 2015, 98, 3684–3689. [Google Scholar] [CrossRef] [PubMed]
- Santos, A.; San Mauro, M.; Sanchez, A.; Torres, J.M.; Marquina, D. The antimicrobial properties of different strains of Lactobacillus spp. isolated from kefir. Syst. Appl. Microbiol. 2003, 26, 434–437. [Google Scholar] [CrossRef] [PubMed]
- Da Cruz Pedrozo Miguel, M.G.; Cardoso, P.G.; de Assis Lago, L.; Schwan, R.F. Diversity of bacteria present in milk kefir grains using culture-dependent and culture-independent methods. Food Res. Int. 2010, 43, 1523–1528. [Google Scholar] [CrossRef]
- Nalbantoglu, U.; Cakar, A.; Dogan, H.; Abaci, N.; Ustek, D.; Sayood, K.; Can, H. Metagenomic analysis of the microbial community in kefir grains. Food Microbiol. 2014, 41, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Chouraqui, J.P.; Van Egroo, L.D.; Fichot, M.C. Acidified milk formula supplemented with Bifidobacterium lactis: Impact on infant diarrhea in residential care settings. J. Pediatr. Gastroenterol. Nutr. 2004, 38, 288–292. [Google Scholar] [CrossRef] [PubMed]
- Gaón, D.; García, H.; Winter, L.; Rodríguez, N.; Quintás, R.; González, S.N.; Oliver, G. Effect of Lactobacillus strains and Saccharomyces boulardii on persistent diarrhea in children. Medicina (Buenos Aires) 2003, 63, 293–298. [Google Scholar]
- Pohjavuori, E.; Viljanen, M.; Korpela, R.; Kuitunen, M.; Tiittanen, M.; Vaarala, O.; Savilahti, E. Lactobacillus GG effect in increasing IFN-gamma production in infants with cow’s milk allergy. J. Allergy Clin. Immunol. 2004, 114, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Bourlioux, P.; Koletzko, B.; Guarner, F.; Braesco, V. The intestine and its microflora are partners for the protection of the host: Report on the Danone Symposium “The Intelligent Intestine,” held in Paris, June 14, 2002. Am. J. Clin. Nutr. 2003, 78, 675–683. [Google Scholar] [CrossRef] [PubMed]
- Azcárate-Peril, M.A.; Sikes, M.; Bruno-Bárcena, J.M. The intestinal microbiota, gastrointestinal environment and colorectal cancer: A putative role for probiotics in prevention of colorectal cancer? Am. J. Physiol. Gastrointest. Liver Physiol. 2011, 301, G401–G424. [Google Scholar] [CrossRef] [PubMed]
- Del Carmen, S.; de LeBlanc, A.M.; Miyoshi, A.; Rocha, C.S.; Azevedo, V.; LeBlanc, J.G. Potential application of probiotics in the prevention and treatment of inflammatory bowel diseases. Ulcers 2011, 2011, 841651. [Google Scholar] [CrossRef]
- Rafter, J.J. The role of lactic acid bacteria in colon cancer prevention. Scand. J. Gastroenterol. 1995, 30, 497–502. [Google Scholar] [CrossRef] [PubMed]
- Hirayama, K.; Rafter, J. The role of lactic acid bacteria in colon cancer prevention: Mechanistic considerations. Antonie Van Leeuwenhoek 1999, 76, 391–394. [Google Scholar] [PubMed]
- Nagao, F.; Nakayama, M.; Muto, T.; Okumura, K. Effects of a fermented milk drink containing Lactobacillus casei strain Shirota on the immune system in healthy human subjects. Biosci. Biotechnol. Biochem. 2000, 64, 2706–2708. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.; Rowland, I.; Tuohy, K.M.; Thomas, L.V.; Yaqoob, P. Selective effects of Lactobacillus casei Shirota on T cell activation, natural killer cell activity and cytokine production. Clin. Exp. Immunol. 2010, 161, 378–388. [Google Scholar] [CrossRef] [PubMed]
- Nomura, M.; Kobayashi, M.; Okamoto, T. Rapid PCR-Based Method Which Can Determine Both Phenotype and Genotype of Lactococcus lactis Subspecies. Appl. Environ. Microbiol. 2002, 68, 2209–2213. [Google Scholar] [CrossRef] [PubMed]
- Beimfohr, C.; Ludwig, W.; Schleifer, K.H. Rapid Genotypic Differentiation of Lactococcus lactis Subspecies and Biovar. Syst. Appl. Microbiol. 1997, 20, 216–221. [Google Scholar] [CrossRef]
- Feutry, F.; Torre, P.; Arana, I.; Garcia, S.; Desmasures, N.; Casalta, E. Lactococcus lactis strains from raw ewe’s milk samples from the PDO Ossau-Iraty cheese area: Levels, genotypic and technological diversity. Dairy Sci. Technol. 2012, 92, 655–670. [Google Scholar] [CrossRef]
- Kahala, M.; Maki, M.; Lehtovaara, A.; Tapanainen, J.M.; Katiska, R.; Juuruskorpi, M.; Juhola, J.; Joutsjoki, V. Characterization of starter lactic acid bacteria from the Finnish fermented milk product viili. J. Appl. Microbiol. 2008, 105, 1929–1938. [Google Scholar] [CrossRef] [PubMed]
- Torriani, S.; Felis, G.E.; Dellaglio, F. Differentiation of Lactobacillus plantarum, L. pentosus, and L. paraplantarum by recA gene sequence analysis and multiplex PCR assay with recA gene-derived primers. Appl. Environ. Microbiol. 2001, 67, 3450–3454. [Google Scholar] [CrossRef] [PubMed]
- Ward, L.J.H.; Timmins, M.J. Differentiation of Lactobacillus casei, Lactobacillus paracasei and Lactobacillus rhamnosus by polymerase chain reaction. Lett. Appl. Microbiol. 1999, 29, 90–92. [Google Scholar] [CrossRef] [PubMed]
- Zhong, L.; Zhang, X.; Covasa, M. Emerging roles of lactic acid bacteria in protection against colorectal cancer. World J. Gastroenterol. 2014, 20, 7878–7886. [Google Scholar] [CrossRef] [PubMed]
- De Montijo-Prieto, S.; Moreno, E.; Bergillos-Meca, T.; Lasserrot, A.; Ruiz-López, M.D.; Ruiz-Bravo, A.; Jiménez-Valera, M.A. Lactobacillus plantarum strain isolated from kefir protects against intestinal infection with Yersinia enterocolitica O9 and modulates immunity in mice. Res. Microbiol. 2015, 166, 626–632. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, S.E.; Keating, N.; Belz, G.T. Natural killer cells and anti-tumor immunity. Mol. Immunol. 2017, in press. [Google Scholar] [CrossRef] [PubMed]
- Voskoboinik, I.; Whisstock, J.C.; Trapani, J.A. Perforin and granzymes: Function, dysfunction and human pathology. Nat. Rev. Immunol. 2015, 15, 388–400. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.H.; Kang, H.; Cho, H. Natural killer cells and tumor metastasis. Arch. Pharm. Res. 2017, 40, 1037–1049. [Google Scholar] [CrossRef] [PubMed]
- Imai, K.; Matsuyama, S.; Miyake, S.; Suga, K.; Nakachi, K. Natural cytotoxic activity of peripheral-blood lymphocytes and cancer incidence: An 11-year follow-up study of a general population. Lancet 2000, 356, 1795–1799. [Google Scholar] [CrossRef]
- Tokatlı, M.; Gülgör, G.; Bağder Elmacı, S.; Arslankoz İşleyen, N.; Özçelik, F. In Vitro Properties of Potential Probiotic Indigenous Lactic Acid Bacteria Originating from Traditional Pickles. BioMed Res. Int. 2015, 2015, 315819. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Wang, Y.; Tan, Z.; Li, Z.; Jiao, Z.; Huang, Q. Screening of Probiotic Activities of Lactobacilli Strains Isolated from Traditional Tibetan Qula, A Raw Yak Milk Cheese. Asian-Australas. J. Anim. Sci. 2016, 29, 1490–1499. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, H.; Old, L.J.; Schreiber, R.D. The roles of IFN gamma in protection against tumor development and cancer immunoediting. Cytokine Growth Factor Rev. 2002, 13, 95–109. [Google Scholar]
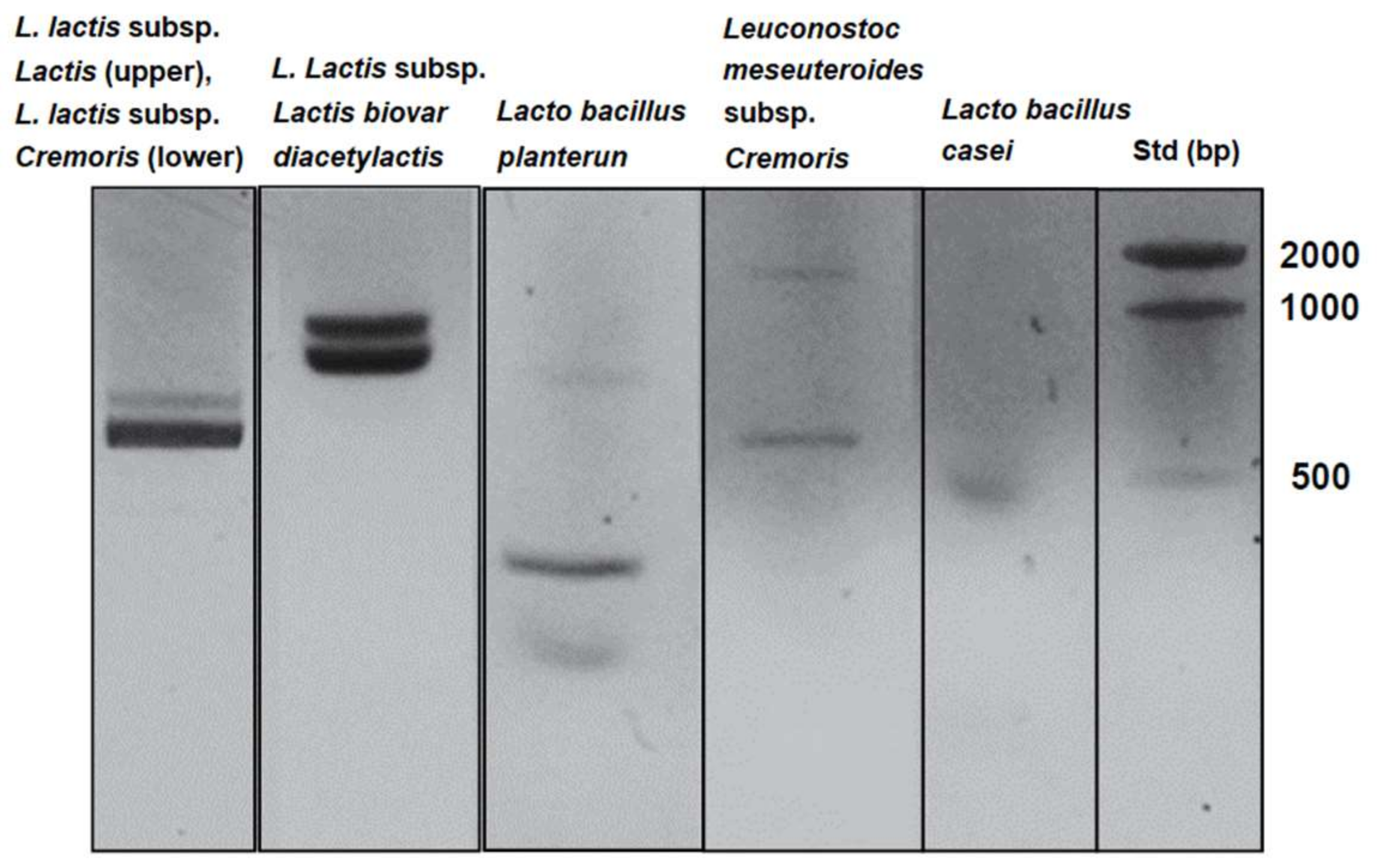

| Strain | Primer Sequence | PCR Condition | Ref. | |
|---|---|---|---|---|
| L. lactis subsp. Lactis | sense | 5′-CGTTATGGATTTGATGGATATAAAGC-3′ | 94 °C 9 min, 94 °C 30 s, 50 °C 30 s, 72 °C 60 s × 45 cycles, 72 °C 7 min | [27] |
| antisense | 5′-ACTCTTCTTAAGAACAAGTTTAACAGC-3′ | |||
| L. lactis subsp. Cremoris | sense | 5′-CGTTATGGATTTGATGGATATAAAGC-3′ | 94 °C 9 min, 94 °C 30 s, 50 °C 30 s, 72 °C 60 s × 45 cycles, 72 °C 7 min | |
| antisense | 5′-ACTCTTCTTAAGAACAAGTTTAACAGC-3′ | |||
| L. lactis subsp. lactis biovar diacetylactis | sense | 5′-CTTCGTTATGATTTTACA-3′ | 94 °C 30 s, 94 °C 30 s, 46 °C 60 s, 72 °C 60 s × 30 cycles, 72 °C 2 min | [28,29] |
| antisense | 5′-AATATCAACAATTCCATG-3′ | |||
| Leuconostoc mesenteroides subsp. Cremoris (Genus-specific primers for Leuconostoc) | sense | 5′-AACTTAGTGTCGCATGAC-3′ | 94 °C 5 min, 94 °C 60 s, 60 °C 60 s, 72 °C 60 s × 30 cycles, 72 °C 10 min | [30] |
| antisense | 5′-AGTCGAGTTACAGACTACAA-3′ | |||
| Lactobacillus plantarum | sense | 5′-CCGTTTATGCGGAACACCTA-3′ | 94 °C 3 min, 94 °C 30 s, 54 °C 10 s, 72 °C 30 s × 30 cycles, 72 °C 5 min | [31] |
| antisense | 5′-TCGGGATTACCAAACATCAC-3′ | |||
| Lactobacillus casei | sense | 5-TGCACTGAGATTCGACTTAA-3 | 94 °C 3 min, 94 °C 45 s, 45 °C 45 s, 72 °C 60 s × 30 cycles, 72 °C 5 min | [32] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yamane, T.; Sakamoto, T.; Nakagaki, T.; Nakano, Y. Lactic Acid Bacteria from Kefir Increase Cytotoxicity of Natural Killer Cells to Tumor Cells. Foods 2018, 7, 48. https://doi.org/10.3390/foods7040048
Yamane T, Sakamoto T, Nakagaki T, Nakano Y. Lactic Acid Bacteria from Kefir Increase Cytotoxicity of Natural Killer Cells to Tumor Cells. Foods. 2018; 7(4):48. https://doi.org/10.3390/foods7040048
Chicago/Turabian StyleYamane, Takuya, Tatsuji Sakamoto, Takenori Nakagaki, and Yoshihisa Nakano. 2018. "Lactic Acid Bacteria from Kefir Increase Cytotoxicity of Natural Killer Cells to Tumor Cells" Foods 7, no. 4: 48. https://doi.org/10.3390/foods7040048
APA StyleYamane, T., Sakamoto, T., Nakagaki, T., & Nakano, Y. (2018). Lactic Acid Bacteria from Kefir Increase Cytotoxicity of Natural Killer Cells to Tumor Cells. Foods, 7(4), 48. https://doi.org/10.3390/foods7040048




