Recent Advances in Physical Post-Harvest Treatments for Shelf-Life Extension of Cereal Crops
Abstract
1. Introduction
2. Modified Atmosphere Packaging (MAP)
3. Thermal Treatments
3.1. Dry Heat Treatments
3.2. Wet Heat Treatments
4. Ionizing Irradiation
4.1. Gamma Irradiation
4.2. Electron Beam Irradiation
5. Non-Ionizing Irradiation
5.1. Ultraviolet (UV) Light
5.2. Microwave Treatments
5.3. Ultrasonication
6. High Hydrostatic Pressure (HHP)
7. Conclusion and Future Trends
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Freita-Silva, O.; de Oliveira, P.S.; Freire Júnior, M. Potential of Electron Beams to Control Mycotoxigenic Fungi in Food. Food Eng. Rev. 2014, 160–170. [Google Scholar] [CrossRef]
- Oerke, E.-C. Crop losses to pests. J. Agric. Sci. 2006, 144, 31. [Google Scholar] [CrossRef]
- Siciliano, I.; Spadaro, D.; Prelle, A.; Vallauri, D.; Cavallero, M.C.; Garibaldi, A.; Gullino, M.L. Use of cold atmospheric plasma to detoxify hazelnuts from aflatoxins. Toxins 2016, 8, 125. [Google Scholar] [CrossRef] [PubMed]
- Magan, N.; Hope, R.; Cairns, V.; Aldred, D. Post-harvest fungal ecology: Impact of fungal growth and mycotoxin accumulation in stored grain. Eur. J. Plant Pathol. 2003, 109, 723–730. [Google Scholar] [CrossRef]
- Oliveira, P.M.; Zannini, E.; Arendt, E.K. Cereal fungal infection, mycotoxins, and lactic acid bacteria mediated bioprotection: From crop farming to cereal products. Food Microbiol. 2014, 37, 78–95. [Google Scholar] [CrossRef] [PubMed]
- Magan, N.; Aldred, D. Post-Harvest Control Strategies: Minimizingmycotoxins in the Food Chain. Int. J. Food Microbiol. 2007, 119, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Horstmann, S.; De Colli, L.; Danaher, M.; Speer, K.; Zannini, E.; Arendt, E.K. Impact of fungal contamination of wheat on grain quality criteria. J. Cereal Sci. 2016, 69, 95–103. [Google Scholar] [CrossRef]
- Audenaert, K.; Monbaliu, S.; Deschuyffeleer, N.; Maene, P.; Vekeman, F.; Haesaert, G.; De Saeger, S.; Eeckhout, M. Neutralized electrolyzed water efficiently reduces Fusarium spp. in vitro and on wheat kernels but can trigger deoxynivalenol (DON) biosynthesis. Food Control 2012, 23, 515–521. [Google Scholar] [CrossRef]
- Cheli, F.; Pinotti, L.; Rossi, L.; Dell’Orto, V. Effect of milling procedures on mycotoxin distribution in wheat fractions: A review. LWT—Food Sci. Technol. 2013, 54, 307–314. [Google Scholar] [CrossRef]
- Vidal, A.; Marín, S.; Ramos, A.J.; Cano-Sancho, G.; Sanchis, V. Determination of aflatoxins, deoxynivalenol, ochratoxin A and zearalenone in wheat and oat based bran supplements sold in the Spanish market. Food Chem. Toxicol. 2013, 53, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Magan, N.; Aldred, D. Food Spoilage Microorganisms; Woodhead Publishing: Sawston, UK, 2006. [Google Scholar]
- Balasubramaniam (Bala), V.M.; Martínez-Monteagudo, S.I.; Gupta, R. Principles and Application of High Pressure–Based Technologies in the Food Industry. Annu. Rev. Food Sci. Technol. 2015, 6, 435–462. [Google Scholar] [CrossRef] [PubMed]
- Crowley, S.; Mahony, J.; Van Sinderen, D. Current perspectives on antifungal lactic acid bacteria as natural bio-preservatives. Trends Food Sci. Technol. 2013, 33, 93–109. [Google Scholar] [CrossRef]
- Pawlowska, A.M.; Zannini, E.; Coffey, A.; Arendt, E.K. “Green Preservatives”: Combating Fungi in the Food and Feed Industry by Applying Antifungal Lactic Acid Bacteria. Adv. Food Nutr. Res. 2012, 66, 217–238. [Google Scholar] [CrossRef] [PubMed]
- Magan, N.; Lacey, J. Effects of gas composition and water activity on growth of field and storage fungi and their interactions. Trans. Br. Mycol. Soc. 1984, 82, 305–314. [Google Scholar] [CrossRef]
- Taniwaki, M.H.; Hocking, A.D.; Pitt, J.I.; Fleet, G.H. Growth of fungi and mycotoxin production on cheese under modified atmospheres. Int. J. Food Microbiol. 2001, 68, 125–133. [Google Scholar] [CrossRef]
- Gupta, A.; Sinha, S.N.; Atwal, S.S. Modified Atmosphere Technology in Seed Health Management: Laboratory and Field Assay of Carbon Dioxide against Storage Fungi in Paddy. Plant Pathol. J. 2014, 13, 193–199. [Google Scholar] [CrossRef]
- Savi, G.D.; Scussel, V.M. Effects of Ozone Gas Exposure on Toxigenic Fungi Species from Fusarium, Aspergillus, and Penicillium Genera. Ozone Sci. Eng. J. Int. Ozone Assoc. 2014, 36, 144–152. [Google Scholar] [CrossRef]
- O’Donnell, C.P.; Tiwari, B.K.; Bourke, P.; Cullen, P.J. Effect of ultrasonic processing on food enzymes of industrial importance. Trends Food Sci. Technol. 2010, 21, 358–367. [Google Scholar] [CrossRef]
- Klein, J.D.; Lurie, S. Postharvest heat treatment and fruit quality. Postharvest News Inf. 1991, 2, 15–19. [Google Scholar]
- Lan, S. Effects of Post-Harvest Treatment and Heat Stress on the Antioxidant Properties of Wheat. Master’s Thesis, University of Maryland, College Park, MD, USA, 3 August 2006. [Google Scholar]
- Lehtinen, P.; Kiiliäinen, K.; Lehtomäki, I.; Laakso, S. Effect of Heat Treatment on Lipid Stability in Processed Oats. J. Cereal Sci. 2003, 37, 215–221. [Google Scholar] [CrossRef]
- Rose, D.J.; Bianchini, A.; Martinez, B.; Flores, R.A. Methods for reducing microbial contamination of wheat flour and effects on functionality. Cereal Foods World 2012, 57, 104–109. [Google Scholar] [CrossRef]
- Bari, L.; Ohki, H.; Nagakura, K.; Ukai, M. Application of Ultra Superheated Steam Technology (USST) to Food Grain Preservation at Ambient Temperature for Extended Periods of Time. Adv. Food Technol. Nutr. Sci. 2015, SE1, S14–S21. [Google Scholar] [CrossRef]
- Gilbert, J.; Woods, S.M.; Turkington, T.K.; Tekauz, A. Effect of heat treatment to control Fusarium graminearum in wheat seed. Can. J. Plant Pathol. 2005, 27, 448–452. [Google Scholar] [CrossRef]
- Chang, Y.; Li, X.-P.; Liu, L.; Ma, Z.; Hu, X.; Zhao, W.; Gao, G. Effect of Processing in Superheated Steam on Surface Microbes and Enzyme Activity of Naked Oats. J. Food Process. Preserv. 2015, 39, 2753–2761. [Google Scholar] [CrossRef]
- Hu, Y.; Nie, W.; Hu, X.; Li, Z. Microbial decontamination of wheat grain with superheated steam. Food Control 2016, 62, 264–269. [Google Scholar] [CrossRef]
- Ban, G.-H.; Kang, D.-H. Effectiveness of superheated steam for inactivation of Escherichia coli O157:H7, Salmonella Typhimurium, Salmonella Enteritidis phage type 30, and Listeria monocytogenes on almonds and pistachios. Int. J. Food Microbiol. 2016, 220, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Cenkowski, S.; Pronyk, C.; Zmidzinska, D.; Muir, W.E. Decontamination of food products with superheated steam. J. Food Eng. 2007, 83, 68–75. [Google Scholar] [CrossRef]
- Clear, R.M.; Patrick, S.K.; Wallis, R.; Turkington, T.K. Effect of dry heat treatment on seed-borne Fusarium graminearum and other cereal pathogens. Can. J. Plant Pathol. 2002, 24, 489–498. [Google Scholar] [CrossRef]
- Bond, W.W.; Favero, M.S.; Petersen, N.J.; Marshall, J.H. Dry-heat inactivation kinetics of naturally occurring spore populations. Appl. Microbiol. 1970, 20, 573–578. [Google Scholar] [PubMed]
- Nielsen, K.F.; Holm, G.; Uttrup, L.P.; Nielsen, P.A. Mould growth on building materials under low water activities. Influence of humidity and temperature on fungal growth and secondary metabolism. Int. Biodeterior. Biodegrad. 2004, 54, 325–336. [Google Scholar] [CrossRef]
- Miller, J.D.; Trenholm, H.L. Mycotoxins in Grain Compounds Other than Aflatoxin; Eagan Press: Saint Paul, MN, USA, 1997. [Google Scholar]
- Bretz, M.; Knecht, A.; Göckler, S.; Humpf, H.U. Structural elucidation and analysis of thermal degradation products of the Fusarium mycotoxin nivalenol. Mol. Nutr. Food Res. 2005, 49, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Vidal, A.; Sanchis, V.; Ramos, A.J.; Marín, S. Thermal stability and kinetics of degradation of deoxynivalenol, deoxynivalenol conjugates and ochratoxin A during baking of wheat bakery products. Food Chem. 2015, 178, 276–286. [Google Scholar] [CrossRef] [PubMed]
- Boudra, H.; Lebars, P.; Lebars, J. Thermostability of Ochratoxin A in Wheat under 2 Moisture Conditions. Appl. Environ. Microbiol. 1995, 61, 1156–1158. [Google Scholar] [PubMed]
- Syamaladevi, R.M.; Tang, J.; Villa-Rojas, R.; Sablani, S.; Carter, B.; Campbell, G. Influence of Water Activity on Thermal Resistance of Microorganisms in Low-Moisture Foods: A Review. Compr. Rev. Food Sci. Food Saf. 2016, 15, 353–370. [Google Scholar] [CrossRef]
- Ban, G.H.; Yoon, H.; Kang, D.H. A comparison of saturated steam and superheated steam for inactivation of Escherichia coli O157: H7, Salmonella Typhimurium, and Listeria monocytogenes biofilms on polyvinyl chloride and stainless steel. Food Control 2014, 40, 344–350. [Google Scholar] [CrossRef]
- Alfy, A.; Kiran, B.V.; Jeevitha, G.C.; Hebbar, H.U. Recent Developments in Superheated Steam Processing of Foods—A Review. Crit. Rev. Food Sci. Nutr. 2016, 56, 2191–2208. [Google Scholar] [CrossRef] [PubMed]
- Jalili, M. A Review on Aflatoxins Reduction in Food. Iran. J. Health Saf. Environ. 2015, 3, 445–459. [Google Scholar]
- Nemţanu, M.R.; Braşoveanu, M.; Karaca, G.; Erper, I. Inactivation effect of electron beam irradiation on fungal load of naturally contaminated maize seeds. J. Sci. Food Agric. 2014, 94, 2668–2673. [Google Scholar] [CrossRef] [PubMed]
- Dev, S.R.S.; Birla, S.L.; Raghavan, G.S.V.; Subbiah, J. Microbial decontamination of food by microwave (MW) and radiao frequency (RF). In Microbial Decontamination in the Food Industry; Novel Methods and Applications; Woodhead Publishing: Sawston, UK, 2012; pp. 274–299. [Google Scholar]
- FAO/IAEA Training Manual on Food Irradiation Technology and Techniques. In Technical Reports Series, Proceedings of the International Atomic Energy Commission; IAEA Publications: Vienna, Austria, 1982; p. 224.
- Mahmoud, N.S.; Awad, S.H.; Madani, R.M.A.; Osman, F.A.; Elmamoun, K.; Hassan, A.B. Effect of γ radiation processing on fungal growth and quality characteristcs of millet grains. Food Sci. Nutr. 2016, 4, 342–347. [Google Scholar] [CrossRef] [PubMed]
- D’Ovidio, K.L.; Trucksess, M.W.; Devries, J.W.; Bean, G. Effects of irradiation on fungi and fumonisin B1 in corn, and of microwave-popping on fumonisins in popcorn. Food Addit. Contam. 2007, 24, 735–743. [Google Scholar] [CrossRef] [PubMed]
- Lung, H.-M.; Cheng, Y.-C.; Chang, Y.-H.; Huang, H.-W.; Yang, B.B.; Wang, C.-Y. Microbial decontamination of food by electron beam irradiation. Trends Food Sci. Technol. 2015, 44, 66–78. [Google Scholar] [CrossRef]
- Supriya, P.; Sridhar, K.R.; Ganesh, S. Fungal decontamination and enhancement of shelf life of edible split beans of wild legume Canavalia maritima by the electron beam irradiation. Radiat. Phys. Chem. 2014, 96, 5–11. [Google Scholar] [CrossRef]
- Salem, E.A.; Soliman, S.A.; El-Karamany, A.M.; El-shafea, Y.M.A. Effect of Utilization of Gamma Radiation Treatment and Storage on Total Fungal Count, Chemical Composition and Technological Properties Wheat Grain. Egypt. J. Biol. Pest Control 2016, 26, 163–171. [Google Scholar]
- Aziz, N.H.; El-Far, F.M.; Shahin, A.A.M.; Roushy, S.M. Control of Fusarium moulds and fumonisin B1 in seeds by gamma-irradiation. Food Control 2007, 18, 1337–1342. [Google Scholar] [CrossRef]
- Carocho, M.; Antonio, A.L.; Barreira, J.C.M.; Rafalski, A.; Bento, A.; Ferreira, I.C.F.R. Validation of Gamma and Electron Beam Irradiation as Alternative Conservation Technology for European Chestnuts. Food Bioprocess Technol. 2014, 7, 1917–1927. [Google Scholar] [CrossRef]
- Carocho, M.; Barros, L.; Antonio, A.L.; Barreira, J.C.M.; Bento, A.; Kaluska, I.; Ferreira, I.C.F.R. Analysis of organic acids in electron beam irradiated chestnuts (Castanea sativa Mill.): Effects of radiation dose and storage time. Food Chem. Toxicol. 2013, 55, 348–352. [Google Scholar] [CrossRef] [PubMed]
- Akueche, E.C.; Anjorin, S.T.; Harcourt, B.I.; Kana, D. Studies on fungal load, total aflatoxins and ochratoxin a contents of gamma-irradiated and non-irradiated Sesamum indicum grains from Abuja markets, Nigeria. Kasetsart J. Nat. Sci. 2012, 46, 371–382. [Google Scholar]
- Herzallah, S.; Alshawabkeh, K.; Al Fataftah, A. Aflatoxin decontamination of artificially contaminated feeds by sunlight, γ-radiation, and microwave heating. J. Appl. Poult. Res. 2008, 17, 515–521. [Google Scholar] [CrossRef]
- Jalili, M.; Jinap, S.; Noranizan, M.A. Aflatoxins and ochratoxin a reduction in black and white pepper by gamma radiation. Radiat. Phys. Chem. 2012, 81, 1786–1788. [Google Scholar] [CrossRef]
- Mehrez, A.; Maatouk, I.; Romero-González, R.; Ben Amara, A.; Kraiem, M.; Garrido Frenich, A.; Landoulsi, A. Assessment of ochratoxin A stability following gamma irradiation: Experimental approaches for feed detoxification perspectives. World Mycotoxin J. 2016, 9, 289–298. [Google Scholar] [CrossRef]
- Hooshmand, H.; Klopfenstein, C.F. Effects of gamma irradiation on mycotoxin disappearance and amino acid contents of corn, wheat, and soybeans with different moisture contents. Plant Foods Hum. Nutr. 1995, 47, 227–238. [Google Scholar] [CrossRef] [PubMed]
- Carocho, M.; Barreira, J.C.; Antonio, A.L.; Bento, A.; Kaluska, I.; Ferreira, I.C. Effects of electron beam radiation on nutritional parameters of Portuguese chestnuts (Castanea sativa mill.). J. Agric. Food Chem. 2012, 60, 7754–7760. [Google Scholar] [CrossRef] [PubMed]
- El-Naggar, S.M.; Mikhaiel, A.A. Disinfestation of stored wheat grain and flour using gamma rays and microwave heating. J. Stored Prod. Res. 2011, 47, 191–196. [Google Scholar] [CrossRef]
- Melki, M.; Marouani, A. Effects of gamma rays irradiation on seed germination and growth of hard wheat. Environ. Chem. Lett. 2010, 8, 307–310. [Google Scholar] [CrossRef]
- Deberghes, P.; Betbeder, A.M.; Boisard, F.; Blanc, R.; Delaby, J.F.; Krivobok, S.; Steiman, R.; Seigle-Murandi, F.; Creppy, E.E. Detoxification of ochratoxin A, a food contaminant: Prevention of growth of Aspergillus ochraceus and its production of ochratoxin A. Mycotoxin Res. 1995, 11, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Di Stefano, V.; Pitonzo, R.; Cicero, N.; D’Oca, M.C. Mycotoxin contamination of animal feedingstuff: Detoxification by gamma-irradiation and reduction of aflatoxins and ochratoxin A concentrations. Food Addit. Contam. Part A 2014, 31, 2034–2039. [Google Scholar] [CrossRef] [PubMed]
- Aquino, S.; Ferreira, F.; Ribeiro, D.H.B.; Corrêa, B.; Greiner, R.; Villavicencio, A.L.C.H. Evaluation of viability of Aspergillus flavus and aflatoxins degradation in irradiated samples of maize. Braz. J. Microbiol. 2005, 36, 352–356. [Google Scholar] [CrossRef]
- Farag, R.S.; El-Baroty, G.S.; Abo-Hagger, A.A. Aflatoxin destruction and residual toxicity of contaminated-irradiated yellow corn and peanuts on rats. Adv. Food Sci. 2004, 26, 122–129. [Google Scholar]
- Prado, G.; de Carvalho, E.P.; Oliveira, M.S.; Madeira, J.G.C.; Morais, V.D.; Correa, R.F.; Cardoso, V.N.; Soares, T.V.; da Silva, J.F.M.; Gonçalves, R.C.P. Effect of gamma irradiation on the inactivation of aflatoxin B1 and fungal flora in peanut. Braz. J. Microbiol. 2003, 34, 138–140. [Google Scholar] [CrossRef]
- Wang, F.; Xie, F.; Xue, X.; Wang, Z.; Fan, B.; Ha, Y. Structure elucidation and toxicity analyses of the radiolytic products of aflatoxin B1 in methanol-water solution. J. Hazard. Mater. 2011, 192, 1192–1202. [Google Scholar] [CrossRef] [PubMed]
- Kume, T.; Furuta, M.; Todoriki, S.; Uenoyama, N.; Kobayashi, Y. Status of food irradiation in the world. Radiat. Phys. Chem. 2009, 78, 222–226. [Google Scholar] [CrossRef]
- Kottapalli, B.; Wolf-Hall, C.E.; Schwarz, P. Effect of electron-beam irradiation on the safety and quality of Fusarium-infected malting barley. Int. J. Food Microbiol. 2006, 110, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Stepanik, T.; Kost, D.; Nowicki, T.; Gaba, D. Effects of electron beam irradiation on deoxynivalenol levels in distillers dried grain and solubles and in production intermediates. Food Addit. Contam. 2007, 24, 1001–1006. [Google Scholar] [CrossRef] [PubMed]
- Lanza, C.M.; Mazzaglia, A.; Paladino, R.; Auditore, L.; Barnà, D.; Loria, D.; Trifirò, A.; Trimarchi, M.; Bellia, G. Characterization of peeled and unpeeled almond (Prunus amygdalus) flour after electron beam processing. Radiat. Phys. Chem. 2013, 86, 140–144. [Google Scholar] [CrossRef]
- Schoeller, N.P.; Ingham, S.C.; Ingham, B.H. Assessment of the Potential for Listeria monocytogenes Survival and Growth during Alfalfa Sprout Production and Use of Ionizing Radiation as a Potential Intervention Treatment. J. Food Prot. 2002, 8, 1259–1266. [Google Scholar] [CrossRef]
- Kikuchi, O.K.; Todoriki, S.; Saito, M.; Hayashi, T. Efficacy of soft-electron (low-energy electron beam) for soybean decontamination in comparison with gamma-rays. J. Food Sci. 2003, 68, 649–652. [Google Scholar] [CrossRef]
- Farkas, J.; Mohacsi-Farkas, C. History and future of food irradiation. Trends Food Sci. Technol. 2011, 22, 121–126. [Google Scholar] [CrossRef]
- Oms-Oliu, G.; Martín-Belloso, O.; Soliva-Fortuny, R. Pulsed light treatments for food preservation. A review. Food Bioprocess Technol. 2010, 3, 13–23. [Google Scholar] [CrossRef]
- Aron Maftei, N.; Ramos-Villarroel, A.Y.; Nicolau, A.I.; Martín-Belloso, O.; Soliva-Fortuny, R. Pulsed light inactivation of naturally occurring moulds on wheat grain. J. Sci. Food Agric. 2014, 94, 721–726. [Google Scholar] [CrossRef] [PubMed]
- Nicorescu, I.; Nguyen, B.; Moreau-Ferret, M.; Agoulon, A.; Chevalier, S.; Orange, N. Pulsed light inactivation of Bacillus subtilis vegetative cells in suspensions and spices. Food Control 2013, 31, 151–157. [Google Scholar] [CrossRef]
- Schmidt-Heydt, M.; Cramer, B.; Graf, I.; Lerch, S.; Hunpf, H.-U.; Geisen, R. Wavelength-dependent degradation of ochratoxin and citrinin by light in vitro and in vivo and its implications on Penicillium. Toxins 2012, 4, 1535–1551. [Google Scholar] [CrossRef] [PubMed]
- Jubeen, F.; Bhatti, I.A.; Khan, M.Z.; Shahid, M. Effect of UVC Irradiation on Aflatoxins in Ground Nut (Arachis hypogea) and Tree Nuts (Juglans regia, Prunus duclus and Pistachio vera). Chem. Soc. Pak. 2012, 34, 1–10. [Google Scholar]
- Liu, R.; Jin, Q.; Tao, G.; Shan, L.; Huang, J.; Liu, Y.; Wang, X.; Mao, W.; Wang, S. Photodegradation kinetics and byproducts identification of the Aflatoxin B1 in aqueous medium by ultra-performance liquid chromatography-quadrupole time-of-flight mass spectrometry. J. Mass Spectrom. 2010, 45, 553–559. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Chang, M.; Jin, Q.; Huang, J.; Liu, Y.; Wang, X. Degradation of aflatoxin B1 in aqueous medium through UV irradiation. Eur. Food Res. Technol. 2011, 233, 1007–1012. [Google Scholar] [CrossRef]
- Fang, Y.; Hu, J.; Xiong, S.; Zhao, S. Effect of low-dose microwave radiation on Aspergillus parasiticus. Food Control 2011, 22, 1078–1084. [Google Scholar] [CrossRef]
- Ursu, M.-P. Usage of Microwaves for Decontamination of Sensible Materials and Cereal Seeds. Rev. Tehnol. Neconv. 2015, 19, 60–64. [Google Scholar]
- Kabak, B.; Dobson, A.D.; Var, I. Strategies to Prevent Mycotoxin Contamination of Food and Animal Feed: A Review. Crit. Rev. Food Sci. Nutr. 2006, 46, 593–619. [Google Scholar] [CrossRef] [PubMed]
- Basaran, P.; Akhan, Ü. Microwave irradiation of hazelnuts for the control of aflatoxin producing Aspergillus parasiticus. Innov. Food Sci. Emerg. Technol. 2010, 11, 113–117. [Google Scholar] [CrossRef]
- Feng, H.; Yang, W.; Hielscher, T. Power Ultrasound. Food Sci. Technol. Int. 2008, 14, 433–436. [Google Scholar] [CrossRef]
- Butz, P.; Tauscher, B. Emerging technologies: Chemical aspects. Food Res. Int. 2002, 35, 279–284. [Google Scholar] [CrossRef]
- Chemat, F.; Zill-E-Huma; Khan, M.K. Applications of ultrasound in food technology: Processing, preservation and extraction. Ultrason. Sonochem. 2011, 18, 813–835. [Google Scholar] [CrossRef] [PubMed]
- Bilek, S.E.; Turantaş, F. Decontamination efficiency of high power ultrasound in the fruit and vegetable industry, a review. Int. J. Food Microbiol. 2013, 166, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Scouten, A.J.; Beuchat, L.R. Combined effects of chemical, heat and ultrasound treatments to kill Salmonella and Escherichia coli O157:H7 on alfalfa seeds. J. Appl. Microbiol. 2002, 92, 668–674. [Google Scholar] [CrossRef] [PubMed]
- Seymour, I.J.; Burfoot, D.; Smith, R.L.; Cox, L.A.; Lockwood, A. Ultrasound decontamination of minimally processed fruits and vegetables. Int. J. Food Sci. Technol. 2002, 37, 547–557. [Google Scholar] [CrossRef]
- Herceg, Z.; Jambrak, R.R.; Vukušić, T.; Stulić, V.; Stanzer, D.; Milošević, S. The effect of high-power ultrasound and gas phase plasma treatment on Aspergillus spp. and Penicillium spp. count in pure culture. J. Appl. Microbiol. 2015, 118, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Lindner, W.; Hasenhuti, K. Decontamination and Detoxification of Corn Which Was Contaminated with Trichothecenes Applying Ultrasonication (Abstr.). In Proceedings of the IX Internat IUPAC Symposium on Mycotoxins and Phytotoxins, Rome, Italy, 7–31 May 1996; p. 182. [Google Scholar]
- Heinz, V.; Buckow, R. Food preservation by high pressure. J. Verbrauch. Lebensmittelsich. 2009, 5, 73–81. [Google Scholar] [CrossRef]
- Polydera, A.C.; Stoforos, N.G.; Taoukis, P.S. Comparative shelf life study and vitamin C loss kinetics in pasteurised and high pressure processed reconstituted orange juice. J. Food Eng. 2003, 60, 21–29. [Google Scholar] [CrossRef]
- Wannasawat Ratphitagsanti, M. Approaches for Enhancing Lethality of Bacterial Spores Treated by Pressure-Assisted Thermal Processing; ProQuest Dissertations Publishing: Ann Arbor, MI, USA, 2009. [Google Scholar]
- Patterson, M.F. Microbiology of pressure-treated foods. J. Appl. Microbiol. 2005, 98, 1400–1409. [Google Scholar] [CrossRef] [PubMed]
- O’Reilly, C.E.; O’Connor, P.M.; Kelly, A.L.; Beresford, T.P.; Murphy, P.M. Use of hydrostatic pressure for inactivation of microbial contaminants in cheese. Appl. Environ. Microbiol. 2000, 66, 4890–4896. [Google Scholar] [CrossRef] [PubMed]
- Willford, J.; Mendonca, A.; Goodridge, L. Water Pressure Effectively Reduces Salmonella enterica Serovar Enteritidis on the Surface of Raw Almonds. J. Food Prot. 2008, 4, 825–829. [Google Scholar] [CrossRef]
- Bello, E.F.T.; Martínez, G.G.; Klotz Ceberio, B.F.; Rodrigo, D.; López, A.M. High Pressure Treatment in Foods. Foods 2014, 3, 476–490. [Google Scholar] [CrossRef] [PubMed]
- Black, E.P.; Setlow, P.; Hocking, A.D.; Stewart, C.M.; Kelly, A.L.; Hoover, D.G. Response of spores to high-pressure processing. Compr. Rev. Food Sci. Food Saf. 2007, 6, 103–119. [Google Scholar] [CrossRef]
- Hao, H.; Zhou, T.; Koutchma, T.; Wu, F.; Warriner, K. High hydrostatic pressure assisted degradation of patulin in fruit and vegetable juice blends. Food Control 2016, 62, 237–242. [Google Scholar] [CrossRef]
- Martínez-Rodríguez, Y.; Acosta-Muñiz, C.; Olivas, G.I.; Guerrero-Beltrán, J.; Rodrigo-Aliaga, D.; Mujica-Paz, H.; Welti-Chanes, J.; Sepulveda, D.R. Effect of high hydrostatic pressure on mycelial development, spore viability and enzyme activity of Penicillium Roqueforti. Int. J. Food Microbiol. 2014, 168–169, 42–46. [Google Scholar] [CrossRef] [PubMed]
- Smith, K.; Mendonca, A.; Jung, S. Impact of high-pressure processing on microbial shelf-life and protein stability of refrigerated soymilk. Food Microbiol. 2009, 26, 794–800. [Google Scholar] [CrossRef] [PubMed]
- Torres, J.A.; Saraiva, J.A.; Guerra-Rodríguez, E.; Aubourg, S.P.; Vázquez, M. Effect of combining high-pressure processing and frozen storage on the functional and sensory properties of horse mackerel (Trachurus trachurus). Innov. Food Sci. Emerg. Technol. 2014, 21, 2–11. [Google Scholar] [CrossRef]
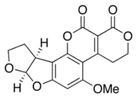
| Aspergillus spp. | Aspergillus spp./Penicillium spp. | Fusarium spp. |
|---|---|---|
 |  | |
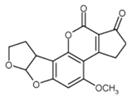
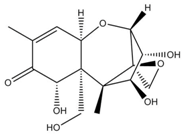
| Aflatoxin B1 | Deoxynivalenol | |
 | 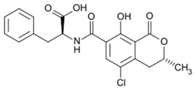 |  |
| Aflatoxin B2 | Ochratoxin A | Nivalenol |
 | 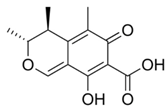 | 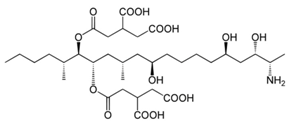 |
| Aflatoxin G1 | Citrinin | Fumonisin B1 |
 | 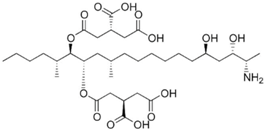 | |
| Aflatoxin G2 | Fumonisin B2 | |
 | ||
| Zearalenone |
| Target Organism/Toxin | Treatment | Sample Matrix | Technological Impact | References |
|---|---|---|---|---|
| Natural microbial load | Dry air 9 day/100 °C | Various cereals | No impact | [21,22] |
| Natural microbial load | Steam 60 min/82 °C | corn | No impact | [23] |
| Natural microbial load | Steam 210–250 °C/15 s | Wheat, barley, rye | Not investigated | [24] |
| F. graminearum | dry air 15 day/60 °C; 5 day/70 °C; 2 day/80 °C | Wheat | No impact | [25] |
| F. graminearum | Dry air 5 day/90 °C | Wheat | Reduced seed viability | [25] |
| F. graminearum | Dry air 21 day/60 °C; 9 day/70 °C; 5 day/80 °C | Barley | Reduced viability for 9 day/70 deg; 5 day/80 deg | [25] |
| Aspergillus spp., Penicillium spp., Fusarium spp., E. coli, L. Monocytogenes, Salmonella spp. | Steam 170–200 °C/<60 s | Various cereals | No impact | [26,27,28] |
| Geobacillus stearothermophilus spores | Steam 20 min/160 °C | Dried spore pellet-sand mixture | Not investigated | [29] |
| DON (50% reduction) | Steam 6 min/185 °C | wheat | Not investigated | [29] |
| Target Organism/Toxin | Treatment | Sample Matrix | Technological Impact | References |
|---|---|---|---|---|
| Natural fungal population | 0.75 kGy gamma | millet | none | [44] |
| Natural microbial load | 6 kGy e-beam | chestnuts | No effect on nutritional value | [45,46] |
| L. monocytogenes | 3.3 kGy e-beam (soft electrons) | Alfalfa sprouts | No quality deterioration | [47] |
| Aspergillus spp., Alternaria spp., Fusarium spp., Curvularia spp., Helminthosporium spp. | 1.5–3.5 kGy gamma | wheat | Reduced quality for doses >2.5 kGy | [48] |
| Fusarium spp. | 4 kGy gamma | Barley | Reduced quality | [49] |
| Fusarium spp. | 6 kGy gamma | Wheat and maize | Reduced quality | [49] |
| Aspergillus spp., Penicillium spp. | 10–15 kGy e-beam | Dry split beans | No quality deterioration (10 kGy) | [50] |
| Penicillium spp., Fusarium spp., Aspergillus spp. | 1.7–4.8 kGy e-beam | corn | Not investigated | [41] |
| Fusarium spp. and DON | 6–10 kGy e-beam | Barley, malt | Not investigated | [51] |
| OTA and aflatoxins | 15 kGy gamma | Wheat and sesame | Not investigated | [52,53,54] |
| OTA | 2 kGy gamma | Aqueous solution | - | [55] |
| DON, ZEN, T-2, FB1 | 10 kGy gamma | Soy beans, corn, wheat | Not investigated | [56] |
| FB1 | 7 kGy gamma | Barley, wheat, maize | Not investigated | [49] |
| aflatoxins | 1.5 kGy e-beam | Ground almond flour | Not investigated | [57] |
| Parameters | Gamma Irradiation | E-Beam Irradiation |
|---|---|---|
| Irradiation Time | Slow | Fast |
| Doses (kGy) | Higher doses | Lower doses |
| Source | Radioactive material | Electricity to generate electrons |
| Flexibility | Inflexible (cannot be turned off) | More flexible (can be turned off) |
| Penetration | Good penetration | Lower penetration power |
| Target Organism/Toxin | Treatment | Sample Matrix | Technological Impact | References |
|---|---|---|---|---|
| Different food spoilage bacteria and fungi | US (ultrasound) > 60 W/cm2 | Aqueous solution | - | [73] |
| A. parasiticus | Microwave: 900 W, 2.45 GHz, 1–5 min | Aqueous solution | - | [74] |
| Aspergillus spp. and Penicillium spp. | US: 6 min, 60 °C, 20–39 W/cm2 | Culture medium | - | [75] |
| Aspergillus spp. | 51.2 J/g pulsed white light | wheat | 15% reduced seed viability | [76] |
| Aspergillus spp. | Microwave: 120 s, 2450 MHz, 1.25 kW | Cereals and nuts | Not investigated | [77,78] |
| Bacillus subtilis | 1.0 J/cm2 * 10 pulses light with 200–1100 nm | spices | No quality deterioration | [79] |
| OTA, OTB (Ochratoxin B), citrinin | Light: 455 nm/470 nm for 5 day | Aqueous solution | - | [80] |
| Aflatoxins | UV (Ultraviolet)-light: 265 nm for 15–45 min | nuts | Not investigated | [81] |
| trichothecenes | US > 200 W/cm2 | corn | No quality deterioration | [82] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schmidt, M.; Zannini, E.; Arendt, E.K. Recent Advances in Physical Post-Harvest Treatments for Shelf-Life Extension of Cereal Crops. Foods 2018, 7, 45. https://doi.org/10.3390/foods7040045
Schmidt M, Zannini E, Arendt EK. Recent Advances in Physical Post-Harvest Treatments for Shelf-Life Extension of Cereal Crops. Foods. 2018; 7(4):45. https://doi.org/10.3390/foods7040045
Chicago/Turabian StyleSchmidt, Marcus, Emanuele Zannini, and Elke K. Arendt. 2018. "Recent Advances in Physical Post-Harvest Treatments for Shelf-Life Extension of Cereal Crops" Foods 7, no. 4: 45. https://doi.org/10.3390/foods7040045
APA StyleSchmidt, M., Zannini, E., & Arendt, E. K. (2018). Recent Advances in Physical Post-Harvest Treatments for Shelf-Life Extension of Cereal Crops. Foods, 7(4), 45. https://doi.org/10.3390/foods7040045






