Incidence, Antimicrobial Susceptibility, and Toxin Genes Possession Screening of Staphylococcus aureus in Retail Chicken Livers and Gizzards
Abstract
:1. Introduction
2. Experimental Section
2.1. Isolation of Staphylococcus aureus from Retail Chicken Livers and Gizzards
| Gene | Size (bp) | Primer sequences (5´–3´) | Bacterium | References |
|---|---|---|---|---|
| Sa4221-1 | 108 | AAT CTT TGT CGG TAC ACG ATA TTC TTC ACG | S. aureus | [34] |
| Sa4221-2 | CGT AAT GAG ATT TCA GTA GAT AAT ACA ACA | |||
| mecA-F | 312 | GTT GTA GTT GTC GGG TTT GGCTT CCA CAT ACC ATC TTC TTT AAC | MRSA | [21] |
| mecA-R | CTT CCA CAT ACC ATC TTC TTT AAC | |||
| mecA1F | 533 | AAA ATC GAT GGT AAA GGT TGG C | MRSA | [35] |
| mecA2R | AGT TCT GCA GTA CCG GAT TTG C | |||
| MecHomFW | 356 | TCA CCA GGT TCA AC[Y] CAA AA | MRSA | [36] |
| MecHomRV | CCT GAA TC[W] GCT AAT AAT ATT TC | |||
| mecAM10/0061 F1 | 1800 | CCA GAT ATA GTA GCA TTA TA | MRSA | [37] |
| mecAM10/0061 R1 | AAA GAT GAC GAT ATT GAG |
2.2. DNA Extraction
2.3. PCR Identification
2.4. Antimicrobial Susceptibility Testing
| Antimicrobial Class | Antimicrobials | Conc. 1 * (µg/mL) | Conc. 2 (µg/mL) (Break point) | Conc. 3 (µg/mL) | Conc. 4 (µg/mL) |
|---|---|---|---|---|---|
| β-Lactams | penicillin | 0.125 | 0.25 | 0.5 | 1 |
| ampicillin | 0.25 | 0.5 | 1 | 2 | |
| oxacillin + 2% Nacl | 2 | 4 | 8 | 16 | |
| cefoxitin + 2% Nacl | 4 | 8 | 16 | 32 | |
| Tetracyclines | tetracycline | 8 | 16 | 32 | 64 |
| doxycycline | 8 | 16 | 32 | 64 | |
| Macrolides | azithromycin | 4 | 8 | 16 | 32 |
| erythromycin | 4 | 8 | 16 | 32 | |
| Aminoglycosides | kanamycin | 32 | 64 | 128 | 256 |
| gentamicin | 8 | 16 | 32 | 64 | |
| Fluoroquinolones | ciprofloxacin | 2 | 4 | 8 | 16 |
| Lincosamides | clindamycin | 2 | 4 | 8 | 16 |
| Phenicols | chloramphenicol | 16 | 32 | 64 | 128 |
| Glycopeptides | vancomycin | 16 | 32 | 64 | 128 |
| Rifamycines | rifampin | 2 | 4 | 8 | 16 |
| Sulfonamides | trimethoprim/sulfamethoxazole | 2/38 | 4/76 | 8/152 | 16/304 |
2.5. Detection of Toxin Genes
| Toxin Gene (Toxin group) | Size (bp) | Primer sequences (5´–3´) | Multiplex PCR reaction set | References |
|---|---|---|---|---|
| sea (Enterotoxins) | 521 | GCA GGG AAC AGC TTT AGG C GTT CTG TAG AAG TAT GAA ACA CG | A | [40] |
| seb-sec (Enterotoxins) | 665 | ATG TAA TTT TGA TAT TCG CAG TG TGC AGG CAT CAT ATC ATA CCA | A | [40] |
| sec (Enterotoxins) | 284 | CTT GTA TGT ATG GAG GAA TAA CAA TGC AGG CAT CAT ATC ATA CCA | A | [40] |
| sed (Enterotoxins) | 385 | GTG GTG AAA TAG ATA GGA CTG C ATA TGA AGG TGC TCT GTG G | A | [40] |
| see (Enterotoxins) | 171 | TAC CAA TTA ACT TGT GGA TAG AC CTC TTT GCA CCT TAC CGC | A | [40] |
| seg (Enterotoxins) | 328 | CGT CTC CAC CTG TTG AAG G CCA AGT GAT TGT CTA TTG TCG | A | [40] |
| seh (Enterotoxins) | 359 | CAA CTG CTG ATT TAG CTC AG GTC GAA TGA GTA ATC TCT AGG | B | [40] |
| sei (Enterotoxins) | 466 | CAA CTC GAA TTT TCA ACA GGT AC CAG GCA GTC CAT CTC CTG | B | [40] |
| sej (Enterotoxins) | 142 | CAT CAG AAC TGT TGT TCC GCT AG CTG AAT TTT ACC ATC AAA GGT AC | B | [40] |
| tst (Toxic Shock Syndrome Toxin 1) | 560 | GCT TGC GAC AAC TGC TAC AG TGG ATC CGT CAT TCA TTG TTA A | B | [40] |
| eta (Exfoliative toxins) | 93 | GCA GGT GTT GAT TTA GCA TT AGA TGT CCC TAT TTT TGC TG | B | [41] |
| etb (Exfoliative toxins) | 226 | ACA AGC AAA AGA ATA CAG CG GTT TTT GGC TGC TTC TCT TG | B | [41] |
| lukS-lukF (Panton-Valentine leucocidin (PVL)) | 433 | ATC ATT AGG TAA AAT GTC TGG ACA TGA TCC A GCA TCA AST GTA TTG GAT AGC AAA AGC | C | [42] |
| lukE-lukD (Leucocidin) | 269 | TGA AAA AGG TTC AAA GTT GAT ACG AG TGT ATT CGA TAG CAA AAG CAG TGC A | C | [42] |
| lukM (Leucocidin) | 780 | TGG ATG TTA CCT ATG CAA CCT AC GTT CGT TTC CAT ATA ATG AAT CAC TAC | C | [42] |
| hla (Hemolysins) | 209 | CTG ATT ACT ATC CAA GAA ATT CGA TTG CTT TCC AGC CTA CTT TTT TAT CAG T | C | [42] |
| hlb (Hemolysins) | 309 | GTG CAC TTA CTG ACA ATA GTG C GTT GAT GAG TAG CTA CCT TCA GT | C | [42] |
| hld (Hemolysins) | 111 | AAG AAT TTT TAT CTT AAT TAA GGA AGG AGT G TTA GTG AAT TTG TTC ACT GTG TCG A | C | [42] |
2.6. Molecular Typing Using spa Genotyping
3. Results and Discussion
3.1. Prevalence of Staphylococcus aureus and MRSA in Chicken Livers and Gizzards
| Prevalence of Staphylococcus aureus | ||||||||
|---|---|---|---|---|---|---|---|---|
| Chicken Livers | Chicken Gizzards | Chicken Livers and Gizzards | ||||||
| Brand A np/n * (%) | Brand B np/n (%) | Total np/n (%) | Brand A np/n (%) | Brand B np/n (%) | Total np/n (%) | Brand A np/n (%) | Brand B np/n (%) | Total np/n (%) |
| 26/71 (36.6) | 1/85 (1.2) | 27/156 (17.3) | 9/24 (37.5) | 0/15 (0) | 9/39 (23.1) | 35/95 (36.8) | 1/100 (1) | 36/195 (18.5) |
3.2. Antimicrobial Susceptibility of the Recovered Isolates
| Antimicrobial Resistance | |||
|---|---|---|---|
| Antibiotic | Chicken Livers np/n (%) * | Chicken Gizzards np/n (%) | Chicken Livers and Gizzard np/n (%) |
| azithromycin | 25/87 (28.7) | 22/28 (78.6) | 47/115 (40.9) |
| ciprofloxacin | 13/87 (14.9) | 21/28 (75.0) | 34/115 (29.6) |
| gentamicin | 10/87 (11.5) | 2/28 (7.1) | 12/115 (10.4) |
| oxacillin | 19/87 (21.8) | 18/28 (64.3) | 37/115 (32.2) |
| cefoxitin | 10/87 (11.5) | 12/28 (42.9) | 22/115 (19.1) |
| tetracycline | 59/87 (67.8) | 23/28 (82.1) | 82/115 (71.3) |
| vancomycin | 30/87 (34.5) | 15/28 (53.6) | 45/115 (39.1) |
| doxycycline | 49/87 (56.3) | 24/28 (85.7) | 73/115 (63.5) |
| trimethoprim/sulfamethoxazole | 22/87 (25.3) | 6/28 (21.4) | 28/115 (24.3) |
| clindamycin | 13/87 (14.9) | 1/28 (3.6) | 14/115 (12.2) |
| penicillin | 47/87 (54.0) | 23/28 (82.1) | 70/115 (60.9) |
| ampicillin | 83/87 (95.4) | 19/28 (67.9) | 102/115 (88.9) |
| kanamycin | 17/87 (19.5) | 7/28 (25.0) | 14/115 (12.2) |
| erythromycin | 31/87 (35.6) | 21/28 (75.0) | 52/115 (45.2) |
| rifampin | 25/87 (28.7) | 2/28 (7.1) | 27/115 (23.5) |
| chloramphenicol | 11/87 (12.6) | 1/28 (3.6) | 12/115 (10.4) |
3.3. Toxin Genes Possession Screening of the Recovered Isolates
| Prevalence of Toxin Genes | |||
|---|---|---|---|
| Toxin Gene | Chicken Livers np/n (%) * | Chicken Gizzards np/n (%) | Chicken Livers and Gizzards np/n (%) |
| sea | 0/87 (0) | 0/28 (0) | 0/115 (0) |
| seb-sec | 0/87 (0) | 0/28 (0) | 0/115 (0) |
| sec | 0/87 (0) | 0/28 (0) | 0/115 (0) |
| sed | 1/87 (1.2) | 0/28 (0) | 1/115 (0.9) |
| see | 0/87 (0) | 0/28 (0) | 0/115 (0) |
| seg | 14/87 (16.1) | 20/28 (71.43) | 34/115 (29.6) |
| seh | 5/87 (5.8) | 0/28 (0) | 5/115 (4.3) |
| sei | 23/87 (26.4) | 26/28 (92.9) | 49/115 (42.6) |
| sej | 0/87 (0) | 0/28 (0) | 0/115 (0) |
| tst | 0/87 (0) | 0/28 (0) | 0/115 (0) |
| eta | 0/87 (0) | 0/28 (0) | 0/115 (0) |
| etb | 0/87 (0) | 0/28 (0) | 0/115 (0) |
| lukE-lukD | 32/87 (36.8) | 10/28 (35.7) | 42/115 (36.5) |
| lukM | 0/87 (0) | 0/28 (0) | 0/115 (0) |
| hla | 85/87 (97.7) | 24/28 (85.7) | 109/115 (94.5) |
| hlb | 56/87 (64.4) | 0/28 (0) | 56/115 (48.7) |
| hld | 85/87 (97.7) | 24/28 (85.7) | 109/115 (94.5) |
| lukS-lukF | 0/87 (0) | 0/28 (0) | 0/115 (0%) |
3.4. Genotyping Using spa Typing
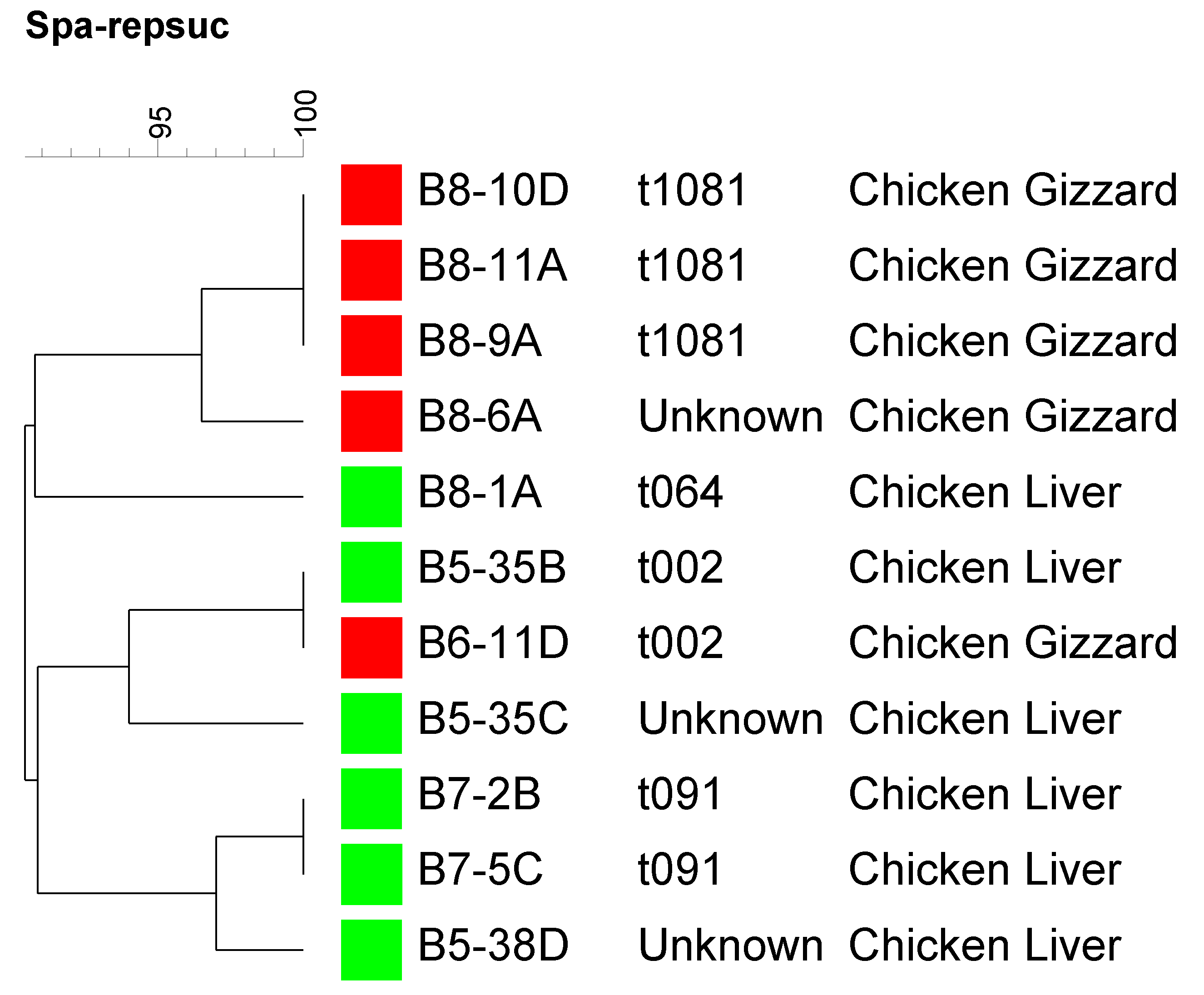
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Noormohamed, A.; Fakhr, M.K. Incidence and antimicrobial resistance profiling of Campylobacter in retail chicken livers and gizzards. Foodborne Pathog. Dis. 2012, 9, 617–624. [Google Scholar] [PubMed]
- Little, C.L.; GormLy, F.J.; Rawal, N.; Richardson, J.F. A recipe for disaster: Outbreaks of campylobacteriosis associated with poultry liver pâté in England and Wales. Epidemiol. Infect. 2010, 138, 1691–1694. [Google Scholar] [CrossRef] [PubMed]
- Abid, M.; Wimalarathna, H.; Mills, J.; Saldana, L.; Pang, W.; Richardson, J.F.; Martin, C.J.M.; McCarthy, N.D. Duck liver-associated outbreak of campylobacteriosis among humans, United Kingdom, 2011. Emerg. Infect. Dis. 2013, 19, 1310. [Google Scholar] [CrossRef] [PubMed]
- O’Leary, M.C.; Harding, O.; Fisher, L.; Cowden, J. A continuous common-source outbreak of campylobacteriosis associated with changes to the preparation of chicken liver pâté. Epidemiol. Infect. 2009, 137, 383–388. [Google Scholar] [CrossRef] [PubMed]
- Forbes, K.J.; GormLey, F.J.; Dallas, J.F.; Labovitiadi, O.; MacRae, M.; Owen, R.J.; Richardson, J.; McGuigan, C.C. Campylobacter immunity and coinfection following a large outbreak in a farming community. J. Clin. Microbiol. 2009, 47, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC). Multistate outbreak of Campylobacter jejuni infections associated with undercooked chicken livers—Northeastern United States, 2012. Morb. Mortal. Wkly. Rep. 2013, 62, 874. [Google Scholar]
- Terry, L. Outbreak Tied to Chicken Livers: 6 Sick, High-End Restaurants “Mortified”. Available online: http://www.oregonlive.com/health/index.ssf/2014/02/post_85.html (accessed on 12 August 2014).
- Centers for Disease Control (CDC). Multistate Outbreak of Human Salmonella Heidelberg Infections Linked to “Kosher Broiled Chicken livers” from Schreiber Processing Corporation. Available online: http://www.cdc.gov/salmonella/heidelberg-chickenlivers/011112/index.htmL (accessed on 12 August 2014).
- Kitai, S.; Shimizu, A.; Kawano, J.; Sato, E.; Nakano, C.; Uji, T.; Kitagawa, H. Characterization of methicillin-resistant Staphylococcus aureus isolated from retail raw chicken meat in Japan. J. Vet. Med. Sci. 2005, 67, 107–110. [Google Scholar] [CrossRef] [PubMed]
- Gundogan, N.; Citak, S.; Yucel, N.; Devren, Y.A. A note on the incidence and antibiotic resistance of Staphylococcus aureus isolated from meat and chicken samples. Meat Sci. 2005, 69, 807–810. [Google Scholar]
- Centers for Disease Control (CDC). CDC Estimates of Foodborne Illness in the United States. 2011. Available online: http://www.cdc.gov/foodborneburden/2011-foodborne-estimates.htmL (accessed on 16 May 2013). [Google Scholar]
- Genigeorgis, C.A. Present state of knowledge on staphylococcal intoxication. Int. J. Food Microbiol. 1989, 9, 327–360. [Google Scholar] [CrossRef] [PubMed]
- Zschöck, M.; Kloppert, B.; Wolter, W.; Hamann, H.P.; LämmLer, C. Pattern of enterotoxin genes seg, seh, sei and sej positive Staphylococcus aureus isolated from bovine mastitis. Vet. Microbiol. 2005, 108, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Ono, H.K.; Omoe, K.; Imanishi, K.; Iwakabe, Y.; Hu, D.L.; Kato, H.; Saito, N.; Nakane, A.; Uchiyama, T.; Shinagawa, K. Identification and characterization of two novel staphylococcal enterotoxin, types S and T. Infect. Immunol. 2008, 76, 4999–5005. [Google Scholar] [CrossRef]
- Wang, S.C.; Wu, C.M.; Xia, S.C.; Qi, Y.H.; Xia, L.N.; Shen, J.Z. Distribution of superantigenic toxin genes in Staphylococcus aureus isolates from milk samples of bovine subclinical mastitis cases in two major diary regions of China. Vet. Microbiol. 2009, 137, 276–281. [Google Scholar] [CrossRef] [PubMed]
- Argudín, M.A.; Mendoza, M.C.; Rodicio, M.R. Food poisoning and Staphylococcus aureus enterotoxins. Toxins 2010, 2, 1751–1773. [Google Scholar] [PubMed]
- Burnside, K.; Lembo, A.; de los Reyes, M.; Iliuk, A.; Binhtran, N.-T.; Connelly, J.E.; Lin, W.-J.; Schmidt, B.Z.; Richardson, A.R.; Fang, F.C.; et al. Regulation of hemolysin expression and virulence of Staphylococcus aureus by a serine/threonine kinase and phosphatase. PLoS ONE 2010, 5, e11071. [Google Scholar] [CrossRef] [PubMed]
- O’Callaghan, R.J.; Callegan, M.C.; Moreau, J.M.; Green, L.C.; Foster, T.J.; Hartford, O.M.; Engel, L.S.; Hill, J.M. Specific roles of alpha-toxin and beta-toxin during Staphylococcus aureus corneal infection. Infect. Immun. 1997, 65, 1571–1578. [Google Scholar] [PubMed]
- Kim, C.S.; Jeon, S.Y.; Min, Y.G.; Rhyoo, C.; Kim, J.W.; Yun, J.B.; Park, S.W.; Kwon, T.Y. Effects of betatoxin of Staphylococcus aureus on ciliary activity of nasal epithelial cells. Laryngoscope 2000, 110, 2085–2088. [Google Scholar] [CrossRef] [PubMed]
- Hayashida, A.; Bartlett, A.H.; Foster, T.J.; Park, P.W. Staphylococcus aureus betatoxin induces lung injury through syndecan-1. Am. J. Pathol. 2009, 174, 509–518. [Google Scholar] [CrossRef] [PubMed]
- De Neeling, A.J.; van Leeuwen, W.J.; Schouls, L.M.; Schot, C.S.; van Veen-Rutgers, A.; Beunders, A.J.; Buiting, A.G.; Hol, C.; Ligtvoet, E.E.; Petit, P.L.; et al. Resistance of staphylococci in The Netherlands: Surveillance by an electronic network during 1989–1995. J. Antimicrob. Chemoth. 1998, 41, 93–101. [Google Scholar] [CrossRef]
- Khanna, T.; Friendship, R.; Dewey, C.; Weese, J.S. Methicillin resistant Staphylococcus aureus colonization in pigs and pig farmers. Vet. Microbiol. 2008, 128, 298–303. [Google Scholar] [CrossRef] [PubMed]
- Voss, A.; Loeffen, F.; Bakker, J.; Klaassen, C.; Wulf, M. Methicillin resistant Staphylococcus aureus in pig farming. Emerg. Infect. Dis. 2005, 11, 1965–1966. [Google Scholar] [CrossRef] [PubMed]
- Van den Eede, A.; Hermans, K.; Lipinska, U.; Struelens, M.; Deplano, A.; Denis, O.; Gasthuys, F.; Haesebrouck, F.; Martens, A. Nasal carriage of methicillin-resistance Staphylococcus aureus in the equine population: Prevalence, typing and antimicrobial resistance, p20. In Proceedings of the 2nd Symposium on Antimicrobial Resistance in Animals and the Environment, ARAE-2007, Tours, France, 17–19 December 2007.
- De Boer, E.; Zwartkruis-Nahuis, J.T.M.; Wit, B.; Huijsdens, X.W.; de Neeling, A.J.; Bosch, T.; van Oosterom, R.A.A.; Vila, A.; Heuvelink, A.E. Prevalence of methicillin-resistant Staphylococcus aureus in meat. Int. J. Food Microbiol. 2009, 134, 52–56. [Google Scholar] [CrossRef]
- Buyukcangaz, E.; Velasco, V.; Sherwood, J.S.; Stepan, R.M.; Koslofsky, R.J.; Logue, C.M. Molecular typing of Staphylococcus aureus and methicillin-resistant S. aureus (MRSA) isolated from animals and retail meat in North Dakota, United States. Foodborne Pathog. Dis. 2013, 10, 608–617. [Google Scholar] [CrossRef] [PubMed]
- Velasco, V.; Sherwood, J.S.; Rojas-García, P.P.; Logue, C.M. Multiplex real-time PCR for detection of Staphylococcus aureus, mecA and Panton-Valentine Leukocidin (PVL) genes from selective enrichments from animals and retail meat. PloS ONE 2014, 9, e97617. [Google Scholar] [CrossRef] [PubMed]
- Jackson, C.R.; Davis, J.A.; Barrett, J.B. Prevalence and characterization of methicillin-resistant Staphylococcus aureus isolates from retail meat and humans in Georgia. J. Clin. Microbiol. 2013, 51, 1199–1207. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, A.M.; Hanson, B.M.; Farina, S.A.; Wu, J.Y.; Simmering, J.E.; Wardyn, S.E.; Smith, T.C. MRSA in conventional and alternative retail pork products. PLoS ONE 2012, 7, e30092. [Google Scholar] [CrossRef] [PubMed]
- Hanson, B.M.; Dressler, A.E.; Harper, A.L.; Scheibel, R.P.; Wardyn, S.E.; Roberts, L.K.; Smith, T.C. Prevalence of Staphylococcus aureus and methicillin-resistant Staphylococcus aureus (MRSA) on retail meat in Iowa. J. Infect. Public Health 2011, 4, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Bhargava, K.; Wang, X.; Donabedian, S.; Zervos, M.; da Rocha, L.; Zhang, Y. Methicillin-resistant Staphylococcus aureus in retail meat, Detroit, Michigan, USA. Emerg. Infect. Dis. 2011, 17, 1135. [Google Scholar] [CrossRef] [PubMed]
- Kelman, A.; Soong, Y.A.; Dupuy, N.; Shafer, D.; Richbourg, W.; Johnson, K.; Meng, J. Antimicrobial susceptibility of Staphylococcus aureus from retail ground meats. J. Food Prot. 2011, 74, 1625–1629. [Google Scholar] [CrossRef] [PubMed]
- Pu, S.; Wang, F.; Ge, B. Isolation and characterization of methicillin-resistant Staphylococcus aureus strains from Louisiana retail meats. Appl. Environ. Microb. 2009, 75, 265–267. [Google Scholar] [CrossRef]
- Martineau, F.; Picard, F.J.; Roy, P.H.; Ouellette, M.; Bergeron, M.G. Species-specific and ubiquitous-DNA-based assays for rapid identification of Staphylococcus aureus. J. Clin. Microbiol. 1998, 36, 618–623. [Google Scholar] [PubMed]
- Murakami, K.; Minamide, W.; Wada, K.; Nakamura, E.; Teraoka, H.; Watanabe, S. Identification of methicillin-resistant strains of staphylococci by polymerase chain reaction. J. Clin. Microbiol. 1991, 29, 2240–2244. [Google Scholar] [PubMed]
- Alvarez, L.G.; Holden, M.T.G.; Lindsay, H.; Webb, C.R.; Brown, D.F.J.; Curran, M.D.; Walpole, E.; Brooks, K.; Pickard, D.J.; Teale, C.; et al. Methicillin-resistant Staphylococcus aureus with a novel mecA homologue in human and bovine populations in the UK and Denmark: A descriptive study. Lancet 2011, 11, 595–603. [Google Scholar] [CrossRef] [PubMed]
- Shore, A.C.; Deasy, E.C.; Slickers, P.; Brennan, G.; Connell, B.O.; Monecke, S.; Ehricht, R.; Coleman, D.C. Detection of staphylococcal cassette chromosome mec Type XI carrying highly divergent mecA, mecI, mecR1, blaZ, and ccr genes in human clinical isolates of clonal complex 130 Methicillin-Resistant Staphylococcus aureus. Antimicrob. Agents Chemother. 2011, 55, 3765–3773. [Google Scholar] [CrossRef] [PubMed]
- Marmur, J. A procedure for the isolation of deoxyribonucleic acid from microorganisms. J. Mol. Bio. 1961, 3, 298–218. [Google Scholar]
- Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing; Twenty-First Informational Supplement. CLSI 2011, 31, 1–172. [Google Scholar]
- Monday, S.R.; Bohach, G.A. Use of multiplex PCR to detect classical and newly described pyrogenic toxin genes in staphylococcal isolates. J. Clin. Microbiol. 1999, 37, 3411–3414. [Google Scholar] [PubMed]
- Mehrotra, M.; Wang, G.; Johnson, W.M. Multiplex PCR for detection of genes for Staphylococcu aureus enterotoxins, exfoliative toxins, toxic shock syndrome toxin 1, and methicillin resistance. J. Clin. Microbiol. 2000, 38, 1032–1035. [Google Scholar] [PubMed]
- Jarraud, S.; Mougel, C.; Thioulouse, J.; Lina, G.; Meugnier, H.; Forey, F.; Nesme, X.; Etienne, J.; Vandenesch, F. Relationships between Staphylococcus aureus genetic background, virulence factors, agr groups (alleles), and human disease. Infect. Immun. 2002, 70, 631–641. [Google Scholar] [CrossRef] [PubMed]
- Shopsin, B.; Gomez, M.; Montgomery, S.O.; Smith, D.H.; Waddington, M.; Dodge, D.E.; Bost, D.E.; Riehman, M.; Naidich, S.; Kreiswirth, B.N. Evaluation of protein A gene polymorphic region DNA sequencing for typing of Staphylococcus aureus strains. J. Clin. Microbiol. 1999, 37, 3556–3563. [Google Scholar] [PubMed]
- Pereira, V.; Lopes, C.; Castro, A.; Silva, J.; Gibbs, P.; Teixeira, P. Characterization for enterotoxin production, virulence factors, and antibiotic susceptibility of Staphylococcus aureus isolates from various foods in Portugal. Food Microbiol. 2009, 26, 278–282. [Google Scholar] [CrossRef] [PubMed]
- Hetem, D.J.; Bootsma, M.C.; Troelstra, A.; Bonten, M.J. Transmissibility of livestock-associated methicillin-resistant Staphylococcus aureus. Emerg. Infect. Dis. 2013, 19, 1797. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdalrahman, L.S.; Fakhr, M.K. Incidence, Antimicrobial Susceptibility, and Toxin Genes Possession Screening of Staphylococcus aureus in Retail Chicken Livers and Gizzards. Foods 2015, 4, 115-129. https://doi.org/10.3390/foods4020115
Abdalrahman LS, Fakhr MK. Incidence, Antimicrobial Susceptibility, and Toxin Genes Possession Screening of Staphylococcus aureus in Retail Chicken Livers and Gizzards. Foods. 2015; 4(2):115-129. https://doi.org/10.3390/foods4020115
Chicago/Turabian StyleAbdalrahman, Lubna S., and Mohamed K. Fakhr. 2015. "Incidence, Antimicrobial Susceptibility, and Toxin Genes Possession Screening of Staphylococcus aureus in Retail Chicken Livers and Gizzards" Foods 4, no. 2: 115-129. https://doi.org/10.3390/foods4020115
APA StyleAbdalrahman, L. S., & Fakhr, M. K. (2015). Incidence, Antimicrobial Susceptibility, and Toxin Genes Possession Screening of Staphylococcus aureus in Retail Chicken Livers and Gizzards. Foods, 4(2), 115-129. https://doi.org/10.3390/foods4020115






