Using Bran of Ancient and Old Grains for Wheat Bread Production
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Materials
2.2. Proximate Chemical Composition of the Studied Wheat Brans
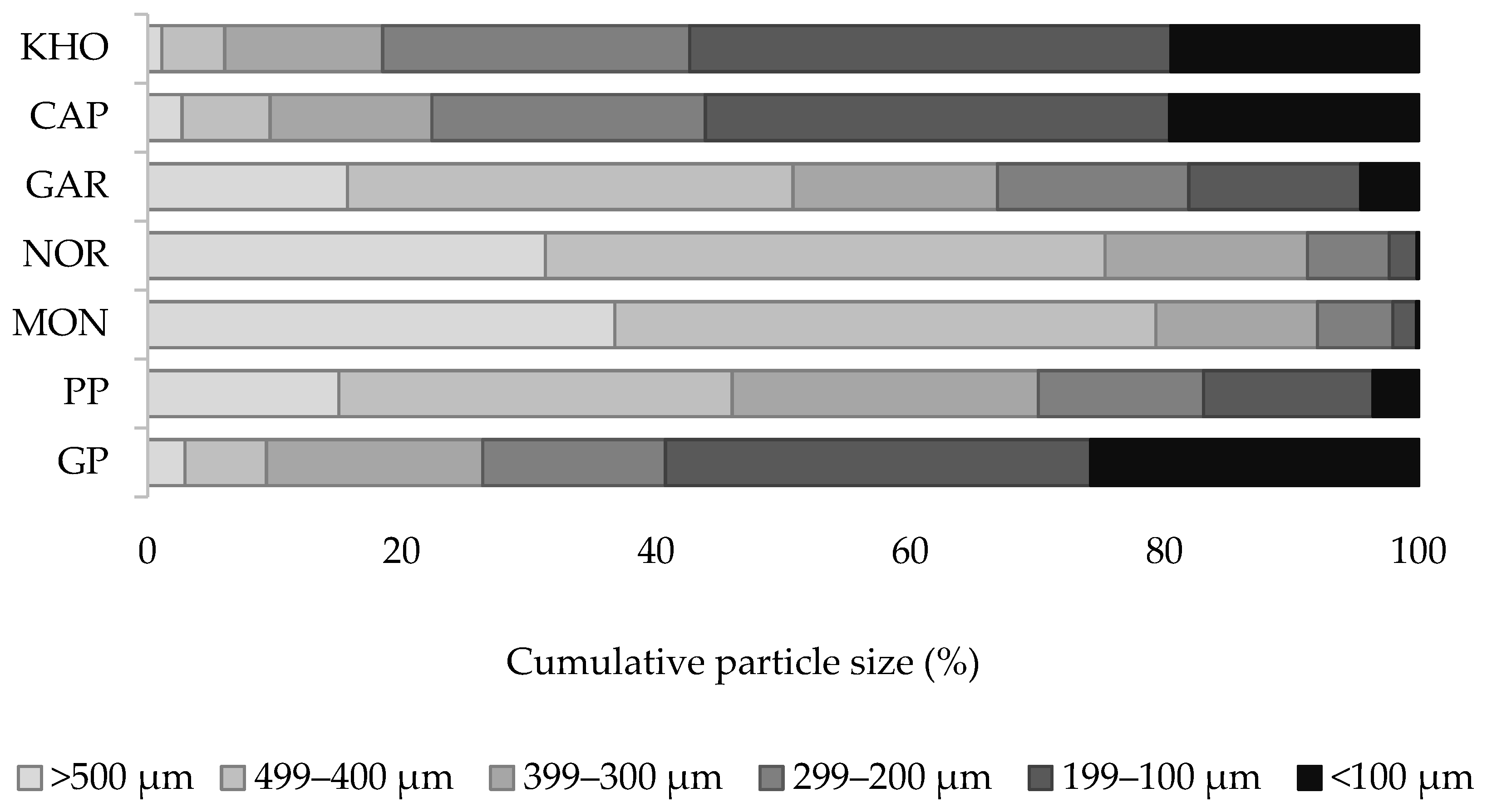
2.3. Particle Size Distribution
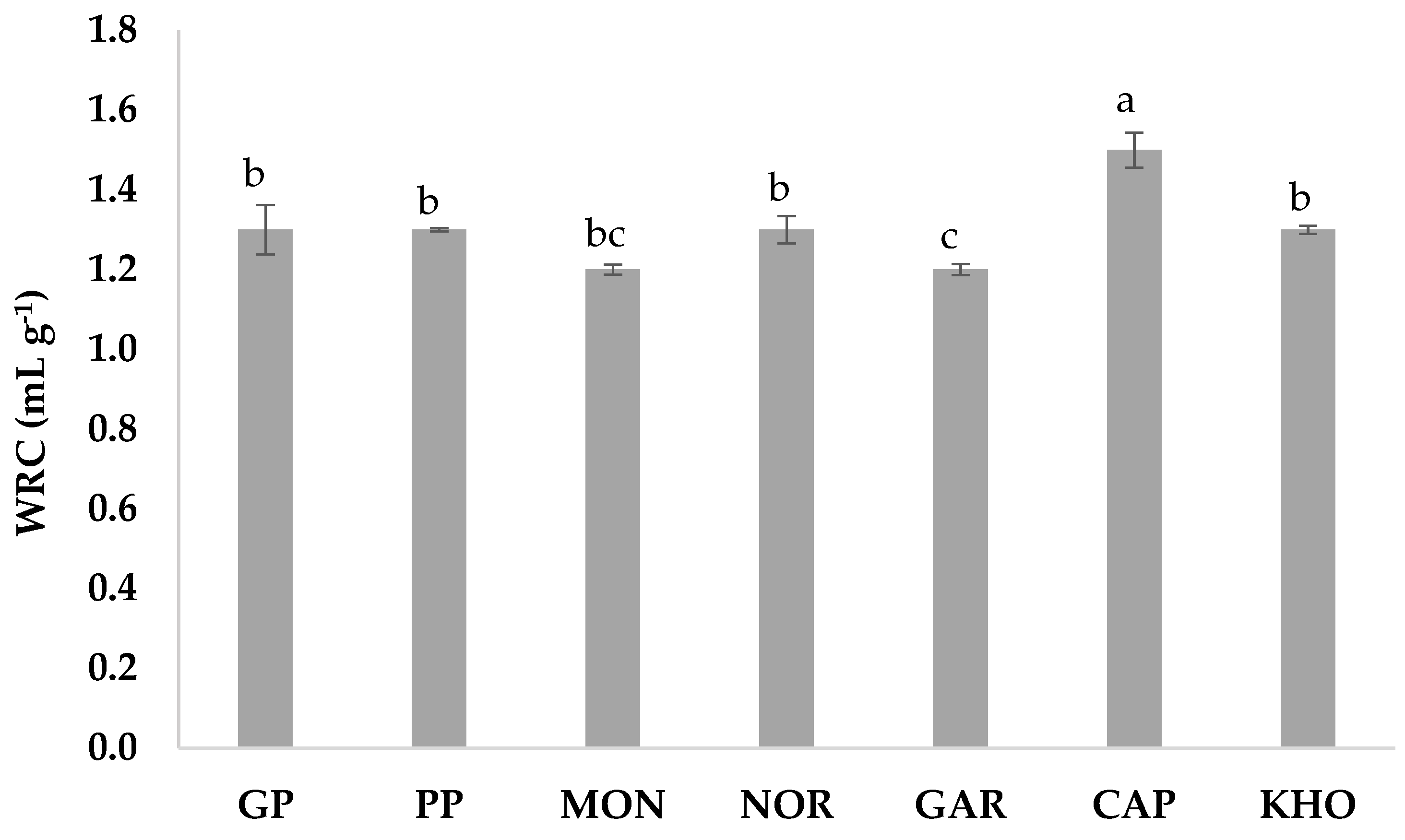
2.4. Water Retention Capacity
2.5. Dough Mixing Properties
2.6. Dough Preparation and Breadmaking Procedure
2.7. Dough Measurements
2.7.1. Dough Viscoelastic Properties
2.7.2. Dough Extensibility Measurements
2.7.3. Rheofermentometer Analysis
2.8. Bread Measurements
2.8.1. Moisture Content
2.8.2. Specific Volume
2.8.3. Textural Properties
2.8.4. Color Determination
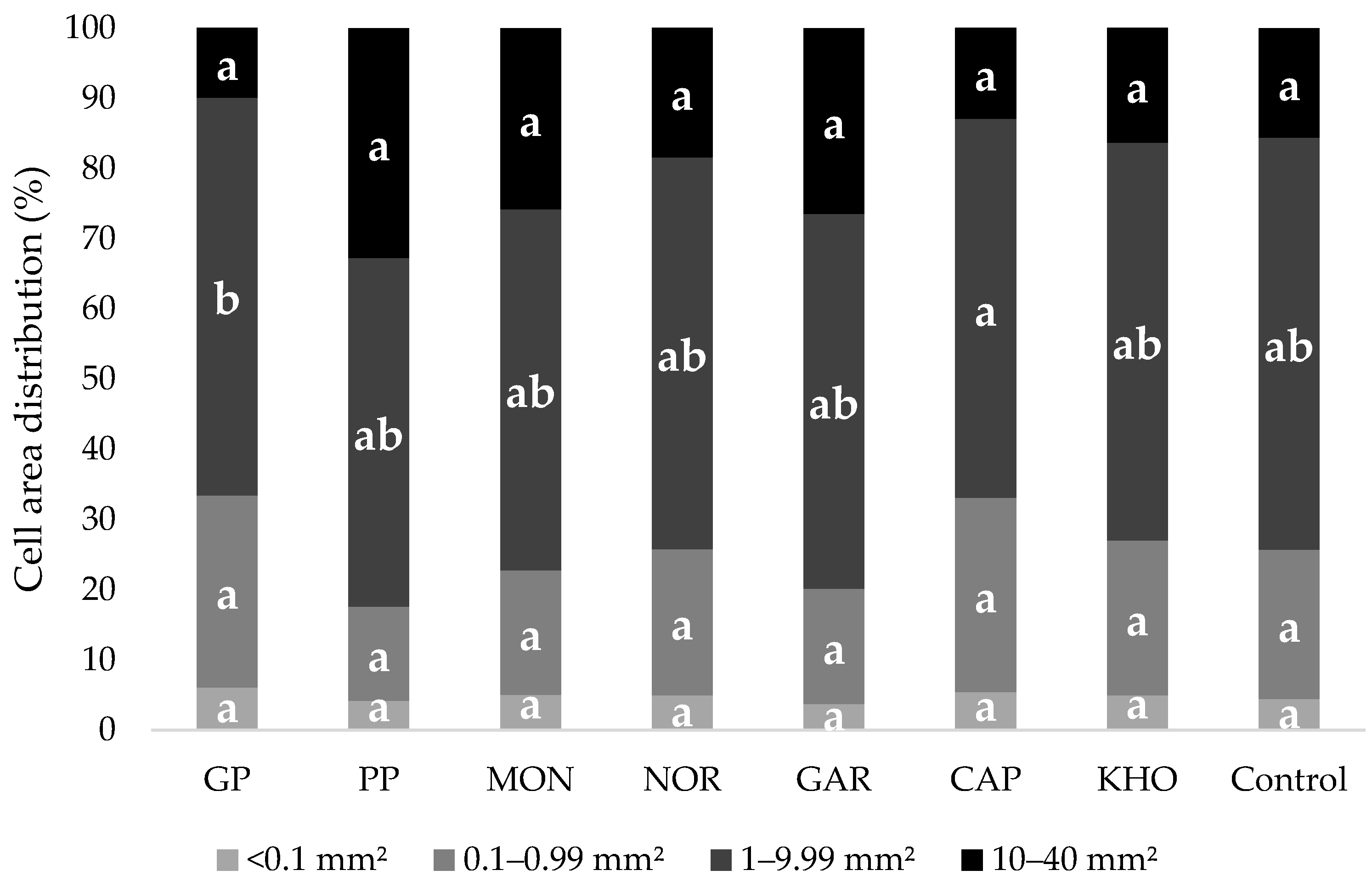
2.8.5. Crumb Grain Characteristics
2.9. Determination of Soluble, Insoluble, and Total Polyphenol Fractions
2.10. Determination of Antioxidant Activity
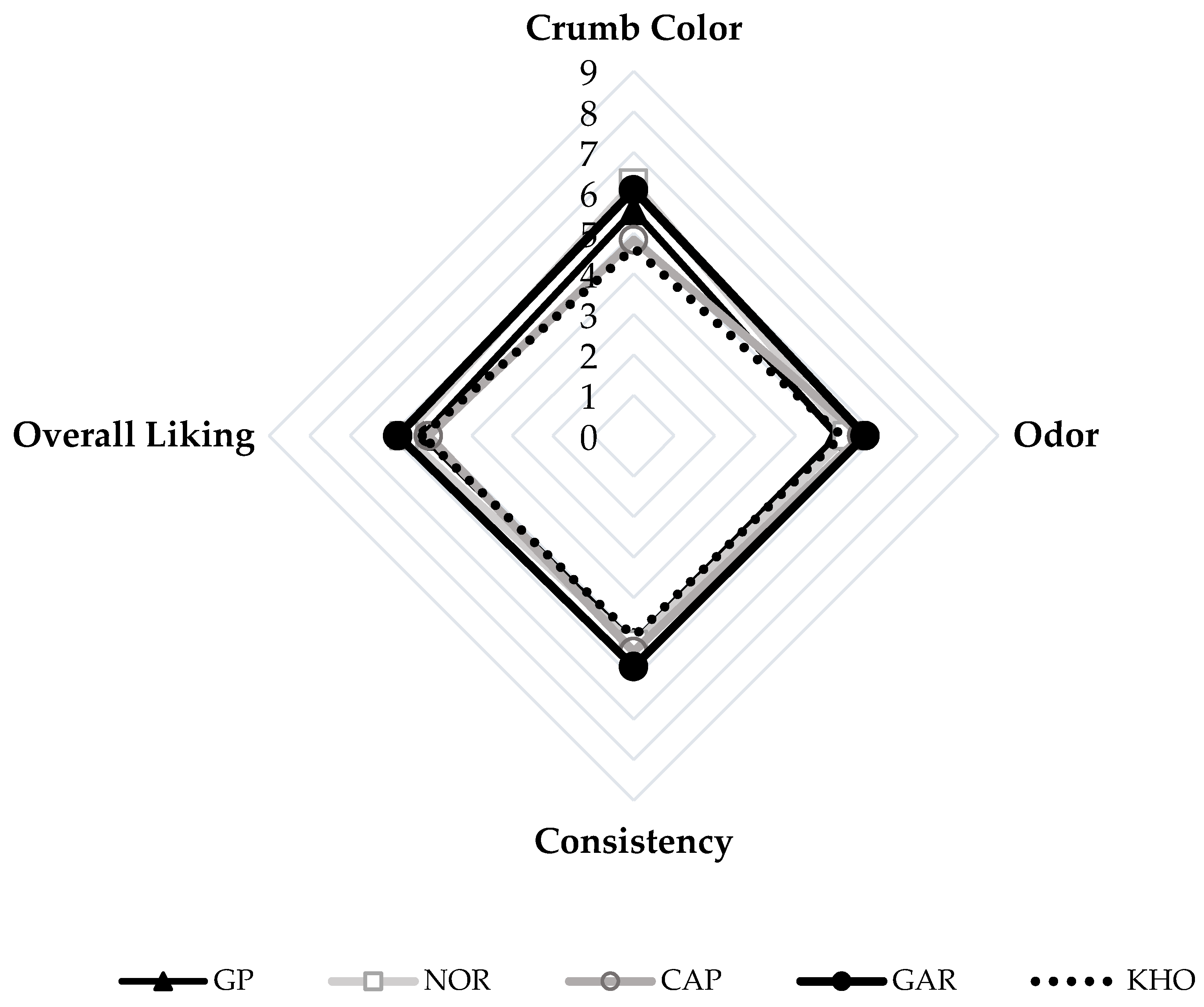
2.11. Sensory Evaluation
2.12. Statistical Analysis
3. Results and Discussion
3.1. Proximate Chemical Composition of Wheat Brans
3.2. Particle Size Distribution
3.3. Water Retention Capacity
3.4. Dough Measurements
3.4.1. Dough Mixing Properties
3.4.2. Dough Viscoelastic Properties and Extensibility Measurements
3.4.3. Rheofermentometer Analysis
3.5. Bread Analyses
3.5.1. Chemical Analysis
3.5.2. Bread Measurements
3.6. Polyphenol Fractions and Antioxidant Activity of Bran and Bread Samples
3.7. Sensory Evaluation
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAOSTAT. FAOSTAT Data. 2022. Available online: https://www.fao.org/faostat/en/#data/QCL (accessed on 31 December 2024).
- Shewry, P.R.; Hey, S. Do “Ancient” Wheat Species Differ from Modern Bread Wheat in Their Contents of Bioactive Components? J. Cereal Sci. 2015, 65, 236–243. [Google Scholar] [CrossRef]
- Faris, J.D.; Zhang, Z.; Chao, S. Map-Based Analysis of the Tenacious Glume Gene Tg-B1 of Wild Emmer and Its Role in Wheat Domestication. Gene 2014, 542, 198–208. [Google Scholar] [CrossRef] [PubMed]
- Doebley, J.F.; Gaut, B.S.; Smith, B.D. The Molecular Genetics of Crop Domestication. Cell 2006, 127, 1309–1321. [Google Scholar] [CrossRef]
- Mefleh, M.; Conte, P.; Fadda, C.; Giunta, F.; Piga, A.; Hassoun, G.; Motzo, R. From Ancient to Old and Modern Durum Wheat Varieties: Interaction among Cultivar Traits, Management, and Technological Quality. J. Sci. Food Agric. 2019, 99, 2059–2067. [Google Scholar] [CrossRef] [PubMed]
- Nocente, F.; Galassi, E.; Taddei, F.; Natale, C.; Gazza, L. Ancient Caucasian Wheats: A Contribution for Sustainable Diets and Food Diversity. Foods 2022, 11, 1209. [Google Scholar] [CrossRef] [PubMed]
- Shewry, P.R. Do Ancient Types of Wheat Have Health Benefits Compared with Modern Bread Wheat? J. Cereal Sci. 2018, 79, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Arzani, A.; Ashraf, M. Cultivated Ancient Wheats (Triticum spp.): A Potential Source of Health-Beneficial Food Products. Compr. Rev. Food Sci. Food Saf. 2017, 16, 477–488. [Google Scholar] [CrossRef]
- Gazza, L.; Galassi, E.; Nocente, F.; Natale, C.; Taddei, F. Cooking Quality and Chemical and Technological Characteristics of Wholegrain Einkorn Pasta Obtained from Micronized Flour. Foods 2022, 11, 2905. [Google Scholar] [CrossRef] [PubMed]
- Cappelli, A.; Cini, E. Challenges and Opportunities in Wheat Flour, Pasta, Bread, and Bakery Product Production Chains: A Systematic Review of Innovations and Improvement Strategies to Increase Sustainability, Productivity, and Product Quality. Sustainability 2021, 13, 2608. [Google Scholar] [CrossRef]
- Giambanelli, E.; Ferioli, F.; Koçaoglu, B.; Jorjadze, M.; Alexieva, I.; Darbinyan, N.; D’Antuono, L.F. A Comparative Study of Bioactive Compounds in Primitive Wheat Populations from Italy, Turkey, Georgia, Bulgaria and Armenia. J. Sci. Food Agric. 2013, 93, 3490–3501. [Google Scholar] [CrossRef] [PubMed]
- Boukid, F.; Folloni, S.; Sforza, S.; Vittadini, E.; Prandi, B. Current Trends in Ancient Grains-Based Foodstuffs: Insights into Nutritional Aspects and Technological Applications. Compr. Rev. Food Sci. Food Saf. 2018, 17, 123–136. [Google Scholar] [CrossRef] [PubMed]
- De Vita, P.; Riefolo, C.; Codianni, P.; Cattivelli, L.; Fares, C. Agronomic and Qualitative Traits of T. turgidum ssp. Dicoccum Genotypes Cultivated in Italy. Euphytica 2006, 150, 195–205. [Google Scholar] [CrossRef]
- Cadeddu, F.; Motzo, R.; Mureddu, F.; Giunta, F. Effects of Clipping on the Nitrogen Economy of Four Triticum Species Grown in a Mediterranean Environment. Field Crops Res. 2021, 267, 108162. [Google Scholar] [CrossRef]
- De Vita, P.; Nicosia, O.L.D.; Nigro, F.; Platani, C.; Riefolo, C.; Di Fonzo, N.; Cattivelli, L. Breeding Progress in Morpho-Physiological, Agronomical and Qualitative Traits of Durum Wheat Cultivars Released in Italy during the 20th Century. Eur. J. Agron. 2007, 26, 39–53. [Google Scholar] [CrossRef]
- Codianni, P.; Galterio, G.; Pogna, N.E.; Fonzo, N. di Mosè and Padre Pio, two new genotypes of hulled wheat. Inf. Agrar. 2000, 56, 37–38. [Google Scholar]
- Hemdane, S.; Jacobs, P.J.; Dornez, E.; Verspreet, J.; Delcour, J.A.; Courtin, C.M. Wheat (Triticum aestivum L.) Bran in Bread Making: A Critical Review. Compr. Rev. Food Sci. Food Saf. 2016, 15, 28–42. [Google Scholar] [CrossRef] [PubMed]
- Antoine, C.; Peyron, S.; Lullien-Pellerin, V.; Abecassis, J.; Rouau, X. Wheat Bran Tissue Fractionation Using Biochemical Markers. J. Cereal Sci. 2004, 39, 387–393. [Google Scholar] [CrossRef]
- Antoine, C.; Peyron, S.; Mabille, F.; Lapierre, C.; Bouchet, B.; Abecassis, J.; Rouau, X. Individual Contribution of Grain Outer Layers and Their Cell Wall Structure to the Mechanical Properties of Wheat Bran. J. Agric. Food Chem. 2003, 51, 2026–2033. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Moore, W.R. Effect of Wheat Bran Particle Size on Dough Rheological Properties. J. Sci. Food Agric. 1997, 74, 490–496. [Google Scholar] [CrossRef]
- Maes, C.; Delcour, J.A. Alkaline Hydrogen Peroxide Extraction of Wheat Bran Non-Starch Polysaccharides. J. Cereal Sci. 2001, 34, 29–35. [Google Scholar] [CrossRef]
- Bataillon, M.; Mathaly, P.; Nunes Cardinali, A.-P.; Duchiron, F. Extraction and Purification of Arabinoxylan from Destarched Wheat Bran in a Pilot Scale. Ind. Crops Prod. 1998, 8, 37–43. [Google Scholar] [CrossRef]
- Verspreet, J.; Dornez, E.; Van den Ende, W.; Delcour, J.A.; Courtin, C.M. Cereal Grain Fructans: Structure, Variability and Potential Health Effects. Trends Food Sci. Technol. 2015, 43, 32–42. [Google Scholar] [CrossRef]
- Nordlund, E.; Aura, A.-M.; Mattila, I.; Kössö, T.; Rouau, X.; Poutanen, K. Formation of Phenolic Microbial Metabolites and Short-Chain Fatty Acids from Rye, Wheat, and Oat Bran and Their Fractions in the Metabolical in Vitro Colon Model. J. Agric. Food Chem. 2012, 60, 8134–8145. [Google Scholar] [CrossRef] [PubMed]
- Hemery, Y.; Rouau, X.; Dragan, C.; Bilici, M.; Beleca, R.; Dascalescu, L. Electrostatic Properties of Wheat Bran and Its Constitutive Layers: Influence of Particle Size, Composition, and Moisture Content. J. Food Eng. 2009, 93, 114–124. [Google Scholar] [CrossRef]
- EFSA Panel on Dietetic Products; NDA. Scientific Opinion on the Substantiation of Health Claims Related to Wheat Bran Fibre and Increase in Faecal Bulk (ID 3066), Reduction in Intestinal Transit Time (ID 828, 839, 3067, 4699) and Contribution to the Maintenance or Achievement of a Normal Body Weight (ID 829) Pursuant to Article 13(1) of Regulation (EC) No 1924/2006. EFSA J. 2010, 8, 1817. [Google Scholar] [CrossRef]
- Lebesi, D.M.; Tzia, C. Effect of the Addition of Different Dietary Fiber and Edible Cereal Bran Sources on the Baking and Sensory Characteristics of Cupcakes. Food Bioprocess Technol. 2011, 4, 710–722. [Google Scholar] [CrossRef]
- Sozer, N.; Cicerelli, L.; Heiniö, R.-L.; Poutanen, K. Effect of Wheat Bran Addition on in Vitro Starch Digestibility, Physico-Mechanical and Sensory Properties of Biscuits. J. Cereal Sci. 2014, 60, 105–113. [Google Scholar] [CrossRef]
- Kaur, G.; Sharma, S.; Nagi, H.P.S.; Dar, B.N. Functional Properties of Pasta Enriched with Variable Cereal Brans. J. Food Sci. Technol. 2012, 49, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Sobota, A.; Rzedzicki, Z.; Zarzycki, P.; Kuzawińska, E. Application of Common Wheat Bran for the Industrial Production of High-Fibre Pasta. Int. J. Food Sci. Technol. 2015, 50, 111–119. [Google Scholar] [CrossRef]
- Almeida, E.L.; Chang, Y.K.; Steel, C.J. Dietary Fibre Sources in Bread: Influence on Technological Quality. LWT Food Sci. Technol. 2013, 50, 545–553. [Google Scholar] [CrossRef]
- Pavlovich-Abril, A.; Rouzaud-Sández, O.; Romero-Baranzini, A.L.; Vidal-Quintanar, R.L.; Salazar-García, M.G. Relationships between Chemical Composition and Quality-Related Characteristics in Bread Making with Wheat Flour–Fine Bran Blends. J. Food Qual. 2015, 38, 30–39. [Google Scholar] [CrossRef]
- Kim, B.-K.; Cho, A.-R.; Chun, Y.-G.; Park, D.-J. Effect of Microparticulated Wheat Bran on the Physical Properties of Bread. Int J. Food Sci. Nutr. 2013, 64, 122–129. [Google Scholar] [CrossRef] [PubMed]
- AACC. Approved Methods; American Association of Cereal Chemists (AACC): Saint Paul, MN, USA, 2000. [Google Scholar]
- AOAC. Official method 2011.25. Insoluble, Soluble, and Total Dietary Fiber in Foods Enzymatic-Gravimetric-Liquid Chromatography First Action. In Official Methods of Analysis of AOAC International, 19th ed.; Horowitz, W., Latimer, G.W.J., Eds.; AOAC International: Gaithersburg, MD, USA, 2011. [Google Scholar]
- ASTM C136-06; Standard Test Method for Sieve Analysis of Fine and Coarse Aggregates. ASTM Standards: West Cochocken, PA, USA, 2006; Volume 4, pp. 1–5.
- AACCI. Method 56-11.02. Solvent Retention Capacity Profile. In Approved Methods of Analysis, 11th ed.; AACC International: St. Paul, MN, USA, 2009. [Google Scholar]
- AACC. AACC International Method 54-21.01. In Methods of the American Association of Cereal Chemists, 10th ed.; AACC: St. Paul, MN, USA, 2000. [Google Scholar] [CrossRef]
- AACC. Approved Methods of Analysis; American Association of Cereal Chemists (AACC): Saint Paul, MN, USA, 2005. [Google Scholar]
- AACC. AACC-I AACC International Approved Methods of Analysis; AACC: St. Paul, MN, USA, 2009. [Google Scholar]
- Collar, C.; Jiménez, T.; Conte, P.; Fadda, C. Impact of Ancient Cereals, Pseudocereals and Legumes on Starch Hydrolysis and Antiradical Activity of Technologically Viable Blended Breads. Carbohydr. Polym. 2014, 113, 149–158. [Google Scholar] [CrossRef]
- Conte, P.; Del Caro, A.; Urgeghe, P.P.; Petretto, G.L.; Montanari, L.; Piga, A.; Fadda, C. Nutritional and Aroma Improvement of Gluten-Free Bread: Is Bee Pollen Effective? LWT 2020, 118, 108711. [Google Scholar] [CrossRef]
- Cannas, M.; Conte, P.; Piga, A.; Del Caro, A. Green Recovery Optimization of Phenolic Compounds from “Spinoso Sardo” Globe Artichoke by-Products Using Response Surface Methodology. Front. Sustain. Food Syst. 2023, 7, 1215809. [Google Scholar] [CrossRef]
- Meilgaard, M.C.; Carr, B.T.; Carr, B.T. Sensory Evaluation Techniques, 4th ed.; CRC Press: Boca Raton, FL, USA, 2006; ISBN 9780429195143. [Google Scholar]
- Stone, H.; Bleibaum, R.N.; Thomas, H.A. Chapter 7—Affective Testing. In Sensory Evaluation Practices, 4th ed.; Stone, H., Bleibaum, R.N., Thomas, H.A., Eds.; Food Science and Technology; Academic Press: San Diego, CA, USA, 2012; pp. 291–325. ISBN 978-0-12-382086-0. [Google Scholar]
- ISO 8589; Standards Sensory Analysis. Methodology. General Guidance. ISO: Geneva, Switzerland, 2006.
- Boita, E.R.F.; Oro, T.; Bressiani, J.; Santetti, G.S.; Bertolin, T.E.; Gutkoski, L.C. Rheological Properties of Wheat Flour Dough and Pan Bread with Wheat Bran. J. Cereal Sci. 2016, 71, 177–182. [Google Scholar] [CrossRef]
- Mefleh, M.; Boukid, F.; Fadda, C. Suitability of Improved and Ancient Italian Wheat for Bread-Making: A Holistic Approach. Life 2022, 12, 1613. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Gao, J.; Jin, X.; Wang, Y.; Dong, Z.; Ying, J.; Zhou, W. Whole-Wheat Flour Particle Size Influences Dough Properties, Bread Structure and in Vitro Starch Digestibility. Food Funct. 2020, 11, 3610–3620. [Google Scholar] [CrossRef]
- Noort, M.W.J.; van Haaster, D.; Hemery, Y.; Schols, H.A.; Hamer, R.J. The Effect of Particle Size of Wheat Bran Fractions on Bread Quality—Evidence for Fibre–Protein Interactions. J. Cereal Sci. 2010, 52, 59–64. [Google Scholar] [CrossRef]
- Jacobs, P.J.; Hemdane, S.; Dornez, E.; Delcour, J.A.; Courtin, C.M. Study of Hydration Properties of Wheat Bran as a Function of Particle Size. Food Chem. 2015, 179, 296–304. [Google Scholar] [CrossRef]
- Zhu, K.; Huang, S.; Peng, W.; Qian, H.; Zhou, H. Effect of Ultrafine Grinding on Hydration and Antioxidant Properties of Wheat Bran Dietary Fiber. Food Res. Int. 2010, 43, 943–948. [Google Scholar] [CrossRef]
- Liu, N.; Ma, S.; Li, L.; Wang, X. Study on the Effect of Wheat Bran Dietary Fiber on the Rheological Properties of Dough. Grain Oil Sci. Technol. 2019, 2, 1–5. [Google Scholar] [CrossRef]
- Gómez, M.; Jiménez, S.; Ruiz, E.; Oliete, B. Effect of Extruded Wheat Bran on Dough Rheology and Bread Quality. LWT Food Sci. Technol. 2011, 44, 2231–2237. [Google Scholar] [CrossRef]
- Sanz Penella, J.M.; Collar, C.; Haros, M. Effect of Wheat Bran and Enzyme Addition on Dough Functional Performance and Phytic Acid Levels in Bread. J. Cereal Sci. 2008, 48, 715–721. [Google Scholar] [CrossRef]
- Laurikainen, T.; Härkönen, H.; Autio, K.; Poutanen, K. Effects of Enzymes in Fibre-Enriched Baking. J. Sci. Food Agric. 1998, 76, 239–249. [Google Scholar] [CrossRef]
- Chen, H.; Rubenthaler, G.L.; Schanus, E.G. Effect of Apple Fiber and Cellulose on the Physical Properties of Wheat Flour. J. Food Sci. 1988, 53, 304–305. [Google Scholar] [CrossRef]
- Jin, X.; Lin, S.; Gao, J.; Wang, Y.; Ying, J.; Dong, Z.; Zhou, W. How Manipulation of Wheat Bran by Superfine-Grinding Affects a Wide Spectrum of Dough Rheological Properties. J. Cereal Sci. 2020, 96, 103081. [Google Scholar] [CrossRef]
- Wang, C.C.; Yang, Z.; Xing, J.J.; Guo, X.N.; Zhu, K.X. Effects of Insoluble Dietary Fiber and Ferulic Acid on the Rheological Properties of Dough. Food Hydrocoll. 2021, 121, 107008. [Google Scholar] [CrossRef]
- Liu, D.; Song, S.; Tao, L.; Yu, L.; Wang, J. Effects of Common Buckwheat Bran on Wheat Dough Properties and Noodle Quality Compared with Common Buckwheat Hull. LWT 2022, 155, 112971. [Google Scholar] [CrossRef]
- Packkia-Doss, P.P.; Chevallier, S.; Pare, A.; Le-Bail, A. Effect of Supplementation of Wheat Bran on Dough Aeration and Final Bread Volume. J. Food Eng. 2019, 252, 28–35. [Google Scholar] [CrossRef]
- Liu, W.; Brennan, M.; Serventi, L.; Brennan, C. Effect of Wheat Bran on Dough Rheology and Final Quality of Chinese Steamed Bread. Cereal Chem. 2017, 94, 581–587. [Google Scholar] [CrossRef]
- Wang, N.; Hou, G.G.; Kweon, M.; Lee, B. Effects of Particle Size on the Properties of Whole-Grain Soft Wheat FLour and Its Cracker Baking Performance. J. Cereal Sci. 2016, 69, 187–193. [Google Scholar] [CrossRef]
- Liu, T.; Hou, G.G.; Cardin, M.; Marquart, L.; Dubat, A. Quality Attributes of Whole-Wheat Flour Tortillas with Sprouted Whole-Wheat Flour Substitution. LWT 2017, 77, 1–7. [Google Scholar] [CrossRef]
- Wang, J.; Rosell, C.M.; Benedito de Barber, C. Effect of the Addition of Different Fibres on Wheat Dough Performance and Bread Quality. Food Chem. 2002, 79, 221–226. [Google Scholar] [CrossRef]
- Bressiani, J.; Oro, T.; Santetti, G.S.; Almeida, J.L.; Bertolin, T.E.; Gómez, M.; Gutkoski, L.C. Properties of Whole Grain Wheat Flour and Performance in Bakery Products as a Function of Particle Size. J. Cereal Sci. 2017, 75, 269–277. [Google Scholar] [CrossRef]
- Majzoobi, M.; Farahnaky, A.; Nematolahi, Z.; Hashemi, M.M.; Taghipour, M. Effect of Different Levels and Particle Sizes of Wheat Bran on the Quality of Flat Bread. J. Agric. Sci. Technol. 2013, 15, 115–123. [Google Scholar]
- de Kock, S.; Taylor, J.; Taylor, J.R.N. Effect of Heat Treatment and Particle Size of Different Brans on Loaf Volume of Brown Bread. LWT Food Sci. Technol. 1999, 32, 349–356. [Google Scholar] [CrossRef]
- Sun, R.; Zhang, Z.; Hu, X.; Xing, Q.; Zhuo, W. Effect of Wheat Germ Flour Addition on Wheat Flour, Dough and Chinese Steamed Bread Properties. J. Cereal Sci. 2015, 64, 153–158. [Google Scholar] [CrossRef]
- Zhang, D.; Moore, W.R. Wheat Bran Particle Size Effects on Bread Baking Performance and Quality. J. Sci. Food Agric. 1999, 79, 805–809. [Google Scholar] [CrossRef]
- Azizi, M.; Sayeddain, S.M.; Payghambardoost, S.H. Effect of Flour Extraction Rate on Flour Composition, Dough Rheological Characteristics and Quality of Flat Bread. J. Agric. Sci. Technol. 2006, 8, 323–330. [Google Scholar]
- Heiniö, R.L.; Noort, M.W.J.; Katina, K.; Alam, S.A.; Sozer, N.; de Kock, H.L.; Hersleth, M.; Poutanen, K. Sensory Characteristics of Wholegrain and Bran-Rich Cereal Foods—A Review. Trends Food Sci. Technol. 2016, 47, 25–38. [Google Scholar] [CrossRef]
- Kim, K.-H.; Tsao, R.; Yang, R.; Cui, S.W. Phenolic Acid Profiles and Antioxidant Activities of Wheat Bran Extracts and the Effect of Hydrolysis Conditions. Food Chem. 2006, 95, 466–473. [Google Scholar] [CrossRef]
- Yadav, M.P.; Kaur, A.; Singh, B.; Simon, S.; Kaur, N.; Powell, M.; Sarker, M. Extraction and Characterization of Lipids and Phenolic Compounds from the Brans of Different Wheat Varieties. Food Hydrocoll. 2021, 117, 106734. [Google Scholar] [CrossRef]
- Verma, B.; Hucl, P.; Chibbar, R.N. Phenolic Content and Antioxidant Properties of Bran in 51 Wheat Cultivars. Cereal Chem. 2008, 85, 544–549. [Google Scholar] [CrossRef]
- Adom, K.K.; Sorrells, M.E.; Liu, R.H. Phytochemicals and Antioxidant Activity of Milled Fractions of Different Wheat Varieties. J. Agric. Food Chem. 2005, 53, 2297–2306. [Google Scholar] [CrossRef]
- Zhang, L.; Gao, W.; Chen, X.; Wang, H. The Effect of Bioprocessing on the Phenolic Acid Composition and Antioxidant Activity of Wheat Bran. Cereal Chem. 2014, 91, 255–261. [Google Scholar] [CrossRef]
- Nguyen, S.N.; Beta, T. Cereal-Derived Polyphenols and Their Bioactive Properties. Curr. Opin. Food Sci. 2024, 56, 101136. [Google Scholar] [CrossRef]
- Dziki, D.; Różyło, R.; Gawlik-Dziki, U.; Świeca, M. Current Trends in the Enhancement of Antioxidant Activity of Wheat Bread by the Addition of Plant Materials Rich in Phenolic Compounds. Trends Food Sci. Technol. 2014, 40, 48–61. [Google Scholar] [CrossRef]
- Alvarez-Jubete, L.; Wijngaard, H.; Arendt, E.K.; Gallagher, E. Polyphenol Composition and in Vitro Antioxidant Activity of Amaranth, Quinoa Buckwheat and Wheat as Affected by Sprouting and Baking. Food Chem. 2010, 119, 770–778. [Google Scholar] [CrossRef]
- Han, H.M.; Koh, B.-K. Effect of Phenolic Acids on the Rheological Properties and Proteins of Hard Wheat Flour Dough and Bread. J. Sci. Food Agric. 2011, 91, 2495–2499. [Google Scholar] [CrossRef]


| Wheat Type | Genotypes | Code | Scientific Names |
|---|---|---|---|
| Improved emmer | Padre Pio | PP | T. turgidum ssp. dicoccum Schubler var. Padre Pio |
| Giovanni Paolo | GP | T. turgidum ssp. dicoccum Schubler var. Giovanni Paolo | |
| Improved einkorn | Monlis | MON | T. monocuccum ssp. monocuccum var. Monlis |
| Norberto | NOR | T. monocuccum ssp. monocuccum var. Norberto | |
| Old wheat | Cappelli | CAP | T. turgidum ssp. durum (Desf.) |
| Emmer landrace | Garfagnana | GAR | T. turgidum ssp. dicoccum |
| Ancient wheat | Khorasan | KHO | T. turgidum ssp. turanicum |
| Samples * | Protein | Ash | Fat | TDF |
|---|---|---|---|---|
| GP | 22.58 ± 0.56 a | 3.34 ± 0.12 d | 2.46 ± 0.31 c | 19.10 ± 0.20 f |
| PP | 19.78 ± 1.29 d | 4.85 ± 0.94 c | 4.01 ± 1.01 b | 22.89 ± 0.06 d |
| MON | 21.87 ± 0.07 b | 5.57 ± 0.16 b | 5.67 ± 0.38 a | 25.14± 0.35 b |
| NOR | 23.21 ±0.11 a | 6.28 ±0.18 a | 5.52 ±0.17 a | 27.17 ±0.05 a |
| GAR | 18.54 ± 0.12 e | 4.78 ± 0.03 c | 4.29 ±0.32 b | 21.97 ± 0.15 e |
| CAP | 20.40 ± 0.12 cd | 3.42 ± 0.05 d | 2.09 ± 0.11 c | 24.46 ± 0.34 bc |
| KHO | 18.56 ± 0.04 e | 3.42 ± 0.10 d | 2.28 ± 0.02 c | 24.27 ± 0.01 b |
| Samples * | Water Absorption (%) | Dough Development Time (min) | Stability (min) | Dough Softening (BU) |
|---|---|---|---|---|
| GP | 59.8 a | 1.7 ± 1 b | 20.0 ± 0 a | 14.5 ± 2 ab |
| PP | 57.5 a | 1.4 ± 1 b | 19.2 ± 2 a | 19.0 ± 7 ab |
| MON | 57.5 a | 2.8 ± 2 ab | 19.1 ± 2 a | 8.0 ± 2 ab |
| NOR | 58.5 a | 5.1 ± 0 a | 19.1 ± 1 a | 11.0 ± 1 ab |
| GAR | 57 a | 2.1 ± 0 ab | 20.0 ± 0 a | 5.5 ± 0 b |
| CAP | 59 a | 1.4 ± 0 b | 12.2 ± 0 b | 16.5 ± 6 ab |
| KHO | 58.5 a | 1.4 ± 0 b | 11.0 ± 0 b | 22.5 ± 3 a |
| Control | 54 b | 1.8 ± 0 b | 19.0 ± 0 a | 9.0 ± 0 ab |
| Viscoelastic Parameters | Extensibility Parameters | ||||
|---|---|---|---|---|---|
| Samples * | G′ (Pa) | G″ (Pa) | Tan δ | R (N) | E (mm) |
| GP | 26,388 ± 1268 a | 9658 ± 332 a | 0.367 ± 0.001 ab | 0.15 ± 0.02 a | 48.07 ± 0.04 b |
| PP | 22,267 ± 65 bc | 8057 ± 466 bcd | 0.362 ± 0.020 b | 0.17 ± 0.01 a | 34.51 ± 0.60 d |
| MON | 18,150 ± 32 de | 7319 ± 175 de | 0.405 ± 0.001 a | 0.10 ± 0.00 b | 35.77 ± 0.12 d |
| NOR | 17,101 ± 815 e | 6709 ± 114 e | 0.393 ± 0.001 ab | 0.11 ± 0.00 b | 33.78 ± 0.02 d |
| GAR | 23,658 ± 163 b | 8642 ± 95 bc | 0.365 ± 0.000 b | 0.15 ± 0.00 a | 39.91 ± 0.51 c |
| CAP | 20,767 ± 690 cd | 7904 ± 185 cd | 0.380 ± 0.000 ab | 0.17 ± 0.00 a | 35.15 ± 0.09 d |
| KHO | 23,781 ± 924 ab | 8971 ± 175 ab | 0.377 ± 0.001 ab | 0.15 ± 0.00 a | 41.66 ± 1.55 c |
| Control | 14,201 ± 240 f | 5640 ± 143 f | 0.398 ± 0.013 ab | 0.09 ± 0.00 b | 54.91 ± 5.47 a |
| Dough Development | Gas Release | ||||||
|---|---|---|---|---|---|---|---|
| Samples * | Hm (mm) | T’2 (min) | H’m (mm) | T’1 (min) | V-TOT (mL) | V-REL (mL) | RC (%) |
| GP | 26.05 ± 0.78 b | 38.15 ± 1.14 c | 76.40 ± 2.69 a | 62.15 ± 3.11 cd | 1839 ± 67 a | 559 ± 58 a | 70 ± 2 d |
| PP | 19.10 ± 0.14 c | 29.00 ± 2.50 d | 71.95 ± 2.76 ab | 114.45 ± 7.25 ab | 1580 ± 39 cde | 333 ± 52 cd | 79 ± 4 ab |
| MON | 20.75 ± 0.49 bc | 29.15 ± 1.04 d | 80.75 ± 0.64 a | 99.45 ± 24.24 abc | 1711 ± 11 b | 485 ± 8 ab | 72 ± 0 cd |
| NOR | 25.75 ± 1.91 b | 38.15 ± 1.04 c | 79.00 ± 1.98 a | 120.00 ± 4.15 ab | 1721 ± 2 ab | 503 ± 5 ab | 71 ± 0 d |
| GAR | 20.50 ± 0.71 bc | 34.30 ± 4.15 cd | 71.30 ± 4.53 ab | 81.45 ± 13.47 bcd | 1615 ± 27 bcd | 424 ± 21 bcd | 74 ± 1 abcd |
| CAP | 24.35 ± 2.05 bc | 54.00 ± 2.07 b | 64.90 ± 0.85 b | 129.45 ± 7.25 a | 1512 ± 26 de | 304 ± 22 d | 80 ± 2 a |
| KHO | 24.15 ± 2.76 bc | 30.45 ± 1.04 cd | 79.70 ± 3.82 a | 54.45 ± 5.18 d | 1639 ± 11 bc | 453 ± 27 abc | 73 ± 2 bcd |
| Control | 47.40 ± 0.69 a | 66.01 ± 3.11 a | 71.90 ± 2.56 ab | 85.01 ± 12.10 bcd | 1472 ± 21 e | 322 ± 21 d | 78 ± 2 abc |
| Samples * | Moisture | Protein | Ash | Fat | TDF |
|---|---|---|---|---|---|
| GP | 37.84 ± 0.22 a | 19.6 ± 0.3 a | 4.18 ± 0.08 bc | 0.36 ± 0.01 c | 14.59 ± 0.05 b |
| PP | 36.57 ± 0.38 b | 19.2 ± 0.1 ab | 4.28 ± 0.05 b | 0.42 ± 0.01 b | 10.77 ± 0.15 d |
| MON | 37.11 ± 0.20 ab | 19.6 ± 0.2 a | 4.34 ± 0.10 b | 0.51 ± 0.02 a | 11.71 ± 0.60 cd |
| NOR | 37.20 ± 0.02 ab | 19.1 ± 0.1 ab | 4.63 ± 0.04 a | 0.44 ± 0.02 b | 15.08 ± 0.16 ab |
| GAR | 36.53 ± 0.03 b | 18.5 ± 0.1 c | 4.30 ± 0.03 b | 0.23 ± 0.01 d | 15.51 ± 0.07 ab |
| CAP | 36.31 ± 0.38 b | 18.9 ± 0.1 bc | 4.04 ± 0.06 c | 0.24 ± 0.01 d | 12.32 ± 0.25 c |
| KHO | 37.74 ± 0.16 a | 18.5 ± 0.1 c | 4.24 ± 0.03 b | 0.37 ± 0.01 c | 16.09 ± 0.10 a |
| Control | 34.86 ± 0.17 c | 15.4 ± 0.4 d | 3.44 ± 0.08 d | 0.07 ± 0.02 e | 10.75 ± 0.24 d |
| Samples * | Specific Volume (mL g−1) | Hardness (N) | Chewiness |
|---|---|---|---|
| GP | 2.46 ± 0.04 d | 17 ± 0.75 a | 12.77 ± 1.46 ab |
| PP | 2.47 ± 0.03 cd | 18.98 ± 1.33 a | 15.39 ± 1.2 a |
| MON | 2.6 ± 0.02 b | 14.19 ± 0.21 ab | 11.79 ± 0.08 ab |
| NOR | 2.55 ± 0.02 bcd | 15.63 ± 2.74 ab | 12.83 ± 1.75 ab |
| GAR | 2.57 ± 0.03 bc | 15.17 ± 2.11 ab | 12.21 ± 1.50 ab |
| CAP | 2.62 ± 0.02 b | 15.71 ± 0.01 ab | 13.09 ± 0.00 ab |
| KHO | 2.64 ± 0.01 b | 16.43 ± 0.39 ab | 13.66 ± 0.15 ab |
| Control | 2.97 ± 0.03 a | 11.44 ± 0.07 b | 9.99 ± 0.16 b |
| Crust Color | Crumb Color | ||||
|---|---|---|---|---|---|
| Samples * | Lcrust | acrust | Lcrumb | acrumb | bcrumb |
| GP | 56.11 ± 2.76 bc | 12.27 ± 0.80 ab | 60.19 ± 0.74 bc | 3.78 ± 0.02 b | 18.91 ± 0.09 b |
| PP | 54.28 ± 0.67 bc | 13.18 ± 0.36 a | 61.54 ± 1.31 bc | 5.27 ± 0.10 a | 19.37 ± 0.27 b |
| MON | 51. 69± 1.64 bc | 13.17 ± 0.65 a | 60.30 ± 0.59 bc | 4.84 ± 0.31 a | 21.33 ± 0.36 a |
| NOR | 51.34 ± 1.19 c | 13.33 ± 0.72 a | 59.30 ± 1.79 c | 5.22 ± 0.02 a | 21.56 ± 0.05 a |
| GAR | 55.47 ± 0.88 bc | 11.83 ± 0.39 ab | 60.26 ± 1.50 bc | 5.00 ± 0.11 a | 18.66 ± 0.05 b |
| CAP | 56.11 ± 1.37 bc | 12.34 ± 0.37 a | 62.79 ± 1.32 bc | 2.27 ± 0.57 c | 20.25 ± 0.33 ab |
| KHO | 57.6 ± 1.50 ab | 11.23 ± 0.74 ab | 64.04 ± 0.06 b | 2.17 ± 0.29 c | 20.30 ± 0.84 ab |
| Control | 62.50 ± 1.15 a | 10.02 ± 0.25 b | 69.90 ± 0.74 a | - 0.57 ± 0.04 d | 14.82 ± 0.65 c |
| Samples * | Polyphenol Fractions (mg GAE 100 g−1 dm) | Antioxidant Activity (μmol TE g−1 dm) | |||
|---|---|---|---|---|---|
| Soluble | Insoluble | IP/SP | Total | ABTS | |
| GP | 102 ± 2 d | 331 ± 12 bc | 3.24 ± 0.04 a | 433 ± 2 d | 73.0 b |
| PP | 125 ± 2 c | 326 ± 5 cd | 2.61 ± 0.05 b | 451 ± 9 c | 70.6 b |
| MON | 163 ± 2 b | 342 ± 7 a | 2.10 ± 0.01 d | 506 ± 3 b | 104.8 a |
| NOR | 178 ± 6 a | 339 ± 2 ab | 1.91 ± 0.08 e | 517 ± 9 a | 106.7 a |
| GAR | 124 ± 1 c | 301 ± 4 e | 2.42 ± 0.05 c | 426 ± 2 de | 58.3 c |
| CAP | 100 ± 4 d | 320 ± 4 d | 3.20 ± 0.03 a | 420 ± 5 e | 48.3 d |
| KHO | 92 ± 0 e | 291 ± 5 f | 3.18 ± 0.00 a | 382 ± 7 f | 45.5 e |
| Samples * | Polyphenol Fractions (mg GAE 100 g−1 dm) | Antioxidant Activity (μmol TE g−1 dm) | |||
|---|---|---|---|---|---|
| Soluble | Insoluble | IP/SP | Total | ABTS | |
| GP | 47 ± 2 c | 308 ± 7 d | 6.54 ± 0.2 c | 355 ± 6 c | 60.9 b |
| PP | 60 ± 2 b | 321 ± 12 bc | 5.38 ± 0.1 d | 380 ± 13 b | 47.9 f |
| MON | 65 ± 6 a | 332 ± 2 a | 5.08 ± 0.3 d | 398 ± 1 a | 51.1 e |
| NOR | 61 ± 4 b | 329 ± 4 ab | 5.40 ± 0.2 d | 390 ± 3 ab | 71.6 a |
| GAR | 47 ± 0 c | 307 ± 5 d | 6.48 ± 0.1 c | 354 ± 5 c | 58.4 c |
| CAP | 45 ± 1 c | 317 ± 4 cd | 6.97 ± 0.1 b | 362 ± 4 c | 56.7 d |
| KHO | 46 ± 2 c | 296 ± 5 e | 6.41 ± 0.0 c | 342 ± 5 d | 52 e |
| Control | 28 ± 2 d | 254 ± 5 f | 9.16 ± 0.5 a | 281 ± 5 e | 45.4 g |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Toumi, O.; Fadda, C.; Del Caro, A.; Conte, P. Using Bran of Ancient and Old Grains for Wheat Bread Production. Foods 2025, 14, 860. https://doi.org/10.3390/foods14050860
Toumi O, Fadda C, Del Caro A, Conte P. Using Bran of Ancient and Old Grains for Wheat Bread Production. Foods. 2025; 14(5):860. https://doi.org/10.3390/foods14050860
Chicago/Turabian StyleToumi, Oumayma, Costantino Fadda, Alessandra Del Caro, and Paola Conte. 2025. "Using Bran of Ancient and Old Grains for Wheat Bread Production" Foods 14, no. 5: 860. https://doi.org/10.3390/foods14050860
APA StyleToumi, O., Fadda, C., Del Caro, A., & Conte, P. (2025). Using Bran of Ancient and Old Grains for Wheat Bread Production. Foods, 14(5), 860. https://doi.org/10.3390/foods14050860







