Toward Nutritionally Sound Plant-Based Meat Analogues: Expert Consensus from a Delphi Study
Abstract
1. Introduction
2. Methods
2.1. Expert Panel
2.2. Delphi Methodology
2.2.1. Round One
2.2.2. Round Two
2.2.3. Round Three
2.3. Statistic Analysis
3. Results and Discussion
3.1. Panel Expert Characteristics
3.2. Delphi Round One
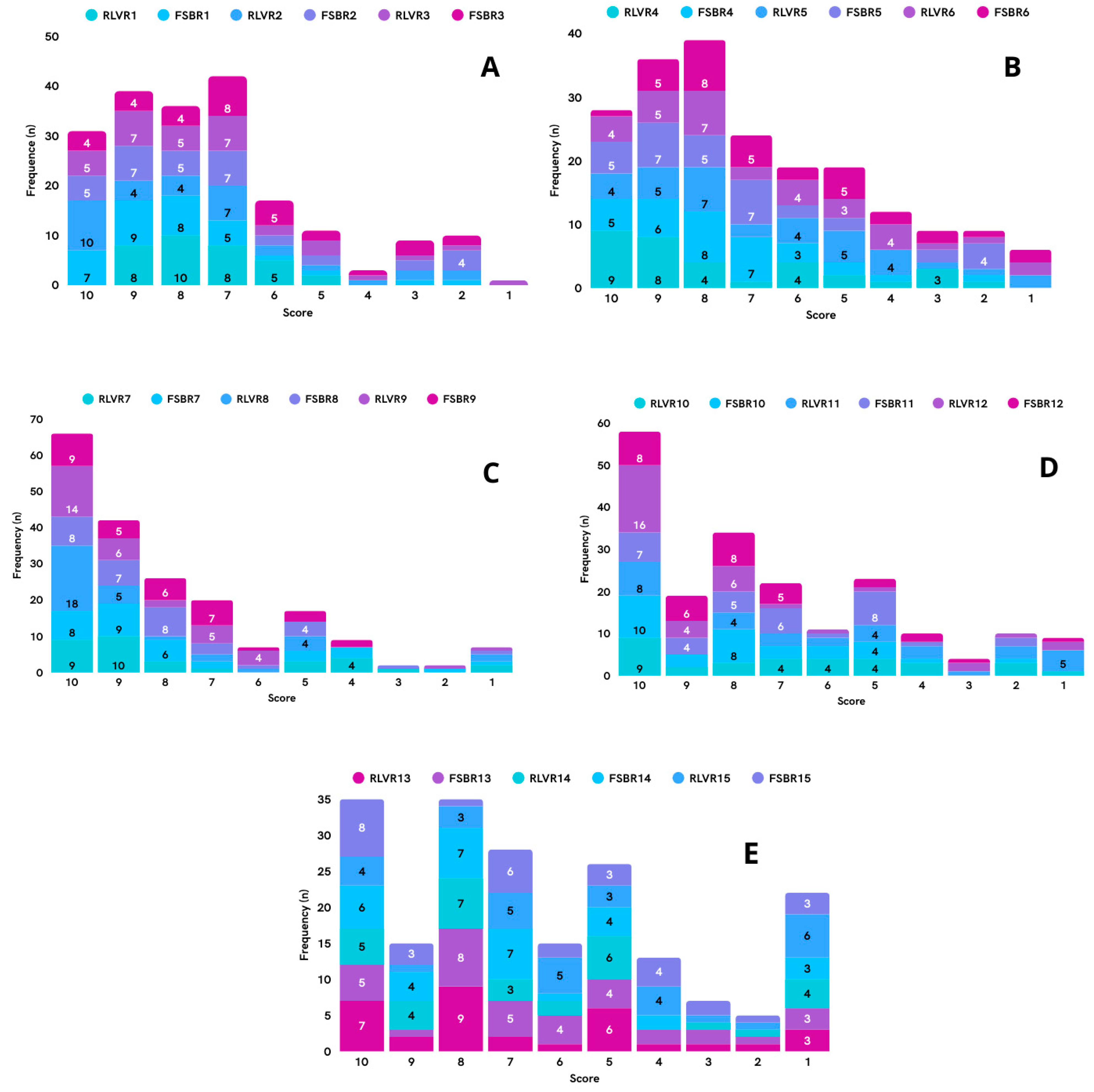
3.3. Delphi Round Two
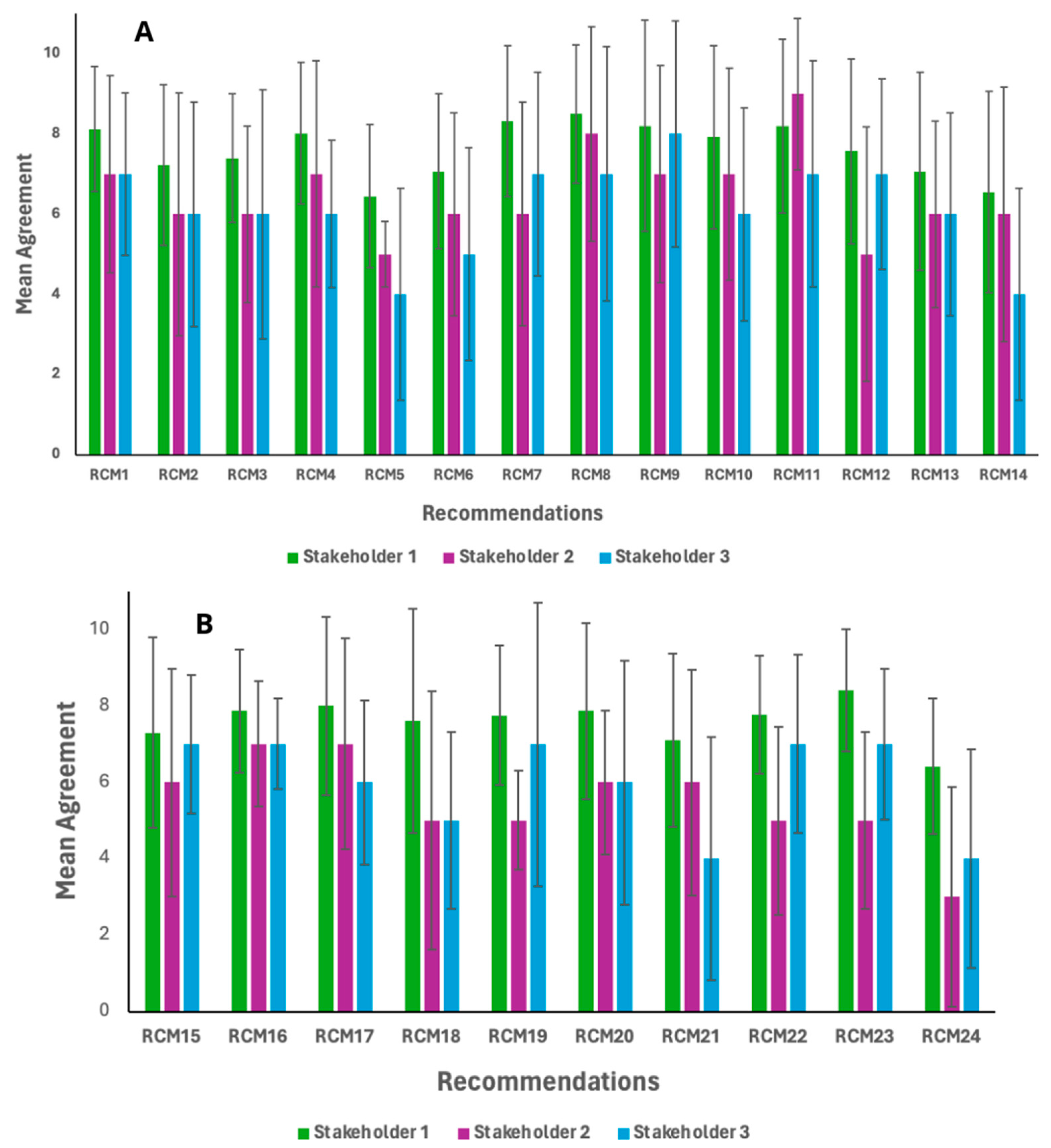
3.4. Delphi Round Three
3.5. Practical Implications
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Correction Statement
References
- Trishita, D. Veganism Statistics 2025 By Plants, Diet, Lifestyle. Available online: https://media.market.us/veganism-statistics/ (accessed on 27 May 2025).
- The Good Food Institute. O Consumidor Brasileiro e o Mercado “Plant-Based”; The Good Food Institute: São Paulo, Brazil, 2022. [Google Scholar]
- Camila, L.; Raquel, C. The Brazilian Consumer and the Plant-Based Market, 1st ed.; The Good Food Institute Brazil: São Paulo, Brazil, 2025; Volume 1. [Google Scholar]
- Ghaffari, M.; Rodrigo, P.G.K.; Ekinci, Y.; Pino, G. Consumers’ Motivations for Adopting a Vegan Diet: A Mixed-Methods Approach. Int. J. Consum. Stud. 2022, 46, 1193–1208. [Google Scholar] [CrossRef]
- Jeżewska-Zychowicz, M.; Sajdakowska, M.; Gębski, J.; Kosicka-Gębska, M.; Gutkowska, K. Diet Quality and Past Changes in Food Consumption as Determinants of Intentions to Eat Less Meat and More Plant-Based Foods. Foods 2024, 13, 3767. [Google Scholar] [CrossRef]
- Ministry of Health. Guia Alimentar Para População Brasileira; Ministry of Health: Brasilia, Brazil, 2014.
- WHO; FAO-Word Health Organization; Food and Agriculture Organization of United Nations. Diet, Nutrition and the Prevention of Chronic Diseases: Report of a Joint WHO/FAO Expert Consultation; WHO/FAO-Word Health Organization/Food and Agriculture Organization of United Nations: Geneve, Switzerland, 2002. [Google Scholar]
- Perez-Cueto, F.J.A.; Rini, L.; Faber, I.; Rasmussen, M.A.; Bechtold, K.B.; Schouteten, J.J.; De Steur, H. How Barriers towards Plant-Based Food Consumption Differ According to Dietary Lifestyle: Findings from a Consumer Survey in 10 EU Countries. Int. J. Gastron. Food Sci. 2022, 29, 100587. [Google Scholar] [CrossRef]
- Ambika, S.; Bhupathiraju, S.N.; Spiegelman, D.; Chiuve, S.E.; Manson, J.E.; Willett, W.; Rexrode, K.M.; MPH; Rimm, E.B.; Hu, F.B. Healthful and Unhealthful Plant-Based Diets and the Risk of Coronary Heart Disease in U.S. Adults. JACC 2017, 70, 411–422. [Google Scholar] [CrossRef]
- Vellinga, R.E.; Rippin, H.L.; Gonzales, G.B.; Temme, E.H.M.; Farrand, C.; Halloran, A.; Clough, B.; Wickramasinghe, K.; Santos, M.; Fontes, T.; et al. Nutritional Composition of Ultra-Processed Plant-Based Foods in the out-of-Home Environment: A Multi-Country Survey with Plant-Based Burgers. Br. J. Nutr. 2024, 131, 1691–1698. [Google Scholar] [CrossRef]
- Kirchner, J.; Leet-Otley, T. Plant-Based Meat Consumer Segmentation Introduction and Summary. Available online: https://gfi.org/event/decoding-the-market-plant-based-meat-consumer-segmentation/ (accessed on 29 August 2025).
- Newton, P.; Eichhorst, W.; Hegwood, M.; Morais-da-Silva, R.L.; Heidemann, M.S.; Hoffmann, A.; Reis, G.G. Price above All Else: An Analysis of Expert Opinion on the Priority Actions to Scale up Production and Consumption of Plant-Based Meat in Brazil. Front. Sustain. Food Syst. 2024, 8. [Google Scholar] [CrossRef]
- Kent, G.; Kehoe, L.; Flynn, A.; Walton, J. Plant-Based Diets: A Review of the Definitions and Nutritional Role in the Adult Diet. Proc. Nutr. Soc. 2022, 81, 62–74. [Google Scholar] [CrossRef]
- Tuso, P.J.; Ismail, M.H.; Ha, B.P.; Bartolotto, C. Nutritional Update for Physicians: Plant-Based Diets. Perm. J. 2013, 17, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Toribio-Mateas, M.A.; Bester, A.; Klimenko, N. Impact of Plant-Based Meat Alternatives on the Gut Microbiota of Consumers: A Real-World Study. Foods 2021, 10, 2040. [Google Scholar] [CrossRef]
- Jensen, C.F. Vitamin B12 Levels in Children and Adolescents on Plant-Based Diets: A Systematic Review and Meta-Analysis. Nutr. Rev. 2023, 81, 951–966. [Google Scholar] [CrossRef] [PubMed]
- García-Maldonado, E.; Zapatera, B.; Alcorta, A.; Vaquero, M.P. Metabolic and Nutritional Biomarkers in Adults Consuming Lacto-Ovo Vegetarian, Vegan and Omnivorous Diets in Spain. A Cross-Sectional Study. Food Funct. 2023, 14, 1608–1616. [Google Scholar] [CrossRef]
- Kesse-Guyot, E.; Allès, B.; Brunin, J.; Fouillet, H.; Dussiot, A.; Mariotti, F.; Langevin, B.; Berthy, F.; Touvier, M.; Julia, C.; et al. Nutritionally Adequate and Environmentally Respectful Diets Are Possible for Different Diet Groups: An Optimized Study from the NutriNet-Santé Cohort. Am. J. Clin. Nutr. 2022, 116, 1621–1633. [Google Scholar] [CrossRef]
- He, J.; Shen, X.; Fang, A.; Song, J.; Li, H.; Guo, M.; Li, K. Association between Predominantly Plant-Based Diets and Iron Status in Chinese Adults: A Cross-Sectional Analysis. Br. J. Nutr. 2016, 116, 1621–1632. [Google Scholar] [CrossRef]
- Hargreaves, S.M.; Araújo, W.M.C.; Nakano, E.Y.; Zandonadi, R.P. Brazilian Vegetarians Diet Quality Markers and Comparison with the General Population: A Nationwide Cross-Sectional Study. PLoS ONE 2020, 15, e0235991. [Google Scholar] [CrossRef]
- Neufingerl, N.; Eilander, A. Nutrient Intake and Status in Adults Consuming Plant-Based Diets Compared to Meat-Eaters: A Systematic Review. Nutrients 2022, 14, 29. [Google Scholar] [CrossRef]
- Higuera, J.M.; Santos, H.M.; Oliveira, A.F.; Nogueira, A.R.A. Animal and Vegetable Protein Burgers: Bromatological Analysis, Mineral Composition, and Bioaccessibility Evaluation. ACS Food Sci. Technol. 2021, 1, 1821–1829. [Google Scholar] [CrossRef]
- Luz, G.M.; Orlando, E.A.; Rebellato, A.P.; Greiner, R.; Pallone, J.A.L. Essential Minerals and Anti-Nutritional Compounds in Plant-Based Burgers Using the Infogest in Vitro Digestion Protocol. J. Food Compos. Anal. 2024, 135, 106574. [Google Scholar] [CrossRef]
- Arranz, E.; Segat, A.; Velayos, G.; Flynn, C.; Brodkorb, A.; Giblin, L. Dairy and Plant Based Protein Beverages: In Vitro Digestion Behaviour and Effect on Intestinal Barrier Biomarkers. Food Res. Int. 2023, 169, 112815. [Google Scholar] [CrossRef] [PubMed]
- Buss, D. Food Technology Magazine. Available online: https://www.ift.org/news-and-publications/food-technology-magazine/issues/2023/february/features/1-rebuilding-plant-based-meat (accessed on 29 August 2025).
- Rizzolo-Brime, L.; Orta-Ramirez, A.; Puyol Martin, Y.; Jakszyn, P. Nutritional Assessment of Plant-Based Meat Alternatives: A Comparison of Nutritional Information of Plant-Based Meat Alternatives in Spanish Supermarkets. Nutrients 2023, 15, 1325. [Google Scholar] [CrossRef] [PubMed]
- De Marchi, M.; Costa, A.; Pozza, M.; Goi, A.; Manuelian, C.L. Detailed Characterization of Plant-Based Burgers. Sci. Rep. 2021, 11, 2049. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, M.; Qureshi, S.; Akbar, M.H.; Siddiqui, S.A.; Gani, A.; Mushtaq, M.; Hassan, I.; Dhull, S.B. Plant-Based Meat Alternatives: Compositional Analysis, Current Development and Challenges. Appl. Food Res. 2022, 2, 100154. [Google Scholar] [CrossRef]
- Locatelli, N.T.; Chen, G.F.N.; Batista, M.F.; Furlan, J.M.; Wagner, R.; Bandoni, D.H.; de Rosso, V.V. Nutrition Classification Schemes for Plant-Based Meat Analogues: Drivers to Assess Nutritional Quality and Identity Profile. Curr. Res. Food Sci. 2024, 9, 100796. [Google Scholar] [CrossRef] [PubMed]
- Bryngelsson, S.; Moshtaghian, H.; Bianchi, M.; Hallström, E. Nutritional Assessment of Plant-Based Meat Analogues on the Swedish Market. Int. J. Food Sci. Nutr 2022, 73, 889–901. [Google Scholar] [CrossRef]
- Biazotto, K.R.; Xavier, A.C.H.; de Mattos, R.R.; Furlan, J.M.; Wagner, R.; Bandoni, D.H.; de Rosso, V.V. Plant-Based Burgers in the Spotlight: A Detailed Composition Evaluation and Comprehensive Discussion on Nutrient Adequacy. Foods 2025, 14, 372. [Google Scholar] [CrossRef] [PubMed]
- Ložnjak Švarc, P.; Jensen, M.B.; Langwagen, M.; Poulsen, A.; Trolle, E.; Jakobsen, J. Nutrient Content in Plant-Based Protein Products Intended for Food Composition Databases. J. Food Compos. Anal. 2022, 106, 104332. [Google Scholar] [CrossRef]
- Cutroneo, S.; Prandi, B.; Faccini, A.; Pellegrini, N.; Sforza, S.; Tedeschi, T. Comparison of Protein Quality and Digestibility between Plant-Based and Meat-Based Burgers. Food Res. Int. 2023, 172, 113183. [Google Scholar] [CrossRef]
- Cutroneo, S.; Angelino, D.; Tedeschi, T.; Pellegrini, N.; Martini, D. Nutritional Quality of Meat Analogues: Results From the Food Labelling of Italian Products (FLIP) Project. Front. Nutr. 2022, 9, 852831. [Google Scholar] [CrossRef]
- Latunde-Dada, G.O.; Kajarabille, N.; Rose, S.; Arafsha, S.M.; Kose, T.; Aslam, M.F.; Hall, W.L.; Sharp, P.A. Content and Availability of Minerals in Plant-Based Burgers Compared with a Meat Burger. Nutrients 2023, 15, 2732. [Google Scholar] [CrossRef]
- Swing, C.; Thompson, T.; Guimaraes, O.; Geornaras, I.; Engle, T.; Belk, K.; Gifford, C.; Nair, M. Nutritional Composition of Novel Plant-Based Meat Alternatives and Traditional Animal-Based Meats. Food Sci. Nutr. 2021, 7, 109. [Google Scholar] [CrossRef]
- He, J.; Liu, H.; Balamurugan, S.; Shao, S. Fatty Acids and Volatile Flavor Compounds in Commercial Plant-Based Burgers. J. Food Sci. 2021, 86, 293–305. [Google Scholar] [CrossRef]
- Cuhls, K.E. Argumentative Delphi Surveys: Lessons for Sociological Research. Am. Sociol. 2024, 55, 120–141. [Google Scholar] [CrossRef]
- Delbecq, A.L.; Van de Ven, A.H.; Gustafson, D.H. Group Techniques for Program Planning: A Guide to Nominal Group and Delphi Processes; Wageningen University: Wageningen, The Netherlands, 1976. [Google Scholar]
- Guseva Canu, I.; Marca, S.C.; Dell’Oro, F.; Balázs, Á.; Bergamaschi, E.; Besse, C.; Bianchi, R.; Bislimovska, J.; Bjelajac, A.K.; Bugge, M.; et al. Harmonized Definition of Occupational Burnout: A Systematic Review, Semantic Analysis, and Delphi Consensus in 29 Countries. Scand. J. Work. Environ. Health 2021, 47, 95–107. [Google Scholar] [CrossRef]
- Seemann, N.M.; Moulton, E.; Humphrey-Murto, S. Capturing the Complexity of Acute Stress in the Health Professions: A Review of Methods for Measuring Stress and Considerations for Moving Forward. Artic. Issues 2016, 91, S11–S12. [Google Scholar] [CrossRef]
- Wu, F.; Qin, Y.; Hou, D.; Xie, S.; Zhang, G.; Zhang, D.; Yang, M. Prepackaged Foods Healthiness Ranking Index: Developing Front-of-Pack Labeling to Facilitate the Assessment and Validation of Healthiness Rankings of Prepackaged Foods in China. BMC Med. 2024, 22, 402. [Google Scholar] [CrossRef]
- Zickafoose, A.; Lu, P.; Baker, M. Forecasting Food Innovations with a Delphi Study. Foods 2022, 11, 3723. [Google Scholar] [CrossRef] [PubMed]
- Gallegos, D.; Booth, S.; Pollard, C.M.; Chilton, M.; Kleve, S. Food Security Definition, Measures and Advocacy Priorities in High-Income Countries: A Delphi Consensus Study. Public Health Nutr. 2023, 26, 1986–1996. [Google Scholar] [CrossRef]
- Lamm, K.W.; Randall, N.L.; Fluharty, F.L. Critical Issues Facing the Animal and Food Industry: A Delphi Analysis. Transl. Anim. Sci. 2021, 5, txaa213. [Google Scholar] [CrossRef] [PubMed]
- Lamm, K.W.; Randall, N.L.; Diez-Gonzalez, F. Critical Food Safety Issues Facing the Food Industry: A Delphi Analysis. J. Food Prot. 2021, 84, 680–687. [Google Scholar] [CrossRef]
- Aschemann-Witzel, J.; Perez-Cueto, F.J.A.; Niedzwiedzka, B.; Verbeke, W.; Bech-Larsen, T. Transferability of Private Food Marketing Success Factors to Public Food and Health Policy: An Expert Delphi Survey. Food Policy 2012, 37, 650–660. [Google Scholar] [CrossRef]
- Lupetti, C.; Guadagnini, G.; Casselli, R.; Cardoso, F.; Lupetti, C. The Brazilian and the Plant-Based-2023/2024. Available online: https://gfi.org.br/en/resources/o-consumidor-brasileiro-e-o-mercado-plant-based-2023-2024/ (accessed on 29 August 2025).
- World Health Organization. WHO Global Report on Sodium Intake Reduction; World Health Organization: Geneva, Switzerland, 2023. [Google Scholar]
- World Health Organization. Carbohydrate Intake for Adults and Children WHO Guideline; World Health Organization: Geneve, Switzerland, 2023. [Google Scholar]
- World Health Organization. Report of a Series of Expert Meetings on Developing WHO Global Sodium Benchmarks; World Health Organization: Geneve, Switzerland, 2020. [Google Scholar]
- World Health Organization. Saturated Fatty Acid and Trans-Fatty Acid Intake for Adults and Children: WHO Guideline; World Health Organization: Geneve, Switzerland, 2023. [Google Scholar]
- World Health Organization. Total Fat Intake for the Prevention of Unhealthy Weight Gain in Adults and Children: WHO Guideline; World Health Organization: Geneve, Switzerland, 2023. [Google Scholar]
- World Health Organization. Nutrition Labelling: Policy Brief; World Health Organization: Geneve, Switzerland, 2022. [Google Scholar]
- World Health Organization. Global Action Plan for the Prevention and Control of Noncommunicable Diseases 2013–2020; World Health Organization: Geneve, Switzerland, 2013; ISBN 9789241506236. [Google Scholar]
- World Health Organization. REPLACE Trans Fat: An Action Package to Eliminate Industrially Produced Trans-Fatty Acids; World Health Organization: Geneve, Switzerland, 2021. [Google Scholar]
- World Health Organization. Sodium Intake for Adults and Children; World Health Organization: Geneve, Switzerland, 2012. [Google Scholar]
- World Health Organization. International Agency for Research of Cancer Monographs Evaluate Consumption of Red Meat and Processed Meat; World Health Organization: Lyon, France, 2015. [Google Scholar]
- Americam Diet Association. Position of the American Dietetic Association: Vegetarian Diets. J. Am. Diet. Assoc. 2009, 109, 1266–1282. [Google Scholar] [CrossRef]
- Institute of Medicine. Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids (Macronutrients); National Academies Press: Washington, DC, USA, 2005; ISBN 030908525X. [Google Scholar]
- Institute of Medicine. Dietary Reference Intakes: The Essential Guide to Nutrient Requirements; National Academies Press: Washington, DC, USA, 2006; ISBN 978-0-309-15742-1. [Google Scholar]
- Ministry of Health; National Health Surveillance Agency. Resolução Da Diretoria Colegiada No 429-Dispõe Sobre a Rotulagem Dos Alimentos; Ministry of Health: Brasilia, Brazil; National Health Surveillance Agency: Brasilia, Brazil, 2020.
- Ministry of Health; National Health Surveillance Agency. Instrução Normativa No 75. Estabelece Os Requisitos Técnicos Para Declaração Rotulagem Nutricional Nos Alimentos; Ministry of Health: Brasilia, Brazil; National Health Surveillance Agency: Brasilia, Brazil, 2020.
- Ministry of Health; National Health Surveillance Agency. Resolução Da Diretoria Colegiada-RDC N° 740. Autoriza o Uso de Aditivos e Coadjuvantes de Tecnologia Em Diversas categorias de Alimentos; Ministry of Health: Brasilia, Brazil; National Health Surveillance Agency: Brasilia, Brazil, 2022.
- Pan American Health Organization. Pan American Health Organization Nutrient Profile Model; Pan American Health Organization: Washington, DC, USA, 2016. [Google Scholar]
- Melina, V.; Craig, W.; Levin, S. Position of the Academy of Nutrition and Dietetics: Vegetarian Diets. J. Acad. Nutr. Diet. 2016, 116, 1970–1980. [Google Scholar] [CrossRef]
- Silva, A.M.M.; Noguera, N.H.; Carmo, A.S.; Malagodi, S.S.; Netto, F.M.; Gigante, M.L.; Sato, A.C.K. Study of National Plant Proteins with Potential for Application in Plant Based Products; The Good Food Institute Brazil: São Paulo, Brazil, 2024. [Google Scholar]
- Leitão, A.E.; Esteves, G.P.; Mazzolani, B.C.; Smaira, F.I.; Santini, M.H.; Santo André, H.C.; Gualano, B.; Roschel, H. Protein and Amino Acid Adequacy and Food Consumption by Processing Level in Vegans in Brazil. JAMA Netw. Open 2024, 7, e2418226. [Google Scholar] [CrossRef] [PubMed]
- Herreman, L.; Nommensen, P.; Pennings, B.; Laus, M.C. Comprehensive Overview of the Quality of Plant- And Animal-Sourced Proteins Based on the Digestible Indispensable Amino Acid Score. Food Sci. Nutr. 2020, 8, 5379–5391. [Google Scholar] [CrossRef]
- Van Den Berg, L.A.; Mes, J.J.; Mensink, M.; Wanders, A.J. Protein Quality of Soy and the Effect of Processing: A Quantitative Review. Front. Nutr. 2022, 9, 100475. [Google Scholar] [CrossRef]
- Food and Agricultural Organization-Nations United. Experts’ Recommendations on Fats and Oils in Human Nutrition; Food and Agricultural Organization-Nations United: Roma, Italy, 1994. [Google Scholar]
- Schuchardt, J.P.; Beinhorn, P.; Hu, X.F.; Chan, H.M.; Roke, K.; Bernasconi, A.; Hahn, A.; Sala-Vila, A.; Stark, K.D.; Harris, W.S. Omega-3 World Map: 2024 Update. Prog. Lipid Res. 2024, 95, 101286. [Google Scholar] [CrossRef]
- Ministério da Saúde; Agência Nacional de Vigilância Sanitária. Resolução Da Diretoria Colegiada-RDC N°514. Altera a RDC No 332, Que Define Os Requisitos Para Uso de Gorduras Trans Industriais Em Alimentos; Ministry of Health: Brasilia, Brazil; National Health Surveillance Agency: Brasilia, Brazil, 2021.
- Barroso, N.G.; Okuro, P.K.; Ribeiro, A.P.B.; Cunha, R.L. Tailoring Properties of Mixed-Component Oleogels: Wax and Monoglyceride Interactions towards Flaxseed Oil Structuring. Gels 2020, 6, 5. [Google Scholar] [CrossRef]
- Yang, F.; Zhang, B.; Chen, B.; Yang, N.; Wang, R.; Zhang, X.; Li, G. A Lipidomic Approach for Profiling and Distinguishing Seed Oils of Hibiscus manihot L., Flaxseed, and Oil Sunflower. JAOCS J. Am. Oil Chem. Soc. 2022, 99, 203–213. [Google Scholar] [CrossRef]
- MacIntosh, S.C.; Shaw, M.; Connelly, M.; Yao, Z.J. Food and Feed Safety of NS-B5ØØ27-4 Omega-3 Canola (Brassica Napus): A New Source of Long-Chain Omega-3 Fatty Acids. Front Nutr. 2021, 8, 716659. [Google Scholar] [CrossRef]
- Tokuşoglu, Ö.; üUnal, M.K. Biomass Nutrient Profiles of Three Microalgae: Spirulina Platensis, Chlorella Vulgaris, and Isochrisis Galbana. Food Sci. 2003, 68, 1144–1148. [Google Scholar] [CrossRef]
- Kamani, M.H.; Neji, C.; Fitzsimons, S.M.; Fenelon, M.A.; Murphy, E.G. Unlocking the Nutritional and Functional Potential of Legume Waste to Produce Protein Ingredients. Crit. Rev. Food Sci. Nutr. 2024, 64, 7311–7329. [Google Scholar] [CrossRef] [PubMed]
- Carbas, B.; Machado, N.; Pathania, S.; Brites, C.; Rosa, E.A.S.; Barros, A.I.R.N.A. Potential of Legumes: Nutritional Value, Bioactive Properties, Innovative Food Products, and Application of Eco-Friendly Tools for Their Assessment. Food Rev. Int. 2023, 39, 160–188. [Google Scholar] [CrossRef]
- Carbonaro, M.; Maselli, P.; Nucara, A. Structural Aspects of Legume Proteins and Nutraceutical Properties. Food Res. Int. 2015, 76, 19–30. [Google Scholar] [CrossRef]
- Ministry of Agriculture, Livestock and Supply. Aprova o Regulamento Técnico de Identidade e Qualidade Do Hambúrguer; Ministry of Agriculture, Livestock and Supply: Brasilia, Brazil, 2023.
- van der Maaden, T.; van der Steen, J.T.; de Vet, H.C.W.; Achterberg, W.P.; Boersma, F.; Schols, J.M.G.A.; van Berkel, J.F.J.M.; Mehr, D.R.; Arcand, M.; Hoepelman, A.I.M.; et al. Development of a Practice Guideline for Optimal Symptom Relief for Patients with Pneumonia and Dementia in Nursing Homes Using a Delphi Study. Int. J. Geriatr. Psychiatry 2015, 30, 487–496. [Google Scholar] [CrossRef]
- Hung, Y.; Hieke, S.; Grunert, K.G.; Verbeke, W. Setting Policy Priorities for Front-of-Pack Health Claims and Symbols in the European Union: Expert Consensus Built by Using a Delphi Method. Nutrients 2019, 11, 403. [Google Scholar] [CrossRef]
- United Kingdom Department of Health. Nutrient Profiling Technical Guidance; United Kingdom Department of Health: London, UK, 2011.
- Health Canada. Front-of-Package Nutrition Symbol Labelling Guide for Industry Front-of-Package Nutrition Symbol Labelling Guide for Industry (Version 2) | 2; Health Canada: Ottawa, ON, Canada, 2023. [Google Scholar]
- Kronsteiner-Gicevic, S.; Bogl, L.H.; Wakolbinger, M.; Müller, S.; Dietrich, J.; De Keyzer, W.; Bullón-Vela, V.; Selinger, E.; Keller, V.; Martínez Tabar, A.; et al. Development of the VEGANScreener, a Tool for a Quick Diet Quality Assessment among Vegans in Europe. Nutrients 2024, 16, 1344. [Google Scholar] [CrossRef] [PubMed]
- Drewnowski, A.; Bruins, M.J.; Besselink, J.J.F. Comparing Nutrient Profiles of Meat and Fish with Plant-Based Alternatives: Analysis of Nutrients, Ingredients, and Fortification Patterns. Nutrients 2024, 16, 2725. [Google Scholar] [CrossRef] [PubMed]
- Williams, E.; Vardavoulia, A.; Lally, P.; Gardner, B. Experiences of Initiating and Maintaining a Vegan Diet among Young Adults: A Qualitative Study. Appetite 2023, 180, 106357. [Google Scholar] [CrossRef]
- Egea, M.B.; de Oliveira Filho, J.G.; Campos, S.B.; Lemes, A.C. The Potential of Baru (Dipteryx alata Vog.) and Its Fractions for the Alternative Protein Market. Front. Sustain. Food Syst. 2023, 7, 1148291. [Google Scholar] [CrossRef]
- Fernandes, D.C.; dos Santos, G.F.; Borges, M.O.; Dias, T.; Naves, M.M.V. Blend of Baru (Dipteryx alata Vog.) By-Products as Nutritive and Healthy Food Ingredients: Chemical Composition, Functional Properties and Application in Plant-Based Burger. Plant Foods Hum. Nutr. 2024, 79, 578–585. [Google Scholar] [CrossRef]
- Mercês, Z.d.C.d.; Salvadori, N.M.; Evangelista, S.M.; Cochlar, T.B.; Rios, A.d.O.; Oliveira, V.R.d. Hybrid and Plant-Based Burgers: Trends, Challenges, and Physicochemical and Sensory Qualities. Foods 2024, 13, 3855. [Google Scholar] [CrossRef]
- Ogunwolu, S.O.; Henshaw, F.O.; Mock, H.P.; Santros, A.; Awonorin, S.O. Functional Properties of Protein Concentrates and Isolates Produced from Cashew (Anacardium occidentale L.). Nut. Food Chem. 2009, 115, 852–858. [Google Scholar] [CrossRef]
- da Cruz, K.S.; da Silva, M.A.; de Freitas, O.D.; Neves, V.A. Partial Characterization of Proteins from Baru (Dipteryx alata Vog) Seeds. J. Sci. Food Agric. 2011, 91, 2006–2012. [Google Scholar] [CrossRef] [PubMed]
- Michel, F.; Hartmann, C.; Siegrist, M. Consumers’ Associations, Perceptions and Acceptance of Meat and Plant-Based Meat Alternatives. Food Qual. Prefer. 2021, 87, 104063. [Google Scholar] [CrossRef]
- Starowicz, M.; Kubara Poznar, K.; Zieliński, H. What Are the Main Sensory Attributes That Determine the Acceptance of Meat Alternatives? Curr. Opin. Food Sci. 2022, 48, 100924. [Google Scholar] [CrossRef]
- Clark, L.F.; Bogdan, A.M. The Role of Plant-Based Foods in Canadian Diets: A Survey Examining Food Choices, Motivations and Dietary Identity. J. Food Prod. Mark. 2019, 25, 355–377. [Google Scholar] [CrossRef]
- Ministry of Agriculture and Livestock; Secretariat of Agricultural Defense. PORTARIA SDA/MAPA No 831; Ministry of Agriculture and Livestock; Secretariat of Agricultural Defense: Brasilia, Brazil, 2023.
- Food and Drug Administration. Labeling of Plant-Based Alternatives to Animal-Derived Foods: Draft Guidance for Industry Draft Guidance; Food and Drug Administration: Rockville, MD, USA, 2025. [Google Scholar]
- Jang, J.; Lee, D.W. Advancements in Plant Based Meat Analogs Enhancing Sensory and Nutritional Attributes. NPJ Sci. Food 2024, 8, 50. [Google Scholar] [CrossRef] [PubMed]
| Nutritional Aspects | Questions |
|---|---|
| Proteins | 1. What is your assessment of the protein adequacy in plant-based meat analogues, both in quantitative and qualitative terms? (Your response may address aspects such as total protein content, amino acid composition, protein digestibility, and other relevant indicators of protein quality.) |
| Fats | 1. What is your assessment of the adequacy of fat content in plant-based meat analogues, considering both qualitative and quantitative aspects? (Your response may address total fat content, levels of saturated, monounsaturated, polyunsaturated, and trans fats, as well as the overall fatty acid composition.) |
| Carbohydrates | 1. What is your assessment of the adequacy of carbohydrate content in plant-based meat analogues, both in qualitative and quantitative terms? (Your response may address aspects such as total carbohydrates, soluble and insoluble dietary fiber, and other relevant carbohydrate-related parameters.) |
| Sodium | What are your thoughts on the sodium content in plant-based meat analogues? |
| Vitamins and Minerals | 1. How would you evaluate the adequacy of vitamins and minerals content in plant-based meat products, considering both qualitative and quantitative aspects? (Your response may encompass B vitamins as well as fat-soluble vitamins (A, D, E, and K) and for minerals (Iron and Zinc) and may address factors such as bioaccessibility). |
| Sensory Aspects | Questions |
| 1. Have you ever tasted vegetable products that are similar to meat products sold in Brazil? | |
| How would you evaluate the sensory quality of plant-based meat analogue products currently available on the Brazilian market? | |
| 2. In your opinion, what are the primary sensory limitations or challenges observed in plant-based meat analogue products currently available on the market? | |
| Technological Aspects | Questions |
| 1. Please list any guidelines you consider important regarding the use of ingredients in the formulation of plant-based products. In your response, you may address aspects related to the selection and application of protein sources, lipid components, dietary fibers, and other functional or nutritional ingredients that are critical to product development. | |
| 2. In your opinion, should any specific ingredients or additives be restricted or prohibited in the formulation of plant-based meat products? If so, please specify which ones and provide the rationale for their limitation or exclusion. | |
| Innovative Aspects | Questions |
| 1. In your opinion, what potential innovations could be applied in the development of plant-based meat products to address existing sensory and nutritional limitations? | |
| 2. In the development of new plant-based meat products, which characteristics do you believe should be prioritized to enhance product quality and consumer acceptance? | |
| Regulatory Aspects | Questions |
| Plant-based products definition provided by Ministry of Agriculture and Livestock, the regulatory authority for animal and plant-based products in Brazil [1]: “A food product composed of raw materials of plant origin that bears a relationship to a corresponding animal-based product”. 1. Do you agree with the definition mentioned? Justify your answer. | |
| 2. What expectations do you have regarding a product that includes the term ‘analogue’ in its sales name? |
| Characteristics | Round 1 (n = 10) | Round 2 (n = 20) | Round 3 (n = 34) |
|---|---|---|---|
| n (%) | n (%) | n (%) | |
| Stakeholder | |||
| 1. Researchers from institutes and/or universities | 4 (40%) | 10 (50%) | 20 (58%) |
| 2. Representatives from scientific societies, non-profit organizations, civil society, and government agencies | 3 (30%) | 5 (25%) | 8 (24%) |
| 3. Representatives from associations linked to the food industry | 3 (30%) | 5 (25%) | 6 (18%) |
| Education background | |||
| Graduate | 0 (0%) | 2 (10%) | 6 (17%) |
| Postgraduate specialization | 1 (10%) | 2 (10%) | 3 (9%) |
| Master’s degree | 2 (20%) | 4 (20%) | 6 (17%) |
| PhD (doctorate) | 5 (50%) | 8 (40%) | 9 (26%) |
| Post-doctoral | 2 (20%) | 4 (20%) | 10 (29%) |
| Professional experience time | |||
| 1 to 5 years | 1 (10%) | 3 (15%) | 3 (9%) |
| 5 to 10 years | 1 (10%) | 3 (15%) | 5 (15%) |
| 10 to 15 years | 1 (10%) | 4 (20%) | 4 (11%) |
| 15 to 20 years | 2 (20%) | 2 (10%) | 5 (15%) |
| More than 20 years | 5 (50%) | 8 (40%) | 17 (50%) |
| Knowledge rating in a plant-based topic (1 to 10) | |||
| Level 9 | 4 (40%) | 7 (35%) | 10 (30%) |
| Level 8 | 3 (30%) | 6 (30%) | 9 (26%) |
| Level 7 | 2 (20%) | 4 (20%) | 8 (23%) |
| Level 6 | 1 (10%) | 3 (15%) | 5 (15%) |
| Level 5 | 0 (0%) | 0 (0%) | 2 (6%) |
| Stated level of interest in the topic | |||
| I have a strong interest in the topic, given its direct relevance to my professional practice. | 7 (70%) | 14 (70%) | 24 (70%) |
| I am interested in the topic, as it is indirectly related to my professional practice. | 3 (30%) | 6 (30%) | 10 (30%) |
| Nutritional Aspects | Questions | Responses |
|---|---|---|
| Proteins | 1. What is your assessment of the protein adequacy in plant-based meat analogues, both in quantitative and qualitative terms? | 1. “Ensuring adequate protein content in plant-based analogues is generally more straightforward than achieving an optimal amino acid profile; nonetheless, it remains feasible. However, it is important to question whether consumers prioritize such nutritional characteristics or are primarily guided by the sensory attributes of these products. It should be emphasized that plant-based analogues are intended to complement a broader dietary pattern and are unlikely to serve as the sole source of protein for consumers. Although protein digestibility is a critical nutritional parameter, it remains largely inaccessible as information to the general public—not only in the context of meat analogues but also for food products more broadly.” 2. “I do not consider protein standardization to be necessary. Plant-based meat analogues are expected to have nutritional profiles that differ from those of conventional meat products, just as various meat products themselves can exhibit considerable nutritional variation. Therefore, aligning the protein composition of analogues to that of meat is not essential.” 3. “Establishing minimum parameters for protein quantity and amino acid quality, differentiated by type of analogue, could represent a viable strategy to address this gap. Such an approach is already mandated, for instance, in the context of nutritional claims for protein content according to Brazilian nutrition labeling law and is also employed as a compositional requirement for certain foods intended for special dietary purposes. However, the inclusion of digestibility as a criterion for protein quality remains overly complex, as evidenced by ANVISA’s (Brazilian Health Surveillance Agency) regulatory experience with foods targeted at athletes.” 4. “For meat analogue products, it is essential that their formulation is based on protein-rich matrices such as soy, wheat, and other legumes. From a quantitative perspective, while increased protein content may add market value, I do not believe that protein quantity and bioavailability parameters should mirror those used for animal-based products. In the context of a plant-based diet, protein intake is understood as a component of the overall dietary pattern rather than being concentrated in specific key items, as is typically the case with meat and eggs in omnivorous diets. Therefore, I advocate for a unified standard for plant-based meat analogues—one that ensures the use of vegetable protein sources while establishing a minimum protein content based on total protein per serving and the profile of essential amino acids, such as leucine. A useful reference for this standard could be the amino acid adequacy table outlined in Brazilian nutrition labeling law, which defines aminograms for protein-rich foods and could be adapted to the specific characteristics of plant-based proteins.” |
| Fats | 1. What is your assessment of the adequacy of fat content in plant-based meat analogues, considering both qualitative and quantitative aspects? | 1. “In general, plant-based analogues of protein-rich animal products tend to exhibit a more favorable lipid profile, primarily due to the use of vegetable oils and fats, which are typically lower in saturated fatty acids and higher in unsaturated fatty acids. As a result, the fat composition of these products is not typically considered a nutritional concern. It is important to note that consumers have access to information on total fat, saturated fat, and trans fat content, as these nutrients are mandatorily declared on nutrition labels in accordance with Brazilian nutrition labeling law. The amounts of monounsaturated and polyunsaturated fats may be declared voluntarily. Nutritional claims related to fat content are also permitted on a voluntary basis, provided that the product complies with the compositional and labeling requirements established by Brazilian nutrition labeling law. Notably, the use of partially hydrogenated oils—the primary source of industrial trans fats—is prohibited in food formulations, and the maximum allowable trans fatty acid content in refined vegetable oils is limited to 2%, as established by Brazilian food law. Therefore, the presence of trans fats in plant-based foods available in Brazil is not considered a significant public health concern. However, the authorization and regulation of plant-based analogues that replicate high-fat animal products require further consideration, particularly as they may fall under additional or separate regulatory frameworks.” 2. “In my assessment, fats represent a greater nutritional and technological challenge than proteins in the development of plant-based meat analogues. Unlike animal-derived meats, which naturally contain saturated fats that contribute to cohesion, texture, and lubricity, these functional lipids are absent in the protein matrices typically used in plant-based formulations. As a result, fats must be added to replicate the structural and sensory roles of animal fat, with palm fat often being the most accessible and technologically effective option. Despite its functional advantages, the widespread use of palm fat in ultra-processed plant-based products raises nutritional concerns due to its high saturated fat content. It is therefore essential to identify strategies that ensure the inclusion of fats—crucial from a culinary standpoint—without compromising the nutritional quality of the final product. In this context, the use of hydrocolloids presents a promising alternative for enhancing texture and matrix cohesion, potentially reducing the reliance on palm fat in an excessive manner.” 3. “Vegetable fats are generally considered nutritionally more favorable, and their incorporation into the formulation of plant-based analogues does not pose significant challenges from a nutritional standpoint.” |
| Carbohydrates | 1. What is your assessment of the adequacy of carbohydrate content in plant-based meat analogues, both in qualitative and quantitative terms? | 1. “I do not consider carbohydrate equivalence between plant-based meat analogues and conventional meat products to be necessary. Plant-based analogues may possess distinct nutritional profiles, just as various meat products differ among themselves. Therefore, aligning carbohydrate content is not essential.” 2. “In general, the technological formulation of plant-based analogues often relies on the combination of carbohydrates and vegetable fats, which contributes to the structural and sensory properties of these products. As a result, the presence of carbohydrates in such formulations is typically higher.” 3. “Plant-based analogues of protein-rich animal foods tend to have a higher carbohydrate content, particularly in terms of dietary fiber. This is attributed both to the intrinsic composition of the plant-derived ingredients and their functional roles in product development. Consumers are provided with information on carbohydrate content—including total sugars and added sugars—on the nutrition label, in accordance with Brazilian nutrition labeling law. Furthermore, the voluntary use of nutrition claims related to sugars is permitted, provided the product complies with the compositional and labeling criteria outlined in Brazilian nutrition labeling law. Nevertheless, it remains important to consider whether analogues of animal-based foods containing added sugars will be authorized under current regulatory frameworks, given that such products are already subject to specific legislation.” |
| Sodium | 1. What are your thoughts on the sodium content in plant-based meat analogues? | 1. “It is hoped that these products contain low levels per 100 g of critical nutrients and, consequently, do not receive a front-of-package warning label on their nutritional information”. 2. “I do not consider sodium adjustment to be necessary. Plant-based products designed to mimic meat may naturally present different nutritional profiles from their animal-based counterparts, just as nutritional variability exists among different types of meat products themselves. Consequently, standardizing sodium levels across these categories is unwarranted. Moreover, according to Brazilian nutrition labeling law, products containing sodium levels above the established threshold are required to display front-of-package warning labels. Thus, the implementation of a specific sodium standard for plant-based analogues is unnecessary”. 3. “Considering that the sodium content of foods is disclosed in the nutritional information panel, and that products with high sodium levels are required to display front-of-pack nutritional labeling in accordance with Brazilian nutrition labeling law, there appears to be no specific concern regarding the sodium content of these products in comparison to other processed foods”. 4. “In the context of the high sodium intake associated with the Western dietary pattern prevalent in Brazil, the elevated sodium content of plant-based analogues may further compromise the overall quality of a plant-based diet. Therefore, alternative formulation strategies are needed to ensure these products retain desirable flavor characteristics without excessive sodium content”. |
| Vitamins and Minerals | 1. How would you evaluate the adequacy of vitamin and minerals content in plant-based meat products, considering both qualitative and quantitative aspects? | 1. “Concerns regarding the nutritional implications of replacing animal-derived foods with analogous plant-based products suggest the potential necessity for mandatory enrichment of these products with selected micronutrients. This would involve establishing minimum and maximum allowable limits, as well as specifying authorized compounds, tailored to each product category. Alternatively, specific guidelines could be developed to regulate the voluntary enrichment of these products”. 2. “It is well established that, quantitatively, plant-based meat analogs contain lower levels of certain vitamins compared to their animal-based counterparts. Therefore, during the formulation of plant-based analogs, supplementation with bioactive forms of these vitamins is essential, in amounts equivalent to those found in the products they aim to replicate”. 3. “Vitamin supplementation in analogous products can help ensure nutritional equivalence; however, it is imperative to assess the stability of these added vitamins throughout the product’s shelf life and consumption. |
| Sensory Aspects | ||
| 1. In your opinion, what are the primary sensory limitations or challenges observed in plant-based meat analogue products currently available on the market? | 1. “It is difficult to generalize, as there is considerable variation among products currently available on the market. However, plant-based meat analogues often exhibit a saltier flavor profile. Additionally, some products lack the melt-in-the-mouth sensation typically associated with animal fat, which may affect overall sensory experience”. 2. “Residual flavors from protein concentrates and other ingredients, the use of overly artificial-tasting flavor maskers, and challenges in replicating the texture of solid products represent significant sensory limitations in the development of plant-based meat analogues”. 3. “Currently, there are no additives or processing aids authorized for use in foods classified as plant-based analogue products by Brazil’s Ministry of Agriculture, Livestock and Food Supply (MAPA). This regulatory gap poses a significant barrier to product formulation, given the essential role of various additives in enhancing the sensory attributes of processed foods. Consequently, one of the primary limitations identified in the proposed regulatory framework is the lack of approval for additives and adjuvants that are critical for achieving sensory characteristics comparable to those of animal-based products.” | |
| Technological Aspects | ||
| 1. Please list any guidelines you consider important regarding the use of ingredients in the formulation of plant-based products. | 1.” Technological innovation in plant-based products should not be unduly restricted, as this is an emerging field in which premature regulatory limitations may hinder the development of novel technologies and ingredients. Such innovations have the potential to significantly enhance the sensory attributes, nutritional quality, and environmental sustainability of plant-based meat analogues.” 2. “The use of food additives and processing aids is governed by established principles in Brazilian health legislation. These principles stipulate that such substances must be proven safe for human consumption under their intended conditions of use, employed in the minimum effective quantities to achieve the desired technological function, and must not mislead consumers. Additionally, only those substances included in the positive lists defined by Brazilian health legislation—organized by food category and authorized by ANVISA—are permitted for use. At present, there are no additives specifically authorized for the food category encompassing plant-based analogue products. It is also important to consider that, depending on the nature of label claims associated with these products, the use of certain types or classes of additives may be deemed inappropriate”. | |
| 2. In your opinion, should any specific ingredients or additives be restricted or prohibited in the formulation of plant-based meat products? | 1. “Ingredients that pose safety risks or have the potential to mislead or confuse consumers—particularly in relation to the claims made on product labels—should not be permitted for use in food products”. 2. “I do not believe that any additive currently permitted for use in animal-based products within the same category should be prohibited in plant-based analogues”. | |
| Innovative Aspects | ||
| 1. In your opinion, what potential innovations could be applied in the development of plant-based meat products to address existing sensory and nutritional limitations? | 1. “Exploration of underutilized plant-based ingredients and emerging technologies, such as precision fermentation, is essential for advancing innovation in the development of plant-based products”. 2. “The regulatory framework can be notably restrictive regarding formulation options for plant-based analogue products. Such limitations are likely to hinder the development of products with sensory and nutritional characteristics comparable to those of animal-based counterparts. Consequently, a comprehensive review of the proposed regulation is warranted”. | |
| 2. In the development of new plant-based meat products, which characteristics do you believe should be prioritized to enhance product quality and consumer acceptance? | 1. “Sustainability, along with improvements in texture and flavor, are critical factors for ensuring consumer acceptance and the successful integration of these products into the diet”. 2. “Sensory and nutritional characteristics”. 3. “Key considerations include product safety, nutritional quality, and regulatory compliance with labeling standards.” 4. “Striking a balance between nutritional quality and sensory attributes should be a central priority in the development of plant-based analogue products”. | |
| Regulatory Aspects | ||
| Plant-based products definition: “A food product composed of raw materials of plant origin that bears a relationship to a corresponding animal-based product regulated by the Ministry of Agriculture and Livestock”. 1. Do you agree with the definition mentioned? Justify your answer. | 1. “No. The proposal requires substantial improvement to ensure that the regulation of this emerging product category is both effective and proportionate in addressing the current challenges of the plant-based food market. This includes careful consideration of the compositional and presentation characteristics of these products, the potential for future innovations, existing regulations on food identity and quality standards, as well as relevant international regulatory frameworks. The main issues associated with this definition are:
2. “I agree with the proposed definition, as it provides a clear and structured framework for categorizing plant-based analogue products while aligning with current scientific and regulatory standards”. | |
| 2. What expectations do you have regarding a product that includes the term ‘analogue’ in its sales name? | 1. “The term ‘vegetable analog of’ may not be easily understood by consumers, as it is not commonly used in the everyday vocabulary of Brazilians. Therefore, I suggest adopting a more consumer-friendly and transparent nomenclature, such as that already in use—for example, ‘burger made with (corresponding vegetable)”. 2. “That it exhibits sensory characteristics and functional performance equivalent to those of the corresponding animal-based product”. 3. “The term ‘analogous’ implies a product that is similar to another. Although this designation is qualified by the term ‘vegetable,’ consumers may interpret it as indicating that the product shares similar sensory and/or nutritional characteristics, or that it can be used in the same manner and for the same culinary purposes as its animal-based counterpart. However, based on the proposed compositional requirements, there is neither a guarantee nor, in some cases, a possibility of achieving nutritional equivalence or similarity in usage and preparation”. | |
| Recommendations | AC Relevance | AC Adequacy |
|---|---|---|
| Nutritional Aspects | ||
| 1. The minimum protein content in plant-based meat analogue products should be equivalent to that of conventional meat products, according to Brazilian Identity and Quality Standard. For example: the minimum protein content in a burger should be set at 15% [81]. | 80% | 80% |
| 2. The minimum protein content in plant-based meat analogue products should be 20% of Daily Value (%DV) equivalent to 10 g per serving size [62,63]. | 80% | 85% |
| 3. The selection of raw materials for the development of plant-based meat analogue products must ensure a composition of essential amino acids that meets the nutritional requirements of adults, in accordance with the standards set forth in Brazilian Food Labeling Law [60]. An amino acid composition is considered adequate when no limiting amino acids are present. The required amino acid profile per gram of protein is as follows: histidine–15 mg, isoleucine–30 mg, leucine–59 mg, lysine–45 mg, methionine + cysteine–22 mg, phenylalanine + tyrosine–38 mg, threonine–23 mg, tryptophan–6 mg, and valine–39 mg. | 65% | 60% |
| 4. The selection of plant-based raw materials used to supply lipids in the formulation of plant-based meat analogue products should predominantly provide monounsaturated and polyunsaturated fatty acids. | 85% | 85% |
| 5. The selection of plant-based raw materials intended to supply lipids in the formulation of plant-based meat analogue products should ensure the inclusion of omega-3 fatty acids, particularly at levels sufficient to qualify as a source of alpha-linolenic acid (ALA), defined as a minimum of 300 mg per serving size [62,63]. | 65% | 65% |
| 6. The selection of plant-based raw materials used to supply lipids in the formulation of plant-based meat analogue products should ensure the provision of polyunsaturated fatty acids, with an omega-6: omega-3 ratio not exceeding 10:1 [72]. | 70% | 70% |
| 7. The total fat content in plant-based meat analogue products should not exceed 30% DV for total energy value [52]. | 90% | 90% |
| 8. The saturated fat content in plant-based meat analogue products should not exceed 10% DV for total energy value [52]. | 90% | 90% |
| 9. The saturated fat content in plant-based meat analogue products should remain below the threshold established for the application of front-of-package nutritional warning labels for solid and semi-solid foods, as defined by Brazilian Food Labeling Law (equivalent to 6 g per 100 g) [62,63]. | 90% | 90% |
| 10. Plant-based meat analogue products must contain dietary fiber levels that meet at least the minimum requirement to be classified as a source of fiber, as defined by Brazilian Food Labeling Law (equivalent to 10% DV, or 2.5 g of fiber per serving size) [62,63]. | 80% | 85% |
| 11. Plant-based meat analogue products must contain dietary fiber levels that meet at least the minimum requirement to be classified as a high content of fiber, as defined by Brazilian Food Labeling Law (equivalent to 20% DV, or 5.0 g of fiber per serving size) [62,63]. | 45% * | 50% * |
| 12. The sodium content in plant-based meat analogue products must be below the threshold established for front-of-package nutritional warning labeling for solid and semi-solid foods, in accordance with Brazilian Food Labeling Law, which sets the limit at 600 mg per 100 g [62,63]. | 90% | 85% |
| 13. The sodium content in plant-based meat analogue products should be less than 1 mg per kilocalorie, in accordance with PAHO/WHO nutritional guidelines [65]. | 65% | 60% |
| 14. Plant-based meat analogue products should be fortified with minerals (iron and zinc) and B vitamins (B6 and B12) at levels sufficient to qualify as sources of these nutrients, in accordance with Brazilian Food Labeling Law [62,63], which defines this threshold as 15% of DV per serving size. | 75% | 80% |
| 15. Plant-based meat analogue products should be fortified with minerals (iron and zinc) and B vitamins (B6 and B12) at levels sufficient to qualify as sources of these nutrients, in accordance with Brazilian Food Labeling Law [62,63], which defines this threshold as 30% of DV per serving size. | 65% | 65% |
| Sensory | ||
| 16. When the objective is to replicate meat products, the sensory attributes of plant-based analogues should be prioritized over nutritional aspects. | 65% | 65% |
| 17. When the objective is to replicate meat products, the nutritional quality of plant-based analogues should take precedence over sensory. | 45% * | 50% * |
| 18. When the objective is to replicate meat products, sensory and nutritional attributes should be given equal importance to effectively achieve product equivalence. | 85% | 85% |
| Technology | ||
| 19. Plant-based meat analogue products should be recognized as a distinct food category within legislation governing the use of food additives and processing aids. | 85% | 85% |
| Regulatory | ||
| 20. A food product formulated from plant-based raw materials that exhibits sensory characteristics comparable to its corresponding animal-derived product regulated by the Ministry of Agriculture and Livestock. | 92% | 92% |
| 21. A food product formulated from plant-based raw materials that demonstrates nutritional equivalence to the corresponding animal-derived product regulated by the Ministry of Agriculture and Livestock. | 75% | 75% |
| 22. A food product formulated from plant-based raw materials that corresponds to the consumption format of the analogous animal-derived product regulated by the Ministry of Agriculture and Livestock. | 97% | 97% |
| 23. Mandatory inclusion of the statement: “This product may have a different nutritional value than the corresponding animal-derived product” on plant-based analogue products. | 85% | 85% |
| 24. Mandatory inclusion of the statement: “This product does not replace its animal-based counterpart in nutritional or functional terms” on plant-based analogue products. | 65% | 65% |
| Recommendations | Relevance (Mean ± SD) | Feasibility (Mean ± SD) | Total Agreement (Mean ± SD) | Consensus (%) |
|---|---|---|---|---|
| Nutritional Aspects | ||||
| 1. The minimum protein content in plant-based meat analogue products should be equivalent to that of conventional meat products, according to Brazilian Identity and Quality Standard. For example: the minimum protein content in a burger should be set at 15% [73]. | 8.06 ± 1.92 | 7.50 ± 2.48. | 7.78 ± 1.81 | 82% all stakeholders |
| 2. The minimum protein content in plant-based meat analogue products should be 20% of Daily Value (%DV) equivalent to 10 g per serving size [60]. | 7.00 ± 2.62 | 6.93 ± 1.41 | 6.96 ± 2.31 | 67% all stakeholders |
| 3. The selection of raw materials for the development of plant-based meat analogue products must ensure a composition of essential amino acids that meets the nutritional requirements of adults, in accordance with the standards set forth in Brazilian Food Labeling Law [60]. An amino acid composition is considered adequate when no limiting amino acids are present. The required amino acid profile per gram of protein is as follows: histidine–15 mg, isoleucine–30 mg, leucine–59 mg, lysine–45 mg, methionine + cysteine–22 mg, phenylalanine + tyrosine–38 mg, threonine–23 mg, tryptophan–6 mg, and valine–39 mg. | 7.24 ± 2.34 | 6.69 ± 2.32 | 6.69 ± 2.13 | 73% stakeholder 1 |
| 4. The selection of plant-based raw materials used to supply lipids in the formulation of plant-based meat analogue products should predominantly provide monounsaturated and polyunsaturated fatty acids. | 7.57 ± 2.50 | 7.60 ± 1.85 | 7.59 ± 1.97 | 76% all stakeholders |
| 5. The selection of plant-based raw materials intended to supply lipids in the formulation of plant-based meat analogue products should ensure the inclusion of omega-3 fatty acids, particularly at levels sufficient to qualify as a source of alpha-linolenic acid (ALA), defined as a minimum of 300 mg per serving size. | 5.69 ± 2.83 | 5.75 ± 2.71 | 5.72 ± 2.48 | 50% * stakeholder 1 |
| 6. The selection of plant-based raw materials used to supply lipids in the formulation of plant-based meat analogue products should ensure the provision of polyunsaturated fatty acids, with an omega-6: omega-3 ratio not exceeding 10:1 [56]. | 6.57 ± 2.63 | 6.33 ± 2.44 | 6.45 ± 2.25 | 60% stakeholders 1 and 2 |
| 7. The total fat content in plant-based meat analogue products should not exceed 30% DV for total energy value [50,55]. | 7.48 ± 2.80 | 7.66 ± 2.49 | 7.57 ± 2.49 | 73% all stakeholders |
| 8. The saturated fat content in plant-based meat analogue products should not exceed 10% DV for total energy value [50,54,55]. | 8.33 ± 2.58 | 7.81 ± 2.29 | 8.05 ± 2.27 | 76% all stakeholders |
| 9. The saturated fat content in plant-based meat analogue products should remain below the threshold established for the application of front-of-package nutritional warning labels for solid and semi-solid foods, as defined by Brazilian Food Labeling Law (equivalent to 6 g per 100 g) [60]. | 8.12 ± 2.42 | 7.90 ± 1.86 | 8.01 ± 1.91 | 79% all stakeholders |
| 10. Plant-based meat analogue products must contain dietary fiber levels that meet at least the minimum requirement to be classified as a source of fiber, as defined by Brazilian Food Labeling Law (equivalent to 10% DV, or 2.5 g of fiber per serving size) [60]. | 6.75 ± 2.78 | 7.75 ± 2.12 | 7.25 ± 2.33 | 73% stakeholders 1 and 2 |
| 11. The sodium content in plant-based meat analogue products must be below the threshold established for front-of-package nutritional warning labeling for solid and semi-solid foods, in accordance with Brazilian Food Labeling Law, which sets the limit at 600 mg per 100 g [60]. | 8.00 ± 2.88 | 7.27 ± 2.15 | 7.86 ± 2.39 | 76% all stakeholders |
| 12. The sodium content in plant-based meat analogue products should be less than 1 mg per kilocalorie, in accordance with PAHO/WHO nutritional guidelines [50] | 6.72 ± 2.85 | 6.51 ± 2.62 | 6.62 ± 2.55 | 70% stakeholders 1 and 3 |
| 13. Plant-based meat analogue products should be fortified with minerals (iron and zinc) and B vitamins (B6 and B12) at levels sufficient to qualify as sources of these nutrients, in accordance with Brazilian Food Labeling Law [60] which defines this threshold as 15% of DV per serving size. | 6.50 ± 2.90 | 7.00 ± 2.62 | 6.74 ± 2.42 | 64% stakeholders 1 and 2 |
| 14. Plant-based meat analogue products should be fortified with minerals (iron and zinc) and B vitamins (B6 and B12) at levels sufficient to qualify as sources of these nutrients, in accordance with Brazilian Food Labeling Law [60] which defines this threshold as 30% of DV per serving size. | 5.45 ± 2.92 | 6.39 ± 2.99 | 5.92 ± 2.73 | 53% * stakeholder 1 |
| Sensory Aspects | ||||
| 15. When the objective is to replicate meat products the sensory parameters of plant-based analogues should take precedence over nutritional quality. | 7.12 ± 2.64 | 7.12 ± 2.57 | 7.24 ± 2.30 | 73% all stakeholders |
| 16. When the objective is to replicate meat products the sensory and nutritional attributes should be given equal importance to effectively achieve product equivalence. | 8.09 ± 1.81 | 7.27 ± 1.62 | 7.68 ± 1.51 | 82% all stakeholders |
| Technology Aspects | ||||
| 17. The use of alternative proteins derived from Brazil’s rich biodiversity—including resources from various biomes and marine environments—should be prioritized in the development of plant-based products designed to mimic meat. | 7.69 ± 2.67 | 7.24 ± 2.30 | 7.46 ± 2.39 | 80% all stakeholders |
| 18. Formulations of plant-based meat products should aim to limit or avoid the inclusion of artificial additives and technological adjuvants, promoting cleaner and more natural ingredient profiles. | 6.90 ± 3.07 | 6.15 ± 2.75 | 6.55 ± 2.76 | 67% * stakeholder 1 |
| Regulatory Aspects | ||||
| 19. Plant-based meat analogue products should be recognized as a distinct food category within legislation governing the use of food additives and processing aids. | 7.30 ± 3.21 | 7.45 ± 2.68 | 7.30 ± 2.84 | 71% stakeholders 1 and 3 |
| 20. A food product formulated from plant-based raw materials that exhibits sensory characteristics comparable to its corresponding animal-derived product regulated by the Ministry of Agriculture and Livestock. | 7.03 ± 2.92 | 6.72 ± 2.97 | 6.87 ± 2.87 | 65% * stakeholder 1 |
| 21. A food product formulated from plant-based raw materials that demonstrates nutritional equivalence to the corresponding animal-derived product regulated by the Ministry of Agriculture and Livestock. | 6.60 ± 2.76 | 6.30 ± 2.82 | 6.45 ± 2.73 | 56% * stakeholders 1 and 2 |
| 22. A food product formulated from plant-based raw materials that corresponds to the consumption format of the analogous animal-derived product regulated by the Ministry of Agriculture and Livestock. | 7.30 ± 2.98 | 7.54 ± 2.73 | 7.37 ± 2.84 | 70% stakeholders 1 and 3 |
| 23. Mandatory inclusion of the statement: “This product may have a different nutritional value than the corresponding animal-derived product” on plant-based analogue products. | 7.33 ± 3.13 | 7.81 ± 3.13 | 7.57 ± 2.63 | 79% stakeholders 1 and 3 |
| 24. Mandatory inclusion of the statement: “This product does not replace its animal-based counterpart in nutritional or functional terms” on plant-based analogue products. | 5.21 ± 3.25 | 5.93 ± 3.31 | 5.75 ± 3.12 | 41% * stakeholder 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Locatelli, N.T.; Polezi, S.; Batista, M.F.; Bandoni, D.H.; de Rosso, V.V. Toward Nutritionally Sound Plant-Based Meat Analogues: Expert Consensus from a Delphi Study. Foods 2025, 14, 3068. https://doi.org/10.3390/foods14173068
Locatelli NT, Polezi S, Batista MF, Bandoni DH, de Rosso VV. Toward Nutritionally Sound Plant-Based Meat Analogues: Expert Consensus from a Delphi Study. Foods. 2025; 14(17):3068. https://doi.org/10.3390/foods14173068
Chicago/Turabian StyleLocatelli, Nathalia Tarossi, Sarah Polezi, Mariana Frazão Batista, Daniel Henrique Bandoni, and Veridiana Vera de Rosso. 2025. "Toward Nutritionally Sound Plant-Based Meat Analogues: Expert Consensus from a Delphi Study" Foods 14, no. 17: 3068. https://doi.org/10.3390/foods14173068
APA StyleLocatelli, N. T., Polezi, S., Batista, M. F., Bandoni, D. H., & de Rosso, V. V. (2025). Toward Nutritionally Sound Plant-Based Meat Analogues: Expert Consensus from a Delphi Study. Foods, 14(17), 3068. https://doi.org/10.3390/foods14173068








