Development and Prediction of a Non-Destructive Quality Index (Qi) for Stored Date Fruits Using VIS–NIR Spectroscopy and Artificial Neural Networks
Abstract
1. Introduction
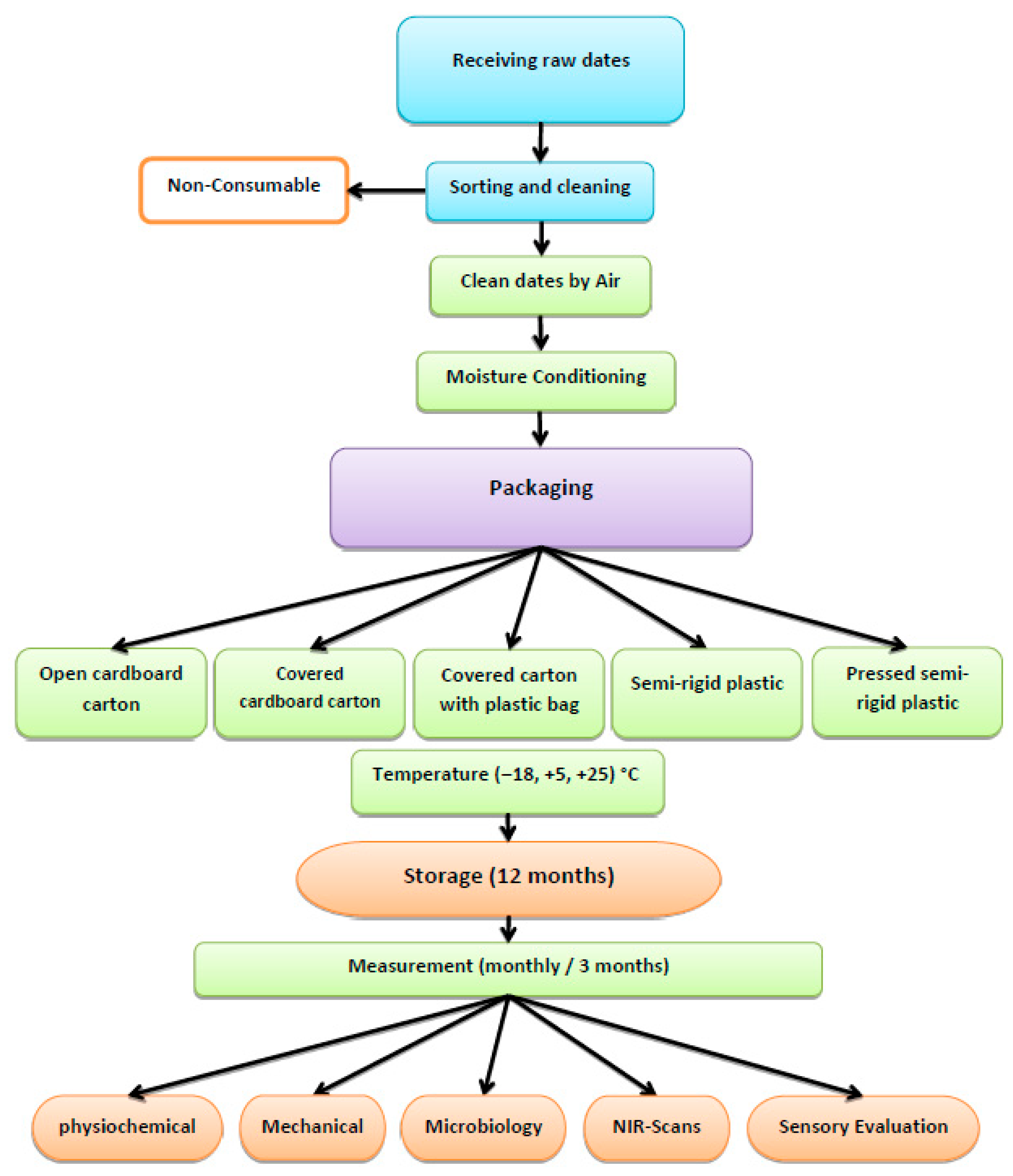
2. Materials, Methods, and Measurements
2.1. Materials
2.1.1. Dates
2.1.2. Packaging Materials
2.2. Methods
Storage of the Dates
2.3. Measurements
- (a)
- Physical properties
- (b)
- Mechanical properties
- (c)
- Chemical analysis
- (d)
- Assessment of Microbial Quality
- (e)
- Sensory evaluation
2.4. Modeling
- (a)
- Quality Index (Qi) Prediction
- (b)
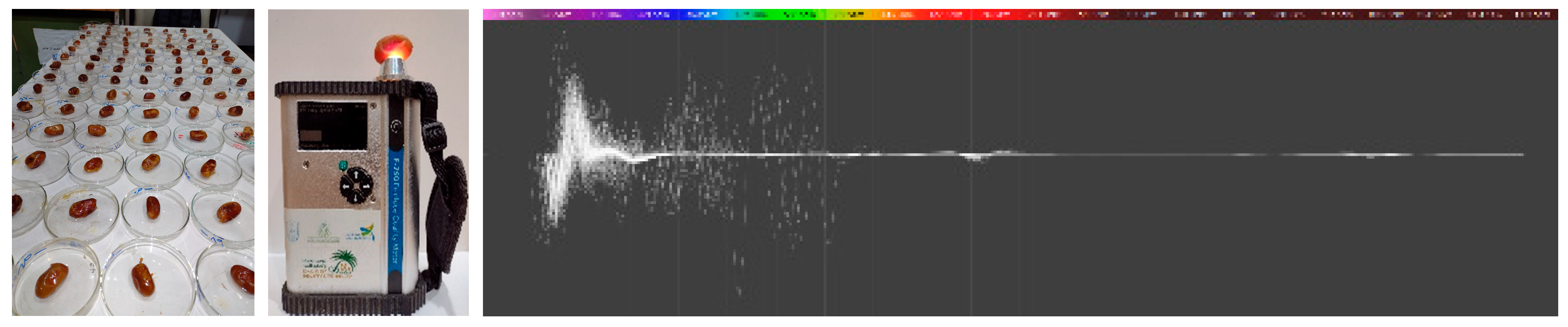
- The VIS-NIR Technique
2.5. Statistical Analysis
3. Results and Discussions
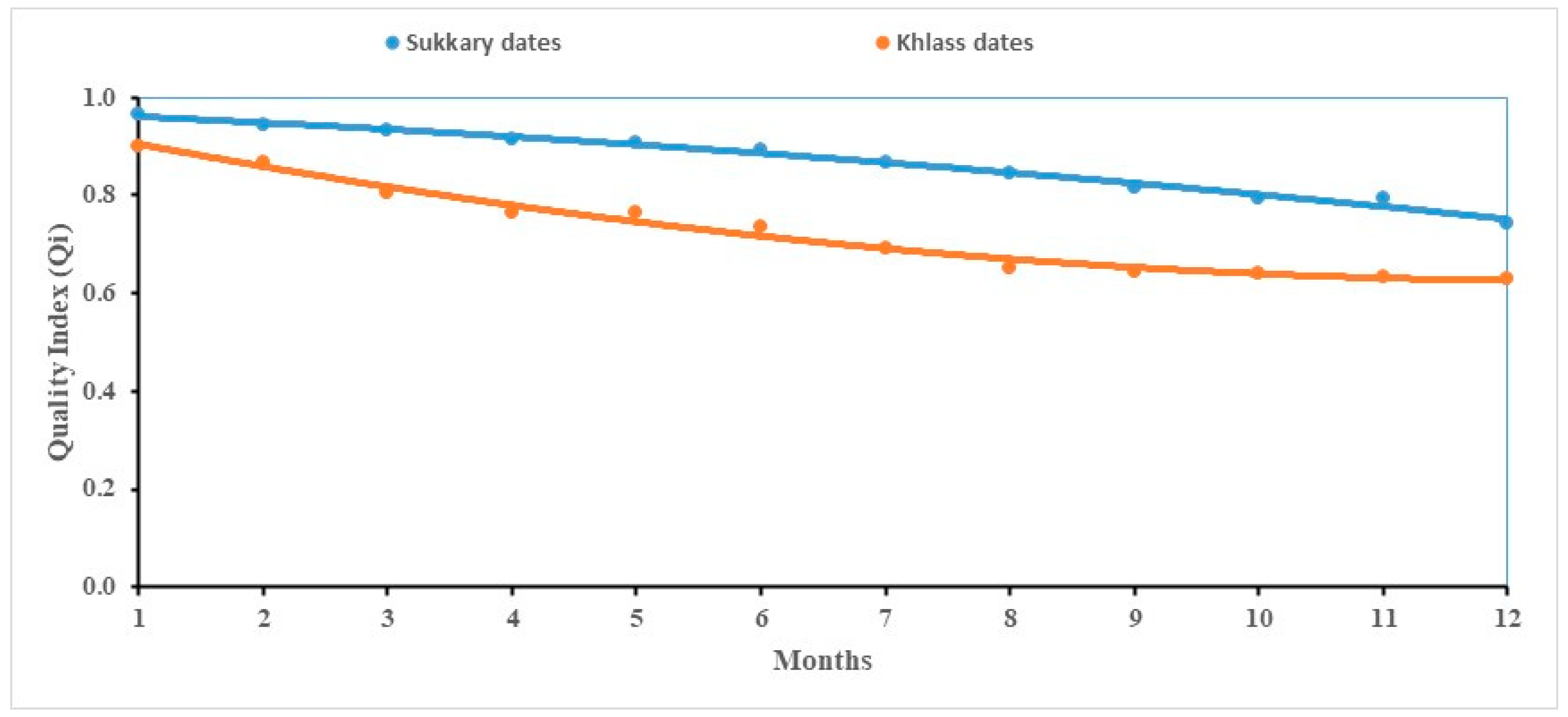
3.1. Modeling of the Quality Index (Qi)
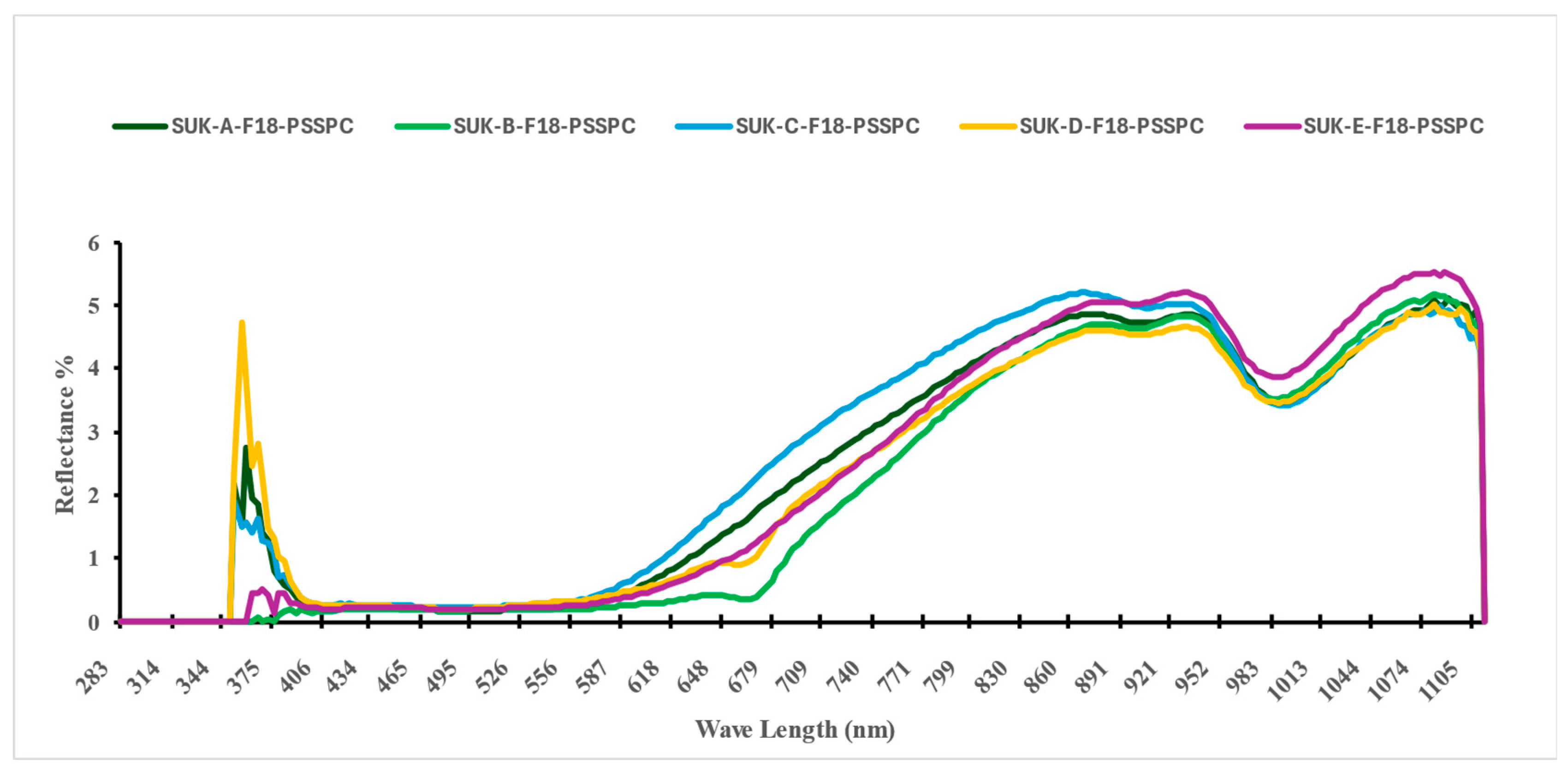
3.2. The VIS-NIR Spectral Analysis
3.3. Quality Index (Qi) Modeling Correlated with the VIS-NIR Spectra
3.3.1. Partial Least Squares Regression (PLSR)
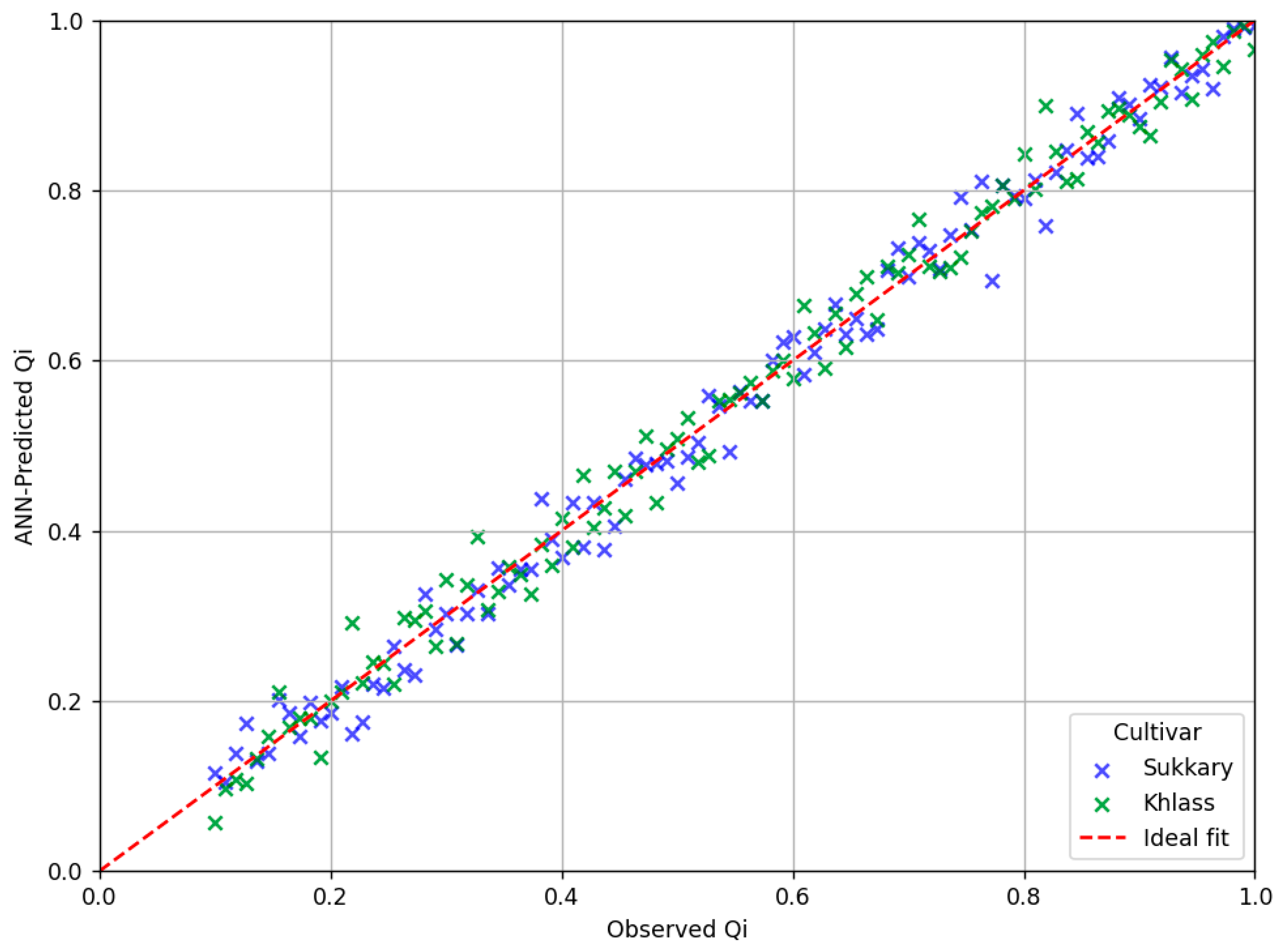
3.3.2. Artificial Neural Network (ANN) Analysis
3.3.3. An Evaluation of the Prediction Error Metrics for the Chemometric Models
3.3.4. The Comparative Evaluation of the Predictive Accuracy Metrics
3.4. Evaluation of the Physicochemical Properties of the Date Fruits During Storage
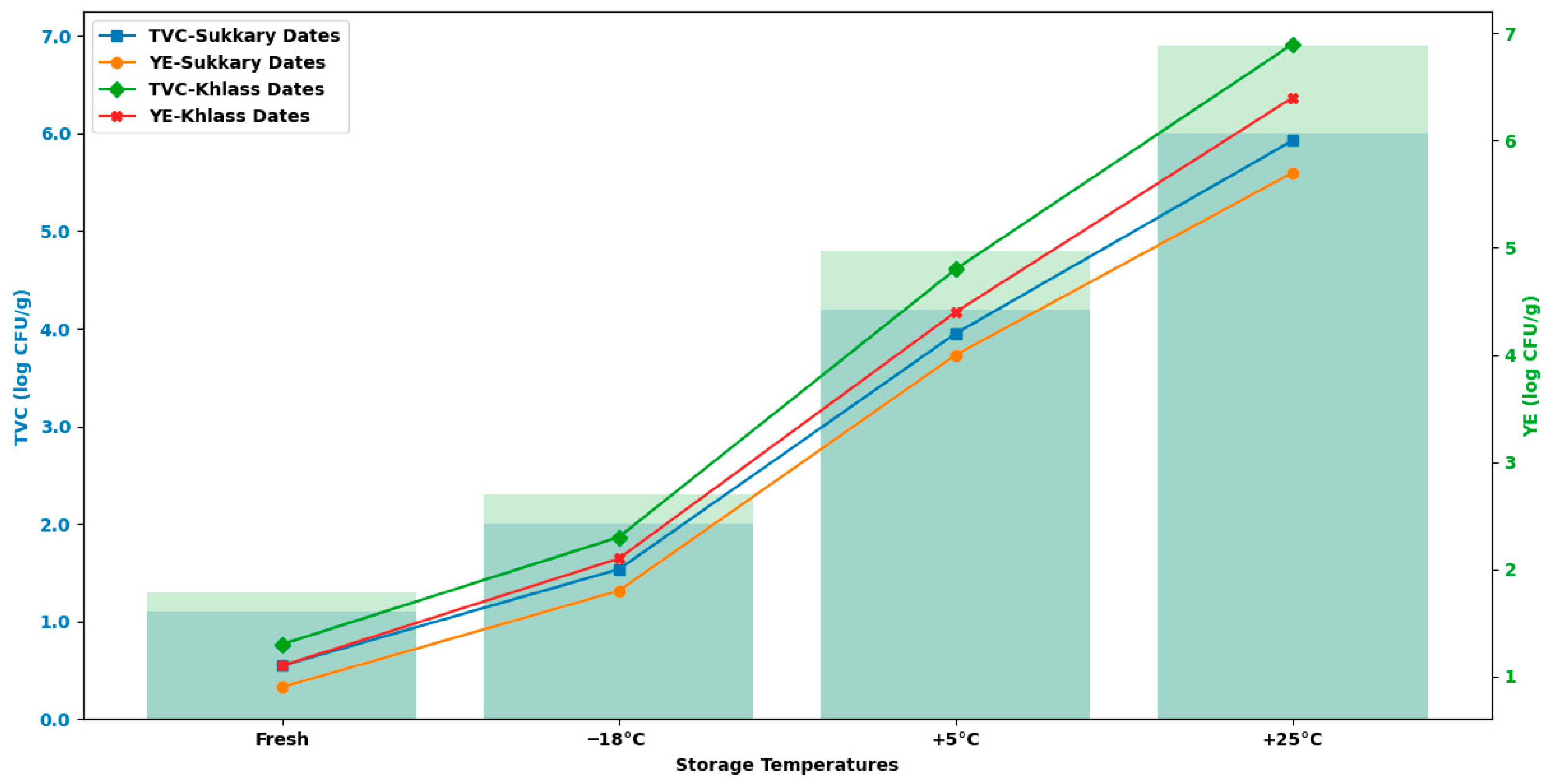
3.5. Assessment of Microbial Quality
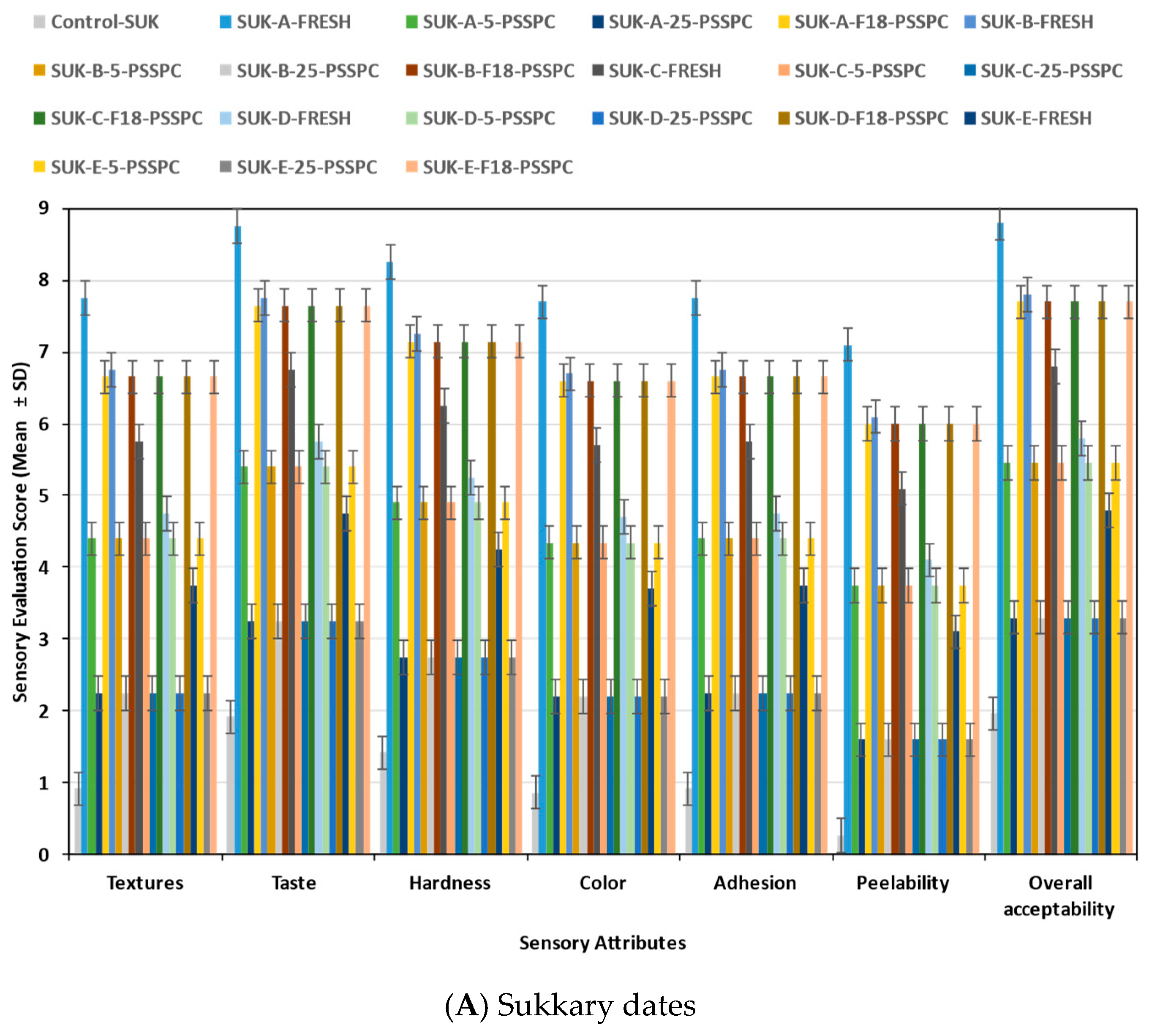
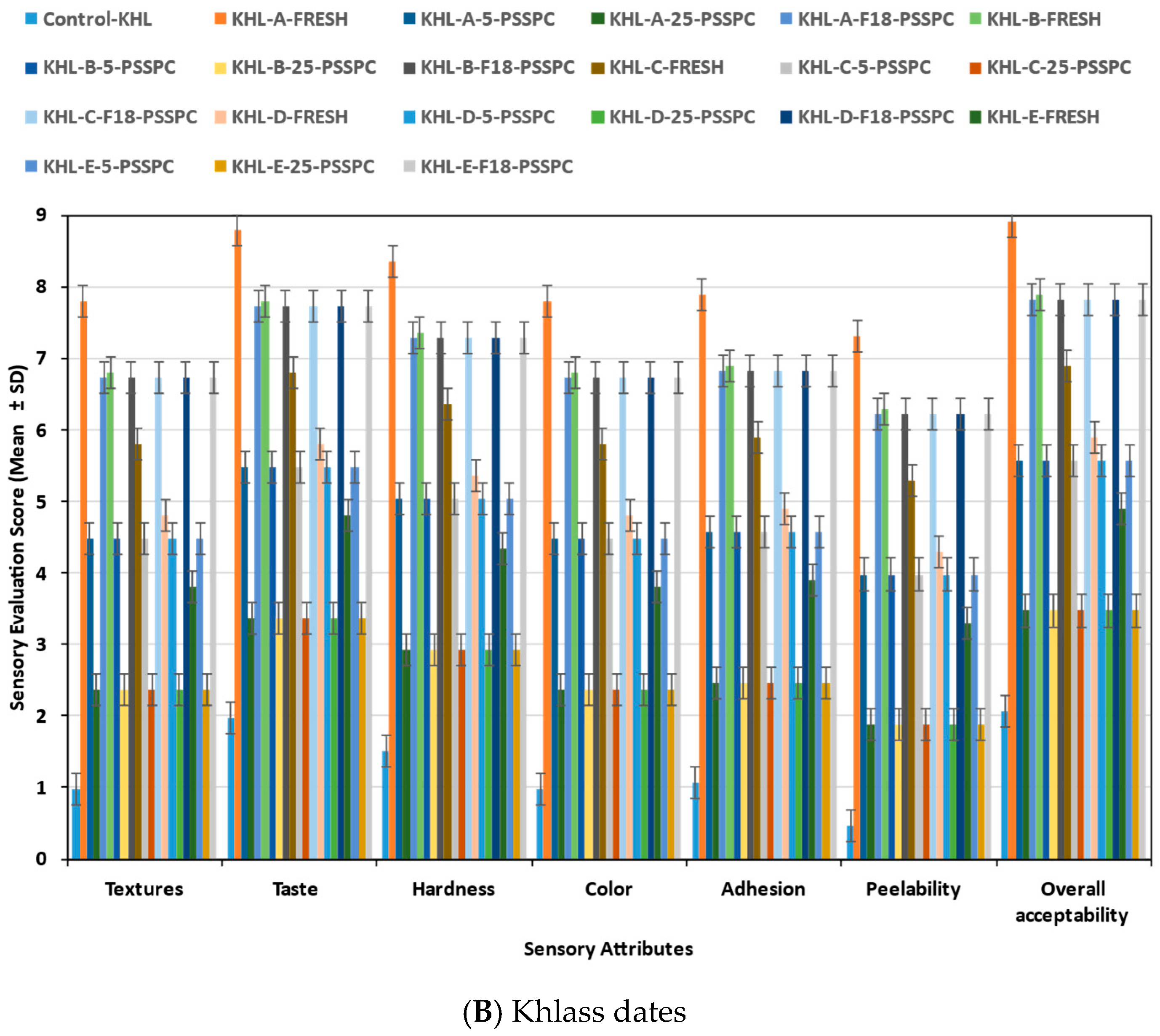
3.6. Sensory Evaluation of Stored Date Fruits (SDFs) During Storage
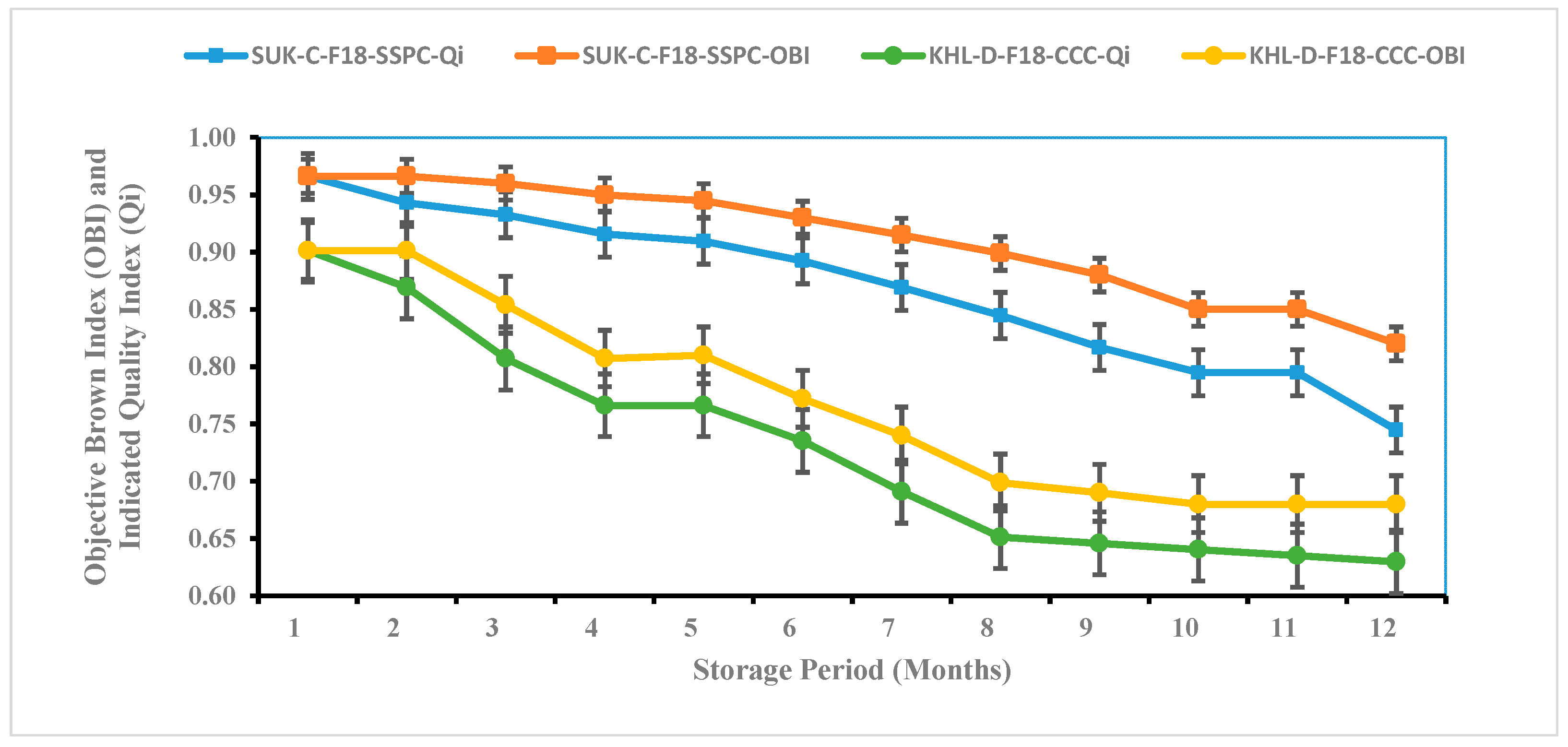
3.7. The Application of the NIR and Qi Models to Subjective and Objective Assessments
3.8. Limitations and Considerations in Non-Destructive Spectroscopic Assessments of Date Fruits’ Shelf Lives
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chao, C.T.; Krueger, R.R. The Date palm (Phoenix dactylifera L.): Overview of Biology, Uses, and Cultivation. Horts 2007, 42, 1077–1082. [Google Scholar] [CrossRef]
- Krueger, R.R. Date palm (Phoenix dactylifera L.) Biology and Utilization. In The Date Palm Genome; Al-Khayri, J.M., Jain, S.M., Johnson, D.V., Eds.; Compendium of Plant Genomes; Springer International Publishing: Cham, Switzerland, 2021; Volume 1, pp. 3–28. ISBN 978-3-030-73745-0. [Google Scholar]
- Zaid, A.; Arias-Jiménez, E.J. Date palm Cultivation; Food and Agriculture Organization of the United Nations: Rome, Italy, 2002; ISBN 978-92-5-104863-4. [Google Scholar]
- FAO. FAO’s Plant Production and Protection Division; FAO: Rome, Italy, 2024; ISBN 978-92-5-137003-2. [Google Scholar]
- Elamshity, M.G.; Alhamdan, A.M. Novel Analytical Approach for Quality Index Modeling of Physiochemical and Phytochemical Properties of Dates at Various Ripening Stages Using Heat Map Visualization, VIS-NIR Spectroscopy, and Artificial Neural Networks. Horticulturae, 2025; under review for publication. [Google Scholar]
- Al-Qurashi, A.D.; Awad, M.A. Quality Characteristics of Bisir ‘Barhee’ Dates during Cold Storage as Affected by Postharvest Dipping in Gibberellic Acid, Naphthaleneacetic Acid and Benzyladenine. Fruits 2011, 66, 343–352. [Google Scholar] [CrossRef]
- Mrázková, M.; Sumczynski, D.; Orsavová, J. Influence of Storage Conditions on Stability of Phenolic Compounds and Antioxidant Activity Values in Nutraceutical Mixtures with Edible Flowers as New Dietary Supplements. Antioxidants 2023, 12, 962. [Google Scholar] [CrossRef] [PubMed]
- Tembo, D.T.; Holmes, M.J.; Marshall, L.J. Effect of Thermal Treatment and Storage on Bioactive Compounds, Organic Acids and Antioxidant Activity of Baobab Fruit (Adansonia digitata) Pulp from Malawi. J. Food Compos. Anal. 2017, 58, 40–51. [Google Scholar] [CrossRef]
- Kapusta-Duch, J.; Kusznierewicz, B.; Leszczyńska, T.; Borczak, B. Effect of Package Type on Selected Parameters of Nutritional Quality of Chill-Stored White Sauerkraut. Pol. J. Food Nutr. Sci. 2017, 67, 137–144. [Google Scholar] [CrossRef]
- Van Ooijen, I.; Fransen, M.L.; Verlegh, P.W.J.; Smit, E.G. Atypical Food Packaging Affects the Persuasive Impact of Product Claims. Food Qual. Prefer. 2016, 48, 33–40. [Google Scholar] [CrossRef]
- Muratore, G.; Restuccia, C.; Licciardello, F.; Lombardo, S.; Pandino, G.; Mauromicale, G. Effect of Packaging Film and Antibrowning Solution on Quality Maintenance of Minimally Processed Globe Artichoke Heads. Innov. Food Sci. Emerg. Technol. 2015, 31, 97–104. [Google Scholar] [CrossRef]
- Abbott, J.A. Quality Measurement of Fruits and Vegetables. Postharvest Biol. Technol. 1999, 15, 207–225. [Google Scholar] [CrossRef]
- Teppola, P. Near-Infrared Spectroscopy: Principles, Instruments, Applications; Siesler, H.W., Ozaki, Y., Kawata, S., Heise, H.M., Eds.; Wiley-VCH: Weinheim, Germany, 2002; ISBN 3-527-30149-6. [Google Scholar]
- Burns, D.A.; Ciurczak, E.W. (Eds.) Handbook of Near-Infrared Analysis, 3rd ed.; Practical Spectroscopy; CRC Press: Boca Raton, FL, USA, 2008; ISBN 978-0-8493-7393-0. [Google Scholar]
- Marini, F.; Bucci, R.; Magrì, A.L.; Magrì, A.D. Artificial Neural Networks in Chemometrics: History, Examples and Perspectives. Microchem. J. 2008, 88, 178–185. [Google Scholar] [CrossRef]
- Hanrahan, G. Artificial Neural Networks in Biological and Environmental Analysis; CRC Press: Boca Raton, FL, USA, 2011; ISBN 978-0-429-14892-7. [Google Scholar]
- Salehi, F. Recent Advances in the Modeling and Predicting Quality Parameters of Fruits and Vegetables during Postharvest Storage: A Review. Int. J. Fruit Sci. 2020, 20, 506–520. [Google Scholar] [CrossRef]
- Kusumiyati, K.; Hadiwijaya, Y.; Putri, I.E. Non-Destructive Classification of Fruits Based on Vis-Nir Spectroscopy and Principal Component Analysis. J. Biodjati 2019, 4, 89–95. [Google Scholar] [CrossRef]
- Nicolaï, B.M.; Defraeye, T.; De Ketelaere, B.; Herremans, E.; Hertog, M.L.A.T.M.; Saeys, W.; Torricelli, A.; Vandendriessche, T.; Verboven, P. Nondestructive Measurement of Fruit and Vegetable Quality. Annu. Rev. Food Sci. Technol. 2014, 5, 285–312. [Google Scholar] [CrossRef]
- Wang, H.; Peng, J.; Xie, C.; Bao, Y.; He, Y. Fruit Quality Evaluation Using Spectroscopy Technology: A Review. Sensors 2015, 15, 11889–11927. [Google Scholar] [CrossRef]
- Ozaki, Y.; McClure, W.F.; Christy, A.A. (Eds.) Near-Infrared Spectroscopy in Food Science and Technology; Wiley-Interscience: Hoboken, NJ, USA, 2007; ISBN 978-0-471-67201-2. [Google Scholar]
- Tian, Z.; Tan, Z.; Li, Y.; Yang, Z. Rapid Monitoring of Flavonoid Content in Sweet Tea (Lithocarpus litseifolius (Hance) Chun) Leaves Using NIR Spectroscopy. Plant Methods 2022, 18, 44. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Zuo, Y.; Wu, Q.; Wang, J.; Ban, L.; Yang, H.; Bai, Z. Development and Validation of Near-Infrared Methods for the Quantitation of Caffeine, Epigallocatechin-3-Gallate, and Moisture in Green Tea Production. J. Anal. Methods Chem. 2021, 2021, 9563162. [Google Scholar] [CrossRef] [PubMed]
- Pandiselvam, R.; Kaavya, R.; Martinez Monteagudo, S.I.; Divya, V.; Jain, S.; Khanashyam, A.C.; Kothakota, A.; Prasath, V.A.; Ramesh, S.V.; Sruthi, N.U.; et al. Contemporary Developments and Emerging Trends in the Application of Spectroscopy Techniques: A Particular Reference to Coconut (Cocos nucifera L.). Molecules 2022, 27, 3250. [Google Scholar] [CrossRef] [PubMed]
- Da Silva Araújo, C.; Vimercati, W.C.; Macedo, L.L.; Ferreira, A.; Prezotti, L.C.; Teixeira, L.J.Q.; Saraiva, S.H. Predicting the Electric Conductivity and Potassium Leaching of Coffee by NIR Spectroscopy Technique. Food Anal. Methods 2020, 13, 2312–2320. [Google Scholar] [CrossRef]
- Cano-Reinoso, D.M.; Purwanto, Y.A.; Budiastra, I.W.; Kuroki, S.; Widodo, S.; Kamanga, B.M. Determination of α-Guaiene and Azulene Chemical Content in Patchouli Aromatic Oil (Pogostemon Cablin Benth.) from Indonesia by Near-Infrared Spectroscopy. Indian J. Nat. Prod. Resour. 2021, 12, 256–262. [Google Scholar] [CrossRef]
- Liu, Y.; Ying, Y. Use of FT-NIR Spectrometry in Non-Invasive Measurements of Internal Quality of ‘Fuji’ Apples. Postharvest Biol. Technol. 2005, 37, 65–71. [Google Scholar] [CrossRef]
- Alhamdan, A.M. Utilizing VIS-NIR Technology to Generate a Quality Index (Qi) Model of Barhi Date Fruits at the Khalal Stage Stored in a Controlled Environment. Foods 2024, 13, 345. [Google Scholar] [CrossRef]
- Alhamdan, A.M. NIR Spectroscopy Assessment of Quality Index of Fermented Milk (Laban) Drink Flavored with Date Syrup during Cold Storage. Fermentation 2022, 8, 438. [Google Scholar] [CrossRef]
- Elamshity, M.G.; Alhamdan, A.M. Non-Destructive Evaluation of the Physiochemical Properties of Milk Drink Flavored with Date Syrup Utilizing VIS-NIR Spectroscopy and ANN Analysis. Foods 2024, 13, 524. [Google Scholar] [CrossRef]
- Ahmed, Z.F.R.; Al Shaibani, F.Y.Y.; Kaur, N.; Maqsood, S.; Schmeda-Hirschmann, G. Improving Fruit Quality, Bioactive Compounds, and Storage Life of Date palm (Phoenix dactylifera L., Cv. Barhi) Using Natural Elicitors. Horticulturae 2021, 7, 293. [Google Scholar] [CrossRef]
- Hussain, M.I.; Farooq, M.; Syed, Q.A. Nutritional and Biological Characteristics of the Date palm Fruit (Phoenix dactylifera L.) A Review. Food Biosci. 2020, 34, 100509. [Google Scholar] [CrossRef]
- Lin, H.; Ying, Y. Theory and Application of Near Infrared Spectroscopy in Assessment of Fruit Quality: A Review. Sens. Instrumen. Food Qual. 2009, 3, 130–141. [Google Scholar] [CrossRef]
- Arévalo-Pinedo, A.; Murr, F.E.X. Kinetics of Vacuum Drying of Pumpkin (Cucurbita Maxima): Modeling with Shrinkage. J. Food Eng. 2006, 76, 562–567. [Google Scholar] [CrossRef]
- Al-Muhtaseb, A.H.; McMinn, W.A.M.; Magee, T.R.A. Water Sorption Isotherms of Starch Powders. J. Food Eng. 2004, 61, 297–307. [Google Scholar] [CrossRef]
- Zaid, A.; Arias-Jiménez, E. Date palm Cultivation. In Plant Production and Protection; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2002; p. 156. [Google Scholar]
- Doymaz, İ. Drying Characteristics and Kinetics of Okra. J. Food Eng. 2005, 69, 275–279. [Google Scholar] [CrossRef]
- Maskan, M. Kinetics of Colour Change of Kiwifruits during Hot Air and Microwave Drying. J. Food Eng. 2001, 48, 169–175. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis, 17th ed.; AOAC International: Gaithersburg, MD, USA, 2000; ISBN 978-0-935584-83-7. [Google Scholar]
- Buntinx, M.; Willems, G.; Knockaert, G.; Adons, D.; Yperman, J.; Carleer, R.; Peeters, R. Evaluation of the Thickness and Oxygen Transmission Rate before and after Thermoforming Mono- and Multi-Layer Sheets into Trays with Variable Depth. Polymers 2014, 6, 3019–3043. [Google Scholar] [CrossRef]
- Xie, L.-G.; Zhao, X.; Dou, S.-H.; Tang, L.; Sun, H.-M. A New Standard Reference Film for Oxygen Gas Transmission Measurements. R. Soc. Open Sci. 2019, 6, 190142. [Google Scholar] [CrossRef]
- Seong, D.M.; Lee, H.; Kim, J.; Chang, J.H. High Oxygen and Water-Vapor Transmission Rate and In Vitro Cytotoxicity Assessment with Illite-Polyethylene Packaging Films. Materials 2020, 13, 2382. [Google Scholar] [CrossRef] [PubMed]
- Jarvis, K.L.; Evans, P.J.; Cooling, N.A.; Vaughan, B.; Habsuda, J.; Belcher, W.J.; Bilen, C.; Griffiths, G.; Dastoor, P.C.; Triani, G. Comparing Three Techniques to Determine the Water Vapour Transmission Rates of Polymers and Barrier Films. Surf. Interfaces 2017, 9, 182–188. [Google Scholar] [CrossRef]
- Pan, Z.; Bora, M.; Gee, R.; Dauskardt, R.H. Water Vapor Transmission Rate Measurement for Moisture Barriers Using Infrared Imaging. Mater. Chem. Phys. 2023, 308, 128289. [Google Scholar] [CrossRef]
- Buchwalder, S.; Nicolier, C.; Hersberger, M.; Bourgeois, F.; Hogg, A.; Burger, J. Development of a Water Transmission Rate (WTR) Measurement System for Implantable Barrier Coatings. Polymers 2023, 15, 2557. [Google Scholar] [CrossRef]
- Karamfilova, E. Food and Drug Administration General Food-Contact Statement. Food Contact Materials Regulation (EC) 1935/2004. European Implementation Assessment. Brussels © European Union. PE 581.411. Food Contact Materials-Regulation (EC) 1935/2004. 2016. Available online: https://www.europarl.europa.eu/RegData/etudes/STUD/2016/581411/EPRS_STU(2016)581411_EN.pdf (accessed on 17 August 2025).
- Official Journal of the European Union. Regulation (EC) No 1935/2004 of the European Parliament and of the Council of 27 October 2004 on Materials and Articles Intended to Contact Food and Repealing Directives 80/590/EEC and 89/109/EEC. Document 32004R1935. L 338/4. Part Paper & Board (OCC/CCC). FDA 21 CFR Part 176 (176.170, 176.180). Regulation-1935/2004-EN-EUR-Lex. 2004. Available online: https://eur-lex.europa.eu/eli/reg/2004/1935/oj/eng (accessed on 17 August 2025).
- Official Journal of the European Union. Regulation (EC) No 1935/2004 of the European Parliament and of the Council of 27 October 2004 on Materials and Articles Intended to Contact Food and Repealing Directives 80/590/EEC and 89/109/EEC. Document 32004R1935. L 338/4. Plastics (PP/PE, PET). FDA 21 CFR. 177.1520 (Olefin Polymers), 177.1630 (Polyethylene Phthalate Polymers). Regulation-1935/2004-EN-EUR-Lex. 2004. Available online: https://eur-lex.europa.eu/eli/reg/2004/1935/oj/eng (accessed on 17 August 2025).
- Official Journal of the European Union. Regulation (EC) No 1935/2004 of the European Parliament and of the Council of 27 October 2004 on Materials and Articles Intended to Contact Food and Repealing Directives 80/590/EEC and 89/109/EEC. Document 32004R1935. L 338/4. Statements Involving Specific Migration Limits (SML). (SML Framework & Union List) + Relevant FDA 21 CFR Part 177 Section(s). Regulation-1935/2004-EN-EUR-Lex. 2004. Available online: https://eur-lex.europa.eu/eli/reg/2004/1935/oj/eng (accessed on 17 August 2025).
- Brody, A.L. Packaging of Foods. In Encyclopedia of Food Microbiology; Elsevier: Amsterdam, The Netherlands, 2014; pp. 1017–1027. ISBN 978-0-12-384733-1. [Google Scholar]
- Goswami, T.K.; Mangaraj, S. Advances in Polymeric Materials for Modified Atmosphere Packaging (MAP). In Multifunctional and Nanoreinforced Polymers for Food Packaging; Elsevier: Amsterdam, The Netherlands, 2011; pp. 163–242. ISBN 978-1-84569-738-9. [Google Scholar]
- Rosato, D.V.; Rosato, D.V.; Rosato, M.V. Plastic Property. In Plastic Product Material and Process Selection Handbook; Elsevier: Amsterdam, The Netherlands, 2004; pp. 40–129. ISBN 978-1-85617-431-2. [Google Scholar]
- Morris, B.A. Barrier. In The Science and Technology of Flexible Packaging; Elsevier: Amsterdam, The Netherlands, 2017; pp. 259–308. ISBN 978-0-323-24273-8. [Google Scholar]
- McKeen, L.W. Introduction to Use of Plastics in Food Packaging. In Plastic Films in Food Packaging; Elsevier: Amsterdam, The Netherlands, 2013; pp. 1–15. ISBN 978-1-4557-3112-1. [Google Scholar]
- Morris, B.A. Commonly Used Resins and Substrates in Flexible Packaging. In The Science and Technology of Flexible Packaging; Elsevier: Amsterdam, The Netherlands, 2017; pp. 69–119. ISBN 978-0-323-24273-8. [Google Scholar]
- Saudi Food & Drug Authority (SFDA) Packing and Packaging Materials 2010. SFDA Announcement Concerning the Import Requirements for Food Contact Packing and Packaging Materials|The Official Website of the Saudi Food and Drug Authority. Available online: https://www.sfda.gov.sa/en/circulars/60309 (accessed on 17 August 2025).
- Motarjemi, Y. Moy, Gerald., Todd, Ewen. Encyclopedia of Food Safety; Elsevier: Amsterdam, The Netherlands, 2014; eBook; ISBN 9780123786135. [Google Scholar]
- Saudi Food & Drug Authority (SFDA). Expiration Dates for Food Products—Part 1: Mandatory Expiration Dates; Saudi Food & Drug Authority (SFDA): Riyadh, Saudi Arabia, 2018. [Google Scholar]
- Codex Alimentarius: Food Labelling Complete Texts, Rev. 2001, ed.; FAO, Ed.; Food and Agriculture Organization of the United Nations: Rome, Italy; World Health Organization: Geneva, Switzerland, 2001; ISBN 978-92-5-104679-1. [Google Scholar]
- U.S. Food and Drug Administration (FDA). Human Foods Program; FDA: Silver Spring, MD, USA, 2024; ISBN 1-888-723-3366. [Google Scholar]
- Official Methods of Analysis. Official Methods of Analysis of AOAC International, 22nd ed.; Latimer, G.W., Ed.; Oxford University Press: New York, NY, USA, 2023; ISBN 978-0-19-761013-8. [Google Scholar]
- Elamshity, M.G. Development of a Nutritional Drink from Cow’s and Camel’s Milk with Date Syrup. Master’s Thesis, King Saud University, Riyadh, Saudi Arabia, 2014. [Google Scholar]
- Fairchild, M.D. Color Appearance Models. Wiley-IS&T Series in Imaging Science and Technology, 3rd ed.; Wiley: Chichester, UK, 2013; ISBN 978-1-119-96703-3. [Google Scholar]
- Webvision: The Organization of the Retina and Visual System; Kolb, H., Fernandez, E., Nelson, R., Eds.; University of Utah Health Sciences Center: Salt Lake City, UT, USA, 1995. [Google Scholar]
- Aly, M.M.A.; Seleem, H.A. Gluten-Free Flat Bread and Biscuits Production by Cassava, Extruded Soy Protein and Pumpkin Powder. Food Nutr. Sci. 2015, 6, 660–674. [Google Scholar] [CrossRef]
- Bourne, M.C. Food Texture and Viscosity: Concept and Measurement, 2nd ed.; Food Science and Technology International Series; Academic Press: San Diego, CA, USA, 2002; ISBN 978-0-12-119062-0. [Google Scholar]
- Gupta, R.K.; Sharma, A.; Sharma, R. Instrumental Texture Profile Analysis (TPA) of Shelled Sunflower Seed Caramel Snack Using Response Surface Methodology. Food Sci. Technol. Int. 2007, 13, 455–460. [Google Scholar] [CrossRef]
- Meullenet, J.; Lyon, B.G.; Carpenter, J.A.; Lyon, C.E. Relationship Between Sensory and Instrumental Texture Profile Attributes. J. Sens. Stud. 1998, 13, 77–93. [Google Scholar] [CrossRef]
- Moskowitz, H.R.; Kapsalis, J.G. The Texture Profile: Its Foundations and Outlook. J. Texture Stud. 1975, 6, 157–166. [Google Scholar] [CrossRef]
- Nishinari, K.; Kohyama, K.; Kumagai, H.; Funami, T.; Bourne, M.C. Parameters of Texture Profile Analysis. Food Sci. Technol. Res. 2013, 19, 519–521. [Google Scholar] [CrossRef]
- Peleg, M. Texture Profile Analysis Parameters Obtained by an Instron Universal Testing Machine. J. Food Sci. 1976, 41, 721–722. [Google Scholar] [CrossRef]
- Rivera, S.; Kerckhoffs, H.; Sofkova-Bobcheva, S.; Hutchins, D.; East, A. Data of Texture Profile Analysis Performed by Different Input Settings on Stored ‘Nui’ and ‘Rahi’ Blueberries. Data Brief. 2021, 38, 107313. [Google Scholar] [CrossRef] [PubMed]
- ISO 4833-1:2013; Microbiology of the Food Chain—Horizontal Method for the Enumeration of Microorganisms—Part 1: Colony Count at 30 °C by the Pour Plate Technique. International Organization for Standardization (ISO): Geneva, Switzerland, 2013.
- ISO 21527-2:2008; Microbiology of Food and Animal Feeding Stuffs—Horizontal Method for the Enumeration of Yeasts and Moulds—Part 2: Colony Count Technique in Products with Water Activity Less Than or Equal to 0.95. International Organization for Standardization (ISO): Geneva, Switzerland, 2008.
- Daroub, H.; Olabi, A.; Toufeili, I. Designing and Testing of an Arabic Version of the Hedonic Scale for Use in Acceptability Tests. Food Qual. Prefer. 2010, 21, 33–43. [Google Scholar] [CrossRef]
- Larmond, E. Laboratory Methods for Sensory Evaluation of Food; Agriculture Canada, Research Branch: Ottawa, ON, Canada, 1977; ISBN 978-0-662-01271-9. [Google Scholar]
- Lawless, H.T.; Heymann, H. Sensory Evaluation of Food: Principles and Practices; Food Science Text Series; Springer: New York, NY, USA, 2010; ISBN 978-1-4419-6487-8. [Google Scholar]
- Lim, J. Hedonic Scaling: A Review of Methods and Theory. Food Qual. Prefer. 2011, 22, 733–747. [Google Scholar] [CrossRef]
- Meilgaard, M.C.; Carr, B.T.; Civille, G.V. Sensory Evaluation Techniques; CRC Press: Boca Raton, FL, USA, 1999; ISBN 978-1-003-04072-9. [Google Scholar]
- Nicolas, L.; Marquilly, C.; O’Mahony, M. The 9-Point Hedonic Scale: Are Words and Numbers Compatible? Food Qual. Prefer. 2010, 21, 1008–1015. [Google Scholar] [CrossRef]
- Silva, A.N.D.; Silva, R.D.C.D.S.N.D.; Ferreira, M.A.M.; Minim, V.P.R.; Costa, T.D.M.T.D.; Perez, R. Performance of Hedonic Scales in Sensory Acceptability of Strawberry Yogurt. Food Qual. Prefer. 2013, 30, 9–21. [Google Scholar] [CrossRef]
- ISO 8589; Sensory Analysis. International Organization for Standardization: Geneva, Switzerland, 2007.
- Meilgaard, M.C.; Civille, G.V.; Carr, B.T. Sensory Evaluation Techniques, 5th ed.; CRC Press: Boca Raton, FL, USA, 2016; ISBN 978-1-4987-2225-7. [Google Scholar]
- MacFie, H.J.H.; Bratchell, N.; Greenhoff, K.; Vallis, L.V. Designs to Balance the Effects of Order of Presentation and First-Order Carry-over Effects in Sensory Evaluation. Food Qual. Prefer. 1989, 1, 117–125. [Google Scholar]
- Williams, E.J. Experimental Designs Balanced for the Estimation of Residual Effects of Treatments. Aust. J. Sci. Res. Ser. A 1949, 2, 149–168. [Google Scholar] [CrossRef]
- Aleid, S.M.; Hassan, B.H.; Almaiman, S.A.; Al-Kahtani, S.H.; Ismail, S.M. Microbial Loads and Physicochemical Characteristics of Fruits from Four Saudi Date palm Tree Cultivars: Conformity with Applicable Date Standards. Food Nutr. Sci. 2014, 5, 316–327. [Google Scholar] [CrossRef][Green Version]
- Zamir, R.; Islam, A.B.M.N.; Rahman, A.; Ahmed, S.; Omar Faruque, M. Microbiological Quality Assessment of Popular Fresh Date Samples Available in Local Outlets of Dhaka City, Bangladesh. Int. J. Food Sci. 2018, 2018. [Google Scholar] [CrossRef]
- Alp, D.; Bulantekin, Ö. The Microbiological Quality of Various Foods Dried by Applying Different Drying Methods: A Review. Eur. Food Res. Technol. 2021, 247, 1333–1343. [Google Scholar] [CrossRef]
- Berthold-Pluta, A.; Garbowska, M.; Stefańska, I.; Stasiak-Różańska, L.; Aleksandrzak-Piekarczyk, T.; Pluta, A. Microbiological Quality of Nuts, Dried and Candied Fruits, Including the Prevalence of Cronobacter spp. Pathogens 2021, 10, 900. [Google Scholar] [CrossRef] [PubMed]
- Codex Alimentarius Commission. CXC 75-2015: Code of Hygienic Practice for Low-Moisture Foods; FAO/WHO Codex Alimentarius: Rome, Italy, 2015. [Google Scholar]
- CXS 143-1985; Codex Codex Standard for Dates. Food and Agriculture Organization of the United Nations: Rome, Italy; World Health Organization: Geneva, Switzerland, 1985.
- Codex Alimentarius Commission. Codex Committee on Food Hygiene (CCFH47)—Working Document FH47/07 (Draft); FAO/WHO Codex Alimentarius: Rome, Italy, 2001. [Google Scholar]
- Gorris, L. 2024 ICMSF Workshop (CCFH54): Opening Key Concepts; ICMSF: Mendota Heights, MN, USA, 2024. [Google Scholar]
- Swanson, K.M.J.; Buchanan, R.L.; Cole, M.B.; Cordier, J.L.; Flowers, R.S.; Gorris, L.G.M.; Gorris, M.H.; Taniwaki, R.B.; Zwietering, M.H. Use of Data for Assessing Process Control and Product Acceptance; Microorganisms in Foods; ICMSF, Ed.; Springer: New York, NY, USA, 2011; ISBN 978-1-4419-9373-1. [Google Scholar]
- Roberts, T.; van Schothorst, M.; Sharpe, A.; BairdParker, A.; Bryan, F.; Buchanan, R.; Busta, F.; Doyle, M.; Farkas, J.; Grau, F.; et al. The International Commission on Microbiological Specifications for Foods (ICMSF): Update. Food Control 1996, 7, 99–101. [Google Scholar] [CrossRef]
- ICMSF International Commission on Microbiological Specifications for Foods (ICMSF) Official Website. Available online: https://www.icmsf.org/ (accessed on 15 August 2025).
- International Organization for Standardization (ISO) ISO 7218—General Requirements and Guidance (ISO/TC 34/SC 9 Landing Page). Available online: https://committee.iso.org/sites/tc34sc9/home/general-standards/content-left-area/culture-media/iso-7218-general-requirements-an.html (accessed on 15 August 2025).
- National Advisory Committee on Microbiological Criteria for Foods (NACMCF). NACMCF: Testing—FDA Text, Appendices A–F (Draft); USDA Food Safety and Inspection Service (FSIS): Washington, DC, USA, 2021. [Google Scholar]
- Food & Drug Administration (Philippines). FDA Circular No. 2022-12-2; FDA Philippines: Muntinlupa City, Philippines, 2022. [Google Scholar]
- Al-Qarawi, A.A.; Ali, B.H.; Al-Mougy, S.A.; Mousa, H.M. Gastrointestinal Transit in Mice Treated with Various Extracts of Date (Phoenix dactylifera L.). Food Chem. Toxicol. 2003, 41, 37–39. [Google Scholar] [CrossRef] [PubMed]
- Camarinha-Matos, L.M. Collaborative Smart Grids—A Survey on Trends. Renew. Sustain. Energy Rev. 2016, 65, 283–294. [Google Scholar] [CrossRef]
- Jayalakshmi, T.; Santhakumaran, A. Statistical Normalization and Back Propagationfor Classification. Int. J. Comput. Theory Eng. 2011, 3, 89–93. [Google Scholar] [CrossRef]
- Janecek, M.; Moses, W.W. Optical Reflectance Measurements for Commonly Used Reflectors. IEEE Trans. Nucl. Sci. 2008, 55, 2432–2437. [Google Scholar] [CrossRef]
- Jones, A.; Smith, B. Reflectance Measurement Techniques. J. Opt. Sci. 2019, 14, 123–135. [Google Scholar]
- Kortüm, G. Reflectance Spectroscopy: Principles, Methods, Applications, 1st ed.; Softcover Repr. of the Hardcover 1969; Springer: New York, NY, USA, 2014; ISBN 978-3-642-88073-5. [Google Scholar]
- Brown, C.; White, D.; Johnson, E. Relative Reflectance Measurements: Principles and Applications. J. Appl. Opt. 2020, 8, 321–333. [Google Scholar]
- Gene, J.; Jeon, M.Y.; Lim, S.D. Reflectometers for Absolute and Relative Reflectance Measurements in the Mid-IR Region at Vacuum. Sensors 2021, 21, 1169. [Google Scholar] [CrossRef]
- Wallace, M.B.; Wax, A.; Roberts, D.N.; Graf, R.N. Reflectance Spectroscopy. Gastrointest. Endosc. Clin. N. Am. 2009, 19, 233–242. [Google Scholar] [CrossRef]
- Basile, T.; Marsico, A.D.; Perniola, R. Use of Artificial Neural Networks and NIR Spectroscopy for Non-Destructive Grape Texture Prediction. Foods 2022, 11, 281. [Google Scholar] [CrossRef]
- Whitbeck, M.R. Second Derivative Infrared Spectroscopy. Appl. Spectrosc. 1981, 35, 93–95. [Google Scholar] [CrossRef]
- Barnes, R.J.; Dhanoa, M.S.; Lister, S.J. Standard Normal Variate Transformation and De-Trending of Near-Infrared Diffuse Reflectance Spectra. Appl. Spectrosc. 1989, 43, 772–777. [Google Scholar] [CrossRef]
- Isaksson, T.; Næs, T. The Effect of Multiplicative Scatter Correction (MSC) and Linearity Improvement in NIR Spectroscopy. Appl. Spectrosc. 1988, 42, 1273–1284. [Google Scholar] [CrossRef]
- Rinnan, Å.; Berg, F.V.D.; Engelsen, S.B. Review of the Most Common Pre-Processing Techniques for near-Infrared Spectra. TrAC Trends Anal. Chem. 2009, 28, 1201–1222. [Google Scholar] [CrossRef]
- Nordey, T.; Joas, J.; Davrieux, F.; Chillet, M.; Léchaudel, M. Robust NIRS Models for Non-Destructive Prediction of Mango Internal Quality. Sci. Hortic. 2017, 216, 51–57. [Google Scholar] [CrossRef]
- Kusumiyati, K.; Munawar, A.A.; Suhandy, D. Department of Agrotechnology, Padjadjaran University, Bandung-Indonesia; Department of Agricultural Engineering, Syiah Kuala University, Banda Aceh-Indonesia; Department of Agricultural Engineering, Lampung University Bandar Lampung-Indonesia Fast. Simultaneous and Contactless Assessment of Intact Mango Fruit by Means of Near Infrared Spectroscopy. AIMS Agric. Food 2021, 6, 172–184. [Google Scholar] [CrossRef]
- Silalahi, D.D.; Reaño, C.E.; Lansigan, F.P.; Panopio, R.G.; Bantayan, N.C. Using Genetic Algorithm Neural Network on Near Infrared Spectral Data for Ripeness Grading of Oil Palm (Elaeis guineensis Jacq.) Fresh Fruit. Inf. Process. Agric. 2016, 3, 252–261. [Google Scholar] [CrossRef]
- American Association of Cereal Chemists. Near-Infrared Technology in the Agricultural and Food Industries; Williams, P., Norris, K.H., American Association of Cereal Chemists, Eds.; American Association of Cereal Chemists: St. Paul, MN, USA, 1987; ISBN 978-0-913250-49-5. [Google Scholar]
- De Jong, S. Multivariate calibration, H. Martens and T. Naes, Wiley, New York, 1989. ISBN 0 471 90979 3. Price: £75.00, US$138.00. No. of pages: 504. J. Chemom. 1990, 4, 441. [Google Scholar] [CrossRef]
- Blanco, M.; Villarroya, I. NIR Spectroscopy: A Rapid-Response Analytical Tool. TrAC Trends Anal. Chem. 2002, 21, 240–250. [Google Scholar] [CrossRef]
- SAS Institute Inc. SAS/STAT® 9.4, version 9.4; Computer Software: Cary, NC, USA, 2023.
- Python Software Foundation Python Language Reference. Python, version 3.12.4; 2024. Available online: https://www.python.org/downloads/release/python-3124/ (accessed on 6 June 2024).
- Virtanen, P.; Gommers, R.; Oliphant, T.E.; Haberland, M.; Reddy, T.; Cournapeau, D.; Burovski, E.; Peterson, P.; Weckesser, W.; Bright, J.; et al. SciPy 1.0: Fundamental Algorithms for Scientific Computing in Python. Nat. Methods 2020, 17, 261–272. [Google Scholar] [CrossRef]
- Reback, J.; McKinney, W.; Jbrockmendel; Bossche, J.V.D.; Augspurger, T.; Cloud, P.; Gfyoung; Sinhrks; Klein, A.; Hawkins, S.; et al. pandas-dev/pandas: Pandas 1.4.2. Zenodo: Geneva, Switzerland, 2022; Available online: https://zenodo.org/records/6408044 (accessed on 17 August 2025).
- Waskom, M.L. Seaborn: Statistical Data Visualization. J. Open Source Softw. 2021, 6, 3021. [Google Scholar] [CrossRef]
- Harris, C.R.; Millman, K.J.; Van Der Walt, S.J.; Gommers, R.; Virtanen, P.; Cournapeau, D.; Wieser, E.; Taylor, J.; Berg, S.; Smith, N.J.; et al. Array Programming with NumPy. Nature 2020, 585, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Microsoft Corporation Microsoft Office 365. Microsoft Computer Software; Microsoft Corporation Microsoft Office 365: Redmond, WA, USA, 2023. [Google Scholar]
- Al-Habsi, N.A.; Al-Kalbani, B.; Al-Farsi, M.; Rahman, M.S. Quality Changes in Khalas Dates during Storage under Ambient and Refrigerated Conditions. Food Chem. 2021, 352, 131–168. [Google Scholar]
- Mohamed, S.A.; El-Zayat, S.A.; El-Morsy, M.M. Postharvest Quality Changes of Barhi Dates during Cold Storage. Postharvest Biol. Technol. 2020, 168, 111–181. [Google Scholar]
- Garcia, L.M.; Patel, R.K. Utilizing Polynomial Models to Assess the Relationship Between Sensory Evaluation and Physicochemical Characteristics in Food Products. Food Chem. 2021, 98, 732–7445. [Google Scholar]
- Smith, J.K.; Johnson, A.B. Integration of Polynomial Models for Descriptive Sensory Evaluation and Physicochemical Properties in Food Product Analysis. J. Food Sci. 2020, 57, 210–225. [Google Scholar]
- Burnham, K.P.; Anderson, D.R. (Eds.) Model Selection and Multimodel Inference; Springer: New York, NY, USA, 2004; ISBN 978-0-387-95364-9. [Google Scholar]
- Schabenberger, O.; Pierce, F.J. Contemporary Statistical Models for the Plant and Soil Sciences; CRC Press: Boca Raton, FL, USA, 2001; ISBN 978-0-429-12065-7. [Google Scholar]
- Huang, Y.; Kangas, L.J.; Rasco, B.A. Applications of Artificial Neural Networks (ANNs) in Food Science. Crit. Rev. Food Sci. Nutr. 2007, 47, 113–126. [Google Scholar] [CrossRef]
- Ivorra, B.; Fraguela, A.; Infante, J.A.; Rey, J.M.; Smith, N.; Olmo, A.M.R.D. Mathematical Modelling in Food Engineering. In Numerical Simulation in Physics and Engineering; Universidad de la Coruña: La Coruña, Spain, 2010; pp. 283–301. [Google Scholar] [CrossRef]
- Kumar, M.; Vatsa, S.; Madhumita, M.; Prabhakar, P.K. Mathematical Modeling of Food Processing Operations: A Basic Understanding and Overview. Turk. J. Agric. Eng. Res. (TURKAGER) 2021, 2, 472–492. [Google Scholar] [CrossRef]
- Leena, N.K. Predicting Food Shelf Life: Advances in Modeling and Predictive Analytics. Afr. J. Food Sci. Technol. 2024, 15, 1–2. [Google Scholar]
- Lončar, B.; Pezo, L. Mathematical Modeling Approach and Simulation in Food Drying Applications. Foods 2024, 13, 384. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Q.; Chen, Y.; Guo, L.; Fei, T.; Qi, K. Estimating Soil Organic Carbon of Cropland Soil at Different Levels of Soil Moisture Using VIS-NIR Spectroscopy. Remote Sens. 2016, 8, 755. [Google Scholar] [CrossRef]
- Bakeev, K.A. (Ed.) Process Analytical Technology: Spectroscopic Tools and Implemented Strategies for the Chemical and Pharmaceutical Industries, 2nd ed.; Wiley: Chichester, UK, 2010; ISBN 978-0-470-72207-7. [Google Scholar]
- Bhargava, R.; Levin, I. (Eds.) Spectrochemical Analysis Using Infrared Multichannel Detectors; Blackwell Pub: Ames, IA, USA, 2005; ISBN 978-1-4051-2504-8. [Google Scholar]
- Carlini, P.; Massantini, R.; Mencarelli, F. Vis-NIR Measurement of Soluble Solids in Cherry and Apricot by PLS Regression and Wavelength Selection. J. Agric. Food Chem. 2000, 48, 5236–5242. [Google Scholar] [CrossRef]
- Peirs, A.; Schenk, A.; Nicolai, B.M. Effect of Natural Variability among Apples on the Accuracy of VIS-NIR Calibration Models for Optimal Harvest Date Predictions. Postharvest Biol. Technol. 2005, 35, 1–13. [Google Scholar] [CrossRef]
- Schaare, P.N.; Fraser, D.G. Comparison of Reflectance, Interactance and Transmission Modes of Visible-near Infrared Spectroscopy for Measuring Internal Properties of Kiwifruit (Actinidia chinensis). Postharvest Biol. Technol. 2000, 20, 175–184. [Google Scholar] [CrossRef]
- Al-Rawahi, A.S.; Rahman, M.S.; Guizani, N.; Essa, M.M. Chemical Composition, Water Sorption Isotherm, and Phenolic Contents in Fresh and Dried Pomegranate Peels. Dry. Technol. 2013, 31, 257–263. [Google Scholar] [CrossRef]
- Altinkok, N.; Koker, R. Use of Artificial Neural Network for Prediction of Physical Properties and Tensile Strengths in Particle Reinforced Alüminum Matrix Composites. J. Mater. Sci. 2005, 40, 1767–1770. [Google Scholar] [CrossRef]
- Funes, E.; Allouche, Y.; Beltrán, G.; Jiménez, A. A Review: Artificial Neural Networks as Tool for Control Food Industry Process. J. Sens. Technol. 2015, 5, 28–43. [Google Scholar] [CrossRef]
- Gorbachev, V.; Nikitina, M.; Velina, D.; Mutallibzoda, S.; Nosov, V.; Korneva, G.; Terekhova, A.; Artemova, E.; Khashir, B.; Sokolov, I.; et al. Artificial Neural Networks for Predicting Food Antiradical Potential. Appl. Sci. 2022, 12, 6290. [Google Scholar] [CrossRef]
- Goyal, S. Artificial Neural Networks (ANNs) in Food Science—A Review. Int. J. Sci. World 2013, 1, 19–28. [Google Scholar] [CrossRef]
- Inyang, U.E. Artificial Neural Network and Their Applications in Food Materials: A Review. Eng. Technol. J. 2022, 7, 235–1248. [Google Scholar] [CrossRef]
- Elakiya, N.; Keerthana, G. Application of Artificial Neural Networks in Soil Science Research. Arch. Curr. Res. Int. 2024, 24, 1–15. [Google Scholar] [CrossRef]
- Przybył, K.; Gawałek, J.; Koszela, K. Application of Artificial Neural Network for the Quality-Based Classification of Spray-Dried Rhubarb Juice Powders. J. Food Sci. Technol. 2023, 60, 809–819. [Google Scholar] [CrossRef]
- Burns, D.A.; Ciurczak, E.W. (Eds.) Handbook of Near-Infrared Analysis; CRC Press: Boca Raton, FL, USA, 2007; ISBN 978-0-429-12301-6. [Google Scholar]
- Cen, H.; He, Y. Theory and Application of near Infrared Reflectance Spectroscopy in Determination of Food Quality. Trends Food Sci. Technol. 2007, 18, 72–83. [Google Scholar] [CrossRef]
- Torkashvand, A.M.; Ahmadi, A.; Nikravesh, N.L. Prediction of Kiwifruit Firmness Using Fruit Mineral Nutrient Concentration by Artificial Neural Network (ANN) and Multiple Linear Regressions (MLR). J. Integr. Agric. 2017, 16, 1634–1644. [Google Scholar] [CrossRef]
- Saeys, W.; Mouazen, A.M.; Ramon, H. Potential for On-Site and on-Line Analysis of Pig Manure Using Visible and Near Infrared Reflectance Spectroscopy. Biosyst. Eng. 2005, 91, 393–402. [Google Scholar] [CrossRef]
- Huang, H.; Yu, H.; Xu, H.; Ying, Y. Near Infrared Spectroscopy for on/in-Line Monitoring of Quality in Foods and Beverages: A Review. J. Food Eng. 2008, 87, 303–313. [Google Scholar] [CrossRef]
- Shrestha, S.; Deleuran, L.C. Prediction of Moisture Content in Tomato Using Near Infrared Spectroscopy and Artificial Neural Network. Comput. Electron. Agric. 2014, 103, 72–77. [Google Scholar]
- Liu, Y.; Sun, X.; Zhang, H.; Zhang, L. Use of VIS-NIR Spectroscopy for Predicting Chemical and Sensory Attributes of Fruits: A Review. Postharvest Biol. Technol. 2016, 118, 60–72. [Google Scholar]
- Nicolaï, B.M.; Beullens, K.; Bobelyn, E.; Peirs, A.; Saeys, W.; Theron, K.I.; Lammertyn, J. Nondestructive Measurement of Fruit and Vegetable Quality by Means of NIR Spectroscopy: A Review. Postharvest Biol. Technol. 2007, 46, 99–118. [Google Scholar] [CrossRef]
- Aleid, S.M.; Al-Khayri, J.M.; Al-Bahrany, A.M. Date palm Status and Perspective in Saudi Arabia. In Date palm Genetic Resources and Utilization; Al-Khayri, J.M., Jain, S.M., Johnson, D.V., Eds.; Springer: Dordrecht, The Netherlands, 2015; pp. 49–95. ISBN 978-94-017-9706-1. [Google Scholar]
- Al-Abdoulhadi, I.A. Assessing Fruit Characteristics to Standardize Quality Norms in Date Cultivars of Saudi Arabia. Indian J. Sci. Technol. 2011, 4, 1262–1266. [Google Scholar] [CrossRef]
- Ahmed, J.; Ramaswamy, H.S.; Ayad, A. Effect of Temperature on Rheological Properties of Date Paste. J. Food Eng. 2013, 60, 359–366. [Google Scholar]
- Al Jasser, S.M. Effect of storage temperatures on microbial load of some dates palm fruit sold in Saudi Arabia market. J. Food Qual. Afr. J. Food Sci. 2010, 4, 359–363. [Google Scholar]
- Masamba, M. Impact of Food Packaging Materials on the Shelf-Life and Quality of Packaged Food Products. Int. J. Food Sci. 2024, 5, 15–28. [Google Scholar] [CrossRef]
- Singh, J.; Gupta, R.; Kumar, S. Modified Atmosphere Packaging in Post-Harvest Technology. Crit. Rev. Food Sci. Nutr. 2021, 61, 1396–1415. [Google Scholar]
- Baltazari, A.; Mtui, H.D.; Mwatawala, M.W.; Chove, L.M.; Msogoya, T.; Samwel, J.; Subramanian, J. Effects of Storage Conditions, Storage Duration and Post-Harvest Treatments on Nutritional and Sensory Quality of Orange (Citrus sinensis (L.) Osbeck) Fruits. Int. J. Fruit Sci. 2020, 20, 737–749. [Google Scholar] [CrossRef]
- Dixit, D.; Maurya, D.; Kishore, S.; Singh, S.; Bhati, D.; Jatav, V. Influence of Different Packaging Materials on Physical and Physiological Attributes of Tomato Fruits during Storage. Ann. Agri Bio Res. 2023, 28, 278–282. [Google Scholar]
- Al-Farsi, M.A.; Lee, C.Y. Nutritional and Functional Properties of Dates: A Review. Crit. Rev. Food Sci. Nutr. 2008, 48, 877–887. [Google Scholar] [CrossRef] [PubMed]
- Habib, H.M.; Ibrahim, W.H. Nutritional Quality of 18 Date Fruit Varieties. Int. J. Food Sci. Nutr. 2011, 62, 544–551. [Google Scholar] [CrossRef]
- Abdelzaher Radwan, E.; El-Salhy, A.-F.; Hussein, H.M.; Sayed, M.K. Effect of Different Packing Types on Storage and Quality of Saidy Dates. New Val. J. Agric. Sci. 2023, 3, 858–868. [Google Scholar] [CrossRef]
- Azene, M.; Workneh, T.S.; Woldetsadik, K. Effect of Packaging Materials and Storage Environment on Postharvest Quality of Papaya Fruit. J. Food Sci. Technol. 2014, 51, 1041–1055. [Google Scholar] [CrossRef]
- Mahajan, B.V.C.; Dhillon, W.S.; Kumar, M.; Singh, B. Effect of Different Packaging Films on Shelf Life and Quality of Peach under Super and Ordinary Market Conditions. J. Food Sci. Technol. 2014, 52, 3756–3762. [Google Scholar] [CrossRef] [PubMed]
- Mortazavi, S.M.H.; Arzani, K.; Barzegar, M. Effect of Vacuum and Modified Atmosphere Packaging on the Postharvest Quality and Shelf Life of Date Fruits in Khalal Stage. Acta Hortic. 2007, 736, 471–477. [Google Scholar] [CrossRef]
- Ramadan, B.R.; Magda, A.A.; Zeinab, M. Influence of Packaging and Cold Storage Conditions on the Physio-Chemical Properties of Barhi Date Fruits. Assiut J. Agric. Sci. 2020, 51, 44. [Google Scholar] [CrossRef]
- Al-Hooti, S.; Sidhu, J.S.; Qabazard, H. Physicochemical Characteristics of Five Date Fruit Cultivars Grown in the United Arab Emirates. Plant Food Hum. Nutr. 1997, 50, 101–113. [Google Scholar] [CrossRef]
- Yahia, E.M.; Basyuni, M.; Abd-Rabou, S. Quality Changes of Dates during Storage—A review. Agric. Rev. 2017, 38, 195–206. [Google Scholar]
- Al-Bulushi, I.M.; Al-Habsi, N.A.; Al-Busaidi, M.A. Microbial Quality of Date Fruits in Oman. J. Food Qual. 2013, 36, 121–126. [Google Scholar]
- Al-Rimawi, F.; Rababah, T.; Rababah, A.; Ereifej, K. Microbiological Quality and Safety of Palestinian Dates. Food Sci. Nutr. 2018, 6, 1140–1145. [Google Scholar]
- Al-Said, F.A.; Othman, A.A.; Al-Zahrani, H.S. Microbiological Safety of Dates in Saudi Arabia: A Study of Retail Samples of Various Types of Dates. J. Food Prot. 2015, 78, 353–357. [Google Scholar]
- Al-Hamdan, A.; Ibrahim, M.; El Zubeir, I.E.M. Microbiological Safety of Dates Sold in the United Arab Emirates. Food Sci. Nutr. 2019, 7, 268–276. [Google Scholar]
- Al-Shaikh, K.S.; Al-Shammari, T.A.; Al-Osaimi, H.A. Microbiological Evaluation of Date Fruits under Different Storage Conditions. Saudi J. Biol. Sci. 2017, 24, 1553–1558. [Google Scholar]
- Al-Sheikh, H.M.; El-Messery, T.M.; Al-Ali, S.K. Microbiological Quality and Safety of Date palm Fruits Sold in Qatar. J. Appl. Sci. 2016, 16, 92–99. [Google Scholar]
- Al-Yahyai, R.; Khan, M.M. Date palm Status and Perspective in Oman. In Date palm Genetic Resources and Utilization; Al-Khayri, J.M., Jain, S.M., Johnson, D.V., Eds.; Springer: Dordrecht, The Netherlands, 2015; pp. 207–240. ISBN 978-94-017-9706-1. [Google Scholar]
- Doyle, M.P.; Beuchat, L.R. (Eds.) Food Microbiology: Fundamentals and Frontiers, 3rd ed.; ASM Press: Washington, DC, USA, 2007; ISBN 978-1-55581-407-6. [Google Scholar]
- Elamshity, M.G. Unveiling Microbial Dynamics in Diverse Date Varieties Stored: Influence of Moisture, Packaging, and Temperature Fluctuations. Food Packag. Shelf Life, 2025; under review for publication. [Google Scholar]
- Hamad, S.H. Microbial Spoilage of Date Rutab Collected from the Markets of Al-Hofuf City in the Kingdom of Saudi Arabia. J. Food Prot. 2008, 71, 1406–1411. [Google Scholar] [CrossRef] [PubMed]
- Altman, D.; Machin, D.; Bryant, T.; Gardner, M.J. Statistics with Confidence: Confidence Intervals and Statistical Guidelines, 2nd ed.; Wiley: Hoboken, NJ, USA, 2013; ISBN 978-0-7279-1375-3. [Google Scholar]
- Montgomery, D.C. Design and Analysis of Experiments, 9th ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2017; ISBN 978-1-119-11347-8. [Google Scholar]
- Field, A. Discovering Statistics Using IBM SPSS Statistics: And Sex and Drugs and Rock “n” Roll, 5th ed.; MobileStudy; Sage: Los Angeles, CA, USA; London, UK; New Delhi, India; Singapore; Washington, DC, USA, 2013; ISBN 978-1-4462-4918-5. [Google Scholar]
- Abbas, Q.; Al-Habsi, N.; Iqbal, R.F. A Review of Postharvest Handling and Storage Technologies for Date Fruit. Sci. Hortic. 2020, 272, 109506. [Google Scholar] [CrossRef]
- Jayasena, V.; Cameron, I. Non-Destructive Spectroscopic Techniques for Quality Evaluation of Fruits and Vegetables. Food Chem. 2015, 179, 85–90. [Google Scholar]
- ElMasry, G.; Nakauchi, S. Image Analysis and Hyperspectral Imaging for Non-Destructive Evaluation of Agricultural Products. J. Food Eng. 2013, 118, 10–25. [Google Scholar]
- Xu, L.; Du, P.; Zhang, B. Enhancing Fruit Quality Assessment Using Fusion of VIS–NIR Spectroscopy and Machine Learning. Sensors 2022, 22, 2267. [Google Scholar]


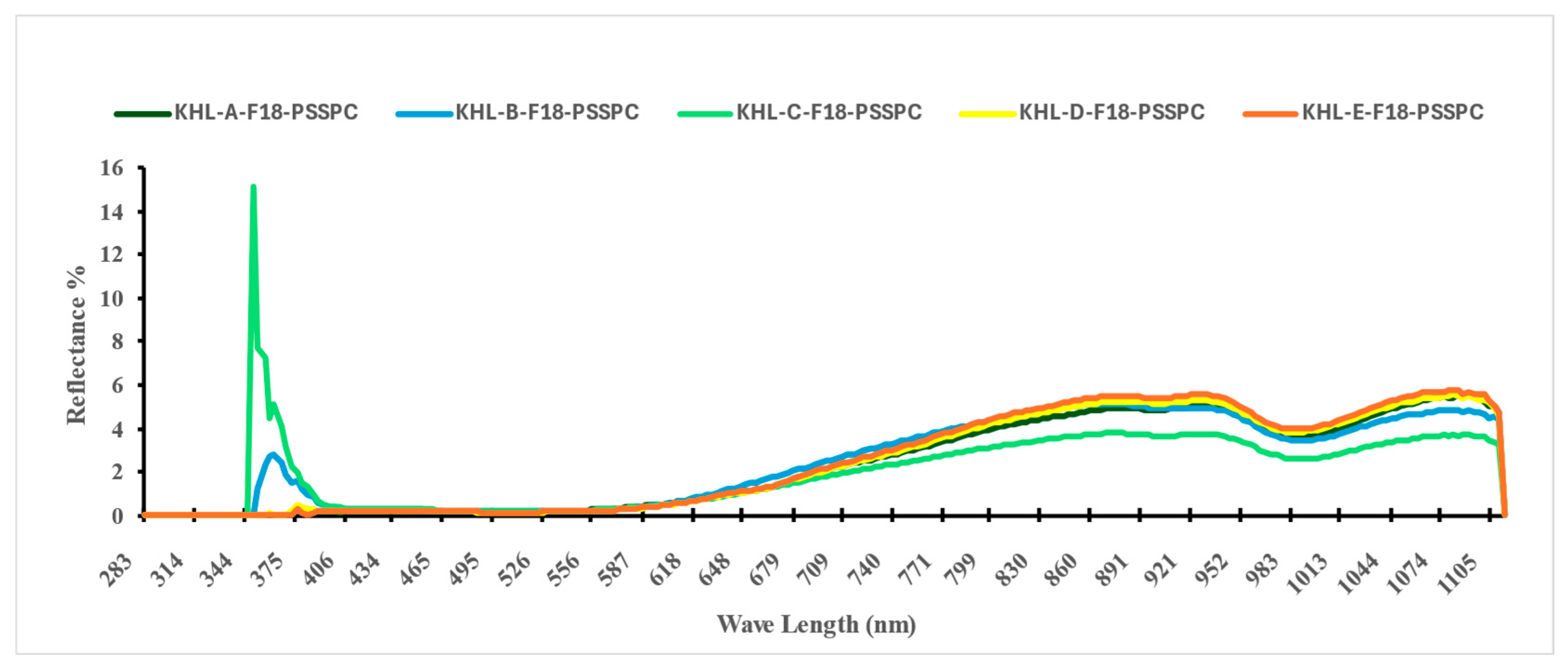
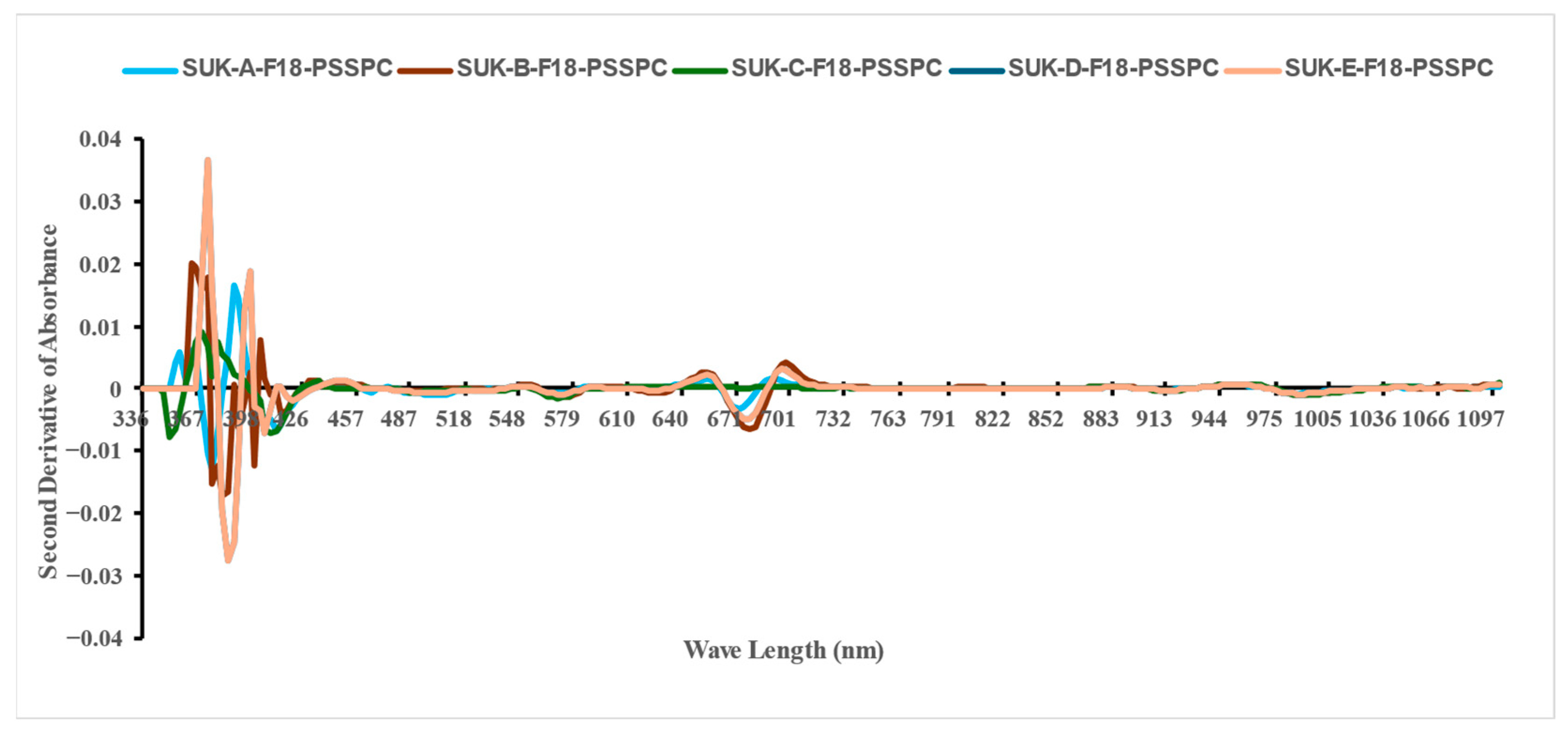
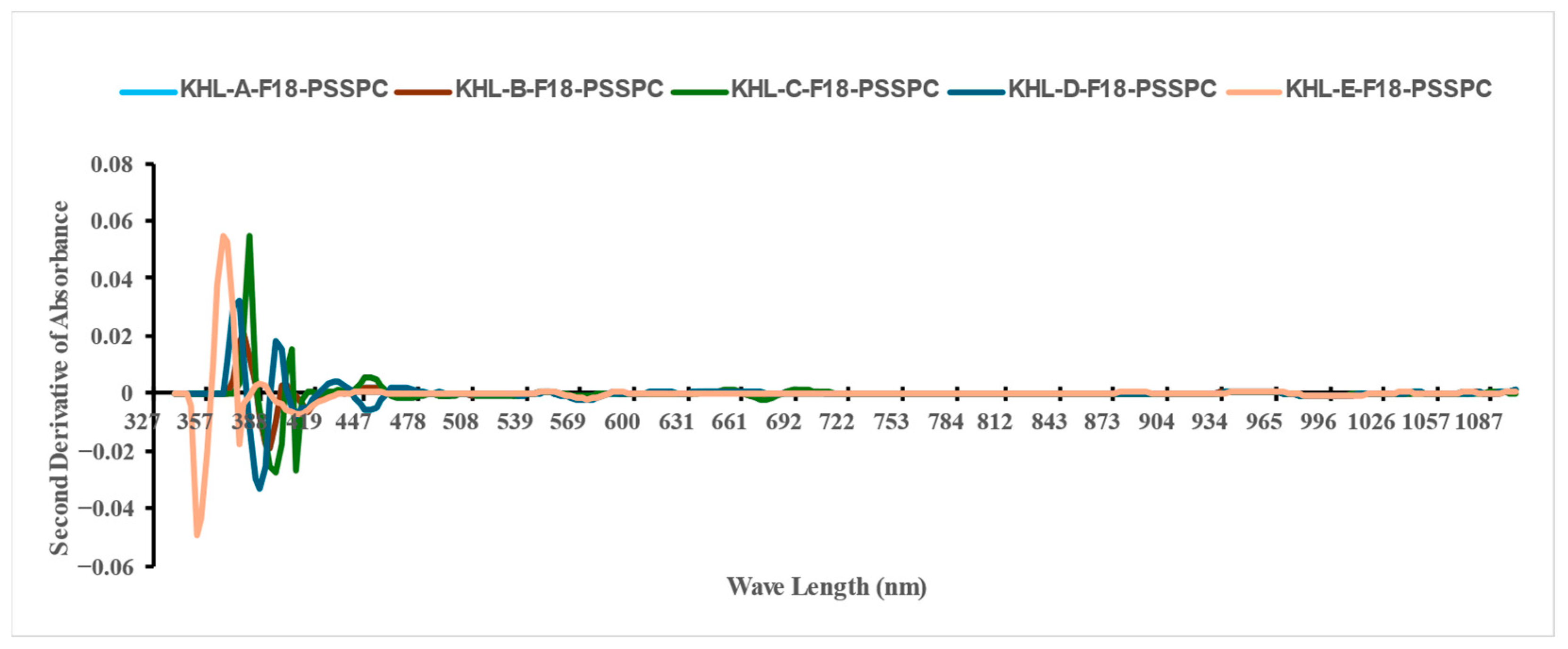
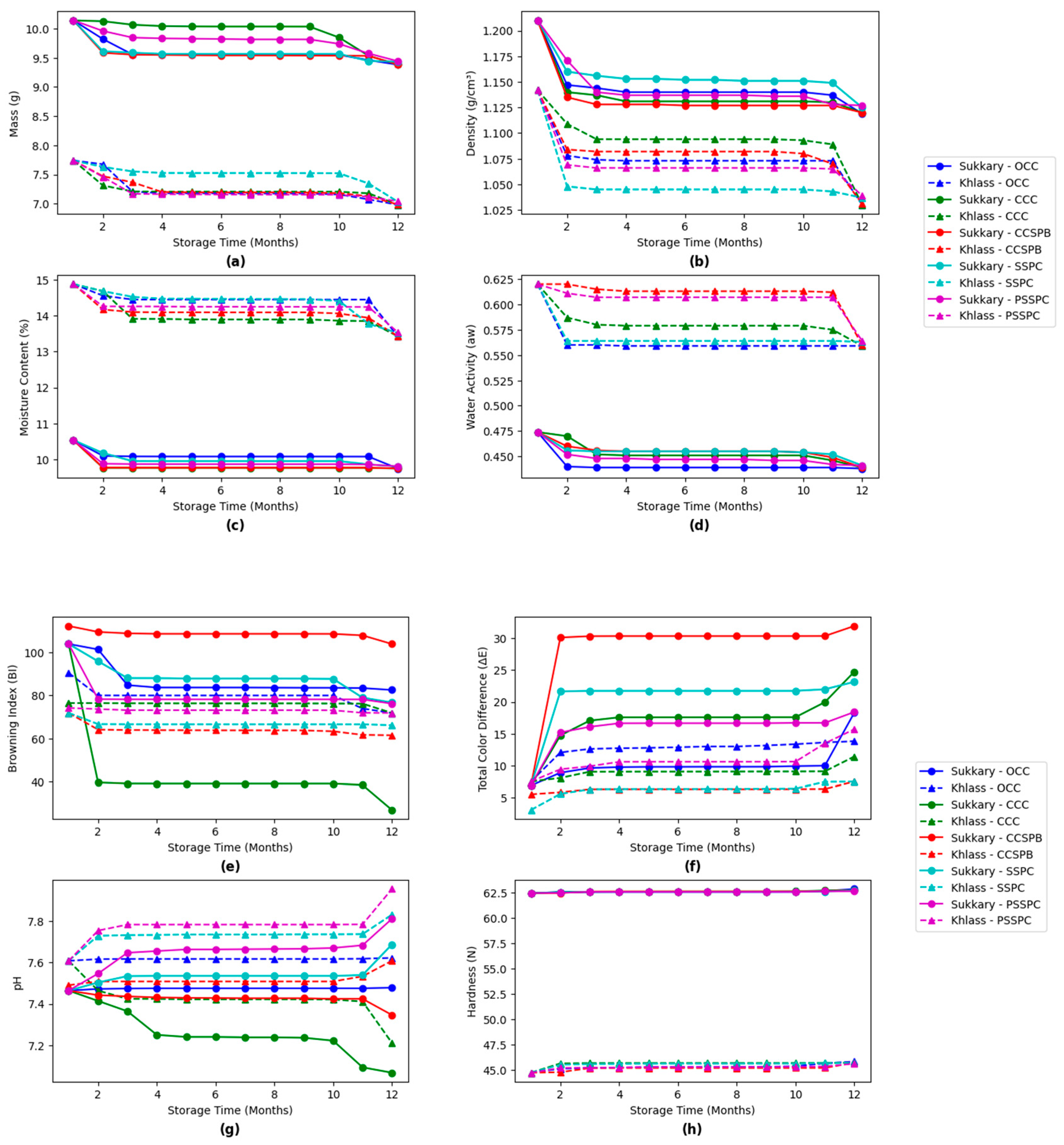
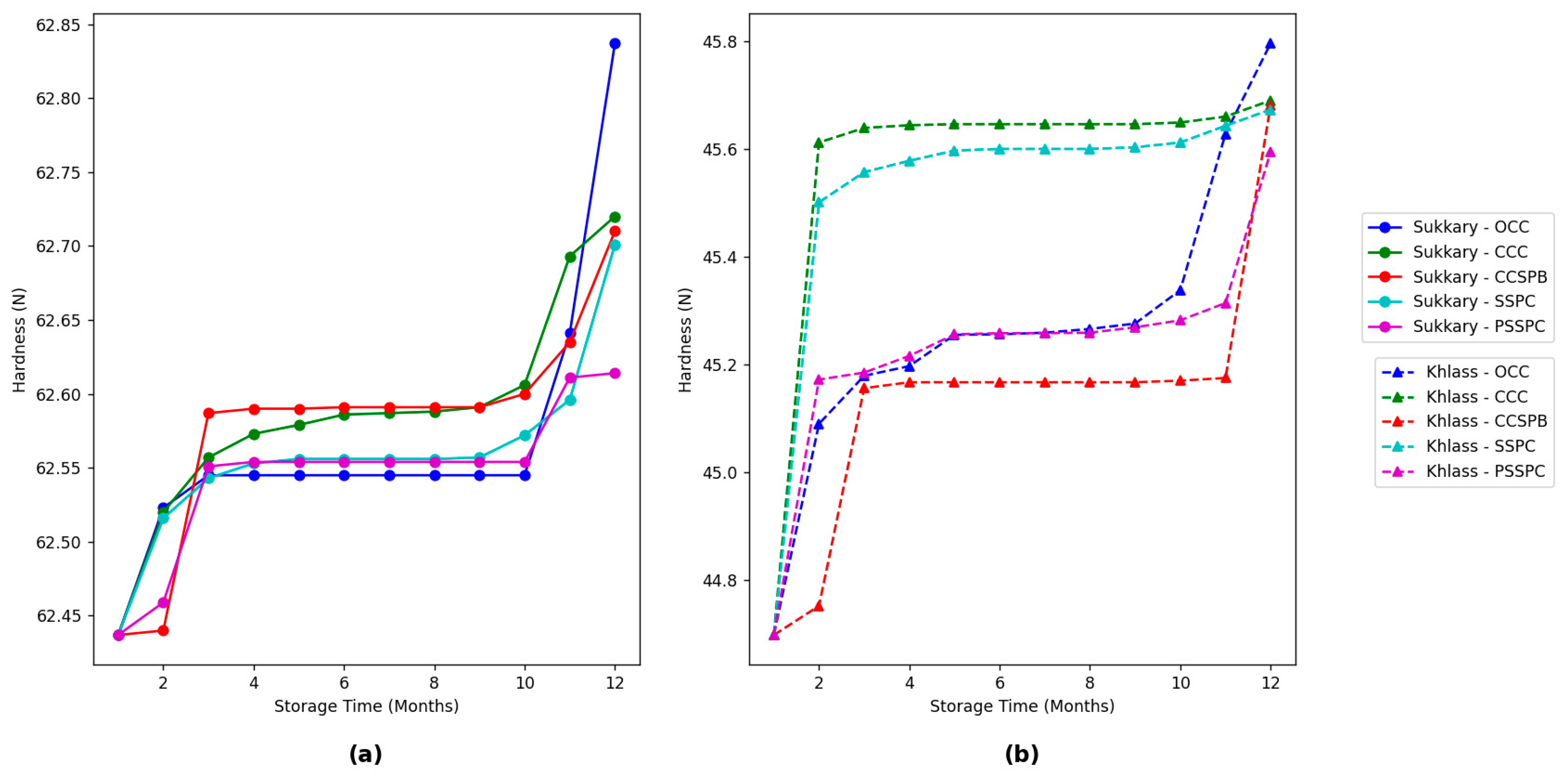
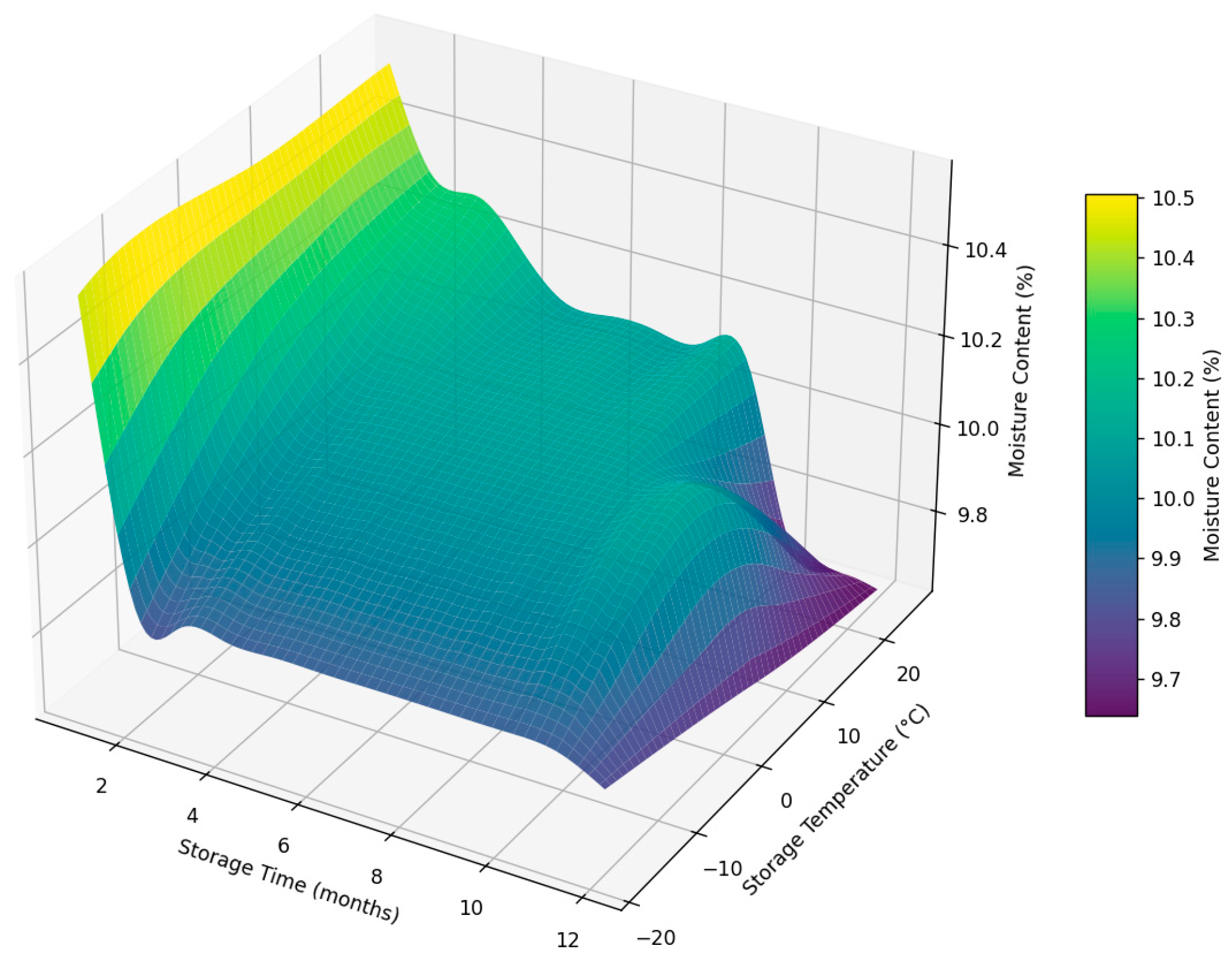
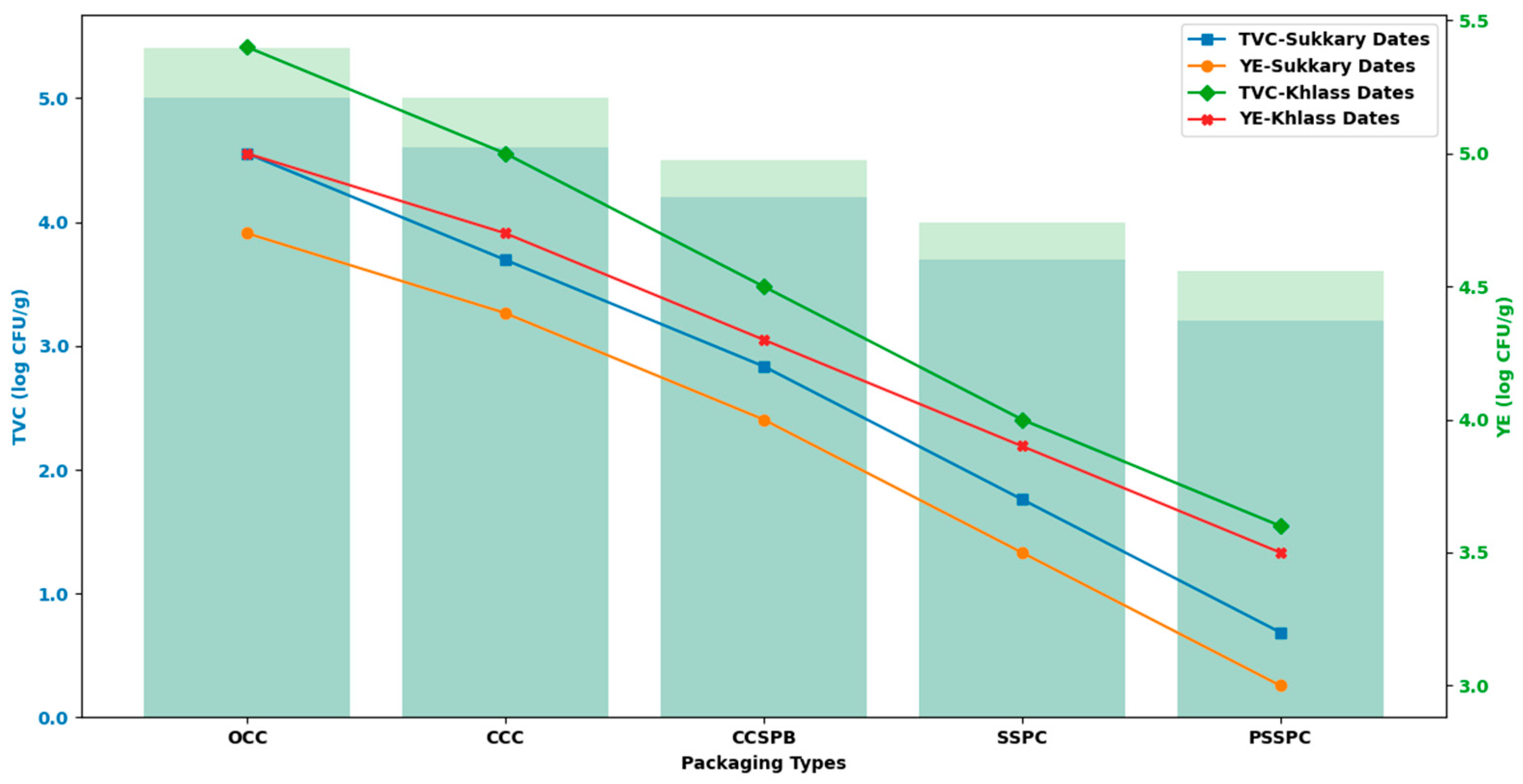
| Class | Moisture Content (db.) (%) | |
|---|---|---|
| Sukkary | Khlass | |
| Control | 4.357 f ± 0.015 | 6.235 f ± 0.021 |
| Group A | 10.531 e ± 0.035 | 14.877 e ± 0.023 |
| Group B | 13.181 d ± 0.011 | 21.781 d ± 0.017 |
| Group C | 16.606 c ± 0.014 | 26.807 c ± 0.036 |
| Group D | 20.489 b ± 0.054 | 32.062 b ± 0.046 |
| Group E | 27.343 a ± 0.016 | 38.697 a ± 0.025 |
| Packaging Materials | Construction /Polymer | Nominal Thickness/ Grammage | WVTR (g·m−2·day−1) @38 °C/90% RH | OTR (cm3·m−2·day−1·bar−1) @23 °C/0% RH | Material Code | Lot/Batch | * CoC/DoC (ID) | Food-Contact Compliance [46] |
|---|---|---|---|---|---|---|---|---|
| OCC | Corrugated Kraft paperboard | [440–600 g·m−2] | 300–1500 | >10,000 (not a gas barrier) | [Carton-200/112/200 B-flute] | [LOT 23B-4719] | [CoC #A-2024-118] | FDA 21 CFR (paper and board guidance), EU 1935/2004 (general) [47] |
| CCC | Paperboard with lid/overwrapping | [440–600 g·m−2] | 250–1200 | >10,000 | [Carton-200/125/200 E-flute] | [LOT 23E-5120] | [CoC #A-2024-119] | FDA 21 CFR; EU 1935/2004 [47] |
| CCSPB | Paperboard + heat-sealable LDPE liner (≈50–70 µm) | LDPE: [50–70 µm] | 5–15 | 2000–8000 | [LDPE-60 µm grade] | [LOT 24-05-18-07] | [DoC #B-10/2011-221] | FDA 21 CFR 177.1520; EU 1935/2004; EU 10/2011 (plastics) [48] |
| SSPC | PET or PP tub (wall ≈ 0.4–0.8 mm) | PET/PP: [0.5–0.8 mm] | 1–5 | 2–20 (PET lower; PP higher) | [PET 700 µm grade] | [LOT KSA-240713-02] | [CoC #C-177.1630-045] | FDA 21 CFR 177.1630 (PET)/177.1520 (PP); EU 1935/2004; EU 10/2011 [48] |
| PSSPC | Multilayer PET/EVOH/PE (co-extruded) | Wall: [0.5–0.8 mm]; EVOH core [~3–10%] | 0.1–0.8 | 0.1–1.0 | [PET/EVOH/PE 650 µm spec] | [LOT EVOH-241009-17] | [DoC #D-10/2011-508] | FDA 21 CFR; EU 1935/2004; EU 10/2011 (incl. specific migration) [49] |
| Analyte | Method and Matrix | Acceptance Limit | Investigate/Hold | Reject (Do Not Serve) |
|---|---|---|---|---|
| Total Viable Count (TVC) | ISO 4833-1 (aerobic plate count, 30 °C); dates, ready-to-eat [73] | ≤5.0 log CFU g−1 (≤105) | 5.0–6.0 log CFU g−1 | >6.0 log CFU g−1 |
| Yeast Enumeration (YE) | ISO 21527-2 (yeasts and molds for low-aw foods); dates, ready-to-eat [74] | ≤4.0 log CFU g−1 (≤104) | 4.0–5.0 log CFU g−1 | >5.0 log CFU g−1 |
| Cultivar | Qi = at2 − bt + c | R2 | ||
|---|---|---|---|---|
| A | b | c | ||
| Sukkary | −0.0007 | 0.0102 | 0.9719 | 0.988 |
| Khlass | 0.0021 | 0.0521 | 0.9554 | 0.984 |
| Cultivar | Model Type | Correlation Coefficient (R2) | Akaike Information Criterion (AIC) | Analysis of Variance (ANOVA) p-Value |
|---|---|---|---|---|
| Sukkary | Linear | 0.976 | 120.3 | - |
| Sukkary | Quadratic | 0.988 | 110.5 | <0.05 |
| Khlass | Linear | 0.927 | 135.2 | - |
| Khlass | Quadratic | 0.984 | 119.8 | <0.05 |
| Cultivar | Parameter | Calibration | Cross-Validation | ||
|---|---|---|---|---|---|
| R2 | RMSEC | R2 | RMSECV | ||
| Sukkary | M.C.S | 0.834 | 0.833 | 0.976 | 0.756 |
| awS | 0.830 | 0.819 | 0.972 | 0.742 | |
| TSSS | 0.832 | 0.822 | 0.974 | 0.745 | |
| BIS | 0.821 | 0.789 | 0.963 | 0.712 | |
| ΔES | 0.824 | 0.799 | 0.966 | 0.722 | |
| pHS | 0.814 | 0.812 | 0.956 | 0.735 | |
| HardnessS | 0.815 | 0.853 | 0.957 | 0.776 | |
| QiS | 0.790 | 0.320 | 0.932 | 0.243 | |
| Khlass | M.C.K | 0.817 | 0.834 | 0.959 | 0.757 |
| awK | 0.813 | 0.820 | 0.955 | 0.743 | |
| TSSK | 0.815 | 0.823 | 0.957 | 0.746 | |
| BIK | 0.804 | 0.790 | 0.946 | 0.713 | |
| ΔEK | 0.807 | 0.800 | 0.949 | 0.723 | |
| pHK | 0.797 | 0.813 | 0.939 | 0.736 | |
| HardnessK | 0.798 | 0.854 | 0.940 | 0.777 | |
| QiK | 0.773 | 0.321 | 0.915 | 0.244 | |
| Cultivar | Parameter | Calibration | Cross-Validation | ||
|---|---|---|---|---|---|
| R2 | RMSEC | R2 | RMSECV | ||
| Sukkary | M.C.S | 0.988 | 0.846 | 0.986 | 0.816 |
| awS | 0.984 | 0.832 | 0.982 | 0.802 | |
| TSSS | 0.986 | 0.835 | 0.984 | 0.805 | |
| BIS | 0.975 | 0.802 | 0.973 | 0.772 | |
| ΔES | 0.978 | 0.812 | 0.976 | 0.782 | |
| pHS | 0.968 | 0.825 | 0.966 | 0.795 | |
| HardnessS | 0.969 | 0.866 | 0.967 | 0.836 | |
| QiS | 0.944 | 0.333 | 0.942 | 0.303 | |
| Khlass | M.C.K | 0.971 | 0.847 | 0.969 | 0.817 |
| awK | 0.967 | 0.833 | 0.965 | 0.803 | |
| TSSK | 0.969 | 0.836 | 0.967 | 0.806 | |
| BIK | 0.958 | 0.803 | 0.956 | 0.773 | |
| ΔEK | 0.961 | 0.813 | 0.959 | 0.783 | |
| pHK | 0.951 | 0.826 | 0.949 | 0.796 | |
| HardnessK | 0.952 | 0.867 | 0.950 | 0.837 | |
| QiK | 0.927 | 0.334 | 0.925 | 0.304 | |
| Model | Cultivar | RMSEP | REP (%) | RER |
|---|---|---|---|---|
| PLSR | Sukkary | 0.256 | 8.12 | 10.6 |
| PLSR | Khlass | 0.266 | 8.75 | 9.8 |
| PLSR | Combined | 0.261 | 8.44 | 10.2 |
| ANN | Sukkary | 0.304 | 6.22 | 13.2 |
| ANN | Khlass | 0.317 | 6.85 | 12.7 |
| ANN | Combined | 0.310 | 6.54 | 13.0 |
| Study/Reference | Product/Model | RMSEP | REP (%) | RER | Notes |
|---|---|---|---|---|---|
| Current study (Sukkary, ANNs) | Date fruits/ANN | 0.304 | 6.22 | 13.2 | High precision, strong RER ≥ 10, low REP < 7% indicates excellent predictability |
| Current study (Khlass, PLSR) | Date fruits/PLSR | 0.266 | 8.75 | 9.8 | Moderate performance; just below the ideal RER threshold, acceptable error levels |
| Huang et al. (2008) [156] | Firmness of apples/PLSR | 1.24 | ~9–11 | ~8.5 | Moderate performance, good for bulk grading |
| Carlini et al. (2000) [141] | Soluble solids in apricot/PLSR | 0.68 | ~10 | ~9.5 | An early benchmark in VIS-NIR modeling |
| Zhang et al. (2021) [23] | Moisture in green tea/NIR | 0.235 | ~7 | 12.1 | High performance using NIR + ANNs in controlled processing settings |
| Nicolaï et al. (2007) [159] | Internal quality of peaches | 0.290 | 8–10 | ~10 | Review: High variability in fruit matrices affects the general RER |
| Goyal (2013) [148] | ANN in various fruits | – | 6–9 | >10 | General trend: the ANN outperforms linear models, especially for nonlinear degradation |
| Moisture Content | Duration | TVC | TVC | TVC | YE | YE | YE |
|---|---|---|---|---|---|---|---|
| (log CFU/g) | (log CFU/g) | (log CFU/g) | (log CFU/g) | (log CFU/g) | (log CFU/g) | ||
| (%) | (Months) | −18 °C | +5 °C | +25 °C | −18 °C | +5 °C | +25 °C |
| Control (4.357) | 3 | 1.1 d ± 0.21 | 2.0 d ± 0.02 | 3.5 d ± 0.03 | 0.9 d ± 0.01 | 1.8 d ± 0.01 | 3.2 d ± 0.02 |
| 6 | 1.2 c ± 0.11 | 2.2 c ± 0.02 | 3.8 c ± 0.03 | 1.0 c ± 0.01 | 2.0 c ± 0.02 | 3.4 c ± 0.03 | |
| 9 | 1.3 b ± 0.21 | 2.4 b ± 0.02 | 4.0 b ± 0.13 | 1.1 b ± 0.01 | 2.2 b ± 0.02 | 3.6 b ± 0.03 | |
| 12 | 1.4 a ± 0.11 | 2.6 a ± 0.02 | 4.3 a ± 0.13 | 1.2 a ± 0.01 | 2.4 a ± 0.02 | 3.8 a ± 0.03 | |
| Group A (10.531) | 3 | 1.6 d ± 0.31 | 3.0 d ± 0.02 | 4.7 d ± 0.13 | 1.3 d ± 0.01 | 2.6 d ± 0.02 | 4.2 d ± 0.03 |
| 6 | 1.8 c ± 0.21 | 3.3 c ± 0.23 | 5.0 c ± 0.04 | 1.5 c ± 0.01 | 2.9 c ± 0.03 | 4.5 c ± 0.04 | |
| 9 | 2.0 b ± 0.12 | 3.6 b ± 0.13 | 5.3 b ± 0.04 | 1.7 b ± 0.02 | 3.2 b ± 0.13 | 4.8 b ± 0.04 | |
| 12 | 2.2 a ± 0.42 | 4.0 a ± 0.13 | 5.6 a ± 0.04 | 1.9 a ± 0.02 | 3.5 a ± 0.13 | 5.1 a ± 0.04 | |
| Group B (13.181) | 3 | 1.9 d ± 0.22 | 3.4 d ± 0.23 | 5.2 d ± 0.04 | 1.6 d ± 0.12 | 3.1 d ± 0.03 | 4.7 d ± 0.24 |
| 6 | 2.1 c ± 0.42 | 3.7 c ± 0.13 | 5.5 c ± 0.04 | 1.8 c ± 0.02 | 3.4 c ± 0.03 | 5.0 c ± 0.14 | |
| 9 | 2.4 b ± 0.42 | 4.0 b ± 0.03 | 5.8 b ± 0.14 | 2.0 b ± 0.02 | 3.7 b ± 0.03 | 5.3 b ± 0.14 | |
| 12 | 2.6 a ± 0.12 | 4.4 a ± 0.13 | 6.2 a ± 0.14 | 2.2 a ± 0.02 | 4.0 a ± 0.13 | 5.6 a ± 0.14 | |
| Group C (16.606) | 3 | 2.3 d ± 0.02 | 4.2 d ± 0.13 | 5.9 d ± 0.24 | 1.9 d ± 0.02 | 3.8 d ± 0.03 | 5.4 d ± 0.24 |
| 6 | 2.5 c ± 0.02 | 4.5 c ± 0.13 | 6.2 c ± 0.44 | 2.1 c ± 0.02 | 4.1 c ± 0.13 | 5.7 c ± 0.24 | |
| 9 | 2.8 b ± 0.02 | 4.9 b ± 0.13 | 6.5 b ± 0.24 | 2.4 b ± 0.02 | 4.4 b ± 0.03 | 6.0 b ± 0.24 | |
| 12 | 3.0 a ± 0.12 | 5.3 a ± 0.13 | 6.9 a ± 0.44 | 2.6 a ± 0.02 | 4.8 a ± 0.03 | 6.4 a ± 0.34 | |
| Group D (20.489) | 3 | 2.7 d ± 0.22 | 4.8 d ± 0.13 | 6.6 d ± 0.44 | 2.3 d ± 0.02 | 4.3 d ± 0.03 | 5.9 d ± 0.24 |
| 6 | 3.0 c ± 0.12 | 5.1 c ± 0.23 | 7.0 c ± 0.44 | 2.5 c ± 0.02 | 4.6 c ± 0.03 | 6.2 c ± 0.24 | |
| 9 | 3.4 b ± 0.32 | 5.5 b ± 0.13 | 7.4 b ± 0.74 | 2.8 b ± 0.02 | 5.0 b ± 0.03 | 6.6 b ± 0.24 | |
| 12 | 3.7 a ± 0.12 | 6.0 a ± 0.14 | 7.9 a ± 0.25 | 3.1 a ± 0.02 | 5.4 a ± 0.03 | 7.1 a ± 0.14 | |
| Group E (27.343) | 3 | 3.6 d ± 0.32 | 5.8 d ± 0.04 | 7.6 d ± 0.05 | 3.0 d ± 0.12 | 5.3 d ± 0.03 | 6.8 d ± 0.14 |
| 6 | 3.9 c ± 0.22 | 6.1 c ± 0.04 | 8.0 c ± 0.05 | 3.3 c ± 0.12 | 5.6 c ± 0.3 | 7.2 c ± 0.4 | |
| 9 | 4.2 b ± 0.03 | 6.5 b ± 0.04 | 8.4 b ± 0.05 | 3.6 b ± 0.13 | 6.0 b ± 0.4 | 7.6 b ± 0.4 | |
| 12 | 4.5 a ± 0.03 | 7.0 a ± 0.05 | 8.9 a ± 0.15 | 3.9 a ± 0.13 | 6.4 a ± 0.4 | 8.1 a ± 0.4 |
| Moisture Content | Duration | TVC | YE |
|---|---|---|---|
| (%) | (Months) | (log CFU/g) | (log CFU/g) |
| Control (6.235) | 3 | 1.5 d ± 0.01 | 1.2 d ± 0.01 |
| 6 | 1.8 c ± 0.02 | 1.5 c ± 0.02 | |
| 9 | 2.2 b ± 0.02 | 1.8 b ± 0.02 | |
| 12 | 2.5 a ± 0.03 | 2.2 a ± 0.03 | |
| Group A (14.877) | 3 | 2.0 d ± 0.02 | 1.8 d ± 0.02 |
| 6 | 2.5 c ± 0.03 | 2.2 c ± 0.03 | |
| 9 | 3.0 b ± 0.03 | 2.7 b ± 0.03 | |
| 12 | 3.5 a ± 0.04 | 3.2 a ± 0.04 | |
| Group B (21.781) | 3 | 2.5 d ± 0.03 | 2.2 d ± 0.03 |
| 6 | 3.0 c ± 0.03 | 2.7 c ± 0.03 | |
| 9 | 3.5 b ± 0.04 | 3.2 b ± 0.04 | |
| 12 | 4.0 a ± 0.04 | 3.7 a ± 0.04 | |
| Group C (26.807) | 3 | 3.0 d ± 0.03 | 2.7 d ± 0.03 |
| 6 | 3.5 c ± 0.04 | 3.2 c ± 0.04 | |
| 9 | 4.0 b ± 0.04 | 3.7 b ± 0.04 | |
| 12 | 4.5 a ± 0.04 | 4.2 a ± 0.04 | |
| Group D (32.062) | 3 | 3.5 d ± 0.04 | 3.2 d ± 0.04 |
| 6 | 4.0 c ± 0.04 | 3.7 c ± 0.04 | |
| 9 | 4.5 b ± 0.04 | 4.2 b ± 0.04 | |
| 12 | 5.0 a ± 0.05 | 4.7 a ± 0.05 | |
| Group E (38.697) | 3 | 4.0 d ± 0.04 | 3.7 d ± 0.04 |
| 6 | 4.5 c ± 0.04 | 4.2 c ± 0.04 | |
| 9 | 5.0 b ± 0.05 | 4.7 b ± 0.25 | |
| 12 | 5.5 a ± 0.05 | 5.2 a ± 0.25 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elamshity, M.G.; Alhamdan, A.M. Development and Prediction of a Non-Destructive Quality Index (Qi) for Stored Date Fruits Using VIS–NIR Spectroscopy and Artificial Neural Networks. Foods 2025, 14, 3060. https://doi.org/10.3390/foods14173060
Elamshity MG, Alhamdan AM. Development and Prediction of a Non-Destructive Quality Index (Qi) for Stored Date Fruits Using VIS–NIR Spectroscopy and Artificial Neural Networks. Foods. 2025; 14(17):3060. https://doi.org/10.3390/foods14173060
Chicago/Turabian StyleElamshity, Mahmoud G., and Abdullah M. Alhamdan. 2025. "Development and Prediction of a Non-Destructive Quality Index (Qi) for Stored Date Fruits Using VIS–NIR Spectroscopy and Artificial Neural Networks" Foods 14, no. 17: 3060. https://doi.org/10.3390/foods14173060
APA StyleElamshity, M. G., & Alhamdan, A. M. (2025). Development and Prediction of a Non-Destructive Quality Index (Qi) for Stored Date Fruits Using VIS–NIR Spectroscopy and Artificial Neural Networks. Foods, 14(17), 3060. https://doi.org/10.3390/foods14173060







