Optimization of Marinating Process and Evaluation of Storage Stability in Bovine By-products
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Experimental Design for Optimization of Base Marinating Formulation
2.3. Comparative Analysis of Oxidative Properties of Natural Antioxidants
2.3.1. ABTS Cation Radical Scavenging Activity
2.3.2. DPPH Free Radical Scavenging Activity
2.3.3. Reducing Power
2.4. Optimization of the Amount of Natural Antioxidant
2.5. Determination of Nutritional Components
2.6. Evaluation of Storage Stability of Bovine By-products During Storage
2.6.1. Edible Quality
Color Measurement
pH
Sensory Evaluation
Texture Properties
2.6.2. Processing Yield of Pre-Prepared Bovine By-products
2.6.3. Oxidative Property
Determination of TBARS Value
Determination of Carbonyl Content
2.6.4. Microbial Analysis
2.7. Statistical Analysis
3. Results and Discussion
3.1. Analysis of Experimental Results from Basic Marinating Studies
3.1.1. Optimization of Marinating Formulas Incorporating Natural Antioxidants
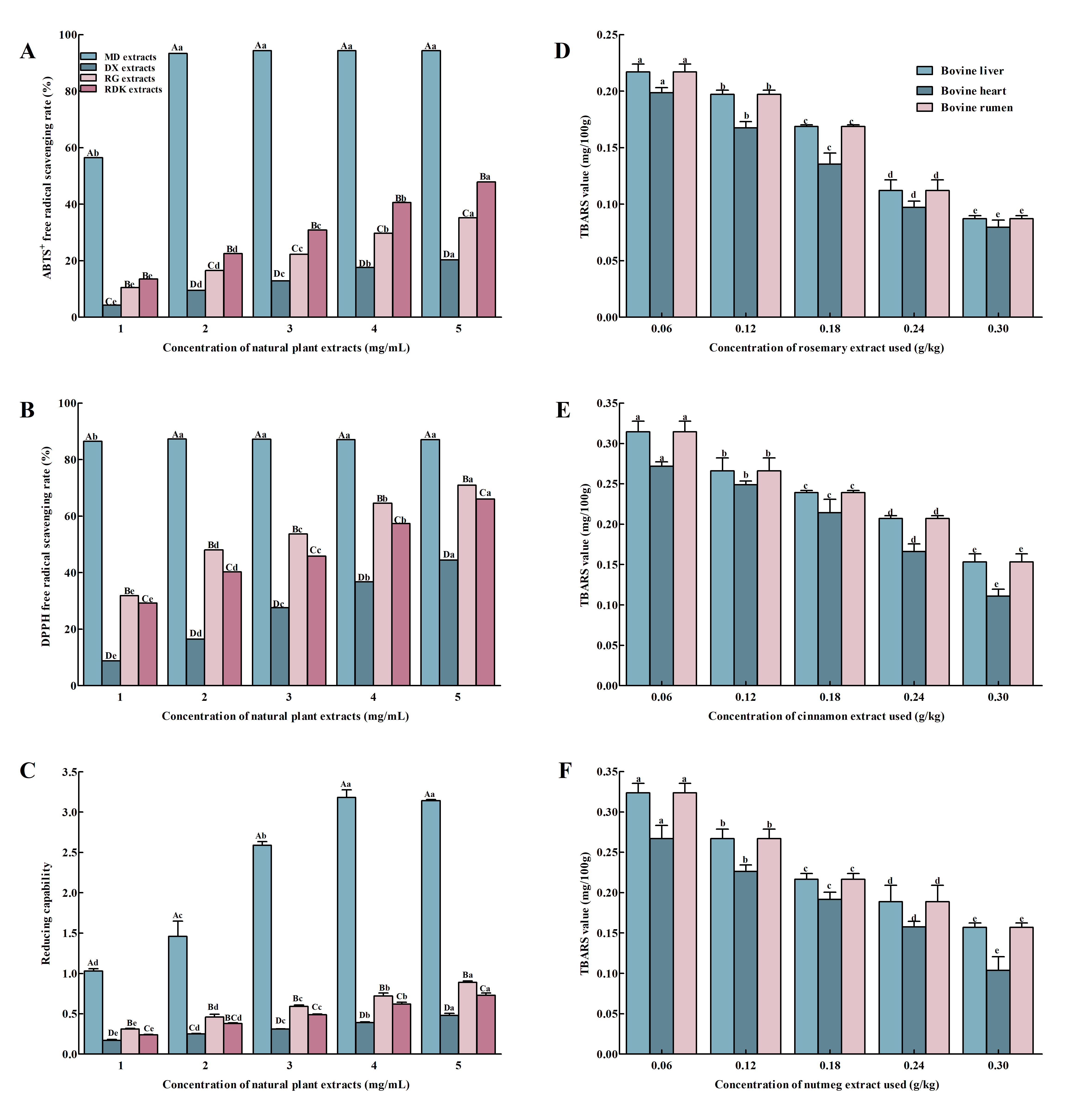
Antioxidant Activities of Natural Antioxidants
Single-Factor Analysis of Natural Antioxidants
3.2. Nutritional Components of Marinated Bovine By-products
3.3. Effect of Refrigeration on Edible Quality
3.3.1. Color Analysis
3.3.2. pH Analysis
3.3.3. Sensory Score Analysis
3.3.4. Product Yield Analysis
3.3.5. Texture Properties Analysis
3.4. Effect of Refrigeration on Oxidative Properties
3.4.1. TBARS Values Analysis
3.4.2. Carbonyl Content Analysis
3.5. TVC Analysis
3.6. Study Limitations
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhao, Y.; Zhang, M.; Law, C.L.; Yang, C. New technologies and products for livestock and poultry bone processing: Research progress and application prospects: A review. Trends Food Sci. Technol. 2024, 144, 104343. [Google Scholar] [CrossRef]
- Mariutti, L.R.; Bragagnolo, N. Influence of salt on lipid oxidation in meat and seafood products: A review. Food Res. Int. 2017, 94, 90–100. [Google Scholar] [CrossRef]
- Hao, S.Q.; Du, J.L.; Sun, X.Y.; Hu, G.H.; Sun, E.K.; Li, X.T.; Jin, Y.; Zhao, L.H. Characteristic flavor analysis of inner mongolia air-dried meat and the impact of vacuum tumbling curing on flavor. J. Food Biochem. 2024, 2024, 4077505. [Google Scholar] [CrossRef]
- Zhou, Y.; Hu, M.; Wang, L. Effects of different curing methods on edible quality and myofibrillar protein characteristics of pork. Food Chem. 2022, 387, 132872. [Google Scholar] [CrossRef]
- Kumar, Y.; Yadav, D.N.; Ahmad, T.; Narsaiah, K. Recent trends in the use of natural antioxidants for meat and meat products. Compr. Rev. Food Sci. Food Saf. 2015, 14, 796–812. [Google Scholar] [CrossRef]
- Fan, X.J.; Liu, S.Z.; Li, H.; He, J.; Feng, J.T.; Zhang, X.; Yan, H. Effects of Portulaca oleracea L. extract on lipid oxidation and color of pork meat during refrigerated storage. Meat Sci. 2019, 147, 82–90. [Google Scholar] [CrossRef]
- Estévez, M. Critical overview of the use of plant antioxidants in the meat industry: Opportunities, innovative applications and future perspectives. Meat Sci. 2021, 181, 108610. [Google Scholar] [CrossRef]
- Petcu, C.D.; Mihai, O.D.; Tăpăloagă, D.; Gheorghe-Irimia, R.-A.; Pogurschi, E.N.; Militaru, M.; Borda, C.; Ghimpeteanu, O.-M. Effects of plant-based antioxidants in animal diets and meat products: A review. Foods 2023, 12, 1334. [Google Scholar] [CrossRef]
- Malik, A.; Najda, A.; Bains, A.; Nurzyńska-Wierdak, R.; Chawla, P. Characterization of citrus nobilis peel methanolic extract for antioxidant, antimicrobial, and anti-inflammatory activity. Molecules 2021, 26, 4310. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Xing, L.J.; Zhou, G.H.; Zhang, W.G. Antioxidative and antibacterial activities of rosemary extract in raw ground pork patties. J. Food Nutr. Res. 2016, 4, 806–813. [Google Scholar] [CrossRef]
- Rajeev, P.S.; Johannah, N.M.; Gopakumar, G.; Maliakel, B.; Krishnakumar, I.M. Optimization of antioxidant efficacy of a deflavored and decolorized rosemary extract: Effect of carnosol content on the oxidative stability of paprika colored beef patties. J. Food Sci. Technol. 2017, 54, 1665–1677. [Google Scholar] [CrossRef] [PubMed]
- Yeddes, M.; Rybak, K.; Rebey, I.B.; Pietrzak, D.; Adamczak, L.; Hammami, M.; Wannes, W.A.; Witrowarajchert, D.; Tounsi, M.S.; Tixier, A.S.F. Lipid oxidation and barrier properties of the coated freeze-dried chicken meat with gelatin-chitosan film enriched with rosemary (Rosmarinus officinalis L.) extract. Foods 2025, 14, 1127. [Google Scholar] [CrossRef]
- Sirocchi, V.; Devlieghere, F.; Peelman, N.; Sagratini, G.; Maggi, F.; Vittori, S.; Ragaert, P. Effect of Rosmarinus officinalis L. essential oil combined with different packaging conditions to extend the shelf life of refrigerated beef meat. Food Chem. 2017, 221, 1069–1076. [Google Scholar] [CrossRef]
- Parvin, R.; Seo, J.K.; Eom, J.U.; Ahamed, Z.; Yang, H.S. Inhibitory and antioxidative capacity of nutmeg extracts on reduction of lipid oxidation and heterocyclic amines in pan-roasted beef patties. Meat Sci. 2023, 197, 109064. [Google Scholar] [CrossRef] [PubMed]
- Moirangthem, S.; Patra, G.; Biswas, S.; Das, A.; Nath, S.; Verma, A.K.; Pal, S.; Chatterjee, N.; Bandyopadhyay, S.; Nanda, P.K. Effect of nutmeg (Myristica fragrans) and tea tree (Melaleuca alternifolia) essential oils on the oxidative and microbial stability of chicken fillets during refrigerated storage. Foods 2024, 13, 4139. [Google Scholar] [CrossRef]
- Shoqairan, Y.I.; Darwish, H.K.; Hamami, M.A.H.; Al-Juhaimi, F.Y.; Ahmed, I.A.M.; Babiker, E.E. The influence of cinnamon powder on the antioxidant and antimicrobial properties of beef burger during refrigerated storage. LWT Food Sci. Technol. 2023, 188, 115422. [Google Scholar] [CrossRef]
- Hussain, Z.; Li, X.; Zhang, D.; Hou, C.; Ijaz, M.; Bai, Y.; Xiao, X.; Zheng, X. Influence of adding cinnamon bark oil on meat quality of ground lamb during storage at 4 °C. Meat Sci. 2021, 171, 108269. [Google Scholar] [CrossRef]
- Zahid, M.A.; Choi, J.Y.; Seo, J.K.; Parvin, R.; Ko, J.; Yang, H.S. Effects of clove extract on oxidative stability and sensory attributes in cooked beef patties at refrigerated storage. Meat Sci. 2020, 161, 107972. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Kang, Y.H. Antioxidant and enzyme inhibitory activities of Plebeian Herba (Salvia plebeia R. Br.) under different cultivation conditions. J. Agric. Food Chem. 2014, 62, 2190–2197. [Google Scholar] [CrossRef]
- Ghasemian, S.O.; Ahmadi-Dastgerdi, A.; Abdollahi, A.; Tirtashi, F.E.; Zokaei, M.; Fallah, N.; NajafAbadi, P.I.; Dolatyari, F.; Verma, A.K. The Effect of active packaging film based on chitosan containing rosemary (Rosmarinus officinalis L.) extract on cheese shelf life. J. Food Biochem. 2024, 2024, 2108707. [Google Scholar] [CrossRef]
- Gu, F.L.; Abbas, S.; Zhang, X.M. Optimization of Maillard reaction products from casein–glucose using response surface methodology. LWT Food Sci. Technol. 2009, 42, 1374–1379. [Google Scholar] [CrossRef]
- GB 5009.3-2016; Determination of Moisture Content in Food. National Standard of the People’s Republic of China: Beijing, China, 2016.
- GB 5009.4-2016; Determination of Ash Content in Food. National Standard of the People’s Republic of China: Beijing, China, 2016.
- GB 5009.5-2016; Determination of Protein Content in Food. National Standard of the People’s Republic of China: Beijing, China, 2016.
- GB 5009.6-2016; Determination of Fat Content in Food. National Standard of the People’s Republic of China: Beijing, China, 2016.
- GB/T 22210-2008; Criterion for Sensory Evaluation of Meat and Meat Products. National Standard of the People’s Republic of China: Beijing, China, 2008.
- Tan, C.; Li, X.; Yu, Y.; Nie, S.; Wen, Q.; Tu, Z.; Zhang, L. Effects of five thermal processing methods on the physicochemical properties and flavor characteristics of grass carp meat. LWT Food Sci. Technol. 2024, 206, 116599. [Google Scholar] [CrossRef]
- GB 4789.2-2022; Food Microbiological Examination—Determination of Total Plate Count. National Standard of the People’s Republic of China: Beijing, China, 2022.
- Ballantyne, K.N.; van Oorschot, R.A.; Mitchell, R.J. Reduce optimisation time and effort: Taguchi experimental design methods. Forensic Sci. Int. Genet. 2008, 1, 7–8. [Google Scholar] [CrossRef]
- Fu, R.; Zhang, Y.; Guo, Y.; Liu, F.; Chen, F. Determination of phenolic contents and antioxidant activities of extracts of Jatropha curcas L. seed shell, a by-product, a new source of natural antioxidant. Ind. Crops Prod. 2014, 58, 265–270. [Google Scholar] [CrossRef]
- Ghorbani, A.; Mahmoudifar, K.; Shokri, S.; Mazaheri, Y.; Shamloo, E.; Rezagholizade-shirvan, A.; Elhamirad, A.H. Effect of Allium Jesdianum’s extract on the physicochemical, antioxidant, antimicrobial and sensory properties of sausage characteristics. Food Chem. X 2024, 22, 101461. [Google Scholar] [CrossRef]
- Li, Y.; Jiang, B.; Zhang, T.; Mu, W.; Liu, J. Antioxidant and free radical-scavenging activities of chickpea protein hydrolysate (CPH). Food Chem. 2008, 106, 444–450. [Google Scholar] [CrossRef]
- Huang, M.; Wang, H.; Xu, X.; Lu, X.; Song, X.; Zhou, G. Effects of nanoemulsion-based edible coatings with composite mixture of rosemary extract and ε-poly-L-lysine on the shelf life of ready-to-eat carbonado chicken. Food Hydrocoll. 2020, 102, 105576. [Google Scholar] [CrossRef]
- Sun, Y.; Zhang, M.; Bhandari, B.; Bai, B. Nanoemulsion-based edible coatings loaded with fennel essential oil/cinnamaldehyde: Characterization, antimicrobial property and advantages in pork meat patties application. Food Control 2021, 127, 108151. [Google Scholar] [CrossRef]
- Wang, Z.F.; He, Z.F.; Zhang, D.; Li, H.J.; Wang, Z.M. Using oxidation kinetic models to predict the quality indices of rabbit meat under different storage temperatures. Meat Sci. 2020, 162, 10842. [Google Scholar] [CrossRef]
- GB 2760-2024; Standard for Use of Food Addditives. National Standard of the People’s Republic of China: Beijing, China, 2024.
- Tomasevic, I.; Djekic, I.; Furnol, M.F.I.; Terjung, N.; Lorenzo, J.M. Recent advances in meat color research. Curr. Opin. Food Sci. 2021, 41, 81–87. [Google Scholar] [CrossRef]
- Faustman, C.; Cassens, R.G. The biochemical basis for discoloration in fresh meat: A review. J. Muscle Foods 1990, 1, 217–243. [Google Scholar] [CrossRef]
- Li, X.; Zhang, Y.; Li, Z.; Li, M.; Liu, Y.; Zhang, D. The effect of temperature in the range of −0.8 to 4 °C on lamb meat color stability. Meat Sci. 2017, 134, 28–33. [Google Scholar] [CrossRef]
- Wei, Z.X.; Zhang, J.J.; Zhang, H.C.; Zhang, N.; Zhang, R.Y.; Li, L.J.; Liu, G.Q. Effect of nanoemulsion loading a mixture of clove essential oil and carboxymethyl chitosan-coated ε-polylysine on the preservation of donkey meat during refrigerated storage. J. Food Process. Preserv. 2021, 45, e15733. [Google Scholar] [CrossRef]
- Sujiwo, J.; Kim, D.; Jang, A. Relation among quality traits of chicken breast meat during cold storage: Correlations between freshness traits and torrymeter values. Poult. Sci. 2018, 97, 2887–2894. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, P.; Pradhan, S.R.; Das, A.; Nanda, P.K.; Bandyopadhyay, N.; Das, A.K. Inhibition of lipid and protein oxidation in raw ground pork by Terminalia arjuna fruit extract during refrigerated storage. Asian Australas. J. Anim. Sci. 2019, 32, 265–273. [Google Scholar] [CrossRef]
- Vaz-Pires, P.; Seixas, P.P.; Mota, M.; Lapa-Guimarães, J.; Pickova, J.; Lindo, A.; Silva, T. Sensory, microbiological, physical and chemical properties of cuttlefish (Sepia officinalis) and broadtail shortfin squid (Illex coindetii) stored in ice. LWT Food Sci. Technol. 2008, 41, 1655–1664. [Google Scholar] [CrossRef]
- Chaijan, M.; Benjakul, S.; Visessanguan, W.; Faustman, C. Changes of pigments and color in sardine (Sardinella gibbosa) and mackerel (Rastrelliger kanagurta) muscle during iced storage. Food Chem. 2005, 93, 607–617. [Google Scholar] [CrossRef]
- Jiang, Z.Y.; Woollard, A.C.S.; Wolff, S.P. Lipid hydroperoxide measurement by oxidation of Fe2+ in the presence of xylenol orange. comparison with the TBA assay and an lodometric method. Lipid 1991, 26, 853–856. [Google Scholar] [CrossRef]
- Van Buren, J.B.; Epperson, B.; Jepsen, S.; Heimbuch, M.; Oliver, K.; Nasados, J.; Bass, P.D.; Colle, M.J. Acerola cherry and rosemary extracts improve color and delay lipid oxidation in previously frozen beef. Foods 2024, 13, 1476. [Google Scholar] [CrossRef] [PubMed]
- Szmańko, T.; Lesiów, T.; Górecka, J. The water-holding capacity of meat: A reference analytical method. Food Chem. 2021, 357, 129727. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Xie, J.; Chu, Y.M. Degradation and evaluation of myofibril proteins induced by endogenous protease in aquatic products during storage: A review. Food Sci. Biotechnol. 2023, 32, 1005–1018. [Google Scholar] [CrossRef]
- Liu, J.; Arner, A.; Puolanne, E.; Ertbjerg, P. On the water-holding of myofibrils: Effect of sarcoplasmic protein denaturation. Meat Sci. 2016, 119, 32–40. [Google Scholar] [CrossRef]
- Colle, M.C.; Richard, R.P.; Smith, D.M.; Colle, M.J.; Loucks, W.I.; Gray, S.J.; Reynolds, Z.D.; Sutton, H.A.; Nasados, J.A.; Doumit, M.E. Dry potato extracts improve water holding capacity, shelf life, and sensory characteristics of fresh and precooked beef patties. Meat Sci. 2019, 149, 156–162. [Google Scholar] [CrossRef]
- Shariatmadari, F.; Ahmadi, H. An overview of rosemary in modern poultry nutrition and production. World’s Poult. Sci. J. 2025, 2025, 1–16. [Google Scholar] [CrossRef]
- Zhuang, X.; Han, M.; Bai, Y.; Liu, Y.; Xing, L.; Xu, X.L.; Zhou, G.H. Insight into the mechanism of myofibrillar protein gel improved by insoluble dietary fiber. Food Hydrocoll. 2018, 74, 219–226. [Google Scholar] [CrossRef]
- Li, F.; Zhong, Q.; Kong, B.; Wang, B.; Pan, N.; Xia, X. Deterioration in quality of quick-frozen pork patties induced by changes in protein structure and lipid and protein oxidation during frozen storage. Food Res. Int. 2020, 133, 109142. [Google Scholar] [CrossRef]
- Xiao, X.C.; Lin, D.; Cao, K.Y.; Sun, L.C.; Chen, Y.L.; Weng, L.; Zhang, L.J.; Cao, M.J. Properties of Pacific white shrimp (Litopenaeus vannamei) collagen and its degradation by endogenous proteinases during cold storage. Food Chem. 2023, 419, 136071. [Google Scholar] [CrossRef] [PubMed]
- Cáceres, E.; García, M.L.; Selgas, M.D. Effect of pre-emulsified fish oil-as source of PUFA n-3- on microstructure and sensory properties of mortadella, a Spanish bologna-type sausage. Meat Sci. 2008, 80, 183–193. [Google Scholar] [CrossRef]
- Hajlaoui, H.; Arraouadi, S.; Mighri, H.; Chaaibia, M.; Gharsallah, N.; Ros, G.; Nieto, G.; Kadri, A. Phytochemical constituents and antioxidant activity of Oudneya Africana L. leaves extracts: Evaluation effects on fatty acids and proteins oxidation of beef burger during refrigerated storage. Antioxidants 2019, 8, 442. [Google Scholar] [CrossRef]
- Karabagias, I.; Badeka, A.; Kontominas, M.G. Shelf life extension of lamb meat using thyme or oregano essential oils and modified atmosphere packaging. Meat Sci. 2011, 88, 109–116. [Google Scholar] [CrossRef]
- Babaolu, A.S.; Poan, H.B.; Ainiwaer, T.; Zkan, H.; Mutlu, E.K.; Karakaya, M. Assessment of garlic and onion powder as natural antioxidant on the physicochemical properties, lipid-protein oxidation and sensorial characteristics of beef and chicken during frozen storage. J. Food Saf. Food Qual. 2023, 74, 101–128. [Google Scholar] [CrossRef]
- Bellucci, E.R.B.; Munekata, P.E.S.; Pateiro, M.; Lorenzo, J.M.; da Silva Barretto, A.C. Red pitaya extract as natural antioxidant in pork patties with total replacement of animal fat. Meat Sci. 2021, 171, 108284. [Google Scholar] [CrossRef]
- Hernández-Hernández, E.; Ponce-Alquicira, E.; Jaramillo-Flores, M.E.; Guerrero Legarreta, I. Antioxidant effect rosemary (Rosmarinus officinalis L.) and oregano (Origanum vulgare L.) extracts on TBARS and colour of model raw pork batters. Meat Sci. 2009, 81, 410–417. [Google Scholar] [CrossRef]
- Chen, X.H.; Shang, S.F.; Yan, F.; Jiang, H.; Zhao, G.J.; Tian, S.; Chen, R.; Chen, D.J.; Dang, Y.F. Antioxidant activities of essential oils and their major components in scavenging free radicals, inhibiting lipid oxidation and reducing cellular oxidative stress. Molecules 2023, 28, 4559. [Google Scholar] [CrossRef] [PubMed]
- Brewer, M.S. Natural antioxidants: Sources, compounds, mechanisms of action, and potential applications. Compr. Rev. Food Sci. Food Saf. 2011, 10, 221–247. [Google Scholar] [CrossRef]
- Wang, C.; Wang, Y.; Song, Y.; Ren, M.; Gao, Z.; Ren, J. Effect of onion skin powder on color, lipid, and protein oxidative stability of premade beef patty during cold storage. Sci. Rep. 2024, 14, 20816. [Google Scholar] [CrossRef]
- Mtibaa, A.C.; Smaoui, S.; Ben Hlima, H.; Sellem, I.; Ennouri, K.; Mellouli, L. Enterocin BacFL31 from a safety Enterococcus faecium FL31: Natural preservative agent used alone and in combination with aqueous peel onion (Allium cepa) extract in ground beef meat storage. BioMed Res. Int. 2019, 2019, 4094890. [Google Scholar] [CrossRef]
- Lee, M.A.; Kim, T.K.; Hwang, K.E.; Choi, Y.J.; Park, S.H.; Kim, C.J.; Choi, Y.S. Kimchi extracts as inhibitors of colour deterioration and lipid oxidation in raw ground pork meat during refrigerated storage. J. Sci. Food Agric. 2019, 99, 2735–2742. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Peng, X.; Li, X.; Wu, J.; Guo, X. The application of clove extract protects chinese-style sausages against oxidation and quality deterioration. Korean J. Food Sci. Anim. Resour. 2017, 37, 114–122. [Google Scholar] [CrossRef]
- SB/T 10482-2008; Quality Safety Requirement of Prepared Meat Products. Domestic Trade Industry Standard of the People’s Republic of China: Beijing, China, 2008.
- Corral-Lugo, A.; Daddaoua, A.; Ortega, A.; Espinosa-Urgel, M.; Krell, T. So different and still so similar: The plant compound rosmarinic acid mimics bacterial homoserine lactone quorum sensing signals. Commun. Integr. Biol. 2016, 9, e1156832. [Google Scholar] [CrossRef]



| Evaluation Projects | Evaluation Criteria | Score |
|---|---|---|
| Texture (20) | Firm and elastic, with rapid recovery from finger indentation | 15–20 |
| Slightly elastic, with slower recovery from finger indentation | 8–14 | |
| Slightly elastic, with slower recovery from finger indentation | 0–7 | |
| Tissue state (20) | Fine texture, intact shape, smooth surface | 15–20 |
| Somewhat rough texture, muscles not tightly packed, locally loose | 8–14 | |
| Rough texture, rough surface, large cracks, loose muscle tissue | 0–7 | |
| Color (20) | Glossy surface, even color | 15–20 |
| Indicates poor luster and average color | 8–14 | |
| No luster on the surface, poor color | 0–7 | |
| Flavor (20) | The overall flavor is intense, with almost no fishy flavor | 15–20 |
| The overall flavor is light, slightly fishy | 8–14 | |
| The overall flavor is single, fishy taste is very heavy | 0–7 | |
| Overall acceptability (20) | Uniform color, strong aroma, overall good impression | 15–20 |
| Relatively uniform color, weaker aroma, generally average impression | 8–14 | |
| Uneven color, off-flavors present, overall poor impression | 0–7 |
| By-products | Treatment | Protein Content (g/100 g) | Fat Content (g/100 g) | Moisture Content (g/100 g) | Ash Content (g/100 g) |
|---|---|---|---|---|---|
| Bovine liver | NG-XX | 21.62 ± 0.03 a | 3.96 ± 0.07 a | 68.42 ± 0.50 a | 1.23 ± 0.07 a |
| NG-CK | 19.35 ± 0.50 b | 3.97 ± 0.01 a | 70.26 ± 0.29 a | 1.23 ± 0.07 a | |
| NG-MD | 20.42 ± 0.19 b | 4.03 ± 0.07 a | 68.77 ± 0.67 a | 1.18 ± 0.10 a | |
| NG-RG | 19.45 ± 0.49 b | 4.04 ± 0.07 a | 69.19 ± 0.88 a | 1.17 ± 0.05 a | |
| NG-RDK | 20.48 ± 0.22 b | 4.17 ± 0.07 a | 69.25 ± 0.60 a | 1.19 ± 0.08 a | |
| Bovine heart | NX-XX | 19.60 ± 0.31 a | 3.98 ± 0.08 a | 67.91 ± 1.11 a | 1.22 ± 0.14 a |
| NX-CK | 17.88 ± 0.29 b | 4.00 ± 0.06 a | 67.76 ± 1.58 a | 1.15 ± 0.07 a | |
| NX-MD | 18.68 ± 0.61 ab | 3.92 ± 0.06 a | 67.93 ± 0.85 a | 1.12 ± 0.12 a | |
| NX-RG | 18.44 ± 0.18 b | 3.90 ± 0.12 a | 68.00 ± 1.43 a | 1.10 ± 0.11 a | |
| NX-RDK | 17.50 ± 0.16 b | 3.84 ± 0.19 a | 67.92 ± 1.68 a | 1.16 ± 0.06 a | |
| Bovine rumen | ND-XX | 13.87 ± 0.17 ab | 3.30 ± 0.06 a | 79.17 ± 1.35 a | 1.11 ± 0.08 a |
| ND-CK | 14.97 ± 0.46 a | 3.32 ± 0.07 a | 77.93 ± 1.58 a | 1.09 ± 0.13 a | |
| ND-MD | 15.43 ± 0.33 a | 3.18 ± 0.09 a | 76.62 ± 2.64 a | 1.15 ± 0.09 a | |
| ND-RG | 15.04 ± 0.59 a | 3.29 ± 0.14 a | 77.34 ± 3.02 a | 1.10 ± 0.08 a | |
| ND-RDK | 13.28 ± 0.67 b | 3.23 ± 0.02 a | 78.99 ± 1.33 a | 1.16 ± 0.10 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qu, Y.; Deng, D.; Zhang, L. Optimization of Marinating Process and Evaluation of Storage Stability in Bovine By-products. Foods 2025, 14, 3036. https://doi.org/10.3390/foods14173036
Qu Y, Deng D, Zhang L. Optimization of Marinating Process and Evaluation of Storage Stability in Bovine By-products. Foods. 2025; 14(17):3036. https://doi.org/10.3390/foods14173036
Chicago/Turabian StyleQu, Yuling, Dan Deng, and Li Zhang. 2025. "Optimization of Marinating Process and Evaluation of Storage Stability in Bovine By-products" Foods 14, no. 17: 3036. https://doi.org/10.3390/foods14173036
APA StyleQu, Y., Deng, D., & Zhang, L. (2025). Optimization of Marinating Process and Evaluation of Storage Stability in Bovine By-products. Foods, 14(17), 3036. https://doi.org/10.3390/foods14173036




