Changes in Umami-Enhancing Nucleotides in White Mullet (Ophiocephalus argus var. Kimnra) Meat Stored at Ice Temperature
Abstract
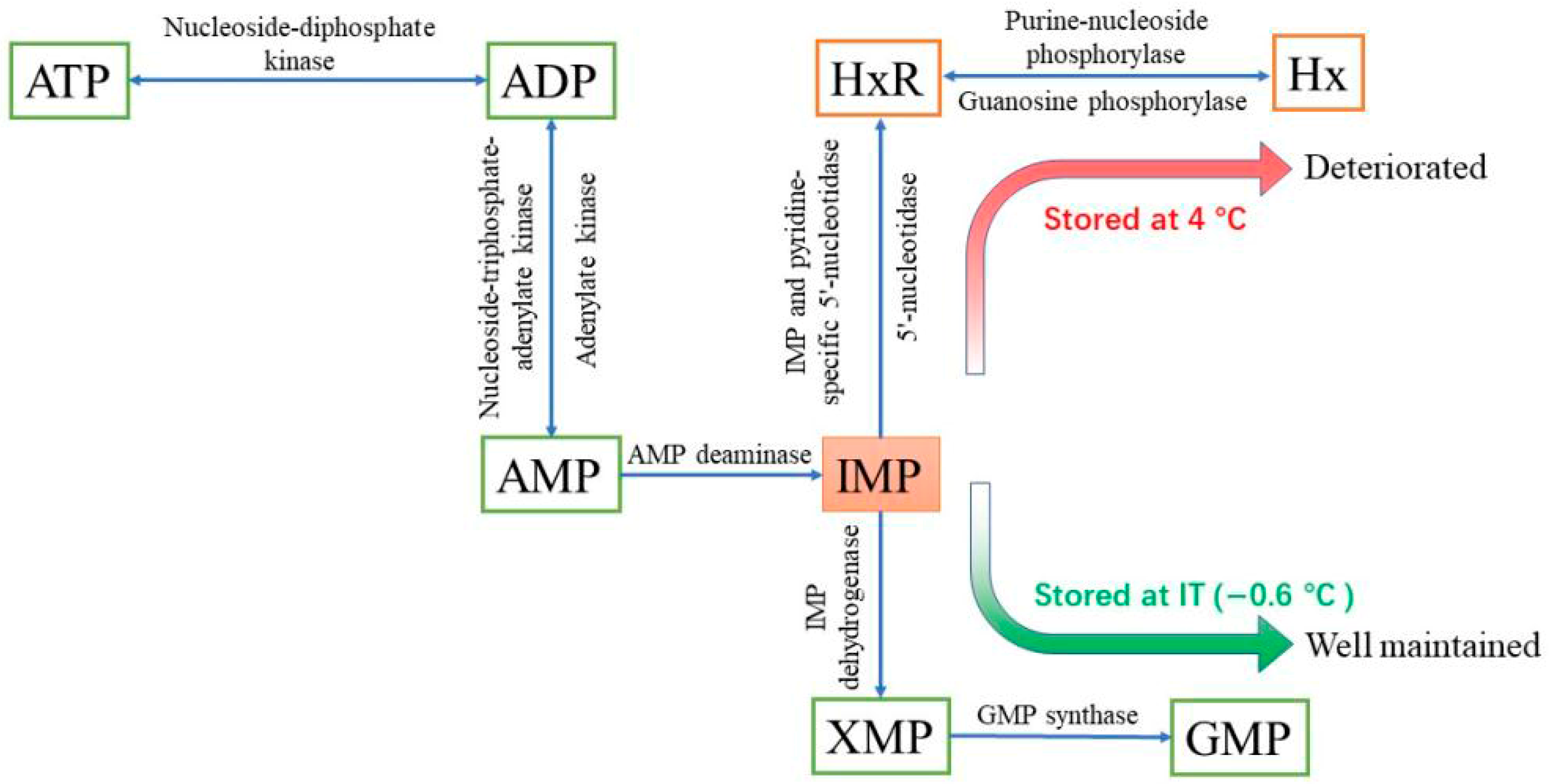
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Experimental Design
2.2.2. Measurement of IT
2.2.3. Determination of Nucleotide Levels
2.2.4. Determination of pH, Color, Water-Holding Capacity (WHC), and Cooking Loss Rate
PH
Color
Water-Holding Capacity (WHC)
Cooking Loss Rate
2.2.5. Determination of 5′-NT Activity
2.2.6. Determination of Glycogen, Pyruvate, and Lactic Acid (LD)
2.2.7. Determination of Pyruvate Kinase (PK) and Lactate Dehydrogenase (LDH) Levels
2.2.8. Data Analysis
3. Results and Analysis
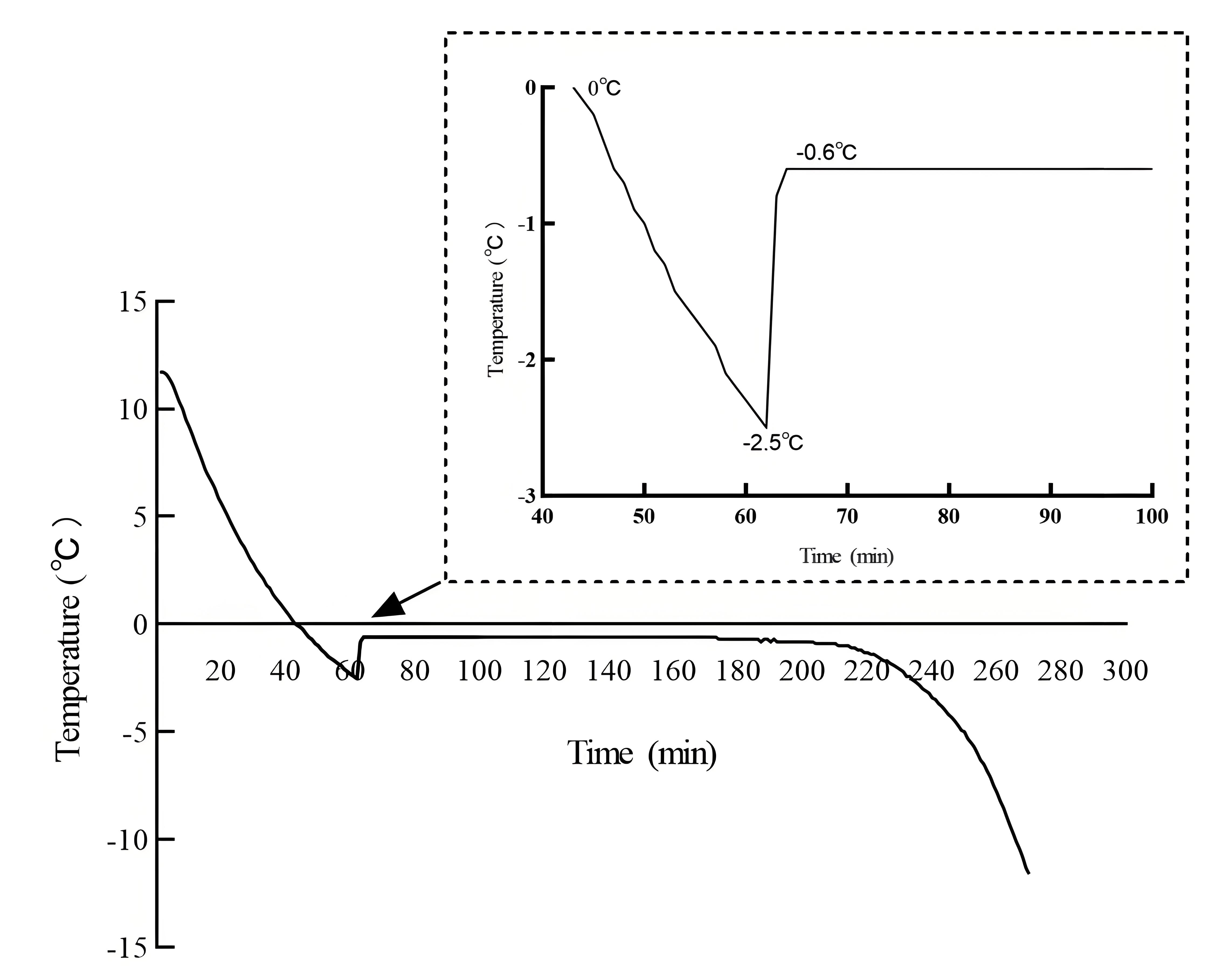
3.1. Measurement Results of Ice Temperature
3.2. Nucleotides
3.2.1. Effect of Storage Temperature on Nucleotide Content
3.2.2. Correlation of Nucleotides
3.3. Effect of IT Storage on pH, WHC, Cooking Loss, and Color of White Mullet Meat
3.3.1. PH, WHC, and Cooking Loss
3.3.2. Muscle Color
3.4. 5′-NT Activity
3.5. Glycogen, Lactic Acid (LD), and Pyruvate
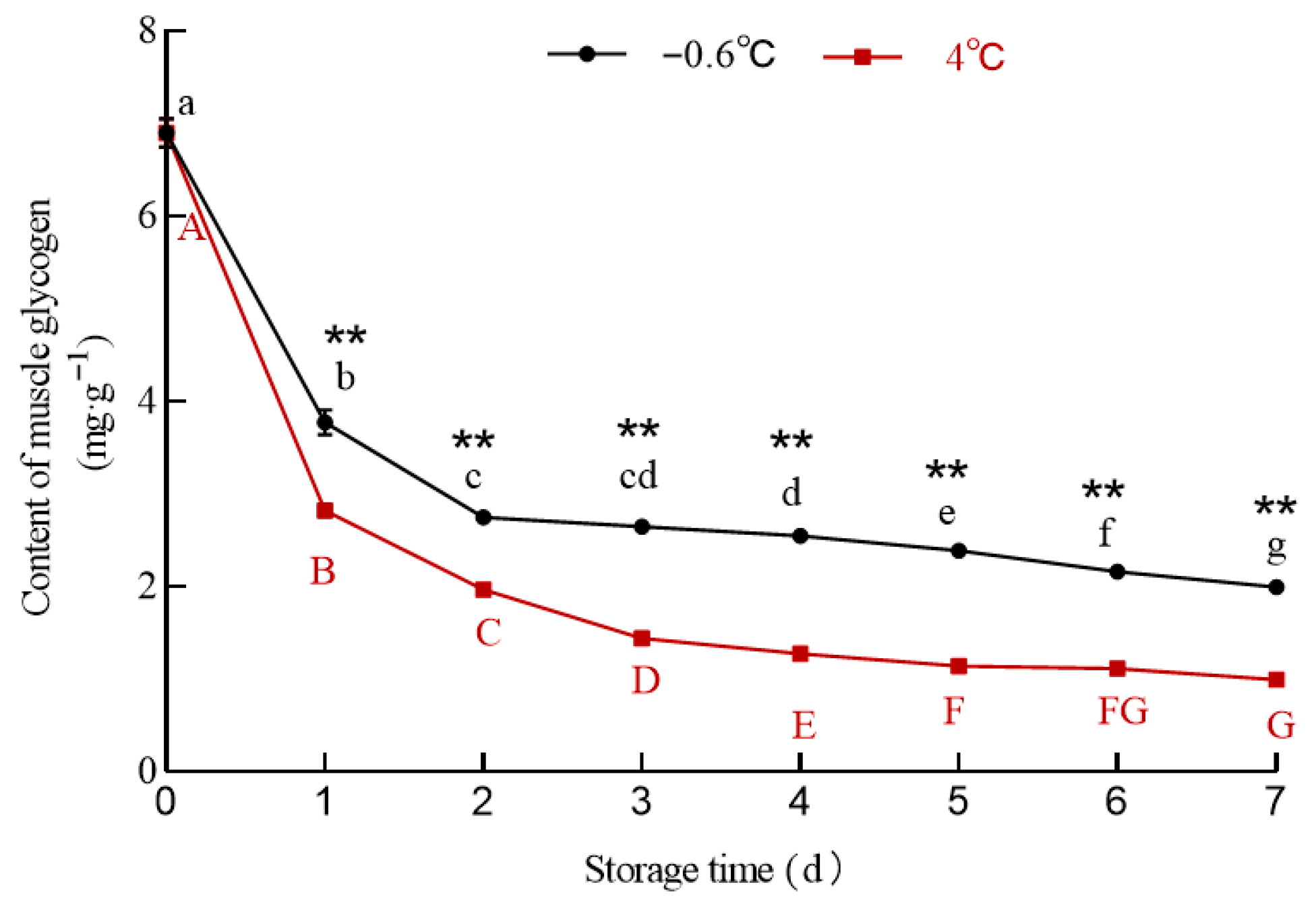
3.5.1. Glycogen
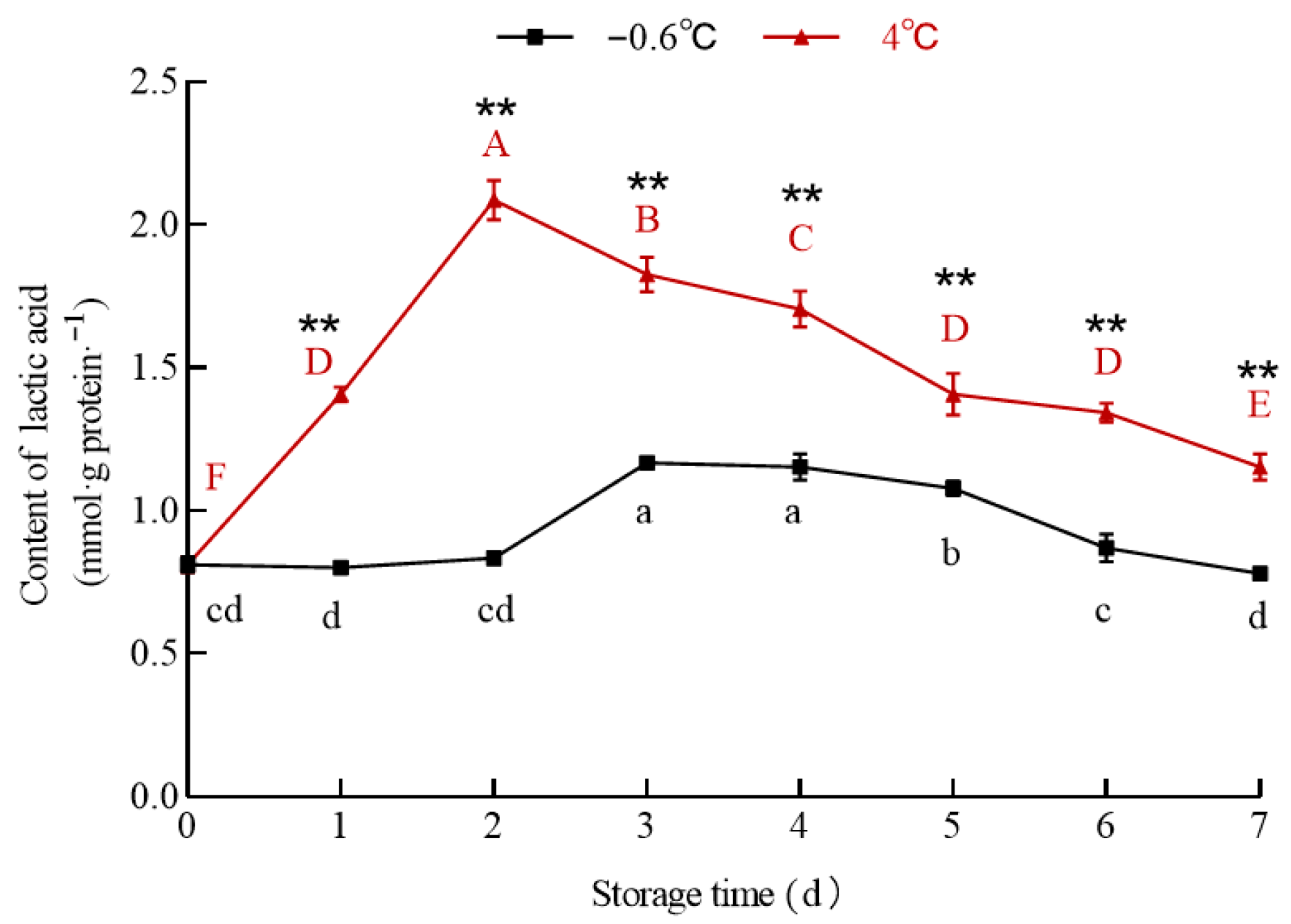
3.5.2. Lactic Acid
3.5.3. Pyruvate

3.6. PK and LDH Activities
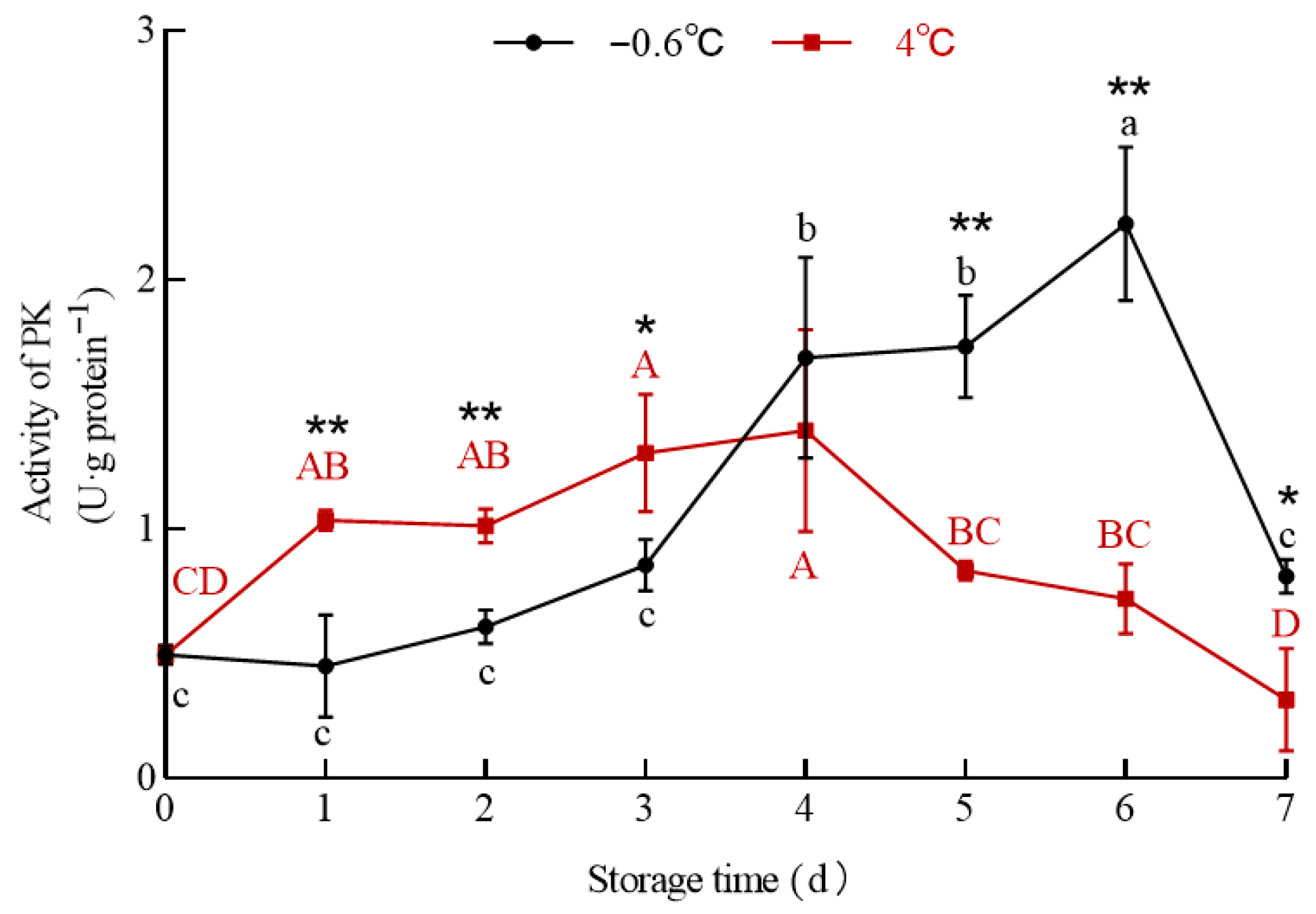
3.6.1. PK
3.6.2. LDH

4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zeng, Q.; Ma, H.; Chen, Q.; Chen, G.; Wang, L.; Zhang, P.; Jia, J.; Zhang, Y. Research Progress on the Application of New Freezing Technology in Meat and Its Products. Food Res. Dev. 2024, 45, 197–201. [Google Scholar]
- Sun, Y.; Tian, Z.; Li, X.; Zhang, X.; Shang, T. Research Progress on the Regulation and Application of Ice-Temperature Preservation Technology to Meat. Food Ind. 2024, 45, 233–237. [Google Scholar]
- Xu, L.; Guan, W.; Zhao, F.; Jing, H. Quality Properties of Chicken Breasts during Ice Temperature Storage. Food Sci. Technol. 2015, 40, 96–102. [Google Scholar] [CrossRef]
- Ji, H.; Hou, X.; Zhang, L.; Wang, X.; Chen, F. Effect of ice-temperature storage on some properties of salt-soluble proteins and gel from chicken breast muscles. CyTA-J. Food 2021, 19, 521–531. [Google Scholar] [CrossRef]
- Gu, X.; Sun, S.; Yang, F.; Li, B.; Yang, L.; Luo, Z. Effects of Ice Temperature or Chilled Storage on Quality and Moisture Migration of Yak Meat. Trans. Chin. Soc. Agric. Eng. 2019, 35, 343–350. [Google Scholar]
- Cao, R.; Yan, L.; Xiao, S.; Hou, B.; Zhou, X.; Wang, W.; Bai, T.; Zhu, K.; Cheng, J.; Zhang, J. Effects of different low-temperature storage methods on the quality and processing characteristics of fresh beef. Foods 2023, 12, 782. [Google Scholar] [CrossRef]
- Tian, Z.; Gulizeiba, Y.; Wu, S.; Liu, D.; Bao, Y.; Wu, Z. Preservation Effect of Ice Temperature Storage on Post Slaughter Horse Meat. Food Sci. Technol. 2025, 12, 147–153. [Google Scholar]
- Qin, Y.; Ayejoripani, A.; Liu, B. Effect of Ice Temperature Storage Combined with Bio-preservatives on the Quality Characteristics of Rainbow Trout Meat. J. Refrig. 2024, 45, 160–166. [Google Scholar]
- Zhang, Y.; Zhang, Y.; Li, H.; Bai, T.; Qian, Q.; Peng, H.; Mu, Y.; Wang, L.; Liu, B.; Chen, J. Effect of 4 °C and ice temperature on umami-enhancing nucleotides of conditioned pork. Food Chem. 2023, 401, 134146. [Google Scholar] [CrossRef]
- Lee, D.; You, Y.; Ho, K.K.; Li, Y.; Jun, S. Impact of supercooling storage on physical and chemical properties of yellowfin tuna (Thunnus albacares). J. Food Eng. 2024, 373, 111818. [Google Scholar] [CrossRef]
- Zhu, Y.; Zhou, X.; Chen, Y.P.; Liu, Z.; Jiang, S.; Chen, G.; Liu, Y. Exploring the relationships between perceived umami intensity, umami components and electronic tongue responses in food matrices. Food Chem. 2022, 368, 130849. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Lv, S.; Liu, Y.; Bi, S.; Zhang, Y. Determination of umami compounds in edible fungi and evaluation of salty enhancement effect of Antler fungus enzymatic hydrolysate. Food Chem. 2022, 387, 132890. [Google Scholar] [CrossRef] [PubMed]
- Hong, H.; Regenstein, J.M.; Luo, Y. The importance of ATP-related compounds for the freshness and flavor of post-mortem fish and shellfish muscle: A review. Crit. Rev. Food Sci. Nutr. 2017, 57, 1787–1798. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Zhuang, S.; Peng, Y.; Tan, Y.; Hong, H.; Luo, Y. Mechanism of inosine monophosphate degradation by specific spoilage organism from grass carp in fish juice system. Foods 2022, 11, 2672. [Google Scholar] [CrossRef]
- Jiang, S.; Ouyang, Z.; Cai, Y.; Lin, Y.; Zheng, S. Transcription factor based whole-cell biosensor for inosinic acid in Corynebacterium stationis. Biochem. Eng. J. 2024, 205, 109248. [Google Scholar] [CrossRef]
- Zhang, Y.; Zeng, Q.; Zhang, Y.; Zhang, P.; Li, Q.; Zhou, J.; Dong, L.; Pan, Z. New Method for 5′−Nucleotidase Preparation and Evaluation of Its Catalytic Activity. Foods 2024, 13, 708. [Google Scholar] [CrossRef]
- Pacheco-Aguilar, R.; Ramirez-Suarez, J.; Castillo-Yañez, F.; Peña-Ramos, E.; Valenzuela-Soto, E.; Marquez-Rios, E. Isolation and properties of 5′-nucleotidase isolated from jumbo squid (Dosidicus gigas) mantle muscle from the Gulf of California, Mexico. Food Chem. 2009, 112, 880–884. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, P.; Chen, Q.; Wang, A.; Dong, L.; Zhang, L. Preliminary Study on the Functions of Peptides Obtained from White Mullet (Ophiocephalus argus var. Kimnra) Meat. Foods 2025, 14, 1322. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, Y.; Chen, P.; Chen, T.; Fang, J.; Wang, L.; Ke, H.; Xiong, W. Heat Resistance and Identification of Characteristic Strain of Thermophilic Bacteria from Epidermal Mucus of White Mullet Fish (Opniocepnalus argus var. Kimnra). Mod. Food Sci. Technol. 2018, 34, 77–82. [Google Scholar]
- Zhang, L.; Chen, P.; Wang, L.; Chen, T.; Zhang, Y.; Xiong, W.; Guo, S. Preliminary Study of Anti-Bacterial Activity of Epidermal Mucus from White Mullet Fish. J. Chengdu Univ. (Nat. Sci. Ed.) 2018, 37, 155–158. [Google Scholar]
- Zhang, L.; Zhang, Y.; Zhang, P.; Li, Y.; Tian, M.; Wang, X.; Ke, H.; Xiong, W. Effect of Carbon Monoxide Treatment on Quality of White Mullet Fish (Opniocepnalus argus var. Kimnra) during Storage. Mod. Food Sci. Technol. 2019, 35, 95–100. [Google Scholar]
- Jiang, L.; Meng, X.; Li, Y.; Ji, L.; Jiao, X. Research on Processing Technology of Convenient Fish Soup Powder of White Mullet. Chin. Condiments 2020, 45, 85–87. [Google Scholar]
- Jiao, X.; Su, J.; Luo, Y.; Fan, W.; Shu, C.; Wu, J.; Jiang, L. Study on the Processing Technology of Fish Cake with Opniocepnalus argus var. Kimnra. Chin. Condiments 2022, 47, 163–165. [Google Scholar]
- Jiang, L.; Shu, C.; Xu, Y.; Sun, Q.; Jiao, X.; Meng, X. Study on Processing Technology of White Mullet Flavor Fish Floss. Chin. Condiments 2022, 47, 109–113. [Google Scholar]
- Deng, Y.; Jiao, X.; Yang, Z.; Wu, J.; Zhang, W.; Zhang, Y. Study on the Processing Technology of White Mullet Fish Balls. Trop. Agric. Eng. 2024, 48, 75–78. [Google Scholar]
- Zhang, L. Preparation of Degradable Polylactic Acid Filmand Its Application in Storage of White MulletFish (Opniocepnalus argus var. Kimnra). Master’s Thesis, Chengdu University, Chengdu, China, 2020. [Google Scholar]
- Cen, X. Construction of Low Frequency Electric Field Superchilling Preservation Platform and Its Effects on Tilapia (Oreochromis niloticus) Meat. Master’s Thesis, Huazhong Agricultural University, Wuhan, China, 2024. [Google Scholar]
- Cai, L. Effect of Ultra-High Pressure on Cyprinus Carpio Sausages Gel Property and Quality Changes during Refrigerated Storage. Chin. Food Addit. 2022, 33, 112–116. [Google Scholar]
- Guo, R.; Feng, Y.; Liu, C.; Qiu, J.; Liang, W. Effect of Different Packaging Methods on the Quality Characteristicsof Buffalo Meat during Refrigerated Storage. Food Ind. Sci. Technol. 2023, 44, 369–377. [Google Scholar]
- Skirnisdottir, S.; Knobloch, S.; Lauzon, H.L.; Ólafsdóttir, A.; Steinþórsson, P.; Bergsten, P.; Marteinsson, V.Þ. Impact of onboard chitosan treatment of whole cod (Gadus morhua) on the shelf life and spoilage bacteria of loins stored superchilled under different atmospheres. Food Microbiol. 2021, 97, 103723. [Google Scholar] [CrossRef]
- Sun, K.; Pan, C.; Chen, S.; Hu, X.; Deng, J.; Li, C. Research Progress on Formation and Properties of Ice Crystals During Partial Freezing Storage of Aquatic Products. J. Guangdong Ocean Univ. 2021, 41, 147–152. [Google Scholar]
- Cui, S.; Liu, K.; Sun, M.; Yang, J.; Zhang, D.; Kang, C.; Zhan, C. Research Progress on the Deterioration Mechanism and Preservation Technologies for Aquatic Products. Meat Res. 2024, 38, 55–62. [Google Scholar]
- Camici, M.; Allegrini, S.; Tozzi, M.G. Interplay between adenylate metabolizing enzymes and AMP-activated protein kinase. FEBS J. 2018, 285, 3337–3352. [Google Scholar] [CrossRef]
- Li, J.; Zhou, G.; Xue, P.; Dong, X.; Xia, Y.; Regenstein, J.; Du, M.; Sun, L. Spoilage microbes: Effect on freshness and IMP degradation in sturgeon fillets during chilled storage. Food Biosci. 2021, 41, 101008. [Google Scholar] [CrossRef]
- Lin, Y.; Zhang, L.; Han, H.; Tian, Y.; Yamashita, T.; Yuan, C.; Wang, Z. Evaluation of Vacuum Microwave Thawing on Quality and Flavor Profiles of North Pacific Krill (Euphausia pacifica) for Potential Human Consumption. Food Bioprocess Technol. 2025, 18, 6504–6519. [Google Scholar] [CrossRef]
- Décout, J.L.; Maurel, M.C. Purine Chemistry in the Early RNA World at the Origins of Life: From RNA and Nucleobases Lesions to Current Key Metabolic Routes. Chem. Bio. Chem. 2025, 26, e202500035. [Google Scholar] [CrossRef] [PubMed]
- Huff-Lonergan, E.; Lonergan, S.M. Mechanisms of water-holding capacity of meat: The role of postmortem biochemical and structural changes. Meat Sci. 2005, 71, 194–204. [Google Scholar] [CrossRef]
- Zhu, Q. Study on the Effects and the Mechanism of Protein S-Nitrosylationon the Development of High Ultimate pH Beef. Master’s Thesis, Nanjing Agricultural University, Nanjing, China, 2024. [Google Scholar]
- Zhang, Y.; Puolanne, E.; Ertbjerg, P. Mimicking myofibrillar protein denaturation in frozen-thawed meat: Effect of pH at high ionic strength. Food Chem. 2020, 338, 128017. [Google Scholar] [CrossRef]
- Liu, Q.; Long, Y.; Zhang, Y.; Zhang, Z.; Yang, B.; Chen, C.; Huang, L.; Su, Y. Phenotypic and genetic correlations of pork myoglobin content with meat colour and other traits in an eight breed-crossed heterogeneous population. Animal 2021, 15, 100364. [Google Scholar] [CrossRef] [PubMed]
- Bai, C.; Xiong, G.; Xu, P.; Li, N.; Wang, J.; Liao, T. Effect of cold-anesthetization rate on blood biochemical parameters and muscle composition during live channel catfish Ictalurus punctatus waterless preservation. Fish. Sci. 2020, 86, 1043–1053. [Google Scholar] [CrossRef]
- Ramos, P.M.; Pedrão, M.R.; Bell, L.C.; Scheffler, T.L.; Ramos, P.M. Early postmortem metabolism and protease activation in fast glycolytic and slow oxidative bovine muscles. Meat Muscle Biol. 2021, 5, 1–10. [Google Scholar] [CrossRef]
- Zhu, X.; Lu, H.; Zheng, Z.; Li, Y.; Hang, G.; Ming, L. Study on Glycolysis Potential and Phosphorylated Proteomics of Camel Meat at Postmortem Aging. Food Ferment. Ind. 2025, 51, 226–233. [Google Scholar]
- Kiyimba, F.; Hartson, S.D.; Mafi, G.G.; Ramanathan, R. Glycogen Supplementation in Vitro Promotes pH Decline in Dark-Cutting Beef by Reverting Muscle’s Metabolome toward a Normal Postmortem Muscle State. J. Agric. Food Chem. 2024, 72, 25275–25285. [Google Scholar] [CrossRef]
- Lin, Y.; Wang, Y.; Li, P.-f. Mutual regulation of lactate dehydrogenase and redox robustness. Front. Physiol. 2022, 13, 1038421. [Google Scholar] [CrossRef]
- Huang, X.; Chen, L.; Ren, C.; Bai, Y.; Hou, C.; Li, X.; Luo, R.; Zhang, D. Research Progress on the Effect of Post-Translational Modification of Pyruvate Kinase on Meat Quality. Food Sci. 2024, 45, 303–310. [Google Scholar]
- Spinicci, K.; Jacquet, P.; Powathil, G.; Stéphanou, A. Modeling the role of HIF in the regulation of metabolic key genes LDH and PDH: Emergence of Warburg phenotype. Comput. Syst. Oncol. 2022, 2, e1040. [Google Scholar] [CrossRef]
- Roosterman, D.; Cottrell, G.S. Rethinking the citric acid cycle: Connecting pyruvate carboxylase and citrate synthase to the flow of energy and material. Int. J. Mol. Sci. 2021, 22, 604. [Google Scholar] [CrossRef]



| Temperature (°C) | Time (d) | AMP | IMP | HxR | Hx | XMP | GMP | UMP | CMP |
|---|---|---|---|---|---|---|---|---|---|
| 4 | 0 | 42.93 ± 8.36 a | 22.73 ± 0.01 e | 3.44 ± 0.03 h | 0.34 ± 0.05 h | 0.25 ± 0.01 d | 22.33 ± 0.01 a | 0.01 ± 0.00 e | 2.15 ± 0.73 a,b |
| 1 | 35.64 ± 0.19 b,* | 45.54 ± 0.03 a | 25.69 ± 0.02 g,* | 3.33 ± 0.02 g,* | 0.19 ± 0.00 f | 20.28 ± 0.18 b,* | 0.16 ± 0.08 d | 2.61 ± 0.01 a | |
| 2 | 19.99 ± 0.15 c | 35.09 ± 0.06 c | 42.28 ± 0.01 f,* | 6.13 ± 0.02 f,* | 0.12 ± 0.01 g | 12.43 ± 0.30 c,* | 0.54 ± 0.00 a | 2.06 ± 0.05 a,b | |
| 3 | 21.86 ± 0.17 c | 45.70 ± 0.16 a | 44.40 ± 0.05 e,* | 8.59 ± 0.01 e,* | 0.22 ± 0.00 e | 10.28 ± 0.19 d,e | 0.34 ± 0.00 b | 2.05 ± 0.02 a,b | |
| 4 | 21.18 ± 0.32 c | 36.93 ± 0.17 b | 54.84 ± 0.24 b,* | 10.04 ± 0.02 d,* | 0.26 ± 0.00 d | 10.61 ± 0.16 d | 0.24 ± 0.00 c | 2.10 ± 0.02 a,b | |
| 5 | 21.33 ± 0.38 c | 29.10 ± 0.20 d | 53.10 ± 0.06 c | 11.23 ± 0.02 c,* | 0.28 ± 0.00 c | 9.99 ± 0.28 e | 0.19 ± 0.00 c,d | 2.03 ± 0.02 b | |
| 6 | 23.02 ± 0.43 c | 10.02 ± 0.04 f | 56.29 ± 0.08 a | 15.11 ± 0.02 b,* | 0.37 ± 0.00 b,* | 8.37 ± 0.38 g | 0.04 ± 0.00 e | 1.92 ± 0.02 b | |
| 7 | 21.89 ± 0.53 c | 4.98 ± 0.01 g | 50.23 ± 0.07 d | 18.26 ± 0.03 a,* | 0.499 ± 0.00 a,* | 8.98 ± 0.42 f | 0.00 ± 0.00 e | 1.96 ± 0.02 b | |
| −0.6 | 0 | 42.93 ± 8.36 a | 22.74 ± 0.01 g | 3.44 ± 0.00 h | 0.34 ± 0.05 h | 0.25 ± 0.01 a,b,c | 22.33 ± 0.01 a | 0.01 ± 0.00 b | 2.15 ± 0.73 a |
| 1 | 24.62 ± 0.26 b | 61.48 ± 3.03 e,* | 16.08 ± 0.13 f | 1.46 ± 0.01 f | 0.17 ± 0.04 c | 15.27 ± 0.36 b | 0.09 ± 0.01 b | 2.53 ± 0.11 a | |
| 2 | 17.94 ± 4.78 b | 74.07 ± 2.35 c,* | 14.35 ± 0.12 g | 1.19 ± 0.01 g | 0.18 ± 0.012 b,c,* | 7.27 ± 0.34 e | 0.98 ± 0.85 a,* | 2.41 ± 0.02 a | |
| 3 | 22.18 ± 2.08 b | 115.93 ± 2.64 a,* | 32.62 ± 0.46 e | 2.81 ± 0.04 e | 0.20 ± 0.06 b,c | 10.77 ± 0.24 c | 1.32 ± 0.11 a,* | 2.31 ± 1.13 a | |
| 4 | 24.81 ± 4.39 b | 80.363 ± 1.372 b,* | 48.95 ± 0.52 d | 5.11 ± 0.04 d | 0.31 ± 0.02 a | 10.97 ± 0.35 c | 0.823 ± 0.11 a,* | 2.19 ± 0.08 a | |
| 5 | 23.73 ± 3.84 b | 69.20 ± 1.32 d,* | 57.90 ± 0.71 b,* | 6.84 ± 0.09 c | 0.27 ± 0.05 a,b | 10.84 ± 0.26 c,* | 0.96 ± 0.24 a,* | 2.24 ± 0.24 a | |
| 6 | 27.71 ± 9.68 b | 66.53 ± 0.73 d,* | 56.90 ± 0.42 c,* | 7.34 ± 0.32 b | 0.29 ± 0.01 a | 9.86 ± 0.78 d,* | 1.18 ± 0.05 a,* | 2.13 ± 0.01 a | |
| 7 | 23.28 ± 5.09 b | 41.15 ± 0.47 f,* | 61.89 ± 0.39 a,* | 10.07 ± 0.09 a | 0.26 ± 0.08 a,b,c | 10.77 ± 0.34 c,* | 1.01 ± 0.47 a,* | 2.06 ± 0.30 a |
| Temperature (°C) | Factors | AMP | IMP | HxR | Hx | XMP | GMP | UMP | CMP |
|---|---|---|---|---|---|---|---|---|---|
| 4 | AMP | 1 | 0.068 | −0.896 ** | −0.693 ** | −0.154 | 0.898 ** | −0.443 * | 0.247 |
| IMP | 0.068 | 1 | −0.182 | −0.624 ** | −0.832 ** | 0.312 | 0.657 ** | 0.418 * | |
| HxR | −0.896 ** | −0.182 | 1 | 0.833 ** | 0.341 | −0.953 ** | 0.211 | −0.378 | |
| Hx | −0.693 ** | −0.624 ** | 0.833 ** | 1 | 0.787 ** | −0.876 ** | −0.248 | −0.450 * | |
| XMP | −0.154 | -.832 ** | 0.341 | 0.787 ** | 1 | −0.432 * | −0.749 ** | −0.347 | |
| GMP | 0.898 ** | 0.312 | −0.953 ** | −0.876 ** | −0.432 * | 1 | −0.174 | 0.514 * | |
| UMP | −0.443 * | 0.657 ** | 0.211 | −0.248 | −0.749 ** | −0.174 | 1 | 0.017 | |
| CMP | 0.247 | 0.418 * | −0.378 | −0.450 * | −0.347 | 0.514 * | 0.017 | 1 | |
| −0.6 | AMP | 1 | −0.582 ** | −0.304 | −0.224 | 0.232 | 0.768 ** | −0.606 ** | −0.322 |
| IMP | −0.582 ** | 1 | 0.209 | −0.014 | −0.172 | −0.641 ** | 0.587 ** | 0.14 | |
| HxR | −0.304 | 0.209 | 1 | 0.957 ** | 0.516 ** | −0.553 ** | 0.552 ** | −0.197 | |
| Hx | −0.224 | −0.014 | 0.957 ** | 1 | 0.487 * | −0.453 * | 0.465 * | −0.224 | |
| XMP | 0.232 | −0.172 | 0.516 ** | 0.487 * | 1 | −0.004 | 0.114 | −0.453 * | |
| GMP | 0.768 ** | −0.641 ** | −0.553 ** | −0.453 * | −0.004 | 1 | −0.706 ** | −0.038 | |
| UMP | −0.606 ** | 0.587 ** | 0.552 ** | 0.465 * | 0.114 | −0.706 ** | 1 | 0.023 | |
| CMP | −0.322 | 0.14 | −0.197 | −0.224 | 0.453 * | −0.038 | 0.023 | 1 |
| Temperature (°C) | Time (d) | L* | a* | b* |
|---|---|---|---|---|
| 4 | 0 | 43.35 ± 0.90 a | 2.65 ± 0.37 a | 1.31 ± 0.40 b,c |
| 1 | 44.19 ± 1.39 a | 2.32 ± 0.44 a,b | 2.10 ± 0.48 a | |
| 2 | 45.06 ± 2.78 a | 1.66 ± 0.85 a,b,c | 1.97 ± 0.30 a,b | |
| 3 | 44.38 ± 1.20 a | 1.49 ± 0.51 b,c | 1.54 ± 0.12 a,b,c | |
| 4 | 43.77 ± 4.13 a | 1.22 ± 0.72 b,c | 1.69 ± 0.06 a,b,c | |
| 5 | 42.07 ± 1.90 a | 1.14 ± 0.17 c | 1.40 ± 0.35 a,b,c | |
| 6 | 44.69 ± 0.25 a | 1.27 ± 0.64 b,c | 1.30 ± 0.51 b,c | |
| 7 | 46.04 ± 2.66 a | 1.32 ± 0.36 b,c | 1.21 ± 0.15 c | |
| −0.6 | 0 | 43.35 ± 0.90 a | 2.65 ± 0.37 a | 1.31 ± 0.40 a |
| 1 | 44.69 ± 0.47 a | 2.47 ± 0.62 a,b | 1.95 ± 0.42 a | |
| 2 | 45.63 ± 2.81 a | 1.81 ± 0.15 a,b | 1.58 ± 0.33 a | |
| 3 | 43.28 ± 1.20 a | 1.51 ± 0.35 a,b | 1.44 ± 0.41 a | |
| 4 | 41.39 ± 2.55 a | 1.31 ± 1.16 b | 1.74 ± 0.17 a | |
| 5 | 43.82 ± 2.08 a | 1.96 ± 0.20 a,b | 1.37 ± 0.46 a | |
| 6 | 44.00 ± 2.91 a | 2.46 ± 0.31 a,b | 1.47 ± 1.34 a | |
| 7 | 45.57 ± 3.97 a | 2.48 ± 0.62 a,b | 1.39 ± 0.14 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.; Li, Q.; Zeng, Q.; Hu, H.; Zhang, L.; Dong, L.; Zhou, J.; Lin, Y. Changes in Umami-Enhancing Nucleotides in White Mullet (Ophiocephalus argus var. Kimnra) Meat Stored at Ice Temperature. Foods 2025, 14, 3022. https://doi.org/10.3390/foods14173022
Zhang Y, Li Q, Zeng Q, Hu H, Zhang L, Dong L, Zhou J, Lin Y. Changes in Umami-Enhancing Nucleotides in White Mullet (Ophiocephalus argus var. Kimnra) Meat Stored at Ice Temperature. Foods. 2025; 14(17):3022. https://doi.org/10.3390/foods14173022
Chicago/Turabian StyleZhang, Yin, Qing Li, Qing Zeng, Hongling Hu, Longyi Zhang, Li Dong, Jiao Zhou, and Yuzhu Lin. 2025. "Changes in Umami-Enhancing Nucleotides in White Mullet (Ophiocephalus argus var. Kimnra) Meat Stored at Ice Temperature" Foods 14, no. 17: 3022. https://doi.org/10.3390/foods14173022
APA StyleZhang, Y., Li, Q., Zeng, Q., Hu, H., Zhang, L., Dong, L., Zhou, J., & Lin, Y. (2025). Changes in Umami-Enhancing Nucleotides in White Mullet (Ophiocephalus argus var. Kimnra) Meat Stored at Ice Temperature. Foods, 14(17), 3022. https://doi.org/10.3390/foods14173022





