Salmonella Biofilm Formation under Fluidic Shear Stress on Different Surface Materials
Abstract
1. Introduction
2. Materials and Methods
2.1. Salmonella Strains
2.2. Surface Materials
2.3. Bacterial Suspension Preparation
2.4. Crystal Violet Assay
2.5. Enumeration of Biofilm Attached Cells
2.6. Scanning Electron Microscopy
2.7. Statistical Analysis
3. Results
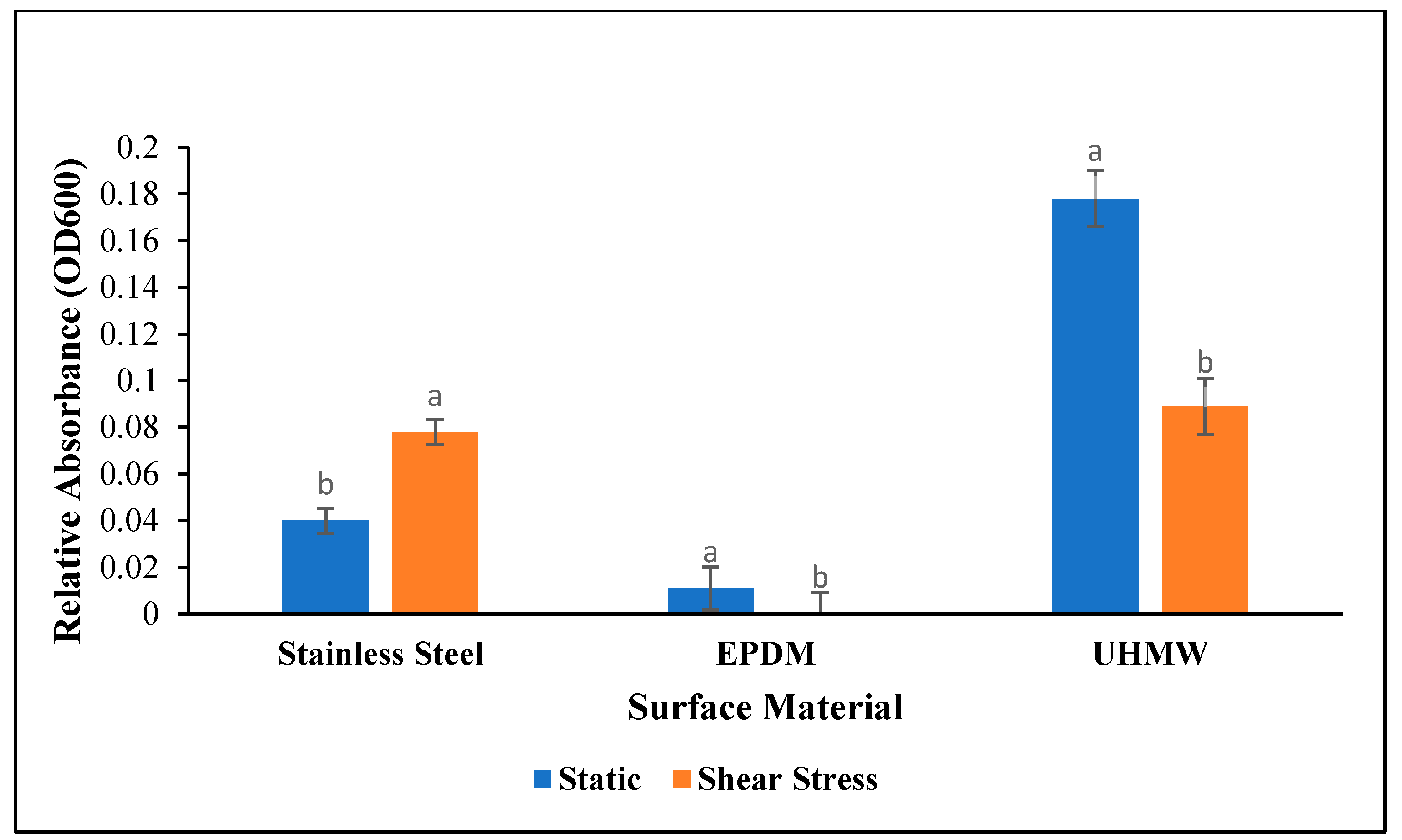
3.1. Crystal Violet Assay
3.2. Enumeration of Biofilm Attached Cells
3.3. Scanning Electron Microscopy
4. Discussion
4.1. Biofilm Density
4.2. Enumeration of Biofilm Attached Cells
4.3. Scanning Electron Microscopy
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Wessels, K.; Rip, D.; Gouws, P. Salmonella in chicken meat: Consumption, outbreaks, characteristics, current control methods and the potential of bacteriophage use. Foods 2021, 10, 1742. [Google Scholar] [CrossRef] [PubMed]
- Salmonella Homepage. Centers for Disease Control and Prevention. 16 June 2022. Available online: https://www.cdc.gov/salmonella/index.html (accessed on 23 January 2023).
- Wibisono, F.M.; Wibisono, F.J.; Effendi, M.H.; Plumeriastuti, H.; Hidayatullah, A.R.; Hartadi, E.B.; Sofiana, E.D. A review of salmonellosis on poultry farms: Public health importance. Syst. Rev. Pharm. 2020, 11, 481–486. [Google Scholar]
- Dar, M.A.; Ahmad, S.M.; Bhat, S.A.; Ahmed, R.; Urwat, U.; Mumtaz, P.T.; Bhat, S.A.; Dar, T.A.; Shah, R.A.; Ganai, N.A. Salmonella typhimurium in poultry: A review. World’s Poult. Sci. J. 2017, 73, 345–354. [Google Scholar] [CrossRef]
- Micciche, A.C.; Foley, S.L.; Pavlidis, H.O.; McIntyre, D.R.; Ricke, S.C. A review of prebiotics against Salmonella in poultry: Current and future potential for microbiome research applications. Front. Vet. Sci. 2018, 5, 191. [Google Scholar] [CrossRef] [PubMed]
- Thames, H.T.; Sukumaran, A.T. A Review of Salmonella and Campylobacter in Broiler Meat: Emerging Challenges and Food Safety Measures. Foods 2020, 9, 776. [Google Scholar] [CrossRef]
- Obe, T.; Nannapaneni, R.; Schilling, W.; Zhang, L.; McDaniel, C.; Kiess, A. Prevalence of Salmonella enterica on poultry processing equipment after completion of sanitization procedures. Poult. Sci. 2020, 99, 4539–4548. [Google Scholar] [CrossRef]
- Pokhrel, D.; Thames, H.T.; Zhang, L.; Dinh, T.T.; Schilling, W.; White, S.B.; Ramachandran, R.; Theradiyil Sukumaran, A. Roles of Aerotolerance, Biofilm Formation, and Viable but Non-Culturable State in the Survival of Campylobacter jejuni in Poultry Processing Environments. Microorganisms 2022, 10, 2165. [Google Scholar] [CrossRef]
- Siceloff, A.T.; Waltman, D.; Shariat, N.W. Regional Salmonella Differences in United States Broiler Production from 2016 to 2020 and the Contribution of Multiserovar Populations to Salmonella Surveillance. Appl. Environ. Microbiol. 2022, 88, e00204-22. [Google Scholar] [CrossRef]
- Thames, H.T.; Fancher, C.A.; Colvin, M.G.; McAnally, M.; Tucker, E.; Zhang, L.; Kiess, A.S.; Dinh, T.T.N.; Sukumaran, A.T. The Prevalence of Salmonella and Campylobacter on Broiler Meat at Different Stages of Commercial Poultry Processing. Animals 2022, 12, 2460. [Google Scholar] [CrossRef]
- Hassan, R.; Buuck, S.; Noveroske, D.; Medus, C.; Sorenson, A.; Laurent, J.; Rotstein, D.; Schlater, L.; Freiman, J.; Douris, A.; et al. Multistate outbreak of Salmonella infections linked to raw turkey products—United States, 2017–2019. Morb. Mortal. Wkly. Rep. 2019, 68, 1045. [Google Scholar] [CrossRef]
- Kumar, A.; Alam, A.; Rani, M.; Ehtesham, N.Z.; Hasnain, S.E. Biofilms: Survival and defense strategy for pathogens. Int. J. Med. Microbiol. 2017, 307, 481–489. [Google Scholar] [CrossRef]
- Merino, L.; Procura, F.; Trejo, F.M.; Bueno, D.J.; Golowczyc, M.A. Biofilm formation by Salmonella sp. in the poultry industry: Detection, control and eradication strategies. Food Res. Int. 2019, 119, 530–540. [Google Scholar] [CrossRef]
- Milanov, D.; Ljubojević, D.; Čabarkapa, I.; Karabasil, N.; Velhner, M. Biofilm as risk factor for Salmonella contamination in various stages of poultry production. Eur. Poult. Sci. 2017, 81. [Google Scholar] [CrossRef]
- Steenackers, H.; Hermans, K.; Vanderleyden, J.; De Keersmaecker, S.C. Salmonella biofilms: An overview on occurrence, structure, regulation and eradication. Food Res. Int. 2012, 45, 502–531. [Google Scholar] [CrossRef]
- Joseph, B.; Otta, S.K.; Karunasagar, I.; Karunasagar, I. Biofilm formation by Salmonella spp. on food contact surfaces and their sensitivity to sanitizers. Int. J. Food Microbiol. 2001, 64, 367–372. [Google Scholar] [CrossRef]
- Nguyen HD, N.; Yuk, H.G. Changes in resistance of Salmonella Typhimurium biofilms formed under various conditions to industrial sanitizers. Food Control 2013, 29, 236–240. [Google Scholar] [CrossRef]
- Xu, Y.; Dhaouadi, Y.; Stoodley, P.; Ren, D. Sensing the unreachable: Challenges and opportunities in biofilm detection. Curr. Opin. Biotechnol. 2020, 64, 79–84. [Google Scholar] [CrossRef]
- Silva, N.B.S.; Marques, L.A.; Röder, D.D.B. Diagnosis of biofilm infections: Current methods used, challenges and perspectives for the future. J. Appl. Microbiol. 2021, 131, 2148–2160. [Google Scholar] [CrossRef]
- Kragh, K.N.; Alhede, M.; Kvich, L.; Bjarnsholt, T. Into the well—A close look at the complex structures of a microtiter biofilm and the crystal violet assay. Biofilm 2019, 1, 100006. [Google Scholar] [CrossRef]
- Stiefel, P.; Rosenberg, U.; Schneider, J.; Mauerhofer, S.; Maniura-Weber, K.; Ren, Q. Is biofilm removal properly assessed? Comparison of different quantification methods in a 96-well plate system. Appl. Microbiol. Biotechnol. 2016, 100, 4135–4145. [Google Scholar] [CrossRef]
- Díez-García, M.; Capita, R.; Alonso-Calleja, C. Influence of serotype on the growth kinetics and the ability to form biofilms of Salmonella isolates from poultry. Food Microbiol. 2012, 31, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Corcoran, M.; Morris, D.; De Lappe, N.; O’connor, J.; Lalor, P.; Dockery, P.; Cormican, M. Commonly used disinfectants fail to eradicate Salmonella enterica biofilms from food contact surface materials. Appl. Environ. Microbiol. 2014, 80, 1507–1514. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Bumunang, E.W.; Stanford, K.; Bie, X.; Niu, Y.D.; McAllister, T.A. Biofilm formation by shiga toxin-producing Escherichia coli on stainless steel coupons as affected by temperature and incubation time. Microorganisms 2019, 7, 95. [Google Scholar] [CrossRef] [PubMed]
- Obe, T.; Nannapaneni, R.; Schilling, W.; Zhang, L.; Kiess, A. Antimicrobial tolerance, biofilm formation, and molecular characterization of Salmonella isolates from poultry processing equipment. J. Appl. Poult. Res. 2021, 30, 100195. [Google Scholar] [CrossRef]
- Shen, C.; Luo, Y.; Nou, X.; Bauchan, G.; Zhou, B.; Wang, Q.; Millner, P. Enhanced inactivation of Salmonella and Pseudomonas biofilms on stainless steel by use of T-128, a fresh-produce washing aid, in chlorinated wash solutions. Appl. Environ. Microbiol. 2012, 78, 6789–6798. [Google Scholar] [CrossRef]
- Yammine, J.; Gharsallaoui, A.; Karam, L.; Ismail, A.; Fadel, A.; Chihib, N.E. Dynamic Salmonella Enteritidis biofilms development under different flow conditions and their removal using nanoencapsulated thymol. Biofilm 2022, 4, 100094. [Google Scholar] [CrossRef]
- Dygico, L.K.; Gahan, C.G.; Grogan, H.; Burgess, C.M. The ability of Listeria monocytogenes to form biofilm on surfaces relevant to the mushroom production environment. Int. J. Food Microbiol. 2020, 317, 108385. [Google Scholar] [CrossRef]
- Nguyen, H.D.N.; Yang, Y.S.; Yuk, H.G. Biofilm formation of Salmonella Typhimurium on stainless steel and acrylic surfaces as affected by temperature and pH level. J. Food Sci. Technol. 2014, 55, 383–388. [Google Scholar] [CrossRef]
- Sadekuzzaman, M.; Mizan, M.F.R.; Yang, S.; Kim, H.S.; Ha, S.D. Application of bacteriophages for the inactivation of Salmonella spp. in biofilms. Food Sci. Technol. Int. 2018, 24, 424–433. [Google Scholar] [CrossRef]
- Ronner, A.B.; Wong, A.C. Biofilm development and sanitizer inactivation of Listeria monocytogenes and Salmonella typhimurium on stainless steel and Buna-n rubber. J. Food Prot. 1993, 56, 750–758. [Google Scholar] [CrossRef]
- Nahar, S.; Ha AJ, W.; Byun, K.H.; Hossain, M.I.; Mizan MF, R.; Ha, S.D. Efficacy of flavourzyme against Salmonella Typhimurium, Escherichia coli, and Pseudomonas aeruginosa biofilms on food-contact surfaces. Int. J. Food Microbiol. 2021, 336, 108897. [Google Scholar] [CrossRef]
- Wang, H.; Ding, S.; Wang, G.; Xu, X.; Zhou, G. In situ characterization and analysis of Salmonella biofilm formation under meat processing environments using a combined microscopic and spectroscopic approach. Int. J. Food Microbiol. 2013, 167, 293–302. [Google Scholar] [CrossRef]
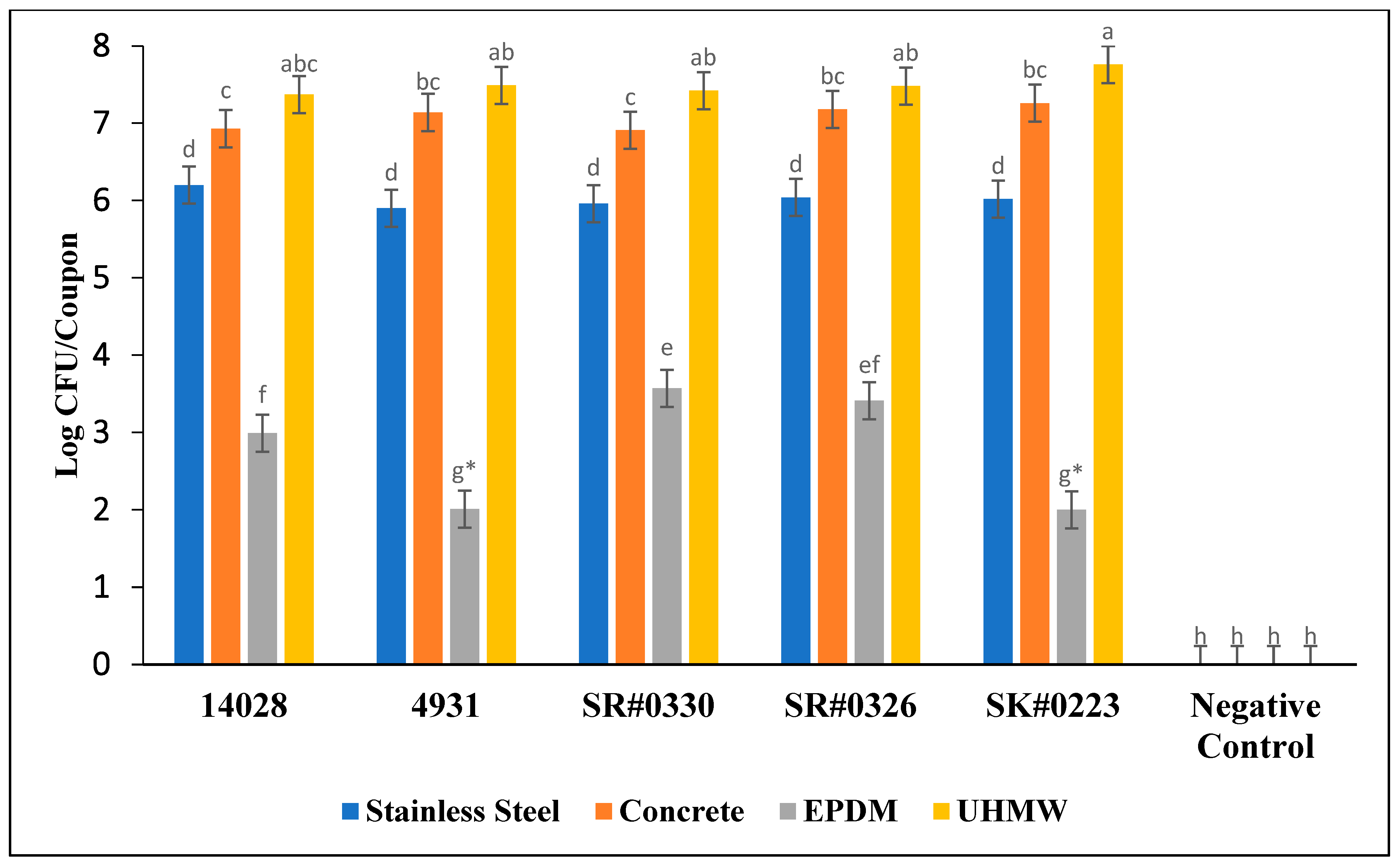

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thames, H.T.; Pokhrel, D.; Willis, E.; Rivers, O.; Dinh, T.T.N.; Zhang, L.; Schilling, M.W.; Ramachandran, R.; White, S.; Sukumaran, A.T. Salmonella Biofilm Formation under Fluidic Shear Stress on Different Surface Materials. Foods 2023, 12, 1918. https://doi.org/10.3390/foods12091918
Thames HT, Pokhrel D, Willis E, Rivers O, Dinh TTN, Zhang L, Schilling MW, Ramachandran R, White S, Sukumaran AT. Salmonella Biofilm Formation under Fluidic Shear Stress on Different Surface Materials. Foods. 2023; 12(9):1918. https://doi.org/10.3390/foods12091918
Chicago/Turabian StyleThames, Hudson T., Diksha Pokhrel, Emma Willis, Orion Rivers, Thu T. N. Dinh, Li Zhang, Mark W. Schilling, Reshma Ramachandran, Shecoya White, and Anuraj T. Sukumaran. 2023. "Salmonella Biofilm Formation under Fluidic Shear Stress on Different Surface Materials" Foods 12, no. 9: 1918. https://doi.org/10.3390/foods12091918
APA StyleThames, H. T., Pokhrel, D., Willis, E., Rivers, O., Dinh, T. T. N., Zhang, L., Schilling, M. W., Ramachandran, R., White, S., & Sukumaran, A. T. (2023). Salmonella Biofilm Formation under Fluidic Shear Stress on Different Surface Materials. Foods, 12(9), 1918. https://doi.org/10.3390/foods12091918





