Exploring Plants with Flowers: From Therapeutic Nutritional Benefits to Innovative Sustainable Uses
Abstract
1. Introduction
2. Conceptualization
2.1. Use of Flowers for Therapeutic and Nutritional Purposes throughout History
2.2. Post-Harvest Treatments Applied to Flowers
2.3. Forms of Consumption
2.4. Relevant Compounds with an Interest in Nutrition, Health Promotion, and Cosmetics
2.5. Beneficial Effects
2.6. Antinutrients or Toxic Compounds
2.7. Flowers, Climate Change, Sustainability, and the Circular Economy
2.7.1. Flowers and Biodiversity
2.7.2. Flowers and Phytoremediation of Soils and Wastewater
2.7.3. Flowers and Dying Fabrics
2.7.4. Other Potential Uses
3. Conclusions and Future Recommendations
- -
- How bioavailable are health-promoting compounds from flowers? And how do post-harvest, industrial, or culinary treatments affect such bioavailability? Can ingesting large amounts of flowers significantly raise plasma and tissue levels of health-promoting compounds?
- -
- Can post-harvest, industrial, or culinary treatments make potentially toxic flowers edible? Can biorefinery approaches be used to obtain added-value products from potentially toxic flowers?
- -
- What plants can be used for phytoremediation and to provide health-promoting rich flowers without posing health risks due to excessive accumulation of pollutants in edible parts?
- -
- What amount of floral waste is necessary for its alternative circular use to be economically viable? How can smaller amounts of waste be used alternatively?
- -
- What floral species can be used to cultivate areas with harsh edaphoclimatic conditions and/or enrich biodiversity by attracting pollinators?
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- De-Ron, A.; Martínez, A. Geología y Biología; Madrid, España, 2003; ISBN 84-665-1891-6. Available online: https://books.google.es/books?id=D13gGEL1ZhwC&lpg=PA18&dq=c%C3%B3mo%20se%20define%20una%20planta%20ornamental&hl=es&pg=PA18#v=onepage&q=c%C3%B3mo%20se%20define%20una%20planta%20ornamental&f=false (accessed on 8 October 2023).
- Fernandes, L.; Saraiva, J.A.; Pereira, J.A.; Casal, S.; Ramalhosa, E. Post-Harvest Technologies Applied to Edible Flowers: A Review: Edible Flowers Preservation. Food Rev. Int. 2019, 35, 132–154. [Google Scholar] [CrossRef]
- Lu, B.; Li, M.; Yin, R. Phytochemical Content, Health Benefits, and Toxicology of Common Edible Flowers: A Review (2000–2015). Crit. Rev. Food Sci. Nutr. 2016, 56, S130–S148. [Google Scholar] [CrossRef] [PubMed]
- Barros, L.; Sarmento, K.; Bisconsin-Junior, A.; Santos, J.; Magnani, M.; Rodriguez, I.; Rodrigo, N.; Tiengo, A.; Maróstica, M. The Use of Alternative Food Sources to Improve Health and Guarantee Access and Food Intake. Food Res. Int. 2021, 149, 110709. [Google Scholar] [CrossRef]
- Ohmiya, A. Diversity of Carotenoid Composition in Flower Petals. JPN Agric. Res. Q. 2011, 45, 163–171. [Google Scholar] [CrossRef]
- Pires, T.; Dias, M.; Barros, L.; Calhelha, R.; Alves, M.; Oliveira, M.; Santos-Buelga, C.; Ferreira, I. Edible Flowers as Sources of Phenolic Compounds with Bioactive Potential. Food Res. Int. 2018, 105, 580–588. [Google Scholar] [CrossRef]
- Del-Rio, D.; Rodriguez-Mateos, A.; Spencer, J.; Tognolini, M.; Borges, G.; Crozier, A. Dietary (Poly)Phenolics in Human Health: Structures, Bioavailability, and Evidence of Protective Effects against Chronic Diseases. Antioxid. Redox Signal. 2013, 18, 1818–1892. [Google Scholar] [CrossRef]
- Albuquerque, B.; Heleno, S.; Oliveira, M.; Barros, L.; Ferreira, I. Phenolic Compounds: Current Industrial Applications, Limitations and Future Challenges. Food Funct. 2021, 12, 14–29. [Google Scholar] [CrossRef]
- Meléndez-Martínez, A.; Böhm, V.; Borge, G.; Cano, M.; Fikselová, M.; Gruskiene, R.; Lavelli, V.; Loizzo, M.; Mandić, A.; Mapelli-Brahm, P.; et al. Carotenoids: Considerations for Their Use in Functional Foods, Nutraceuticals, Nutricosmetics, Supplements, Botanicals and Novel Foods in the Context of Sustainability, Circular Economy and Climate Change. Annu. Rev. Food Sci. Technol. 2021, 12, 433–460. [Google Scholar] [CrossRef]
- Meléndez-Martínez, A.J.; Mandić, A.I.; Bantis, F.; Böhm, V.; Borge, G.I.A.; Brnčić, M.; Bysted, A.; Cano, M.P.; Dias, M.G.; Elgersma, A.; et al. A Comprehensive Review on Carotenoids in Foods and Feeds: Status Quo, Applications, Patents, and Research Needs. Crit. Rev. Food Sci. Nutr. 2022, 62, 1999–2049. [Google Scholar] [CrossRef]
- Wessinger, C. A Genetic Route to Yellow Flowers. New Phytol. 2015, 206, 1193–1195. [Google Scholar] [CrossRef]
- Mlcek, J.; Rop, O. Fresh Edible Flowers of Ornamental Plant—A New Source of Nutraceutical Foods. Trends Food Sci. Technol. 2011, 22, 561–569. [Google Scholar] [CrossRef]
- Alasalvar, C.; Pelvan, E.; Özdemir, K.S.; Kocada, T. Compositional, Nutritional, and Functional Characteristics of Instant Teas Produced from Low- and High-Quality Black. J. Agric. Food Chem. 2013, 61, 7529–7536. [Google Scholar] [CrossRef] [PubMed]
- Coyago, E.; Bonilla, S. Absorción de Plomo de Suelos Altamente Contaminados en Especies Vegetativas Usadas Para Consumo Animal y Humano. La Granja 2016, 23, 35–46. [Google Scholar] [CrossRef]
- Guimarães, R.; Barros, L.; Carvalho, A.; Ferreira, I. Infusions and Decoctions of Mixed Herbs Used in Folk Medicine: Synergism in Antioxidant Potential. Phyther. Res. 2011, 25, 1209–1214. [Google Scholar] [CrossRef] [PubMed]
- Petrovska, B. Historical Review of Medicinal Plants’ Usage. Pharmacogn. Rev. 2012, 6, 1. [Google Scholar] [CrossRef] [PubMed]
- Willett, W.; Rockström, J.; Loken, B.; Springmann, M.; Lang, T.; Vermeulen, S.; Garnett, T.; Tilman, D.; DeClerck, F.; Wood, A.; et al. Food in the Anthropocene: The EAT–Lancet Commission on Healthy Diets from Sustainable Food Systems. Lancet 2019, 393, 447–492. [Google Scholar] [CrossRef]
- WFO. The World Flora Online. Available online: https://wfoplantlist.org/plant-list (accessed on 8 October 2023).
- Poole, R.; Runger, W. Handbook of Flowering, 1st ed.; CRC Press: Boca Raton, FL, USA, 2017; ISBN 9781351072533. [Google Scholar]
- Coyago-Cruz, E.; Baldeón, M. Novel List of Ecuadorian Flowers with Antimicrobial Activity. In Medicinal Plants of Ecuador; CRC Press: Boca Raton, FL, USA; Taylor Francis Group: New York, NY, USA, 2022; ISBN 9780367775865. [Google Scholar]
- Awan, A.; Ahmed, C.; Uzair, M.; Aslam, M.; Farooq, U.; Ishfaq, K. Family Acanthaceae and Genus Aphelandra: Ethnopharmacological and Phytochemical Review. Int. J. Pharm. Pharm. Sci. 2014, 6, 44–55. [Google Scholar]
- Baumeler, A.; Hesse, M.; Werner, C. Benzoxazinoids-Cyclic Hydroxamic Acids, Lactams and Their Corresponding Glucosides in the Genus Aphelandra (Acanthaceae). Phytochemistry 2000, 53, 213–222. [Google Scholar] [CrossRef]
- Dätwyler, P.; Bosshardt, H.; Bernhard, H.O.; Hesse, M.; Johne, S. Die Struktur Des Spermin-alkaloides Aphelandrin Aus Aphelandra squarrosa NEES. Helv. Chim. Acta 1978, 61, 2646–2671. [Google Scholar] [CrossRef]
- Todorova, M.; Werner, C.; Hesse, M. Enzymatic Phenol Oxidation and Polymerization of the Spermine Alkaloid Aphelandrine. Phytochemistry 1994, 37, 1251–1256. [Google Scholar] [CrossRef]
- Javaid, A.; Shahzad, M.; Bashir, C.; Uzair, M. In-Vitro Biological Evaluation of Crude Extract of Aerial Parts of Aphelandra squarrosa. Indian Res. J. Pharm. Sci. 2014, 1, 75–79. [Google Scholar]
- Corrêa, G.; Alcântara, A. Chemical Constituents and Biological Activities of Species of Justicia: A Review. Rev. Bras. Farmacogn. 2012, 22, 220–238. [Google Scholar] [CrossRef]
- Rahmatullah, M.; Khatun, A.; Morshed, N.; Kumar, P.; Ahmed, S.; Hossan, S.; Junrut, M.; Jahan, R.; Rahmatullah, M.; Khatun, M.A.; et al. A Randomized Survey of Medicinal Plants Used by Folk Medicinal Healers of Sylhet Division, Bangladesh. Adv. Nat. Appl. Sci. 2010, 4, 52–62. [Google Scholar]
- Abdela, J. In Vivo Antimalarial Activity of Solvent Fractions of the Leaf of Justicia Schimperiana Hochst. Ex Nees (Acanthaceae) against. Master’s Thesis, Adaba University, Addis, Ethiopia, 2014. [Google Scholar]
- Johnson, A.; Johnson, S. Garden Plants Poisonous to People. Primefact 2006, 9, 12. [Google Scholar]
- Matak, S.; Hashemabadi, D.; Kaviani, B. Changes in Postharvest Physio-Biochemical Characteristics and Antioxidant Enzimes Activity of Cut Alsteroemeria Aurantiaca Flower as Affected by Cycloheximide, Coconut Water and 6-Benzyladenine. Biosci. J. Uberlandia 2017, 33, 321–332. [Google Scholar]
- Estomba, D.; Ladio, A.; Lozada, M. Medicinal Wild Plant Knowledge and Gathering Patterns in a Mapuche Community from North-Western Patagonia. J. Ethnopharmacol. 2006, 103, 109–119. [Google Scholar] [CrossRef]
- Sanso, A. El Género Alstroemeria (Alstroemeriaceae) en Argentina. Darwiniana 2016, 1996, 349–382. [Google Scholar]
- Surse, S.N.; Shrivastava, B.; Sharma, P.; Gide, P.S.; Sana, A. Celosia Cristata: Potent Pharmacotherapeutic Herb—A Review. Int. J. Pharm. Phytopharm. Res. 2015, 6084, 787–796. [Google Scholar] [CrossRef]
- Tobar-Vargas, A.; Gavio, B.; Fernández, J.L. New Records of Plants for San Andres and Old Providence Islands (International Biosphere Reserve Seaflower), Caribbean Colombia. Check List 2013, 9, 1361–1366. [Google Scholar] [CrossRef]
- Varadharaj, V.; Muniyappan, J. Phytochemical and Phytotherapeutic Properties of Celosia Species—A Review. Int. J. Pharmacogn. Phytochem. Res. 2017, 9, 820–825. [Google Scholar] [CrossRef]
- Badawy, E.S.; Kandil, M.; Mahgoub, M.; Shanan, N.; Hegazi, N. Chemical Constituents of Celosia argentea va. Cristata L. Plants as Affected by Foliar Application of Putrescine and Alpha-Tocopherol. Int. J. ChemTech Res. 2015, 8, 464–470. [Google Scholar]
- Islam, S.; Shajib, M.S.; Ahmed, T. Antinociceptive Effect of Methanol Extract of Celosia cristata Linn. in Mice. BMC Complement. Altern. Med. 2016, 16, 400. [Google Scholar] [CrossRef] [PubMed]
- Santhoshkumar, M.; Mahakavi, T.; Baskaran, L. Isolation and Identification of Bacteria from Chlorpyrifos Polluted Soil. Int. Lett. Nat. Sci. 2015, 45, 23–26. [Google Scholar] [CrossRef]
- Kim, Y.S.; Hwang, J.W.; Sung, S.H.; Jeon, Y.J.; Jeong, J.H.; Jeon, B.T.; Moon, S.H.; Park, P.J. Antioxidant Activity and Protective Effect of Extract of Celosia cristata L. Flower on Tert-Butyl Hydroperoxide-Induced Oxidative Hepatotoxicity. Food Chem. 2015, 168, 572–579. [Google Scholar] [CrossRef] [PubMed]
- Andarwulan, N.; Kurniasih, D.; Apriady, R.A.; Rahmat, H.; Roto, A.V.; Bolling, B.W. Polyphenols, Carotenoids, and Ascorbic Acid in Underutilized Medicinal Vegetables. J. Funct. Foods 2012, 4, 339–347. [Google Scholar] [CrossRef]
- Al-Snafi, A.E. Pharmacological Effects of Allium Species Grown in Iraq. An Overview. Int. J. Pharm. Health Care Res. 2013, 1, 132–155. [Google Scholar]
- Al-Snafi, A. Phenolics and Flavonoids Contents of Medicinal Plants, as Natural Ingredients for Many Therapeutic Purposes—A Review. IOSR J. Pharm. 2020, 10, 42–81. [Google Scholar]
- Kong, H. The Use of Plants and Wildflowers as Bioremediation for Contaminated Soils in the Hong Kong S.A.R. Open J. Soil Sci. 2014, 4, 305–311. [Google Scholar] [CrossRef]
- Parvu, A.E.; Parvu, M.; Vlase, L.; Miclea, P.; Mot, A.C. Anti-Inflammatory Effects of Allium schoenoprasum L. Leaves. J. Physiol. Pharmacol. 2014, 65, 309–315. [Google Scholar]
- Rose, P.; Whiteman, M.; Moore, K.; Zhun, Y. Bioactive S-Alk(En)Yl Cysteine Sulfoxide Metabolites in the Genus Allium: The Chemistry of Potential Therapeutic Agents. Nat. Prod. Rep. 2005, 22, 351–368. [Google Scholar] [CrossRef]
- Singh, Y.; Baijnath, H. Agapanthus Campanulatus. Curtis’s Bot. Mag. 2018, 35, 106–124. [Google Scholar] [CrossRef]
- Duncan, A.; Jager, A.; Van-Staden, J. Screening of Zulu Medicinal Plants for Angiotensin Converting Enzyme (ACE) Inhibitors. J. Ethnopharmacol. 1999, 68, 63–70. [Google Scholar] [CrossRef]
- Sandoval-Herazo, L.C.; Alvarado-Lassman, A.; Marín-Muñiz, J.L.; Méndez-Contreras, J.M.; Zamora-Castro, S.A. Effects of the Use of Ornamental Plants and Different Substrates in the Removal of Wastewater Pollutants through Microcosms of Constructed Wetlands. Sustainability 2018, 10, 1594. [Google Scholar] [CrossRef]
- Miyahara, T.; Takahashi, M.; Ozeki, Y.; Sasaki, N. Isolation of an Acyl-Glucose-Dependent Anthocyanin 7-O-Glucosyltransferase from the Monocot Agapanthus africanus. J. Plant Physiol. 2012, 169, 1321–1326. [Google Scholar] [CrossRef] [PubMed]
- Kamara, B.I.; Manong, D.T.L.; Brandt, E.V. Isolation and Synthesis of a Dimeric Dihydrochalcone from Agapanthus africanus. Phytochemistry 2005, 66, 1126–1132. [Google Scholar] [CrossRef]
- Kaido, T.L.; Veale, D.J.H.; Havlik, I.; Rama, D.B.K. Preliminary Screening of Plants Used in South Africa as Traditional Herbal Remedies during Pregnancy and Labour. J. Ethnopharmacol. 1997, 55, 185–191. [Google Scholar] [CrossRef] [PubMed]
- University-of-Pretoria. Background Information on the Selected Plants. 2018. Available online: https://repository.up.ac.za/bitstream/handle/2263/25744/02chapter2.pdf?sequence=3&isAllowed=y (accessed on 8 October 2023).
- Van-Wyk, B. A Review of Commercially Important African Medicinal Plants. J. Ethnopharmacol. 2015, 176, 118–134. [Google Scholar] [CrossRef]
- Shawky, E. Phytochemical and Biological Investigation of Clivia Nobilis Flowers Cultivated in Egypt. Iran. J. Pharm. Res. 2016, 15, 531–535. [Google Scholar]
- Pérez, V.; Cambi, V.; Rueda, M.; Calfuán, M. Consequences of the Loss of Traditional Knowledge: The Risk of Injurious and Toxic Plants Growing in Kindergartens. Etnobotany Res. Appl. 2012, 10, 77–94. [Google Scholar] [CrossRef]
- Cimmino, A.; Masi, M.; Evidente, M.; Superchi, S.; Evidente, A. Amaryllidaceae Alkaloids: Absolute Configuration and Biological Activity. Chirality 2017, 29, 486–499. [Google Scholar] [CrossRef]
- Onder, A. Coriander and Its Phytoconstituents for the Beneficial Effects. IntechOpen 2018, 9, 165–185. [Google Scholar] [CrossRef]
- Barros, L.; Dueñas, M.; Dias, M.I.; Sousa, M.J.; Santos-Buelga, C.; Ferreira, I.C.F.R. Phenolic Profiles of In Vivo and In Vitro Grown Coriandrum sativum L. Food Chem. 2012, 132, 841–848. [Google Scholar] [CrossRef]
- Sahib, N.G.; Anwar, F.; Gilani, A.; Hamid, A.A.; Saari, N.; Alkharfy, K.M. Coriander (Coriandrum sativum L.): A Potential Source of High-Value Components for Functional Foods and Nutraceuticals—A Review. Phyther. Res. 2012, 27, 1439–1456. [Google Scholar] [CrossRef] [PubMed]
- Gaur, N.; Kukreja, A.; Yadav, M.; Tiwari, A. Assessment of Phytoremediation Ability of Coriander Sativum for Soil and Water Co-Contaminated with Lead and Arsenic: A Small-Scale Study. 3 Biotech 2017, 7, 196. [Google Scholar] [CrossRef] [PubMed]
- Laskar, M.; Ali, S.; Siddiqui, S. The Potential of Coriandrum sativum L. Seeds in the Remediation of Waste Water. Int. J. Adv. Sci. Technol. 2016, 86, 41–50. [Google Scholar] [CrossRef]
- Al-Snafi, A. Medicinal Plants with Central Nervous Effects (Part 2): Plant Based Review. Sch. Acad. J. Pharm. 2016, 5, 175–193. [Google Scholar] [CrossRef]
- Aydogan, A.; Sezer, K.; Ozmen, O.; Haligur, M.; Albay, M. Clinical and Pathological Investigations of Accidental Catharanthus Roseus Toxicity in Sheep. Isr. J. Vet. Med. 2015, 70, 51–56. [Google Scholar]
- Ikeura, H.; Kawasaki, Y.; Kaimi, E.; Nishiwaki, J.; Noborio, K.; Tamaki, M. Screening of Plants for Phytoremediation of Oil-Contaminated Soil. Int. J. Phytoremediat. 2016, 18, 460–466. [Google Scholar] [CrossRef]
- Ehsan, N.; Nawaz, R.; Ahmad, S.; Musaa, M.; Hayat, J.; Ehsan, N.; Nawaz, R.; Ahmad, S.; Khan, M.M.; Hayat, J. Phytoremediation of Chromium-Contaminated Soil by an Ornamental Plant, Vinca (Vinca rosea L.). J. Environ. Agric. Sci. 2016, 7, 29–34. [Google Scholar]
- Ramulondi, M.; Wet, H.D.; Vuuren, S. Van Toxicology of Medicinal Plants and Combinations Used in Rural Northern KwaZulu-Natal (South Africa) for the Treatment of Hypertension. Perspect. Med. 2018, 16, 100251. [Google Scholar] [CrossRef]
- Nugraha, A.; Keller, P.A. Revealing Indigenous Indonesian Traditional Medicine: Anti-Infective Agents. Nat. Prod. Commun. 2011, 6, 1953–1966. [Google Scholar] [CrossRef] [PubMed]
- Castillo, F.; Hernández, D.; Gallegos, G.; Rodríguez, R.; Aguilar, C. Antifungal Properties of Bioactive Compounds Form Plants. Intech Open 2014, 2, 64. [Google Scholar] [CrossRef]
- Bandara, V.; Weinstein, S.A.; White, J.; Eddleston, M. A Review of the Natural History, Toxinology, Diagnosis and Clinical Management of Nerium oleander (Common Oleander) and Thevetia peruviana (Yellow Oleander) Poisoning. Toxicon 2010, 56, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Tiwari, S.; Hasan, A.; Saxena, V.; Pandey, L.M. Recent Advances in Conventional and Contemporary Methods for Remediation of Heavy Metal—Contaminated Soils. 3 Biotech 2018, 8, 216. [Google Scholar] [CrossRef] [PubMed]
- Amrita, P.; Krishnaveni, K.; Neha, K.; Neethu, T.; Shanmugasundaram, R.; Sambathkumar, R. A Review on Management of Common Oleander and Yellow Oleander Poisoning. World J. Pharm. Pharm. Sci. 2016, 5, 493–503. [Google Scholar] [CrossRef]
- Arya, V.; Kumar, D.; Gautam, M. Phytopharmacological Review on Flowers: Source of Inspiration for Drug Discovery. Biomed. Prev. Nutr. 2014, 4, 45–51. [Google Scholar] [CrossRef]
- Balkan, I.A.; Doğan, H.T.; Zengin, G.; Colak, N.; Ayaz, F.A.; Gören, A.C.; Kırmızıbekmez, H.; Yeşilada, E. Enzyme Inhibitory and Antioxidant Activities of Nerium oleander L. Flower Extracts and Activity Guided Isolation of the Active Components. Ind. Crops Prod. 2018, 112, 24–31. [Google Scholar] [CrossRef]
- Zhang, J.; Yin, Z.-Q.; Liang, J.-Y. A New Isoflavonoid Glycoside from the Aerial Parts of Trachelospermum jasminoides. Chin. J. Nat. Med. 2013, 11, 274–276. [Google Scholar] [CrossRef]
- Sheu, M.J.; Chou, P.Y.; Cheng, H.C.; Wu, C.H.; Huang, G.J.; Wang, B.S.; Chen, J.S.; Chien, Y.C.; Huang, M.H. Analgesic and Anti-Inflammatory Activities of a Water Extract of Trachelospermum jasminoides (Apocynaceae). J. Ethnopharmacol. 2009, 126, 332–338. [Google Scholar] [CrossRef]
- Zhao, Z.; He, X.; Zhao, Y.; Sun, Y.; Chen, X.; Cun, Y.; Huang, L.; Bai, Y.; Zheng, X. Phytochemistry, Pharmacology and Traditional Uses of Plants from the Genus Trachelospermum L. Molecules 2017, 22, 1406. [Google Scholar] [CrossRef]
- Swith, S. In Vitro Studies with Aglaonema Commutatum Schott. 2014. Available online: https://plants.ces.ncsu.edu/plants/aglaonema-commutatum/ (accessed on 8 October 2023).
- Dahanayake, K.; Chow, C. Moisture Content, Ignitability, and Fire Risk of Vegetation in Vertical Greenery Systems. Fire Ecol. 2018, 14, 125–142. [Google Scholar] [CrossRef]
- Fokou, P.V.T.; Nyarko, A.K.; Appiah-Opong, R.; Yamthe, L.R.T.; Addo, P.; Asante, I.K.; Boyom, F.F. Ethnopharmacological Reports on Anti-Buruli Ulcer Medicinal Plants in Three West African Countries. J. Ethnopharmacol. 2015, 172, 297–311. [Google Scholar] [CrossRef]
- Clark, B.; Bliss, B.; Suzuki, J.; Borris, R. Chemotaxonomy of Hawaiian Anthurium Cultivars Based on Multivariate Analysis of Phenolic Metabolites. J. Agric. Food Chem. 2014, 62, 11323–11334. [Google Scholar] [CrossRef]
- Li, C.; Yang, G.; Huang, S.; Lü, D.; Wang, C.; Chen, J.; Yin, J. Characterisation of Flavonoids in Anthurium Spathes and Their Contribution to Spathe Colouration. J. Hortic. Sci. Biotechnol. 2013, 88, 208–215. [Google Scholar] [CrossRef]
- Benitez, M. Huertos Familiares y Alimentación de Grupos Domésticos Cafetaleros de La Sierra Madre de Chiapas. ECOSUR El Col. La Front. Sur 2017, 80. Available online: https://dialnet.unirioja.es/servlet/articulo?codigo=7556620 (accessed on 8 October 2023).
- Mounika, K.; Panja, B.; Saha, J. Diseases of Peace Lily [Spathiphyllum sp.] Caused by Fungi, Bacteria and Viruses: A Review. Pharma Innov. J. 2017, 6, 103–106. [Google Scholar]
- Rajkapoor, B.; Burkan, Z.E.; Senthilkumar, R. Oxidants and Human Diseases: Role of Antioxidant Medicinal Plants-A Review. Pharmacologyonline 2010, 1, 1117–1131. [Google Scholar]
- Bondareva, N.; Timchenko, L.; Dobrynya, Y.; Avanesyan, S.; Piskov, S.; Rzhepakovsky, I.; Kozlova, M.; Areshidze, D.; Lyhvar, A. Influence of the Chlorophytum Comosum Leaves Hydroalcoholic Extract on Some Representatives of Intestinal Microflora of Rats. J. Pharm. Sci. Res. 2017, 9, 874–877. [Google Scholar]
- Chauhan, R.; Quraishi, A.; Jadhav, S.K.; Keshavkant, S. A Comprehensive Review on Pharmacological Properties and Biotechnological Aspects of Genus Chlorophytum. Acta Physiol. Plant. 2016, 38, 116. [Google Scholar] [CrossRef]
- Pettit, T.; Irga, P.J.; Torpy, F.R. Towards Practical Indoor Air Phytoremediation: A Review. Chemosphere 2018, 208, 960–974. [Google Scholar] [CrossRef]
- Das, S.; Shil, P. Phytoremediation: A Cost-Effective Clean up Technique for Soil and Ground Water Contaminants. J. Environ. Res. Dev. 2012, 6, 1087–1091. [Google Scholar]
- Gawrońska, H.; Bakera, B. Phytoremediation of Particulate Matter from Indoor Air by Chlorophytum Comosum L. Plants. Air Qual. Atmos. Health 2015, 8, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Aberoumand, A. Assessment of Proximate and Phytochemical Composition for Evaluation of Nutritive Values of Some Plant Foods Obtained from Iran and India; Nokkoul, D.R., Ed.; In Tech: Rijeka, Croatia, 2011; ISBN 0958-3440. [Google Scholar]
- Machaca, F. Efecto Toxicológico Del Jincho Jincho (Heracium Neoherrerae), Altamisa (Ambrosia Arborescens), Diente de León (Taraxacum Officinale), Huira Huira (Pseudognaphalium Spicatum) y Mishico (Bidens Andicola) En Ratas (Wistar). Rev. Investig. Altoandinas-J. High Andean Res. 2014, 16, 43–50. [Google Scholar] [CrossRef]
- Bussmann, R.; Paniagua, N.; Moya, L.; Hart, R. Changing Markets-Medicinalplants in the Markets of La Paz and El Alto, Bolivia. J. Ethnopharmacol. 2016, 193, 76–95. [Google Scholar] [CrossRef] [PubMed]
- De-Feo, V.; Urrunaga, R. Medicinal Plants and Phytotherapy in Traditional Medicine of Paruro Province, Cusco Department, Peru. Pharmacologyonline 2012, 1, 154–219. [Google Scholar]
- Vinueza, D.R.; López, E.; Acosta, K.; Abdo, S. Assessment of Anti-Inflammatory Activity and Cytotoxicity of Freeze Dried Hydroalcoholic Extract of Bidens Andicola on Isolated Neutrophils. Asian J. Pharm. Clin. Res. 2017, 10, 160–163. [Google Scholar] [CrossRef]
- Ordónez, P.; Vega, M.; Malagón, O. Phytochemical Study of Native Plant Species Used in Traditional Medicine in Loja Province. Lyonia A J. Ecol. Appl. 2006, 10, 65–71. [Google Scholar]
- Castillo, M.; Quinatoa, E.; Risco, D.; Arnelas, I. Preliminary Phytochemical Screening of Some Andean Plants. Prelim. Phytochem. Screen. Some Andean Plants 2011, 2, 35–37. [Google Scholar]
- Shah, P.J.; Williamson, M.T. Antibacterial and Synergistic Activity of Calendula Officinalis Methanolic Petal Extract on Klebsiella Pneumoniae Co-Producing ESBL and AmpC Beta Lactamase. Int. J. Curr. Microbiol. Appl. Sci. 2015, 4, 107–117. [Google Scholar]
- Baskaran, K. Pharmacological Activities of Calendula Officinalis. Int. J. Sci. Res. 2015, 6, 43–47. [Google Scholar]
- Sausserde, R.; Kampuss, K. Composition of Carotenoids in Calendula (Calendula officinalis L.) Flowers. Foodbalt 2014, 2014, 13–18. [Google Scholar] [CrossRef]
- Hristozkova, M.; Geneva, M.; Stancheva, I.; Boychinova, M.; Djonova, E. Contribution of Arbuscular Mycorrhizal Fungi in Attenuation of Heavy Metal Impact on Calendula officinalis Development. Appl. Soil Ecol. 2016, 101, 57–63. [Google Scholar] [CrossRef]
- Santos, C.; Pereyra, A.; Patriarca, A.; Mazzobre, M.; Polak, T.; Abram, V.; Buera, M.; Poklar, N. Phenolic Compounds in Extracts from Eucalyptus Globulus Leaves and Calendula officinalis Flowers. J. Nat. Prod. Resour. 2016, 2, 53–57. [Google Scholar]
- Toiu, A.; Benedic, D.; Duda, M.; Hanganu, D.; Oniga, I. Determination of Total Carotenoid Content in Some Calendula officinalis and Tagestes patula Varieties. Hop Med. Plants 2016, XXIV, 57–62. [Google Scholar]
- Honório, I.C.G.; Bonfim, F.P.G.; Montoya, S.G.; Casali, V.V.D.; Leite, J.P.V.; Cecon, P.R. Growth, Development and Content of Flavonoids in Calendula (Calendula officinalis L.). Acta Sci. Agron. 2016, 38, 69. [Google Scholar] [CrossRef]
- Shoeb, M. Cytotoxic Compounds from the Genus Centaurea. Ph.D. Thesis, University of Aberdeen, Aberdeen, Scotland, 2017. [Google Scholar]
- Bruno, M.; Bancheva, S.; Rosselli, S.; Maggio, A. Sesquiterpenoids in Subtribe Centaureinae (Cass.) Dumort (Tribe Cardueae, Asteraceae): Distribution, 13C NMR Spectral Data and Biological Properties. Phytochemistry 2013, 95, 19–93. [Google Scholar] [CrossRef]
- Azzini, E.; Maiani, G.; Garaguso, I.; Polito, A.; Foddai, M.S.; Venneria, E.; Durazzo, A.; Intorre, F.; Palomba, L.; Rauseo, M.L.; et al. The Potential Health Benefits of Polyphenol-Rich Extracts from Cichorium intybus L. Studied on Caco-2 Cells Model. Oxid. Med. Cell. Longev. 2016, 2016, 1594616. [Google Scholar] [CrossRef] [PubMed]
- Bahmani, M.; Shahinfard, N.; Rafieian-Kopaei, M.; Saki, K.; Shahsavari, S.; Taherikalani, M.; Ghafourian, S.; Baharvand-Ahmadi, B. Chicory: A Review on Ethnobotanical Effects of Cichorium intybus L. J. Chem. Pharm. Sci. 2015, 8, 672–682. [Google Scholar]
- Derakhshani, Z.; Hassani, A.; Pirzad, A.; Abdollahi, R. Evaluation of Phenolic Content and Antioxidant Capacity in Some Medicinal Herbs Cultivated in Iran. Bot. Serbica 2012, 36, 117–122. [Google Scholar]
- Montefusco, A.; Semitaio, G.; Marrese, P.P.; Iurlaro, A.; De Caroli, M.; Piro, G.; Dalessandro, G.; Lenucci, M.S. Antioxidants in Varieties of Chicory (Cichorium intybus L.) and Wild Poppy (Papaver rhoeas L.) of Southern Italy. J. Chem. 2015, 2015, 923142. [Google Scholar] [CrossRef]
- Park, C.; Chae, S.; Park, S.; Kim, J.; Kim, Y.; Chung, S.; Arasu, M.; Al-Dhabi, N.; Park, S. Anthocyanin and Carotenoid Contents in Different Cultivars of Chrysanthemum (Dendranthema grandiflorum Ramat.) Flower. Molecules 2015, 20, 11090–11102. [Google Scholar] [CrossRef] [PubMed]
- Han, A.-R.; Kim, H.; So, Y.; Nam, B.; Lee, I.-S.; Nam, J.-W.; Jo, Y.; Kim, S.; Kim, J.-B.; Kang, S.-Y.; et al. Quantification of Antioxidant Phenolic Compounds in a New Chrysanthemum Cultivar by High-Performance Liquid Chromatography with Diode Array Detection and Electrospray Ionization Mass Spectrometry. Int. J. Anal. Chem. 2017, 2017, 1254721. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.; Wei, S. Factors Influencing Consumers’ Attitudes towards the Consumption of Edible Flowers. Food Qual. Prefer. 2017, 56, 93–100. [Google Scholar] [CrossRef]
- Sandhya, D.; Sowjanya, L.; Laxmi, S.; Sulakshana, M. Edible Flowers—A Review Article. Int. J. Adv. Res. Sci. Technol. 2014, 3, 51–57. [Google Scholar]
- Liang, Y.; Liu, J.; Zhang, S.P.; Wang, S.J.; Guo, W.H.; Wang, R.Q. Genetic Diversity of the Invasive Plant Coreopsis Grandiflora at Different Altitudes in Laoshan Mountain, China. Can. J. Plant Sci. 2008, 88, 831–837. [Google Scholar] [CrossRef]
- Valadon, L.; Summery, R. Carotenoids of Compositae Flowers. Phytochemistry 1971, 10, 2349–2353. [Google Scholar] [CrossRef]
- Hua, C.; Jian, L.; Yongli, Z.; Qiang, W.; Xiuli, G.; Yinghua, W.; Renqing, W. Influence of Invasive Plant Coreopsis Grandiflora on Functional Diversity of Soil Microbial Communities. J. Environ. Biol. 2011, 32, 567–572. [Google Scholar]
- Alsayari, A.; Muhsinah, A.B.; Hassan, M.Z.; Ahsan, M.J.; Alshehri, J.A.; Begum, N. Aurone: A Biologically Attractive Scaffold as Anticancer Agent. Eur. J. Med. Chem. 2019, 166, 417–431. [Google Scholar] [CrossRef]
- Eser, F.; Yaglioglu, A.S.; Dolarslan, M.; Aktas, E.; Onal, A. Dyeing, Fastness, and Cytotoxic Properties, and Phenolic Constituents of Anthemis tinctoria Var. Tinctoria (Asteraceae). J. Text. Inst. 2016, 108, 1489–1495. [Google Scholar] [CrossRef]
- Ozturk, M.; Sakcali, S.; Gucel, S.; Tombuloglu, H. Boron and Plants. In Plant Adaptation and Phytoremediation; Springer: Dordrecht, The Netherland, 2010; ISBN 978-90-481-9369-1. [Google Scholar]
- Rodrigues, N.; Ueda-Nakamura, T.; Dias, B.; Nakamura, C. Antitrypanosomal Activity of a Semi-Purified Subfraction Rich in Labdane Sesquiterpenes, Obtained from Flowers of Anthemis tinctoria, against Trypanosoma cruzi. Pharmacol. Pharm. 2011, 02, 47–55. [Google Scholar] [CrossRef]
- Hanganu, D.; Pintea, A.; Marculescu, A.; Toma, C.; Mirel, S. Chemical Research of Carotenoids from Anthemis tinctoria L. (Asteracea). Farmacia 2008, 56, 344–351. [Google Scholar]
- Lara-Cortés, E.; Martín-Belloso, O.; Osorio-Díaz, P.; Barrera-Necha, L.L.; Sánchez-López, J.A.; Bautista-Baños, S. Actividad Antioxidante, Composición Nutrimetal y Funcional de Flores Comestibles de Dalia. Rev. Chapingo Ser. Hortic. 2014, 20, 101–116. [Google Scholar] [CrossRef]
- Zhang, K.; Wang, J.; Guo, M.; Du, W.; Wu, R.; Wang, X. Short-Day Signals Are Crucial for the Induction of Anthocyanin Biosynthesis in Begonia semperflorens under Low Temperature Condition. J. Plant Physiol. 2016, 204, 1–7. [Google Scholar] [CrossRef]
- Lara-Cortés, E.; Troncoso-Rojas, R.; Hernández-López, M.; Bautista-Baños, S. Evaluation of the Antimicrobial Activity of Cinnamaldehyde in the Preservation of Edible Dahlia Flowers, under Different Storage Conditions. Rev. Chapingo Ser. Hortic. 2016, XXII, 177–189. [Google Scholar] [CrossRef]
- Munhoz, V.M.; Longhini, R.; Souza, J.R.; Zequi, J.A.; Mello, E.V.; Lopes, G.C.; Mello, J.C. Extraction of Flavonoids from Tagetes Patula: Process Optimization and Screening for Biological Activity. Braz. J. Pharmacogn. 2014, 24, 576–583. [Google Scholar] [CrossRef]
- Ortiz, D. Plantas Ornamentales de Noguera (Teruel) Angiospermas Dicotiledoneas (Ii). 2009, pp. 67–83. Available online: https://dialnet.unirioja.es/descarga/articulo/3097850.pdf (accessed on 8 October 2023).
- Santos, P.C.; Santos, V.H.M.; Mecina, G.F.; Andrade, A.R.; Fegueiredo, P.A.; Moraes, V.M.O.; Silva, L.P.; Silva, R.M.G. Phytotoxicity of Tagetes erecta L. and Tagetes patula L. on Plant Germination and Growth. South Afr. J. Bot. 2015, 100, 114–121. [Google Scholar] [CrossRef]
- Patel, M.; Newkar, B.; Baghel, P.; Pandey, B. Phytoremediation of Chemicals by Wedeliatrilobata, Tecomastans and Tageteserecta. Indian J. Sci. Res. 2014, 4, 165–169. [Google Scholar]
- Wei, J.L.; Lai, H.Y.; Chen, Z.S. Chelator Effects on Bioconcentration and Translocation of Cadmium by Hyperaccumulators, Tagetes patula and Impatiens walleriana. Ecotoxicol. Environ. Saf. 2012, 84, 173–178. [Google Scholar] [CrossRef]
- Park, Y.J.; Park, S.Y.; Arasu, M.V.; Al-Dhabi, N.A.; Ahn, H.G.; Kim, J.K.; Park, S.U.; Iriti, M. Accumulation of Carotenoids and Metabolic Profiling in Different Cultivars of Tagetes Flowers. Molecules 2017, 22, 313. [Google Scholar] [CrossRef]
- Gong, Y.; Liu, X.; He, W.; Xu, H.; Yuan, F.; Gao, Y. Investigation into the Antioxidant Activity and Chemical Composition of Alcoholic Extracts from Defatted Marigold (Tagetes erecta L.) Residue. Fitoterapia 2012, 83, 481–489. [Google Scholar] [CrossRef]
- Gakuubi, M.; Wanzala, W.; Wagacha, J. Bioactive Properties of Tagetes minuta L. (Asteraceae) Essential Oils: A Review. Am. J. Essent. Oils Nat. Prod. 2016, 4, 27–36. [Google Scholar]
- Deshmukh, V.; Rothe, S. Exotic Medicinal Plants from West Vidarbha Region VI. Biolife 2013, 2, 387–391. [Google Scholar]
- Jerves-Andrade, L.; León-Tamariz, F.; Peñaherrera, E.; Cuzco, N.; Tobar, V.; Ansaloni, R.; Maes, L.; Wilches, I. Medicinal Plants Used in South Ecuador for Gastrointestinal Problems: An Evaluation of Their Antibacterial Potential. J. Med. Plant Res. 2014, 8, 1310–1320. [Google Scholar] [CrossRef]
- Uqab, B.; Mudasir, S.; Nazir, R. Review on Bioremediation of Pesticides. J. Bioremediation Biodegrad. 2016, 7, 3–7. [Google Scholar] [CrossRef]
- Saciragie, S. Taraxacum Officinale Weber as Heavy Metal Absorber in Phytoremediation of Agricultural Land. Herbol. Int. J. Weed Res. Control 2011, 12, 20–21. [Google Scholar]
- Martínez, M.; Poirrier, P.; Chamy, R.; Prufer, D.; Schulze-GRonover, C.; Jorquera, L.; Ruiz, G. Taraxacum Officinale and Related Species an Ethnopharmacological Review and Its Potential as a Commercial Medicinal Plant. J. Ethnopharmacol. 2015, 169, 244–262. [Google Scholar] [CrossRef]
- Pădureţ, S.; Amariei, S.; Gutt, G.; Piscuc, B. The Evaluation of Dandelion (Taraxacum officinale) Properties as a Valuable Food Ingredient. Rom. Biotechnol. Lett. 2016, 21, 11569–11575. [Google Scholar]
- Huang, J.; Huang, C.; Liebman, M.; Assimos, D.G.; Huang, J.; Huang, C.; Liebman, M. Oxalate Contents of Commonly Used Chinese Medicinal Herbs. J. Tradit. Chin. Med. 2015, 196, 137–138. [Google Scholar] [CrossRef]
- Jaramillo, C.J.; Espinoza, A.J.; D’Armas, H.; Troccoli, L.; de Astudillo, L.R. Concentraciones de Alcaloides, Glucósidos Cianogénicos, Polifenoles y Saponinas En Plantas Medicinales Seleccionadas En Ecuador y Su Relación Con La Toxicidad Aguda Contra Artemia Salina. Rev. Biol. Trop. 2016, 64, 1171–1184. [Google Scholar] [CrossRef]
- Mohamed, A.H.; Ahmed, F.A.; Ahmed, O.K. Hepatoprotective and Antioxidant Activity of Zinnia Elegans Leaves Ethanolic Extract. Int. J. Sci. Eng. Res. 2015, 6, 154–161. [Google Scholar]
- Szewczyk, K.; Zidorn, C.; Biernasiuk, A.; Komsta, Ł.; Granica, S. Polyphenols from Impatiens (Balsaminaceae) and Their Antioxidant and Antimicrobial Activities. Ind. Crops Prod. 2016, 86, 262–272. [Google Scholar] [CrossRef]
- Grzeszczuk, M.; Stefaniak, A.; Anna, P.; Pachlowska, A.; Grzeszcsuk, M. Biological Value of Various Edible Flower Species. Acta Sci. Pol. Hortorum Cultus 2016, 15, 109–119. [Google Scholar]
- Sandgrid, S. Breeding of Begonia Tuberhybrida Using Modern Biotechnology. Masther´s Thesis, Norwegian University of Life Sciences, Aas, Norway, 2017. Available online: https://nmbu.brage.unit.no/nmbu-xmlui/bitstream/handle/11250/2459605/Sandgrind%20Sjur%20Master%27s%20thesis%20150817%20Breeding%20of%20Begonia%20tuberhybrida%20using%20modern%20biotechnology.pdf?sequence=1&isAllowed=y (accessed on 22 October 2023).
- Darwish, M.; Nassar, R.; Abdel-aziz, N.; Abdel-aal, A. Riboflavin Minimizes the Deleterious Effects of Salinity Stress on Growth, Chloroplast Pigments, Free Proline, Activity of Antioxidant Enzyme Catalase and Leaf Anatomy of Tecoma capensis (Thumb.) Lindl. Middle East J. Agric. 2017, 6, 757–765. [Google Scholar]
- Saini, N.K.; Singhal, M.; Srivastava, B. Evaluation of Antioxidant Activity of Tecomaria Capensis Leaves Extract. Inven. Rapid Ethnopharmacol 2014, 2011, 117. [Google Scholar]
- Verloove, F.; Salas-Pascual, M.; Rodríguez, Á.M. New Records of Alien Plants for the Flora of Gran Canaria (Canary Islands, Spain). Flora Mediterr. 2018, 28, 119–135. [Google Scholar] [CrossRef]
- Gidey, M.; Beyene, T.; Signorini, M.A.; Bruschi, P.; Yirga, G. Traditional Medicinal Plants Used by Kunama Ethnic Group in Northern Ethiopia. J. Med. Plants Res. 2015, 9, 494–509. [Google Scholar] [CrossRef]
- Hamed, M.; Mohamed, M.; Ibrahim, M. Cytotoxic Activity Assesment of Secondary Metabolites from Tecomaria capensis v. Aurea. Int. J. Pharmacogn. Phytochem. Res. 2016, 8, 1173–1182. [Google Scholar]
- Stafford, G.I.; Pedersen, M.E.; van Staden, J.; Jäger, A.K.; Staden, J.V.; Jäger, A.K.; van Staden, J.; Jäger, A.K. Review on Plants with CNS-Effects Used in Traditional South African Medicine against Mental Diseases. J. Ethnopharmacol. 2008, 119, 513–537. [Google Scholar] [CrossRef]
- Aguilar-Santamaría, L.; Ramírez, G.; Nicasio, P.; Alegría-Reyes, C.; Herrera-Arellano, A. Antidiabetic Activities of Tecoma stans (L.) Juss. Ex Kunth. J. Ethnopharmacol. 2009, 124, 284–288. [Google Scholar] [CrossRef]
- Sbihi, H.M.; Mokbli, S.; Nehdi, I.A. Physico-Chemical Properties of Tecoma stans Linn. Seed Oil: A New Crop for Vegetable Oil. Nat. Prod. Res. 2015, 29, 1249–1255. [Google Scholar] [CrossRef]
- Govindappa, M.; Sadananda, T.S.; Channabasava, R.; Jeevitha, M.K.; Pooja, K.S.; Vinay, B. Antimicrobial, Antioxidant Activity and Phytochemical Screening of Tecoma stans (L.) Juss. Ex Kunth. J. Phytol. 2011, 38, 68–76. [Google Scholar]
- Kays, S.J.; Hatch, J.; Dong, S.Y. Volatile Floral Chemistry of Heliotropium arborescens L. ‘Marine’. HortScience 2005, 40, 1237–1238. [Google Scholar] [CrossRef]
- Subramanian, P.; Rao, P.; Sudhakar, P.; Reddy, T.; Reddy, P. Pharmacognostic Evaluation of Heliotropium peruvianum L.: A Homoeopathic Drug. Indian J. Res. Homoeopath. 2013, 7, 103. [Google Scholar] [CrossRef]
- Zozomová-lihová, J.; Marhold, K.; Španiel, S. Taxonomy and Evolutionary History of Alyssum Montanum (Brassicaceae) and Related Taxa in Southwestern Europe and Morocco: Diversification Driven by Polyploidy, Geographic and Ecological Isolation. Taxon 2014, 63, 562–591. [Google Scholar] [CrossRef]
- Barzanti, R.; Colzi, I.; Arnetoli, M.; Gallo, A.; Pignattelli, S.; Gabbrielli, R.; Gonnelli, C. Cadmium Phytoextraction Potential of Different Alyssum Species. J. Hazard. Mater. 2011, 196, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Rajkumar, M.; Prasad, M.N.V.; Swaminathan, S.; Freitas, H. Climate Change Driven Plant-Metal-Microbe Interactions. Environ. Int. 2013, 53, 74–86. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Bueno, R.P.; Rincón-Cervera, M.A.; González-Fernández, M.J.; Guil-Guerrero, J.L. Phytochemical Composition and Antitumor Activities of New Salad Greens: Rucola (Diplotaxis tenuifolia) and Corn Salad (Valerianella locusta). Plant Foods Hum. Nutr. 2016, 71, 197–203. [Google Scholar] [CrossRef]
- Nicoletti, R.; Raimo, F.; Miccio, G. Diplotaxis Tenuifolia: Biology, Production and Properties. Eur. J. Plant Sci. Biotechnol. 2007, 1, 36–43. [Google Scholar]
- Ozturk, M.; Sakcali, S.; Celik, A. A Biomonitor of Heavy Metals on Ruderal Habitats in Turkey-Diplotaxis tenuifolia (L.) DC. Sains Malays. 2013, 42, 1371–1376. [Google Scholar]
- Bennett, R.; Rosa, E.; Mellon, F.; Kroon, P. Ontogenic Profiling of Glucosinolates, Flavonoids, and Other Secondary Metabolites in Eruca sativa (Salad Rocket), Diplotaxis erucoides (Wall Rocket), Diplotaxis tenuifolia (Wild Rocket), and Bunias orientalis (Turkish Rocket). J. Agric. Food Chem. 2006, 54, 4005–4015. [Google Scholar] [CrossRef]
- Pasini, F.; Verardo, V.; Caboni, M.F.; D’Antuono, L.F. Determination of Glucosinolates and Phenolic Compounds in Rocket Salad by HPLC-DAD-MS: Evaluation of Eruca sativa Mill. and Diplotaxis tenuifolia L. Genetic Resources. Food Chem. 2012, 133, 1025–1033. [Google Scholar] [CrossRef]
- Giordano, S.; Molinaro, A.; Spagnuolo, V.; Muscariello, L.; Ferrara, R.; Cennamo, G.; Aliotta, G. In Vitro Allelopathic Properties of Wild Rocket (Diplotaxis tenuifolia DC) Extract and of Its Potential Allelochemical S-Glucopyranosyl Thiohydroximate. J. Plant Interact. 2005, 1, 51–60. [Google Scholar] [CrossRef]
- Rasool, N.; Afzal, S.; Riaz, M.; Rashid, U.; Rizwan, K.; Zubair, M.; Ali, S.; Shahid, M. Evaluation of Antioxidant Activity, Cytotoxic Studies and GC-MS Profiling of Matthiola incana (Stock Flower). Legum. Res. 2013, 36, 21–32. [Google Scholar]
- Hadland, S.; Knigh, J.; Harris, S. Medical Marijuana: Review of the Science and Implications for Developmental Behavioral Pediatric Practice. J. Dev Behav Peatric 2015, 36, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Citterio, S.; Santagostino, A.; Fumagalli, P.; Prato, N.; Ranalli, P.; Sgorbati, S. Heavy Metal Tolerance and Accumulation of Cd, Cr and Ni by Cannabis sativa L. Plant Soil 2016, 1621, 36–43. [Google Scholar] [CrossRef]
- Tatsuzawa, F.; Saito, N.; Toki, K.; Shinoda, K.; Honda, T. Flower Colors and Their Anthocyanins in Matthiola Incana Cultivars (Brassicaceae). J. Jpn. Soc. Hortic. Sci. 2012, 81, 91–100. [Google Scholar] [CrossRef]
- Anna, S.; Premoli, M.; Tambaro, S.; Kumar, A.; Maccarinelli, G.; Memo, M.; Mastinu, A. Cannabis Sativa: A Comprehensive Ethnopharmacological Review of a Medicinal Plant with a Long History. J. Ethnopharmacol. 2018, 227, 300–315. [Google Scholar] [CrossRef]
- Koduru, S.; Grierson, D.S.; Afolayan, A.J. Ethnobotanical Information of Medicinal Plants Used for Treatment of Cancer in the Eastern Cape Province, South Africa. Curr. Sci. 2007, 92, 906–908. [Google Scholar] [CrossRef]
- Mansouri, H.; Asrar, Z.; Szopa, J. Effects of ABA on Primary Terpenoids and Δ9-Tetrahydrocannabinol in Cannabis sativa L. at Flowering Stage. Plant Growth Regul. 2009, 58, 269–277. [Google Scholar] [CrossRef]
- Andrade-Mahecha, M.; Tapia-Blácido, D.; Menegalli, F. Physical-Chemical, Thermal, and Functional Properties of Achira (Canna indica L.) Flour and Starch from Different Geographical Origin. Starch/Staerke 2012, 64, 348–358. [Google Scholar] [CrossRef]
- George, J. Screening and Antimicrobial Activity of Canna Indica against Clinical Pathogens Bioactive. Int. J. Life Sci. Educ. Res. 2014, 2, 85–88. [Google Scholar]
- Al-Snafi, A. Bioactive Components and Pharmacological Effects of Canna Indica—An Overview. Int. J. Pharmacol. Toxicol. 2015, 5, 71–75. [Google Scholar]
- Gaur, A.; Boruah, M.; Tyagi, D.K. Antimicrobial Potentials of Canna Indica Linn. Extracts against Selected Bacteria. J. Environ. Sci. Toxicol. Food Technol. 2014, 8, 22–23. [Google Scholar] [CrossRef]
- Chandra, S.; Rawat, D.S.; Chandra, D.; Rastogi, J. Nativity, Phytochemistry, Ethnobotany and Pharmacology of Dianthus caryophyllus. Res. J. Med. Plant 2016, 10, 1–9. [Google Scholar] [CrossRef][Green Version]
- Ohmiya, A.; Tanase, K.; Hirashima, M.; Yamamizo, C.; Yagi, M. Analysis of Carotenogenic Gene Expression in Petals and Leaves of Carnation (Dianthus caryophyllus L.). Plant Breed. 2013, 132, 423–429. [Google Scholar] [CrossRef]
- Amrouche, T.; Yang, X.; Capanoglu, E.; Huang, W.; Chen, Q.; Wu, L.; Zhu, Y.; Liu, Y.; Wang, Y. Contribution of Edible Flowers to the Mediterranean Diet: Phytonutrients, Bioactivity Evaluation and Applications. Food Front. 2022, 3, 592–630. [Google Scholar] [CrossRef]
- Leven, M.; Berghe, D.; Mertens, F.; Vlietinck, A.; Lammens, E. Screening of Higher Plants for Biological Activities I. Antimicrobial Activity. J. Med. Plant Res. 1979, 36, 311–321. [Google Scholar] [CrossRef]
- Rop, O.; Mlcek, J.; Jurikova, T.; Neugebauerova, J.; Vabkova, J. Edible Flowers—A New Promising Source of Mineral Elements in Human Nutrition. Molecules 2012, 17, 6672–6683. [Google Scholar] [CrossRef]
- Nho, K.J.; Chun, J.M.; Kim, H.K. Ethanol Extract of Dianthus chinensis L. Induces Apoptosis in Human Hepatocellular Carcinoma HepG2 Cells in Vitro. Evid. Based Complement. Altern. Med. 2012, 2012, 573527. [Google Scholar] [CrossRef]
- Mahgoub, M.H.; Aziz, G.A.; Abu-zied, K.M.; Mohamed, T.K. Effect of Khellinone Derivatives on Growth and Flowering of Dianthus chinensis L. Plant. Int. J. Adv. Res. 2015, 3, 130–138. [Google Scholar]
- Han, J.; Wang, Z.; Zheng, Y.; Zeng, G.; He, W.; Tan, N. New Dicyclopeptides from Dianthus chinensis. Acta Pharm. Sin. 2014, 49, 656–660. [Google Scholar] [CrossRef]
- Chou, S.-C.; Everngam, M.; Beck, J. Allelochemical Phenolic Acids from Gypsophila paniculata. J. Undergrad. Chem. Res. 2008, 7, 2–4. [Google Scholar]
- Mustafa, K.; Hasan, O. Economic Importance of Gypsophila L., Ankyropetalum fe nzl and Saponaria L. (Caryophyllaceae) Taxa of Turkey. Afr. J. Biotechnol. 2011, 10, 9533–9541. [Google Scholar] [CrossRef]
- Yücekutlu, A.N.; Bildacı, I. Determination of Plant Saponins and Some of Gypsophila Species: A Review of the Literature. Hacet. J. Biol. Chem. 2008, 36, 129–135. [Google Scholar]
- Smułek, W.; Zdarta, A.; Pacholak, A.; Zgoła-Grześkowiak, A.; Marczak, Ł.; Jarzębski, M.; Kaczorek, E. Saponaria officinalis L. Extract: Surface Active Properties and Impact on Environmental Bacterial Strains. Colloids Surf. B Biointerfaces 2017, 150, 209–215. [Google Scholar] [CrossRef]
- Kucukkurt, I.; Ince, S.; Enginar, H.; Eryavuz, A.; Fidan, A.F.; Kargioglu, M. Protective Effects of Agrostemma githago L. and Saponaria officinalis L. Extracts against Ionizing Radiation-Induced Oxidative Damage in Rats. Rev. Med. Vet. Toulouse 2011, 162, 289–296. [Google Scholar]
- Sengul, M.; Ercisli, S.; Yildiz, H.; Gungor, N.; Kavaz, A.; Cetin, B. Antioxidant, Antimicrobial Activity and Total Phenolic Content within the Aerial Parts of Artemisia absinthum, Artemisia santonicum and Saponaria officinalis. Iran. J. Pharm. Res. 2011, 10, 49–56. [Google Scholar] [PubMed]
- Ruiz-Riaguas, A.; Fernández-de Córdova, M.L.; Llorent-Martínez, E.J. Phenolic Profile and Antioxidant Activity of Euonymus japonicus Thunb. Nat. Prod. Res. 2020, 36, 3445–3449. [Google Scholar] [CrossRef]
- Chen, F.; Yao, Q.; Tian, J. Review of Ecological Restoration Technology. Eng. Rev. 2016, 36, 115–121. [Google Scholar]
- Jinbo, Z.; Mingan, W.; Wenjun, W.; Zhiqing, J.; Zhaonong, H. Insecticidal Sesquiterpene Pyridine Alkaloids from Euonymus Species. Phytochemistry 2002, 61, 699–704. [Google Scholar] [CrossRef]
- Hrichi, S.; Chaabane-Banaoues, R.; Giuffrida, D.; Mangraviti, D.; Oulad El Majdoub, Y.; Rigano, F.; Mondello, L.; Babba, H.; Mighri, Z.; Cacciola, F. Effect of Seasonal Variation on the Chemical Composition and Antioxidant and Antifungal Activities of Convolvulus althaeoides L. Leaf Extracts. Arab. J. Chem. 2020, 13, 5651–5668. [Google Scholar] [CrossRef]
- Hassine, M.; Znati, M.; Flamini, G.; Ben, H. Chemical Composition, Antibacterial and Cytotoxic Activities of the Essential Oil from the Flowers of Tunisian Convolvulus althaeoides L. Nat. Prod. Res. 2014, 28, 769–775. [Google Scholar] [CrossRef] [PubMed]
- Hassawi, D.; Kharma, A. Antimicrobial Activity of Some Medicinal Plants against Candida albicans. J. Biol. Sci. 2006, 6, 109–114. [Google Scholar]
- Al-Snafi, A.E. The Chemical Constituents and Pharmacological Effects of Convolvulus arvensis and Convolvulus scammonia—A Review. Indian J. Pharm. Sci. Res. 2015, 5, 172–185. [Google Scholar]
- Tawfeeq, A.; Hassan, I.; Mohammed, H.; AbdulHaffiD, Z.; Tawfeeq, A.; Hassan, I.; Al-Deen, H.; Kadhim, M.T.; AbdulHaffiD, Z. Convolvulus scammonia Crude Alkaloids Extract Induces Apoptosis through Microtubules Destruction in Mice Hepatoma H22 Cell Line. Iraqi J. Cancer Med. Genet. 2007, 5, 1. [Google Scholar] [CrossRef]
- Kulligowska, K.; Lutken, H.; Lopes, L.; Dibbern, M.; Nymark, J. Kalanchoe. Ornam. Crops 2018, 11, 453–479. [Google Scholar]
- Milad, R.; El-ahmady, S.; Singab, A.N. Genus Kalanchoe (Crassulaceae): A Review of Its Ethnomedicinal, Botanical, Chemical and Pharmacological Properties. Eur. J. Med. Plants 2014, 4, 86–104. [Google Scholar] [CrossRef]
- Nielsen, A.H.; Olsen, C.E. Flavonoids in Flowers of 16 Kalanchoë Blossfeldiana Varieties. Phytochemistry 2005, 66, 2829–2835. [Google Scholar] [CrossRef]
- Habibur, R.; Puramsetti, P.; Thumma, L.; Nukabathini, S.; Kumar, P.R. A Review on Ethnobotany, Phytochemistry and Pharmacology of Citrullus lanatus L. Int. Res. J. Pharm. Appl. Sci. 2013, 3, 77–81. [Google Scholar]
- Harith, S.S.; Mazlun, M.H.; Mydin, M.M.; Nawi, L.; Saat, R. Studies on Phytochemical Constituents and Antimicrobial Properties of Citrullus Lanatus Peels. Malays. J. Anal. Sci. 2018, 22, 151–156. [Google Scholar]
- Erhirhie, E.; Ekene, N. Medicinal Values on Citrullus lanatus (Watermelon): Pharmacological Review. Int. J. Res. Pharm. Biomed. Sci. 2013, 4, 1305–1312. [Google Scholar]
- Giwa, A.; Bello, I.; Oladipo, M.; Adeoye, D. Removal of Cadmium from Waste—Water by Adsorption Using the Husk of Melon (Citrullus Lanatus) Seed. Int. J. Basic Appl. Sci. 2013, 2, 110–123. [Google Scholar]
- Azevedo-Meleiro, C.H.; Rodriguez-Amaya, D.B. Qualitative and Quantitative Differences in Carotenoid Composition among Cucurbita moschata, Cucurbita maxima, and Cucurbita pepo. J. Agric. Food Chem. 2007, 55, 4027–4033. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Yu, X.; Maninder, M.; Xu, B. Total Phenolics and Antioxidants Profiles of Commonly Consumed Edible Flowers in China. Int. J. Food Prop. 2018, 21, 1524–1540. [Google Scholar] [CrossRef]
- Popescu, R.; Kopp, B. The Genus Rhododendron: An Ethnopharmacological and Toxicological Review. J. Ethnopharmacol. 2013, 147, 42–62. [Google Scholar] [CrossRef]
- Han, J.; He, G.-W.; Chen, Z.-W. Protective Effect and Mechanism of Total Flavones from Rhododendron simsii Planch on Endothelium-Dependent Dilatation and Hyperpolarization in Cerebral Ischemia-Reperfusion and Correlation to Hydrogen Sulphide Release in Rats. Evid. Based Complement. Altern. Med. 2014, 2014, 904019. [Google Scholar] [CrossRef]
- Gapuz, M.; Besagas, R. Phytochemical Profiles and Antioxidant Activities of Leaf Extracts of Euphorbia Species. Phytochem. Profiles Antioxid. Act. Leaf Extr. Euphorbia Species 2018, 12, 59–65. [Google Scholar]
- Pascal, O.; Virgyle, A.; Esaie, T.; Mawulé, H.-A.; Eloi, A. A Review of the Ethnomedical Uses, Phytochemistry and Pharmacology of the Euphorbia Genus. Pharma Innov. J. 2017, 6, 34–39. [Google Scholar] [CrossRef]
- Qaisar, M.; NaeemuddinGilani, S.; Farooq, S.; Rauf, A.; Naz, R.; Perveez, S. Preliminary Comparative Phytochemical Screening of Euphorbia Species. J. Agric. Environ. Sci. 2012, 12, 1056–1060. [Google Scholar] [CrossRef]
- Sierra, L.; Mejía, J.; Gualteros, L.; Córdoba, Y.; Martínez, J.; Stashenko, E. Combinación de Técnicas de Extracción y de Análisis Cromatográfico Para La Determinación de Metabolitos Secundarios en Flores Tropicales. Sci. Chromatogr. 2018, 10, 21–38. [Google Scholar]
- Bergius, P. Brownea Rosa-de-Monte. Láminas X XI XII XIII XIV 1916, 63, 2795. [Google Scholar]
- Coyago-Cruz, E.; Corell, M.; Meléndez-Martínez, A. Estudio Sobre el Contenido en Carotenoides y Compuestos Fenólicos de Tomates y Flores en el Contexto de la Alimentación Funcional; Punto Rojo Libros, S.L.: Sevilla, España, 2017; ISBN 9788417148096. [Google Scholar]
- Elansary, H.; Szopa, A.; Kubica, P.; Ekiert, H.; Ali, H.; Elshikh, M.; Abdel-salam, E.; El-Esawi, M.; El-Ensary, D. Bioactivities of Traditional Medicinal Plants in Alexandria. Evid. Based Complement. Altern. Med. 2018, 2018, 1463579. [Google Scholar] [CrossRef] [PubMed]
- Yuniarto, A.; Sukandar, E.; Fidrianny, I.; Setiawan, F.; Adnyana, I. Antiobesity, Antidiabetic and Antioxidant Activities of Senna (Senna alexandrina Mill.) and Pomegranate (Punica granatum L.) Leaves Extracts and Its Fractions. Int. J. Pharm. Phytopharm. Res. 2018, 8, 18–24. [Google Scholar]
- Alcaráz, L.; Mattana, C.; Satorres, S.; Petenatti, E.; Petenatti, M.; Del-Vitto, L.; Laciar, A. Antibacterial Activity of Extracts Obtained from Senna Corymbosa and Tipuana Tipu. Pharmacologyonline 2012, 3, 158–161. [Google Scholar]
- Del-Vitto, L.; Petenatti, E.; Petenatti, M. Recursos Herbolarios de San Luis (República Argentina) Primera Parte: Plantas Nativas. Mutequina 1997, 6, 49–66. [Google Scholar]
- Jeruto, P.; Arama, P.; Anyango, B.; Maroa, G. Phytochemical Screening and Antibacterial Investigations of Crude Methanol Extracts of Senna. J. Appl. Biosci. 2017, 11357–11367. [Google Scholar] [CrossRef]
- Nyamwamu, L.B.; Ngeiywa, M.; Mulaa, M.; Lelo, A.E.; Ingonga, J.; Kimutai, A. Acute Toxicity of Senna Didymobotrya Fresen Irwin Roots Used as a Traditional Medicinal Plant in Kenya. Am. Int. J. Contemp. Res. 2015, 5, 74–77. [Google Scholar]
- Nyaberi, M. Studies on the Use of Herbs to Preserve Meat and Milk among the Pastoral Communities of West Pokot in Kenya; Jomo Kenyatta University of Agriculture and Technology: Nairobi, Kenya, 2009. [Google Scholar]
- Chinchilla, M.; Valerio, I.; Sánchez, R.; Mora, V.; Bagnarello, V.; Martínez, L.; Gonzalez, A.; Vanegas, J.C.; Apestegui, Á. In Vitro Antimalarial Activity of Extracts of Some Plants from a Biological Reserve in Costa Rica. Rev. Biol. Trop. 2012, 60, 881–891. [Google Scholar] [CrossRef]
- Thabit, S.; Handoussa, H.; Roxo, M.; Azevedo, B.C.D.; Sayed, N.S.E.E.; Wink, M. Styphnolobium japonicum (L.) Schott Fruits Increase Stress Resistance and Exert Antioxidant Properties in Caenorhabditis Elegans and Mouse Models. Molecules 2019, 24, 2633. [Google Scholar] [CrossRef]
- He, X.; Bai, Y.; Zhao, Z.; Wang, X.; Fang, J.; Huang, L.; Zeng, M.; Zhang, Q.; Zhang, Y.; Zheng, X. Local and Traditional Uses, Phytochemistry, and Pharmacology of Sophora japonica L.: A Review. J. Ethnopharmacol. 2016, 187, 160–182. [Google Scholar] [CrossRef]
- Amer, M.; Ahmad, R.; Awwad, A. Biosorption of Cu(II), Ni(II), Zn(II) and Pb(II) Ions from Aqueous Solution by Sophora japonica Pods Powder. Int. J. Ind. Chem. 2015, 6, 67–75. [Google Scholar] [CrossRef]
- Yang, J.; Yao, D.; Li, X.; Zhang, Z. Research on Effect of Woody Plants Remediation Heavy Metal; Springer: Berlin/Heidelberg, Germany, 2013. [Google Scholar]
- Wang, F.; Miao, M.; Xia, H.; Yang, L.; Wang, S.; Sun, G. Antioxidant Activities of Aqueous Extracts from 12 Chinese Edible Flowers In Vitro and In Vivo. Food Nutr. Res. 2016, 61, 1265324. [Google Scholar] [CrossRef] [PubMed]
- Tava, A.; Pecio, Ł.; Scalzo, R.L.; Stochmal, A.; Pecetti, L. Phenolic Content and Antioxidant Activity in Trifolium germplasm from Different Environments. Molecules 2019, 24, 298. [Google Scholar] [CrossRef] [PubMed]
- Kolodziejczyk-Czepas, J. Trifolium Species—The Latest Findings on Chemical Profile, Ethnomedicinal Use and Pharmacological Properties Phytochemical Profile of Trifolium. Pharm. Pharmacol. 2016, 68, 845–861. [Google Scholar] [CrossRef] [PubMed]
- Saraswathi, J.; Venkatesh, K.; Baburao, N.; Hilal, M.H.; Rani, A.R. Phytopharmacological Importance of Pelargonium Species. J. Med. Plants Res. 2011, 5, 2587–2598. [Google Scholar]
- Mohammed, F. Phytochemical and Biological Study of Pelargonium peltatum (L.) L ’ Her. Family Geraniaceae Cultivated in Egypt. Master´s Thesis, Cairo University, Giza, Egypt, 2019. [Google Scholar]
- Singab, A.N.B.; El-Hefnawy, H.M.; El-Kolobby, D.G. Biological Studies Concerning the Antioxidant and Antimicrobial Activities of Pelargonium Species Cultivated in Egypt (Family-Geraniaceae). Int. Curr. Pharm. J. 2015, 4, 340–342. [Google Scholar] [CrossRef]
- Zawadzińska, A.; Salachna, P. Growth, Flowering and Photosyntetic Pigments of Pelargonium × Hortorum L.H. Bailey “Survivor Hot Pink” and “Graffiti Fire” Grown in Substrates Containing Sewage Sludge Compost. J. Ecol. Eng. 2015, 16, 168–176. [Google Scholar] [CrossRef]
- Gul, I.; Manzoor, M.; Ahmad, I.; Kallerhoff, J.; Arshad, M. Phytoaccumulation of Cadmium by Pelargonium × Hortorum—Tolerance and Metal Recovery. Environ. Sci. Pollut. Res. 2023, 30, 32673–32682. [Google Scholar] [CrossRef]
- Li, A.N.; Li, S.; Li, H.B.; Xu, D.P.; Xu, X.R.; Chen, F. Total Phenolic Contents and Antioxidant Capacities of 51 Edible and Wild Flowers. J. Funct. Foods 2014, 6, 319–330. [Google Scholar] [CrossRef]
- Wang, Z.; Ren, J.; Wang, Z. Current Status and Future Direction of Chinese Herbal Medicine. Trends Pharmacol. Sci. 2002, 23, 347–348. [Google Scholar] [CrossRef]
- Held, D.W.; Potter, D.A. Characterizing Toxicity of Pelargonium spp. and Two Other Reputedly Toxic Plant Species to Japanese Beetles (Coleoptera: Scarabaeidae). Plant Insect Interact. 2003, 32, 873–880. [Google Scholar] [CrossRef]
- He, B.; Guo, T.; Huang, H.; Xi, W.; Chen, X. Physiological Responses of Scaevola Aemula Seedlings under High Temperature Stress. South Afr. J. Bot. 2017, 112, 203–209. [Google Scholar] [CrossRef]
- Jo, S.; Kim, J.; Lee, N. Anti-Inflammatory and Anti-Bacterial Active Ingredients Derived from the Extract of the Leaves of Hydrangea Petiolaris. J. Soc. Cosmet. Sci. Korea 2020, 46, 207–218. [Google Scholar] [CrossRef]
- Cantor, M.; Tolety, J. Gladiolus. In Wild Crop Relatives: Genomic and Breeding Resources; Springer: Berlin/Heidelberg, Germany, 2020; pp. 1–23. ISBN 2013206534. [Google Scholar]
- Hasan, H.; Battikhi, A.; Qrunfleh, M. Impacts of Treated Wastewater Reuse on Some Soil Properties and Production of Gladiolus Communis. J. Hortic. 2014, 01, 1103–1118. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, Z.; Xia, P. Rugao Area Pterocarya Stenoptera Leaves Tannin Extraction Technology Research. Adv. Mater. Res. 2015, 1076, 210–215. [Google Scholar] [CrossRef]
- Cheng, H.-Y.; Lin, T.-C.; Yang, C.-M.; Wang, K.-C.; Lin, C.-C. Mechanism of Action of the Suppression of Herpes Simplex Virus Type 2 Replication by Pterocarnin A. Microbes Infect. 2004, 6, 738–744. [Google Scholar] [CrossRef]
- Kuo, P.-L.; Hsu, Y.-L.; Lin, T.-C.; Lin, L.-T.; Lin, C.-C. Induction of Apoptosis in Human Breast Adenocarcinoma MCF-7 Cells by Pterocarnin A from the Bark of Pterocarya Stenoptera via the Fas-Mediated Pathwar. Preclin. Rep. 2007, 18, 555–562. [Google Scholar] [CrossRef]
- Zielińska, S.; Matkowski, A. Phytochemistry and Bioactivity of Aromatic and Medicinal Plants from the Genus Agastache (Lamiaceae). Phytochem. Rev. 2014, 13, 391–416. [Google Scholar] [CrossRef]
- Ebadollahi, A.; Safaralizadeh, M.; Pourmirza, A.; Gheibi, S. Toxicity of Essential Oil of Agastache Foeniculum (Pursh) Kuntze to Oryzephilus surinamensis L. and Lasioderna serricorne F. J. Plant Prot. Res. 2010, 50, 215–219. [Google Scholar] [CrossRef]
- Yadikar, N.; Bobakulov, K.M.; Eshbakova, K.A.; Aisa, H.A. Phenolic Compounds from Lavandula angustifolia. Chem. Nat. Compd. 2017, 53, 562–564. [Google Scholar] [CrossRef]
- Nurzyńska-Wierdak, R.; Zawiślak, G. Chemical Composition and Antioxidant Activity of Lavender (Lavandula angustifolia Mill) Aboveground Parts. Acta Sci. Pol. Hortorum Cultus 2016, 15, 225–241. [Google Scholar]
- González-Tejero, M.; Casares-Porcel, M.; Sánchez-Rojas, C.; Ramiro-Gutiérrez, J.; Molero-Mesa, J.; Pieroni, A.; Giusti, M.; Censorii, E.; Pasquale, C.; Della, A.; et al. Medicinal Plants in the Mediterranean Area: Synthesis of the Results of the Project Rubia. J. Ethnopharmacol. 2008, 116, 341–357. [Google Scholar] [CrossRef] [PubMed]
- Del-Vitto, L.; Petenatti, E.; Petenatti, M. Recursos Herbolarios de San Luis (Argentina). Segunda Parte. Plantas Exóticas Cultivadas, Adventicias y/o Naturalizadas. Mutequina 1998, 7, 29–48. [Google Scholar]
- Pietrella, D.; Angiolella, L.; Vavala, E.; Rachini, A.; Mondello, F.; Ragno, R.; Bistoni, F.; Vecchiarelli, A. Beneficial Effect of Mentha Suaveolens Essential Oil in the Treatment of Vaginal Candidiasis Assessed by Real-Time Monitoring of Infection. BMC Complement. Altern. Med. 2011, 11, 18. [Google Scholar] [CrossRef]
- Oumzil, H.; Ghoulami, S.; Rhajaoui, M.; Ilidrissi, A.; Fkih-Tetouani, S.; Paid, M.; Benjouad, A. Antibacterial and Antifungal Activity of Essential Oils of Mentha Suaveolens. Phyther. Res. 2002, 16, 727–731. [Google Scholar] [CrossRef]
- Straumite, E.; Kruma, Z.; Galoburda, R. Pigments in Mint Leaves and Stems. Agron. Res. 2015, 13, 1104–1111. [Google Scholar]
- Ziarati, P.; Iranzad-asl, S.; Asgarpanah, J. Companion Pelargonium roseum and Rosmarinus officinalis in Cleaning up Contaminated Soil by Phytoextraction Technique the Role of Companion Plants in Boosting Phytoremediation Potential. Int. J. Plant Anim. Environ. Sci. 2014, 4, 424–430. [Google Scholar]
- Dumbravă, D.; Moldovan, C.; Raba, D.; Popa, M. Vitamin C, Chlorophylls, Carotenoids and Xanthophylls Content in Some Basil (Ocimum basilicum L.) and Rosemary (Rosmarinus officinalis L.) Leaves Extracts. J. Agroaliment. Process. Technol. 2012, 18, 253–258. [Google Scholar]
- Pradhita, O.; Manurung, R.; Inderaja, B.M.; Abduh, M.Y.; Hanifah, R. Factors Affecting Biomass Growth and Production of Essential Oil from Leaf and Flower of Salvia leucantha Cav. J. Essent. Oil Bear. Plants 2018, 21, 1021–1029. [Google Scholar] [CrossRef]
- Shaheen, U.Y.; Hussain, M.H.; Ammar, H.A. Cytotoxicity and Antioxidant Activity of New Biologically Active Constituents from Salvia lanigra and Salvia splendens. Pharmacogn. J. 2011, 3, 36–48. [Google Scholar] [CrossRef]
- Marchioni, I.; Najar, B.; Ruffoni, B.; Copetta, A.; Pistelli, L.; Pistelli, L. Bioactive Compounds and Aroma Profile of Some Lamiaceae Edible Flowers. Plants 2020, 9, 691. [Google Scholar] [CrossRef] [PubMed]
- Kandil, M.M.; Ibrahim, M.M.; El-Hanafy, S.H.; El-Sabwah, M.M. Effect of Putrescine and Uniconazole on Some Flowering Characteristics, and Some Chemical Constituents of Salvia splendens F. Plant. Int. J. ChemTech Res. 2015, 8, 174–186. [Google Scholar]
- Mathew, J.; Thoppil, J.E. Chemical Composition and Mosquito Larvicidal Activities of Salvia Essential Oils. Pharm. Biol. 2011, 49, 456–463. [Google Scholar] [CrossRef] [PubMed]
- Rajendran, R.; Prabha, A.L. AgNPs Synthesis, Characterization and Antibacterial Activity from Salvia splendens Sellow Ex Roem. & Schult. Plant Extract. Int. J. Sci. Res. 2013, 14, 2319–7064. [Google Scholar]
- Adrover, M.; Vadell, J.; Moya, G.; Martinez, A. Selection of Woody Species for Wastewater Enhancement and Restoration of Riparian Woodlands. J. Environ. Biol. 2008, 29, 357–361. [Google Scholar]
- Latoui, M.; Aliakbarian, B.; Casazza, A.A.; Seffen, M.; Converti, A.; Perego, P. Extraction of Phenolic Compounds from Vitex agnus-castus L. Food Bioprod. Process. 2012, 90, 748–754. [Google Scholar] [CrossRef]
- Van-Die, M.; Burger, H.; Teede, H.; Bone, K. Vitex Agnus-Castus Extracts for Female Reproductive Disorders: A Systematic Review of Clinical Trials. Planta Medica 2012, 79, 562–575. [Google Scholar] [CrossRef]
- Ghannadi, A.; Bagherinejad, M.R.; Abedi, D.; Jalali, M.; Absalan, B.; Sadeghi, N. Antibacterial Activity and Composition of Essential Oils from Pelargonium Graveolens L’Her and Vitex agnus-castus L. Iran. J. Microbiol. 2012, 4, 171–176. [Google Scholar]
- Souto, E.B.; Durazzo, A.; Nazhand, A.; Lucarini, M.; Zaccardelli, M.; Souto, S.B.; Silva, A.M.; Severino, P. Vitex agnus-castus L.: Main Features and Nutraceutical Perspectives. Forests 2020, 11, 761. [Google Scholar] [CrossRef]
- Wang, C.; Chen, L.; Yang, L. Antitumor Activity of Four Macrocyclic Ellagitannins from Cuphea hyssopifolia. Cancer Lett. 1999, 140, 195–200. [Google Scholar] [CrossRef]
- Elgindi, M.; Ayoub, N.; Milad, R.; Mekky, R. Antioxidant and Cytotoxic Activities of Cuphea hyssopifolia Kunth (Lythraceae) Cultivated in Egypt. J. Pharmacogn. Phytochem. 2012, 1, 68–78. [Google Scholar]
- Wang, S.; Wang, P.; Gao, L.; Yang, R.; Li, L.; Zhang, E.; Wang, Q.; Li, Y.; Yin, Z. Characterization and Complementation of a Chlorophyll-Less Dominant Mutant GL1 in Lagerstroemia indica. DNA Cell Biol. 2017, 36, 354–366. [Google Scholar] [CrossRef] [PubMed]
- Sivakumar, D.; Dhananjeyan, V.; Sathish Kumar, S.; Ashok Kumar, S.M. Fluoride Removal from Groundwater Using Lagerfstroemia indica L. Seed Powder. J. Chem. Pharm. Sci. 2015, 8, 784–789. [Google Scholar]
- Ashnagar, A.; Motakefpour, M.; Abbas, A.; Mehregan, I.; Grannadi, A. Persian Common Crape Myrtle Leaves; Phytochemical Screening and Flavonoid Patterns. J. Curr. Chem. Pharm. Sci. 2012, 2, 240–243. [Google Scholar]
- Chandra, M. Antimicrobial Activity of Medicinal Plants against Human Pathogenic Bacteria. Int. J. Biotechnol. Bioeng. Res. 2013, 4, 653–658. [Google Scholar]
- Wafa, B.A.; Makni, M.; Ammar, S.; Khannous, L.; Hassana, A.B.; Bouaziz, M.; Es-Safi, N.E.; Gdoura, R. Antimicrobial Effect of the Tunisian Nana Variety Punica granatum L. Extracts against Salmonella enterica (Serovars Kentucky and Enteritidis) Isolated from Chicken Meat and Phenolic Composition of Its Peel Extract. Int. J. Food Microbiol. 2017, 241, 123–131. [Google Scholar] [CrossRef]
- Kuppusamy, S.; Thavamani, P.; Megharaj, M.; Naidu, R. Bioremediation Potential of Natural Polyphenol Rich Green Wastes: A Review of Current Research and Recommendations for Future Directions. Environ. Technol. Innov. 2015, 4, 17–28. [Google Scholar] [CrossRef]
- Hajimahmoodi, M.; Moghaddam, G.; Ranjbar, A.M.; Khazani, H.; Sadeghi, N.; Oveisi, M.R.; Jannat, B. Total Phenolic, Flavonoids, Tannin Content and Antioxidant Power of Some Iranian Pomegranate Flower Cultivars (Punica granatum L.). Am. J. Plant Sci. 2013, 2013, 1815–1820. [Google Scholar] [CrossRef]
- Shaygannia, E.; Bahmani, M.; Zamanzad, B.; Rafieian-Kopaei, M. A Review Study on Punica granatum L. J. Evid. Based Complement. Altern. Med. 2015, 21, 221–227. [Google Scholar] [CrossRef]
- Sokkar, N.M.; Rabeh, M.A.; Ghazal, G.; Slem, A.M. Determination of Flavonoids in Stamen, Gynoecium, and Petals of Magnolia grandiflora L. and Their Associated Antioxidant and Hepatoprotection Activities. Quim. Nova 2014, 37, 667–671. [Google Scholar] [CrossRef]
- Malik, S.; Ahmad, M.; Khan, F. Qualititative and Quantitative Estimation of Terpenoid Contents in Some Important. Pak. J. Sci. 2017, 69, 2017. [Google Scholar]
- Krüger, C.; Malheiros, C.; Sayonara, J.; Silva, B.; Hofmann, T.C.; Messina, T.M.; Manfredini, V.; Escobar, C.; Flávio, L.; Oliveira, S.; et al. Preliminary in Vitro Assessment of the Potential Toxicity and Antioxidant Activity of Ceiba speciosa (A. St. -Hill) Ravenna (Paineira). Braz. J. Pharm. Sci. 2015, 53, 1–12. [Google Scholar]
- Vankar, P.; Sarswat, R.; Sahu, R. Biosorption of Zinc Ions from Aqueous Solutions onto Natural Dye Waste of Hibiscus rosa Sinensis: Thermodynamic and Kinetic Studies. Environ. Prog. Sustain. Energy 2011, 79, 975–979. [Google Scholar] [CrossRef]
- Pillai, S.S.; Mini, S. Polyphenols Rich Hibiscus Rosa Sinensis Linn. Petals Modulate Diabetic Stress Signalling Pathways in Streptozotocin-Induced Experimental Diabetic Rats. J. Funct. Foods 2016, 20, 31–42. [Google Scholar] [CrossRef]
- Sharma, K.; Pareek, A.; Chauhan, E. Evaluation of Hyperglycemic and Hyperlipidemic Mitigating Impact of Hibiscus rosa Sinensis (Gudhal) Flower in Type II Diabetes Mellitus Subjects. Int. J. Appl. Biol. Pharm. Technol. 2016, 2000, 223–229. [Google Scholar]
- Pooja, A.; Shruti, D.; Rohit, C.; Hemalata, S.; Vikas, B.; Vinod, C.; Anil, K. Antimicrobial Activity of Ethno-Medicinal Plants against Cariogenic Pathogens. J. Med. Plants Stud. 2016, 4, 283–290. [Google Scholar]
- Mohd-Esa, N.; Hern, F.S.; Ismail, A.; Yee, C.L. Antioxidant Activity in Different Parts of Roselle (Hibiscus sabdariffa L.) Extracts and Potential Exploitation of the Seeds. Food Chem. 2010, 122, 1055–1060. [Google Scholar] [CrossRef]
- Da-Costa-Rocha, I.; Bonnlaender, B.; Sievers, H.; Pischel, I.; Heinrich, M. Hibiscus sabdariffa L.—A Phytochemical and Pharmacological Review. Food Chem. 2014, 165, 424–443. [Google Scholar] [CrossRef]
- Younes, M.A.; Nicaise, L.A.; Bertrand, M.B. Polymerization Degree of Phytochelatin in Contaminated Soil Phytoremediation of Manganese in Hibiscus sabdariffa Linn Var Sabdariffa. Eur. Sci. J. 2016, 12, 482–492. [Google Scholar] [CrossRef][Green Version]
- Riaz, G.; Chopra, R. A Review on Phytochemistry and Therapeutic Uses of Hibiscus sabdariffa L. Biomed. Pharmacother. 2018, 102, 575–586. [Google Scholar] [CrossRef]
- Teixeira, M.; Tao, W.; Fernandes, A.; Faria, A.; Ferreira, I.M.; He, J.; de Freitas, V.; Mateus, N.; Oliveira, H. Anthocyanin-Rich Edible Flowers, Current Understanding of a Potential New Trend in Dietary Patterns. Trends Food Sci. Technol. 2023, 138, 708–725. [Google Scholar] [CrossRef]
- Yang, J.E.; Ngo, H.T.T.; Hwang, E.; Seo, S.A.; Park, S.W.; Yi, T.H. Dietary Enzyme-Treated Hibiscus syriacus L. Protects Skin against Chronic UVB-Induced Photoaging via Enhancement of Skin Hydration and Collagen Synthesis. Arch. Biochem. Biophys. 2019, 662, 190–200. [Google Scholar] [CrossRef] [PubMed]
- Vasudeva, N.; Sharma, S.K. Biologically Active Compounds from the Genus Hibiscus. Pharm. Biol. 2008, 46, 145–153. [Google Scholar] [CrossRef]
- Sharma, A.; Flores-Vallejo, R.; Cardoso-Taketa, A.; Villarreal, M. Antibacterial Activities of Medicinal Plants Used in Mexican Traditional Medicine. J. Ethnopharmacol. 2017, 208, 264–329. [Google Scholar] [CrossRef]
- Abdelhafez, O.; Fawzy, M.; Fahim, J.; Desoukey, S.; Krischke, M.; Mueller, M.; Abdelmohsen, U. Hepatoprotective Potential of Malvaviscus Arboreus against Carbon Tetrachloride-Induced Liver Injury in Rats. PLoS ONE 2018, 13, e0202362. [Google Scholar] [CrossRef] [PubMed]
- Yasunaka, K.; Abe, F.; Nagayama, A.; Okabe, H.; Lozada-P, L.; Edith, L.; Mu, E.E. Antibacterial Activity of Crude Extracts from Mexican Medicinal Plants and Purified Coumarins and Xanthones. J. Ethnopharmacol. 2005, 97, 293–299. [Google Scholar] [CrossRef]
- Allah, M.; Mohamed, B.E.; Halim, A. Phytochemical and Biological Study of Bougainvillea Spectabilis Family Nyctaginaceae Growing in Egypt. Master´s Thesis, Cairo University, Giza, Egypt, 2016. [Google Scholar]
- Juson, A.; Martinez, M.; Ching, J. Accumulation and Distribution of Heavy Metals in Leucaena Leucocephala Lam. and Bougainvillea spectabilis Willd. Plant Systems. J. Exp. Biol. Agric. Sci. 2016, 4, 1–6. [Google Scholar] [CrossRef]
- Petrova, I.; Petkova, N.; Ivanov, I. Five Edible Flowers—Valuable Source of Antioxidants in Human Nutrition. Int. J. Pharmacogn. Phytochem. Res. 2016, 8, 604–610. [Google Scholar] [CrossRef][Green Version]
- Bhat, M.; Zinjarde, S.; Bhargava, S.; Kumar, A.; Joshi, B. Antidiabetic Indian Plants: A Good Source of Potent Amylase Inhibitors. Evid. Based Complement. Altern. Med. 2011, 2011, 810207. [Google Scholar] [CrossRef]
- Khalid, D.; Sarray, A.; Horiacha, L.M.; Zhuravel, I.O.; Fedosov, A.I. HPLC Study of Phenolic Compounds in Mirabilis jalapa Raw Material. Pharmacia 2020, 67, 145–152. [Google Scholar] [CrossRef]
- Gogoi, J.; Nakhuru, K.; Policegoudra, R.; Chattopadhyay, P.; Rai, A.; Veer, V. Isolation and Characterization of Bioactive Components from Mirabilis jalapa L. Radix. J. Tradit. Complement. Med. 2016, 6, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Nath, L.; Manjunath, K.; Savadi, R.; Akki, K. Anti-Inflammatory Activity of Mirabilis jalapa Linn. Leaves. J. Basic Clin. Pharm. 2010, 1, 93–96. [Google Scholar] [PubMed]
- Sabharwal, S.; Sudan, S.; Ranjan, V. Jasminum sambac Linn (Motia) a Review. Int. J. Pharm. Res. Bio Sci. 2013, 2, 108–130. [Google Scholar]
- Mandal, A.; Purakayastha, T.J.; Ramana, S.; Neenu, S.; Bhaduri, D.; Chakraborty, K.; Manna, M.C.; Rao, A.S. Status on Phytoremediation of Heavy Metals in India—A Review. Int. J. Bio Resour. Stress Manag. 2014, 5, 553. [Google Scholar] [CrossRef]
- Tom, K.M.; Benny, P.J. A Review of Phytopharmacological Studies on Some Common Flowers. Int. J. Curr. Pharm. Rev. Res. 2016, 7, 171–180. [Google Scholar]
- Jordheim, M.; Skaar, I.; Lunder, H.; Andersen, Ø.M. Anthocyanins from Fuchsia Flowers. Nat. Prod. Commun. 2011, 6, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Lucrecia, A.N.A.; John, E.D.E.M.Y. Diversidad, Distribución e Importancia Económica de Passifloraceae de Guatemala. Biodiversidad 2014, 2, 17–33. Available online: https://www.researchgate.net/publication/263149293_Diversidad_distribucion_e_importancia_economica_de_Passifloraceae_de_Guatemala (accessed on 8 October 2023).
- Abourashed, E.A.; Vanderplank, J.R.; Khan, I.A. High-Speed Extraction and HPLC Fingerprinting of Medicinal Plants—I. Application to Passiflora Flavonoids. Pharm. Biol. 2002, 40, 81–91. [Google Scholar] [CrossRef]
- González-Barrio, R.; Periago, M.J.; Luna-Recio, C.; Garcia-Alonso, F.J.; Navarro-González, I. Chemical Composition of the Edible Flowers, Pansy (Viola wittrockiana) and Snapdragon (Antirrhinum majus) as New Sources of Bioactive Compounds. Food Chem. 2018, 252, 373–380. [Google Scholar] [CrossRef]
- Cui, S.; Zhang, T.; Zhao, S.; Li, P. Evaluation of Three Ornamental Plants for Phytoremediation of Pb Contamined Soil. Int. J. Phytoremediation 2013, 15, 299–306. [Google Scholar] [CrossRef]
- Liu, R.; Jadeja, R.; Zhou, Q.; Liu, Z. Treatment and Remediation of Petroleum-Contaminated Soils Using Selective Ornamental Plants. Environ. Eng. Sci. 2012, 29, 494–501. [Google Scholar] [CrossRef] [PubMed]
- Schrader, K.K.; Andolfi, A.; Cantrell, C.L.; Cimmino, A.; Duke, S.O.; Osbrink, W.; Wedge, D.E.; Evidente, A. A Survey of Phytotoxic Microbial and Plant Metabolites as Potential Natural Products for Pest Management. Chem. Biodivers. 2010, 7, 2261–2280. [Google Scholar] [CrossRef] [PubMed]
- Graf, B.; Rojas-Silva, P.; Baldeón, M. Discovering the Pharmacological Potential of Ecuadorian Market Plants Using a Screens-to-Nature Participatory Approach. J. Biodivers. Bioprospect. Dev. 2016, 3, 1–9. [Google Scholar] [CrossRef]
- Mahmood, Q.; Rashid, A.; Ahmad, S.; Azim, M.; Bilal, M. Current Status of Toxic Metals Addition to Environment and Its Consequences. Environmental Pollition. 2021, pp. 1–27. Available online: https://link.springer.com/chapter/10.1007/978-94-007-3913-0_2 (accessed on 8 October 2023).
- Fitsiou, E.; Mitropoulou, G.; Spyridopoulou, K.; Tiptiri-Kourpeti, A.; Vamvakias, M.; Bardouki, H.; Panayiotidis, M.; Galanis, A.; Kourkoutas, Y.; Chlichlia, K.; et al. Phytochemical Profile and Evaluation of the Biological Activities of Essential Oils Derived from the Greek Aromatic Plant Species Ocimum basilicum, Mentha spicata, Pimpinella anisum and Fortunella margarita. Molecules 2016, 21, 1069. [Google Scholar] [CrossRef] [PubMed]
- Lichtenthaler, H.K. Chlorophylls and Carotenoids: Pigments of Photosynthetic Biomembranes. Methods Enzymol. 1987, 148, 350–382. [Google Scholar] [CrossRef]
- Chen, G.-L.; Chen, S.-G.; Xiao, Y.; Fu, N.-L. Antioxidant Capacities and Total Phenolic Contents of 30 Flowers. Ind. Crops Prod. 2018, 111, 430–445. [Google Scholar] [CrossRef]
- Evci, G.; Pekcan, V.; Yilmaz, I.; Citak, N.; Tuna, N.; Ay, O.; Pilash, A.; Kaya, Y. Determination of Yield Performances of Oleic Type Sunflower (Helianthus annuus L.) Hybrids Resistant to Broomrape and Downy Mildew. Ekin J. Crop Breed. Genet. 2016, 2, 45–50. [Google Scholar]
- Nirola, R.; Megharaj, M.; Beecham, S.; Aryal, R.; Thavamani, P.; Vankateswarlu, K.; Saint, C. Remediation of Metalliferous Mines, Revegetation Challenges and Emerging Prospects in Semi-Arid and Arid Conditions. Environ. Sci. Pollut. Res. 2016, 23, 20131–20150. [Google Scholar] [CrossRef]
- Jaradat, N.A.; Zaid, A.N.; Hussein, F. Investigation of the Antiobesity and Antioxidant Properties of Wild Plumbago Europaea and Plumbago Auriculata from North Palestine. Chem. Biol. Technol. Agric. 2016, 3, 31. [Google Scholar] [CrossRef]
- Singh, K.; Naidoo, Y.; Baijnath, H. A Comprehensive Review on the Genus Plumbago with Focus on Plumbago auriculata (Plumbaginaceae). Afr. J. Tradit. Complement. Altern. Med. 2018, 15, 199–215. [Google Scholar] [CrossRef]
- Tudorel Olaru, O.; Iuliana Anghel, A.; Istudor, V.; Viorel Ancuceanu, R.; Dinu, M.; Tudorel, O.; Anghel, A.I.; Istudor, V.; Ancuceanu, R.V. Contributions to the Pharmacognostical and Phytobiological Study of Fallopia aubertii (L. Henry) Holub. (Polygonaceae). Farmacia 2013, 61, 991–999. [Google Scholar]
- Alyazouri, A.; Jewsbury, R.; Tayim, H.; Humphreys, P.; Al-Sayah, M. Phytoextraction of Cr ( VI ) from Soil Using Portulaca oleracea. Toxicol. Environ. Chem. 2013, 95, 1338–1347. [Google Scholar] [CrossRef]
- Wang, Y.; Fujinami, K.; Zhang, R.; Wan, C.; Wang, N.; Ba, Y.; Koumoto, K. Antibacterial Attributes of Apigenin, Isolated from Portulaca oleracea L. Int. J. Bacteriol. 2014, 3, 031101. [Google Scholar] [CrossRef]
- Habibian, M.; Sadeghi, G.; Karimi, A. Phytochemicals and Antioxidant Properties of Solvent Extracts from Purslane (Portulaca oleracea L.): A Preliminary Study. Food Sci. Eng. 2020, 1, 1–12. [Google Scholar] [CrossRef]
- Ramadan, B.K.; Schaalan, M.F.; Tolba, A.M. Hypoglycemic and Pancreatic Protective Effects of Portulaca Oleracea Extract in Alloxan Induced Diabetic Rats. BMC Complement. Altern. Med. 2017, 17, 37. [Google Scholar] [CrossRef]
- Takamura, T.; Morino, T. Coloration and Pigmentation in Petals of Some Cyanic Ranunculus Cultivars. Acta Hortic. 2019, 1237, 251–257. [Google Scholar] [CrossRef]
- Ali, B.; Shanab, A.; Adwan, G.M.; Adwan, K.M.; Bassam, F. Efficacy of Aqueous and Ethanol Extracts of Some Palestinian Medicinal Plants for Potential Antibacterial Activity. Islam. Univ. J. 2008, 16, 77–86. [Google Scholar]
- Giampieri, F.; Alvarez-Suarez, J.; Mazzoni, L.; Romandini, S.; Bompadre, S.; Diamanti, J.; Capocasa, F.; Mezzetti, B.; Quiles, J.; Ferreiro, M.; et al. The Potential Impact of Strawberry on Human Health. Nat. Prod. Res. 2013, 27, 448–455. [Google Scholar] [CrossRef]
- Dos-Santos, A.; Silva, E.; Dos-Santos, W.; Da-Silva, E.; Dos-Santos, L.; Bruna, B.; Maria, M.; Dos-Santos, W. Evaluation of Minerals, Toxic Elements and Bioactive Compounds in Rose Petals (Rosa spp.) Using Chemometric Tools and Artificial Neural Networks. Microchem. J. 2018, 138, 98–108. [Google Scholar] [CrossRef]
- Schmitzer, V.; Veberic, R.; Stampar, F. Prohexadione-Ca Application Modifies Flavonoid Composition and Color Characteristics of Rose (Rosa hybrida L.) Flowers. Sci. Hortic. 2012, 146, 14–20. [Google Scholar] [CrossRef]
- Lee, J.H.; Lee, H.J.; Choung, M.G. Anthocyanin Compositions and Biological Activities from the Red Petals of Korean Edible Rose (Rosa hybrida Cv. Noblered). Food Chem. 2011, 129, 272–278. [Google Scholar] [CrossRef]
- Uddin, R.; Saha, M.R.; Subhan, N.; Hossain, H.; Jahan, I.A.; Akter, R. HPLC-Analysis of Polyphenolic Compounds in Gardenia jasminoides and Determination of Antioxidant Activity by Using Free Radical Scavenging Assays. Adv. Pharm. Bull. 2014, 4, 273–281. [Google Scholar] [PubMed]
- Zheng, N.; Zhao, Y.; Song, Q.; Jia, L.; Fang, W. Biomass Assisted Synthesis of Alumina by Gardenia jasminoides Ellis and Their Application for Removal of Ni (II) from Aqueous Solution. J. Hazard. Mater. 2013, 260, 1057–1063. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Li, M.; Yang, Z.; Tao, W.; Wang, P.; Tian, X.; Li, X.; Wang, W. Gardenia jasminoides Ellis: Ethnopharmacology, Phytochemistry, and Pharmacological and Industrial Applications of an Important Traditional Chinese Medicine. J. Ethnopharmacol. 2020, 257, 112829. [Google Scholar] [CrossRef] [PubMed]
- Dontha, S.; Kamurthy, H.; Mantripragada, B. Phytochemical and Pharmacological Profile of Ixora: A Review. Int. J. Pharm. Sci. Res. 2015, 6, 567–584. [Google Scholar] [CrossRef]
- Bhosale, S.; Rathor, B.; Dhabe, A. Survey of Medicinal Plants Used on Wounds by Local People of Shirol Tahasil of Maharashtra State, India. Int. J. Curr. Res. Acad. Rev. 2016, 4, 81–86. [Google Scholar] [CrossRef][Green Version]
- Anolieto, G.; Ikhajiagbe, B.; Okonokhua, B.; Edegbai, B.; Obasuyi, D. Metal Tolerant Species Distribution and Richness in and around the Metal Based Industries: Possible Candidates for Phytoremediation. Full Length Res. Pap. 2008, 2, 360–370. [Google Scholar]
- Duncan, E.J. A Review on Warszewiczia Coccinea (Vahl) Klotzsch—The ‘Chaconia’. Living World J. Trinidad Tobago F. Nat. Club 2007, 1, 1–7. [Google Scholar]
- Sadilova, E.; Stintzing, F.C.; Carle, R. Anthocyanins, Colour and Antioxidant Properties of Eggplant (Solanum melongena L.) and Violet Pepper (Capsicum annuum L.) Peel Extracts. Z. Fur Naturforsch. Sect. C J. Biosci. 2006, 61, 527–535. [Google Scholar] [CrossRef]
- Alcántara, E.; Barra, R.; Benlloch, M.; Ginhas, A.; Jorrín, J.; López, J.; Lora, Á.; Ojeda, M.; Puig, M.; Pujadas, A.; et al. Phytoremediation of a Metal Contaminated Area in Southern Spain. Minerva Biotecnol. 2001, 13, 33–36. [Google Scholar]
- Teerakun, M.; Reungsang, A. Determination of Plant Species for the Phytoremediation of Carbofuran Residue in Rice Field Soils. Songklanakarin J. Sci. Technol. 2005, 27, 967. [Google Scholar]
- Srinivasan, K. Biological Activities of Red Pepper (Capsicum annuum) and Its Pungent Principle Capsaicin: A Review. Crit. Rev. Food Sci. Nutr. 2015, 56, 1488–1500. [Google Scholar] [CrossRef] [PubMed]
- Rashed, K.; Sahuc, M.-E.; Deloison, G.; Calland, N.; Brodin, P.; Rouillé, Y.; Séron, K. Potent Antiviral Activity of Solanum Rantonnetii and the Isolated Compounds against Hepatitis C Virus In Vitro. J. Funct. Foods 2014, 11, 185–191. [Google Scholar] [CrossRef]
- Bondareva, L.; Teisserenc, R.; Pakharkova, N.; Shubin, A.; Le Dantec, T.; Renon, L.; Svoboda, I. Assessment of the Bioavailability of Cu, Pb, and Zn through Petunia axillaris in Contaminated Soils. Int. J. Ecol. 2014, 2014, 378642. [Google Scholar] [CrossRef]
- Cna’ani, A.; Shavit, R.; Ravid, J.; Aravena-Calvo, J.; Skaliter, O.; Masci, T.; Vainstein, A. Phenylpropanoid Scent Compounds in Petunia × Hybrida Are Glycosylated and Accumulate in Vacuoles. Front. Plant Sci. 2017, 8, 1898. [Google Scholar] [CrossRef]
- Jonsson, L.M.V.; Aarsman, M.E.G.; Schram, A.W.; Bennink, G.J.H. Methylation of Anthocyanins by Cell-Free Extracts of Flower Buds of Petunia hybrida. Phytochemistry 1982, 21, 2457–2459. [Google Scholar] [CrossRef]
- Molesini, B.; Treggiari, D.; Dalbeni, A.; Minuz, P. Plant Cystine-Knot Peptides: Pharmacological Perspectives. Br. J. Clin. Pharmacol. 2017, 83, 63–70. [Google Scholar] [CrossRef]
- Dorais, M.; Ehret, D.; Papadopoulos, A. Tomato (Solanum lycopersicum) Health Components: From the Seed to the Consumer. Phytochem. Rev. 2008, 7, 231–250. [Google Scholar] [CrossRef]
- Bergougnoux, V. The History of Tomato: From Domestication to Biopharming. Biotechnol. Adv. 2014, 32, 170–189. [Google Scholar] [CrossRef]
- Uera, R.B.; Paz-Alberto, A.M.; Sigua, G.C. Phytoremediation Potentials of Selected Tropical Plants for Ethidium Bromide. Environ. Sci. Pollut. Res. 2007, 14, 505–509. [Google Scholar] [CrossRef]
- Malamas, M.; Marselos, M. The Tradition of Medicinal Plants in Zagori, Epirus (Northwestern Greece). J. Ethnopharmacol. 1992, 37, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Bahramsoltani, R.; Rostamiasrabadi, P.; Shahpiri, Z.; Marques, A.M.; Rahimi, R.; Farzaei, M.H. Aloysia Citrodora Paláu (Lemon verbena): A Review of Phytochemistry and Pharmacology. J. Ethnopharmacol. 2018, 222, 34–51. [Google Scholar] [CrossRef] [PubMed]
- Zoghlami, R.I.; Hamdi, H.; Boudabbous, K.; Hechmi, S.; Khelil, M.N.; Jedidi, N. Seasonal Toxicity Variation in Light-Textured Soil Amended with Urban Sewage Sludge: Interaction Effect on Cadmium, Nickel, and Phytotoxicity. Environ. Sci. Pollut. Res. 2018, 25, 3608–3615. [Google Scholar] [CrossRef] [PubMed]
- Benzarti, S.; Lahmayer, I.; Dallali, S.; Chouchane, W.; Hamdi, H. Allelopathic and Antimicrobial Activities of Leaf Aqueous and Methanolic Extracts of Verbena officinalis L. and Aloysia citrodora L. (Verbenaceae): A Comparative Study. Med. Aromat. Plants 2016, 5, 1–9. [Google Scholar] [CrossRef]
- Oukerrou, M.A.; Tilaoui, M.; Mouse, H.A.; Leouifoudi, I.; Jaafari, A.; Zyad, A. Chemical Composition and Cytotoxic and Antibacterial Activities of the Essential Oil of Aloysia Citriodora Palau Grown in Morocco. Adv. Pharmacol. Sci. 2017, 2017, 7801924. [Google Scholar] [CrossRef]
- Kalita, S.; Kumar, G.; Karthik, L.; Rao, K.V.B. A Review on Medicinal Properties of Lantana camara Linn. Res. J. Pharm. Technol. 2012, 5, 711–715. [Google Scholar]
- Waoo, A.A.; Khare, S.; Ganguly, S. Evaluation of Phytoremediation Potential of Lantana Camara for Heavy Metals in an Industrially Polluted Area in Bhopal, India. Int. J. Eng. Appl. Sci. 2014, 1, 258035. [Google Scholar]
- Ndossi, B.; Chacha, M. Comparative Antibacterial and Antifungal Efficacy of Selected Tanzania Medicinal Plants. Eur. J. Med. Plants 2016, 14, 1–10. [Google Scholar] [CrossRef]
- Ghisalberti, E. Review Lantana camara L. (Verbenaceae). Fitoterapia 2000, 71, 467–486. [Google Scholar] [CrossRef]
- Skowyra, M.; Calvo, M.I.; Gallego, M.G.; Azman, N.A.M.; Almajano, M.P. Characterization of Phytochemicals in Petals of Different Colours from Viola × Wittrockiana Gams. and Their Correlation with Antioxidant Activity. J. Agric. Sci. 2014, 6, 93–105. [Google Scholar] [CrossRef]
- Vega, J.; Ruiz-Espinosa, H.; Luna-Guevara, J.; Luna-Guevara, M.; Hernández-Carranza, P.; Ávila-Sosa, R.; Ochoa-Velasco, C. Effect of Solvents and Extraction Methods on Total Anthocyanins, Phenolic Compounds and Antioxidant Capacity of Renealmia alpinia (Rottb.) Maas Peel. Czech J. Food Sci. 2017, 35, 456–465. [Google Scholar] [CrossRef]
- Santa-Cruz, Ó. Validación Del Extracto Del Exocarpo de Renealmia Alpinia (Kumpia) Como Colorante Nuclear Tisular. Bachelor’s Thesis, Universidad Norbert Wiener, Lima, Peru, 2014. Available online: https://repositorio.uwiener.edu.pe/handle/20.500.13053/96?locale-attribute=es (accessed on 8 October 2023).
- Gómez-Betancur, I.; Benjumea, D. Traditional Use of the Genus Renealmia and Renealmia alpinia (Rottb.) Maas (Zingiberaceae)—A Review in the Treatment of Snakebites. Asian Pac. J. Trop. Med. 2014, 7, S574–S582. [Google Scholar] [CrossRef] [PubMed]
- Collaço, R.; Cogo, J.; Rodrigues-Simioni, L.; Rocha, T.; Oshima-Franco, Y.; Randazzo-Moura, P. Protection by Mikania laevigata (Guaco) Extract against the Toxicity of Philodryas Olfersii Snake Venom. Toxicon 2012, 60, 614–622. [Google Scholar] [CrossRef] [PubMed]
- Fernández, M.; Ortiz, W.; Pereáñez, J.; Martínez, D. Evaluación de Las Propiedades Antiofídicas Del Extracto Etanólico y Fracciones Obtenidas de Renealmia Alpinia (Rottb) Mass (Zingiberaceae) Cultivad in Vitro. Vitae Rev. La Fac. Química Farm. 2010, 17, 75–82. [Google Scholar]
- Jimenez-Gonzalez, O.; Ruiz-Espinosa, H.; Luna-Guevara, J.J.; Ochoa-Velasco, C.E.; Luna Vital, D.; Luna-Guevara, M.L. A Potential Natural Coloring Agent with Antioxidant Properties: Microencapsulates of Renealmia alpinia (Rottb.) Maas Fruit Pericarp. NFS J. 2018, 13, 1–9. [Google Scholar] [CrossRef]
- Meléndez-Martínez, A.; Benítez, A.; Corell, M.; Hernanz, D.; Mapelli-Brahm, P.; Stinco, C.; Coyago-Cruz, E. Screening for Innovative Sources of Carotenoids and Phenolic Antioxidants among Flowers. Foods 2021, 10, 2625. [Google Scholar] [CrossRef]
- Li, S.; Li, S.-K.; Gan, R.-Y.; Song, F.-L.; Kuang, L.; Li, H.-B. Antioxidant Capacities and Total Phenolic Contents of Infusions from 223 Medicinal Plants. Ind. Crops Prod. 2013, 51, 289–298. [Google Scholar] [CrossRef]
- Grzeszczuk, M.; Weso, A.; Jadczak, D.; Jakubowska, B. Nutritional Value of Chive Edible Flowers. Acta Sci. Pol. Hortorum Cultus 2011, 10, 85–94. [Google Scholar] [CrossRef]
- López-García, J.; Kuceková, Z.; Humpolícek, P.; Mlcek, J.; Sáha, P. Polyphenolic Extracts of Edible Flowers Incorporated onto Atelocollagen Matrices and Their Effect on Cell Viability. Molecules 2013, 18, 13435–13445. [Google Scholar] [CrossRef]
- Bloor, S.; Falshaw, R. Covalently Linked Anthocyanin-Flavonol Pigments from Blue Agapanthus Flowers. Phytochemistry 2000, 53, 575–579. [Google Scholar] [CrossRef]
- Viljoen, C.D.; Snyman, M.C.; Spies, J.J. Identification and Expression Analysis of Chalcone Synthase and Dihydroflavonol 4-Reductase in Clivia Miniata. South Afr. J. Bot. 2013, 87, 18–21. [Google Scholar] [CrossRef]
- Divya, P.; Puthusseri, B.; Neelwarne, B. Carotenoid Content, Its Stability during Drying and the Antioxidant Activity of Commercial Coriander (Coriandrum sativum L.) Varieties. Food Res. Int. 2012, 45, 342–350. [Google Scholar] [CrossRef]
- Divya, P.; Puthusseri, B.; Neelwarne, B. The Effect of Plant Regulators on the Concentration of Carotenoids and Phenolic Compounds in Foliage of Coriander. LWT Food Sci. Technol. 2014, 56, 101–110. [Google Scholar] [CrossRef]
- Martins, N.; Barros, L.; Santos-Buelga, C.; Ferreira, I.C.F.R. Antioxidant Potential of Two Apiaceae Plant Extracts: A Comparative Study Focused on the Phenolic Composition. Ind. Crops Prod. 2016, 79, 188–194. [Google Scholar] [CrossRef]
- Misra, N.N.; Misra, R.; Mariam, A.; Yusuf, K.; Yusuf, L. Salicylic Acid Alters Antioxidant and Phenolics Metabolism in Catharanthus Roseus Grown under Salinity Stress. Afr. J. Tradit. Complement. Altern. Med. 2014, 11, 118. [Google Scholar] [CrossRef] [PubMed]
- Jaleel, C.A.; Manivannan, P.; Lakshmanan, G.M.A.; Gomathinayagam, M.; Panneerselvam, R. Alterations in Morphological Parameters and Photosynthetic Pigment Responses of Catharanthus Roseus under Soil Water Deficits. Colloids Surf. B Biointerfaces 2008, 61, 298–303. [Google Scholar] [CrossRef] [PubMed]
- Demmig, B.; Winter, K.; Kruger, A.; Czygan, F.-C. Zeaxanthin and the Heat Dissipation of Excess Light Energy in Nerium Oleander Exposed to a Combination of High Light and Water Stress. Plant Physiol. 2008, 87, 17–24. [Google Scholar] [CrossRef]
- Knoth, R. Ultrastructure of Lycopene Containing Chromoplasts in Fruits of Aglaonema commutatum Schott (Araceae). Protoplasma 1981, 106, 249–250. [Google Scholar] [CrossRef]
- Collette, V.; Jameson, P.; Schwinn, K.; Umaharan, P.; Davies, K. Temporal and Spatial Expression of Flavonoid Biosynthetic Genes in Flowers of Anthurium andraeanum. Physiol. Plant. 2004, 122, 297–304. [Google Scholar] [CrossRef]
- Gupta, S.; Agarwal, A.; Kumar, K.; Arya, M.; Nasim, M. Physiochemical Response of Air Purifying Indoor Plants under Cold Stress. J. Biochem. Int. 2015, 2, 1–7. [Google Scholar]
- Zheng, L.; Van Labeke, M.C. Chrysanthemum Morphology, Photosynthetic Efficiency and Antioxidant Capacity Are Differentially Modified by Light Quality. J. Plant Physiol. 2017, 213, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Khalil, M.; Raila, J.; Ali, M.; Islam, K.M.S.; Schenk, R.; Krause, J.P.; Schweigert, F.J.; Rawel, H. Stability and Bioavailability of Lutein Ester Supplements from Tagetes Flower Prepared under Food Processing Conditions. J. Funct. Foods 2012, 4, 602–610. [Google Scholar] [CrossRef]
- Dudek, G.; Strzelewicz, A.; Krasowska, M.; Rybak, A.; Turczyn, R. A Spectrophotometric Method for Plant Pigments Determination and Herbs Classification. Chem. Pap. 2014, 68, 579–583. [Google Scholar] [CrossRef]
- Kucekova, Z.; Mlcek, J.; Humpolicek, P.; Rop, O. Edible Flowers—Antioxidant Activity and Impact on Cell Viability. Cent. Eur. J. Biol. 2013, 8, 1023–1031. [Google Scholar] [CrossRef]
- Kishimoto, S.; Sumitomo, K.; Yagi, M.; Nakayama, M.; Ohmiya, A.; Nakamura, M.; Ohmiya, A. Three Routes to Orange Petal Color via Carotenoid Components in 9 Compositae Species. J. Jpn. Soc. Hortic. Sci. 2007, 76, 250–257. [Google Scholar] [CrossRef]
- Szewczyk, K.; Olech, M. Optimization of Extraction Method for LC–MS Based Determination of Phenolic Acid Profiles in Different Impatiens Species. Phytochem. Lett. 2017, 20, 322–330. [Google Scholar] [CrossRef]
- Muthu, K.; Borse, L.; Thangatripathi, D.R.; Borse, S. Antimicrobial Activity of Heartwood of Tecoma Stans. Int. J. Pharm. Pharm. Sci. 2012, 4, 384–386. [Google Scholar]
- Wiszniewska, A.; Hanus-Fajerska, E.; Muszyńska, E.; Smoleń, S. Comparative Assessment of Response to Cadmium in Heavy Metal-Tolerant Shrubs Cultured In Vitro. Water. Air. Soil Pollut. 2017, 228, 304. [Google Scholar] [CrossRef] [PubMed]
- Žnidarčič, D.; Ban, D.; Šircelj, H. Carotenoid and Chlorophyll Composition of Commonly Consumed Leafy Vegetables in Mediterranean Countries. Food Chem. 2011, 129, 1164–1168. [Google Scholar] [CrossRef]
- Ferrante, A.; Vernieri, P.; Serra, G.; Tognoni, F. Changes in Abscisic Acid during Leaf Yellowing of Cut Stock Flowers. Plant Growth Regul. 2004, 43, 127–134. [Google Scholar] [CrossRef]
- Chen, T.; He, J.; Zhang, J.; Li, X.; Zhang, H.; Hao, J.; Li, L. The Isolation and Identification of Two Compounds with Predominant Radical Scavenging Activity in Hempseed (Seed of Cannabis sativa L.). Food Chem. 2012, 134, 1030–1037. [Google Scholar] [CrossRef] [PubMed]
- Tinoi, J.; Rakariyatham, N.; Deming, R. Determination of Major Carotenoid Constituents in Petal Extracts of Eight Selected Flowering Plants in the North of Thailand. Chiang Mai J. Sci. 2006, 33, 327–334. [Google Scholar]
- Vankar, P.S.; Srivastava, J. Ultrasound-Assisted Extraction in Different Solvents for Phytochemical Study of Canna Indica. Int. J. Food Eng. 2010, 6. [Google Scholar] [CrossRef]
- Stefaniak, A.; Grzeszczuk, M. Nutritional and Biological Value of Five Edible Flower Species. Not. Bot. Horti Agrobot. Cluj Napoca 2019, 47, 128–134. [Google Scholar] [CrossRef]
- Lee, J.; Seo, Y.; Lee, J.; Ju, J. Antioxidant Activities of Dianthus chinensis L. Extract and Its Inhibitory Activities against Nitric Oxide Production and Cancer Cell Growth and Adhesion. J. Korean Soc. Food Sci. Nutr. 2016, 45, 44–51. [Google Scholar] [CrossRef]
- Price, H.; Zuker, A.; Danziger, G. Gypsophila Plants Having Elevated Amount of Beta-Carotene and Methods for Obtaining the Same. U.S. Patent 10,918,043, 28 December 2017. [Google Scholar]
- Al-Rifai, A.A.; Aqel, A.; Al-Warhi, T.; Wabaidur, S.M.; Al-Othman, Z.A.; Badjah-Hadj-Ahmed, A.Y. Antibacterial, Antioxidant Activity of Ethanolic Plant Extracts of Some Convolvulus Species and Their DART-ToF-MS Profiling. Evid. Based Complement. Altern. Med. 2017, 2017, 5694305. [Google Scholar] [CrossRef]
- Chandrika, U.; Fernando, K.; Ranaweera, K. Carotenoid Content and in Vitro Bioaccessibility of Lycopene from Guava (Psidium guajava) and Watermelon (Citrullus lanatus) by High-Performance Liquid Chromatography Diode Array Detection. Int. J. Food Sci. Nutr. 2009, 60, 558–566. [Google Scholar] [CrossRef] [PubMed]
- Saadawy, F.; Abdel-Moniem, A. Effect of Some Factors on Growth and Development of Euphorbia milii Var. Longifolia. Middle East J. Agric. Res. 2015, 4, 613–628. [Google Scholar]
- Biesiada, A.; Sokół-Łetowska, A.; Kucharska, A. The Effect of Nitrogen Fertilization on Yielding and Antioxidant Activity of Lavender (Lavandula angustifolia Mill.). Acta Sci. Pol. Hortorum Cultus 2008, 7, 33–40. [Google Scholar]
- Carmen, S.; Mărghitaş, L.A. Changes in Major Bioactive Compounds with Antioxidant Activity of Agastache foeniculum, Lavandula angustifolia, Melissa officinalis and Nepeta cataria: Effect of Harvest Time and Plant Species. Ind. Crops Prod. 2015, 77, 499–507. [Google Scholar] [CrossRef]
- Radulescu, C.; Stihi, C.; Ilie, M.; Lazurcă, D.; Gruia, R.; Olaru, O.T.; Bute, O.C.; Dulama, I.D.; Stirbescu, R.M.; Teodorescu, S.; et al. Characterization of Phenolics in Lavandula Angustifolia. Anal. Lett. 2017, 50, 2839–2850. [Google Scholar] [CrossRef]
- Morales-serna, A.; Morales-serna, J.A.; García-ríos, E.; Madrigal, D.; Cárdenas, J.; Salmón, M. Constituents of Organic Extracts of Cuphea hyssopifolia. J. Mex. Chem. Soc. 2011, 55, 62–64. [Google Scholar]
- Sravanthi, T.; Rao, G. Evaluation of Antioxidant-Phytochemical Compounds in Punica grantum. Int. J. Pharm. Sci. Res. 2015, 6, 5295–5300. [Google Scholar] [CrossRef]
- Rummun, N.; Somanah, J.; Ramsaha, S.; Bahorun, T.; Neergheen-Bhujun, V. Bioactivity of Nonedible Parts of Punica granatum L.: A Potential Source of Functional Ingredients. Int. J. Food Sci. 2013, 2013, 602312. [Google Scholar] [CrossRef] [PubMed]
- Fellah, B.; Bannour, M.; Rocchetti, G.; Lucini, L.; Ferchichi, A. Phenolic Profiling and Antioxidant Capacity in Flowers, Leaves and Peels of Tunisian Cultivars of Punica granatum L. J. Food Sci. Technol. 2018, 55, 3606–3615. [Google Scholar] [CrossRef]
- Wang, N.; Zhang, C.; Bian, S.; Chang, P.; Xuan, L.; Fan, L.; Yu, Q.; Liu, Z.; Gu, C. Flavonoid Components of Different Color Magnolia Flowers and Their Relationship to Cultivar Selections. Am. Soc. Hortic. Sci. 2019, 54, 404–408. [Google Scholar] [CrossRef]
- Afify, A.; Hassan, H. Free Radical Scavenging Activity of Three Different Flowers-Hibiscus Rosa-Sinensis, Quisqualis Indica and Senna Surattensis. Asian Pac. J. Trop. Biomed. 2016, 6, 771–777. [Google Scholar] [CrossRef]
- Kostic, A.; Milincic, D.; Gasic, U.; Nedic, N.; Stanojevic, S.; Tesic, Z.; Pesic, M. Polyphenolic Profile and Antioxidant Properties of Bee-Collected Pollen from Sunflower (Helianthus annuus L.) Plant. LWT Food Sci. Technol. 2019, 112, 108244. [Google Scholar] [CrossRef]
- Rezende, F.; Sande, D.; Coelho, A.C.; Geane, P.; Amélia, M.; Boaventura, D.; Takahashi, J.A. Edible Flowers as Innovative Ingredients for Future Food Development: Anti-Alzheimer, Antimicrobial and Antioxidant Potential. Chem. Eng. Trans. 2019, 75, 337–342. [Google Scholar] [CrossRef]
- Matea, C.T.; Soran, M.-L.; Pintea, A.; Bele, C. Analytical Determination of Carotenoids in Inland Ocimum basilicum L. Bull. UASVM Agric. 2010, 67, 1843–5386. [Google Scholar] [CrossRef]
- Singh, S.; Singh, R.; Ashish, N.; Salim, K.M. Estimation of Phytochemicals and Antioxidant Activity in Hibiscus Sabdariffa L. Progress. Hortic. 2013, 45, 174–181. [Google Scholar]
- Jung, E.; Joo, N. Physicochemical Properties and Antimicrobial Activity of Roselle (Hibiscus sabdariffa L.). J. Sci. Food Agric. 2013, 93, 3769–3776. [Google Scholar] [CrossRef] [PubMed]
- Kandylis, P. Phytochemicals and Antioxidant Properties of Edible Flowers. Appl. Sci. 2022, 12, 9937. [Google Scholar] [CrossRef]
- Hanny, B.; Henson, R.; Thompson, A.; Gueldner, R.; Hedin, P. Identification of Carotenoid Constituents in Hibiscus syriacus L. J. Agric. Food Chem. 1972, 20, 914–916. [Google Scholar] [CrossRef]
- Geng, M.; Ren, M.; Liu, Z.; Shang, X. Free Radical Scavenging Activities of Pigment Extract from Hibiscus syriacus L. Petals in Vitro. Afr. J. Biotechnol. 2012, 11, 429–435. [Google Scholar] [CrossRef]
- Zachariah, S.M.; Aleykutty, N.A.; Viswanad, V.; Jacob, S.; Prabhakar, V. In-Vitro Antioxidant Potential of Methanolic Extracts of Mirabilis jalapa Linn. Free Radic. Antioxid. 2011, 1, 82–86. [Google Scholar] [CrossRef]
- Johnson, C.E.; Lin, L.; Harnly, J.M.; Oladeinde, F.O. Identification of the Phenolic Components of Vernonia amygdalina and Russelia equisetiformis. J. Nat. Prod. 2011, 4, 57–64. [Google Scholar]
- Hanhineva, K.; Rogachev, I.; Kokko, H.; Mintz-Oron, S.; Venger, I.; Kärenlampi, S.; Aharoni, A. Non-Targeted Analysis of Spatial Metabolite Composition in Strawberry (Fragaria × Ananassa) Flowers. Phytochemistry 2008, 69, 2463–2481. [Google Scholar] [CrossRef]
- Torey, A.; Sasidharan, S.; Latha, L.Y.; Sudhakaran, S.; Ramanathan, S. Antioxidant Activity and Total Phenolic Content of Methanol Extracts of Ixora coccinea. Pharm. Biol. 2010, 48, 1119–1123. [Google Scholar] [CrossRef]
- Matteo, R.; Luis, B.; Manuel, P.; Cerda, J.; Andrea, T.; Andrea, R.; Matteo, C. Determinación de Polifenoles En Cinco Especies Amazónicas Con Potencial Antioxidante. Rev. Amaz. Cienc. Y Tecnol. 2017, 6, 55–64. [Google Scholar]
- Murakami, Y.; Fukui, Y.; Watanabe, H.; Kokubun, H.; Toya, Y.; Ando, T. Distribution of Carotenoids in the Flower of Non-Yellow Commercial Petunia. J. Hortic. Sci. Biotechnol. 2003, 78, 127–130. [Google Scholar] [CrossRef]
- Gamsjaeger, S.; Baranska, M.; Schulz, H.; Heiselmayer, P.; Musso, M. Discrimination of Carotenoid and Flavonoid Content in Petals of Pansy Cultivars (Viola × Wittrockiana) by FT-Raman Spectroscopy. J. Raman Spectrosc. 2011, 42, 1240–1247. [Google Scholar] [CrossRef]
- Moliner, C.; Barros, L.; Dias, M.I.; Reigada, I.; Ferreira, I.C.; López, V.; Langa, E.; Rincón, C.G. Viola × Wittrockiana: Phenolic Compounds, Antioxidant and Neuroprotective Activities on Caenorhabditis Elegans. J. Food Drug Anal. 2019, 27, 849–859. [Google Scholar] [CrossRef] [PubMed]
- Chae, S.; Lee, S.; Kim, J.; Park, W.; Uddin, M.; Kim, H.; Park, S.; Chaem, S.; Lee, S.; Kim, J.; et al. Variation of Carotenoid Content in Agastache rugosa and Agastache foeniculum. Asian J. Chem. 2013, 25, 4364–4366. [Google Scholar] [CrossRef]
- Ohmiya, A. Qualitative and Quantitative Control of Carotenoid Accumulation in Flower Petals. Sci. Hortic. 2013, 163, 10–19. [Google Scholar] [CrossRef]
- Nan, M.; Pintea, A.; Bunea, A.; Esianu, S.; Tamas, M. HPLC Analysis of Carotenids from Inula helenium L. Flowers and Leaves. Farmacia 2012, 60, 501–509. [Google Scholar]
- Lafountain, A.M.; Frank, H.A.; Yuan, Y.W. Carotenoid Composition of the Flowers of Mimulus lewisii and Related Species: Implications Regarding the Prevalence and Origin of Two Unique, Allenic Pigments. Arch. Biochem. Biophys. 2015, 573, 32–39. [Google Scholar] [CrossRef]
- Kishimoto, S.; Oda-Yamamizo, C.; Ohmiya, A. Comparison of Petunia and Calibrachoa in Carotenoid Pigmentation of Corollas. Breed. Sci. 2019, 69, 117–126. [Google Scholar] [CrossRef]
- Yuan, H.; Zhang, J.; Nageswaran, D.; Li, L. Carotenoid Metabolism and Regulation in Horticultural Crops. Hortic. Res. 2015, 2, 15036. [Google Scholar] [CrossRef]
- Zhang, C.; Wang, Y.; Fu, J.; Bao, Z.; Zhao, H. Transcriptomic Analysis and Carotenogenic Gene Expression Related to Petal Coloration in Osmanthus Fragrans ‘Yanhong Gui’. Trees Struct. Funct. 2016, 30, 1207–1223. [Google Scholar] [CrossRef]
- Xiao, Z.; Su, J.; Liu, X.; Sun, X.; He, L.; Zhou, H.; Li, C. Overexpression of RmLCYB from Rhododendron Molle Increases Carotenoid in Nicotiana Tabacum. Acta Physiol. Plant. 2022, 44, 71. [Google Scholar] [CrossRef]
- Qing, H.S.; Qian, J.Y.; Chen, J.H.; Jiang, L.L.; Fu, J.X.; Huang, X.Q.; Zhang, C. Carotenoid Analysis and Functional Characterization of Lycopene Cyclases in Zinnia elegans L. Ind. Crops Prod. 2022, 188, 115724. [Google Scholar] [CrossRef]
- Fernandes, L.; Pereira, J.A.; Saraiva, J.A.; Ramalhosa, E.; Casal, S. Phytochemical Characterization of Borago officinalis L. and Centaurea cyanus L. during Flower Development. Food Res. Int. 2019, 123, 771–778. [Google Scholar] [CrossRef] [PubMed]
- Alarcón-Alonso, J.; Zamilpa, A.; Alarcón, F.; Herrera-Ruiz, M.; Tortoriello, J.; Jimenez-Ferrer, E. Pharmacological Characterization of the Diuretic Effect of Hibiscus sabdariffa Linn (Malvaceae) Extract. J. Ethnopharmacol. 2012, 139, 751–756. [Google Scholar] [CrossRef]
- Hegde, A.S.; Gupta, S.; Sharma, S.; Srivatsan, V.; Kumari, P. Edible Rose Flowers: A Doorway to Gastronomic and Nutraceutical Research. Food Res. Int. 2022, 162, 111977. [Google Scholar] [CrossRef]
- Niizu, P.; Rodriguez-Amaya, D. Flowers and Leaves of Tropaeolum majus L. as Rich Sources of Lutein. J. Food Sci. 2005, 70, 605–609. [Google Scholar] [CrossRef]
- Cornea-Cipcigan, M.; Bunea, A.; Bouari, C.M.; Pamfil, D.; Páll, E.; Urcan, A.C.; Mărgăoan, R. Anthocyanins and Carotenoids Characterization in Flowers and Leaves of Cyclamen Genotypes Linked with Bioactivities Using Multivariate Analysis Techniques. Antioxidants 2022, 11, 1126. [Google Scholar] [CrossRef]
- Leonti, M.; Verpoorte, R. Traditional Mediterranean and European Herbal Medicines. J. Ethnopharmacol. 2017, 199, 161–167. [Google Scholar] [CrossRef]
- Srivastava, J.K.; Shankar, E.; Gupta, S. Chamomile: A Herbal Medicine of the Past with a Bright Future (Review). Mol. Med. Rep. 2010, 3, 895–901. [Google Scholar]
- Madras, B.K. Update of Cannabis and Its Medical Use. Alcohol. Drug Abus. Res. 2015, 5, 1–41. [Google Scholar]
- Garcia-Oliveira, P.; Barral, M.; Carpena, M.; Gullón, P.; Fraga-Corral, M.; Otero, P.; Prieto, M.; Simal-Gandara, J. Traditional Plants from Asteraceae Family as Potential Candidates for Functional Food Industry. Food Funct. 2021, 12, 2850–2873. [Google Scholar] [CrossRef]
- Rodrigues, H.; Spence, C. Looking to the Future, by Studying the History of Edible Flowers. Int. J. Gastron. Food Sci. 2023, 34, 100805. [Google Scholar] [CrossRef]
- Fernandes, L.; Casal, S.; Pereira, J.A.; Saraiva, J.A.; Ramalhosa, E. Edible Flowers: A Review of the Nutritional, Antioxidant, Antimicrobial Properties and Effects on Human Health. J. Food Compos. Anal. 2017, 60, 38–50. [Google Scholar] [CrossRef]
- Vega, G.; Dumas, C.; Kahn, B.; Mana, J. KaBloom!: Revolution in the Flower Industry. N. Engl. J. Entrep. 2011, 14, 61–77. [Google Scholar] [CrossRef]
- Cunningham, E. What Nutritional Contribution Do Edible Flowers Make? J. Acad. Nutr. Diet. 2015, 115, 856. [Google Scholar] [CrossRef]
- Lara-Cortés, E.; Osorio-Díaz, P.; Jiménez-Aparicio, A.; Bautista-Baños, S. Contenido Nutricional, Propiedades Funcionales y Conservación de Flores Comestibles. Revisión. Arch. Latinoam. Nutr. 2013, 63, 197–208. [Google Scholar]
- Fallahi, H.-R.; Aghhavani-Shajari, M.; Sahabi, H.; Behdani, M.; Sayyari-Zohan, M.; Vatandoost, S. Influence of Some Pre and Post-Harvest Practices on Quality of Saffron Stigmata. Sci. Hortic. Amst. 2021, 278, 109846. [Google Scholar] [CrossRef]
- Terry, M.I.; Ruiz-Hernández, V.; Águila, D.; Weiss, J.; Egea-Cortines, M. The Effect of Post-Harvest Conditions in Narcissus sp. Cut Flowers Scent Profile. Front. Plant Sci. 2021, 11, 540821. [Google Scholar] [CrossRef]
- Kelley, K.M.; Cameron, A.C.; Biernbaum, J.A.; Poff, K.L. Effect of Storage Temperature on the Quality of Edible Flowers. Postharvest Biol. Technol. 2003, 27, 341–344. [Google Scholar] [CrossRef]
- Tai, C.; Chen, B.H. Analysis and Stability of Carotenoids in the Flowers of Daylily (Hemerocallis Disticha) as Affected by Various Treatments. J. Agric. Food Chem. 2000, 48, 5962–5968. [Google Scholar] [CrossRef]
- Siriamornpun, S.; Kaisoon, O.; Meeso, N. Changes in Colour, Antioxidant Activities and Carotenoids (Lycopene, β-Carotene, Lutein) of Marigold Flower (Tagetes erecta L.) Resulting from Different Drying Processes. J. Funct. Foods 2012, 4, 757–766. [Google Scholar] [CrossRef]
- Lin, S.-D.; Sung, J.-M.; Chen, C.-L. Effect of Drying and Storage Conditions on Caffeic Acid Derivatives and Total Phenolics of Echinacea Purpurea Grown in Taiwan. Food Chem. 2011, 125, 226–231. [Google Scholar] [CrossRef]
- Diva, F.; Almeida, L.; Faria, W.; Souza, R.; Tiwari, B.K.; Cullen, P.J.; Maria, J.; Bourke, P.; Fernandes, F.A.N.; Rodrigues, S. Fructooligosaccharides Integrity after Atmospheric Cold Plasma and High—Pressure Processing of a Functional Orange Juice. Food Res. Int. 2017, 102, 282–290. [Google Scholar] [CrossRef]
- Sotelo, A.; López-García, S.; Basurto-Peña, F. Content of Nutrient and Antinutrient in Edible Flowers of Wild Plants in Mexico. Plant Foods Hum. Nutr. 2007, 62, 133–138. [Google Scholar] [CrossRef]
- Weisz, G.M.; Kammerer, D.R.; Carle, R. Identification and Quantification of Phenolic Compounds from Sunflower (Helianthus annuus L.) Kernels and Shells by HPLC-DAD/ESI-MS N. Food Chem. 2009, 115, 758–765. [Google Scholar] [CrossRef]
- Liu, F.; Wang, M.; Wang, M. Phenolic Compounds and Antioxidant Activities of Flowers, Leaves and Fruits of Five Crabapple Cultivars (Malus Mill. Species). Sci. Hortic. 2018, 235, 460–467. [Google Scholar] [CrossRef]
- Tocci, N.; Weil, T.; Perenzoni, D.; Narduzzi, L.; Madriñán, S.; Crockett, S.; Nürk, N.M.; Cavalieri, D.; Mattivi, F. Phenolic Profile, Chemical Relationship and Antifungal Activity of Andean Hypericum Species. Ind. Crops Prod. 2018, 112, 32–37. [Google Scholar] [CrossRef]
- Navarro-González, I.; González-Barrio, R.; García-Valverde, V.; Bautista-Ortín, A.; Periago, M. Nutritional Composition and Antioxidant Capacity in Edible Flowers: Characterisation of Phenolic Compounds by HPLC-DAD-ESI/MSn. Int. J. Mol. Sci. 2015, 16, 805–822. [Google Scholar] [CrossRef]
- Lewis, D.H.; Arathoon, H.S.; Swinny, E.E.; Huang, S.C.; Funnell, K.A. Anthocyanin and Carotenoid Pigments in Spathe Tissue from Selected Zantedeschia Hybrids. XXVI Int. Hortic. Congr. Elegant Sci. Floric. 2002, 624, 147–154. [Google Scholar] [CrossRef]
- Mapelli-Brahm, P.; Meléndez-Martínez, A. The Colourless Carotenoids Phytoene and Phytofluene: Sources, Consumption, Bioavailability and Health Effects. Curr. Opin. Food Sci. 2021, 41, 201–209. [Google Scholar] [CrossRef]
- Meléndez-Martínez, A. An Overview of Carotenoids, Apocarotenoids and Vitamin A in Agro-Food, Nutrition, Health and Disease. Mol. Nutr. Food Res. 2019, 63, 1801045. [Google Scholar] [CrossRef]
- Meléndez-Martínez, A.; Stinco, C.; Mapelli-Brahm, P. Review Skin Carotenoids in Public Health and Nutricosmetics: The Emerging Roles and Applications of the UV Radiation-Absorbing Colourless Carotenoids Phytoene and Phytofluene. Nutrients 2019, 11, 1093. [Google Scholar] [CrossRef]
- Pérez-Sánchez, A.; Barrajón-Catalán, E.; Herranz-López, M.; Micol, V. Nutraceuticals for Skin Care: A Comprehensive Review of Human Clinical Studies. Nutrients 2018, 10, 403. [Google Scholar] [CrossRef]
- Chagas, R.; Santana, J.; Correa, U.; Santos, C.; Ribeiro, Y.; Matos, T.; Pedreira, J.; Carvalho, N.; Santos, H.; Narain, N. Phytochemicals Screening, Antioxidant Capacity and Chemometric Characterization of Four Edible Flowers from Brazil. Food Res. Int. 2020, 130, 108899. [Google Scholar] [CrossRef]
- Cai, Y.-Z.; Xing, J.; Sun, M.; Zhan, Z.-Q.; Corke, H. Phenolic Antioxidants (Hydrolyzable Tannins, Flavonols, and Anthocyanins) Identified by LC-ESI-MS and MALDI-QIT-TOF MS from Rosa Chinensis Flowers. J. Agric. Food Chem. 2005, 53, 9940–9948. [Google Scholar] [CrossRef]
- Chang, Y.; Huang, H.; Hsu, J.; Yang, S.; Wang, C. Hibiscus Anthocyanins Rich Extract-Induced Apoptotic Cell Death in Human Promyelocytic Leukemia Cells. Toxicol. Appl. Pharmacol. 2005, 205, 201–212. [Google Scholar] [CrossRef]
- Jakubczyk, K.; Koprowska, K.; Gottschling, A.; Janda-Milczarek, K. Edible Flowers as a Source of Dietary Fibre (Total, Insoluble and Soluble) as a Potential Athlete’s Dietary Supplement. Nutrients 2022, 14, 2470. [Google Scholar] [CrossRef]
- Kim, I.S.; Koppula, S.; Park, P.-J.; Kim, E.H.; Gil Kim, C.; Choi, W.S.; Lee, K.H.; Choi, D.-K. Chrysanthemum Morifolium Ramat (CM) Extract Protects Human Neuroblastoma SH-SY5Y Cells against MPP+ -Induced Cytotoxicity. J. Ethnopharmacol. 2009, 126, 447–454. [Google Scholar] [CrossRef]
- Johnson, E. Role of Lutein and Zeaxanthin in Visual and Cognitive Function throughout the Lifespan. Nutr. Rev. 2014, 72, 605–612. [Google Scholar] [CrossRef]
- Nelson, L.S.; Shih, R.D.; Balick, M.J. Handbook of Poisonous and Injurious Plants, 2nd ed.; Nelson, L.S., Shih, R.D., Balick, M.J., Eds.; Springer: New York, NY, USA, 2007; ISBN 9780387312682. [Google Scholar]
- Wilczyńska, A.; Kukułowicz, A.; Lewandowska, A. Preliminary Assessment of Microbial Quality of Edible Flowers. Lwt 2021, 150, 111926. [Google Scholar] [CrossRef]
- Mahadkar, S.; Valvi, S.; Rathod, V. Screening of Anti-Nutritional Factors from Some Wild Edible Plants. J. Nat. Prod. Plant Resourse 2012, 2, 251–255. [Google Scholar]
- Hassan, L.; Bagudo, B.; Aliero, A.; Umar, K.; Sani, N. Evaluation of Nutrient and Anti-Nutrient Contents of Parkia biglobosa (L.) Flower. Niger. J. Basic Appl. Sci. 2011, 19, 76–80. [Google Scholar] [CrossRef]
- Popova, A.; Mihaylova, D. Antinutrients in Plant-Based Foods: A Review. Open Biotechnol. J. 2019, 13, 68–76. [Google Scholar] [CrossRef]
- European-Comission Communication from the Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions. 2014. Available online: https://ec.europa.eu/environment/circular-economy/pdf/circular-economy-communication.pdf (accessed on 8 October 2023).
- FAO. The State of the World’s Biodiversity for Food and Agriculture; Bélanger, J., Pilling, D., Eds.; FAO Commission on Genetic Resources for Food and Agriculture Assessments: Rome, Italy, 2019; ISBN 9789251312704. [Google Scholar]
- Ali, H.; Khan, E.; Sajad, M.A. Phytoremediation of Heavy Metals-Concepts and Applications. Chemosphere 2013, 91, 869–881. [Google Scholar] [CrossRef] [PubMed]
- Sarwar, N.; Imran, M.; Shaheen, M.R.; Ishaque, W.; Kamran, M.A.; Matloob, A.; Rehim, A.; Hussain, S. Phytoremediation Strategies for Soils Contaminated with Heavy Metals: Modifications and Future Perspectives. Chemosphere 2017, 171, 710–721. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.H.A.; Kiyani, A.; Mirza, C.R.; Butt, T.A.; Barros, R.; Ali, B.; Iqbal, M.; Yousaf, S. Ornamental Plants for the Phytoremediation of Heavy Metals: Present Knowledge and Future Perspectives. Environ. Res. 2021, 195, 110780. [Google Scholar] [CrossRef] [PubMed]
- Sabri, M.A.; Ibrahim, T.H.; Khamis, M.I.; Ludwick, A.; Nancarrow, P. Sustainable Management of Cut Flowers Waste by Activation and Its Application in Wastewater Treatment Technology. Environ. Sci. Pollut. Res. 2021, 28, 31803–31813. [Google Scholar] [CrossRef]
- Adeel, S.; Salman, M.; Usama, M.; Rehman, F.; Ahmad, T.; Amin, N. Sustainable Isolation and Application of Rose Petals Based Anthocyanin Natural Dye for Coloration of Bio-Mordanted Wool Fabric: Short Title: Dyeing OfbBio Mordanted Wool with Rose Petal Extract. J. Nat. Fibers 2021, 19, 6089–6103. [Google Scholar] [CrossRef]
- Sheikh, J.; Singh, N.; Pinjari, D. Sustainable Functional Coloration of Linen Fabric Using Kigelia Africana Flower Colorant. J. Nat. Fibers 2021, 18, 888–897. [Google Scholar] [CrossRef]
- Khammee, P.; Unpaprom, Y.; Chaichompoo, C.; Khonkaen, P.; Ramaraj, R. Appropriateness of Waste Jasmine Flower for Bioethanol Conversion with Enzymatic Hydrolysis: Sustainable Development on Green Fuel Production. 3 Biotech 2021, 11, 216. [Google Scholar] [CrossRef]
- Ghaderi, S.; Haddadi, S.A.; Davoodi, S.; Arjmand, M. Application of Sustainable Saffron Purple Petals as an Eco-Friendly Green Additive for Drilling Fluids: A Rheological, Filtration, Morphological, and Corrosion Inhibition Study. J. Mol. Liq. 2020, 315, 113707. [Google Scholar] [CrossRef]
- Veerakumar, P.; Maiyalagan, T.; Raj, B.G.S.; Guruprasad, K.; Jiang, Z.; Lin, K.C. Paper Flower-Derived Porous Carbons with High-Capacitance by Chemical and Physical Activation for Sustainable Applications. Arab. J. Chem. 2020, 13, 2995–3007. [Google Scholar] [CrossRef]
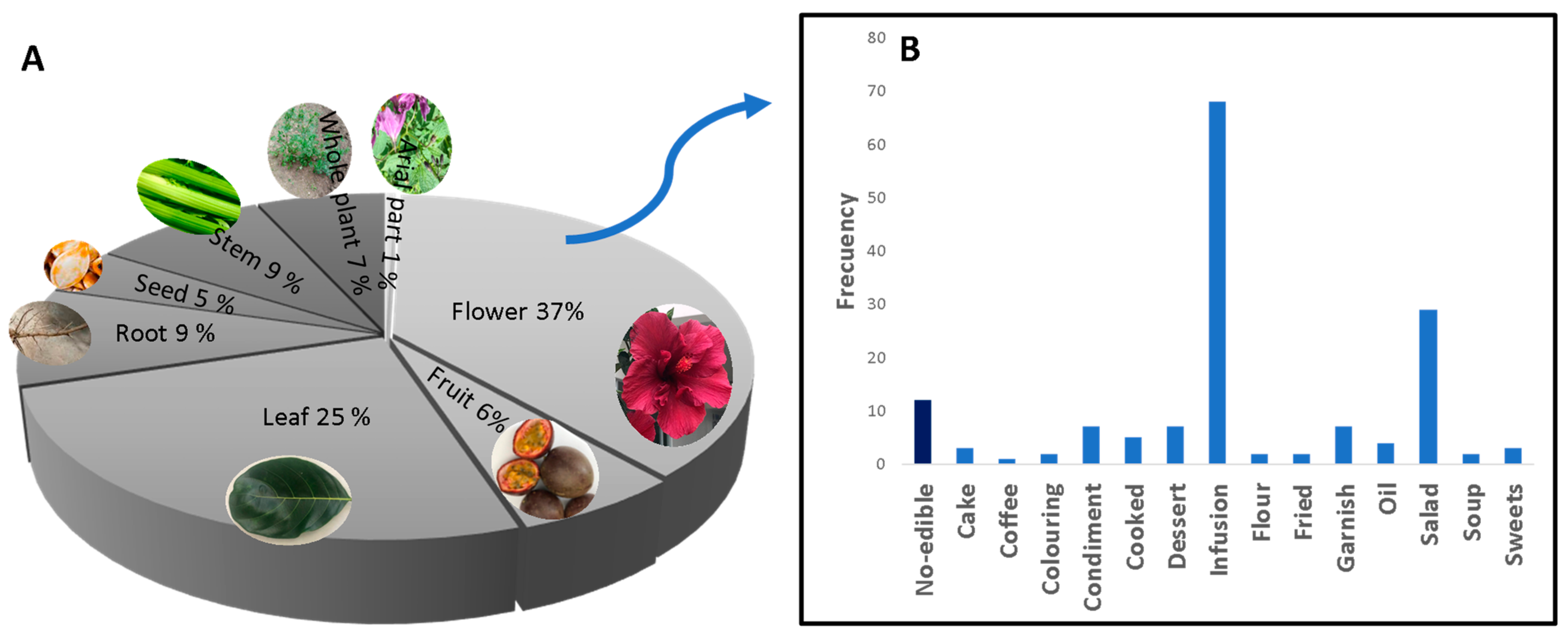
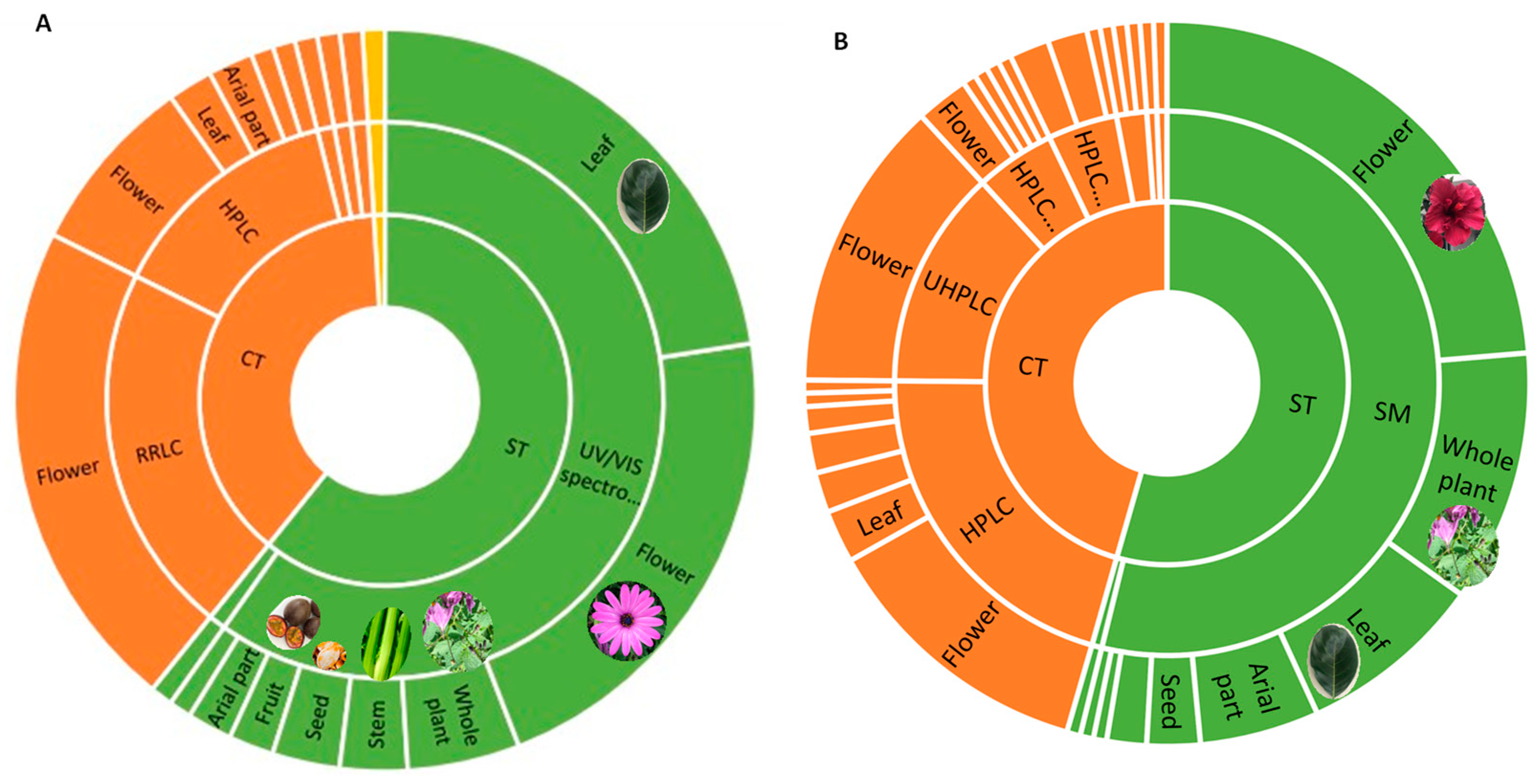
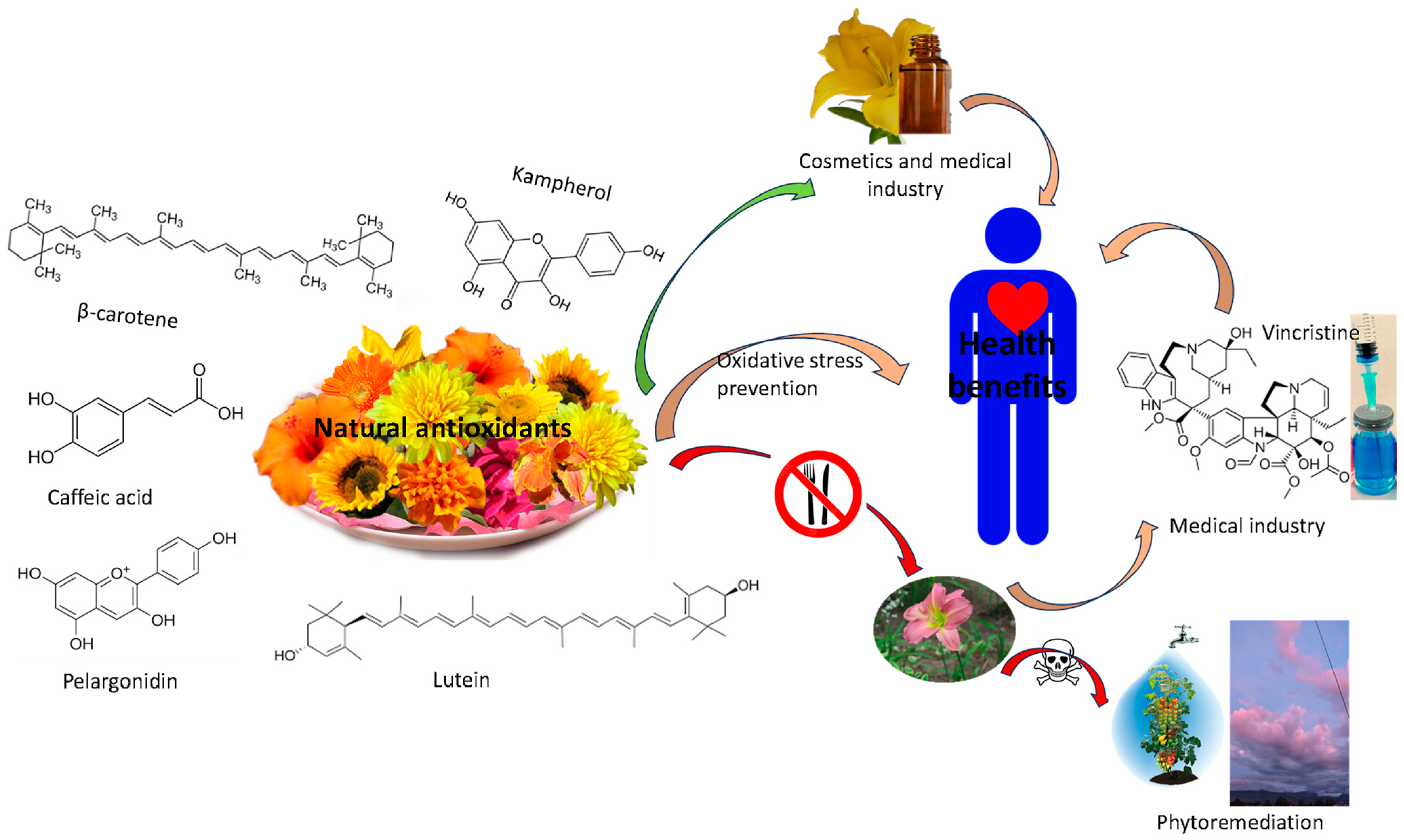
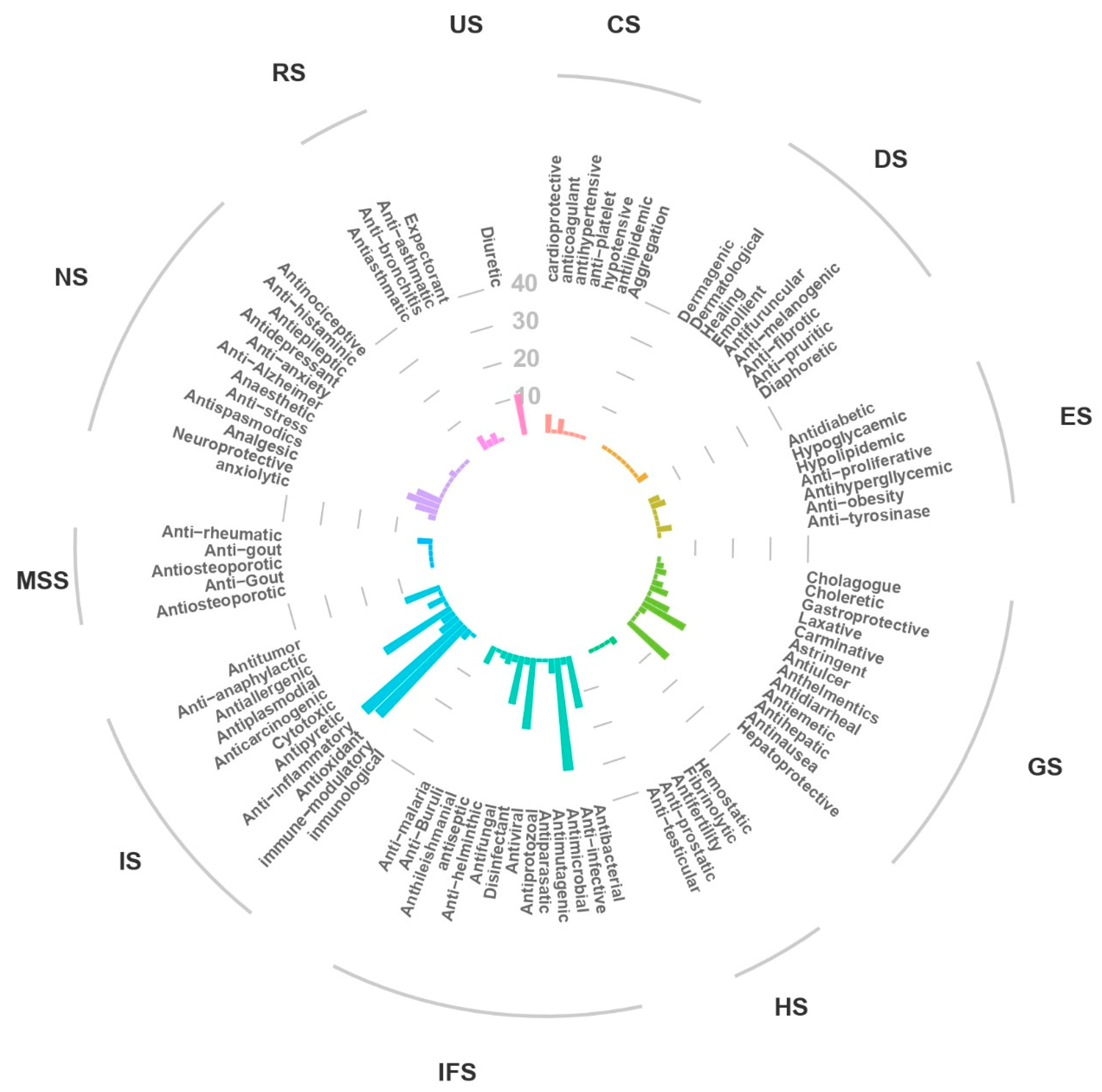
| Family [18] | Species [18] | Common Name | Place of Origin | Most Used Part/Flower Image | Phytoremediation Uses | Main Chemical Groups | Medicinal Use | Gastronomic Uses | |
|---|---|---|---|---|---|---|---|---|---|
| Acanthaceae | Aphelandra squarrosa Nees | Aphelandra, Kuda Belang, zebra plant, saffron spike [19,20] | Central and South America [19,20] | Root, leaf [21] |  | na | Alkaloids (aphelandrine, spermin), phytoanticipins (2-benzoxazolinone (BOA), 2-hydroxy-1,4-benzoxazine-3-one (HBOA)), glucosides (cyclic hydroxamic acids and their corresponding glucosides) [21,22,23,24] Note: It presents allelopathic activity [25] | Activity: antibacterial and antifungal [20,21] | Non-edible |
| Acanthaceae | Justicia aurea Schltdl. | Justicia, Yellow Jacobinia, Brazilian plume [26,27] | Central America [26] | Leaf [27,28] |  | na | na | Treatment: coughs, epilepsy, anxiety, and malaria [26,27,28] | Leaf: juice [27] |
| Alstroemeriaceae | Alstroemeria aurea Graham | Amancay, Peruvian Lily, Lily of the Incas, Parrot Lily, and jingle bell [29,30] | Andean forests [30,31] | Flower, leaf, stem [29,30] |  | na | Glucosides (tuliposide A), tulipalin A, phenols (6-hydroxy pelargonidin glycoside)s [32] | Treatment: gynaecological and obstetric [31] Toxicity: All parts can cause skin allergies [29,32] | Non-edible [29] |
| Amaranthaceae | Celosia argentea L. | Lion hand, velvet, cockscomb, plumón, pluma, plumero rosa, cresta de gallo, celosia [33,34,35] | Asia [34], unknown origin [35] | Flower, seed, leaf [36,37] |  | Soil decontamination [38] | Alkaloids, saponins, tannins, phenols (anthocyanin), glycoproteins [33,35,36,37,39] | Treatment: stop bleeding, liver heat, diseases of the blood, therapeutic eye diseases, and infections of the urinary tract [33,35,37]. Activity: antitumor, antiviral, hepatoprotective, immune-modulatory, antidiarrheal, anti-diabetic, anti-infective, anti-helminthic, anti-inflammatory, antioxidant, antinociceptive [33,35,36,37] | Leaf: vegetables [36] Flower: vegetables, additive food [36,39] Seed: flour [33] |
| Amaryllidaceae | Allium schoenoprasum L. | Wild chives, scallions, garlic chives, brown garlic, leaf onions, kucai [40] | Central Asia [41] | Leaf, root, flower [40,42] |  | Soil decontamination (Pb, Cd, Zn, and polycyclic aromatic hydrocarbons) [43] | Phenols, terpenes (volatile and essential oils), sulphur-compounds [41,42,44,45] Low toxicity (Daily doses: 60 g FW and 120 mg essential oil [41] | Treatment: stop bleeding, lower blood pressure, and prevent infections of the urinary tract [40,44] Activity: antithrombotic, antitumor, hepatoprotective, immune-modulatory, antidiarrheal, anti-diabetic, anti-infective, antioxidant, anti-inflammatory, antimicrobial (antifungal, antibacterial, antiviral, antiprotozoal, anthelmintic) [40,41,44] | Leaf: vegetables, condiment [41,44]. All parts are edible [40] |
| Amaryllidaceae | Agapanthus africanus (L.) Hoffmanns | African Lily, Nile Lily, African agapanthus, love flower [46] | South Africa [46] | Leaf, root, flower [47] |  | Water decontamination (TSS, COD, BOD, TP) [48] | Alkaloids (galantamine, tazatine), terpene (essential oil), tannins, phenolics (flavonoid)s, lipids (lecithin), proteins (polypeptides), saponins [46,49,50] | Treatment: heart diseases, hypertension, pregnancy and labour, cancer, and haemorrhoids [47,49,50,51] | Whole plant: infusion [50,51] |
| Amaryllidaceae | Clivia miniata (Lindl.) Bosse Clivia miniata var. citrina S. Watson | Bush lily, orange lily, umayime [52,53] | South Africa [54]. | Whole plant [52,53,55] |  | Soil decontamination (Pb and carbon). | Alkaloids (galantamine), esters (3a-4-dihydro-lactone), benzopyran ((3,4-g) indole ring system), triazines (atrazine) [52,54,55,56] | Treatment: fever, relieve pain, facilitate childbirth, and as a snake bite remedy [52,54] Activity: antimicrobial, antiviral, uterotonic, antitumor, cytotoxic activities [52,54,56] Toxicity: All plants present high toxicity (alkaloids) [55]. | Non-edible [56] |
| Apiaceae | Coriandrum sativum L. | Cilantro, Chinese parsley, European coriander, cilantrillo [57] | Mediterranean regions [57] | Whole plant [58,59] |  | Soil decontamination (Pb, Cr and As). Water decontamination (Zn (II) ions from aqueous medium) [60,61] | Sugars, alkaloids, phenolics, resins, tannins, anthraquinones, sterols, and terpenes (essential oils) [42,57,58,59] | Activity: antimicrobial, antioxidant, anti-diabetic, anxiolytic, cardioprotective, antiepileptic, anthelmintic, antiulcer, anti-carcinogenic, diuretic, antidepressant, antimutagenic, anti-inflammatory, antilipidemic, antihypertensive, neuroprotective, diuretic [57,58,59,62] This presents cytoprotective effects in gastric epithelial cells. LD50 oil = 4.1 g/Kg [57] | Arial part: several culinary uses [57,58] |
| Apocynaceae | Catharanthus roseus (L.) G. Don | Cape vinca, chavelita, teresita, vinca rosea, Isabelita, nayon-tara [27,63] | Madagascar [53] | Leaf, root [27] |  | Soil decontamination (Cr, Pb, Ni and oil-contaminated soil) [64,65] | Hallucinogen (Ibogaine), alkaloids(ascartharathine, lochnenine, vindoline, vindolinenine, vincristine, vinblastine, reserpine, tetrahydroal-stronine, yohimbine, serpentine) [53,63,66] Note: the leaves have cytotoxicity [66] | Treatment: leukaemia, a popular remedy for diabetes, headache, wasp stings, sore throat, eye irritation, low blood pressure, insomnia, Hodgkin’s disease, hypertension, neuroblastoma, malaria, rhabdomyosarcoma, Wilms tumour, vascular dementia, Alzheimer’s, dermatitis, acne [27,53,63,67] Activity: antifungal [68] | Leaf: juice [27] |
| Apocynaceae | Nerium oleander L. | Adelfa, flower laurel, laurel rose, trinitaria [69] | Mediterranean regions [69] | Leaf, flower, root, stem [69] |  | Soil decontamination (Ni and Cr) [70] | Alkaloids, tannins, steroids, terpenoids, flavonoids, saponins, and cardiac glycosides (nerifolin, peruvosid, vetoxin, thevethin A, thevethin B, ruvosid oleandrin, folinerin, adynerin, and digitoxigenin) [69,71,72] Note: Hazardous compounds (cardioactive steroids or cardiac glycosides) [55] | Treatment: cardiac affections, diabetes, rheumatic pain, epilepsy, asthma, leprosy, nervous regulation, painful menstrual periods, malaria, indigestion, ringworm, venous diseases, skin problems, warts, and chemotherapeutic agents [69,71,73] Activity: antifungal, cytotoxic, anti-inflammatory, antioxidant, analgesic, cardioprotective, neuroprotective, hepatoprotective [73] Toxicity: All plant causes abortions and skin irritant [29,55] | Non-edible [29] |
| Apocynaceae | Trachelospermum jasminoides (Lind.) Len. | Star jazmín, fake jasmíne, milk jasmine, Chinese jasmine | Asia | Leaf, flower, stem [74,75] |  | na | Lignans, alkaloids, triterpenoids, and phenolics [75,76] | Treatment: relieving rheumatic, arthritic pain, fever, gonarthritis, backache, and pharyngitis [76]. Activity: anti-inflammatory, analgesic, antitumor, antioxidant, and antimicrobial [76] | Arial part: infusion [76] |
| Araceae | Aglaonema commutatum Schott | Aglaonema, cafeto ornamental [34] | Southeast Asia [34] | Leaf, fruit [77] |  | Used as a vertical greenery system (VGS) to contribute to improving air quality [78] | Alkaloids (calcium oxalate crystals, polyhydroxy alkaloids), proteins (latex), terpenes (carotenoids) [77] | Treatment: Buruli ulcer (chronic and debilitating infection of the skin) and reduced swellings [79] | Non-edible. Toxic if consumed [34]. Leaf: infusion [79] |
| Araceae | Anthurium andraeanum Linden ex André | Anthurium, capotillo, flower of love, flamenco flower | America [80] | Leaf, flower |  | Water decontamination (COD, P, coliforms) [48] | Alkaloids (calcium oxalate crystals), glycosides (cyanogenic glycosides), and phenolics [80,81] | na | na |
| Araceae | Spathiphyllum montanum (R. A. Baker) Grayum | Spath, peace liliescuina de moisés, guisnay [20,82,83] | Tropical America [20,82,83] | Leaf [84] |  | Air decontamination with toxins [83] | Phenols (flavonoids) [84] | Activity: antioxidant, anti-inflammatory, antimicrobial, anti-carcinogenic [20,84] | na |
| Asparagaceae | Chlorophytum comosum (Thunb.) Jacques | Tape, malamadre, clorofito, lasso of love, spider plant, ribbon plant [85] | Africa [86] | Leaf, flower, stem [85] |  | Soil decontamination (Al, Pb, Cd salt, trichloroethylene, toluene, formaldehyde, particulate matter, and benzene) [86,87,88] Air decontamination (PM) [89] | Saponin (gitogenin, ecogenin, tigogenin), glycosides and alkaloids [85,86] | Treatment: bronchitis, cough, fracture, and burns [85] Activity: antimicrobial, anti-carcinogenic, hepatoprotective, antitumour properties, and cytotoxicity against cancerous cell lines [85,86] | Arial part: infusion [90] |
| Asteraceae | Bidens andicola Kunth | Ñachac, mìshico, quello-ttica, quico, chiri chiri, zumila [20,91,92,93] | South America [20,94] | Whole plant [93,94] |  | na | Alkaloids, phenolics (flavonoids), saponins, tannins, cardiotonics, steroids, terpenoids (sesquiterpene lactones), and chalcones (chalcone ester glycosides) [91,94,95,96] | Treatment: excessive vaginal fluid, postpartum, diarrhoea, cholera, stomachache, nervous afflictions, skin problems, asthma, eye inflammation, and renal affections [92,94,96] Activity: uterine antihaemorrhagic, antirheumatic, anti-inflammatory, anti-allergenic, antibacterial, antidiabetic, antimalarial, antiviral, antihypertensive, antioxidant, antimicrobial activity, and antispasmodic properties [20,94] Toxicity: It presents a moderate toxic effect [91] | Leaf: salad Whole plant: infusion [93] |
| Asteraceae | Calendula officinalis L. | Calendula, African marigold, Common marigold, Zergul, Garden Marigold, Marigold, Pot Marigold [97,98] | Southern Europe [99] | Flower [99] |  | Soil decontamination (Cd, Pb) [100] | Saponins, sterols, terpenes (carotenoids, volatiles oils), tannins, resins, triterpenoids, phenols, coumarins, and quinones [42,72,97,101,102] | Treatment: used as emollient, vulnerary, moisturising, analgesic, cramps, ulcers, jaundice, and haemorrhoids [99,101] Activity: antioxidant, anti-inflammatory, antibacterial, antifungal, antiviral, antipyretic, antiseptic, antispasmodic, astringent, bitter, candidacies, cardiotonic, carminative, cholagogue, dermagenic, diaphoretic, diuretic, haemostatic, immunostimulant, lymphatic, uterotonic, and as a vasodilator [97,98,99,102,103] Toxicity: The leaves can cause phytodermatitis and cytotoxicity activity [55] | It has a slightly bitter and spicy flavour. Flower: infusion [99] |
| Asteraceae | Centaurea seridis L. | Bracera marine, thorny broom | Mediterranean region [104] | na |  | na | Glucosides, sesquiterpenoids [104,105] | Activity: anti-diabetic [104] | na |
| Asteraceae | Cichorium intybus L. | Brussels chicory, coffee chicory, root chicory, cikoria, nigana, cicoria, juju, radicheta [106,107,108] | Western Asia, Europe, and North Africa [106,107] | Whole plant [107] |  | Soil decontamination with DDT (Dichlorodiphenyltrichloroethane) [88] | Sesquiterpenes lactones (lactucin, lactopicrin), aesculetin, Cichorium), coumarin (scopoletin, 6-7-dihydro coumarin, umbelliferone glycosides, terpenes (oils essential), phenolics [106,107,109] | Treatment: cardiovascular, digestive, and skin protection [107] Activity: antioxidant, hypolipidemic, anti-carcinogenic, anti-allergenic, anti-testicular, antidiabetic, diuretic, anti-inflammatory, analgesic, sedative, immunological, antimicrobial, antiprotozoal, hepatoprotective, neuroprotective, and gastroprotective [106,107] | Leaf: salad, infusion Roots: flour [107] |
| Asteraceae | Chrysanthemum morifolium Ramat | Chrysanthema | Asia [3] | Flower [3] |  | Soil decontamination with Pb | Pyrethroids (pyrethrins, deltamethrin), terpenes (sesquiterpene lactones), and phenolics (chrysanthemin) [3,110] | Treatment: used in the detoxification of blood, regulation of pressure, calming nerves, hypertension, angina, digestive system, muscular-skeletal system, respiratory system, arteriosclerosis, hypertension [3,111,112] Activity: antioxidant, anti-inflammatory, anti-carcinogenic [111] Toxicity: the flowers present phytodermatitis [55] | Flower: infusion, food supplement [3,111,113] |
| Asteraceae | Coreopsis grandiflora Hogg ex Sweet | Coreopsis | America [114] | Flower [115] |  | Soil disturbance [116] | Phenolics [117] | Activity: antioxidant, anti-inflammatory, antimicrobial, antimalarial, antileishmanial, and anti-Alzheimer [117] | Flower: food additive |
| Asteraceae | Cota tinctoria (L.) J. Gay | Golden marguerite, yellow chamomile [118] | Mediterranean region | Whole plant [118] |  | Soil decontamination with B [119] | Terpenes (volatile oils), triterpenes, tannins, and phenolics [118,120,121] | Treatment: gastrointestinal disorders, stomach, haemorrhoids, antispasmodics, stimulating menstrual flow, hepatic insufficiency, and jaundice [118] Activity: antimicrobial, anti-inflammatory, antibacterial, antispasmodic, and sedative [118,120] | Flower: meat and dairy colouring [118] |
| Asteraceae | Dahlia coccinea Cav. | Dahlia, mirasol, mountain dahlia, wild dahlia, sunflower [122] | Mexico [122,123] | Flower, root [122] |  | Soil decontamination with oil [64] | Terpenes (essential oils), polysaccharides (inulin), and acetylene compounds [122] | Activity: antioxidant, anti-inflammatory, anti-carcinogenic, anti-obesity, and gastroprotective [122,124] | Flower: salad, dessert, garnish Root: soup [122] |
| Asteraceae | Dahlia pinnata Cav. | Dahlia, heron flower [20] | Mexico It was declared the national flower of Mexico [20,125] | Flower, root [122] |  | Soil decontamination oil [64] | Terpenes (essential oils), proteins (insulin), monosaccharides (fructose), acids (phytin, polyacetylenes, benzoic acid) [126] Note: Root exudates are nematode toxic | Activity: antimicrobial [20] | Flower: salad dessert, garnish Root: soup [122] |
| Asteraceae | Gaillardia × grandiflora Hort. Ex Van Houtte | Gallant, flower blanket, gold button, bloodsucker, topasa dre | na | Flower |  | na | Alkaloids (oxalates) Note: It presents an inhibitory effect on the pathogenic fungi [127] | na | Non-edible [127] |
| Asteraceae | Tagetes erecta L. | Carnation of the moor, flower of the dead, carnation Chinese, damask, flower crest, French marigold [125,126] | Mexico The traditional day of the Dead flower in Mexico [127] | Flower [127] |  | Water decontamination (textile dye blue 160), HgCl, SnCl2 [128]. Soil decontamination with Cd (hyperaccumulator) and oil [64,129] | Organic acids, terpenes (essential oil), alkaloids, and phenolics [125,130,131] Note: Essential oil is cytogenotoxic. It can be harmful in large amounts [132] | Treatment: therapies and aromatherapies, digestive ailments (colic, parasites, discomfort, and diarrhoea), liver diseases, antiseptic, diuretic, depurative [125,127,133] Activity: antioxidant, anti-carcinogenic, anti-inflammatory, disinfectant, healing, and antifungal [125,127,131] Toxicity: the leaves present phytodermatitis [55,127] | Flower: infusion, salad, fried. It is a natural colouring and has a bitter taste [12,113] |
| Asteraceae | Taraxacum campylodes G. E. Haglund | Dandelion, bitter chicory, diente de león [31,91,92] | Europe and Asia [120] | Whole plant [113,134] |  | Soil decontamination (Cu, Zn, Mn, Ni, Cr, Fe, and Pb) [135,136] | Alkaloids (phytosterol, taraxacin, oxalates), phenolic (taraxastero, stigmasterol, chicoric acid, caffeic acid, acopoletin) [137,138,139,140] | Treatment: depuratives help the liver, kidney, stomachache, gall bladder, diuretic effect, constipation, clean skin impurities, acne, and hives [92,134,137,139] Activity: hepatoprotective, antirheumatic, spasmolytic, diuretic, anti-inflammatory, anti-carcinogenic, antirheumatic, anti-allergenic, anticoagulant, and anti-carcinogenic [31,137]. Toxicity: the leaves present phytodermatitis [55] | Leaf: salad, cooked Root: coffee Flower: with olive oil, cakes, fries, and wine Whole plant: infusion [113,137] |
| Asteraceae | Zinnia elegans L. | Guadalajara, mystical rose, paper flower, field chinita | Mexico and Central America [141] | Leaf, flower |  | Soil decontamination (Pb and Cr) [65] | Phenols (flavonoids), glycosides, tannins, and saponins [141] | Treatment: malaria and stomach pain Activity: hepatoprotective, antiparasitic, antifungal, antibacterial, and antioxidant [141] | Flower: salad, infusion |
| Balsaminaceae | Impatiens walleriana Hook. f. | House joy, bear ears, balsam, miramelindo | Africa and Asia | Leaf, stem, flower |  | Soil decontamination with Cd (hyperaccumulator) [129] | Naphthoquinones, phenols (flavonoids), saponins (triterpenoid saponins), alkaloids (phytosterols, proteins, and terpenes (essential oils) [129,142] | Treatment: abdominal pain, ulcers, amenorrhea [129] | Flower: infusion, salad, garrison It has a sweet flavour. |
| Begoniaceae | Begonia cucullata Willd. | Begonia, sugar flower [126] | Brazil [126] | Flower [143] |  | Soil decontamination with oil [64] | Phenolics, terpenes [144] | Activity: antispasmodic, astringent, ophthalmic, poultice, and stomachic activity [143] | na |
| Begoniaceae | Begonia × tuberhybrida Voss | Begonia [12,126] | Andes [126] | Flower [12] |  | na | Alkaloids (oxalic acid, tetracyclic triterpene), phenolics [113,144] | Activity: antispasmodic, astringent, ophthalmic, poultice, and stomachic [12,143] | Petals are edible. This flower has a lemon flavour [12,113,143] |
| Bignoniaceae | Tecoma capensis (Thunb.) Lindl. | Cape Honeysuckle, tecoma [145,146] | South Africa [147] | Leaf, flower, root [148] |  | na | na It is an invasive species [147] | Treatment: pneumonia, enteritis, diarrhoea, fragrance, tonic, eliminating placenta retained in childbirth, snakebite, sleeplessness, induced sleep [148,149,150] Activity: antimicrobial, antifungal, antipyretic, antioxidant [149] | Arial part: infusion [148] |
| Bignoniaceae | Tecoma stans (L.) Juss. ex Kunth | Yellow bell, tronadora, huiztontli, huiztonxochitl [151] | Mexico [147,151] | Leaf, flower [152] |  | Soil and water decontamination (FeCl3, CaCO3) [128] | Alkaloids (tecomine, tecostamine), phenolics, steroids, and tannins [151,153] | Treatment: arterial hypotension, hypoglycaemia, and urinary disorder [151,152] Activity: antidiabetic, antimicrobial, and antioxidant [149,152] | Non-edible |
| Boraginaceae | Heliotropium arborescens L. | Vanilla of garden, heliotrope, grass of the mule, violoncello, cherry pie, heliotrope [154,155] | Peru | Leaf, stem, flower [155] |  | na | Esters (heliotropin, benzyl acetate), alkaloids (heliotrine, oxalates), phenols (vanillin, cynoglossin, caffeic acid), benzaldehydes (benzaldehyde, p-anisaldehyde), lithospermic acid [139,154,155] | Treatment: headache, sun stoke, sinus cancer, mucus relief, diuretic, uterine displacement, fever, migraine, high blood pressure, diarrhoea, breast cancer, kidney infection, pressure in the stomach and sternum, uterine displacement, and dysmenorrhea [139,155] | Arial part: infusion |
| Brassicaceae | Alyssum montanum L. | Spanish, garlic herb, rabies herb, rage herb | Europe [156] | Flower |  | Soil decontamination (Cd, Ni, Pb, and Cu) [157,158] | Glucosinolates (goitrogenic glycosides) | na | Non-edible |
| Brassicaceae | Diplotaxis tenuifolia (L.) DC. | Rucola, yellow flower, rustic [159,160] | Mediterranean region [160] | Leaf |  | Soil decontamination with Pb [161]. | Glucosinolates Note: It presents allelopathic properties (S-glucopyranosyl thiohydroximate) [162,163,164] | Treatment: digestive, diabetes, cardiovascular disorders, and cancer [159] Activity: antitumor [159] | Leaf: salad It is used in the food industry (IV gamma) [159,160] |
| Brassicaceae | Matthiola incana (L.) R. Br. | Alehí, Jasmine ashtray, White viola [134] | South Europe | Flower [134] |  | na | Isoprenoids (tocopherols), proteins (hormones), and anti-pathogens [165] | Treatment: traditional medicine, stomachache, colic, and diarrhoea for frighten [134,165] | Arial part: infusion, garnish, salad, desserts [134] |
| Cannabaceae | Cannabis sativa L. | Marijuana, marihuana, hashish, hachís, hemp [53] | Asia [53] | Whole plant [47,166] |  | Soil decontamination (Cu, Cd, As, Ti, Cr, and Ni) [167]. | Phenols (tannins), cannabinoids, terpenophenols (tetrahydrocannabinol (THC), cannabidiol (CDB)), and alkaloids [168,169] | Treatment: developmental disorder, hypertension, asthma, diabetes, heart conditions, blood pressure, epilepsy, and glaucoma [47,53,170] Activity: analgesic, antiemetic, anti-carcinogenic, antispasmodics [53] Note: the leaves are mutagenic without metabolic activation [66] | Arial part: infusion. It is a source of fibre, food, oil, and medicine [169,171] |
| Cannaceae | Canna indica L. | Achira, achira roja, achera, sago, spark, Indian cane, papantla [20,34,172] | South America [20,34,173] | Root, flowers |  | Water decontamination (Cu, Zn, fertilisers, carbamazepine, and insecticides) [48] | Alkaloids, phenols (tannins) [42,173,174] | Treatment: peptic ulcer, diarrhoea, and ulcerative colitis Activity: antibacterial, anthelmintic, antiviral, anti-inflammatory, hepatoprotective, antidiarrheal, anti-carcinogenic, analgesic, and antioxidant [20,62,173,175] | Root: starch Leaf: food cover [174] |
| Caryophyllaceae | Dianthus caryophyllus L. | Carnation, claveles [176] | Europe and Asia [176] | Flower [134] |  | na | Triterpenes, saponins, terpenoids (carotenoids), and phenolics [42,176,177] | Treatment: HIV, simple herpes, hepatitis, vomiting, and gastric disorders [134,176,178] Activity: antibacterial, anti-fungal, antiviral, cardiotonic, diaphoretic, vermifuge, gastroprotective, anti-carcinogenic [176,179] | It has a slightly bitter flavour. Flower: salad, butter, garnish [180] |
| Caryophyllaceae | Dianthus chinensis L. | Dianthus, carnation, Chinese carnation, pae-raeng-ee-kot [181] | China [182] | Leaf, stem, flower |  | na | Phenols (eugenol), alcohols (phenyl ethyl alcohol), glycosides (melosides A and L, dianchinenosides A, B, C, and D), saponins [181,183] | Treatment: menostasis, gonorrhoea, diuretics, emmenagogue, and cough [181,182,183] Activity: anti-inflammatory, diuretic, analgesic, anti-hepatotoxic, hypotensive, anthelmintic, intestinal peristaltic, antitumor, antioxidant, antitumor, antibacterial, antifungal [181,183] | It is slightly bitter. Flower: infusion, salad, desserts, garnish [113] |
| Caryophyllaceae | Gypsophila paniculata L. | Cloud, bridal veil, paniculata, baby’s breath, sabunotu, Tibbi sabunotu [184] | Turkey, Caucasia, and Iran [185] | Leaf, stem, flower |  | Soil decontamination (B) [185] | Allelochemical phenolic acids and saponins (triterpenoid saponins) Note: It presents insecticidal activity. It is an invasive perennial plant [184,186] | Treatment: cough, respiration system, bronchitis, stomach disorders, bone deformations, pimples, bile disorders, liver problems, rheumatism, and skin diseases [185]. Activity: antimicrobial [184] | Arial part: infusion |
| Caryophyllaceae | Saponaria officinalis L. | Soap dish, soap flower, sabunotu, tibbi sabunotu, karga sabunu, soapwort [185,187] | Turkey, Caucasia, and Iran | Root, leaf [185] |  | Soil and water decontamination (hydrocarbon, Cd(II), Zn(II), Cu (II) [187] | Triterpenoid, saponins (saponarioside A/B) [185,187,188] | Treatment: influenza, stomach disorders, simple herpes, bone deformations, cough, bronchitis, rheumatism, pimples, skin diseases, bile disorders and hepatic eruptions, venereal ulcers, diuretic, diaphoretic, cholagogue, and hepatic eruptions [185,187,188] Activity: anti-microbial, antipyretic, antiseptic, anthelmintic, tonic, diuretic, anti-diabetic [185,187,189] | Arial part: infusion [189] |
| Celastraceae | Euonymus japonicus Thunb. | Evonimo, bonetero | Japan [190] | Fruit, leaf, seed [190] |  | Air decontamination [191] | Alkaloids, terpenes, phenolics [190,191,192] | na | Fruit: the powder is a natural colouring for butter [190] |
| Convolvulaceae | Convolvulus althaeoides L. | Bells of the virgin, carriguela, correhuela, bindweed, leblab elhokul [193] | Mediterranean region [193,194] | Leaf, root, flowers [194,195] |  | na | Alkaloids, saponins, phenolics, chlorophylls, and terpenes (carotenoids) [193] Note: Essential oil presents cytotoxic activities and is considered “weed” [194] | Treatment: wound healing, asthma Activity: laxative, purgative, antimalarial, antimicrobial, antioxidant [193,194,195] | Arial part: infusion |
| Convolvulaceae | Convolvulus pseudoscammonia C. Koch | Meadow bell, scammony Syrian bidweed, Purgin bindweed. | Asia [196] | Leaf, stem, flower [196] |  | na | Alkaloids, saponins, terpenes (resin), phenols (dihydroxy cinnamic acid, flavonols), and coumarins (beta-methyl-aesculetin) [62,196,197] | Treatment: uterotonic, abortifacient, treatment of oedema, ascites, simple obesity, lung fever, ardent fever, purgative, vasorelaxant [196] Activity: antimalarial, anti-platelet aggregation, anti-carcinogenic, cell protector effect, anti-carcinogenic [194,196] | Arial part: infusion [196] |
| Crassulaceae | Kalanchoe blossfeldiana Poelln. | Kalanchoe [126] | Madagascar and East and South Africa [126,198] | Flower [198] |  | Soil decontamination with benzene [88]. | Phenolics, coumarins, bufadienolides, triterpenoids, phenanthrenes, sterols, fatty acids, and kalanchosine dimalate salt [199,200] Note: All plants present high toxicity (cardioactive steroids or cardiac glycosides) [55] | Treatment: skin problems, periodontal disease, cheilitis, cracking lips in children, wounds, insect bites, ear infections, dysentery, fever, abscesses, cholera, urinary disorders, arthritis, gastric ulcers, rheumatism, pulmonary disease, rheumatoid arthritis, coughs, gastric ulcers Activity: antimicrobial [199] | Arial part: infusion |
| Cucurbitaceae | Citrullus lanatus (Thunb.) Matsum. and Nakai | Watermelon, Sandia, side, patilla [34,201,202] | Southern Africa [201,203] | Fruit, seed [53,201] |  | Water decontamination with Cd [204] | Saponin, alkaloids, phenols (anthocyanins, tannins, phenolic acids, flavonoids), terpenes (carotenoids, monoterpenes) [201,203] | Activity: antimicrobial, antioxidant, anti-inflammatory, antispasmodic, anti-prostatic, analgesic, antidiabetic, laxative, antiulcer, and hepatoprotective [201,202,203] | Fruit: widely used in the food industry [34] |
| Cucurbitaceae | Cucurbita maxima Duchesne | Squash, pumpkin, flor de calabacín, auyama, calabaza, sapayo, zapallo [34,92] | South Africa [34] | Fruit, leaf [202] |  | na | Saponins, alkaloids (cardenolides), and phenols (flavonoids) [202,205] | Treatment: Seed oil is used for the treatment of benign prostatic hypertrophy, laxative Activity: laxative, antimicrobial [92] Toxicity: LC50 = 4311 µg/mL | Fruit: widely used in the food industry [34] |
| Ericaceae | Rhododendron simsii Planch. | Azalea indica, azalea [206] | China [207] | Leaf, flower [207] |  | na | Alkaloids (grayanotoxane (pollen, nectar, and leaves)), phenols (flavonoids), and benzoic acid derivatives [207] | Treatment: gastrointestinal disorders, asthma, arthritis, skin diseases, cough, resolving sore toxin, amenorrhea, expectorant, and bronchitis [207] Activity: anti-inflammatory, anti-herpes, antioxidant, antiviral, hepatoprotective, and sedative [207,208] | Arial part: infusion |
| Euphorbiaceae | Euphorbia milii Des Moul. | Crown of Christ, corona de espinas, espinas de cristo [34,209] | Madagascar [34] | Whole plant |  | Air decontamination [210] | Terpenes (triterpenoids, diterpenoids), phenols (flavonoid, tannins s), proteins (latex) [210,211] | Treatment: respiratory tract inflammation, diarrhoea, skin ailments, gonorrhoea, tumours, cough, dysentery, asthma, hepatitis, abdominal oedema Activity: sedative and analgesic [211] | Arial part: infusion |
| Fabaceae | Brownea macrophylla Linden | Brownea, Mountain rose, Cross stick, Male cross stick [212] | Colombia, Panama, and Venezuela [213] | Flower [212] |  | na | na | Treatment: haemostatic, for haemorrhages, birth control, and against snake bites [212] | Non-edible |
| Fabaceae | Lathyrus aphaca L. | Yellow pea, aphaca, wild pea, Indian flower [214] | Europe, Asia, Africa [214] | Flower [214] |  | na | Seeds contain toxic amino acids [133] | na | Seed: widely used in the food industry |
| Fabaceae | Senna alexandrina Mill. | Senna, cassia, sen, cassia angustifolia [53,214] | Egypt [214,215] | Leaf, fruit, flower [53,214,216] |  | Soil decontamination (Al, Ba, Mn, and Zn) | Glycosides (anthraquinone derivatives, senna glycosides) [214,215] | Activity: laxative, antipyretic, purgative, diuretic, stomach [53,215,216] | Arial part: infusion |
| Fabaceae | Senna corymbosa (Lam.) H. S. Irwin & Barneby | Buttercup bush, Argentine Senna, sena del campo, rama negra, mata negra [20,217,218] | South America [20,217] | Flower |  | na | Glycosides (anthraquinone glycosides, naphthoquinone), phenols (flavonoids) [217] | Activity: laxative, purgative, antidiabetic, hepatoprotective, antimalarial, antipyretic, antiasthmatic, antiviral, and antibacterial [20,217,218] | Arial part: infusion |
| Fabaceae | Senna didymobotrya (Fresen.) H. S. Irwin & Barneby | Senna, popcorn cassia [219] | na | Whole plant [220,221] |  | na | Alkaloids, anthraquinones, phenols (condensed tannins, hydrolysable tannins), saponins, sterols, and steroids [221] | Treatment: madness, ringworm infections, leprosy, syphilis, diabetes, convulsions, stomach complaints, wound healing, an antidote for snakebites, haemorrhoids, sickle cell anaemia. Activity: purgative, anti-inflammatory, antimalarial, hepatoprotective, antimicrobial [219,220] | Arial part: infusion |
| Fabaceae | Senna papillosa (Britton & Rose) H.S. Irwin & Barneby | Sena, candelillo | na | Flower |  | na | Phenols (flavonoids), coumarins, mellilotic acid, and iridoids | Activity: antimalarial [222] | Non-edible |
| Fabaceae | Styphnolobium japonicum (L.) Schott | Acacia from Japan, sófora, Japanase pagoda tree, Huai, Chinese scholar tree [223] | China [223] | Leaf, root, flower, seed [224] |  | Soil decontamination (Cu, Cr, Cd, Hg, Ni, Zn and Pb) [225,226] | Alkaloids, phenols (isoflavonoids), triterpenoids [223,224] | Treatment: haemorrhoids, uterus problems, intestinal bleeding, arteriosclerosis, hypertension, cooling blood, and haemostasis [223,224] Activity: anti-inflammatory, antibacterial, antiosteoporotic, antihyperglycemic, anti-obesity, and antitumor [223,224] | Arial part: infusion [224] Flower: tea, cake [227] |
| Fabaceae | Trifolium alexandrinum L. | Clover, berseem clover [228] | Mediterranean | Whole plant [229] |  | na | Cyanogenic glycosides [229] | Treatment: bronchitis, asthma, burns, cough, ulcers, sedation, polycystic ovary, heart disorders, colic Activity: anti-diabetic and laxative [229] | na |
| Geraniaceae | Pelargonium domesticum L. H. Bailey | Geranium thinking, ral geranium, malvon thinking [230] | Africa [126] | Whole plant [230] |  | Soil decontamination with benzene [88] | na | Treatment: respiratory infection, sleep disturbance, fatigue, loss of appetite, wound healing [230] Toxicity: the leaves and stems present phytodermatitis [55] | Flower: dessert, cake, drink, salad, flower water, garnish. |
| Geraniaceae | Pelargonium peltatum (L.) L’Hér. | Gitanilla, geranium of ivy, geranio [230] | Africa [126] | Whole plant [230] |  | na | Phenols, terpenes (essential oil) [231] | Treatment: heal wounds Activity: antioxidant and antimicrobial [230,231,232] Toxicity: the leaves and stems present phytodermatitis [55] | Leaf has an astringent and bitter taste. The leaves and stems present gastrointestinal toxins. |
| Geraniaceae | Pelargonium × hortorum L. H. Bailey | Common geranium, malvon, garden geranium, geranio común | Africa [233] | Whole plant |  | Soil decontamination (Cd and Pb) [234] | Triterpenoids, sterols, and phenols (flavonoids, anacardic acids) [235] | Treatment: respiratory infection, sleep disturbance, fatigue, loss of appetite, wound healing Activity: antioxidant and insecticidal [230,236] Toxicity: the flowers induce paralysis [237] | Flower: dessert, cake, drink, salad, flower water, garnish. |
| Goodeniaceae | Scaevola aemula R. Bronw | Fan flower | Australian [238] | Flower |  | na | na | na | Arial part: infusion |
| Hydrangeacea | Hydrangea petiolaris Siebold Zuc | Hydrangea | Himalaya | Flower |  | na | na | Activity: anti-inflammatory, antibacterial [239] | Arial part: infusion |
| Iridaceae | Gladiolus communis L. | Gladiolo [240] | Africa [240] | na |  | Water decontamination (Zn, Pb, Cu) [241] | na | Treatment: obesity, asthma, diabetes, and fertility | It tastes like lettuce. Flower: salad, garnish |
| Juglandaceae | Pterocarya stenoptera C. DC. | Chinese ash, Chinese wingnut, ghost maple, willow, gold trees [242,243] | China | Leaf, bark |  | na | Phenols (tannins) [242] | Treatment: insecticide, remove scabies, eczema, abscesses, rheumatism, cold-damp bone ache, odontia, head pain, haemorrhoids, itch, pyrosis, ulcer [242,243,244] Activity: carminative, anthelmintic, anti-herpes [244] | Arial part: infusion [244] |
| Lamiaceae | Agastache foeniculum (Pursh) Kuntze | Aniseed swab, hyssop [108,245] | North America [245] | Aerial parts, seed, root [245] |  | na | Note: Essential oil presents toxicity with LC50 between 18.8 to 21.6 µL/L [245,246] | Activity: antimicrobial, antiviral, antimutagenic, anti-inflammatory, and antioxidant [245] | The flower has an anise flavour. |
| Lamiaceae | Lavandula angustifolia Mill. | Alucema, lavender [126] | Mediterranean region [126,247] | Leaf, stem, flower |  | na | Terpenes (essential oils, triterpenoids, sesquiterpene, linalool, linalyl acetate), phenols (flavonoids (apigenin, luteolin)), coumarins [247,248] | Treatment: respiratory, muscular-skeletal, and cardiovascular [249] Toxicity: the leaves can produce phytodermatitis [55] | It is an aromatic herb. Arial part: infusion |
| Lamiaceae | Mentha suaveolens Ehrh. | Mastranzo, mint suaveolens [250] | Occidental Mediterranean | Leaf, stem, flower |  | na | Terpenoids (essential oils), phenols (flavonoids) Note: It is toxic in high doses (peppermint oil) [251,252] | Activity: antimicrobial, antiviral, antioxidant, tonic, stimulating, stomachic, carminative, analgesic, choleretic, antispasmodic, sedative, hypotensive, insecticidal, analgesic, anti-inflammatory [251] Toxicity: the leaves can cause phytodermatitis [55] | It is used in the food industry. Arial part: infusion |
| Lamiaceae | Mentha × piperita L. | Peppermint, menta, hierbabuena [126] | Natural hybrid of Mentha aquatica and Mentha spicata [253] | Leaf, stem, flower |  | Fatty acids, terpenes, and phenolics [253] | Treatment: disorders of the mental-nervous, respiratory, digestive, metabolic, and nutritional Activity: antifungal, antiviral, antioxidant, anti-allergenic [253] Toxicity: essential oils and the leaves can cause phytodermatitis [55] | It is used in the food industry. Arial part: seasoning, cold drinks, salads [253] | |
| Lamiaceae | Rosmarinus officinalis L. | Romero, Rosemary [31,108,126] | Mediterranean region [126] | Leaf, stem, flower |  | Soil decontamination (Ni, Cu, Zn, Cr, Co, Pb, and Cd) [254] | Terpenoids (essential oil: pinene, camphene, cineol, borneol, camphor) [255] | Treatment: cardiovascular, skin, muscular-skeletal diseases, sensory, nutritional, reproductive, mental-nervous, digestive, and respiratory systems [31,178,249,250,255] | Arial part: dessert, sorbet, season meet, infusion [227] |
| Lamiaceae | Salvia leucantha Cav. | Mexican bush sage or sage [256] | East Mexican and Tropical America [20] | Flower |  | na | Diterpenoids(salvigenane and isosalvipuberulan) [257] | Treatment: mental, nervous, gastrointestinal, menstrual, digestive disorder, blood circulatory regulator [256] Activity: antibacterial, antiviral, antitumor, spasmolytic, antioxidant, anti-inflammatory [20,256] | Flower: flavouring, condiment |
| Lamiaceae | Salvia microphylla Kunth | Asia | Flower |  | na | Terpenoids (essential oils, diterpenoids, sesquiterpenoids, triterpenoids), phenols (flavonoids) [258] | Treatment: digestive and respiratory problems | Arial part: infusion [257] | |
| Lamiaceae | Salvia splendens Sellow ex Schult. | Red salvia, banderilla, salvia scarlata | Brazil [259] | Flower |  | na | Terpenoids (monoterpenes, diterpenoids, sesquiterpenes, and tanshinones) and phenols (flavonoids, savianin, monardacin, and their demalonyl derivatives). Note: It presents cytotoxic activity [257,260] | Activity: antioxidant, neuroprotective, antimicrobial, antibacterial, anti-carcinogenic, anti-inflammatory, analgesic, anaesthetic, anti-stress, antiulcer, antimutagenic, antidiabetic, diuretic, haemostatic, hypoglycaemic, diaphoretic, and antidepressant [257,260,261] | It Is flavouring. Arial part: infusion, sweet-salty dishes [257] |
| Lamiaceae | Vitex agnus-castus L. | Pepper of the mountains, willow trigger | Mediterranean region | Flower |  | Water decontamination [262] | Phenolics, terpenes (essential oils), alkaloids [263] | Treatment: premenstrual, digestive, respiratory system, premenstrual dysphoric disorder, lactation difficulties, low fertility, menopause [249,263,264,265,266] | It is used in the food industry. Arial part: infusion [266] |
| Lythraceae | Cuphea hyssopifolia Kunth | False brecina, cufea, false Mexican heather, false erica | Central and South America [267] | Flower |  | na | Phenols (tannins, flavonoids, and phenolic acids), sterols, and terpenes (triterpenes) [267] Note: It presents cytotoxic activity [268] | Treatment: stomach pain, syphilis, and cancer Activity: antiviral, antimicrobial, antioxidant, hepatoprotective, antitumor [267,268] | Arial part: infusion |
| Lythraceae | Lagerstroemia indica L. | Jupiter tree, mousse, lilac of the Indies, southern lilac, crepe | China [269] | Flower |  | Water decontamination with fluoride [270] | Alkaloids, glycosides (anthraquinone glycosides), phenols (flavonoids), saponins [271] Note: It presents cytotoxic activity. LC50 = 60 µg/mL and LC90 = 100 60 µg/mL [272] | Treatment: stomach pain, weight loss, lower blood sugar Activity: antioxidant, antibacterial, antiviral, anti-inflammatory, anti-gout, anti-diarrheal, anti-obesity, and anti-fibrotic [271] | Arial part: infusion |
| Lythraceae | Punica granatum L. | Pomegranate, Granada, Dalim gach [27] | Asia and Mediterranean Europe [273] | Fruit, leaf [27] |  | Soil decontamination [274] | Phenols (catechins), alkaloids, [273,275] | Treatment: bronchitis, tuberculosis, diarrhoea, and protecting the kidney [27,249,276] Activity: anti-carcinogenic, antimicrobial, antihypertensive, anti-diabetic, anti-HSV-1, diuretic, antioxidant [67,273,276] | Fruit: widely used in the food industry [276] Flower: infusion Leaf: fried [27] |
| Magnoliaceae | Magnolia grandiflora L. | Magnolia [126,277] | South-eastern United States [126,277] | Flower |  | Soil decontamination (Cu, Cr, Zn, Ni, Cd, Hg) [226] | Phenols (flavonoids), terpenes (sesquiterpenes, essential oils) [277] | Treatment: flatulent dyspepsia, cough, asthma, digestive problems, and emotional distress [277] Activity: antifungal, anti-melanogenic, antioxidant and antimicrobial [277] | Flower: infusion |
| Malvaceae | Ceiba speciosa (A.St.-Hil.) Ravenna | Chorisia, Bottle tree; Drunken tree | South America | Flower |  | na | Phenols (flavonoids), alkaloids, coumarins, terpenes (sesquiterpenes, sesquiterpene lactones, triterpenes), steroids, lignans, cyclopropenium fatty acids, and oxidised naphthalenes [278,279] | Treatment: fever, diabetes, headache, diarrhoea, parasitic infections, rheumatism, and peptic ulcer Activity: anti-inflammatory, antimicrobial, hepatoprotective, cytotoxic, antioxidant, hypoglycaemic, and antipyretic [279] | It is used for a variety of ailments. Seed: culinary and industrial |
| Malvaceae | Gossypium arboreum L. | Cotton, cotonera, coto, algodón | Asia | Leaf, root, seed, flower |  | Soil decontamination with oil [64] | Phenols (gossypetin 8-o-rhamnoside, gossypetin 8-o-glucoside) and terpenes (gossypol). | Treatment: healing of wounds, ulcers, bruises, respiratory, and skin diseases | Non-edible |
| Malvaceae | Hibiscus rosa-sinensis L. | Chinese rose, cayenne, pop, hibiscus, papo, San Joaquín, carnation, Laal joba [27] | Eastern Asia National flower of Malaysia, Dominican Republic, Puerto Rico, Hawaii, Barranquilla, and Barrancabermeja (Colombia) | Flower, root, leaf [27] |  | Water decontamination with zinc ions [280] | Ketones (chloroacetophenone), phenolics (tannins), steroids, proteins (mucilage) [42,72,281] | Treatment: hypertension, inflammations, dysentery, and respiratory tract [27] Activity: antispasmodic, analgesic, astringent, laxative, antioxidant, antimicrobial, anti-diabetic, cardioprotective, and anti-anxiety [281,282,283] Toxicity: the leaves can cause phytodermatitis [55] | Flower: salad, cooked, infusion Root: salad Leaf: juice [27] |
| Malvaceae | Hibiscus sabdariffa L. | Rose of Jamaica, rose of Abyssinia, hibiscus red sorrel, rosella, tart of guinea, alleluia, susur, flor de Jamaica [53,284] | West Africa [285] | Leaf, flower [285] |  | Soil decontamination (Mn and As) [286] | Phenols (protocatechuic acid) [42,284,285] | Treatment: folk remedy for abscesses, bilious conditions, cancer, cough, dysuria, cardiovascular problems, and scurvy [249,284,287] Activity: antioxidant, antiseptic, astringent, diuretic, emollient, purgative, and sedative [53,285,287] | It is a resource for food and medicine. Flower: food colouring, beverages, jams [285,287,288] |
| Malvaceae | Hibiscus syriacus L. | Altea, Syria rose, wasp, hibiscus [34] | Asia It is the national flower of Korea. Origin unknown [34,289] | Leaf, flower, root |  | na | Terpenes (essential oils, pentacyclic triterpene esters), lignans, coumarins, and phenolics [289] | Activity: antioxidant, dermatological, anti-proliferative, anti-carcinogenic, antimicrobial, antiviral, anti-inflammatory, anti-tyrosinase [289,290] | Flower and leaf: salad, cooked, infusion |
| Malvaceae | Malvaviscus arboreus Cav | Marshmallow, false hibiscus, azocopacle, manzanita [291] | South and Central America, Southeastern United States [292] | Flower [292] |  | na | Phenolics, sterols, fatty acids [292] | Treatment: dysentery, stomach pain, ulcers, and coughs [292,293] Activity: antioxidant, antimicrobial, thrombolytic, anti-inflammatory, cytotoxic, hepatoprotective [292] | Arial part: infusion, salads [292] |
| Nyctaginaceae | Bougainvillea spectabilis Willd. | Bougainvillea, bogambilya, bongabilya, great bougainvillea [294] | South America [20,294] | Leaf, stem [294] |  | Soil decontamination (Cu, Zn) [295] | Phenols (flavonoids, tannins), saponins, sterols, triterpenes, and alkaloids [294,296] | Treatment: stomach, hepatitis, cough [294] Activity: analgesic, anti-diabetic, anti-inflammatory, antimicrobial, astringents, diuretics, antifertility [20,283,294,297] | Flower: infusion, salad, fried [294] |
| Nyctaginaceae | Mirabilis jalapa L. | Don Diego at night, dompedros, parakeet, wonder of Peru, carnation [20] | South America [20,126] | Leaf, root, flower [298,299] |  | Soil decontamination (total petroleum hydrocarbons) [64] | Triterpenes, proteins, phenolics, alkaloids, and steroids [298,299] | Treatment: anthrax Activity: antitumor, virus inhibitor, anti-inflammatory, antimicrobial, antioxidant, and antidiarrheal [20,299,300] | Flower: food colouring Leaf: cooked, infusion Root: infusion Seed: infusion |
| Oleaceae | Jasminum sambac (L.) Aiton | Diamela, Arabian Jasmine, Jasmine diamela, Jasmine paper, mostia, Lily jasmine [301] | India National flower of the Philippines. It is one of the three important flowers in Indonesia [301] | Leaf, flower, root [227,301] |  | Soil decontamination with Pb [302] | Alkaloids, phenols (flavonoids, tannins), terpenoids (essential oils), coumarins, glycosides (cardiac glycosides), steroids, saponins, and phytosterols [303] | Treatment: cough, reducing sputum, cancer, uterine bleeding, ulceration, leprosy, skin diseases, and wound healing [227,301] Activity: antioxidant, anti-inflammatory, anti-carcinogenic, anti-obesity, and neuroprotective [301] | Flower: infusion, salad [227] |
| Onagraceae | Fuchsia magellanica Lam. | Fuchsia magellanica [20] | Peru, Chile, and Argentina [20] | Leaf, stem, fruit, flower |  | na | Phenolics [304] | Treatment: scarce menstruation and increased flow of urine [20] | It has a slightly acidic flavour. Fruit and flower: infusion |
| Orchidaceae | Phalaenopsis aphrodite Rchb. f. | Orchid | It is considered an Indonesian national flower. |  | na | Alkaloids (phanaelopsin T), phenolics | Activity: antioxidant | na | |
| Passifloraceae | Passiflora × belotii Pépin | Passionflower | North, Central and South America [305] | Leaf, flower, fruit |  | na | Phenols (flavonoids) [306] | Treatment: sedative, hypnotic, antispasmodic, and hypotensive | Flower: infusion |
| Plantaginaceae | Antirrhinum majus L. | Scrofularia, dragon mouth, bunnies, dragoncitos, gallitos [126,307] | Mediterranean region [126] | Flower |  | Soil decontamination (Pb and petroleum) [308,309] | Saponins, phenols [42] | Treatment: scurvy, liver disorders, tumours, haemorrhages Activity: diuretics [307] | Flower: salad [58] |
| Plantaginaceae | Plantago major L. | Llantén [38] | Europe | Leaf, seed |  | Soil decontamination (Cu, Mn, Zn, Pb and Cr) [214] | Proteins (mucilage), phenols (tannins), chromogenic glycosides (catapol), and alkaloids (noscapid) [309] | Treatment: digestive, stomach upset, intestine inflammation, abscesses, cold pimples, metabolic, muscular-skeletal, respiratory, mental-nervous, liver, kidney, rheumatism, wounds, dysentery, burns, angina, asthma, fever, tuberculosis, whooping cough, chronic renal inflammation, dermal diseases, bronchitis, purgative, arthrosis, skin problems, haemorrhoids, blood pressure, and heart afflictions [134,195,249,310,311] | Arial part: infusion [309] |
| Plantaginaceae | Russelia equisetiformis Schltdl. & Cham. | Ruselia, tears of love, firecracker, coral, fountain plant [312] | Tropical America [313] | Leaf, flower [313] |  | na | Sterols, triterpenes, saponins [314] | Treatment: malaria, cancer, and inflammatory diseases [313] Activity: antimicrobial [314] Toxicity: Its present cytotoxic activity [312] | Arial part: infusion [314] |
| Plumbaginaceae | Limonium sinuatum (L.) Mill. | Blue inmortelle, capitana, Straw flower, limoniun, paper flower [214] | Mediterranean region [315] | Flower [28] |  | Soil decontamination with Pb [28] | na | Treatment: helps prevent the increase in glucose levels [28] Activity: antioxidant [316] | Flower: food additive, infusion [316] |
| Plumbaginaceae | Plumbago auriculata Lam. | Plumbago, Blue Jasmine, azulina, cape leadwort [34] | South Africa [34] | Flower, root [150] |  | Soil decontamination (metalliferous mines and phytoremediation) [317] | Phenolics (tannins) [318] | Treatment: headache, warts, fractures, oedema, malaria, and skin lesions Activity: sedative and antimicrobial [150,319] | Arial part: infusion |
| Polygonaceae | Fallopia aubertii (L. Henry) Holub | Gabriele Falloppio, Fallopius [320] | Turkestan [320] | Flower |  | na | Phenols (flavonoids, tannins), terpenes (carotenoids, triterpenes), sterols, [320] | Activity: antioxidant, anti-carcinogenic, and antimutagenic Toxicity: It presents cytotoxic activity [320] | na |
| Polygonaceae | Polygala vulgaris L. | Common polygala | Europe | Flower |  | na | na | na | na |
| Portulacaceae | Portulaca oleracea L. | Verdolaga, ghotika, pinyin, krokot, little hogweed, purslane [40,126] | India | Leaf, stem, flower [40] |  | Soil decontamination with Cr (VI) [321] | Alkaloids (oxalic acid), coumarins, phenols (flavonoids, tannins), glycosides (cardiac glycosides), anthraquinones, linoleic acid, saponins [322,323] | Treatment: used in musculoskeletal, nutritional, mental-nerve, cardiovascular, haemorrhoids, and gastrointestinal disorders [40,323] Activity: anti-diarrheal, anti-inflammatory, anthelmintic, diuretic, antiasthmatic, anti-bronchitis, anti-Buruli ulcer, antioxidant, and hypoglycaemic [40,79,324] | Arial part: raw or cooked [40,323] |
| Ranunculaceae | Ranunculus asiaticus L. | Mediterranean region [325] | Flower |  | na | Alkaloids | Activity: antibacterial [326] | na | |
| Rosaceae | Fragaria × ananassa (Duchesne ex Weston) Duchesne | Strawberry, fruit billa [250] | Europe | Fruit |  | na | Phenolics, vitamin C [327] | Activity: antimicrobial, anti-allergenic, antihypertensive [327] | Fruit: widely used in the food industry |
| Rosaceae | Rosa hybrid Vill. | Rosa | na | Flower |  | Soil decontamination (As, Co, Mo, and Ni) [328] | Phenols (glycosylated cyanidin’, pelargonidin) [329,330] | Treatment: used in respiratory and dermatological diseases and arthritis. Activity: antioxidant, anti-inflammatory laxative, and astringent [330] | It is sweet and aromatic. Flower: dessert, sweet, savoury dishes |
| Rubiaceae | Gardenia jasminoides J. Ellis | Gardenia, Cape Jasmine | Asia | Fruit [331] |  | Soil decontamination (alumina and aluminium salts) [332] | Phenols (flavonoids), terpenoids, and organic acids [333] | Activity: antioxidant, anti-inflammatory, and fibrinolytic [331,333] Toxicity: the fruit can cause phytodermatitis. It presents a cytotoxic effect [55]. | Fruit: food colouring [333] Flower: tea [331] |
| Rubiaceae | Ixora coccinea L. | Ixora, iosca, Santa Rita, geranium of the jungle, llama of the forests, corralito [334] | Asia [334] | Leaf, stem [334,335] |  | Soil decontamination [336] | Alkaloids, glycosides, phenols (flavonoids, tannins), steroids, triterpenoids, saponins, and proteins (resins) Note: It has cytotoxic activity [334] | Treatment: reduce cholesterol, control blood pressure, regeneration of tissues, reduce obesity Activity: antibacterial, antiviral, antimutagenic, anti-inflammatory, antioxidant, anthelmintic, antileishmanial, anti-asthmatic, hepatoprotective [334] | Arial part: infusion |
| Rubiaceae | Palicourea marcgravii A. St.-Hil. | Crying or golden | Brazil | Flower |  | na | Alkaloids glucosides (croceaine A), triterpenes, coumarins, and phenols (phenolic acids) | Treatment: inflammation of the urinary tract Activity: antimicrobial Toxicity: It presents ictiotoxic and cytotoxic activity | Arial part: infusion |
| Rubiaceae | Warszewiczia coccinea (Vahl) Klotszch | na | Central and South America [337] | Flower |  | na | Triterpenes | Treatment: inhibitors of acetylcholinesterase [337] | na |
| Solanaceae | Capsicum annuum L. | Pepper, chilli, morron | Mediterranean region [338] | Fruit [79] |  | Soil and water decontamination (carbofuran residue and Pb) [339,340] | Carotenoids (capsaicin, capsorubin), alkaloids [338,341] | Treatment: Buruli ulcer and gastrointestinal benefits [79] Activity: anti-haemorrhoidal, antirheumatic, anti-inflammatory, and analgesic [341] | Fruit: macerated fruit tea, sweet and savoury dishes, salad, cooked seasoning [341] |
| Solanaceae | Lycianthes rantonnetii (Carrière) Bitter | Solano of blue flower, perennial dulcamara | Argentina and Paraguay [342] | Flower |  | na | Alkaloids [342] | Treatment: seborrheic dermatitis, bronchitis, cough Activity: antioxidant and anti-hepatic Toxicity: It has high toxicity [342] | Arial part: infusion |
| Solanaceae | Petunia × hybrida Vilm. | Petunia [126] | The hybrid of P. axillaris × P. integrifolia [126] | Flower [214] |  | Soil decontamination (Pb, Cu, and Zn) [343] | Phenols (phenylpropanoids, anthocyanins) [344,345,346] | Activity: antimicrobial and antifungall [214,346] | Flower: garrison [214] |
| Solanaceae | Solanum lycopersicum L. | Tomato, jitomato, gold-apple [34] | Colombia, Peru, Ecuador [214] | Fruit [347,348] |  | Soil decontamination (Cr, As, Zn, Cd, Pb, Cu, and Ni) [340,349] | Solanine (leaves and stems contain high concentrations) [214] | Treatment: cardiovascular diseases and macular degeneration [347,348] Activity: anti-carcinogenic, anti-furuncular [347,350] Toxicity: The leaves present phytodermatitis [55] | Fruit: widely used in the food industry [34] |
| Verbenaceae | Aloysia citriodora Palau | Kidron, lemon verbena, verbena de Indias, María Luisa, Verbena olorosa, Verbena grass Louise, Arabic tea [194,214] | America [214] | Leaf, stem, flower [92,214,351] |  | Soil decontamination (Cd and Ni) [352] | Terpenes (essential oil (neral, geranial, limonene, 1,8-cineole)), verbascosides and derivatives, and phenolics (flavonoids) Note: It has cytotoxic activity and allelopathic properties [351,353] | Treatment: digestive and nervous systems Activity: antioxidant, antifungal, antiasthmatic, antimicrobial, anaesthetic, neuroprotective, spasmolytic, anxiolytic, anti-colitis, antibacterial activity, antispasmodic, stomach, sedative, antipyretic [92,214,351,353,354] | Arial part: infusion [351] Dried leaf: marinated, seasoning, sauces [351,354] |
| Verbenaceae | Lantana camara L. | Lantana, Spanish flag, frutillo, supirrosa, cariaquito [214,355] | America [355] | Leaf, seed, flower [355] |  | Soil decontamination (Pb, Cr, As, Zn, Cd, Cu, Hg, Ni) [356]. | Alkaloids (lanthamine), terpenoids, phytosterols, saponins, phenols (tannins, phycobatannins), and steroids [42,72] | Treatment: fever, flu, stomach problems, asthma, and rheumatism [214,355,357] Activity: antispasmodic, anti-carcinogenic, antitumor, and antimicrobial [214,355,358] Toxicity: the leaves and fruits present gastrointestinal toxins [55] | Flower: infusion [355,358] |
| Verbenaceae | Verbena × hybrid Groenland & Rümpler | Verbena [143] | na | Flower [143] |  | na | Phenols (flavones, flavonols) | na | Flower: raw, cooked, garnished [143] |
| Viola | Viola × wittrockiana Gams | Pansy, Wesel Ice [180] | na | Flower [180] |  | Soil decontamination (As, Cd, Pb, and Se) [307] | na | Treatment: respiratory ailments, relaxation of blood vessels, and reduction of fevers and colds [12,113] Activity: anti-inflammatory [12,359] | It has a sweet flavour [12]. |
| Zingiberaceae | Renealmia alpinia (Rottb.) Maas | x’kijit, Kumpia [360,361] | Mexico [362] | Fruit [361] |  | na | na | Treatment: antiemetic, antinausea, and snake venom neutraliser [360,361,363,364] | Seed: oil food [361,365] |
| Family [18] | Species [18] | Carotenoids Concentration/Quantification Technique | Phenolics Concentration/Quantification Technique |
|---|---|---|---|
| Acanthaceae | Aphelandra squarrosa Nees | Flower: 381.3 µg/g DW, individual carotenoids (RRLC) [366] | Root: 3.5 µmol Benzoxazinoid/g FW)/(HPLC) [22] |
| Acanthaceae | Justicia aurea Schltdl. | Flower: 47.9 µg violaxanthin/g DW (RRLC) [366] | na |
| Alstroemeriaceae | Alstroemeria aurea Graham | Flower: 4.5 to 4.9 µg total carotenoids/g DW (SM) [30]; 30 µg/g DW, individual carotenoids (RRLC) [366]; 536.6 µg/g β-carotene total carotenoids (SM) [214] | Flower: 3 mg GAE/g total phenolics (SM) [214] |
| Amaranthaceae | Celosia argentea L. | Flower: 22.1 and 116.3 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] Leaf: 0.12 to 0.36 mg total carotenoids/g FW (SM) [36] | Flower: 5.01 and 6.06 g total anthocyanin/100 g FW (SM) [36], 58.4 mg GAE/g water extract, 67.6 mg GAE/g ethanol extract (SM) [39]; 13.8 and 7.7 mg total phenolics/g DW, individual phenolics (RRLC) [366] Arial part: 2.2 and 9.4 mg GAE/g DW (SM) [367], 45.2 mg GAE/g extract, and 66.7 mg QE/g extract (SM) [37] |
| Amaryllidaceae | Allium schoenoprasum L. | Flower:58.2 mg total carotenoids/kg FW (SM) [368]; 70.1 µg total carotenoids/g DW, individual carotenoids (RRLC) [366]; 423.2 µg total carotenoids/g DW [214] Root: 0.08 mg β-carotene/100 g FW, 0.65 mg total carotenoids/100 g FW (review) [40] | Flowers: 201.8 µg gallic acid/g DW, 207.3 µg coumaric acid/g DW, 887.4 µg ferulic acid/g DW, 20.3 µg rutin/g DW (HPLC) [369]; 375.8 mg total polyphenols/100 FW (SM) [368]; 9.3 mg total phenolics/g DW, individual phenolics (RRLC) [366]; 28.9 mg GAE/g DW (SM) [214] Leaf: 16.7 mg total flavonoids/g FW [42], 68.5 GAE/g (SM) [44] Root: 2.7 mg myricetin/100 g FW, 4.5 mg quercetin/100 g FW, 7.7 mg kaempferol/100 g FW, 21.0 mg GAE/100 g FW, 0.5 mg anthocyanin/100 g FW (review) [40] |
| Amaryllidaceae | Agapanthus africanus (L.) Hoffmanns | Flower: 8.1 µg total carotenoids/g DW, individual carotenoids (RRLC) [366]; 67.0 and 90.0 µg total carotenoids/g DW (SM) [214] | Flower: Identification of delphinidin, p-coumaroyl, kaempferol, and others (HPLC-MS) [370] 13.7 mg total phenolics/g DW, individual phenolics (RRLC) [366]; 23.6 and 25.7 mg GAE/g DW (SM) [214] |
| Amaryllidaceae | Clivia miniata (Lindl.) Bosse Clivia miniata var. citrina S. Watson | na | Flower: 1.8 total anthocyanin/100 mg FW (SM) [371] |
| Apiaceae | Coriandrum sativum L. | Flower: 267.6 µg total carotenoids/g DW, individual carotenoids (RRLC) [366]; 189.1 µg total carotenoids/g DW (SM) [214] Leaf: 152.8 to 169.2 mg total carotenoids/100 g DW, 38.3 to 58.6 mg β-carotene/100 g DW (before saponification), 27.1 to 47.5 mg β-carotene/100 g DW (after saponification) (SM) [372] Arial part: 2.0 g total carotenoids/kg DW, 0.6 g β-carotene/DW, 1.0 g lutein/kg DW (HPLC) [373] Seed: 1.8 to 2.2 mg total carotenoids/100 g DW, 0.3 to 0.6 mg β-carotene/100 g DW (SM) [372] | Flower: 2.4 mg total phenolics/g DW, individual phenolics (RRLC) [366]; 16.5 mg GAE/g DW (SM) [214] Seed: 12.2 GAE/g, 12.6 total flavonoids (quercetin equivalents)/g, 133.74 µg GAE/mg hydro-alcohol extract, 44.5 µg total flavonoids/mg 70% ethanol extract (SM) [42]; individual phenolics, 2.2 mg/g (HPLC-MS) [374]; individual phenolics, 129.9 mg total phenolics acids/kg DW (HPLC-MS) [58]; Arial part: individual phenolics, 6273.5 mg total phenolics/kg DW (HPLC-MS) [58]; 10.0 g total phenolics/kg DW; 0.5 g chlorogenic acid/kg DW (SM and HPLC) [373] |
| Apocynaceae | Catharanthus roseus (L.) G. Don | Flower: 3.7 µg lutein/g DW (RRLC) [366]; 163.7 and 185.1 µg total carotenoids/g DW (SM) [214] Leaf: 0.5 to 0.7 mg carotenoids/g DW (SM) [375]; 9 to 1.3 mg carotenoid/g FW, 11.9 to 32.1 mg xanthophyll/g FW (SM) [376] | Flower: 26.5 and 29.1 mg total phenolics/g DW, individual phenolics (RRLC) [366]; 67.1 and 55.5 mg GAE/g DW (SM) [214] Leaf: 05.0 to 19.0 mg phenolics/g DW (SM) [375]; 55.3 to 88.0 mg anthocyanin/g FW (SM) [376] |
| Apocynaceae | Nerium oleander L. | Flower: 1.0 to 3.4 µg lutein/g DW (RRLC) [366]; 51.2 to 67.6 µg total carotenoids/g DW (SM) [214] Leaf: 2.3 µmol carotenoids/g DW, 40.7 µmol β-carotene/g DW, 42.4 µmol lutein/g DW (SM) [377] | Flower: 53.8 µg GAE/mg ethanol extract, 34.3 µg quercetin/mg ethanol extract (SM) [73]; 14.0 to 22.0 mg total phenolics/g DW, individual phenolics (RRLC) [366]; 58.0 to 67.1 mg GAE/g DW (SM) [214] |
| Apocynaceae | Trachelospermum jasminoides (Lind.) Len. | Flower: 3.6 µg lutein/g DW (RRLC) [366]; 14.7 µg total carotenoids/g DW (SM) [214] | Flower: 13.2 mg taxifolin/g, 9.5 mg isoquercitrin/g, 7.6 mg chlorogenic acid/g, and 0.2 mg gallic acid/g (HPLC) [75] Aerial part: five compounds were isolated [74]; 4.1 mg total phenolics/g DW, individual phenolics (RRLC) [366]; 110.3 mg GAE/g DW (SM) [214] |
| Araceae | Aglaonema commutatum Schott | Flower: 78.7 µg total carotenoids/g DW, individual carotenoids (RRLC) [366]; 191.6 µg total carotenoids/g DW (SM) [214] Leaf: α and β-carotene [77] Fruit: lycopene, lycoxanthin, violaxanthin,α, β, γ, δ-carotene [77]; 100.0 µg β-carotene/g DW, 110.0 µg cryptoxanthin/g DW, 1100.0 µg lycopene/g DW (polarization microscopy) [378] Seed: lutein [77] | Flower: 12.5 mg GAE/g DW (SM) [367]; 0.6 mg total phenolics/g DW, individual phenolics (RRLC) [366]; 57.9 mg GAE/g DW (SM) [214] |
| Araceae | Anthurium andraeanum Linden ex André | Flower: 14.1 µg total carotenoids/g DW, individual carotenoids (RRLC) [366]; 61.9 µg total carotenoids/g DW (SM) [214] | Flower: Individual flavonoids (HPLC-MS) [80], individual flavonoids, 0.3 to 8.7 mg total anthocyanin/g FW (HPLC-ESI-MS) [81]; 12.0 to 26.0 mg flavonoids/g FW [379]; 7.7 mg total phenolics/g DW, individual phenolics (RRLC) [366]; 117.4 mg GAE/g DW (SM) [214] |
| Araceae | Spathiphyllum montanum (R. A. Baker) Grayum | Flower: 326.6 µg total carotenoids/g DW (SM) [214] | Flower: Phenols and flavonoids [84]; 87.3 mg GAE/g DW (SM) [214] |
| Asparagaceae | Chlorophytum comosum (Thunb.) Jacques | Flower: 93.9 µg total carotenoids/g DW, individual carotenoids (RRLC) [366]; 245.3 µg total carotenoids/g DW (SM) [214] Leaf: 0.5 to 3.0 mg total carotenoid/g FW (SM) [380] | Flower: 21.1 mg total phenolics/g DW, individual phenolics (RRLC) [366]; 42.7 mg GAE/g DW (SM) [214] Arial part: 1.4 mg total phenolic/g [90] |
| Asteraceae | Bidens andicola Kunth | na | Arial part: Phytochemical screening (SM) [96] |
| Asteraceae | Calendula officinalis L. | Flower: 48.0 to 276 mg carotenoids/100 g FW, 270.0 to 3510.0 mg carotenoids/100 g DW [99]; 57.2 µg total carotenoids/g FW (SM) [296]; 50.0 to 350.0 µg carotenoid/g DW [100]; 1.0 to 1.3 mg β-carotene/g DW [102] | Flower: 313.4 mg total polyphenol/g 2% flowers extract, 19.4 mg flavonoid and quercetin/g 2% flowers extract, 28.6 mg total polyphenols/g, 18.8 mg total flavonoids/g, 12.2 mg rutin and narcissin/g [42]; 0.7 to 5.1 mg GAE/g FW, 0.7 to 3.30 mg total flavonoids/g FW [296]; individual phenolics (HPLC-MS), 0.03 to 5.5 mg GAE/g DW [101]; 15.0 to 20.0 mg caffeic acid/g DW [100] |
| Asteraceae | Centaurea seridis L. | na | na |
| Asteraceae | Cichorium intybus L. | Flower: 8.0 to 30.2 µg lutein/g FW, 0.1 to 0.4 µg β-cryptoxanthin/g FW, 3.3 to 14.1 µg β-carotene/g FW [109]; 64.5 µg total carotenoids/g DW (SM) [214] Root: 1.1 to 2.4 mg lutein/kg, 0.2 to 0.5 mg β-carotene/kg (HPLC) [106] | Root: 12.8 to 101.5 mg quercetin/kg, 8.1 to 26.2 mg kaempferol/kg (HPLC-ESI-MS) [106] Flower: 20.0 to 130.0 mg GAE/100 g FW (SM) [109], 10.6 mg GAE/g FW (SM) [108]; 9.9 mg total phenolics/g DW, individual phenolics (RRLC) [366]; 46.0 mg GAE/g DW (SM) [214] Seed: 0.05 to 0.1 g total flavonoids/100 g DW; 0.5 to 2.5 g phenolic acids/100 g DW; 50.8 to 285.0 mg GAE/100 g DW; 43.3 to 150.0 mg total flavonoid/100 DW; sixty-four phenolic acids and flavonoids were extracted (SM, HPLC [42] |
| Asteraceae | Chrysanthemum morifolium Ramat | Flower: 0.1 to 0.2 mg carotenoid/g FW (SM) [381]; 11.8 to 165.5 µg lutein/g DW, 0.1 to 4.4 µg zeaxanthin/g DW, 0.1 to 1.9 µg β-cryptoxanthin/g DW, 0.1 to 2.9 µg 13-cis-β-carotene/g DW, 0.0 to 3.5 µg α-carotene/g DW, 1.4 to 21.9 g trans- β-carotene/g DW, 0.3 to 5.2 µg 9-cis- β-carotene/g DW (HPLC) [110] | Flower: 12.0 mg GAE/g DW (SM) [367]; individual phenols, 3732 to 12,562 mg total phenolics/g (HPLC-ESI) [111]; 0.5 to 5.5 mg total flavonoid/g FW, 0.2 to 2.5 total phenolic/g FW (SM) [381]; individual anthocyanin, 004 to 11.3 mg anthocyanin/g DW (HPLC-ESI-MS) [381] |
| Asteraceae | Coreopsis grandiflora Hogg ex Sweet | Flower: 1060 mg β-carotene/g (SM) [115]; 1060 µg total carotenoids/g DW (SM) [214] | Flower: 6.2 mg GAE/g DW (SM) [214] |
| Asteraceae | Cota tinctoria (L.) J. Gay | Flower: 0.7 mg α-carotene/g FW, 5.1 mg β-carotene/g FW (TLC) [115]; individual carotenoids, 46.9 mg carotenoid/100 g FW, 6.3 mg carotenoids/100 g DW (HPLC) [121] | Flower: 0.9 mg gallic acid/100 g DW, 26.8 mg chlorogenic acid/100 g DW, and other [118] Stem: 3.30 mg 4droxybenzoic acid/100 g DW, 1.3 mg caffeic acid/100 g DW, and other [118] Root: 0.01 mg quercetin/100 g DW and other (HPLC) [118] |
| Asteraceae | Dahlia coccinea Cav. | Flower: 2.5 to 24.0 g carotenoid/g DW (SM) [122] | Flower: 6.4 to 13.7 µg gallic acid/g DW, 0.9 to 4.7 µg caffeic acid/g DW, 2.7 to 16.4 µg chlorogenic acid/g DW, 3.7 to 5.8 µg hydroxybenzoic acid/g DW, 7.2 to 26.3 µg quercetin/g DW (HPLC), 2.0 to 125 mg gallic acid/g DW (SM) [122]; 86.6 mg GAE/g DW (SM) [214]; 15.7 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Asteraceae | Dahlia pinnata Cav. | Flowers: 2.5 to 24.0 g total carotenoids/g DW (SM) [122] | Flower: 6.4 to 13.7 µg gallic acid/g DW, 0.9 to 4.7 µg caffeic acid/g DW, 2.7 to 16.4 µg chlorogenic acid/g DW, 3.7 to 5.8 µg hydroxybenzoic acid/g DW, 7.2 to 26.3 µg quercetin/g DW (HPLC), 2.0 to 125.0 mg gallic acid/g DW (SM) [122] |
| Asteraceae | Gaillardia × grandiflora Hort. Ex Van Houtte | na | na |
| Asteraceae | Tagetes erecta L. | Flower: 2.0 to 52.0 µg violaxanthin/g DW, 10.0 to 305.0 µg zeaxanthin/g DW, 0.2 to 8.2 µg luteolin/g DW, 1.0 to 36.0 µg α-carotene/g DW, 2.0 to 53.0 µg β-carotene/g DW, 1.0 to 3.8 µg 13-cis- β-carotene/g DW (HPLC) [130]; 1.9 to 11.6 mg lutein ester/g DW (HPLC) [382] | Flower: total phenolics and flavonoids, 62.3 mg GAE/g, 97.0 mg rutin equivalent/g (HPLC-MS) [131]; 27.1 to 42.2 mg GAE/g, 20.1 to 41.9 mg quercetin equivalent/g (SM) [127]; 4.6 g gallic acid/kg FW (SM) [180] |
| Asteraceae | Taraxacum campylodes G. E. Haglund | Flower: 41.9 mg total carotenoids/kg [138]; 259.0 µg carotenoids/g DW [383] Leaf: 206.4 mg total carotenoid/kg [138] Stem: 20.5 mg total carotenoid/kg [138] | Flower: 441.4 µg gallic acid/g DW, 18.7 µg rutin/g DW, 274.9 µg resveratrol/g DW, 82.9 µg vanillic acid/g DW, 593.0 µg sinapic acid/g DW (HPLC) [369]; 11.1 mg quercetin/g DW [383]; 441.1 mg gallica acid/kg DW, 274.9 mg resveratrol/kg DW, 593.0 mg sinapic acid/kg DW [384]; 22.3 GAE/g DW [140] |
| Asteraceae | Zinnia elegans L. | Flower: 223.7 to 995.9 µg total carotenoids/g DW (SM) [214] | Flower: 87.5 to 985.0 µg total anthocyanin/g FW (SM) [385]; 22.7 to 25.7 mg GAE/g DW (SM) [214] Leaf: 2.6 mg total phenolics/g DW, 0.6 mg total flavonoids/g DW (SM) [141] |
| Balsaminaceae | Impatiens walleriana Hook. f. | Flower: 148.3 µg total carotenoids/g DW (SM) [214]; 2.9 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 6.8 mg GAE/g (SM), 96.1 mg epicatechin/100 g, 183.5 mg gallic acid/100 g, 83.9 mg protocatechuic acid/100 g [235]; 4.9 g gallic acid/kg FW (SM) [180]; 111.8 mg GAE/g DW (SM) [214]; 8.4 mg total phenolics/g DW, individual phenolics (RRLC) [366] Leaf: 3.4 µg gallic acid/g DW, 71.9 µg protocatechuic acid/g DW, 12.8 µg 4-hydroxy benzoic/g DW, 8.2 µg vanillic acid/g DW, 7.6 µg cis-p-coumaric/g DW, and other (HPLC) [386] Aerial part: individual compounds (UHPLC-MS), 12.2 mg total phenolic/g DW, 2.7 mg total phenolic acids/g DW, 3.9 mg quercetin equivalent/g DW (SM) [142] |
| Begoniaceae | Begonia cucullata Willd. | Flower: 0.03 µg total carotenoids/g FW (SM) [143] | Flower: 448.8 mg total phenolics/100 g FW (SM) [143]; 1.8 to 9.8 µg quercetin/g FW [123] |
| Begoniaceae | Begonia × tuberhybrida Voss | Flower: 0.02 µg total carotenoids/g FW (SM) [143]; 187.6 µg total carotenoids/g DW (SM) [214]; 8.7 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 100.9 mg total phenolics/100 g FW (SM) [143]; 107.2 mg GAE/g DW (SM) [214]; 8.7 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Bignoniaceae | Tecoma capensis (Thunb.) Lindl. | Flower: 238.6 µg total carotenoids/g DW (SM) [214]; 37.6 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] Leaf: 0.3 to 0.6 mg carotenoids/g FW [145] | Flower: 19.2 mg GAE/g DW (SM) [214]; 1.3 mg total phenolics/g DW, individual phenolics (RRLC) [366] Leaf: Phytochemical screening [149] |
| Bignoniaceae | Tecoma stans (L.) Juss. ex Kunth | na | Leaf: 177.0 to 216.0 mg GAE/g DW [153] Whole plant: Phytochemical screening [387] |
| Boraginaceae | Heliotropium arborescens L. | Flower: 30.7 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 1.2 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Brassicaceae | Alyssum montanum L. | Flower: 238.6 µg total carotenoids/g DW (SM) [214]; 81.3 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] Leaf: 0.14 to 0.16 mg total carotenoids/100 g FW (SM) [388] | Flower: 45.8 mg GAE/g DW (SM) [214]; 1.7 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Brassicaceae | Diplotaxis tenuifolia (L.) DC. | Flower: 257.2 µg total carotenoids/g DW (SM) [214]; 105.4 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] Leaf: individual carotenoids, 3520 and 2970 µg total carotenoid/g DW (HPLC-MS) [159]; 5.3 mg lutein/100 g, 0.7 mg violaxanthin/100 g, 0.5 mg/100 g, 0.4 neoxanthin/g (HPLC) [389] | Flower: 60.6 mg GAE/g DW (SM) [214]; 7.7 mg total phenolics/g DW, individual phenolics (RRLC) [366] Leaf: individual phenolics, 68,600 and 139,000 µg phenolics/g DW (UHPLC-Orbitrap-MS) [159]; 4.7 to 19.8 g/kg DW (HPLC-MS) [163] |
| Brassicaceae | Matthiola incana (L.) R. Br. | Flower: 200 to 1500 µg carotenoid/cm2 (SM) [390]; carotenoids identification by TLC and HPLC [168]; 64.6 µg total carotenoids/g DW (SM) [214]; 2.5 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 0.1 mg GAE/g (SM), 11.1 mg protocatechuic acid (HPLC) [235]; 0.3 to 1.9 mg GAE/g [165]; anthocyanin and other flavonoids identification by TLC and HPLC [168]; 27.0 mg GAE/g DW (SM) [214]; 5.7 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Cannabaceae | Cannabis sativa L. | Flower: 2.0 to 2.6 µg carotenoids/g FW [171]; 248.8 µg total carotenoids/g DW (SM) [214]; 19.8 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 33.8 mg GAE/g DW (SM) [214]; 2.2 mg total phenolics/g DW, individual phenolics (RRLC) [366] Seed: 0.4 to 13.9 mg GAE/100 mg [391] |
| Cannaceae | Canna indica L. | Flower: 310.0 mg total carotenoids/kg FW, 189.0 mg total xanthophylls/kg FW, 628.0 mg xanthophylls/kg DW, 1054.0 mg total carotenoids/kg DW (HPLC) [392]; 451.5 and 2453.9 µg total carotenoids/g DW (SM) [214] | Flower: 0.06 to 1.0 mg GAE/100 g, 1.8 to 19.9 mg total flavonoid/100 g [393]; 11.0 and 12.2 mg GAE/g DW (SM) [214] Seeds: 4.8 µg flavonoids/g, 13.8 µg total polyphenols/g, anthocyanin identification [42] |
| Caryophyllaceae | Dianthus caryophyllus L. | Flowers: 1.0 to 10.0 µg total carotenoids/g FW [177]; 83.1 and 75.5 µg total carotenoids/g DW (SM) [214]; 1.6 and 15.8 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] Leaf: 2.0 to 120.0 µg total carotenoid/g FW, individual carotenoids (HPLC) [177] | Flowers: 0.4 mg GAE/g (SM), 52.4 mg cyanidin-3-glucoside/100 g and 150.7 mg protocatechuic acid/100 g (HPLC) [235]; 5.3 g gallic acid/kg FW (SM) [180]; 27.4 and 48.1 mg GAE/g DW (SM) [214]; 10.9 and 15.4 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Caryophyllaceae | Dianthus chinensis L. | Flower: 261.6 µg total carotenoids/g FW (SM) [394]; 84.2 µg total carotenoids/g DW (SM) [214] Leaf: 95.0 µg total carotenoids/g DW [395]; 0.3 to 0.5 mg carotenoid/g DW [182] | Flower: 5.3 mg GAE/g (SM), 73.2 mg catechin/100 g, 110.9 mg epicatechin/100 g (HPLC) [235]; 12.3 mg GAE/g FW, 443.5 mg total anthocyanins/100 g FW [394]; 32.6 mg GAE/g DW (SM) [214]; 9.5 mg total phenolics/g DW, individual phenolics (RRLC) [366] Leaf: 19.0 mg GAE/g DW, 65.7 total flavonoids/g DW [395] |
| Caryophyllaceae | Gypsophila paniculata L. | Flower: 80.0 to 450.0 µg β-carotene/g FW, and other carotenoids [396]; 71.8 µg total carotenoids/g DW (SM) [214]; 33.8 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 44.6 mg GAE/g DW (SM) [214]; 22.2 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Caryophyllaceae | Saponaria officinalis L. | Flower: 84.0 µg total carotenoids/g DW (SM) [214]; 9.5 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] Leaf: 61.5 µg β-carotene/L [188] | Flower: 17.6 mg GAE/g DW (SM) [214]; 10.5 mg total phenolics/g DW, individual phenolics (RRLC) [366] Arial part: 6.5 µg GAE/mg [189] |
| Celastraceae | Euonymus japonicus Thunb. | Flower: 147.8 µg total carotenoids/g DW (SM) [214] | Flower: 88.9 mg GAE/g DW (SM) [214]; 1.9 mg total phenolics/g DW, individual phenolics (RRLC) [366] Leaf: Thirty-two compounds (HPLC-ESI-MS), individual phenolics (UPLC-TOF-MS) [190] |
| Convolvulaceae | Convolvulus althaeoides L. | Flower: 76.1 µg total carotenoids/g DW (SM) [214] Leaf: 2693.9 µg total carotenoid/g, 2655.3 µg total carotenoid/g individual carotenoids (LC-PDA-APCI/MS) [193] | Flower: 38.8 mg GAE/g DW (SM) [214]; 9.5 mg total phenolics/g DW, individual phenolics (RRLC) [366] Leaf: 3904.6 µg total phenolics/g, 2948.0 µg total phenolics/g, individual phenolics (LC-PDA-ESI/MS) [193] Whole plant: 30.3 to 125.4 mg total phenolics/g, 9.2 to 123.9 mg total flavonoids/g (SM) [397] |
| Convolvulaceae | Convolvulus pseudoscammonia C. Koch | na | na |
| Crassulaceae | Kalanchoe blossfeldiana Poelln. | Flower: 487.2 and 496.9 µg total carotenoids/g DW (SM) [214] | Flower: 21.0 to 392.0 µg anthocyanidin/g FW, 1.0 to 99.0 µg quercetin/g FW, individual phenolics (HPLC) [200]; 17 and 14.7 mg GAE/g DW (SM) [214] |
| Cucurbitaceae | Citrullus lanatus (Thunb.) Matsum. & Nakai | Flower: 335.4 µg total carotenoids/g DW (SM) [214] Fruit: 0.3 µg trans-lutein/g FW, 37.2 µg trans-lycopene/g DW, 1.5 µg trans-β-carotene/g DW (HPLC) [398] | Flower: 4.7 mg GAE/g DW (SM) [214] |
| Cucurbitaceae | Cucurbita maxima Duchesne | Flower: 1075.6 µg total carotenoids/g DW (SM) [214] Fruit: 15.4 µg β-carotene/g DW, 10.7 µg lutein/g DW, 20.6 µg violaxanthin/g DW, 9.8 µg neoxanthin/g DW (HPLC-MS) [205] Peel: Phytochemical screening [202] | Flower: 865.3 mg GAE/g DW (SM) [214] Peel: Phytochemical screening [202] |
| Ericaceae | Rhododendron simsii Planch. | Flower: 26.5 µg total carotenoids/g DW (SM) [214]; 79.0 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 0.6 mg GAE/g (SM), 128.4 mg catechin/100 g, 65.3 mg epicatechin/100 g (HPLC) [235], 249.8 mg GAE/g DW, 20.0 mg total flavonoid/g DW (SM) [206]; 93.0 mg GAE/g DW (SM) [214]; 1.1 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Euphorbiaceae | Euphorbia milii Des Moul. | Flower: 151.2 µg total carotenoids/g DW (SM) [214]; 7.4 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] Leaf: 0.2 to 2.9 mg total carotenoid/g FW (SM) [399] Whole plant: Phytochemical screening [211] | Flower: 104.8 mg GAE/g DW (SM) [214]; 7.4 mg total phenolics/g DW, individual phenolics (RRLC) [366] Leaf: Phytochemical screening, 3.6 mg GAE/g DW, 20.3 mg quercetin/g DW [209] Whole plant: Phytochemical screening [211] |
| Fabaceae | Brownea macrophylla Linden | Flower: 377.4 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 3.5 to 579.0 mg GAE/g DW [212] |
| Fabaceae | Lathyrus aphaca L. | Flower: 580.9 µg total carotenoids/g DW (SM) [214] | Flower: 5.7 mg GAE/g DW (SM) [214] |
| Fabaceae | Senna alexandrina Mill. | Flower: 673.5 µg total carotenoids/g DW (SM) [214] | Flower: 2.0 mg GAE/g DW (SM) [214] Whole plant: 15.9 mg gallic acid/100 g DW, 363.2 mg gentilic acid/100 g DW, 187.5 mg epigallocatechin/100 g DW, 137.7 mg kaempferol/100 g DW, and other (HPLC) [215] |
| Fabaceae | Senna corymbosa (Lam.) H. S. Irwin & Barneby | na | na |
| Fabaceae | Senna didymobotrya (Fresen.) H. S. Irwin & Barneby | na | Whole plant: Phytochemical screening [219] |
| Fabaceae | Senna papillosa (Britton & Rose) H.S. Irwin & Barneby | Flower: 2772.2 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | na |
| Fabaceae | Styphnolobium japonicum (L.) Schott | na | Leaf: 192.4 µg GAE/mg DW, individual phenolics (HPLC-PDA-ESI-MS/MS) [223] |
| Fabaceae | Trifolium alexandrinum L. | na | na |
| Geraniaceae | Pelargonium domesticum L. H. Bailey | Flower: 45.4 and 40.0 µg total carotenoids/g DW (SM) [214]; 0.7 and 2.1 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 236.0 and 195.2 mg GAE/g DW (SM) [214]; 32.7 and 37.9 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Geraniaceae | Pelargonium peltatum (L.) L’Hér. | Flower: 129.4 µg total carotenoids/g DW (SM) [214]; 3.3 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 19.4 mg GAE/g, 2.7 mg total flavonoids/g (SM), thirty-one compounds (HPLC-MS) [231]; 139.0 mg GAE/g DW (SM) [214]; 32.4 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Geraniaceae | Pelargonium × hortorum L. H. Bailey | Flower: 86.9 to 132.4 µg total carotenoids/g DW (SM) [214]; 3.5 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] Leaf: 383.4 to 528.4 µg total carotenoids/g FW (SM) [233] | Flower: 2.4 mg GAE/g (SM), 819.0 mg homogentisic acid/100 g, 693.1 mg catechin/100 g (HPLC) [235]; 131.2 to 144.5 mg GAE/g DW (SM) [214]; 39.8 to 68.9 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Goodeniaceae | Scaevola aemula R. Bronw | Flower: 208.2 µg total carotenoids/g DW (SM) [214]; 41.6 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] Leaf: 4.5 to 8.5 mg total carotenoid/g DW (SM) [238] | Flower: 60.2 mg GAE/g DW (SM) [214]; 7.3 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Hydrangeacea | Hydrangea petiolaris Siebold Zuc | Flower: 37.5 µg total carotenoids/g DW (SM) [214]; 7.3 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 99.5 mg GAE/g DW (SM) [214]; 61.4 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Iridaceae | Gladiolus communis L. | Flower: 117.7 µg total carotenoids/g DW (SM) [214]; 3.1 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 49.9 mg GAE/g DW (SM) [214]; 2.4 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Juglandaceae | Pterocarya stenoptera C. DC. | Flower: 133.8 µg total carotenoids/g DW (SM) [214]; 32.7 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 89.7 mg GAE/g DW (SM) [214]; 18.1 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Lamiaceae | Agastache foeniculum (Pursh) Kuntze | Flower: 83.7 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] Arial parts: β-carotene, lutein, zeaxanthin, violaxanthin, antheraxanthin (HPLC) [245] | Flower: 6.6 mg total phenolics/g DW, individual phenolics (RRLC) [366] Arial parts: 1.6 mg apigenin/g [245]; 13.3 mg GAE/g FW [108] |
| Lamiaceae | Lavandula angustifolia Mill. | Flower: 0.2 µg total carotenoids/g FW (SM) [143]; 0.25 mg total carotenoids/g DW (SM) [400]; 418.6 µg total carotenoids/g DW (SM) [214]; 132.3 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] Leaf: 0.3 to 0.45 mg total carotenoids/g DW (SM) [400] | Flower: 472.6 mg total phenolics/g FW (SM) [143]; 47.3 to 92.4 mg total phenolics/100 DW, 2.5 to 5.0 mg flavones/g DW, individual phenolics (HPLC) [401]; 4.0 to 6.0 mg total phenolics/g DW [400]; 29.5 mg GAE/g DW (SM) [214] Leaf: 4.0 to 5.0 mg total phenolics/g DW [400]; 5.6 mg total phenolics/g DW, individual phenolics (RRLC) [366] Whole plant: 13.3 mg GAE/g FW [108]; 1.1 to 32.7 µg total phenolics/mg [402] |
| Lamiaceae | Mentha suaveolens Ehrh. | Flower: 584.2 µg total carotenoids/g DW (SM) [214]; 149.9 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] Leaf: 6.0 mg total carotenoids/100 g (SM) [253] Stem: 7.0 to 9.0 mg total carotenoids/100 g (SM) [253] | Flower: 87.1 mg GAE/g DW (SM) [214]; 3.9 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Lamiaceae | Mentha × piperita L. | Flower: 545.5 µg total carotenoids/g DW (SM) [214]; 148.0 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] Leaf: 4.0 to 7.0 mg total carotenoids/100 g (SM) [253] Stem: 4.0 to 8.0 mg total carotenoids/100 g (SM) [253] | Flower: 109.1 mg GAE/g DW (SM) [214]; 0.5 mg total phenolics/g DW, individual phenolics (RRLC) [366] Whole plant: 19.5 mg GAE/g FW [108] |
| Lamiaceae | Rosmarinus officinalis L. | Flower: 190.7 µg total carotenoids/g DW (SM) [214]; 14.9 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] Whole plant: 259.0 µg total carotenoids/g DW (SM) [383] Leaf: 30.6 and 9.0 mg total carotenoids/L (SM) [255] | Flower: 115.8 mg GAE/g DW (SM) [214]; 7.7 mg total phenolics/g DW, individual phenolics (RRLC) [366] Whole plant: 33.2 mg GAE/g FW [108]; 1.9 mg quercetin/g DW (SM) [383] |
| Lamiaceae | Salvia leucantha Cav. | na | na |
| Lamiaceae | Salvia microphylla Kunth | Whole plant: 4.3 µg total carotenoids/g FW [258] | Whole plant: 2.4 mg GAE/g FW, 0.2 mg total anthocyanins/g FW [258] |
| Lamiaceae | Salvia splendens Sellow ex Schult. | Flowers: 0.04 µg total carotenoids/g FW [143]; 471.5 µg total carotenoids/g DW (SM) [214]; 5.5 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] Leaf: 0.2 and 0.3 mg total carotenoids/g FW (SM) [259] | Flower: 2.6 mg GAE/g (SM), 30.6 mg cyanidin-3-glucoside/100 g, 20.3 mg protocatechuic acid/100 g (HPLC) [235]; 216.2 mg total phenolics/g FW [143]; 67.9 mg GAE/g DW (SM) [214]; 7.2 mg total phenolics/g DW, individual phenolics (RRLC) [366] Leaf: 0.2 and 0.3 g total anthocyanins/g FW (SM) [259] |
| Lamiaceae | Vitex agnus-castus L. | Flower: 73.5 µg total carotenoids/g DW (SM) [214]; 7.5 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 62.9 mg GAE/g DW (SM) [214]; 28.9 mg total phenolics/g DW, individual phenolics (RRLC) [366] Whole plant: 5.0 to 32.0 mg total phenolics/g DW leaf, 2.0 to 20.0 mg total phenolics/g DW seeds, 1.0 to 3.0 mg total phenolics/g DW roots, and other phenolics [263] |
| Lythraceae | Cuphea hyssopifolia Kunth | Flower: 153.1 µg total carotenoids/g DW (SM) [214]; 157.0 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 102.3 mg GAE/g DW (SM) [214]; 11.3 mg total phenolics/g DW, individual phenolics (RRLC) [366] Arial part: individual flavonoids ((Fourier transform (FTIR)) [403] |
| Lythraceae | Lagerstroemia indica L. | Flower: 74.3 µg total carotenoids/g DW (SM) [214]; 12.9 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] Leaf: 1.2 to 1.9 mg total carotenoids/g FW (SM) [269] | Flower: 108.1 mg GAE/g DW (SM) [214]; 3.1 mg total phenolics/g DW, individual phenolics (RRLC) [366] Whole plant: Phytochemical screening [272] |
| Lythraceae | Punica granatum L. | Fruit: 0.5 to 10.1 mg total carotenoids/g FW, 0.5 to 10.2 mg β-carotene/g FW (SM) [404] Flower: 1434.4 µg total carotenoids/g DW (SM) [214]; 33.9 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 179.1 mg GAE/g DW (SM) [214]; 146.9 mg total phenolics/g DW, individual phenolics (RRLC) [366]; 336.5 mg GAE/g [405], 15.2 to 25.9 mg GAE/g DW [275] Leaf: 79.7 mg GAE/100 g DW, and others (SM) [406] Seed: 0.7 mg GAE/g (SM) [405] Fruit: 4.0 to 11.8 mg total phenolics/g DW, and others (SM) [404] |
| Magnoliaceae | Magnolia grandiflora L. | Flower: 32.6 µg total carotenoids/g DW (SM) [214]; 20.4 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: individual phenolics, 32.6 to 140.6 mg sum of phenolics/g DW (HPLC-PDA-MS/MS-ESI) [277]; 1273.9 to 4836.9 µg total flavonols/g FW, and others (HPLC-ESI-MS) [407]; 40.4 mg GAE/g DW (SM) [214]; 0.7 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Malvaceae | Ceiba speciosa (A.St.-Hil.) Ravenna | na | Whole plant: 8.7 and 9.4 mg gallic acid/g, 43.2 and 16.4 mg chlorogenic acid/g, 41.7 and 30.3 mg caffeic acid/g, 19.8 and 3.9 mg quercetin/g, 65.4 and 46.3 mg kaempferol/g [279] |
| Malvaceae | Gossypium arboreum L. | Flower: 203.3 and 206.5 µg total carotenoids/g DW (SM) [214]; 0.7 and 5.2 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 120.7 mg GAE/g DW (SM) [214]; 5.3 and 7.9 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Malvaceae | Hibiscus rosa-sinensis L. | Flower: 162.0 µg total carotenoids/g FW (SM) [408]; 448.3 and 624.3 µg total carotenoids/g DW (SM) [214] | Flower: 0.17 mg flavonoids/g, 0.09 mg total phenolics/g, 4104.0 µg flavonoids/g, 7.6 µg rutin/g, 361.9 µg quercetin/g, 50.7 µg kaempferol and myricetin/g, 61.5 mg GAE/100 g methanol extract, 59.3 mg GAE/100 g ethanol extract, 53.3 mg total flavonoids/100 g methanol extract, 32.3 mg total flavonoids/100 g ethanol extract [42]; 0.1 mg GAE/g (SM), 133.8 mg catechin/100 g (HPLC) [235]; [409]; 0.5 g gallic acid/100 g (SM) [410]; 186.2 to 281.2 mg total phenolics/100 g FW [408]; 6.2 and 8.5 mg GAE/g DW (SM) [214] |
| Lamiacea | Ocimum basilicum L. | Flower: 286.2 µg total carotenoids/g DW (SM) [214]; 88.9 and 504.6 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] Whole plant: 55.0 to 69.0 µg total carotenoids/g [411]; 51.6 and 68.3 µg total carotenoids/g FW (SM) [258] | Flower: 47.5 mg GAE/g DW (SM) [214]; 0.2 mg syringic acid/g DW, individual phenolics (RRLC) [366] Leaf: 25.7 mg GAE/g FW [108] Flower: 15.2 mg GAE/g FW [108] Whole plant: 7.2 and 8.1 mg GAE/g FW, 0.2 and 0.06 mg total anthocyanins/g FW (SM) [258] |
| Malvaceae | Hibiscus sabdariffa L. | Flower: 126.9 mg total carotenoids/100 mg Leaf: 559.4 mg total carotenoids/100 mg Seed: 232.9 and 500.7 mg total carotenoids/100 mg Fruit: 641.9 mg total carotenoids/100 mg Calyx: 507.6 mg total carotenoids/100 mg [412] | Flower: 16.53 mg anthocyanins/g, 7.4 mg phenols/g, 3.5 mg flavonoids/g, individual phenolics [42]; 6.8 to 91.9 mg GAE/g (SM) [413]; 21.1 mg GAE/g DW (SM) [414] Leaf: 312.6 mg total phenolics/100 mg Arial part: 0.9 to 4.9 mg GAE/g (SM) [284]; individual phenolics value [287] |
| Malvaceae | Hibiscus syriacus L. | Flower: 0.8 mg total carotenoids/kg FW (SM) [415]; 35.3 µg total carotenoids/g DW (SM) [214]; 3.8 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] Leaf: 112.9 mg total carotenoids/kg FW, individual carotenoids (SM) [415] | Flower: 63.4 mg rutin equivalents/g, 172.6 mg GAE/g (SM) [416]; 23.6 mg GAE/g DW (SM) [214]; 29.9 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Malvaceae | Malvaviscus arboreus Cav | Flower: 49.5 µg total carotenoids/g DW (SM) [214]; 147.4 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 0.3 mg GAE/g (SM), 38.1 mg catechin/100 g, 36.8 mg epicatechin/100 g, (HPLC) [235]; 64.7 mg GAE/g DW (SM) [214]; 14.8 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Nyctaginaceae | Bougainvillea spectabilis Willd. | Flower: 323.6 µg total carotenoids/g DW (SM) [214]; 45.5 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 6.9 mg GAE/g (SM), 79.6 mg catechin/100 g, 89.3 mg epicatechin/100 g, 39.8 mg gallic acid/100 g (HPLC) [235]; 1.7 to 2.3 mg GAE/g FW, 0.9 to 1.3 mg total flavonoids/g FW (SM) [296]; 15.8 mg total phenolics/g DW, individual phenolics (RRLC) [366] Leaf: 3.9 mg GAE/g [294]; 24.0 mg GAE/g DW (SM) [214] |
| Nyctaginaceae | Mirabilis jalapa L. | Flower: 524.1 µg total carotenoids/g DW (SM) [214]; 4.1 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 88.6 mg GAE/g DW (SM) [214]; 9.5 mg total phenolics/g DW, individual phenolics (RRLC) [366] Arial part: 4.4 mg total flavonoid/g DW (SM) [417] Whole plant: individual phenolics, 2977.4 µg total phenolics/mg flowers, 304.3 µg total phenolics/mg herb, 67.9 µg total phenolics/g fruits, 12.4 µg total phenolics/mg roots [298] |
| Oleaceae | Jasminum sambac (L.) Aiton | Flower: 80.3 µg total carotenoids/g DW (SM) [214]; 4.9 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 39.3 mg GAE/g DW (SM) [214]; 9.2 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Onagraceae | Fuchsia magellanica Lam. | Flower: 95.3 µg total carotenoids/g DW (SM) [214]; 11.8 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: individual anthocyanins [304]; 191.7 mg GAE/g DW (SM) [214]; 42.5 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Orchidaceae | Phalaenopsis aphrodite Rchb. f. | Flower: 50.3 and 257.8 µg total carotenoids/g DW (SM) [214]; 8.3 and 44.4 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 22.7 and 44.6 mg GAE/g DW (SM) [214]; 7.9 and 39.9 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Passifloraceae | Passiflora × belotii Pépin | Flower: 100.3 µg total carotenoids/g DW (SM) [214]; 49.1 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 25.2 mg GAE/g DW (SM) [214]; 6.4 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Plantaginaceae | Antirrhinum majus L. | Flower: 71.6 µg total carotenoids/g DW (SM) [214] | Flower: 10.0 mg GAE/g DW, 1.8 mg total flavonoids/g DW [307]; 74.7 mg GAE/g DW (SM) [214]; 8.9 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Plantaginaceae | Plantago major L. | Flower: 355.0 µg total carotenoids/g DW (SM) [214]; 168.2 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 76.4 mg GAE/g DW (SM) [214]; 22.8 mg total phenolics/g DW, individual phenolics (RRLC) [366] Whole plant: 1.6 mg GAE/g DW [367] |
| Plantaginaceae | Russelia equisetiformis Schltdl. & Cham. | Flower: 173.1 µg total carotenoids/g DW (SM) [214] | Flower: 31.6 mg GAE/g DW (SM) [214] Leaf: 26 phenolics (LC-MS) [418] |
| Plumbaginaceae | Limonium sinuatum (L.) Mill. | Flower: 21.41 µg total carotenoids/g DW (SM) [214]; 3.2 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 34.2 mg GAE/g DW (SM) [235]; 5.6 to 61.4 mg GAE/g DW [315]; 31.5 mg GAE/g DW (SM) [214]; 2.0 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Plumbaginaceae | Plumbago auriculata Lam. | na | Flower: 59.8 mg total phenolics/g DW, individual phenolics (RRLC) [366] Leaf: 24.3 mg GAE/g DW; 87.1 mg total flavonoid/g DW (SM) [318] |
| Polygonaceae | Fallopia aubertii (L. Henry) Holub | Flower: 19.9 µg total carotenoids/g DW (SM) [214]; 3.7 and 5.4 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] Whole plant: phytochemical screening [320] | Flower: 66.3 mg GAE/g DW (SM) [214]; 1.7 and 2.7 mg total phenolics/g DW, individual phenolics (RRLC) [366] Whole plant: phytochemical screening [320] |
| Polygonaceae | Polygala vulgaris L. | Flower: 29.5 µg total carotenoids/g DW (SM) [214]; 1.7 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 40.4 mg GAE/g DW (SM) [214]; 4.5 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Portulacaceae | Portulaca oleracea L. | Flower: 476.5 to 949.5 µg total carotenoids/g DW (SM) [214]; 632.9 and 1012.4 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] Arial part: 0.9 mg β-carotene/100 g FW, 5.5 mg total carotenoids/100 g FW [40]; 45.0 mg total carotenoids/100 g [324]; 18.9 to 36.5 mg total carotenoids/g DW [323] | Flower: 2.0 to 88.4 mg GAE/g DW (SM) [214]; 4.3 mg total phenolics/g DW, individual phenolics (RRLC) [366] Whole plant: 14.1 mg GAE/g DW (SM) [367], 0.3 mg quercetin/100 g FW (HPLC), 82.7 mg GAE/100 g FW, 0.24 mg total anthocyanin/100 g FW (SM) [40] Arial part: 5.9 mg total phenolics/g [90]; 3.2 mg total phenolics/100 g, 6.2 mg total flavonoids/100 g, and other phenolics [324]; 56.2 to 142.2 mg GAE/g DW [323] |
| Ranunculaceae | Ranunculus asiaticus L. | Flower: lutein and zeaxanthin (HPLC) [325] | Flower: Individual flavonoids, anthocyanidin and flavonol aglucone (HPLC) [325] |
| Rosaceae | Fragaria × ananassa (Duchesne ex Weston) Duchesne | Flower: 7.2 µg total carotenoids/g DW (SM) [214] | Flower: Anthocyanidins and flavonols/UHPLC-qTOF-MS [419]; 143.0 mg GAE/g DW (SM) [214]; 41.6 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Rosaceae | Rosa hybrid Vill. | Flower: 80.8 to 106.2 µg total carotenoids/g DW (SM) [214]; 33.2 and 64.2 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 2.6 mg GAE/g (SM), 880.0 mg homogentisic acid/100 g, 587.9 mg protocatechuic acid/100 g, 825.7 mg epicatechin/100 g (HPLC) [235]; quercetin and kaempferol compounds [329]; 106.3 to 153.4 mg GAE/g DW (SM) [214]; 7.8 to 19.2 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Rubiaceae | Gardenia jasminoides J. Ellis | Flower: 21.8 µg total carotenoids/g DW (SM) [214]; 6.9 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 29.6 mg GAE/g DW (SM) [214]; 1.9 mg total phenolics/g DW, individual phenolics (RRLC) [366] Leaf: 9.1 mg gallic acid/100 g DW, 141.0 mg catechin/100 g DW, 72.1 mg rutin hydrate/100 g DW, 19.1 mg quercetin/100 g DW (HPLC) [331] Whole plant: 17.3 mg GAE/g (SM) [367] |
| Rubiaceae | Ixora coccinea L. | Flower: 387.9 µg total carotenoids/g DW (SM) [214] | Flower: 9.5 mg GAE/g DW (SM) [214] Whole plant: 210.6 µg GAE/mg flower, 180.6 µg GAE/mg leaf, 100.3 µg GAE/mg stem [420] |
| Rubiaceae | Palicourea marcgravii A. St.-Hil. | na | na |
| Rubiaceae | Warszewiczia coccinea (Vahl) Klotszch | Flower: 97.2 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Whole plant: 595.0 mg GAE/100 g DW (SM) [421] |
| Solanaceae | Capsicum annuum L. | Flower: 482.6 µg total carotenoids/g DW (SM) [214]; 139.7 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 65.6 mg GAE/g DW (SM) [214]; 9.7 mg total phenolics/g DW, individual phenolics (RRLC) [366] Fruit: 1012.0 to 4135.5 µg GAE/g FW (SM) [42] |
| Solanaceae | Lycianthes rantonnetii (Carrière) Bitter | na | na |
| Solanaceae | Petunia × hybrida Vilm. | Flower: 0.1 to 35.8 µg β-carotene/g FW, 0.0 to 13.9 µg lutein/g FW, and others (HPLC) [422]; 80.8 to 213.0 µg total carotenoids/g DW (SM) [214]; 9.1 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 30.2 to 48.9 mg GAE/g DW (SM) [214]; 4.8 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Solanaceae | Solanum lycopersicum L. | Flower: 748.1 µg total carotenoids/g DW (SM) [214]; 88.1 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 47.8 mg GAE/g DW (SM) [214]; 3.4 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Verbenaceae | Aloysia citriodora Palau | Flower: 50.4 µg total carotenoids/g DW (SM) [214]; 1.7 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 39.1 mg GAE/g DW (SM) [214]; 15.6 mg total phenolics/g DW, individual phenolics (RRLC) [366] Leaf: 260.0 to 350 µg GAE/g DW, 12.0 to 13.0 µg total flavonoids/g DW (SM) [353] |
| Verbenaceae | Lantana camara L. | Flower: β-carotene [5]; 64.8 to 2056.1 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 0.1 mg GAE/g (SM), 56.1 mg catechin/100 g, 48.6 mg cyanidin-3-glucoside/100 g (HPLC) [235]; 10.9 to 22.5 mg total phenolics/g DW, individual phenolics (RRLC) [366] Leaf: 11.1 mg flavonoids/g, 917.6 mg polyphenol/100 g, 328.6 mg polyphenols/100 g [42] Stem: 3.29 mg flavonoids/100 g, 8.0 mg flavonoids/100 g [42] |
| Verbenaceae | Verbena × hybrid Groenland & Rümpler | Flower: 0.05 µg total carotenoids/g FW (SM) [143]; 13.3 to 74.2 µg total carotenoids/g DW (SM) [214]; 12.7 to 106.3 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Flower: 809.0 mg total phenolics/100 g FW (SM) [143]; 35.1 to 44.4 mg GAE/g DW (SM) [214]; 21.3 to 797.0 mg total phenolics/g DW, individual phenolics (RRLC) [366] |
| Viola | Viola × wittrockiana Gams | Flower: β-carotene, lycopene, and xanthophyll (FT-Raman) [423] | Flower: Kaempferol, quercetin dihydrate, lutein, and other anthocyanins (FT-Raman) [423]; 64.0 mg total phenolics/g extract LC-DAD-ESI/MS [424]; 15.20 mg GAE/g DW, 2.3 mg total flavonoids/g, individual phenolics DW [307]; 5.1 g GAE/kg FW [180] |
| Zingiberaceae | Renealmia alpinia (Rottb.) Maas | Flower: 3044.7 µg total carotenoids/g DW, individual carotenoids (RRLC) [366] | Fruit: 9.2 mg GAE/g DW (MS), individual anthocyanin (HPLC) [365] |
| Carotenoids | Edible Flowers |
|---|---|
| Antheraxanthin | Agapanthus africanus [366], Agastache foeniculum, Agastache rugosa [425], Calendula officinalis ‘Alice Orange’ [426], Calendula officinalis ‘Alice Yellow’, Camelia chrysantha [5], Campanula shetleri, Campanula carpatica, Capsicum annuum [366], Celosia cristata [35], Coriandrum sativum, Cuphea hyssopifolia, Diplotaxis tenuifolia, Eschscholzia californica, Fuchsia magellanica, Gazania spp. ‘Daybreak Orange’, Gazania spp. ‘Day Break Yellow’, Gentiana lutea [5], Gypsophila paniculate [366], Helianthus annuus ‘Sunrich Orange’ [5], Inula helenium [427], Lantana camara [366], Lilium spp. ‘Connecticut King’ [426], Lilium spp. ‘Montreux’, Lilium spp. ‘Saija’, Lilium tigrinum ‘Red Night’, Lily Connecticut king, Lotus japonicus [5], Malvaviscus arboreus [366], Mimulus lewisii, Minulus cupreus, Mimulus cardinalis, Minulus verbenacreus, Minulus guttatus, Minulus kelloggil, Mimulus jungermannioides [428], Momordica charantia, Narcissus poeticus ‘Florex Gold’ [5], Nicotiana glauca [5], Passiflora × belotti [366], Petunia × hybrida [429], Phalaenopsis aphrodite, Portulaca oleracea, Rosa × hybrida [366], Scaevola aemula, Solanum laxum [366], Tagetes erecta orange [430], Tagetes patula ‘Safari Tangerine’ [5], Tecoma capensis [366], Tropaeolum majus, Verbena × hybrid, Viola tricolour [5], and Viola witrockiana ‘Pansy’ [307]. |
| Astaxanthin | Adonis aestivalis, Adonis annua, Gazania rigens, Gerbera jamesonii yellow, orange and red, Hieracium aurantiacum, Hieracum pilosella, Hypochoeris radicata, transgenic Lotus japonicus, Senecio scandens, Tagetes erecta yellow and orange [430], Tagetes patula [130]. |
| Capsanthin | Asiatic hybrid Lily, Lilium spp. ‘Saija’, Lilium spp. ‘Red Night’, Tiger Lily [5]. |
| α-Carotene | Acnutea ageratum, Agastache foeniculum, Agastache rugosa [425], Bidens ferulaefolia, Calendula officinalis, ‘Alice Orange’ and ‘Alice Yellow’ [5], Chrysanthemum carinatum, Chrysanthemum segetum, Coreopsis grandiflora, Coreopsis tinctoria, Coreopsis verticillate, Cosmidium brunette, Begonia argentea, twenty-three Dendranthema grandiflorum [110], Dimorphotheca aurantiaca, Helianthus decapitatis var. Loddon Gold, Helichrysum bracteatum, Hypericum perforatum [5], Hypochoeris radicata, Lantana camara [5,366], Lavandula angustifolia [366], Layia elegans, Layia platyglossa, Momordica charantia, Osmanthus Chenghong Dangui, Osmanthus fragrans [5], Osmanthus fragrans ‘Yanhong Gui’ [431], Passiflora × belotti, Renealmia Alpinia [366], Rhododendron mole [432], Rudbeckia speciosum, Santolinas teretifolia, Santolinas viridis, Spathiphyllum montanum [366], Senecio scandens, Solidago sp., Tagetes erecta yellow and orange [430], Tagete patula [130], Taraxacum kok-saghyz, Tragopogon pratensis, Tropaeolum majus [5], Ursinia calenduliflora, Venidium decurrens, Zinnia elegans [433], Zinnia elegans ‘Dreamland Red’, and ‘Dreamland Yellow’ [433]. |
| β-Cryptoxanthin | Camelia chrysantha [426], Canna indica [5], Cosmos bipinnatus, twenty-three Dendranthema grandiflorum [110], Eschscholzia California, Gentiana lutea [5], Gerbera jamesonii yellow, orange, and red, Hemerocallis disticha [5], Hieracium aurantiacum, Ipomoea sp. [5], Ipomoea obscura, Ipomoea nil, Lantana camara [5,366], Melampodium divaricatum, Mimulus tigrinus [5], Momordica charantia, Narcissus pseudonarcissus ‘King Alfred’, Nicotiana glauca, Pyrostegia venusta, Sandersonia aurantiaca, Tabebula chrysantha [5], Tagetes erecta, Tagetes erecta yellow and orange [430], Tagetes patula [130], Tecoma capensis [214], Tropaeolum majus [5], Zinnia elegans [214], Zinnia elegans ‘Dreamland Red’, and ‘Dreamland Yellow’ [433]. |
| Β-Carotene | Adonis aestivalis, Agastache foeniculum, Agastache rugosa [425], Agastache anisate [214], Aglaonema commutatum, Allium schoenoprasum, Alyssum montanum [366], Antirrhinum majus ‘Snapdragon’ [307], Anthurium andreanum [214,366], Allamanda cathartica [5], Begonia argentea, Begonia cavaleriei [366], Borago officinalis [434], Boronia megastigma, Brassica oleraceae, Brownea macrophylla [366], nine Calendula officinalis [426], Calendula officinalis ‘Alice Orange’ [5], Calibrachoa ‘Million Bells Yellow’ [5], Camelia chrysantha [426], Campanula carpatica [366], Canna indica, Cannabis sativa, Capparis spinosa, Capsicum annuum [366], Carica papaya, Celosia argentea, Celosia cristata [366], Centaurea cyanus, Chlorophytum comossum, Coriandrum sativum [366], Cosmos bipinnatus, Multiflora chrysanthemum, Chrysanthemum morifolium [426], Crocus sativus, Cucurbita maxima, Cucumis sativus var. Anguria, Cuphea hyssopifolia [214,366], Cyclamen hederifolium, Cyclamen mirabile, Cyclamen persicum, Dahlia pinnata [366], twenty-three Dendranthema grandiflorum [110], Dianthus caryophyllus, Dimorphotheca aurantiaca, Diplotaxis tenuifolia, Drymonia affinis [366], Ethlingera elatior, Eustoma grandiflorum ‘Azuma-no-yuki’ and ‘Halley Lime’, Gazania × hybrida [5], Gentiana lutea [5], Gerbera jamesonii yellow and orange [5], Guzmania hybrid, Gypsophila paniculate [214], Heliotropium peruvianum [366], Hemerocallis disticha, Hibiscus sabdariffa [435], Hieracium aurantiacum, Hieracium pilosella, Hypochoeris radicata, Hypericum perforatum, Ipomoea sp., Ipomoea obscura [5], Ipomoea nil, Inula helenium [427], Ipomoea obscura, Lagerstroemia indica [366], Lantana camara [5,366], Lavandula angustifolia [214,366], Lilium spp., Lilium spp. ‘Conneticut King’ [426], Lilium spp. ‘Montreux’, Limonium sinuatum [366], Lycium barbarum, Lycoris radiata, Lotus japonicus, Melampodium divaricatum, Mentha × piperita [366], Mentha suaveolens [366], Mimulus cupreus, Mimulus trigrinus [5], Momordica charantia, Narcissus poeticus, ‘Scarlet Elegance’, Narcissus pseudonarcissus, ‘King Alfred’; Nelumbo lutea. ‘American Yellow’, Nicotiana glauca [5], Nicotiana tabacum [432], Ocimun basilicum [366], Osmanthus, Osmanthus fragrans [5], Osmanthus fragans ‘Yanhong Gui’ [431], Passiflora × belotti, Phalaenopsis aphrodite [214,366], Petunia × hybrida [5,366], Portulaca oleracea [366], Pyrostegia venusta [5], Punica granatum [214,366], Renealmia alpinia [366], Rhododendron mole [432], Rosa hybrida [214,436], Rosa hybrida ‘Alister Stella’, Rose ‘American Pillar’, Rose canina, Rose ‘Golden Wings’, Rose moyesii, Rose pomífera, Rose rubrijoli, Rose rubiginosa, Rose ‘Saraband’, Russelia equisetiformis [366], Saccharum edule, Sandersonia aurantiaca, Senna paillosa [366], Senecio scandens, Solanum laxum [366], white Solanum lycopersicum, Solanum rantonnetii [366], Sophora japonica [214], Sphatiphyllum montanum [366], Tagetes erecta yellow and orange [430], Tagetes patula [130], Tobebuia chrysantha, Thumbergia alata [5], and Trifolium hybridum, orange and red Tropaeolum majus [437], Tulipa sp. ‘Golden Harvest’, Verbena × hybrida [214], Viola tricolor, Viola witrockiana ‘Pansy’ [307], Vitex agnus-castus [366], Zinnia elegans, Zinnia elegans ‘Dreamland Red’, and ‘Dreamland Yellow’ [5,433]. |
| Lutein | Agapanthus africanus, Agastache anisate [366], Agastache foeniculum, Agastache rugosa [425], Aglaonema commutatum [366], Allamanda cathartica [5], Allium schoenoprasum [366], Aloysia citrodora, Alyssum montanum, Anthurium andraeanum [366], Antirrhinum majus ‘Snapdragon’ [307], Aphelandra squarrosa, Begonia argentea [366], Begonia × tuberhybrida, Boronia megastigma, Bidens ferulifolia, Bougainvillea spectabilis, Borago officinalis [434], Calendula officinalis ‘Alice Orange’ and ‘Alice Yellow’, Calibrachoa ‘Million Bells Yellow’ [5], Campanula carpatica [366], Campanula shertleri, Cannavis sativa [366], Capparis spinosa, Capsicum annuum [366], Capparis spinose, Canna indica [5], Cannabis sativa, red Catharanthus roseus, Celosia argentea, Celosia cristata [366], Centaurea cyanus, Chlorophytum comossum [366], Chrysanthemum spp., Sunny Orange Chrysanthemum, yellow Paragon Chrysanthemum morifolium [5], Cichorium intybus, Coeopsis grandiflora, Convolvulus scammonia, Coriandrum sativum [366], Cosmos bipinnatus, Cucumis sativas var. Anguria, Cuphea hyssopifolia [366], Cyclamen hederifolium, Cyclamen mirabile, Cyclamen persicum [438], twenty-three Dahlia pinnata [366], Dendranthema grandiflorum [110], Dianthus caryophyllus, Drymonia affinis [366], Drymonia sp., Escallonia rubra, Euonymus japonicus, Euphorbia milii, Eustoma, Eustoma grandiflorum, Eustoma grandiflorum ‘Azuma-no-yuki’ and ‘Halley Lime’, Fallopia aubertii, Fuchsia magellanica, Gladiolus communis, Gazania spp., ‘Day Break Yellow’ [5], Gentiana lutea [426], Gerbera jamesonii ‘Dancer’ [5], Gladiolus communis, Gossypium arboreum, Guzmania hybrid, Gypsophila paniculates [366], Helianthus annuus, Helianthus annuus ‘Sunrich Orange’ [5], Heliotropium peruvianum [366], Hemerocallis disticha [5], Hibiscus sabdariffa [435], Hibiscus syriacus, Hydrangea petiolaris, Impatiens balsamina, Impatiens walleriana, Inulina helenium [427], Ipomoea sp., Ipomoea nil, Ipomoea obscura [5], Ixora coccinea, Jasminum sambac, Kalanchoe blossfeldiana, Lagerstroemia indica [366], Lantana camara, Lavandula angustifolia [366], Lilium spp. [5], Lily connecticut, Lilium spp. ‘Cocceticut King’ [426], Lilium spp. ‘Montreux [5]‘, Lycoris radiata, Lotus japonicus, Malvaviscus arboreun, Mattiola incana, Melampodium divaricatum, Mentha × piperita, Mentha suaveolens [366], Mirabilis jalapa, Momordica charantia, Narcissus poeticus ‘Scarlet Elegance’, Narcissus pseudonarcissus ‘King Alfred’, Narcissus pseudonarcissus [5], Nerium oleander, Nelumbo lutea, ‘American Yellow’, Nicotiana glauca [5], Nicotiana tabacum [432], Ocimun basilicum [366], Osmanthus jingui, Osteospermum ecklonis ‘Jury’, Osteospermun fruticosum, Papaver rhoeas, Passiflora × belotti [366], Pelargonium × domesticum, Pelargonium × hortorum, Pelargonium peltatum, Petunia × hybrida [5,366], Phalaenopsis aphrodite [366], Pterocaya stenoptera, Petunia × hybrida [429], Plantago major, Polygala vulgaris, Punica grantum, Purtulaca oleracea [366], Pyrostegia venusta [5], Rhododendron mole [432], Rhododendron simsii, Rosa hybrida [366], Rose ‘American Pillar’, Rose ‘Golden Wings’, Rose moyesii, Rosmarinus officinalis, Rubiaceae warszewiczia, Russelia equisetiformis [366], Saintpaulia ionantha, Salvia splendens, Saponaria officinalis, Scaevola aemula, Senna alexandrina, Senna papillosa, Solanum laxum [366], Solanum lycopersicum ‘M82’ [5], Sophora japónica, Spathiphyllum montanum [366], Solanum lycopersicum, Solanum rantonnetii [366], Tabebuia chrysantha, Tagetes erecta [5], Lady Tagetes erecta, Tagetes patula [430], Tagetes patula ‘Safari Tangerine’ [5], Tecoma capensis, Trachelospermum jasminoides, Theretia peruviana, Thumbergia alata [5], Trachelospermum jasminoides, Trifolium cernnum, Trifolium hybridum, Tropaeolum majus [437], Tulipa sp. ‘Golden Harvest’ [5], Verbena × hybrida [366], Viola tricolor, Viola witrockiana ‘Pansy’ [307], Vitex agnus-costus [366], Warszewiczia coccinea, Wuonymus japonicus, Wuphorbia milii, Zantedeschia hybrid ‘Florex Gold’, Zinnia elegans [433], Zinnia elegans ‘Dreamland Coral’, Zinnia elegans ‘Dreamland Red’, and ‘Dreamland Yellow ‘ [433]. |
| Lutein epoxide | Aglaonema commutatum [366], Anthemis tinctoria, Calendula officinalis, ‘Alice Orange’ and ‘Alice Yellow’ [5], Capsicum annuum [366], Chrysanthemum spp, Chrysanthemum morifolium, Multiplora chrysanthemum, Sunny Orange Chrysanthemum, Chrysanthemum morifolium, ‘Sunny Orange’ [5], Convolvulus scammonia, Cuphea hyssopifolia [366], Gazania spp. ‘Daybreak Orange’ [5], Guzmania hybrid [366], Diplotaxis tenuifolia, Gazania spp. ‘Day Break Yellow’, Hemerocallis disticha, Hibiscus rosa-sinensis, Hibiscus syriacus, Impatiens walleriana, Inulina helenium [427], Lantana camara [366], Malvaviscus arboreus, Mentha × piperita, Mentha suaveolens [366], Narcissus poeticus ‘Scarlet Elegance’, Narcissus pseudonarcissus ‘King Alfred’, Ocimun basillicum, Phalaenopsis Aphrodite [366], Pterocaya stenoptera, Russelia equisetiformis [366], Scaevola aemula, Solanum laxum [366], Solanum lycopersicum, Solanum rantonnetii [366], Taraxacum officinalis, Tecoma capensis, and Tulipa sp. ‘Golden Harvest’. |
| Lycopene | Calendula officinalis ‘Alice Orange’ and ‘Alice Yellow’ [5], Begonia cavaleriei, Dimorphotheca aurantiaca, Gardenia jasminoide, Gazania spp. [5], Gazania rigens, Osteospermum ecklonis [5], Rose ‘American Pillar’, Rose canina, Rose moyesii, Rose pomífera, Rose rubrijoli, Rose rubiginosa, and Tagetes erecta. |
| Neoxanthin | Allamanda cathartica, Brassica napus [5], Capparis spinosa, Cosmos bipinnatus, Cyclamen hederifolium, Cyclamen mirabile, Cyclamen persicum [438], Eschscholzia California, Eustoma, Gentiana lutea [5], Gerbera jamesonii, Hemerocallis disticha [5], Hypericum perforatum, Ipomoea sp., Ipomoea nil, Ipomoea obscura, Lilium tigrinum ‘Red Night’, Lotus japonicus [5], Melampodium divaricatum, Mimulus lewisii, Mimulus cupreus, Mimulus cardianlis, Mimulus verbenaceus, Mimulus bigelovii, Mimulus bicolour, Mimulus torreyvi, Mimulus guttatus, Mimulus kelloggii, Mimulus jungermannioides [428], Nelumbo lutea ‘American Yellow’, Oncidium ‘Gower Ramsey’, Pyrostegia venusta, wild, old-gold [5], tangerine and yellow Solanum lycopersicum, Tabebuia chrysantha [5], Tagetes erecta ‘Orange Isis’, Theretia peruviana, Zinnia elegans [433], Zinnia elegans ‘Dreamland Red’, and ‘Dreamland Yellow’ [433]. |
| Phytoene | Begonia × semperflorens, Dianthus chinensis, Dimorphotheca aurantiaca, Gardenia jasminides, Gazania rigens, Guzmania hybrid [366], Hieracium aurantiacum, Impatiens balsamina, Lagerstroemia indica, Lantana camara yellow, red, and white [366], Lavandula angustifolia [366], Magnolia grandiflora, Osteospermun fruticosum, Punica granatum, Rosa × hybrida, pink and white [366], tangerine Solanum lycopersicum, Solanum rantonnetii [366], Tagetes erecta yellow and orange [430], Tagetes patula [130], Trifolium cernuum, Zinnia elegans [433], Zinnia elegans ‘Dreamland Red’, and ‘Dreamland Yellow’ [433]. |
| Violaxanthin | Agastache foeniculum, Agastache rugosa [425], Aglaonema commutatum [366], Allamanda cathartica, Allium schoenoprasum, Alyssum montanum [366], Anthemis tinctoria, Aphelandra squarrosa, Bougainvillea spectabilis, Brassica napus, Camelia chrysantha [426], yellow and orange Canna indica, Capparis spinosa, Capsicum annuum [366], Celosia cristata [35], Sunny Orange Chrysanthemum, Multiflora chrysanthemum, Chrysanthemum morifolium, ‘Sunny Orange’ [5], Cosmos bipinnatus, Cyclamen hederifolium, Cyclamen mirabile, Cyclamen persicum [438], Diplotaxis tenuifolia, Drymonia spp., Drymonia affinis [366], Eustoma grandiflorum ‘Azuma-no-yuki’ and ‘Halley Lime’, Dianthus caryophyllus, Gentiana lutea, Gazania spp. ‘Daybreak Orange’, Gazania spp. ‘Day Break Yellow’, [5] Helianthus annuus ‘Sunrich Orange’ [5], Hemerocallis disticha, Hypericum perforatum, Ipomea sp., Ipomea nil, Ipomoea obscura [5], Justicia aurea, red Lantana camara [366], Lilium spp., Lotus japonicus, yellow Lathyrus aphaca, Lilium spp. ‘Conneticut King’ [426], Lilium spp. ‘Montreux’, Melampodium divaricatum, Mentha × piperita, Mentha suaveolens [366], Mimulus lewisii, Mimulus cupreus, Minulus cardinalis, Mimulus whitneyi, Mimulus verbenaceus, Mimulus bigelovii, Mimulus bicolor, Mimulus torreyl, Mimulus guttatus, Mimulus jungermaniodes [428], Nelumbo lutea ‘American Yellow’, Nicotiana glauca [5], Ocimun basilicum [366], Omcidium ‘Gower Ramsey’, Plantago major, Pterocaya stenoptera, Pyrostegia venusta [5], Senna papillosa [366], yellow Solanum lycopersicum [5], wild, old-gold, and tangerine Rosa hybrida, ‘Star of Persia’, Scaevola aemula, Solanum lycopersicum, Solanum rantonnetii [366], Spathiphyllum montanum [366], Tabebuia chrysantha [5], Tagetes erecta ‘Orange Isis’, Gerbera jamesonii, Tagetes patula ‘Safari Tangerine’ [5], Theretia peruviana, Thumbergia alata, Tropaeolum majus, Verbena × hybrida [366], Viola tricolor, Viola wittrockiana [307], Zinnia elegans [5], Warszewiczia coccinea, Zinnia elegans ‘Dreamland Coral’, ‘Dreamland Red’, and ‘Dreamland Yellow’ [433]. |
| Zeaxanthin | Agastache foeniculum, Agastache rugosa [425], Allamanda cathartica [5], Antirrhinum majus ‘Snapdragon’ [307], Brassica oleraceae, Canna indica [5], Cosmos bipinnatus, Cucurbita maxima, Crocus sativus, Cucumis sativus var. Anguria, twenty-three Dendranthema grandiflorum [110], Dianthus caryophyllus, Eustoma [5], Gaillardia × grandiflora, Gazania spp. ‘Daybreak Orange’, Gazania spp. ‘Day Break Yellow [5]‘, Gazania × hybrida, Gentiana lutea [5], Gerbera jamesonii, Helianthus annuus, ‘Sunrich Orange’, Hemerocallis disticha [5], Hibiscus rosa-sinensis, Hypericum perforatum, Ipomoea sp. [5], Ipomoea nil, Ipomoea obscura [5], Lantana camara [366], Lycium barbarum, Lycoris radiata, Melampodium divaricatum, Momordica charantia, Narcissus poeticus ‘Scarlet Elegance’, Narcissus pseudonarcissus ‘King Alfred’ [5], Nicotiana tabacum [432], Petunia × hybrida, Pyrostegia venusta [5], Rhododendron mole [432], Rose moyesii, Rose pomífera, Sandersonia aurantiaca [5], Senecio scanders, high-pigment 3 (hp3), Solanum lycopersicum, Tabebuia chrysantha [5], Tagetes erecta [430], Tagetes patula ‘Safari Tangerine’, Theretia peruviana, Tropaeolum majus [5], Viola witrockiana ‘Pansy’ [307], Zinnia elegans [433], Zinnia elegans ‘Dreamland Coral’ [5], ‘Dreamland Red’, and ‘Dreamland Yellow’ [433]. |
| Phenolics | Edible Flowers |
|---|---|
| Chlorogenic acid | Alyssum montanum, Anthemis tinctoria, Calendula officinalis [101], Campanula portenschlagiana, Carica papaya, Ceiba speciosa [279], Celosia cristata [214], Dahlia anemome, Ethlingera elatior, Fragaria × ananassa, Gossypium arboretum [366], Helianthus annuus [456], Heliotropium peruvianum [366], Ixora parviflora, Lycoris radiata, Malus ‘Royalty’ [457], Malus micromahus ‘Makino’, Malus ‘Pink spire’, Malus ‘Sparkler’, Malus ‘Strawberry Parfait’, Nerium oleander, Pelargonium peltatum, Pterocarya stenoptera [366], Rosa hybrida ‘Scarlet’, Saccharum edule, Solanum laxum, Tecoma stans [214], Trachelospermum jasminoides [366]. |
| Caffeic acid | Calendula officinalis [101], Carica papaya, Celosia cristata [214], Convolvulus althaeoides, Ethlingera elatior, Guzmania hybrid [366], Helianthus annuus [456], Hydrangea petiolaris, Ixora javanica, Lavandula angustifolia, Lycoris radiata, Ocimun basilicum, Pelargonium × hortorum, Pelargonium peltatum [366], Portulaca oleracea [214], Saccharum edule, Salvia splendens, Trachelospermum jasminoides [366]. |
| Ferulic acid | Carica papaya, Catharanthus roseus, Dahlia anemone, Ethlingera elatior, Gardenia jasminoide [366], Helianthus annnuus [456], Ixora javanica, Lycoris radiata, Nerium oleander, Saccharum edule [366]. |
| Quercetrin | Aglaonema commutatum, Aloysia citrodora, Alstroemeria aurea, Anthurium andraeanum, Begonia argentea, Begonia cavaleriei, Begonia × semperflorens, Bougainvillea spectabilis, Cannabis sativa, Capsicum annuum, Celosia cristata, Centaurea sonchifolia, Citrullus lanatus, Coriandrum sativum, Dahlia anemone, Dianthus chinensis, Diplotasis tenuifolia, Escallonia rubra, Euonymus japonicus, Euphorbia milii, Eustoma grandiflorum, Fallopia aubertii, Fragaria ananassa, Fuchsia magellanica, Gossypium arboretum, Gypsophila paniculate, Hibiscus syriacus [366], Hypericum cardonae, Hypericum carinosum, Hypericum cuatrecasii, Hypericum garciae, Hypericum humboldtianum, Hypericum laricifolium, and Hypericum myricarifolium [458], Impatiens balsamina, Lathyrus aphaca, Limonium sinuatum, Mentha suaveolens, Nerium oleander, Pelargonium × domesticum, Pelargonium × hortorum, Petunia × hybrida, Phalaenopsis aphrodite, Plantago major, Plumbajo capensis, Pterocaya stenoptera, Rosa × hybrida [436], Rhododendrom simsii, Saponaria officinalis, Senna alexandrina, Sophora japonica, Tecoma capensis, Trifolium cernuum, Verbena × hybrida [366], Zinnia elegans [214]. |
| Quercetin | Aglaonema commutatum, Begonia argentea, Bunias orientalis, Calendula officinalis, Catharanthus roseus, Celosia cristata, Dianthus chinensis, Diplotaxis tenuifolia, Escallonia rubra [366], Eruca sativa, Eruca ssp., Hypericum cardonae, Hypericum carinosum, Hypericum cuatrecasii, Hypericum garciae, Hypericum humboldtianum, Hypericum laricifolium, Hypericum myricarifolium [458], Ixora arboreoa, Ixora parviflora, Petunia × hybrida, Plumbago capensis, Rosa hybrida [366], Spilanthes oleracea, Verbena × hybrida [214,366]. |
| Galic acid | Begonia cavaleriei, Begonia × semperflorens, Celosia cristata, Coriandrum sativum, Dahlia anemone, Dianthus chinensis, Euonymus japonicus, Euphorbia milii, Fallopia aubertii, Fuchsia magellanica, Gossypium arboreum, Heliotropium peruvianum [366], Hypericum cardonae, Hypericum carinosum, Hypericum cuatrecasii, Hypericum garciae, Hypericum humboldtianum, Hypericum laricifolium, Hypericum myricarifolium [458], Ixora finlaysoniana, Kalanchoe blossfeldiana, Limonium sinuatum, Lycoris radiata, Nerium oleander, Pelargonium × domesticum, Pelargonium × hortorum, Pelargonium peltatum, Phalaenopsis aphrodite, Pterocarya stenoptera, Portulaca oleracea, Rosa hybrida [366], and Trachelospermum jasminoides [214]. |
| Hydroxybenzoic acid | Begonia × semperflorens, Celosia cristata, Chlorophytum comosum, Coriandrum sativum, Dahlia coccinea, Gossypium arboreum, Hypericum cardonae, Hypericum carinosum, Hypericum cuatrecasii, Hypericum garciae, Hypericum humboldtianum, Hypericum laricifolium, Hypericum myricarifolium [458], Ixora coccinea, Lycoris radiata, Mirabilis jalapa [366], Portulaca oleracea [214], Scaevola aemula, Solanum lycopersicum, Solanum rantonnetti, Trachelospermum jasminoides [366]. |
| Cumaric acid | Agapanthus africanus, Agastache anisata, Allium schoenoprasum [369], Begonia cavalerier, Begonia × semperflorens, Celosia cristata, Campanula carpatica, Campanula portenschalagiana, Campanula shetleri, Canna indica, Catharanthus roseus, Cichorium intybus, Coriandrum sativum, Cucurbita maxima, Dahlia anemone, Dianthus caryophyllus, Euonymus japonicus, Euphorbia milii, Fuchsia magellanica, Gaillardia × grandiflora, Gazania × hybrida, Gladiolus communis, Heliotropium peruvianum, Jasminum sambac, Lantana camara, Lycoris radiata, Magnolia grandiflora, Mattiola incana, Nerium oleander, Osteospermun fructicosum, Passiflora × belotti, Pelargonium × domesticum, Pelargonium × hortorum, Pelargonium peltatum, Petunia × hybrida, Polygala vulgaris, Portulaca oleracea, Rosa hybrida, Russelia equisetiformis [366], Taraxacum officinale [214], Verbena × hybrida, and Vitex agnus-castus [366]. |
| Kaempferol | Purple amaranth, Antirrhinum majus, Begonia × semperflorens, Bunias orientalis, red and Orange Capuzin, Catharanthus roseus, Celosia cristata [366], Chrysanthemum spp, Chrysanthemum morifolium [111], Colvolvulus scammonia, Coreopsis grandiflora, Dahlia anemone [366], Dahlia pinnata, Eruca sativa, Eruca ssp., Hypericum cardonae, Hypericum carinosum, Hypericum cuatrecasii, Hypericum garciae, Hypericum humboldtianum, Hypericum laricifolium, Hypericum myricarifolium [458], Impatiens walleriana, Ixora coccinea, Limonium sinuatum, Malvaviscus arboreus [366], Punica granatum [406], Rosa hybrida [366], Spathiphyllum montanum, and Tagetes patula [214]. |
| Anthocyanins | Bidens ferulifolia, Capsicum annuum, Catharanthus roseus [214], twenty-three Dendranthema grandiflorum, Ixora chinensis, Lycoris radiata, Malus ‘Royalty’, Malus micromahus ‘Makino’, Malus ‘Pink spire’, Malus ‘Sparkler’, Malus ‘Strawberry Parfait’ [279], Punica granatum [406], Rosa spp. ‘Mister Lincoln’, ‘Papa Meilland’, Rosa rugosa ‘Veilchenblau’, ‘Better Times’, ‘María Callas’, ‘Queen Elizabeth’, Spilanthes oleracea, Solanum melongena, Tropaeolum majus, Tagetes erecta, JasminePrimu majus [459], Zantedeschia hybrid ‘Albomaculata’, ‘Black Magic’, ‘Florex Gold’, ‘Mango’, ‘Majestic Red’, ‘Chianti’, ‘Treasure’, ‘Pink’, and ‘Persuasion’ [460]. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Coyago-Cruz, E.; Moya, M.; Méndez, G.; Villacís, M.; Rojas-Silva, P.; Corell, M.; Mapelli-Brahm, P.; Vicario, I.M.; Meléndez-Martínez, A.J. Exploring Plants with Flowers: From Therapeutic Nutritional Benefits to Innovative Sustainable Uses. Foods 2023, 12, 4066. https://doi.org/10.3390/foods12224066
Coyago-Cruz E, Moya M, Méndez G, Villacís M, Rojas-Silva P, Corell M, Mapelli-Brahm P, Vicario IM, Meléndez-Martínez AJ. Exploring Plants with Flowers: From Therapeutic Nutritional Benefits to Innovative Sustainable Uses. Foods. 2023; 12(22):4066. https://doi.org/10.3390/foods12224066
Chicago/Turabian StyleCoyago-Cruz, Elena, Melany Moya, Gabriela Méndez, Michael Villacís, Patricio Rojas-Silva, Mireia Corell, Paula Mapelli-Brahm, Isabel M. Vicario, and Antonio J. Meléndez-Martínez. 2023. "Exploring Plants with Flowers: From Therapeutic Nutritional Benefits to Innovative Sustainable Uses" Foods 12, no. 22: 4066. https://doi.org/10.3390/foods12224066
APA StyleCoyago-Cruz, E., Moya, M., Méndez, G., Villacís, M., Rojas-Silva, P., Corell, M., Mapelli-Brahm, P., Vicario, I. M., & Meléndez-Martínez, A. J. (2023). Exploring Plants with Flowers: From Therapeutic Nutritional Benefits to Innovative Sustainable Uses. Foods, 12(22), 4066. https://doi.org/10.3390/foods12224066








