New and Rapid Analytical Method Using HPLC-MS Detection for Acrylamide Determination in Black Ripe Olives
Abstract
:1. Introduction
2. Materials and Methods
2.1. Olives
2.2. Moisture Analysis
2.3. Analysis of Acrylamide in Olive Pulp by HPLC-MS
2.4. Analysis of Acrylamide in Olive Pulp by GC-MS
2.5. Method Validation
2.6. Statistical Analysis
3. Results and Discussion
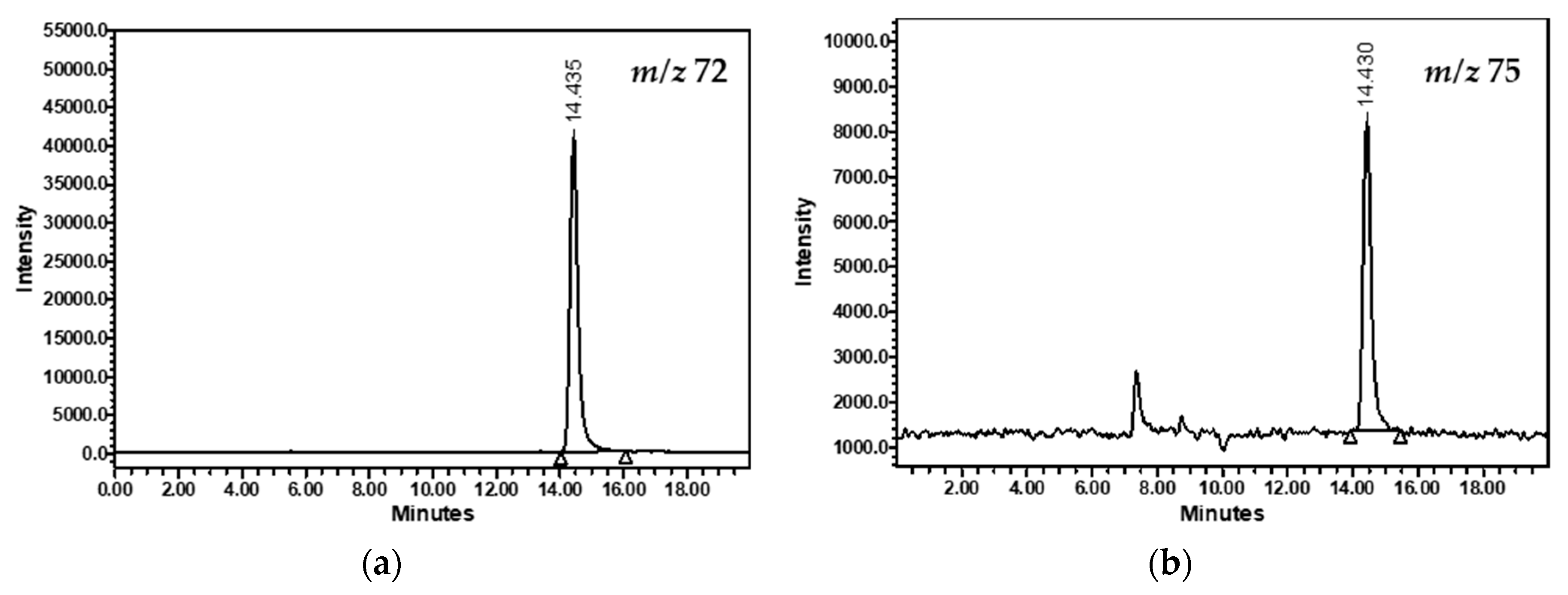
3.1. Method Optimization
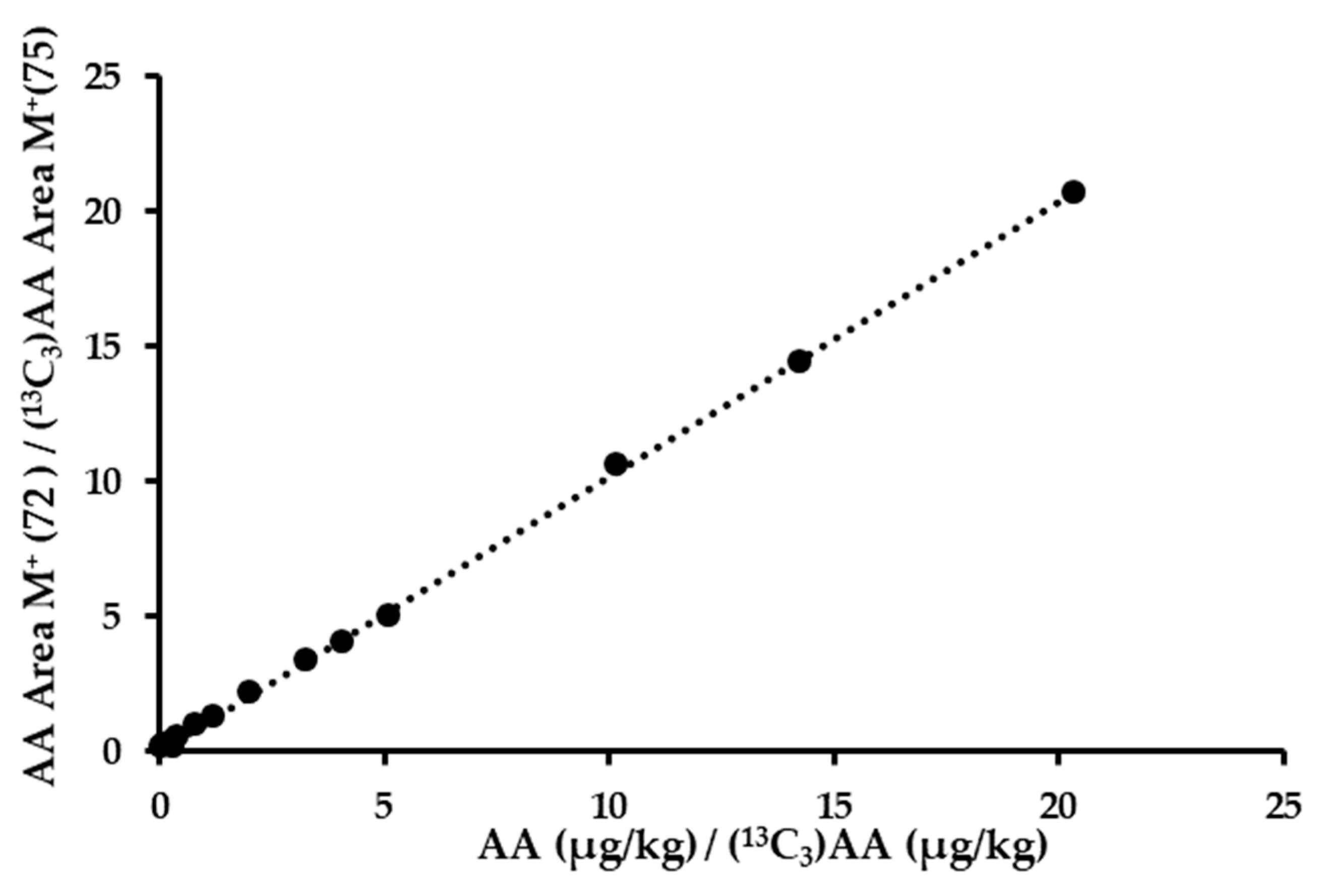
3.2. Validation of the New Method
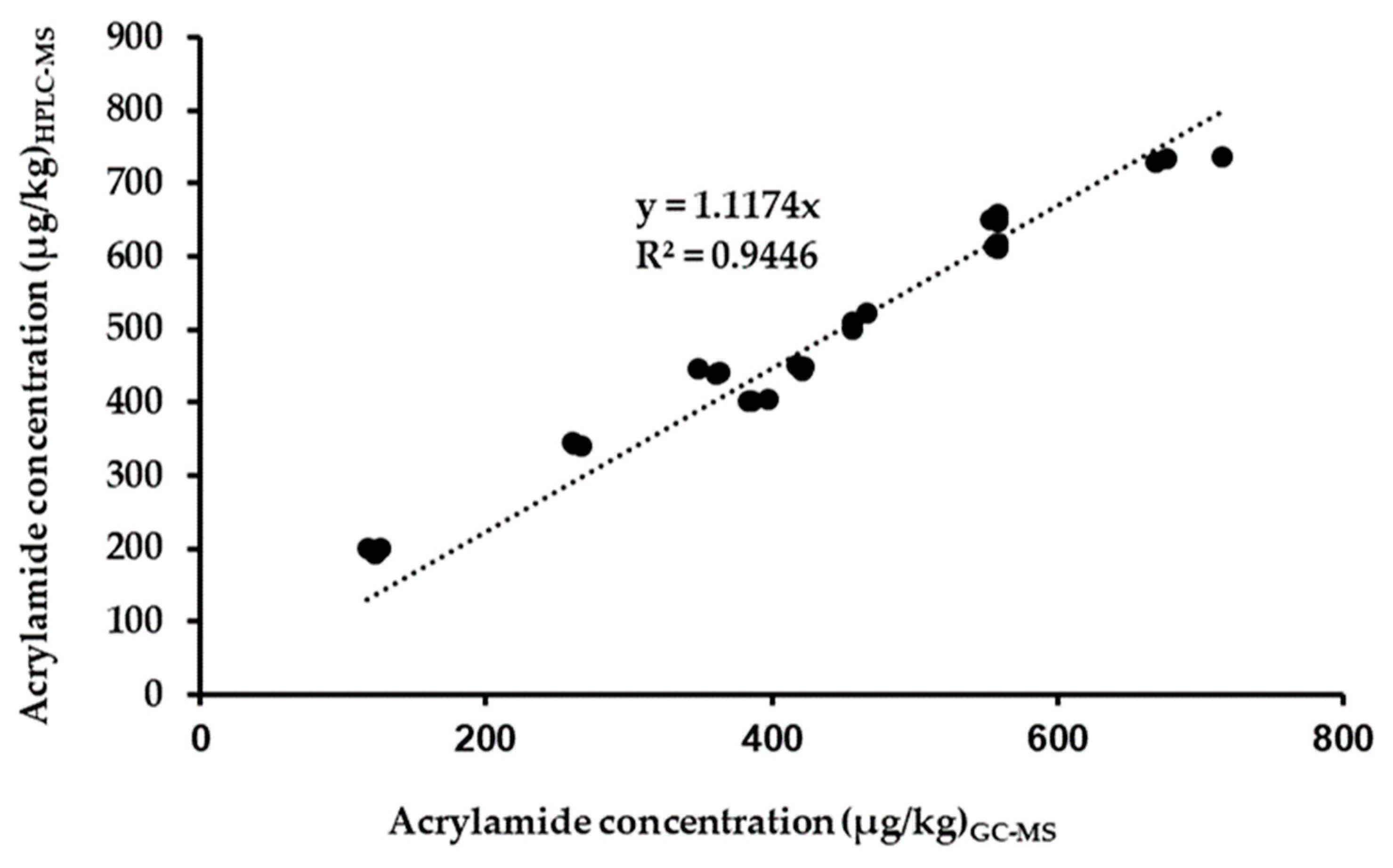
3.3. Comparison between the Proposed Method (HPLC-MS) and the Validated GC-MS
3.4. Analysis of Commercial Samples
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tareke, E.; Rydberg, P.; Karlsson, P.; Eriksson, S.; Törnqvist, M. Analysis of acrylamide, a carcinogen formed in heated foodstuffs. J. Agric. Food Chem. 2002, 50, 4998–5006. [Google Scholar] [CrossRef] [PubMed]
- Roach, J.A.G.; Andrzejewski, D.; Gay, M.L.; Nortrup, D.; Musser, S.M. Rugged LC-MS/MS survey analysis for acrylamide in foods. J. Agric. Food Chem. 2003, 51, 7547–7554. [Google Scholar] [CrossRef] [PubMed]
- EFSA (European Food Safety Authority). Scientific opinion on acrylamide in food. EFSA J. 2015, 13, 4104. [Google Scholar] [CrossRef]
- Casado, F.J.; Montaño, A. Influence of processing conditions on acrylamide content in black ripe olives. J. Agric. Food Chem. 2008, 56, 2021–2027. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.; Avena-Bustillos, R.J.; Lear, M.; Sedej, I.; Holstege, D.M.; Friedman, M.; Mchugh, T.H.; Wang, S.C. Evaluation of thermal processing variables for reducing acrylamide in canned black ripe olives. J. Food Eng. 2016, 191, 124–130. [Google Scholar] [CrossRef]
- Crawford, L.M.; Wang, S.L. Comparative study of four analytical methods for the routine determination of acrylamide in black ripe olives. J. Agric. Food Chem. 2019, 67, 12633–12641. [Google Scholar] [CrossRef]
- Martín-Vertedor, D.; Fernández, A.; Mesías, M.; Martínez, M.; Martín-Tornero, E. Identification of mitigation strategies to reduce acrylamide levels during the production of black olives. J. Food Compos. Anal. 2021, 102, 104009. [Google Scholar] [CrossRef]
- Duedahl-Olesen, K.; Wilde, A.S.; Dagnӕs-Hansen, M.P.; Mikkelsen, A.; Olesen, P.T.; Granby, K. Acrylamide in commercial table olives and the effect of domestic cooking. Food Control 2022, 132, 108515. [Google Scholar] [CrossRef]
- Guth, S.; Baum, M.; Cartus, A.; Diel, P.; Karl-Heinz, E.; Engeli, B.; Epe, B.; Grune, T.; Haller, D.; Heinz, V.; et al. Evaluation of the genotoxic potential of acrylamide: Arguments for the derivation of a tolerable daily intake (TDI value). Food Chem. Toxicol. 2023, 173, 113632. [Google Scholar] [CrossRef]
- Food and Drug Administration (FDA). Guidance for Industry: Acrylamide in Foods; FDA: College Park, MD, USA, 2016. Available online: http://www.fda.gov/FoodGuidances (accessed on 1 September 2023).
- European Commission. Commission Regulation (EU) 2017/2158 of 20 November 2017 establishing mitigation measures and benchmark levels for the reduction of the presence of acrylamide in food. Off. J. Eur. Union 2017, L304, 24–44. Available online: https://data.europa.eu/eli/reg/2017/2158/oj (accessed on 22 August 2023).
- European Commission. Commission Recommendation (EU) 2019/1888 of 7 November 2019 on the monitoring of the presence of acrylamide in certain foods. Off. J. Eur. Union 2019, L290, 31–33. Available online: https://eur-lex.europa.eu/eli/reco/2019/1888/oj (accessed on 22 August 2023).
- Sánchez-Gómez, A.H.; García-García, P.; Rejano-Navarro, L. Elaboration of table olives. Grasas Aceites 2006, 57, 86–94. [Google Scholar] [CrossRef]
- IOC (International Olive Council). Unified Qualitative Standard Applicable for Table Olives in International Trade. RES-2/91-IV/04 2004. 2004. Available online: http://www.internationaloliveoil.org (accessed on 24 August 2023).
- Amrein, T.M.; Andres, L.; Escher, F.; Amadò, R. Occurrence of acrylamide in selected foods and mitigation options. Food Addit. Contam. 2007, 24 (Suppl. S1), 13–15. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Nevado, F.; Cabrera-Bañegil, M.; Repilado, E.; Martillanes, S.; Martín-Vertedor, D. Effect of different baking treatments on the acrylamide formation and phenolic compounds in Californian-style black olives. Food Control 2018, 94, 22–29. [Google Scholar] [CrossRef]
- Martín-Vertedor, D.; Rodrigues, N.; Marx, I.M.G.; Dias, L.G.; Veloso, A.C.A.; Pereira, J.A.; Peres, A.M. Assessing acrylamide content in sterilized Californian-style black table olives using HPLC-MS-QQQ and a potentiometric electronic tongue. LWT-Food Sci. Technol. 2020, 129, 109605. [Google Scholar] [CrossRef]
- Delatour, T.; Stadler, R.H. Two decades of research in dietary acrylamide: What do we know today. Crit. Rev. Food Sci. Nutr. 2022, 19, 1–9. [Google Scholar] [CrossRef]
- Ehling, S.; Hengel, M.; Shibamoto, T. Formation of acrylamide from lipids. In Chemistry and Safety of Acrylamide in Food. Advances in Experimental Medicine and Biology; Friedman, M., Mottram, D., Eds.; Springer: Boston, MA, USA, 2005; Volume 561, pp. 223–233. [Google Scholar]
- Casado, F.J.; Montaño, A.; Carle, R. Contribution of peptides and polyphenols from olive water to acrylamide formation in sterilized table olives. LWT-Food Sci. Technol. 2014, 59, 376–382. [Google Scholar] [CrossRef]
- Charoenprasert, S.; Zweigenbaum, J.A.; Zhang, G.; Mitchell, A.E. The influence of pH and sodium hydroxide exposure time on glucosamine and acrylamide levels in California-style black ripe olives. J. Food Sci. 2017, 82, 1574–1581. [Google Scholar] [CrossRef]
- Charoenprasert, S.; Mitchell, A. Influence of California-style black ripe olive processing on the formation of acrylamide. J. Agric. Food Chem. 2014, 62, 8716–8721. [Google Scholar] [CrossRef]
- Mechi, D.; Fernández, A.; Baccouri, B.; Abaza, L.; Martín-Vertedor, D. Addition of “Chetoui” olive leaf extract to reduce acrylamide in Californian-style black olive. Food Biosci. 2022, 50, 102080. [Google Scholar] [CrossRef]
- Bermudo, E.; Moyano, E.; Puignou, L.; Galceran, M.T. Liquid chromatography coupled to tándem mass spectrometry for the analysis of acrylamide in typical Spanish products. Talanta 2008, 76, 389–394. [Google Scholar] [CrossRef] [PubMed]
- Oellig, C.; Gottstein, E.; Granvogl, M. Analysis of acrylamide in vegetable chips after derivatization with 2-mercaptobenzoic acid by liquid chromatography–mass spectrometry. Eur. Food Res. Technol. 2022, 248, 937–946. [Google Scholar] [CrossRef]
- Kumari, A.; Bhattacharya, B.; Agarwal, T.; Paul, V.; Maurya, V.K.; Chakkaravarthi, S.; Simal-Gandara, J. Method development and validation for acrylamide in potato cutlet by UPLC-MS/MS. Food Control 2023, 151, 109817. [Google Scholar] [CrossRef]
- Martín-Vertedor, D.; Fernández, A.; Mesías, M.; Martínez, M.; Díaz, M.; Martín-Tornero, E. Industrial Strategies to Reduce Acrylamide Formation in Californian-Style Green Ripe Olives. Foods 2020, 9, 1202. [Google Scholar] [CrossRef]
- Fernández, M.J.; Castro, R.; Garrido, A.; González, F.; González Cancho, F.; Nosti, M.; Heredia, A.; Mínguez, M.I.; Rejano, L.; Durán, M.C.; et al. Metodología. In Biotecnología de la Aceituna de Mesa, 1st ed.; CSIC, Ed.; CSIC: Madrid, Spain, 1985; pp. 385–453. [Google Scholar]
- Horwitz, W. Evaluation of analytical methods used for regulation of foods and drugs. Anal. Chem. 1982, 54, 67–76. [Google Scholar] [CrossRef]
- ICH Committee for Medicinal Products for Human Use. Harmonised Guideline: Analytical procedure development Q14 on 31 March 2022. Available online: https://www.ema.europa.eu/en/ich-q14-analytical-procedure-development-scientific-guideline (accessed on 5 September 2023).
- Rufián-Henares, J.A.; Morales, F.J. Determination of acrylamide in potato chips by a reversed-phase LC–MS method based on a stable isotope dilution assay. Food Chem. 2003, 97, 555–562. [Google Scholar] [CrossRef]
- Pena-Pereira, F.; Wojnowski, W.; Tobiszewski, M. AGREE-Analytical GREEnness Metric Approach and Software. Anal. Chem. 2020, 92, 10076–10082. [Google Scholar] [CrossRef]
- Hölzle, E.; Becker, L.; Oellig, C.; Granvogl, M. Heat-Introduced formation of acrylamide in table olives: Analysis of acrylamide, free asparagine, and 3-Aminopropionamide. J. Agric. Food Chem. 2023, 71, 13508–13517. [Google Scholar] [CrossRef]
- Mencin, M.; Abramovič, H.; Vidrih, R.; Schreiner, M. Acrylamide levels in food products on the Slovenian market. Food Control 2020, 114, 107267. [Google Scholar] [CrossRef]
| Statistical | Value |
|---|---|
| Slope | 1.0186 |
| Coefficient of determination (R2) | 0.9995 |
| LOD (µg/kg) | 4 |
| LOQ (µg/kg) | 11 |
| Accuracy 1 (%) | 98.4–104.8 |
| Intraday precision 2 (RSD, %) | 1.7–2.6 |
| Interday precision 2 (RSD, %) | 2.4–18.9 |
| Sample | Alternatives | Acrylamide Concentration (µg/kg) | |
|---|---|---|---|
| Value 1 | Standard Deviation | ||
| Pitted olive | A | 342 a 2 | 5 |
| A + SPE | 323 a | 12 | |
| A + Carrez + SPE | 327 a | 19 | |
| Whole olive | A | 540 a | 17 |
| A + SPE | 529 a | 31 | |
| A + Carrez + SPE | 491 b | 7 | |
| Style | Cultivar | Acrylamide Concentration (µg/kg) | ||
|---|---|---|---|---|
| Value 1 | Standard Deviation | Relative Standard Deviation | ||
| Sliced | Hojiblanca | 720 | 39 | 5 |
| 596 | 54 | 9 | ||
| 515 | 14 | 3 | ||
| 364 | 26 | 7 | ||
| 263 | 46 | 17 | ||
| Pitted | Hojiblanca | 1068 | 174 | 16 |
| 466 | 53 | 11 | ||
| 326 | 14 | 4 | ||
| Cacereña | 108 | 6 | 5 | |
| 79 | 5 | 6 | ||
| Whole | Hojiblanca | 765 | 39 | 5 |
| 589 | 33 | 6 | ||
| 450 | 15 | 3 | ||
| Cacereña | 300 | 51 | 17 | |
| 148 | 13 | 9 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brenes-Álvarez, M.; Ramírez, E.M.; Brenes, M.; García-García, P.; Medina, E.; Romero, C. New and Rapid Analytical Method Using HPLC-MS Detection for Acrylamide Determination in Black Ripe Olives. Foods 2023, 12, 4037. https://doi.org/10.3390/foods12214037
Brenes-Álvarez M, Ramírez EM, Brenes M, García-García P, Medina E, Romero C. New and Rapid Analytical Method Using HPLC-MS Detection for Acrylamide Determination in Black Ripe Olives. Foods. 2023; 12(21):4037. https://doi.org/10.3390/foods12214037
Chicago/Turabian StyleBrenes-Álvarez, Mercedes, Eva María Ramírez, Manuel Brenes, Pedro García-García, Eduardo Medina, and Concepción Romero. 2023. "New and Rapid Analytical Method Using HPLC-MS Detection for Acrylamide Determination in Black Ripe Olives" Foods 12, no. 21: 4037. https://doi.org/10.3390/foods12214037
APA StyleBrenes-Álvarez, M., Ramírez, E. M., Brenes, M., García-García, P., Medina, E., & Romero, C. (2023). New and Rapid Analytical Method Using HPLC-MS Detection for Acrylamide Determination in Black Ripe Olives. Foods, 12(21), 4037. https://doi.org/10.3390/foods12214037






