Microbiological Quality of Polish Artisanal Varietal Honeys
Abstract
:1. Introduction
2. Materials and Methods
2.1. Honey
2.2. Analyses
2.2.1. Physicochemical Measurements
2.2.2. Microbiological Tests
2.3. Statistical Analysis
3. Results
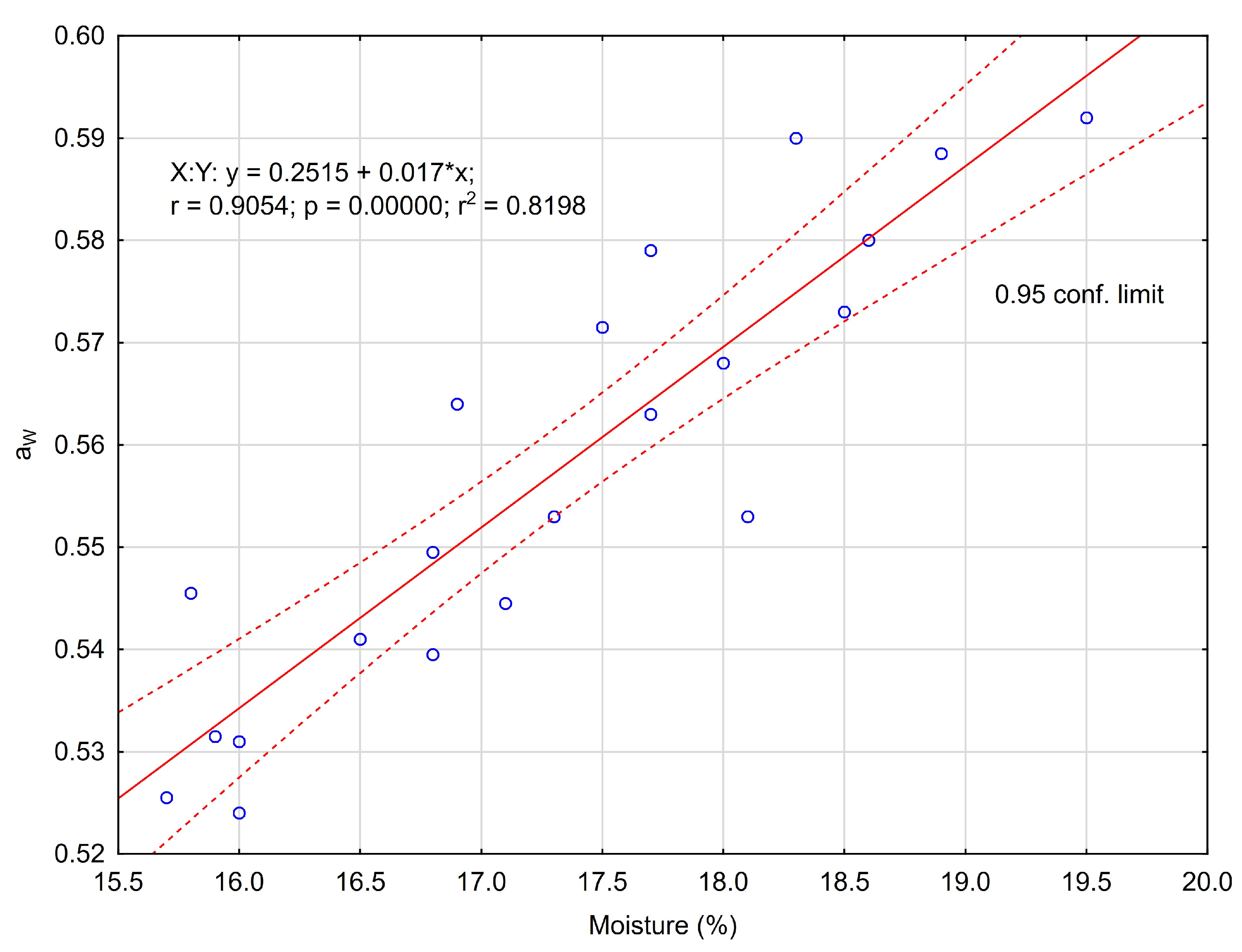
3.1. Physicochemical Properties
3.2. Microbiology Criteria
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Almasaudi, S. The antibacterial activities of honey. Saudi J. Biol. Sci. 2021, 28, 2188–2196. [Google Scholar] [CrossRef]
- Kędzia, B.; Hołderna-Kędzia, E. Contemporary opinions on the mechanism of antimicrobial action of honey. Post. Fitoter. 2017, 18, 290–297. [Google Scholar] [CrossRef]
- Kwakman, P.H.; Zaat, S.A. Antibacterial components of honey. IUBMB Life 2012, 64, 48–55. [Google Scholar] [CrossRef]
- Olaitan, P.B.; Adeleke, O.E.; Iyabo, O.O. Honey: A reservoir for microorganisms and an inhibitory agent for microbes. Afr. Health Sci. 2007, 7, 159–165. [Google Scholar] [PubMed]
- Zulkhairi Amin, F.A.; Sabri, S.; Ismail, M.; Chan, K.W.; Ismail, N.; Mohd Esa, N.; Azmi, M.L.M.; Zawawi, N. Probiotic properties of Bacillus strains isolated from stingless bee (Heterotrigona itama) honey collected across Malaysia. Int. J. Environ. Res. Public Health 2020, 17, 278. [Google Scholar] [CrossRef]
- Hossain, M.L.; Lim, L.Y.; Hammer, K.; Hettiarachchi, D.; Locher, C. Determination of Antioxidant and Antibacterial Activities of Honey-Loaded Topical Formulations: A Focus on Western Australian Honeys. Appl. Sci. 2023, 13, 7440. [Google Scholar] [CrossRef]
- López, A.C.; Alippi, A.M. Phenotypic and genotypic diversity of Bacillus cereus isolates recovered from honey. Int. J. Food Microbiol. 2007, 117, 175–184. [Google Scholar] [CrossRef]
- Rodríguez-Andrade, E.; Stchigel, A.M.; Terrab, A.; Guarro, J.; Cano-Lira, J.F. Diversity of xerotolerant and xerophilic fungi in honey. IMA Fungus 2019, 10, 20. [Google Scholar] [CrossRef]
- Brudzynski, K. Honey as an ecological reservoir of antibacterial compounds produced by antagonistic microbial interactions in plant nectars, honey and honey bee. Antibiotics 2021, 10, 551. [Google Scholar] [CrossRef]
- Rosiak, E.; Jaworska, D. Probiotic and prebiotic properties of bee honeys in terms of their quality and health safety. Żywn. Nauk. Technol. Jakość 2019, 26, 36–48. [Google Scholar] [CrossRef]
- Kačániová, M.; Kňazovická, V.; Felšöciová, S.; Rovná, K. Microscopic fungi recovered from honey and their toxinogenity. J. Environ. Sci. Health Part A 2012, 47, 1659–1664. [Google Scholar] [CrossRef] [PubMed]
- Moliné de la Paz, M.; Fernández, N.J.; Medici, S.K.; Fasce, D.; Gende, L.B. Effect of microwave treatment on microbial contamination of honeys and on their physicochemical and thermal properties. Pol. J. Food Nutr. Sci. 2015, 65, 119–126. [Google Scholar] [CrossRef]
- Agbagwa, O.E.; Otokunefor, T.V.; Frank-Peterside, N. Preliminary detection of Bacillus species in commercial honey. Br. Microbiol. Res. J. 2014, 4, 1370. [Google Scholar] [CrossRef]
- Levine, S.M.; Tang, Y.W.; Pei, Z. Recent advances in the rapid detection of Bacillus anthracis. Rev. Res. Med. Microbiol. 2005, 16, 125–133. [Google Scholar] [CrossRef]
- Pomastowski, P.; Złoch, M.; Rodzik, A.; Ligor, M.; Kostrzewa, M.; Buszewski, B. Analysis of bacteria associated with honeys of different geographical and botanical origin using two different identification approaches: MALDI-TOF MS and 16S rDNA PCR technique. PLoS ONE 2019, 14, e0217078. [Google Scholar] [CrossRef] [PubMed]
- Kędzierska-Matysek, M.; Teter, A.; Skałecki, P.; Topyła, B.; Domaradzki, P.; Poleszak, E.; Florek, M. Residues of Pesticides and Heavy Metals in Polish Varietal Honey. Foods 2022, 11, 2362. [Google Scholar] [CrossRef]
- Regulation of the Minister of Agriculture and Rural Development of 14 January 2009 on the methods of analysis related to the assessment of honey. J. Laws 2009, 17, 2018–2023.
- PN-EN ISO 4833-2:2013-12/AC:2014-04; Microbiology of the Food Chain—Horizontal Method for the Enumeration of Microorganisms—Part 2: Colony-Count at 30 Degrees C by the Surface Plating Technique. ISO: Geneva, Switzerland, 2014.
- PN-EN 15784:2009; Animal Feeding Stuffs—Isolation and Enumeration of Presumptive Bacillus spp. Available online: https://sklep.pkn.pl/pn-en-15784-2009e.html (accessed on 27 May 2023).
- PN-R-64791:1994; Feeds—Requirements and Microbiological Testing. Available online: https://sklep.pkn.pl/pn-r-64791-1994p.html (accessed on 18 June 2023).
- Adenekan, M.O.; Amusa, N.A.; Lawal, A.O.; Okpeze, V.E. Physico-chemical and microbiological properties of honey samples obtained from Ibadan. J. Microbiol. Antimicrob. 2010, 2, 100–104. Available online: https://www.academia.edu/download/33812958/Physico-chemical_and_microbiological_properties_of.pdf (accessed on 26 May 2023).
- Singh, I.; Singh, S. Honey moisture reduction and its quality. J. Food Sci. Technol. 2018, 55, 3861–3871. [Google Scholar] [CrossRef]
- EU Council Directive 2001/110/EC of 20 December 2001 relating to honey. Off. J. Eur. Union 2002, L10, 47–52. Available online: http://data.europa.eu/eli/dir/2001/110/2014-06-23 (accessed on 8 July 2023).
- Bakier, S. Characteristics of water state in some chosen types of honey found in Poland. Acta Agroph. 2006, 7, 7–15. [Google Scholar]
- Chirife, J.M.; Zamora, C.; Motto, A. The correlation between water activity and % moisture in honey. Fundamental aspects and application to Argentine honeys. J. Food Eng. 2006, 72, 287–292. [Google Scholar] [CrossRef]
- Sanz, S.; Gradillas, G.; Jiemeno, F.; Perez, C.; Juan, T. Fermentation Problem in Spanish North-Coast Honey. J. Food Prot. 1994, 58, 515–518. [Google Scholar] [CrossRef]
- Rosiak, E.; Madras-Majewska, B.; Gemba, M. Selected methods of microbiological quality assessment of lime and acacia honeys available on Polish market. Żywn. Nauk. Technol. Jakość 2020, 27, 85–95. [Google Scholar] [CrossRef]
- Kędzierska-Matysek, M.; Florek, M.; Wolanciuk, A.; Skałecki, P. Effect of freezing and room temperatures storage for 18 months on quality of raw rapeseed honey (Brassica napus). J. Food Sci. Technol. 2016, 53, 3349–3355. [Google Scholar] [CrossRef]
- Kačániová, M.; Kňazovická, W.; Melich, M.; Felšöciová, M.; Massanyi, P.; Stawarz, R.; Hascik, P.; Pechociak, T.; Kuczkowska, A.; Putała, A. Environmental concentration of selected elements and relation to physicochemical parameters in honey. J. Environ. Sci. Health Part A 2009, 44, 414–422. [Google Scholar] [CrossRef]
- Cianciosi, D.; Forbes-Hernández, T.Y.; Afrin, S.; Gasparrini, M.; Reboredo-Rodriguez, P.; Manna, P.P.; Zhang, J.; Lamas, L.B.; Flórez, S.M.; Toyos, P.A.; et al. Phenolic compounds in honey and their associated health benefits: A review. Molecules 2018, 23, 2322. [Google Scholar] [CrossRef]
- Xiong, Z.R.; Sogin, J.H.; Worobo, R.W. Microbiome analysis of raw honey reveals important factors influencing the bacterial and fungal communities. Front. Microbiol. 2023, 13, 1099522. [Google Scholar] [CrossRef]
- Mato, I.; Huidobro, J.F.; Simal-Lozano, J.; Sancho, M.T. Significance of nonaromatic organic acids in honey. J. Food Prot. 2003, 66, 2371–2376. [Google Scholar] [CrossRef]
- Molan, P.C.; Rhodes, T. Honey: A biologic wound dressing. Wounds 2015, 27, 141–151. [Google Scholar] [PubMed]
- Dobrinas, S.; Soceanu, A.; Birghila, S.; Birghila, C.; Matei, N.; Popescu, V.; Constanda, L.M. Chemical Analysis and Quality Assessment of Honey Obtained from Different Sources. Processes 2022, 10, 2554. [Google Scholar] [CrossRef]
- Hamdy, A.A.; Esawy, M.A.; Elattal, N.A.; Amin, M.A.; Ali, A.E.; Awad, G.E.A.; Connerton, I.; Mansour, N.M. Complete genome sequence and comparative analysis of two potential probiotics Bacillus subtilis isolated from honey and honeybee microbiomes. J. Genet. Eng. Biotechnol. 2020, 18, 34. [Google Scholar] [CrossRef] [PubMed]
- Ziuzia, P.; Janiec, Z.; Wróbel-Kwiatkowska, M.; Lazar, Z.; Rakicka-Pustułka, M. Honey’s Yeast—New Source of Valuable Species for Industrial Applications. Int. J. Mol. Sci. 2023, 24, 7889. [Google Scholar] [CrossRef] [PubMed]
- Snowdon, J.A.; Cliver, D.O. Microorganisms in honey. Int. J. Food Microbiol. 1996, 31, 1–26. [Google Scholar] [CrossRef]
- Grenda, T.; Kwiatek, K. Clostridium botulinum—Charakterystyka i znaczenie epidemiologiczne. Med. Weter. 2009, 65, 743–746. [Google Scholar]
- Grenda, T.; Grabczak, M.; Sieradzki, Z.; Kwiatek, K.; Pohorecka, K.; Skubida, M.; Bober, A. Clostridium botulinum spores in Polish honey samples. J. Vet. Sci. 2018, 19, 635–642. [Google Scholar] [CrossRef]
- Wojtacka, J.; Wysok, B.; Lipiński, Z.; Gomółka-Pawlicka, M.; Rybak-Chmielewska, H.; Wiszniewska-Łaszczych, A. Clostridium botulinum spores found in honey from small apiaries in Poland. J. Apic. Sci. 2016, 60, 89–100. [Google Scholar] [CrossRef]
| No | Origin Type of Honey | Moisture (%) | Extract (%) | pH | Free Acidity (mval kg−1) | aW |
|---|---|---|---|---|---|---|
| 1 | MF | 16.8 | 81.5 | 3.72 | 48.0 | 0.539 |
| 2 | MF | 15.8 | 82.4 | 3.69 | 42.0 | 0.545 |
| 3 | MF | 17.1 | 81.2 | 4.16 | 38.0 | 0.544 |
| 4 | MF | 17.7 | 80.6 | 3.93 | 34.0 | 0.563 |
| 5 | MF | 16.0 | 82.2 | 3.73 | 36.5 | 0.524 |
| 6 | MF | 17.7 | 80.6 | 3.79 | 25.0 | 0.579 |
| 7 | MF | 18.1 | 80.2 | 3.99 | 27.0 | 0.553 |
| 8 | MF | 18.3 | 80.0 | 4.01 | 16.0 | 0.590 |
| mean ± s.d. | 17.19 ±0.93 ab | 81.09 ± 0.89 ab | 3.88 ± 0.17 ab | 33.31 ± 10.23 ab | 0.555 ± 0.022 ab | |
| 9 | HD | 15.7 | 82.5 | 4.52 | 32.0 | 0.525 |
| 10 | HD | 16.5 | 81.8 | 3.93 | 41.0 | 0.541 |
| 11 | HD | 16.8 | 81.5 | 4.04 | 34.0 | 0.549 |
| mean ± s.d. | 16.33 ± 0.57 a | 81.93 ± 0.51 b | 4.16 ± 0.31 b | 35.67 ± 4.73 bc | 0.539 ± 0.012 a | |
| 12 | RS | 18.5 | 79.8 | 3.80 | 27.0 | 0.573 |
| 13 | RS | 16.9 | 81.4 | 3.68 | 19.5 | 0.564 |
| 14 | RS | 18.9 | 79.4 | 3.92 | 18.0 | 0.588 |
| mean ± s.d. | 18.10 ± 1.06 b | 80.20 ± 1.06 a | 3.80 ± 0.12 a | 21.50 ± 4.82 a | 0.575 ± 0.012 b | |
| 15 | BW | 17.5 | 80.8 | 3.87 | 48.7 | 0.571 |
| 16 | BW | 18.6 | 79.7 | 3.51 | 46.5 | 0.580 |
| 17 | BW | 17.3 | 81.0 | 3.68 | 45.0 | 0.553 |
| 18 | BW | 19.5 | 78.8 | 3.58 | 49.0 | 0.592 |
| mean ± s.d. | 18.23 ± 1.02 b | 80.08 ± 1.02 a | 3.66 ± 0.16 a | 47.30 ± 1.90 c | 0.574 ± 0.016 b | |
| 19 | LI | 16.0 | 82.2 | 4.20 | 21.5 | 0.531 |
| 20 | LI | 15.9 | 82.3 | 3.82 | 42.0 | 0.531 |
| 21 | LI | 18.0 | 80.3 | 3.91 | 35.0 | 0.568 |
| mean ± s.d. | 16.63 ± 1.18 ab | 81.60 ± 1.13 ab | 3.98 ± 0.20 ab | 32.83 ± 10.42 ab | 0.544 ± 0.021 ab | |
| Total | 17.31 ± 1.11 | 80.96 ± 1.07 | 3.88 ± 0.23 | 34.56 ± 10.58 | 0.557 ± 0.022 |
| No | Origin Type of Honey | Total Count of Fungi (cfu/g) | Presumptive Bacillus spp. (cfu/g) | Anaerobic Spore-Forming Bacteria (in 0.1 g) | Total Bacteria Count (cfu/g) |
|---|---|---|---|---|---|
| 1 | MF | <10 | <10 | n.d. | <10 |
| 2 | MF | <10 | <10 | n.d. | 3.0 × 102 |
| 3 | MF | <10 | <10 | n.d. | <10 |
| 4 | MF | <10 | <10 | n.d. | <10 |
| 5 | MF | <10 | <10 | n.d. | 5.0 × 101 |
| 6 | MF | <10 | <10 | n.d. | <10 |
| 7 | MF | <10 | <10 | n.d. | <10 |
| 8 | MF | <10 | <10 | n.d. | 5.0 × 101 |
| 9 | HD | <40 | <10 | n.d. | 1.5 × 102 |
| 10 | HD | <10 | <10 | n.d. | 2.5 × 102 |
| 11 | HD | <10 | <10 | n.d. | 5.0 × 101 |
| 12 | RS | <10 | <10 | n.d. | <10 |
| 13 | RS | <10 | <10 | n.d. | 5.0 × 101 |
| 14 | RS | <10 | <10 | n.d. | 3.5 × 102 |
| 15 | BW | 2.0 × 101 | <10 | detected | 4.5 × 102 |
| 16 | BW | <40 | <10 | n.d. | <10 |
| 17 | BW | 2.0 × 101 | <10 | n.d. | 2.5 × 102 |
| 18 | BW | <10 | <10 | n.d. | <10 |
| 19 | LI | <10 | <10 | n.d. | <10 |
| 20 | LI | <10 | <10 | n.d. | <10 |
| 21 | LI | <10 | <10 | n.d. | <10 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kędzierska-Matysek, M.; Teter, A.; Daszkiewicz, T.; Florek, M. Microbiological Quality of Polish Artisanal Varietal Honeys. Foods 2023, 12, 3349. https://doi.org/10.3390/foods12183349
Kędzierska-Matysek M, Teter A, Daszkiewicz T, Florek M. Microbiological Quality of Polish Artisanal Varietal Honeys. Foods. 2023; 12(18):3349. https://doi.org/10.3390/foods12183349
Chicago/Turabian StyleKędzierska-Matysek, Monika, Anna Teter, Tomasz Daszkiewicz, and Mariusz Florek. 2023. "Microbiological Quality of Polish Artisanal Varietal Honeys" Foods 12, no. 18: 3349. https://doi.org/10.3390/foods12183349
APA StyleKędzierska-Matysek, M., Teter, A., Daszkiewicz, T., & Florek, M. (2023). Microbiological Quality of Polish Artisanal Varietal Honeys. Foods, 12(18), 3349. https://doi.org/10.3390/foods12183349






