Edible Insects in Thailand: An Overview of Status, Properties, Processing, and Utilization in the Food Industry
Abstract
1. Introduction
2. Thai Edible Insects: Sustainable Prospects and Challenges
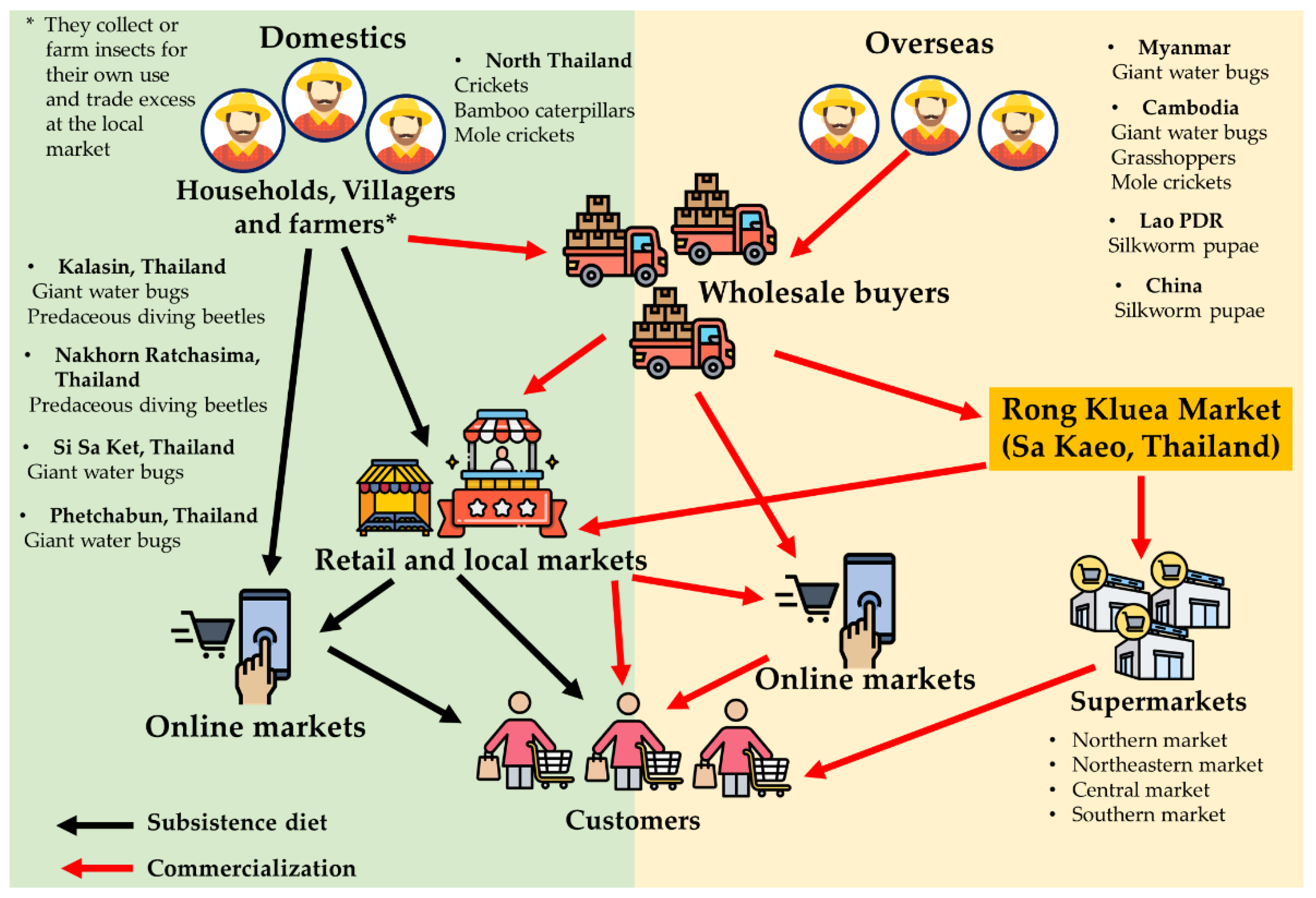
2.1. Commercial and Edible Insects in Thailand
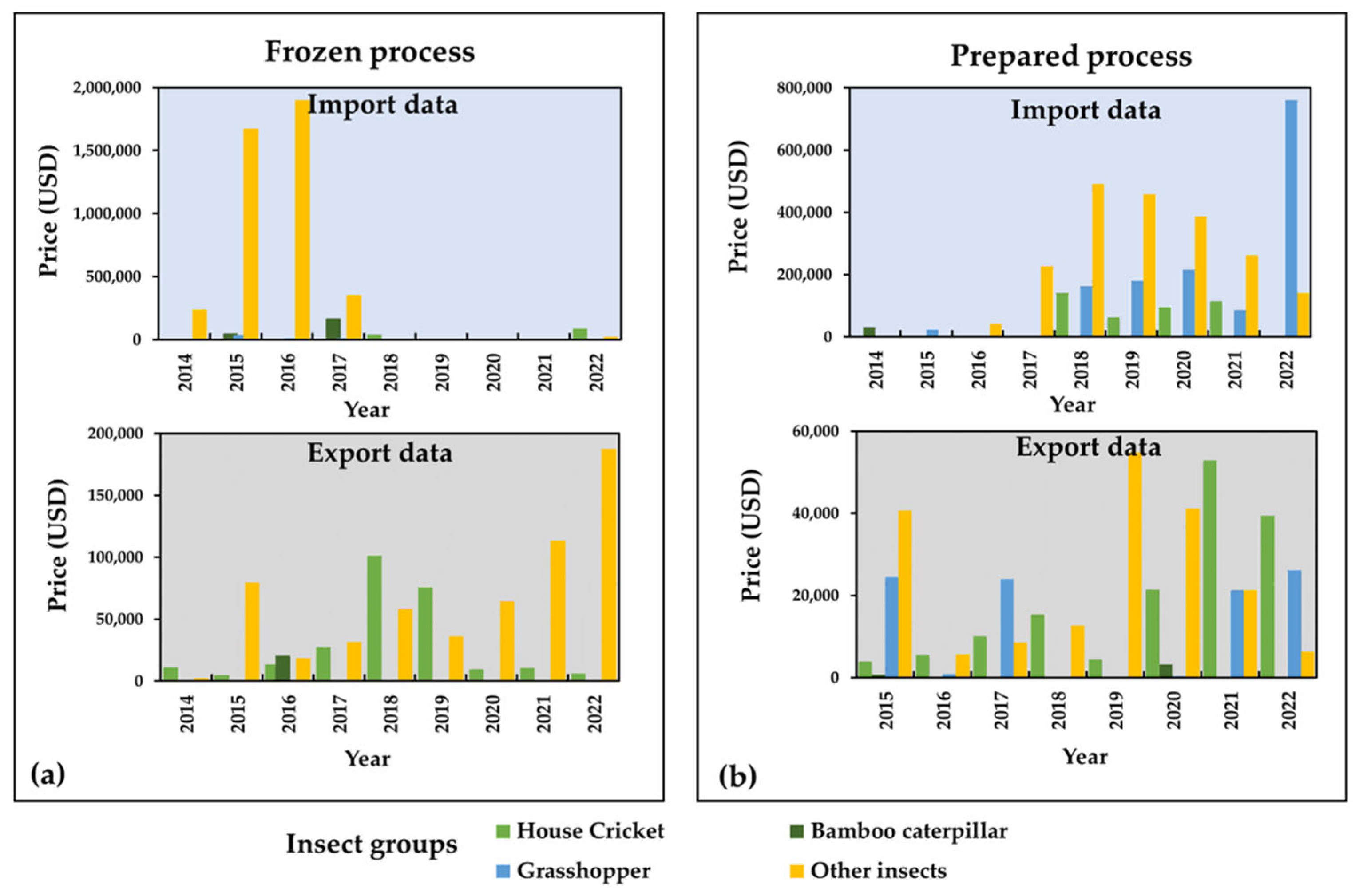
2.2. Edible Insect Market Potential and Food Industry
2.3. New Gastronomic Trends
3. Chemical Composition and Important Biological Activities
3.1. Chemical and Nutritional Composition
3.2. Biological Activities
4. Processing and Utilization in the Food Industry
4.1. Low-Temperature Processing (Refrigeration and Freezing)
4.2. Traditional Processing Techniques
4.3. Edible Insect Powder
4.4. Edible Insect Protein
4.5. Edible Insect Oil Extraction
| Edible Insect | Processing | Products | Effect of Edible Insect Powder | References |
|---|---|---|---|---|
| Cricket | Powder drying | Bread | Bread’s structure was improved, and its hardness was decreased as a result of using cricket powder. The health-promoting properties of bread products was reported. | [93] |
| Mealworms and crickets | Powder drying | Muffin | Increasing the amount of insect powder decreased the lightness. Moreover, it gave a softer texture and a decrease in the hardness, springiness, resilience, cohesiveness, and chewiness of the muffins. | [94] |
| Cricket | Powder drying | Baked chips | The baked chips revealed a higher quantity of protein, iron, and calcium when compared to the control. | [101] |
| Cricket | Powder drying | Cookies | The potential to increase protein content in cookies by using cricket powder. As the quantity of cricket powder used in the formulation increased, the organoleptic acceptability of cookies enriched with cricket powder decreased. | [102] |
| Mealworm | High hydrostatic pressure | Protein | It was discovered that large protein aggregates formed, primarily composed of hexamerin 2 and amylase. | [99] |
| Giant water bug | Solvent Extraction | Essential flavor | The majority of volatile chemicals were created by the oxidation of lipids. Compounds from the Maillard reaction are produced thermally, such as 2-acetyl-1-pyrroline and 2-acetyl-2-thiazoline. The contribution of the two most prevalent volatiles, (E)-2-hexenyl acetate and (E)-2-hexenyl butanoate, to the overall aroma of the bugs was highest. | [103] |
| Yellow mealworm, lesser mealworm, house cricket and Dubia cockroach | Aqueous oil extraction | Oil | Except for the Dubia cockroach, these bug oils contained compounds that were linked to pleasant scents. In the latter oil, several acid compounds linked to unpleasant aromas were identified. The yellow meal worm oil, lesser meal worm oil, and cricket oil all have qualities that make them suitable for use as table oils and food ingredients. | [104] |
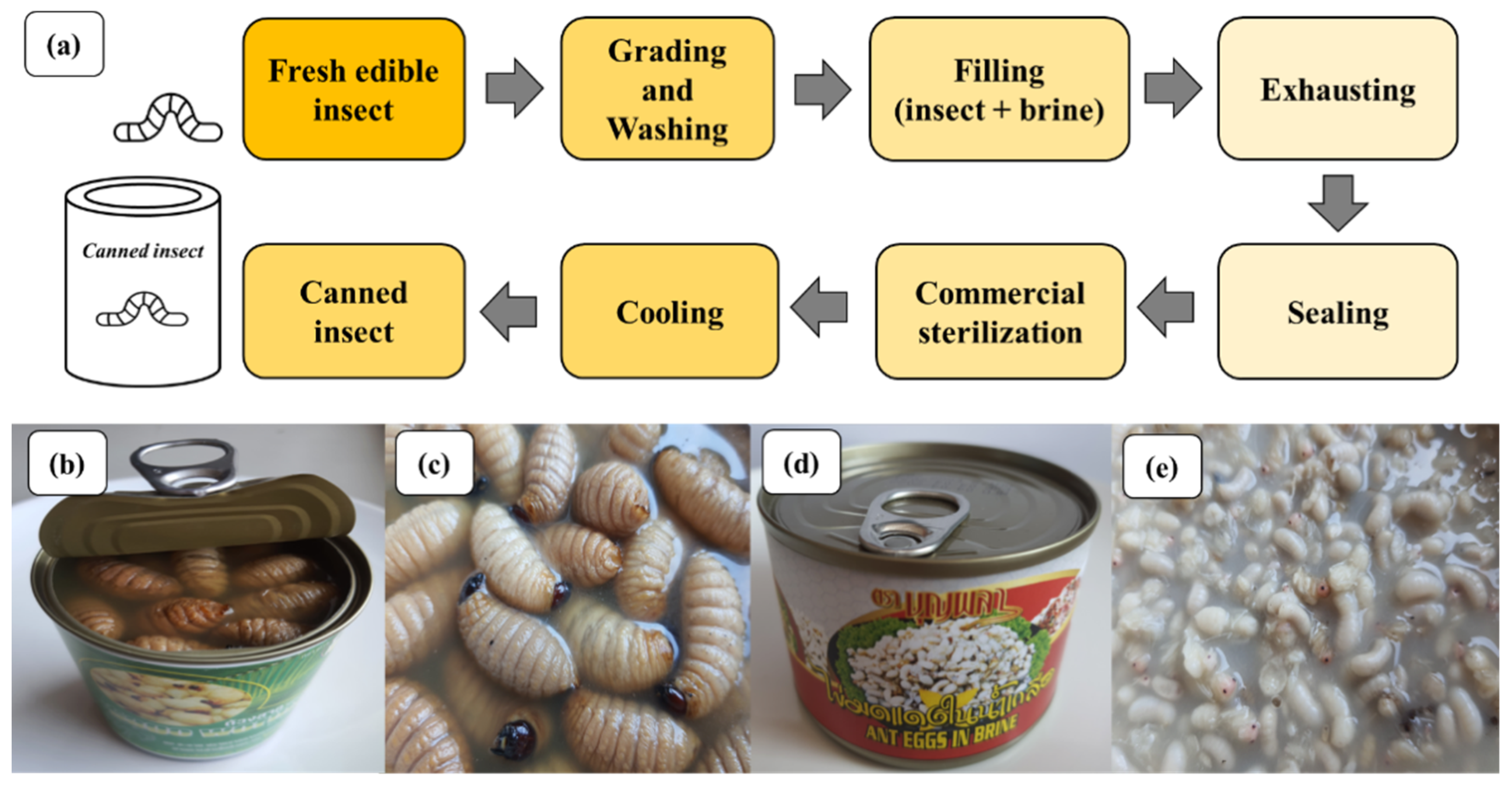
4.6. Canned Edible Insects
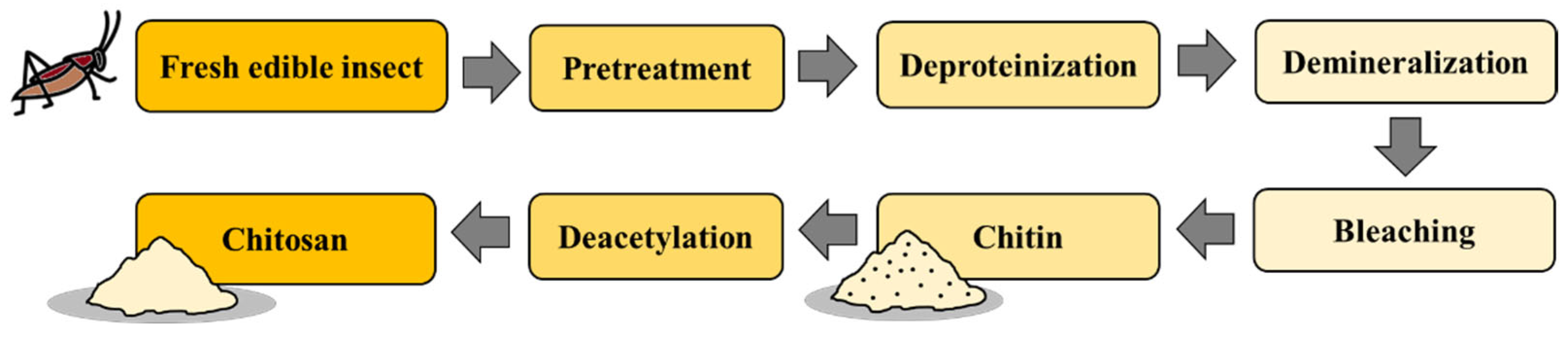
4.7. Chitin Extraction
4.8. Edible Insect Products and Other Uses
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jongema, Y. List of Edible Insect Species of the World; Wageningen University & Research: Wageningen, The Netherlands, 2017. [Google Scholar]
- Pali-Scholl, I.; Binder, R.; Moens, Y.; Polesny, F.; Monso, S. Edible insects-defining knowledge gaps in biological and ethical considerations of entomophagy. Crit. Rev. Food Sci. Nutr. 2019, 59, 2760–2771. [Google Scholar] [CrossRef] [PubMed]
- Park, S.J.; Kim, K.Y.; Baik, M.Y.; Koh, Y.H. Sericulture and the edible-insect industry can help humanity survive: Insects are more than just bugs, food, or feed. Food Sci. Biotechnol. 2022, 31, 657–668. [Google Scholar] [CrossRef] [PubMed]
- Wegener, G. ‘Let food be thy medicine, and medicine be thy food’: Hippocrates revisited. Acta Neuropsychiatr. 2014, 26, 1–3. [Google Scholar] [CrossRef] [PubMed]
- van Huis, A.; Rumpold, B.A.; van der Fels-Klerx, H.J.; Tomberlin, J.K. Advancing edible insects as food and feed in a circular economy. J. Insects Food Feed. 2021, 7, 935–948. [Google Scholar] [CrossRef]
- Dossey, A.T.; Tatum, J.T.; McGill, W.L. Chapter 5—Modern insect-based food industry: Current status, insect processing technology, and recommendations moving forward. In Insects as Sustainable Food Ingredients; Dossey, A.T., Morales-Ramos, J.A., Rojas, M.G., Eds.; Academic Press: San Diego, CA, USA, 2016; pp. 113–152. [Google Scholar]
- Rovai, D.; Michniuk, E.; Roseman, E.; Amin, S.; Lesniauskas, R.; Wilke, K.; Garza, J.; Lammert, A. Insects as a sustainable food ingredient: Identifying and classifying early adopters of edible insects based on eating behavior, familiarity, and hesitation. J. Sens. Stud. 2021, 36, e12681. [Google Scholar] [CrossRef]
- Guine, R.P.F.; Correia, P.; Coelho, C.; Costa, C.A. The role of edible insects to mitigate challenges for sustainability. Open Agric. 2021, 6, 24–36. [Google Scholar] [CrossRef]
- FAO. Thinking about the Future of Food Safety—A Foresight Report; Food and Agriculture Organization: Rome, Italy, 2022. [Google Scholar]
- Baker, M.A.; Shin, J.T.; Kim, Y.W. Customer acceptance, barriers, and preferences in the U.S. In Edible Insects in Sustainable Food Systems; Halloran, A., Flore, R., Vantomme, P., Roos, N., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 387–399. [Google Scholar]
- Kim, T.K.; Yong, H.I.; Kim, Y.B.; Kim, H.W.; Choi, Y.S. Edible insects as a protein source: A review of public perception, processing technology, and research trends. Food Sci. Anim. Resour. 2019, 39, 521–540. [Google Scholar] [CrossRef]
- Halloran, A.; Flore, R. A New world of ingredients: Aspiring chefs’ opinions on insects in gastronomy. In Edible Insects in Sustainable Food Systems; Halloran, A., Flore, R., Vantomme, P., Roos, N., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 129–137. [Google Scholar]
- Yhoung-Aree, J.; Viwatpanich, K. Edible insects in the Laos PDR, Myanmar, Thailand, and Vietnam. In Ecological Implications of Minilivestock: Potential of Insects, Rodents, Frogs and Snails; CRC Press: Boca Raton, FL, USA, 2005; pp. 415–440. [Google Scholar]
- Tagawa, K.; Hosoya, T.; Hyakumura, K.; Suzuki, D.; Yoshizawa, S.; Praxaysombath, B. The effects of season, geography, and urbanization on the diversity of edible insects at food markets in Laos. PLoS ONE 2022, 17, e0267307. [Google Scholar] [CrossRef]
- Sirimungkararat, S.; Saksirirat, W.; Nopparat, T.; Natongkham, A. Edible products from eri and mulberry silkworms in Thailand. Forest insects as food: Humans bite back. RAP Publ. 2010, 1, 189–231. [Google Scholar]
- Hanboonsong, Y.; Tasanee, J.; Patrick, B.D. Six-Legged Livestock: Edible Insect Farming, Collection and Marketing in Thailand; Food and Agriculture Organization of the United Nations: Bangkok, Thailand, 2013. [Google Scholar]
- Yhoung-Aree, J.; Puwastien, P.; Attig, G.A. Edible insects in Thailand: An unconventional protein source? Ecol. Food Nutr. 1997, 36, 133–149. [Google Scholar] [CrossRef]
- Köhler, R.; Kariuki, L.; Lambert, C.; Biesalski, H.K. Protein, amino acid and mineral composition of some edible insects from Thailand. J. Asia Pac. Entomol. 2019, 22, 372–378. [Google Scholar] [CrossRef]
- Durst, P.B.; Hanboonsong, Y. Small-scale production of edible insects for enhanced food security and rural livelihoods: Experience from Thailand and Lao People’s Democratic Republic. J. Insects Food Feed. 2015, 1, 25–31. [Google Scholar] [CrossRef]
- DeFoliart, G.R. Insects as food: Why the western attitude is important. Annu. Rev. Entomol. 1999, 44, 21–50. [Google Scholar] [CrossRef] [PubMed]
- Raheem, D.; Carrascosa, C.; Oluwole, O.B.; Nieuwland, M.; Saraiva, A.; Millán, R.; Raposo, A. Traditional consumption of and rearing edible insects in Africa, Asia and Europe. Crit. Rev. Food Sci. Nutr. 2019, 59, 2169–2188. [Google Scholar] [CrossRef]
- Hanboonsong, Y. Edible insects and associated food habits in Thailand. In Forest Insects as Food: Humans bite Back; Food and Agriculture Organization: Bangkok, Thailand, 2010; pp. 1173–1182. [Google Scholar]
- Jintapitak, M.; Ansari, M.A.; Kamyod, C.; Singkhamfu, W.; Kamthe, N.S.; Temdee, P. Blockchain eco-system for Thai insect industry: A smart contract conceptual framework. In Proceedings of the 2019 22nd International Symposium on Wireless Personal Multimedia Communications (WPMC), Lisbon, Portugal, 24–27 November 2019; pp. 1–4. [Google Scholar]
- Van Huis, A. Edible insects are the future? Proc. Nutr. Soc. 2016, 75, 294–305. [Google Scholar] [CrossRef] [PubMed]
- Chantawannakul, P. From entomophagy to entomotherapy. Front. Biosci. Landmark 2020, 25, 179–200. [Google Scholar] [CrossRef]
- Halloran, A.; Roos, N.; Flore, R.; Hanboonsong, Y. The development of the edible cricket industry in Thailand. J. Insects Food Feed. 2016, 2, 91–100. [Google Scholar] [CrossRef]
- Dobermann, D.; Swift, J.A.; Field, L.M. Opportunities and hurdles of edible insects for food and feed. Nutr. Bull. 2017, 42, 293–308. [Google Scholar] [CrossRef]
- Raheem, D.; Raposo, A.; Oluwole, O.B.; Nieuwland, M.; Saraiva, A.; Carrascosa, C. Entomophagy: Nutritional, ecological, safety and legislation aspects. Food Res. Int. 2019, 126, 108672. [Google Scholar] [CrossRef]
- Yen, A.L. Insects as food and feed in the Asia Pacific region: Current perspectives and future directions. J. Insects Food Feed. 2015, 1, 33–55. [Google Scholar] [CrossRef]
- Phankaew, C. Standard Development of Edible Cricket Production to Good Agriculture Practice of Cricket Farming for Export; The 2nd of Progress Project Report; Department of Entomology, Faculty of Agriculture, Kasetsart University: Bangkok, Thailand, 2019. [Google Scholar]
- Nischalke, S.; Wagler, I.; Tanga, C.; Allan, D.; Phankaew, C.; Ratompoarison, C.; Razafindrakotomamonjy, A.; Kusia, E. How to turn collectors of edible insects into mini-livestock farmers: Multidimensional sustainability challenges to a thriving industry. Glob. Food Sec. 2020, 26, 100376. [Google Scholar] [CrossRef]
- Tonchow, A. Black Soldier Fly Production Manual; Natural Agriculture Research and Development Center, Maejo University: Chiang Mai, Thailand, 2017. [Google Scholar]
- Baiano, A. Edible insects: An overview on nutritional characteristics, safety, farming, production technologies, regulatory framework, and socio-economic and ethical implications. Trends Food Sci. Technol. 2020, 100, 35–50. [Google Scholar] [CrossRef]
- Customs Department of Thailand. Available online: https://www.customs.go.th/statistic_report.php (accessed on 12 February 2023).
- FAO. Edible insects. In Future Prospects for Food and Feed Security; Food and Agriculture Organization: Rome, Italy, 2013; p. 171. [Google Scholar]
- Imathiu, S. Benefits and food safety concerns associated with consumption of edible insects. NFS J. 2020, 18, 1–11. [Google Scholar] [CrossRef]
- Gahukar, R.T. Chapter 4—Edible insects farming: Efficiency and impact on family livelihood, food security, and environment compared with livestock and crops. In Insects as Sustainable Food Ingredients; Dossey, A.T., Morales-Ramos, J.A., Rojas, M.G., Eds.; Academic Press: San Diego, CA, USA, 2016; pp. 85–111. [Google Scholar]
- Halloran, A.; Hanboonsong, Y.; Roos, N.; Bruun, S. Life cycle assessment of cricket farming in north-eastern Thailand. J. Clean. Prod. 2017, 156, 83–94. [Google Scholar] [CrossRef]
- Department of Agricultural Extension Launched an Opening for the Registration of Five Economic Insect Farmers and Promoted it as a New Career to Generate Income (in Thai). Available online: https://www.prd.go.th/th/content/category/detail/id/39/iid/55566 (accessed on 2 December 2022).
- Magara, H.J.O.; Niassy, S.; Ayieko, M.A.; Mukundamago, M.; Egonyu, J.P.; Tanga, C.M.; Kimathi, E.K.; Ongere, J.O.; Fiaboe, K.K.M.; Hugel, S.; et al. Edible Crickets (Orthoptera) Around the World: Distribution, Nutritional Value, and Other Benefits—A Review. Front. Nutr. 2021, 7, 537915. [Google Scholar] [CrossRef]
- Preteseille, N.; Deguerry, A.; Reverberi, M.; Weigel, T. Insects in Thailand: National leadership and regional development, from standards to regulations through association. In Edible Insects in Sustainable Food Systems; Halloran, A., Flore, R., Vantomme, P., Roos, N., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 435–442. [Google Scholar]
- EU Legislation. International Platform of Insects for Food and Feed (IPIFF). Available online: https://ipiff.org/insects-novel-food-eu-legislation (accessed on 22 May 2023).
- van Huis, A.; Oonincx, D.G.A.B. The environmental sustainability of insects as food and feed. A review. Agron. Sustain. Dev. 2017, 37, 43. [Google Scholar] [CrossRef]
- Moruzzo, R.; Mancini, S.; Guidi, A. Edible insects and sustainable development goals. Insects 2021, 12, 557. [Google Scholar] [CrossRef]
- Mishyna, M.; Chen, J.; Benjamin, O. Sensory attributes of edible insects and insect-based foods—Future outlooks for enhancing consumer appeal. Trends Food Sci. Technol. 2020, 95, 141–148. [Google Scholar] [CrossRef]
- Bozic, A.; Milosevic, S. Contemporary trends in the restaurant industry and gastronomy. J. Hosp. Tour. Res. 2021, 45, 905–907. [Google Scholar] [CrossRef]
- Dogan, E.; Cekal, N. Sustainable food alternative in gastronomy: Edible insects (entomophagy). Int. J. Agric. Environ. Food Sci. 2022, 6, 246–253. [Google Scholar]
- Koseckova, P.; Zverina, O.; Pechova, M.; Krulikova, M.; Duborska, E.; Borkovcova, M. Mineral profile of cricket powders, some edible insect species and their implication for gastronomy. J. Food Compos. Anal. 2022, 107, 104340. [Google Scholar] [CrossRef]
- Shah, A.A.; Wanapat, M. Gryllus testaceus walker (crickets) farming management, chemical composition, nutritive profile, and their effect on animal digestibility. Entomol. Res. 2021, 51, 639–649. [Google Scholar] [CrossRef]
- Ghosh, S.; Jung, C.; Meyer-Rochow, V.B. Nutritional value and chemical composition of larvae, pupae, and adults of worker honey bee, Apis mellifera ligustica as a sustainable food source. J. Asia Pac. Entomol. 2016, 19, 487–495. [Google Scholar] [CrossRef]
- Melo-Ruiz, V.; Moreno-Bonett, C.; Sanchez-Herrera, K.; Diaz-Garcia, R.; Gazga-Urioste, C. Macronutrient composition of giant water bug (Lethocerus sp.) edible insect in Mexico and Thailand. J. Agric. Sci. Technol. 2016, 6, 349–354. [Google Scholar]
- Realm, K. Nutrient composition of the Indonesian sago grub (Rhynchophorus bilineatus). Int. J. Trop. Insect Sci. 2020, 40, 677–686. [Google Scholar]
- Adegboye, A.R. Potential use of edible insects in complementary foods for children: A literature review. Int. J. Environ. Res. Public Health. 2022, 19, 4756. [Google Scholar] [CrossRef]
- Mekuria, S.A.; Kinyuru, J.N.; Mokua, B.K.; Tenagashaw, M.W. Nutritional quality and safety of complementary foods developed from blends of staple grains and honey bee larvae (Apis mellifera). Int. J. Food Sci. 2021, 2021, 5581585. [Google Scholar] [CrossRef]
- Meyer-Rochow, V.B.; Gahukar, R.T.; Ghosh, S.; Jung, C. Chemical composition, nutrient quality and acceptability of edible insects are affected by apecies, developmental stage, gender, diet, and processing method. Foods 2021, 10, 1036. [Google Scholar] [CrossRef]
- Yang, Q.; Liu, S.; Sun, J.; Yu, L.; Zhang, C.; Bi, J.; Yang, Z. Nutritional composition and protein quality of the edible beetle Holotrichia parallela. J. Insect. Sci. 2014, 14, 139. [Google Scholar] [CrossRef]
- D’Antonio, V.; Serafini, M.; Battista, N. Dietary modulation of oxidative stress from edible insects: A mini-review. Front. Nutr. 2021, 8, 642551. [Google Scholar] [CrossRef]
- Liu, S.; Sun, J.; Yu, L.; Zhang, C.; Bi, J.; Zhu, F.; Qu, M.; Yang, Q. Antioxidant activity and phenolic compounds of Holotrichia parallela Motschulsky extracts. Food Chem. 2012, 134, 1885–1891. [Google Scholar] [CrossRef] [PubMed]
- Shi, G.; Kang, Z.; Liu, H.; Ren, F.; Zhou, Y. The effects of quercetin combined with nucleopolyhedrovirus on the growth and immune response in the silkworm (Bombyx mori). Arch. Insect Biochem. Physiol. 2021, 108, e21839. [Google Scholar] [CrossRef] [PubMed]
- Yeerong, K.; Sriyab, S.; Somwongin, S.; Punyoyai, C.; Chantawannakul, P.; Anuchapreeda, S.; Prommaban, A.; Chaiyana, W. Skin irritation and potential antioxidant, anti-collagenase, and anti-elastase activities of edible insect extracts. Sci. Rep. 2021, 11, 22954. [Google Scholar] [CrossRef] [PubMed]
- Saiki, M.; Takemoto, N.; Nagata, M.; Matsumoto, M.; Amen, Y.; Wang, D.; Shimizu, K. Analysis of antioxidant and antiallergic active components extracted from the edible insect Oxya yezoensis. Nat. Prod. Commun. 2021, 16, 1934578X211023363. [Google Scholar] [CrossRef]
- Navarro del Hierro, J.; Gutiérrez-Docio, A.; Otero, P.; Reglero, G.; Martin, D. Characterization, antioxidant activity, and inhibitory effect on pancreatic lipase of extracts from the edible insects Acheta domesticus and Tenebrio molitor. Food Chem. 2020, 309, 125742. [Google Scholar] [CrossRef]
- Nino, M.C.; Reddivari, L.; Ferruzzi, M.G.; Liceaga, A.M. Targeted phenolic characterization and antioxidant bioactivity of extracts from edible Acheta domesticus. Foods 2021, 10, 2295. [Google Scholar] [CrossRef]
- Quah, Y.; Tong, S.-R.; Bojarska, J.; Giller, K.; Tan, S.-A.; Ziora, Z.M.; Esatbeyoglu, T.; Chai, T.-T. Bioactive peptide discovery from edible insects for potential applications in human health and agriculture. Molecules 2023, 28, 1233. [Google Scholar] [CrossRef]
- Botella-Martínez, C.; Lucas-González, R.; Pérez-Álvarez, J.A.; Fernández-López, J.; Viuda-Martos, M. Assessment of chemical composition and antioxidant properties of defatted flours obtained from several edible insects. Food Sci. Technol. Int. 2021, 27, 383–391. [Google Scholar] [CrossRef]
- Djouadi, A.; Sales, J.R.; Carvalho, M.O.; Raymundo, A. Development of healthy protein-rich crackers using Tenebrio molitor flour. Foods 2022, 11, 702. [Google Scholar] [CrossRef]
- Dutta, P.; Dey, T.; Manna, P.; Kalita, J. Antioxidant potential of Vespa affinis L., a traditional edible insect species of North East India. PLoS ONE 2016, 11, e0156107. [Google Scholar] [CrossRef]
- Takechi, T.; Wada, R.; Fukuda, T.; Harada, K.; Takamura, H. Antioxidant activities of two sericin proteins extracted from cocoon of silkworm (Bombyx mori) measured by DPPH, chemiluminescence, ORAC and ESR methods. Biomed. Rep. 2014, 2, 364–369. [Google Scholar] [CrossRef] [PubMed]
- Wannee, S.; Luchai, B. 1-Deoxynojirimycin and polyphenolic composition and antioxidant activity of different native Thai silkworm (Bombyx mori) larvae. J. King Saud Univ. Sci. 2020, 32, 2762–2766. [Google Scholar] [CrossRef]
- Pyo, S.J.; Kang, D.G.; Jung, C.; Sohn, H.Y. Anti-thrombotic, anti-oxidant and haemolysis activities of six edible insect species. Foods 2020, 9, 401. [Google Scholar] [CrossRef] [PubMed]
- Alagappan, S.; Chaliha, M.; Sultanbawa, Y.; Fuller, S.; Hoffman, L.C.; Netzel, G.; Weber, N.; Rychlik, M.; Cozzolino, D.; Smyth, H.E.; et al. Nutritional analysis, volatile composition, antimicrobial and antioxidant properties of Australian green ants (Oecophylla smaragdina). Future Foods 2021, 3, 100007. [Google Scholar] [CrossRef]
- Melo da Cunha, J.d.S.; Alfredo, T.M.; dos Santos, J.M.; Alves Junior, V.V.; Rabelo, L.A.; Lima, E.S.; Boleti, A.P.d.A.; Carollo, C.A.; dos Santos, E.L.; de Picoli Souza, K. Antioxidant, antihyperglycemic, and antidiabetic activity of Apis mellifera bee tea. PLoS ONE 2018, 13, e0197071. [Google Scholar] [CrossRef]
- Sidor, E.; Dżugan, M. Drone brood homogenate as natural remedy for treating health care problem: A scientific and practical approach. Molecules 2020, 25, 5699. [Google Scholar] [CrossRef]
- Sidor, E.; Miłek, M.; Tomczyk, M.; Dżugan, M. Antioxidant activity of frozen and freeze-dried drone brood homogenate regarding the stage of larval development. Antioxidants 2021, 10, 639. [Google Scholar] [CrossRef]
- Andreadis, S.S.; Panteli, N.; Mastoraki, M.; Rizou, E.; Stefanou, V.; Tzentilasvili, S.; Sarrou, E.; Chatzifotis, S.; Krigas, N.; Antonopoulou, E. Towards functional insect feeds: Agri-food by-products enriched with post-distillation residues of medicinal aromatic plants in Tenebrio molitor (Coleoptera: Tenebrionidae). Antioxidants 2022, 11, 68. [Google Scholar] [CrossRef]
- Zielinska, E.; Baraniak, B.; Karas, M. Antioxidant and anti-inflammatory activities of hydrolysates and peptide fractions obtained by enzymatic hydrolysis of selected heat-treated edible insects. Nutrients 2017, 9, 970. [Google Scholar] [CrossRef]
- Pu, Y.C.; Xiang, H.J.; Liang, X.Y.; Wang, Y.; Hou, Y.M.; Fu, L.; Wang, R. External immune inhibitory efficiency of external secretions and their metabolic profiling in Red Palm Weevil, Rhynchophorus ferrugineus (Coleoptera: Curculionidae). Front Physiol. 2020, 10, 1624. [Google Scholar] [CrossRef]
- Kim, S.R.; Hong, S.J.; Choi, K.; Kim, S.W.; Jeong, S.T.; Park, S.W. Antibacterial and anti-inflammatory activities of the immune-challenged silkworm (Bombyx mori) hemolymph with Lactobacillus cell wall extracts. Entomol. Res. 2019, 49, 354–362. [Google Scholar] [CrossRef]
- Yun, E.Y.; Lee, J.K.; Kwon, O.Y.; Hwang, J.S.; Kim, I.; Kang, S.W.; Lee, W.J.; Ding, J.L.; You, K.H.; Goo, T.W. Bombyx mori transferrin: Genomic structure, expression and antimicrobial activity of recombinant protein. Dev. Comp. Immunol. 2009, 33, 1064–1069. [Google Scholar] [CrossRef] [PubMed]
- Hwang, D.; Lee, S.H.; Goo, T.W.; Yun, E.Y. Potential of antimicrobial peptide-overexpressed Tenebrio molitor larvae extract as a natural preservative for Korean traditional sauces. Insects 2022, 13, 381. [Google Scholar] [CrossRef] [PubMed]
- Wen, X.; Gongpan, P.; Meng, Y.; Nieh, J.C.; Yuan, H.; Tan, K. Functional characterization, antimicrobial effects, and potential antibacterial mechanisms of new mastoparan peptides from hornet venom (Vespa ducalis, Vespa mandarinia, and Vespa affinis). Toxicon 2021, 200, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.; Wong, N.A.K.; Chae, M.; Auh, J.H. Comparative characterization of protein hydrolysates from three edible insects: Mealworm larvae, adult crickets, and silkworm pupae. Foods 2019, 8, 563. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.W.; Hwang, H.S.; Kim, D.-S.; Sheen, S.H.; Heo, D.H.; Hwang, G.; Kang, S.H.; Kweon, H.; Jo, Y.-Y.; Kang, S.W.; et al. Effect of silk fibroin peptide derived from silkworm Bombyx mori on the anti-inflammatory effect of Tat-SOD in a mice edema model. BMB Rep. 2011, 44, 787–792. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Zhou, S.T.; Wu, X.M.; Pang, X.Q.; Ni, L.L.; Yuan, S.M.; Yang, Z.B.; Li, Y.H.; Xiao, H. GC-MS analysis of Vespa velutina auraria Smith and its anti-inflammatory and antioxidant activities in vitro. Open Chem. 2022, 20, 602–610. [Google Scholar] [CrossRef]
- Ahn, M.Y.; Han, J.W.; Hwang, J.S.; Yun, E.Y.; Lee, B.M. Anti-inflammatory effect of glycosaminoglycan derived from Gryllus bimaculatus (a type of cricket, insect) on adjuvant-treated chronic arthritis rat model. J. Toxicol. Environ. Health A 2014, 77, 1332–1345. [Google Scholar] [CrossRef]
- Son, Y.J.; Choi, S.Y.; Hwang, I.K.; Nho, C.W.; Kim, S.H. Could defatted mealworm (Tenebrio molitor) and mealworm oil be used as food ingredients? Foods 2020, 9, 40. [Google Scholar] [CrossRef]
- Zhao, J.G.; Wang, H.Y.; Wei, Z.G.; Zhang, Y.Q. Therapeutic effects of ethanolic extract from the green cocoon shell of silkworm Bombyx mori on type 2 diabetic mice and its hypoglycaemic mechanism. Toxicol. Res. 2019, 8, 407–420. [Google Scholar] [CrossRef]
- Hall, F.; Reddivari, L.; Liceaga, A.M. Identification and characterization of edible cricket peptides on hypertensive and glycemic In vitro inhibition and their anti-Inflammatory activity on RAW 264.7 macrophage cells. Nutrients 2020, 12, 3588. [Google Scholar] [CrossRef] [PubMed]
- Dong-Hwan, S.E.O.; Hwang, S.Y.; Han, J.; Koh, S.K.; Kim, I.; Ryu, K.S.; Yun, C.Y. Immune-enhancing activity screening on extracts from two crickets, Gryllus bimaculatus and Teleogryllus emma. Entomol. Res. 2004, 34, 207–211. [Google Scholar] [CrossRef]
- Ojha, S.; Bußler, S.; Psarianos, M.; Rossi, G.; Schlüter, O.K. Edible insect processing pathways and implementation of emerging technologies. J. Insects Food Feed. 2021, 7, 877–900. [Google Scholar] [CrossRef]
- De Smet, J.; Lenaerts, S.; Borremans, A.; Scholliers, J.; Van Der Borght, M.; Van Campenhout, L. Stability assessment and laboratory scale fermentation of pastes produced on a pilot scale from mealworms (Tenebrio molitor). LWT 2019, 102, 113–121. [Google Scholar] [CrossRef]
- Tenyang, N.; Tiencheu, B.; Mamat, A.; Mawamba, L.A.; Ponka, R. Effect of cooking methods on the nutritive value and lipid oxidation of two cricket species consumed In Cameroon. Eur. J. Nutr. Food Saf. 2021, 13, 11–23. [Google Scholar] [CrossRef]
- Kowalczewski, P.L.; Walkowiak, K.; Masewicz, L.; Bartczak, O.; Lewandowicz, J.; Kubiak, P.; Baranowska, H.M. Gluten-free bread with cricket powder—Mechanical properties and molecular water dynamics in dough and ready product. Foods 2019, 8, 240. [Google Scholar] [CrossRef]
- Zielinska, E.; Pankiewicz, U.; Sujka, M. Nutritional, physiochemical, and biological value of muffins enriched with edible insects flour. Antioxidants 2021, 10, 1122. [Google Scholar] [CrossRef]
- Wendin, K.; Olsson, V.; Langton, M. Mealworms as food ingredient-sensory investigation of a model system. Foods 2019, 8, 319. [Google Scholar] [CrossRef]
- Liceaga, A.M. Processing insects for use in the food and feed industry. Curr. Opin. Insect Sci. 2021, 48, 32–36. [Google Scholar] [CrossRef]
- Sakadevan, K.; Nguyen, M.L. Chapter four - livestock production and its impact on nutrient pollution and greenhouse gas emissions. In Advances in Agronomy; Sparks, D.L., Ed.; Academic Press: San Diego, CA, USA, 2017; Volume 141, pp. 147–184. [Google Scholar]
- Kim, H.W.; Setyabrata, D.; Lee, Y.J.; Jones, O.G.; Kim, Y.H.B. Pre-treated mealworm larvae and silkworm pupae as a novel protein ingredient in emulsion sausages. Innov. Food Sci. Emerg. Technol. 2016, 38, 116–123. [Google Scholar] [CrossRef]
- Boukil, A.; Marciniak, A.; Mezdour, S.; Pouliot, Y.; Doyen, A. Effect of high hydrostatic pressure intensity on structural modifications in mealworm (Tenebrio molitor) proteins. Foods 2022, 11, 956. [Google Scholar] [CrossRef] [PubMed]
- Laroche, M.; Perreault, V.; Marciniak, A.; Gravel, A.; Chamberland, J.; Doyen, A. Comparison of conventional and sustainable lipid extraction methods for the production of oil and protein isolate from edible insect meal. Foods 2019, 8, 572. [Google Scholar] [CrossRef] [PubMed]
- Cheng, K.; Leong, K.; Chan, S. Cricket as an alternative source of protein in the development of nutritious baked chips. Food Res. 2022, 6, 74–82. [Google Scholar] [CrossRef]
- Tedjakusuma, F.; Linggadiputra, J.; Cahya, A.D.; Surya, R. Development of cricket flour-enriched cookies. In Proceedings of the IOP Conference Series: Earth and Environmental Science, Athens, Greece, 22–23 October 2022; p. 012092. [Google Scholar]
- Kiatbenjakul, P.; Intarapichet, K.-O.; Cadwallader, K.R. Characterization of potent odorants in male giant water bug (Lethocerus indicus Lep. and Serv.), an important edible insect of Southeast Asia. Food Chem. 2015, 168, 639–647. [Google Scholar] [CrossRef]
- Tzompa-Sosa, D.A.; Yi, L.; Van Valenberg, H.; Lakemond, C. Four insect oils as food ingredient: Physical and chemical characterisation of insect oils obtained by an aqueous oil extraction. J. Insects Food Feed. 2019, 5, 279–292. [Google Scholar] [CrossRef]
- Harkin, C.; Mehlmer, N.; Woortman, D.V.; Brück, T.B.; Brück, W.M. Nutritional and additive uses of chitin and chitosan in the food industry. In Sustainable Agriculture Reviews 36: Chitin and Chitosan: Applications in Food, Agriculture, Pharmacy, Medicine and Wastewater Treatment; Crini, G., Lichtfouse, E., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 1–43. [Google Scholar]
- Zainol Abidin, N.A.; Kormin, F.; Zainol Abidin, N.A.; Mohamed Anuar, N.A.; Abu Bakar, M.F. The potential of insects as alternative sources of chitin: An overview on the chemical method of extraction from various sources. Int. J. Mol. Sci. 2020, 21, 4978. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Sun, J.; Yu, L.; Zhang, C.; Bi, J.; Zhu, F.; Qu, M.; Jiang, C.; Yang, Q. Extraction and characterization of chitin from the beetle Holotrichia parallela Motschulsky. Molecules 2012, 17, 4604–4611. [Google Scholar] [CrossRef]
- Van Huis, A.; Van Itterbeeck, J.; Klunder, H.; Mertens, E.; Halloran, A.; Muir, G.; Vantomme, P. Edible Insects: Future Prospects for Food and Feed Security; Food and Agriculture Organization of the United Nations: Rome, Italy, 2013. [Google Scholar]
- Bungthong, C.; Siriamornpun, S. Changes in amino acid profiles and bioactive compounds of Thai silk cocoons as affected by water extraction. Molecules 2021, 26, 2033. [Google Scholar] [CrossRef]



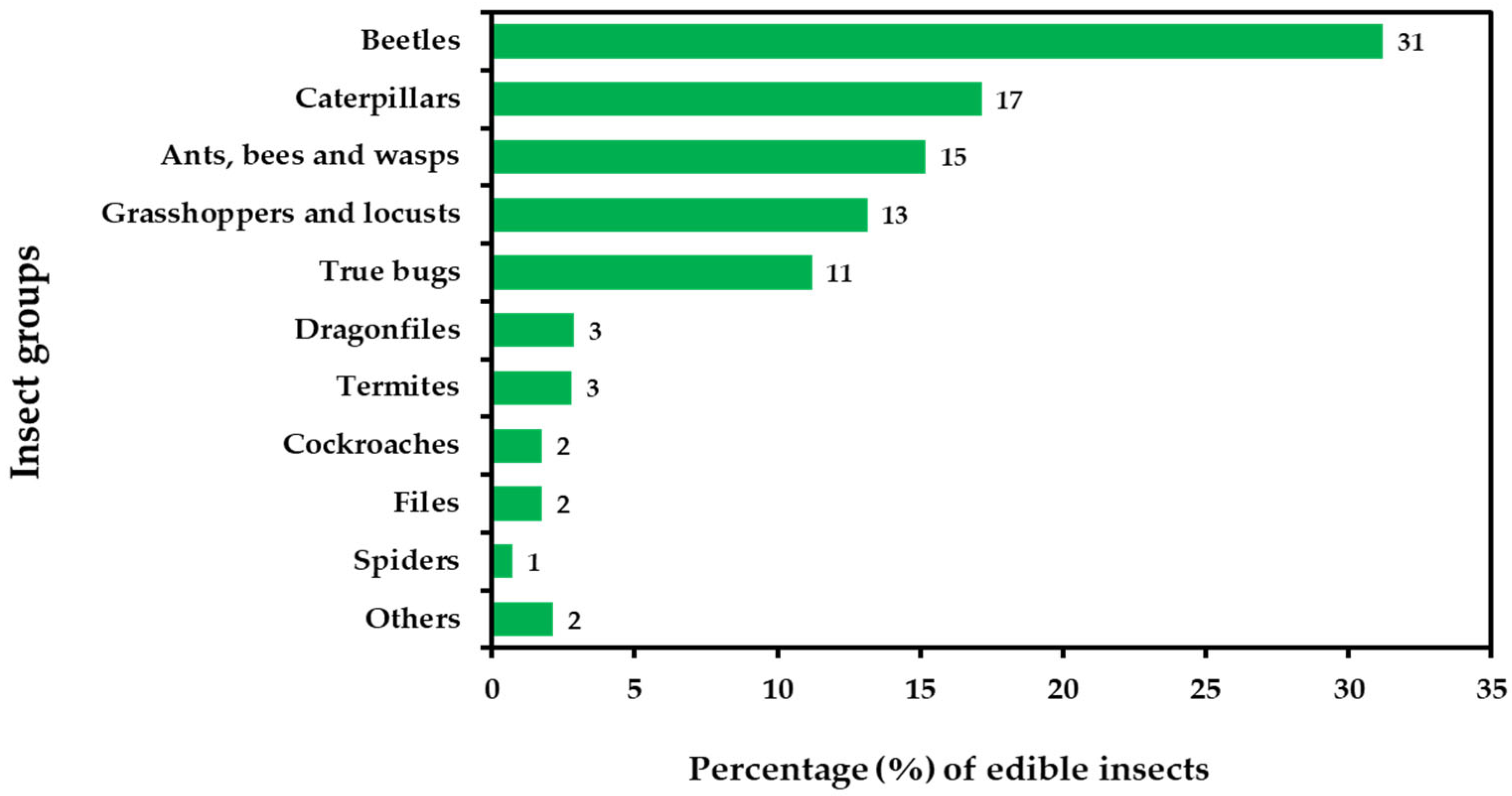
| Common Name (English) | Local Name (Thai to English) a | Scientific Name | Edible Stage * | Harvested Source ** | Price/kg (USD) b |
|---|---|---|---|---|---|
| Bombay locust | Tuck-tan (Patanga) | Patanga succincta (Johannson, 1763) | A | W | 6.47–7.35 |
| Small rice grasshopper | Tuck-tan (lek) | Oxya japonica japonica (Thunberg, 1815) Locusta migratoria manilensis (Meyen, 1835) | A | W | 5.88–7.35 |
| Yellow-backed grasshopper | Tuck-tan (yai) | Cyrtacanthacris tatarica (Linnaeus, 1758) | A | W | 5.88–7.35 |
| Cockchafer scarab beetle/June beetle | Ki-noon | Holotrichia sp. | A | W/F | 5.88–10.30 |
| Red palm weevil/Sago palm weevil | Duang Saku | Rhynchophorus ferrugineus (Olivier, 1790) | L | F | 7.35–8.82 |
| Dung beetle | Kud-jee | Paragymnopleurus aethiops (Sharp, 1875) | P/A | W | 0.30–1.50/each |
| Paper wasps/Lesser banded hornet | Tdaw | Vespa affinis (Linnaeus, 1764) | L/P | W/S | 44.12–58.82 |
| Silkworm | Dug-dae Mai | Bombyx mori (Linnaeus, 1758) | P | F | 5.88–14.40 |
| Bamboo caterpillar | Non Mai-pai | Omphisa fuscidentalis (Hampson, 1896) | L | W | 35.30–73.53 |
| Subterranean ant | Mangmun | Carebara castanea (Smith, 1858) | A/E | W | 35.30–58.82 (adults) 58.82–73.53 (eggs) |
| True water beetle | Tub-toa | Cybister limbatus (Fabricius, 1775) | A | W | 9.70–11.17 |
| Water scavenger beetle | Malaeng niang | Hydrous cavistanum | A | W | n.d. |
| Crickets/African cricket/Mediterranean field cricket/Two-spotted cricket | Jing-reed (Tong dam) | Gryllus bimaculatus (De Geer, 1773) | A | W/F | 2.94–3.53 |
| Ground cricket | Jing-reed Tongdang | Teleogryllus mitratus (Burmeister, 1838) | A | W/F | 2.94–3.53 |
| House cricket | Jing-reed (Tong dang li) (sa-ding)/Ji-reed Ban | Acheta domesticus (Linnaeus, 1758) | A | W/F | 3.82–4.41 |
| Short-tailed cricket | Ji-pome, Ji-koong | Brachytrupes portentosus (Lichtenstein, 1796) | A | W/F | 3.53 |
| Mole cricket | Kra-chon/Ji-zon | Gryllotalpa africana (Palisot de Beauvois, 1820) | A | W/F | 4.41 |
| Red ant/weaver ant | Mod-dang Kai-mod-dang Ma-peng | Oecophylla smaragdina (Fabricius, 1775) | E/P | W/S | 14.70–29.14 |
| Buprestis beetle | Malaeng Tub | Sternocera aequisignata (Saunders, 1866) | A | W/S | n.d. |
| Honey bee | Pung | Apis spp. | L/P | W/F | 11.76–17.65 |
| Cicada | Juk-jan | Meimuna opalifera (Walker, 1850) | A | W | 11.76–14.70 |
| Winged Termite (Alates) | Mang-moa | Termes sp. | A | W | 4.41–5.88 |
| Giant water bug | Mang da na | Lethocerus indicus (Lepeletier et Serville, 1775) | A | W/F | Male 0.29–0.58/each Female 0.23–0.50/each |
| Water scorpion | Mang pong nam | Laccotrephes ruber (Linnaeus, 1764) | A | W | n.d. |
| Scarab beetle/Siamese rhinoceros beetle/Fighting beetle/Rhinoceros beetles | Duang Kwang | Xylotrupes gideon (Linnaeus, 1767) | A | W/F | 11.76–14.70 |
| Mealworm | Non Nok | Tenebrio molitor (Linnaeus, 1758) | L | F | 7.35–8.82 |
| Common skimmer/River skimmer | Mang ra ngum | Crocothemis sp. | N | W | 4.41–5.88 |
| Chemical Composition | Content | |||
|---|---|---|---|---|
| Cricket (g/100 g) [49] | Honeybee Larvae (g/100 g) [50] | Giant Water Bug (g/100 g) [51] | Sago Worms (g/100 g) [52] | |
| Protein | 63.3 | 35.3 | 53.11 | 10.39 |
| Fat | 10.3 | 14.5 | 8.15 | 17.17 |
| Fiber | 5.2 | - | 12.23 | - |
| Ash | 5.6 | 4.1 | - | - |
| Carbohydrate | - | 46.1 | 19.74 | - |
| Minerals | - | - | 6.75 | - |
| Calcium | 10.1 | 0.0849 | - | 0.0149 |
| Phosphorous | 7.9 | 0.7825 | - | 0.1023 |
| Magnesium | 1.2 | 0.1770 | - | 0.0526 |
| Zinc | 0.215 | 0.0116 | - | 0.0029 |
| Copper | 0.015 | 0.0036 | - | 0.0004 |
| Manganese | 0.040 | 0.0012 | - | 0.0004 |
| Iron | 0.116 | 0.0131 | - | 0.0008 |
| Sodium | - | 0.0594 | - | 0.0166 |
| Potassium | - | 1.8719 | - | 0.2046 |
| Bioactive Compounds | Species | References |
|---|---|---|
| Anti-oxidant activity | ||
| Phenolic compounds | Acheta domesticus, Tenebrio molitor | [62] |
| 4-Hydroxybenzoic acid, p-coumaric acid, ferulic acid, syringic acid | Acheta domesticus | [63] |
| Catechin (phenolic compounds) and proteins | Holotrichia parallela | [58] |
| Peptides (CTKKHKPNC) | Oecophylla smaragdina | [64] |
| Total phenolic compounds | Rhynchophorus ferrugineus | [65] |
| Total phenolic compounds | Tenebrio molitor | [66] |
| Crude extract | Vespa affinis | [67] |
| Total phenolic compounds, sericin proteins (silk cocoon), Silkworm pupae protein concentrate, 35-kDa protein, 1-deoxynojirimycin (DNJ) | Bombyx mori | [68,69] |
| Crude extract | Teleogryllus emma | [70] |
| Total phenolic contents | Oecophylla smaragdina | [71] |
| Unknown | Apis mellifera (bee tea) | [72] |
| Total phenolic acids and flavonoids | Apis mellifera (drone brood) | [73,74] |
| Total phenolic and flavonoid compounds, mealworm oil, defatted mealworm, peptides | Tenebrio molitor | [75,76] |
| Crude extract | Euconocephalus sp. | [60] |
| Crude extract | Lethocerus indicus | [60] |
| Crude extract | Patanga succincta | [60] |
| Antibacterial activity | ||
| p-Benzoquinone | Rhynchophorus ferrugineus | [77] |
| Hemolymph, seroin 1, 2, and 3 proteins, cecropin, cecropin B moricin, gloverin, vitellogenin, derived oils, recombinant Bombyx mori transferrin (BmTf) | Bombyx mori | [78,79] |
| Crude extract | Oecophylla smaragdina | [71] |
| Immunized Tenebrio molitor larvae (iTME) | Tenebrio molitor larvae | [80] |
| Mastoparan peptides | Vespa affinis | [81] |
| Anti-inflammatory activity | ||
| Protein/peptide hydrolysates, hemolymph, fibroin peptide | Bombyx mori | [78,82,83] |
| Protein/peptide hydrolysates | Gryllodes sigillatus , and Tenebrio molitor | [76] |
| Locust cyclopeptides (LCPs) | Locusta migratoria | |
| Unknown | Vespa affinis | [84] |
| Glycosaminoglycan | Gryllus bimaculatus | [85] |
| Mealworm oil, defatted mealworm, peptides | Tenebrio molitor | [86] |
| Anti-collagenase activity | ||
| Crude extract | Acheta domesticus | [60] |
| Crude extract | Bombyx mori | [60] |
| Crude extract | Patanga succincta | [60] |
| Crude extract | Euconocephalus sp. | [60] |
| Crude extract | Lethocerus indicus | [60] |
| Crude extract | Omphisa fuscidentalis | [60] |
| Elastase-inhibitory activity | ||
| Crude extract | Acheta domesticus | [60,62] |
| Crude extract | Bombyx mori | [60] |
| Crude extract | Euconocephalus sp. | [60] |
| Crude extract | Lethocerus indicus | [60] |
| Crude extract | Patanga succincta | [60] |
| Peptide hydrolysates | Gryllodes sigillatus | [76] |
| α-Glucosidase-inhibitory activity | ||
| Protein/peptide hydrolysates | Bombyx mori, Tenebrio molitor | [76] |
| Hepatoprotective activity | ||
| Peptide (AGLQFPVGR) | Allomyrina dichotoma | [64] |
| Peptide (LE, AKKHKE) | Tenebrio molitor | [64] |
| Inhibitory activity of pancreatic lipase | ||
| Crude extract | Tenebrio molitor | [62] |
| Antidiabetic/insulin-like/insulin-like peptide (ApILP) | ||
| Sericin (green cocoon shell) | Bombyx mori | [87] |
| Antidiabetic | ||
| Unknown | Apis mellifera (bee tea) | [72] |
| Peptides (EIAQDFKTDL) | Allomyrina dichotoma | [64] |
| Cationic peptide | Gryllodes sigillatus | [88] |
| Angiotensin-converting enzyme (ACE) inhibition | ||
| Peptides (ASL, GNPWM) | Bombyx mori | [64] |
| Immune-enhancing activity | ||
| Crude extract | Gryllus bimaculatus | [89] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krongdang, S.; Phokasem, P.; Venkatachalam, K.; Charoenphun, N. Edible Insects in Thailand: An Overview of Status, Properties, Processing, and Utilization in the Food Industry. Foods 2023, 12, 2162. https://doi.org/10.3390/foods12112162
Krongdang S, Phokasem P, Venkatachalam K, Charoenphun N. Edible Insects in Thailand: An Overview of Status, Properties, Processing, and Utilization in the Food Industry. Foods. 2023; 12(11):2162. https://doi.org/10.3390/foods12112162
Chicago/Turabian StyleKrongdang, Sasiprapa, Patcharin Phokasem, Karthikeyan Venkatachalam, and Narin Charoenphun. 2023. "Edible Insects in Thailand: An Overview of Status, Properties, Processing, and Utilization in the Food Industry" Foods 12, no. 11: 2162. https://doi.org/10.3390/foods12112162
APA StyleKrongdang, S., Phokasem, P., Venkatachalam, K., & Charoenphun, N. (2023). Edible Insects in Thailand: An Overview of Status, Properties, Processing, and Utilization in the Food Industry. Foods, 12(11), 2162. https://doi.org/10.3390/foods12112162