Effect of Fractionation and Processing Conditions on the Digestibility of Plant Proteins as Food Ingredients
Abstract
:1. Introduction
2. Method and Definitions
3. Ingredient Preparation
3.1. Pre-Fractionation Treatment
3.2. Conventional Protein Fractionation
3.3. Alternative Protein Fractionation Strategies
4. Post-Fractionation Processing
4.1. Proteolysis
4.2. Heat Treatment
4.3. High Pressure Processing
5. Crosslinking, Complexation and Other Modifications
5.1. Transglutaminase-Catalysed Polymerization
5.2. Acylation
5.3. Complexation with Phenolic Compounds
5.4. Protein Oxidation
5.5. Other Modifications
6. Structure Formation
6.1. Extrusion and Texturization
6.2. Pre- and Intra-Gastric Gelation
7. Macronutrient Interactions
7.1. Animal- and Plant-Based Protein Hybrid Foods
7.2. Starch
7.3. Fibre
8. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Rockström, J.; Steffen, W.; Noone, K.; Persson, Å.; Chapin, F.S.; Lambin, E.F.; Lenton, T.M.; Scheffer, M.; Folke, C.; Schellnhuber, H.J.; et al. A safe operating space for humanity. Nature 2009, 461, 472–475. [Google Scholar] [CrossRef] [PubMed]
- Aiking, H.; de Boer, J. The next protein transition. Trends Food Sci. Technol. 2020, 105, 515–522. [Google Scholar] [CrossRef]
- Willett, W.; Rockström, J.; Loken, B.; Springmann, M.; Lang, T.; Vermeulen, S.; Garnett, T.; Tilman, D.; DeClerck, F.; Wood, A.; et al. Food in the Anthropocene: The EAT-Lancet Commission on healthy diets from sustainable food systems. Lancet 2019, 393, 447–492. [Google Scholar] [CrossRef]
- Euromonitor International. The Rise of Vegan and Vegetarian Food; Euromonitor: London, UK, 2020. [Google Scholar]
- Smetana, S.; Mathys, A.; Knoch, A.; Heinz, V. Meat alternatives: Life cycle assessment of most known meat substitutes. Int. J. Life Cycle Assess. 2015, 20, 1254–1267. [Google Scholar] [CrossRef]
- Van der Weele, C.; Feindt, P.; Jan van der Goot, A.; van Mierlo, B.; van Boekel, M. Meat alternatives: An integrative comparison. Trends Food Sci. Technol. 2019, 88, 505–512. [Google Scholar] [CrossRef]
- Sim, S.Y.J.; SRV, A.; Chiang, J.H.; Henry, C.J. Plant Proteins for Future Foods: A Roadmap. Foods 2021, 10, 1967. [Google Scholar] [CrossRef] [PubMed]
- Herreman, L.; Nommensen, P.; Pennings, B.; Laus, M.C. Comprehensive overview of the quality of plant- and animal-sourced proteins based on the digestible indispensable amino acid score. Food Sci. Nutr. 2020, 8, 5379–5391. [Google Scholar] [CrossRef]
- Sá, A.G.A.; Moreno, Y.M.F.; Carciofi, B.A.M. Plant proteins as high-quality nutritional source for human diet. Trends Food Sci. Technol. 2020, 97, 170–184. [Google Scholar] [CrossRef]
- Rutherfurd, S.M.; Fanning, A.C.; Miller, B.J.; Moughan, P.J. Protein digestibility-corrected amino acid scores and digestible indispensable amino acid scores differentially describe protein quality in growing male rats. J. Nutr. 2015, 145, 372–379. [Google Scholar] [CrossRef] [Green Version]
- Santos-Hernández, M.; Alfieri, F.; Gallo, V.; Miralles, B.; Masi, P.; Romano, A.; Ferranti, P.; Recio, I. Compared digestibility of plant protein isolates by using the INFOGEST digestion protocol. Food Res. Int. 2020, 137, 109708. [Google Scholar] [CrossRef]
- Drulyte, D.; Orlien, V. The Effect of Processing on Digestion of Legume Proteins. Foods 2019, 8, 224. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, L.H.; Hung, T.V.; Bennett, L. Extraction and characterization of chickpea (Cicer arietinum) albumin and globulin. J. Food Sci. 2008, 73, C299–C305. [Google Scholar] [CrossRef] [PubMed]
- Sathe, S.K.; Iyer, V.; Salunkhe, D.K. Functional Properties of the Great Northern Bean (Phaseolus Vulgaris L.) Proteins. Amino Acid Composition, In Vitro Digestibility, and Application to Cookies. J. Food Sci. 1982, 47, 8–11. [Google Scholar] [CrossRef]
- Wang, X.S.; Tang, C.H.; Yang, X.Q.; Gao, W.R. Characterization, amino acid composition and in vitro digestibility of hemp (Cannabis sativa L.) proteins. Food Chem. 2008, 107, 11–18. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, Z.; Wang, R.; Sui, X.; Qi, B.; Han, F.; Li, Y.; Jiang, L. Secondary Structure and Subunit Composition of Soy Protein in Vitro Digested by Pepsin and Its Relation with Digestibility. BioMed Res. Int. 2016, 2016, 5498639. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mohamed, T.K.; Zhu, K.; Issoufou, A.; Fatmata, T.; Zhou, H. Functionality, in vitro digestibility and physicochemical properties of two varieties of defatted foxtail millet protein concentrates. Int. J. Mol. Sci. 2009, 10, 5224–5238. [Google Scholar] [CrossRef] [Green Version]
- Singh, T.P.; Sogi, D.S. Comparative study of structural and functional characterization of bran protein concentrates from superfine, fine and coarse rice cultivars. Int. J. Biol. Macromol. 2018, 111, 281–288. [Google Scholar] [CrossRef]
- Espinosa-Ramírez, J.; Serna-Saldívar, S.O. Functionality and characterization of kafirin-rich protein extracts from different whole and decorticated sorghum genotypes. J. Cereal Sci. 2016, 70, 57–65. [Google Scholar] [CrossRef]
- Vogelsang-O’Dwyer, M.; Bez, J.; Petersen, I.L.; Joehnke, M.S.; Detzel, A.; Busch, M.; Krueger, M.; Ispiryan, L.; O’Mahony, J.A.; Arendt, E.K.; et al. Techno-functional, nutritional and environmental performance of protein isolates from blue lupin and white lupin. Foods 2020, 9, 230. [Google Scholar] [CrossRef] [Green Version]
- Wally-Vallim, A.P.; Vanier, N.L.; da Rosa Zavareze, E.; Zambiazi, R.C.; de Castro, L.A.S.; Schirmer, M.A.; Elias, M.C. Isoflavone Aglycone Content and the Thermal, Functional, and Structural Properties of Soy Protein Isolates Prepared from Hydrothermally Treated Soybeans. J. Food Sci. 2014, 79, E1351–E1358. [Google Scholar] [CrossRef] [Green Version]
- Marques, M.R.; Soares Freitas, R.A.M.; Corrêa Carlos, A.C.; Siguemoto, É.S.; Fontanari, G.G.; Arêas, J.A.G. Peptides from cowpea present antioxidant activity, inhibit cholesterol synthesis and its solubilisation into micelles. Food Chem. 2015, 168, 288–293. [Google Scholar] [CrossRef] [PubMed]
- Dikshit, M.; Ghadle, M. Effect of sprouting on nutrients, antinutrients and in vitro digestibility of the MACS-13 soybean variety. Plant Foods Hum. Nutr. 2003, 58, 1–11. [Google Scholar] [CrossRef]
- López-Barrios, L.; Antunes-Ricardo, M.; Gutiérrez-Uribe, J.A. Changes in antioxidant and antiinflammatory activity of black bean (Phaseolus vulgaris L.) protein isolates due to germination and enzymatic digestion. Food Chem. 2016, 203, 417–424. [Google Scholar] [CrossRef] [PubMed]
- Aijie, L.; Shouwei, Y.; Li, L. Structure, trypsin inhibitor activity and functional properties of germinated soybean protein isolate. Int. J. Food Sci. Technol. 2014, 49, 911–919. [Google Scholar] [CrossRef]
- Sefatie, R.S.; Fatoumata, T.; Eric, K.; Shi, Y.H.; Guo-Wei, L. In vitro antioxidant activities of protein hydrolysate from germinated black soybean (Glycine max L.). Adv. J. Food Sci. Technol. 2013, 5, 453–459. [Google Scholar] [CrossRef]
- Bartkiene, E.; Sakiene, V.; Bartkevics, V.; Rusko, J.; Lele, V.; Juodeikiene, G.; Wiacek, C.; Braun, P.G. Lupinus angustifolius L. lactofermentation and protein isolation: Effects on phenolic compounds and genistein, antioxidant properties, trypsin inhibitor activity, and protein digestibility. Eur. Food Res. Technol. 2018, 244, 1521–1531. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, Y.; Li, Y.; Dai, C.; Ding, Q.; Hong, C.; He, Y.; He, R.; Ma, H. Effect of alkali concentration on digestibility and absorption characteristics of rice residue protein isolates and lysinoalanine. Food Chem. 2019, 289, 609–615. [Google Scholar] [CrossRef]
- Yu, R.S.T.; Kyle, W.S.A.; Hung, T.V.; Zeckler, R. Characterisation of aqueous extracts of seed proteins of Lupinus albus and Lupinus angustifolius. J. Sci. Food Agric. 1987, 41, 205–218. [Google Scholar] [CrossRef]
- Ruiz, G.A.; Opazo-Navarrete, M.; Meurs, M.; Minor, M.; Sala, G.; van Boekel, M.; Stieger, M.; Janssen, A.E.M. Denaturation and in vitro gastric digestion of heat-treated quinoa protein isolates obtained at various extraction pH. Food Biophys. 2016, 11, 184–197. [Google Scholar] [CrossRef]
- Hughes, G.J.; Ryan, D.J.; Mukherjea, R.; Schasteen, C.S. Protein digestibility-corrected amino acid scores (PDCAAS) for soy protein isolates and concentrate: Criteria for evaluation. J. Agric. Food Chem. 2011, 59, 12707–12712. [Google Scholar] [CrossRef]
- Pedersen, C.; Almeida, J.S.; Stein, H.H. Analysis of published data for standardized ileal digestibility of protein and amino acids in soy proteins fed to pigs. J. Anim. Sci. 2016, 94, 340–343. [Google Scholar] [CrossRef] [Green Version]
- Da Silva Pinto, M.; Lajolo, F.M.; Genovese, M.I. Effect of storage temperature and water activity on the content and profile of isoflavones, antioxidant activity, and in vitro protein digestibility of soy protein isolates and defatted soy flours. J. Agric. Food Chem. 2005, 53, 6340–6346. [Google Scholar] [CrossRef] [PubMed]
- Chamba, M.V.M.; Hua, Y.; Murekatete, N.; Chen, Y. Effects of synthetic and natural extraction chemicals on yield, composition and protein quality of soy protein isolates extracted from full-fat and defatted flours. J. Food Sci. Technol. 2013, 52, 1016–1023. [Google Scholar] [CrossRef] [Green Version]
- Lqari, H.; Vioque, J.; Pedroche, J.; Millán, F. Lupinus angustifolius protein isolates: Chemical composition, functional properties and protein characterization. Food Chem. 2002, 76, 349–356. [Google Scholar] [CrossRef]
- Sánchez-Vioque, R.; Clemente, A.; Vioque, J.; Bautista, J.; Millán, F. Protein isolates from chickpea (Cicer arietinum L.): Chemical composition, functional properties and protein characterization. Food Chem. 1999, 64, 237–243. [Google Scholar] [CrossRef]
- Baker, E.C.; Rackis, J.J. Preparation of unheated soy protein isolates with low trypsin inhibitor content. Adv. Exp. Med. Biol. 1986, 199, 349–355. [Google Scholar] [CrossRef]
- Joehnke, M.S.; Jeske, S.; Ispiryan, L.; Zannini, E.; Arendt, E.K.; Bez, J.; Sørensen, J.C.; Petersen, I.L. Nutritional and anti-nutritional properties of lentil (Lens culinaris) protein isolates prepared by pilot-scale processing. Food Chem. X 2021, 9, 100112. [Google Scholar] [CrossRef]
- Tang, C.H. Functional properties and in vitro digestibility of buckwheat protein products: Influence of processing. J. Food Eng. 2007, 82, 568–576. [Google Scholar] [CrossRef]
- Lin, Y.; Pangloli, P.; Dia, V.P. Physicochemical, functional and bioactive properties of hempseed (Cannabis sativa L.) meal, a co-product of hempseed oil and protein production, as affected by drying process. Food Chem. 2021, 350, 129188. [Google Scholar] [CrossRef]
- De Almeida, N.M.; De Moura Bell, J.M.L.N.; Johnson, L.A. Properties of soy protein produced by countercurrent, two-stage, enzyme-assisted aqueous extraction. JAOCS J. Am. Oil Chem. Soc. 2014, 91, 1077–1085. [Google Scholar] [CrossRef]
- De Mesa-Stonestreet, N.J.; Alavi, S.; Gwirtz, J. Extrusion-enzyme liquefaction as a method for producing sorghum protein concentrates. J. Food Eng. 2012, 108, 365–375. [Google Scholar] [CrossRef]
- Bhatty, R.S.; Christison, G.I. Composition and nutritional quality of pea (Pisum sativum L.), faba bean (Vicia faba L. spp. minor) and lentil (Lens culinaris Medik.) meals, protein concentrates and isolates. Qual. Plant. Plant Foods Hum. Nutr. 1984, 34, 41–51. [Google Scholar] [CrossRef]
- Vogelsang-O’Dwyer, M.; Petersen, I.L.; Joehnke, M.S.; Sørensen, J.C.; Bez, J.; Detzel, A.; Busch, M.; Krueger, M.; O’Mahony, J.A.; Arendt, E.K.; et al. Comparison of Faba bean protein ingredients produced using dry fractionation and isoelectric precipitation: Techno-functional, nutritional and environmental performance. Foods 2020, 9, 322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Opazo-Navarrete, M.; Schutyser, M.A.I.; Boom, R.M.; Janssen, A.E.M. Effect of pre-treatment on in vitro gastric digestion of quinoa protein (Chenopodium quinoa Willd.) obtained by wet and dry fractionation. Int. J. Food Sci. Nutr. 2018, 69, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Martínez-Velasco, A.; Lobato-Calleros, C.; Hernández-Rodríguez, B.E.; Román-Guerrero, A.; Alvarez-Ramirez, J.; Vernon-Carter, E.J. High intensity ultrasound treatment of faba bean (Vicia faba L.) protein: Effect on surface properties, foaming ability and structural changes. Ultrason. Sonochem. 2018, 44, 97–105. [Google Scholar] [CrossRef]
- Çabuk, B.; Nosworthy, M.G.; Stone, A.K.; Korber, D.R.; Tanaka, T.; House, J.D.; Nickerson, M.T. Effect of fermentation on the protein digestibility and levels of non-nutritive compounds of pea protein concentrate. Food Technol. Biotechnol. 2018, 56, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Liu, Z.; Rui, X.; L’Hocine, L.; Zhang, Q.; Li, W.; Dong, M. Assessment of the effect of lactic acid fermentation on the gastroduodenal digestibility and immunoglobulin e binding capacity of soy proteins: Via an in vitro dynamic gastrointestinal digestion model. Food Funct. 2020, 11, 10467–10479. [Google Scholar] [CrossRef]
- Ge, S.J.; Zhang, L.X. Predigestion of soybean proteins with immobilized trypsin for infant formula. Appl. Biochem. Biotechnol. 1993, 43, 199–209. [Google Scholar] [CrossRef]
- Nguyen, T.T.P.; Bhandari, B.; Cichero, J.; Prakash, S. In vitro digestion of infant formulations with hydrolysed and non-hydrolysed proteins from dairy and soybean. Food Funct. 2016, 7, 4908–4919. [Google Scholar] [CrossRef]
- Kaur, L.; Rutherfurd, S.M.; Moughan, P.J.; Drummond, L.; Boland, M.J. Actinidin enhances protein digestion in the small intestine as assessed using an in vitro digestion model. J. Agric. Food Chem. 2010, 58, 5074–5080. [Google Scholar] [CrossRef]
- Rutherfurd, S.M.; Montoya, C.A.; Zou, M.L.; Moughan, P.J.; Drummond, L.N.; Boland, M.J. Effect of actinidin from kiwifruit (Actinidia deliciosa cv. Hayward) on the digestion of food proteins determined in the growing rat. Food Chem. 2011, 129, 1681–1689. [Google Scholar] [CrossRef]
- Montoya, C.A.; Hindmarsh, J.P.; Gonzalez, L.; Boland, M.J.; Moughan, P.J.; Rutherfurd, S.M. Dietary actinidin from kiwifruit (Actinidia deliciosa cv. Hayward) increases gastric digestion and the gastric emptying rate of several dietary proteins in growing rats. J. Nutr. 2014, 144, 440–446. [Google Scholar] [CrossRef] [PubMed]
- Aryee, A.N.A.; Boye, J.I. Improving the Digestibility of Lentil Flours and Protein Isolate and Characterization of Their Enzymatically Prepared Hydrolysates. Int. J. Food Prop. 2016, 19, 2649–2665. [Google Scholar] [CrossRef]
- Fleddermann, M.; Fechner, A.; Rößler, A.; Bähr, M.; Pastor, A.; Liebert, F.; Jahreis, G. Nutritional evaluation of rapeseed protein compared to soy protein for quality, plasma amino acids, and nitrogen balance—A randomized cross-over intervention study in humans. Clin. Nutr. 2013, 32, 519–526. [Google Scholar] [CrossRef] [PubMed]
- Singh, T.P.; Siddiqi, R.A.; Sogi, D.S. Enzymatic modification of rice bran protein: Impact on structural, antioxidant and functional properties. LWT 2021, 138, 110648. [Google Scholar] [CrossRef]
- Clemente, A.; Vioque, J.; Sánchez-Vioque, R.; Pedroche, J.; Bautista, J.; Millán, F. Protein quality of chickpea (Cicer arietinum L.) protein hydrolysates. Food Chem. 1999, 67, 269–274. [Google Scholar] [CrossRef]
- Goertzen, A.D.; House, J.D.; Nickerson, M.T.; Tanaka, T. The impact of enzymatic hydrolysis using three enzymes on the nutritional properties of a chickpea protein isolate. Cereal Chem. 2021, 98, 275–284. [Google Scholar] [CrossRef]
- Tian, T.; Teng, F.; Zhang, S.; Qi, B.; Wu, C.; Zhou, Y.; Li, L.; Wang, Z.; Li, Y. A Study of Structural Change During In Vitro Digestion of Heated Soy Protein Isolates. Foods 2019, 8, 594. [Google Scholar] [CrossRef] [Green Version]
- Chen, F.P.; Li, B.S.; Tang, C.H. Nanocomplexation between Curcumin and Soy Protein Isolate: Influence on Curcumin Stability/Bioaccessibility and in Vitro Protein Digestibility. J. Agric. Food Chem. 2015, 63, 3559–3569. [Google Scholar] [CrossRef]
- Ren, C.; Xiong, W.; Peng, D.; He, Y.; Zhou, P.; Li, J.; Li, B. Effects of thermal sterilization on soy protein isolate/polyphenol complexes: Aspects of structure, in vitro digestibility and antioxidant activity. Food Res. Int. 2018, 112, 284–290. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, S.; Qi, B.; Sui, X.; Jiang, L. Complexation of thermally-denatured soybean protein isolate with anthocyanins and its effect on the protein structure and in vitro digestibility. Food Res. Int. 2018, 106, 619–625. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.A.; Barbeau, W.E. Changes in the nutritive value of soy protein concentrate during autoclaving. Plant Foods Hum. Nutr. 1991, 41, 179–192. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.-H.; Chen, L.; Ma, C.Y. Thermal aggregation, amino acid composition and in vitro digestibility of vicilin-rich protein isolates from three Phaseolus legumes: A comparative study. Food Chem. 2009, 113, 957–963. [Google Scholar] [CrossRef]
- Sathe, S.K.; Deshpande, S.S.; Salunkhe, D.K. Functional Properties of Lupin Seed (Lupinus mutabilis) Proteins and Protein Concentrates. J. Food Sci. 1982, 47, 491–497. [Google Scholar] [CrossRef]
- Sathe, S.K.; Deshpande, S.S.; Salunkhe, D.K. Functional Properties of Winged Bean [Psophocarpus tetragonolobus (L.) DC] Proteins. J. Food Sci. 1982, 47, 503–509. [Google Scholar] [CrossRef]
- Carbonaro, M.; Grant, G.; Cappelloni, M. Heat-induced denaturation impairs digestibility of legume (Phaseolus vulgaris L and Vicia faba L) 7S and 11S globulins in the small intestine of rat. J. Sci. Food Agric. 2005, 85, 65–72. [Google Scholar] [CrossRef]
- Tavano, O.L.; Neves, V.A. Isolation, solubility and in vitro hydrolysis of chickpea vicilin-like protein. LWT Food Sci. Technol. 2008, 41, 1244–1251. [Google Scholar] [CrossRef]
- Neves, V.A.; Lourenço, E.J. Isolation and in vitro hydrolysis of globulin G1 from lentils (lens culinaris, medik). J. Food Biochem. 1995, 19, 109–120. [Google Scholar] [CrossRef]
- Rivera del Rio, A.; Opazo-Navarrete, M.; Cepero-Betancourt, Y.; Tabilo-Munizaga, G.; Boom, R.M.; Janssen, A.E.M. Heat-induced changes in microstructure of spray-dried plant protein isolates and its implications on in vitro gastric digestion. LWT 2020, 118, 108795. [Google Scholar] [CrossRef]
- Pelgrom, P.J.; Berghout, J.A.; van der Goot, A.J.; Boom, R.M.; Schutyser, M.A. Preparation of functional lupine protein fractions by dry separation. LWT Food Sci. Technol. 2014, 59, 680–688. [Google Scholar] [CrossRef]
- Opazo-Navarrete, M.; Tagle Freire, D.; Boom, R.M.; Janssen, A.E.M. The Influence of Starch and Fibre on In Vitro Protein Digestibility of Dry Fractionated Quinoa Seed (Riobamba Variety). Food Biophys. 2019, 14, 49–59. [Google Scholar] [CrossRef] [Green Version]
- Chang, K.C.; Satterlee, L.D. Isolation and Characterization of the Major Protein from Great Northern Beans (Phaseolus vulgaris). J. Food Sci. 1981, 46, 1368–1373. [Google Scholar] [CrossRef]
- Wu, W.; Hettiarachchy, N.S.; Kalapathy, U.; Williams, W.P. Functional properties and nutritional quality of alkali- and heat-treated soy protein isolate. J. Food Qual. 1999, 22, 119–133. [Google Scholar] [CrossRef]
- Savoie, L.; Parent, G.; Galibois, I. Effects of alkali treatment on the in-vitro digestibility of proteins and the release of amino acids. J. Sci. Food Agric. 1991, 56, 363–372. [Google Scholar] [CrossRef]
- Sarwar, G.; L’Abbé, M.R.; Trick, K.; Botting, H.G.; Ma, C.Y. Influence of feeding alkaline/heat processed proteins on growth and protein and mineral status of rats. Adv. Exp. Med. Biol. 1999, 459, 161–177. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Mu, T.; Zhang, M.; Arogundade, L.A. Nutritional assessment and effects of heat processing on digestibility of Chinese sweet potato protein. J. Food Compos. Anal. 2012, 26, 104–110. [Google Scholar] [CrossRef]
- Laguna, L.; Picouet, P.; Guàrdia, M.D.; Renard, C.M.G.C.; Sarkar, A. In vitro gastrointestinal digestion of pea protein isolate as a function of pH, food matrices, autoclaving, high-pressure and re-heat treatments. LWT Food Sci. Technol. 2017, 84, 511–519. [Google Scholar] [CrossRef]
- Yin, S.-W.; Tang, C.-H.; Wen, Q.-B.; Yang, X.-Q.; Li, L. Functional properties and in vitro trypsin digestibility of red kidney bean (Phaseolus vulgaris L.) protein isolate: Effect of high-pressure treatment. Food Chem. 2008, 110, 938–945. [Google Scholar] [CrossRef]
- Mariniello, L.; Giosafatto, C.V.L.; Di Pierro, P.; Sorrentino, A.; Porta, R. Synthesis and Resistance to in Vitro Proteolysis of Transglutaminase Cross-Linked Phaseolin, the Major Storage Protein from Phaseolus vulgaris. J. Agric. Food Chem. 2007, 55, 4717–4721. [Google Scholar] [CrossRef]
- Strauch, R.C.; Lila, M.A. Pea protein isolate characteristics modulate functional properties of pea protein–cranberry polyphenol particles. Food Sci. Nutr. 2021, 9, 3740–3751. [Google Scholar] [CrossRef]
- Tang, C.H.; Li, L.; Yang, X.Q. Influence of transglutaminase-induced cross-linking on in vitro digestibility of soy protein isolate. J. Food Biochem. 2006, 30, 718–731. [Google Scholar] [CrossRef]
- Tang, C.-H.; Sun, X.; Yin, S.-W.; Ma, C.-Y. Transglutaminase-induced cross-linking of vicilin-rich kidney protein isolate: Influence on the functional properties and in vitro digestibility. Food Res. Int. 2008, 41, 941–947. [Google Scholar] [CrossRef]
- Li, Y.; Damodaran, S. In vitro digestibility and IgE reactivity of enzymatically cross-linked heterologous protein polymers. Food Chem. 2017, 221, 1151–1157. [Google Scholar] [CrossRef] [PubMed]
- Sheng, W.W.; Zhao, X.H. Functional properties of a cross-linked soy protein-gelatin composite towards limited tryptic digestion of two extents. J. Sci. Food Agric. 2013, 93, 3785–3791. [Google Scholar] [CrossRef]
- Fu, M.; Zhao, X.H. Modified properties of a glycated and cross-linked soy protein isolate by transglutaminase and an oligochitosan of 5 kDa. J. Sci. Food Agric. 2017, 97, 58–64. [Google Scholar] [CrossRef]
- Zhu, C.Y.; Liu, H.F.; Fu, M.; Zhao, X.H. Structure and property changes of soybean protein isolates resulted from the glycation and cross-linking by transglutaminase and a degraded chitosan. CYTA J. Food 2016, 14, 138–144. [Google Scholar] [CrossRef] [Green Version]
- Gan, C.Y.; Cheng, L.H.; Azahari, B.; Easa, A.M. In-vitro digestibility and amino acid composition of soy protein isolate cross-linked with microbial transglutaminase followed by heating with ribose. Int. J. Food Sci. Nutr. 2009, 60, 99–108. [Google Scholar] [CrossRef]
- Yin, S.W.; Tang, C.H.; Wen, Q.B.; Yang, X.Q. Effects of acylation on the functional properties and in vitro trypsin digestibility of red kidney bean (Phaseolus vulgaris L.) protein isolate. J. Food Sci. 2009, 74, E488–E494. [Google Scholar] [CrossRef]
- El-Adawy, T.A. Functional properties and nutritional quality of acetylated and succinylated mung bean protein isolate. Food Chem. 2000, 70, 83–91. [Google Scholar] [CrossRef]
- Achouri, A.; Zhang, W. Effect of succinylation on the physicochemical properties of soy protein hydrolysate. Food Res. Int. 2001, 34, 507–514. [Google Scholar] [CrossRef]
- De Regil, L.M.; Calderón de la Barca, A.M. Nutritional and technological evaluation of an enzymatically methionine-enriched soy protein for infant enteral formulas. Int. J. Food Sci. Nutr. 2004, 55, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Budryn, G.; Zaczyńska, D.; Rachwał-Rosiak, D.; Oracz, J. Changes in properties of food proteins after interaction with free and β-cyclodextrin encapsulated hydroxycinnamic acids. Eur. Food Res. Technol. 2015, 240, 1157–1166. [Google Scholar] [CrossRef] [Green Version]
- Rohn, S.; Petzke, K.J.; Rawel, H.M.; Kroll, J. Reactions of chlorogenic acid and quercetin with a soy protein isolate—Influence on the in vivo food protein quality in rats. Mol. Nutr. Food Res. 2006, 50, 696–704. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Bao, Z.; Wu, N.; Yang, X.; Lin, W.; Chen, Z.; Wang, J.; Guo, J. Preparation and characterisation of soya milk enriched with isoflavone aglycone fermented by lactic acid bacteria combined with hydrothermal cooking pretreatment. Int. J. Food Sci. Technol. 2015, 50, 1331–1337. [Google Scholar] [CrossRef]
- Zhao, J.; Su, G.; Chen, C.; Sun, X.; Sun, W.; Zhao, M. Physicochemical and Structural Characteristics of Soybean Protein Isolates Induced by Lipoxygenase-Catalyzed Linoleic Acid Oxidation during in Vitro Gastric Digestion. J. Agric. Food Chem. 2020, 68, 12384–12392. [Google Scholar] [CrossRef]
- Chen, N.; Zhao, M.; Sun, W. Effect of protein oxidation on the in vitro digestibility of soy protein isolate. Food Chem. 2013, 141, 3224–3229. [Google Scholar] [CrossRef]
- Sánchez-Vioque, R.; Vioque, J.; Clemente, A.; Pedroche, J.; Bautista, J.; Millán, F. Interaction of chickpea (Cicer arietinum L.) legumin with oxidized linoleic acid. J. Agric. Food Chem. 1999, 47, 813–818. [Google Scholar] [CrossRef]
- Duque-Estrada, P.; Berton-Carabin, C.C.; Nieuwkoop, M.; Dekkers, B.L.; Janssen, A.E.M.; van der Goot, A.J. Protein Oxidation and In Vitro Gastric Digestion of Processed Soy-Based Matrices. J. Agric. Food Chem. 2019, 67, 9591–9600. [Google Scholar] [CrossRef] [Green Version]
- Chen, N.; Zhao, Q.; Sun, W.; Zhao, M. Effects of malondialdehyde modification on the in vitro digestibility of soy protein isolate. J. Agric. Food Chem. 2013, 61, 12139–12145. [Google Scholar] [CrossRef]
- Rhee, K.S.; Rhee, K.C. Nutritional Evaluation of the Protein in Oilseed Products Heated with Sugars. J. Food Sci. 1981, 46, 164–168. [Google Scholar] [CrossRef]
- Wang, H.; Chen, Y.; Hua, Y.; Kong, X.; Zhang, C. Effects of phytase-assisted processing method on physicochemical and functional properties of soy protein isolate. J. Agric. Food Chem. 2014, 62, 10989–10997. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Bhirud, P.R.; Tyler, R.T. Extrusion texturization of air-classified pea protein. J. Food Sci. 1999, 64, 509–513. [Google Scholar] [CrossRef]
- Omosebi, M.O.; Osundahunsi, O.F.; Fagbemi, T.N. Effect of extrusion on protein quality, antinutritional factors, and digestibility of complementary diet from quality protein maize and soybean protein concentrate. J. Food Biochem. 2018, 42, e12508. [Google Scholar] [CrossRef]
- Li, S.; Jiang, Z.; Wang, F.; Wu, J.; Liu, Y.; Li, X. Characterization of rice glutelin fibrils and their effect on in vitro rice starch digestibility. Food Hydrocoll. 2020, 106, 105918. [Google Scholar] [CrossRef]
- Opazo-Navarrete, M.; Altenburg, M.D.; Boom, R.M.; Janssen, A.E.M. The Effect of Gel Microstructure on Simulated Gastric Digestion of Protein Gels. Food Biophys. 2018, 13, 124–138. [Google Scholar] [CrossRef]
- Hall, A.E.; Moraru, C.I. Effect of High Pressure Processing and heat treatment on in vitro digestibility and trypsin inhibitor activity in lentil and faba bean protein concentrates. LWT 2021, 152, 112342. [Google Scholar] [CrossRef]
- Rui, X.; Fu, Y.; Zhang, Q.; Li, W.; Zare, F.; Chen, X.; Jiang, M.; Dong, M. A comparison study of bioaccessibility of soy protein gel induced by magnesiumchloride, glucono-δ-lactone and microbial transglutaminase. LWT Food Sci. Technol. 2016, 71, 234–242. [Google Scholar] [CrossRef]
- Ou, S.; Kwok, K.C.; Kang, Y. Changes in in vitro digestibility and available lysine of soy protein isolate after formation of film. J. Food Eng. 2004, 64, 301–305. [Google Scholar] [CrossRef]
- Hu, B.; Chen, Q.; Cai, Q.; Fan, Y.; Wilde, P.J.; Rong, Z.; Zeng, X. Gelation of soybean protein and polysaccharides delays digestion. Food Chem. 2017, 221, 1598–1605. [Google Scholar] [CrossRef]
- Huang, Z.; Gruen, I.; Vardhanabhuti, B. Intragastric Gelation of Heated Soy Protein Isolate-Alginate Mixtures and Its Effect on Sucrose Release. J. Food Sci. 2018, 83, 1839–1846. [Google Scholar] [CrossRef]
- Khalesi, M.; Fitzgerald, R.J. In vitro digestibility and antioxidant activity of plant protein isolate and milk protein concentrate blends. Catalysts 2021, 11, 787. [Google Scholar] [CrossRef]
- Baugreet, S.; Gomez, C.; Auty, M.A.E.; Kerry, J.P.; Hamill, R.M.; Brodkorb, A. In vitro digestion of protein-enriched restructured beef steaks with pea protein isolate, rice protein and lentil flour following sous vide processing. Innov. Food Sci. Emerg. Technol. 2019, 54, 152–161. [Google Scholar] [CrossRef]
- Oñate Narciso, J.; Brennan, C. Whey and Pea Protein Fortification of Rice Starches: Effects on Protein and Starch Digestibility and Starch Pasting Properties. Starch/Staerke 2018, 70, 1700315. [Google Scholar] [CrossRef]

 represents centrifugation after alkaline extraction and isoelectric precipitation.
represents centrifugation after alkaline extraction and isoelectric precipitation.
 represents centrifugation after alkaline extraction and isoelectric precipitation.
represents centrifugation after alkaline extraction and isoelectric precipitation.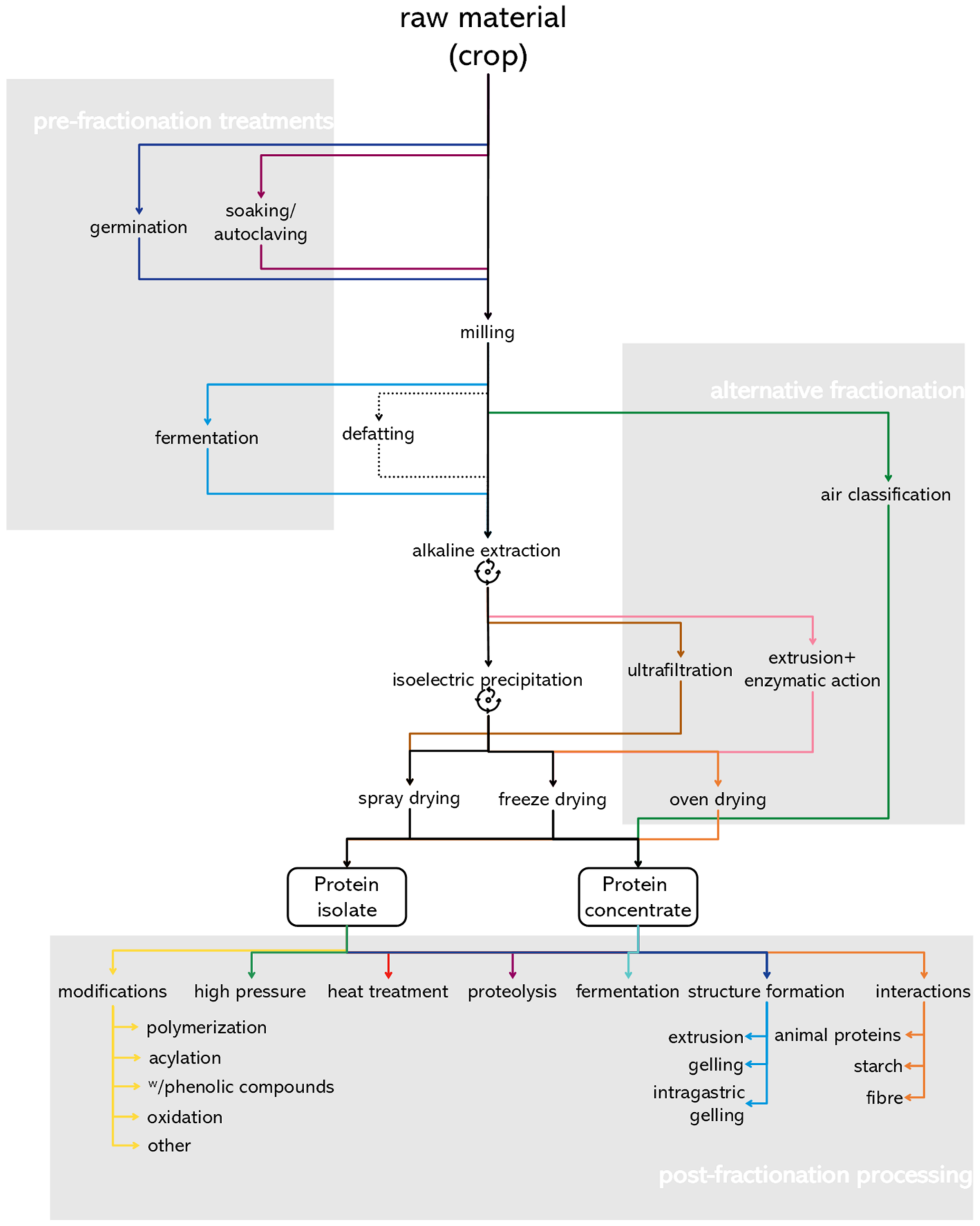
 ,
,  negative;
negative;  ,
,  positive; or
positive; or  neutral effect on protein digestibility. Only processes or ingredients with more than one study reporting on the effect of processing on digestibility were included in this table.
neutral effect on protein digestibility. Only processes or ingredients with more than one study reporting on the effect of processing on digestibility were included in this table.
 ,
,  negative;
negative;  ,
,  positive; or
positive; or  neutral effect on protein digestibility. Only processes or ingredients with more than one study reporting on the effect of processing on digestibility were included in this table.
neutral effect on protein digestibility. Only processes or ingredients with more than one study reporting on the effect of processing on digestibility were included in this table.| Crop | Pre-Fractionation | Fractionation | Processing | Nutrient Interactions Starch | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Germination | Dry Fractionation | Alkaline Treatment | Fermentation | Enzymatic Hydrolysis | Heating | High Pressure | Polymerization | Acylation | Phenolic Compounds | Oxidation | Gelling | Extrusion | ||
| soybean |  | - |  also post-fractionation |  |  |  | - |  |  hydrolysate |  |  |  |  | - |
| black bean |  | - | - | - | - | - | - |  | - | - | - | - | - | - |
| chickpea | - | - | - | - |  |  | - | - | - | - |  | - | - | - |
| fava bean | - |  | - | - | - |  legumin | - | - | - | - | - | - | - | - |
| lentil | - |  | - | - |  |  globulin | - | - | - | - | - | - | - | - |
| lupin | - | - |  |  pre-fractionation | - |  | - | - | - | - | - | - | - | - |
| maize | - | - | - | - |  | - | - | - | - | - | - | - |  | - |
| mung bean | - | - | - | - | - |  | - | - |  | - | - | - | - | - |
| navy bean | - | - |  | - | - |  | - | - | - | - | - | - | - | |
| quinoa | - |  |  | - | - |  | - | - | - | - | - | - | - |  |
| rapeseed | - | - |  | - |  | - | - | - | - | - | - | - | - | - |
| red kidney bean | - | - | - | - | - |  |  |  |  | - | - | - | - | - |
| rice | - | - |  | - |  | - | - | - | - | - | - | - | - | - |
| yellow pea | - |  | - |  | - |  |  | - | - |  | - |  |  |  |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rivera del Rio, A.; Boom, R.M.; Janssen, A.E.M. Effect of Fractionation and Processing Conditions on the Digestibility of Plant Proteins as Food Ingredients. Foods 2022, 11, 870. https://doi.org/10.3390/foods11060870
Rivera del Rio A, Boom RM, Janssen AEM. Effect of Fractionation and Processing Conditions on the Digestibility of Plant Proteins as Food Ingredients. Foods. 2022; 11(6):870. https://doi.org/10.3390/foods11060870
Chicago/Turabian StyleRivera del Rio, Andrea, Remko M. Boom, and Anja E. M. Janssen. 2022. "Effect of Fractionation and Processing Conditions on the Digestibility of Plant Proteins as Food Ingredients" Foods 11, no. 6: 870. https://doi.org/10.3390/foods11060870
APA StyleRivera del Rio, A., Boom, R. M., & Janssen, A. E. M. (2022). Effect of Fractionation and Processing Conditions on the Digestibility of Plant Proteins as Food Ingredients. Foods, 11(6), 870. https://doi.org/10.3390/foods11060870






