Dry but Not Humid Thermal Processing of Aloe vera Gel Promotes Cytotoxicity on Human Intestinal Cells HT-29
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material Extraction
2.2. Molecular Identification through DNA Barcoding
2.3. Alcohol Insoluble Residues
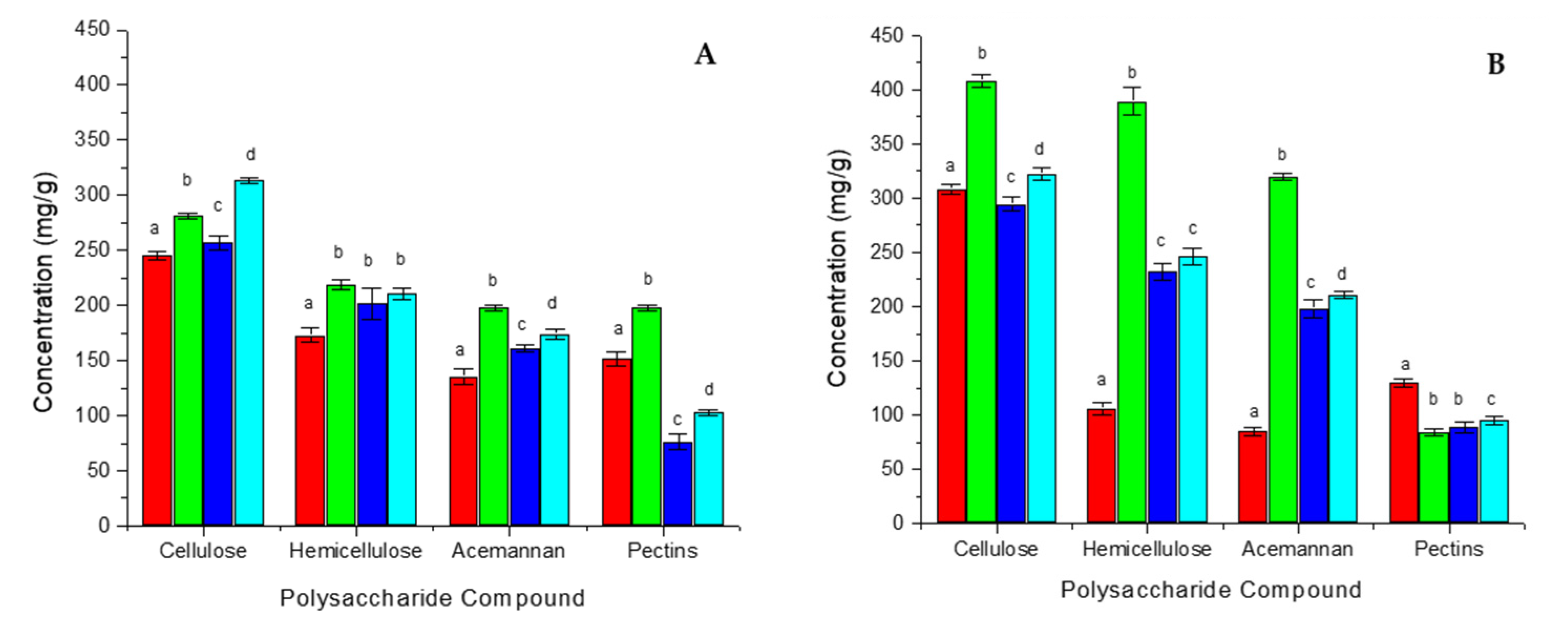
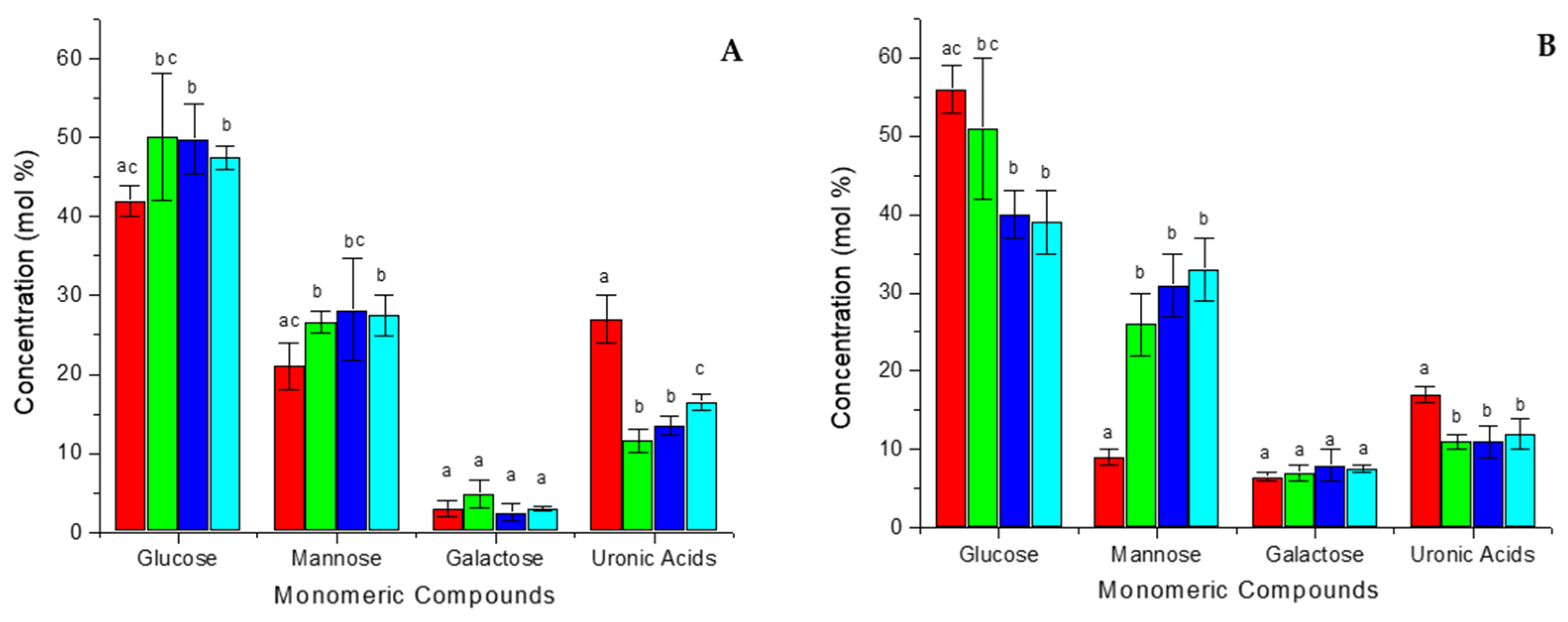
2.4. Analysis of Carbohydrate Composition
2.5. Degree of Acetylation of Acemannan by Nuclear Magnetic Resonance (NMR) Analysis
2.6. Culture Conditions
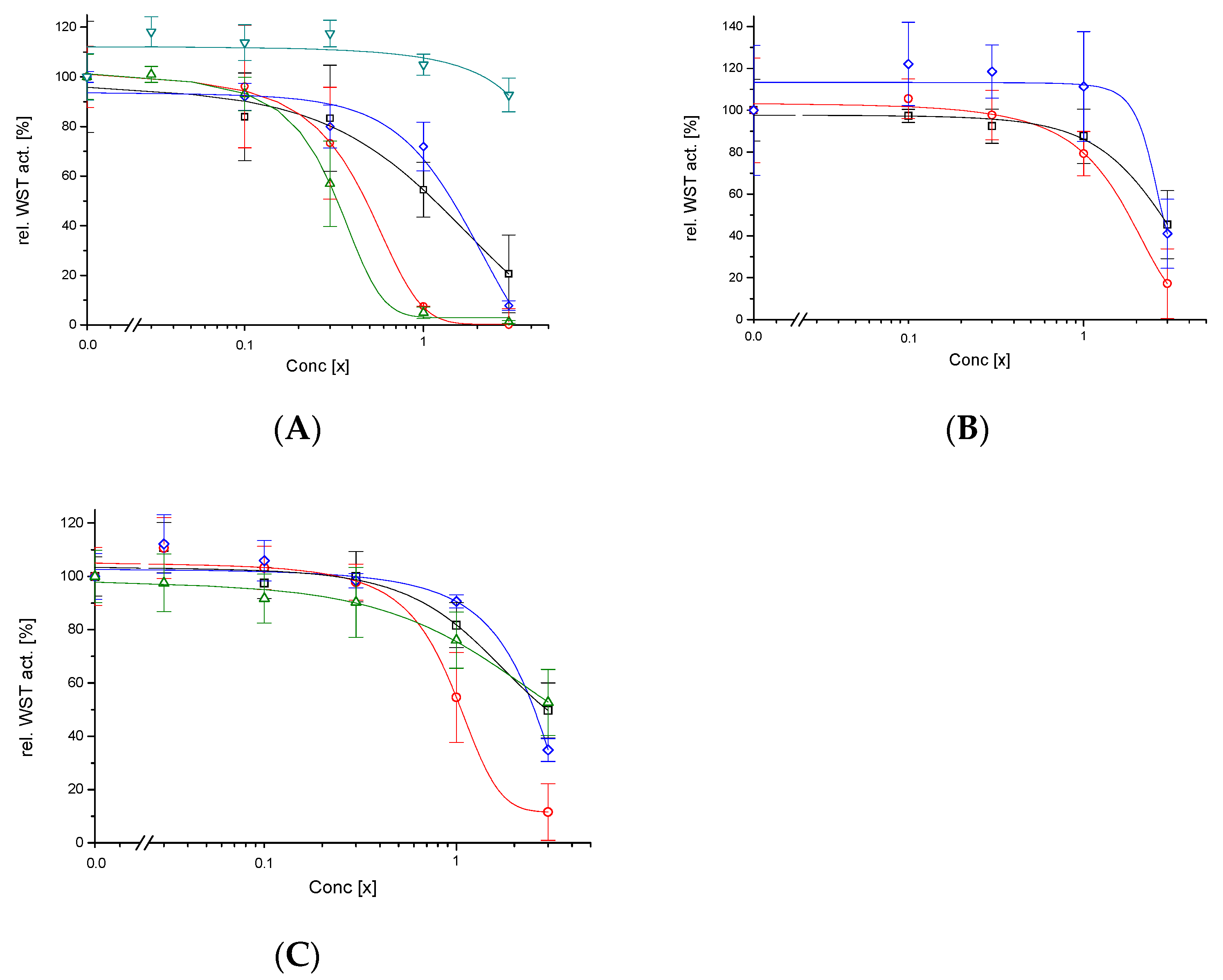
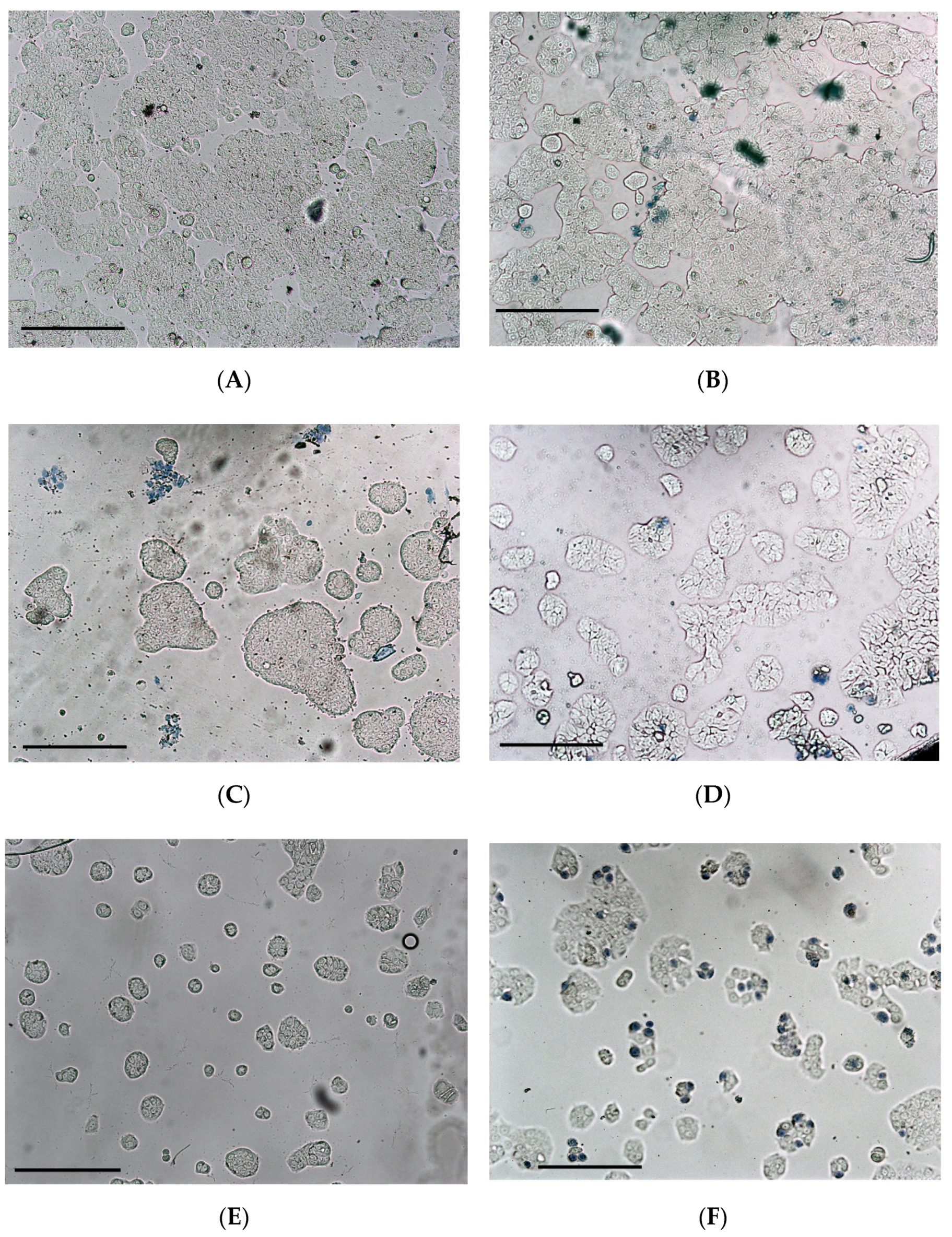
2.7. Cell Viability and Necrosis
2.8. Statistical Analysis
3. Results
3.1. Molecular Identification
3.2. Analysis of Carbohydrate Composition
3.3. Cytotoxicity
3.4. Polysaccharide Modifications
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AAG | AIR-fraction from DAG |
| AILF | AIR-fraction from ILF |
| AIR | Alcohol Insoluble Residue |
| CC50 | Median Cytotoxic Concentration |
| D2O | Heavy water |
| DAG | Dehydrated Aloe vera Gel |
| ILF | Aloe vera Inner Leaf gel, high Fiber content |
| PAG | Pasteurized Aloe vera Gel |
| WST | Water-Soluble Tetrazolium salt (2-(4-iodophenyl)-3-(4-nitrophenyl)-5-(2,4-disulfo-phenyl)-2H-tetrazolium) |
References
- Upton, R.; Axentiev, P. Aloe vera leaf, Aloe vera leaf juice, Aloe vera inner leaf juice. In Standards of Identity, Analysis and Quality Control, 1st ed.; American Herbal Pharmacopoeia: Scotts Valley, CA, USA, 2012; pp. 1–55. [Google Scholar]
- Van Gorkom, B.A.P.; de Vries, E.G.E.; Karrenbeld, A.; Kleibeuker, J.H. Review article: Anthranoid laxatives and their potential carcinogenic effects. Aliment. Pharmacol. Ther. 1999, 13, 443–452. [Google Scholar] [CrossRef] [PubMed]
- Boudreau, M.D.; Mellick, P.W.; Olson, G.R.; Felton, R.P.; Thorn, B.T.; Beland, F.A. Clear evidence of carcinogenic activity by whole-leaf extract of Aloe barbadensis Miller (Aloe vera) in F344/N rats. Toxicol. Sci. 2013, 131, 26–39. [Google Scholar] [CrossRef]
- Femenia, A.; Sánchez, E.S.; Simal, S.; Rosselló, C. Compositional features of polysaccharides from Aloe vera (Aloe barbadensis Miller) plant tissues. Carbohydr. Polym. 1999, 39, 109–117. [Google Scholar] [CrossRef]
- López, Z.; Femenia, A.; Núñez-Jinez, G.; Salazar-Zúñiga, M.N.; Cano, E.M.; Espino, T.; Knauth, P. In vitro imunomodulatory effect of food supplement from Aloe vera. Evid. Based Complement. Alternat. Med. 2019, 2019, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Budai, M.M.; Varga, A.; Milez, S.; Tözsér, J.; Benkö, S. Aloe vera downregulates LPS-induced inflammatory cytokine production and expression of NLRP3 inflammasome in human macrophages. Mol. Immunol. 2013, 56, 471–479. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Minjares-Fuentes, J.R.; Femenia, A. Effect of processing on the bioactive polysaccharides and phenolic compounds from Aloe vera (Aloe barbadensis Miller). In Dietary Fiber Functionality in Food and Nutraceuticals: From Plant to Gut; Hosseinian, F., Oomah, B.D., Campos-Vega, R., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2017; pp. 263–287. [Google Scholar]
- Minjares-Fuentes, R.; Femenia, A.; Comas-Serra, F.; Rodriguez-González, V.M. Compositional and structural features of the main bioactive polysaccharide present in the Aloe vera plant. J. AOAC Int. 2018, 101, 1711–1719. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, T. The compounds in Aloe leaf exudates: A review. Bot. J. Linn. Soc. 1985, 90, 157–177. [Google Scholar] [CrossRef]
- Simões, J.; Nunes, F.M.; Domingues, P.; Coimbra, M.A.; Domingues, M.R. Mass spectroscopy characterization of an Aloe vera mannan presenting immunostimulatory activity. Carbohydr. Polym. 2012, 90, 229–236. [Google Scholar] [CrossRef]
- Chang, X.L.; Wang, C.; Feng, Y.; Liu, Z. Effects of heat treatments on the stabilities of polysaccharides substances and barbaloin in gel juice from Aloe vera Miller. J. Food Eng. 2006, 75, 245–251. [Google Scholar] [CrossRef]
- Minjares-Fuentes, R.; Rodríguez-González, V.M.; González-Laredo, R.F.; Eim, V.; González-Centeno, M.R.; Femenia, A. Effect of different drying procedures on the bioactive polysaccharide acemannan from Aloe vera (Aloe barbadensis Miller). Carbohydr. Polym. 2017, 168, 327–336. [Google Scholar] [CrossRef]
- Femenia, A.; García-Pascual, P.; Simal, S.; Rosselló, C. Effects of heat treatment and dehydration on bioactive polysaccharide acemannan and cell wall polymers from Aloe barbadensis Miller. Carbohydr. Polym. 2003, 51, 397–405. [Google Scholar] [CrossRef]
- Rodríguez-González, V.M.; Femenia, A.; González-Laredo, R.F.; Rocha-Guzmán, N.E.; Gallegos-Infante, J.A.; Candelas-Cadillo, M.G.; Ramírez-Baca, P.; Simal, S.; Rosselló, C. Effects of pasteurization on bioactive polysaccharide acemannan and cell wall polymers from Aloe barbadensis Miller. Carbohydr. Polym. 2011, 86, 1675–1683. [Google Scholar] [CrossRef]
- Sriariyakul, W.; Swasdisevi, T.; Devahastin, S.; Soponronnarit, S. Drying of Aloe vera puree using hot air in combination with far-infrared radiation and high-voltage electric field: Drying kinetics, erergy consumption and product quality evaluation. Food Bioprod. Process. 2016, 100, 391–400. [Google Scholar] [CrossRef]
- Liu, C.; Cui, Y.; Pi, F.; Cheng, Y.; Guo, Y.; Qian, H. Extraction, purificarion, structural characteristics, biological activities and pharmacological applications of acemannan, a polysaccharide from Aloe vera: A review. Molecules 2019, 24, 21. [Google Scholar]
- Chokboribal, J.; Tachaboonyakiat, W.; Sangvanich, P.; Ruangpornvisuti, V.; Jettanacheawchankit, S.; Thunyakitpisal, P. Deacetylation affects the physical properties and bioactivity of acemannan, an extracted polysaccharide from Aloe vera. Carbohydr. Polym. 2015, 133, 556–566. [Google Scholar] [CrossRef]
- Hussain, S.A.; Yadav, V.; Reddi, S.; Patil, G.R.; Singh, R.R.B.; Kapila, S. Thermal processing conditions affect in vitro immunostimulatory activity of Aloe vera juice. J. Appl. Res. Med. Aromat. Plants 2019, 12, 73–77. [Google Scholar] [CrossRef]
- Salah, F.; El-Ghoul, Y.; Madhdhi, A.; Majdoub, H.; Jarroux, N.; Sakli, F. Effect of the deacetylation degree on the antibacterial and biofilm activity of acemannan from Aloe vera. Ind. Crops Prod. 2017, 103, 13–18. [Google Scholar] [CrossRef]
- Guo, X.; Mei, N. Aloe vera: A review of toxicity and adverse clinical effects. J. Environ. Sci. Health Pt. C 2016, 34, 77–96. [Google Scholar] [CrossRef]
- du Plessis, L.H.; Hamman, J.H. In vitro evaluation of the cytotoxic and apoptogenic properties of Aloe whole leaf and gel materials. Drug Chem. Toxicol. 2014, 37, 169–177. [Google Scholar] [CrossRef]
- López, Z.; Núñez-Jinez, G.; Avalos-Navarro, G.; Rivera, G.; Salazar-Flores, J.; Ramírez, J.A.; Ayil-Gutiérrez, B.A.; Knauth, P. Antioxidant and cytotoxicological effects of Aloe vera food supplements. J. Food Qual. 2017, 2017, 10. [Google Scholar] [CrossRef] [Green Version]
- CBOL Plant Working Group; Hollingsworth, P.M.; Forrest, L.L.; Spouge, J.L.; Hajibabaei, M.; Ratnasingham, S.; van der Bank, M.; Chase, M.W.; Cowan, R.S.; Erickson, D.L.; et al. A DNA barcode for land plants. Proc. Nat. Acad. Sci. USA 2009, 106, 12794–12797. [Google Scholar]
- Rosenthal, A.; Coutelle, O.; Craxton, M. Large-scale production of DNA sequencing templates by microtitre format PCR. Nucleic Acids Res. 1993, 21, 173–174. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Knauth, P.; Acevedo-Hernández, G.J.; Cano, M.E.; Gutiérrez-Lomelí, M.; López, Z. In vitro bioactivity of methanolic extracts from Amphipterygium adstringens (Schltdl. Schiede ex Standl.), Chenopodium ambrosioides (L.), Cirsium mexicanum (DC.), Eryngium carlinae (F. Delaroche) and Pithecellobium dulce (Roxb. Benth.) used in traditional medicine in Mexico. Evid. Based Complement. Alternat. Med. 2018, 2018, 11. [Google Scholar]
- Waldron, K.W.; Selvendran, R.R. Composition of the cell wall of different asparagus (Asparagus officinalis) tissues. Physiol. Plant. 1990, 80, 568–575. [Google Scholar] [CrossRef]
- Minjares-Fuentes, R.; Medina-Torres, L.; González-Laredo, R.F.; Rodríguez-González, V.M.; Eim, V.; Femenia, A. Influence of water deficit on the main polysaccharides and the rheological properties of Aloe vera (Aloe barbadensis Miller) mucilage. Ind. Crops Prod. 2017, 109, 644–653. [Google Scholar] [CrossRef]
- Saeman, J.F.; Moore, W.E.; Mitchell, R.I.; Millett, M.A. Techniques for the determination of pulp constituents by quantitative paper chromatography. Tappi J. 1954, 37, 336–343. [Google Scholar]
- Blumenkrantz, N.; Asboe-Hansen, G. New method for quantitative determination of uronic acids. Anal. Biochem. 1973, 54, 484–492. [Google Scholar] [CrossRef]
- Bozzi, A.; Perrin, C.; Austin, S.; Arce-Vera, F. Quality and authenticity of commercial Aloe vera gel powders. Food Chem. 2007, 103, 22–30. [Google Scholar] [CrossRef]
- Jiao, P.; Jia, Q.; Randel, G.; Diehl, B.; Weaver, S.; Milligan, G. Quantitative 1H-NMR spectrometry method for quality control of Aloe vera products. J. AOAC Int. 2010, 93, 842–848. [Google Scholar]
- Yi, Y.; Xu, W.; Wang, H.-X.; Huang, F.; Wang, L.-E. Natural polysaccharides experience physiochemical and functional changes during preparation: A review. Carbohydr. Polym. 2020, 234, 18. [Google Scholar] [CrossRef]
- Han, M.M.; Yi, Y.; Wang, H.X.; Huang, F. Investigation of the Maillard reaction between polysaccharides and proteins from longan pulp and the improvement in activities. Molecules 2017, 22, 938. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Somjai, C.; Siriwoharn, T.; Kulprachakarn, K.; Chaipoot, S.; Phongphisutthinant, R.; Wiriyacharee, P. Utilization of Maillard reaction in moist-dry-heating system to enhance physicochemical and antioxidative properties of dried whole longan fruit. Heliyon 2021, 7, e07094. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Kumar, R. Role of acemannan O-acetyl group in murine radioprotection. Carbohydr. Polym. 2019, 207, 460–470. [Google Scholar] [CrossRef] [PubMed]
- Dotzel, M.M. Status of Certain Additional Over-the-Counter Drug Category II and III Active Ingredients. Final rule, DOCID:fr09my02-7; Docket No. 78N-036L.; RIN 0910-AA01, Department of Health and Human Services (HHS)—Food and Drug Administration (FDA). Fed. Regist. 2002, 67, 31123–31125. [Google Scholar]
- Avila, H.; Rivero, J.; Herrera, F.; Fraile, G. Cytotoxicity of a low molecular weight fraction from Aloe vera (Aloe barbadensis Miller) gel. Toxicon 1997, 35, 1423–1430. [Google Scholar] [CrossRef]

| Samples | Degree of Deacetylation | Deacetylation [%] | Cytotoxicity (CC50) | ||
|---|---|---|---|---|---|
| Dehydrated | AIR | Pasteurized | |||
| Lyophilized Aloe gel | 0.43 ± 0.01 | 0.0 | - | - | - |
| Aloe Gel 60 | 0.37 ± 0.01 | 14.0 | 1.1× | 2.9× | 2.8× |
| Aloe Gel 80 | 0.18 ± 0.01 | 58.1 | 0.4× | 0.9× | 1.6× |
| Aloe Gel 100 | 0.23 ± 0.01 | 46.5 | 1.5× | 2.7× | 2.5× |
| ILF | 0.15 ± 0.01 | 65.1 | 0.3× | >3× | - |
| ILL | 0.08 ± 0.01 | 81.4 | >3× | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
López, Z.; Salazar Zúñiga, M.N.; Femenia, A.; Acevedo-Hernández, G.J.; Godínez Flores, J.A.; Cano, M.E.; Knauth, P. Dry but Not Humid Thermal Processing of Aloe vera Gel Promotes Cytotoxicity on Human Intestinal Cells HT-29. Foods 2022, 11, 745. https://doi.org/10.3390/foods11050745
López Z, Salazar Zúñiga MN, Femenia A, Acevedo-Hernández GJ, Godínez Flores JA, Cano ME, Knauth P. Dry but Not Humid Thermal Processing of Aloe vera Gel Promotes Cytotoxicity on Human Intestinal Cells HT-29. Foods. 2022; 11(5):745. https://doi.org/10.3390/foods11050745
Chicago/Turabian StyleLópez, Zaira, Michelle N. Salazar Zúñiga, Antoni Femenia, Gustavo J. Acevedo-Hernández, Jaime A. Godínez Flores, M. Eduardo Cano, and Peter Knauth. 2022. "Dry but Not Humid Thermal Processing of Aloe vera Gel Promotes Cytotoxicity on Human Intestinal Cells HT-29" Foods 11, no. 5: 745. https://doi.org/10.3390/foods11050745
APA StyleLópez, Z., Salazar Zúñiga, M. N., Femenia, A., Acevedo-Hernández, G. J., Godínez Flores, J. A., Cano, M. E., & Knauth, P. (2022). Dry but Not Humid Thermal Processing of Aloe vera Gel Promotes Cytotoxicity on Human Intestinal Cells HT-29. Foods, 11(5), 745. https://doi.org/10.3390/foods11050745








