Abstract
Seaweeds are traditional food ingredients mainly in seaside regions. Modern food science and nutrition researchers have identified seaweed as a source of functional nutrients, such as dietary soluble and insoluble fibers, proteins, omega-3 fatty acids, prebiotic polysaccharides, polyphenols, and carotenoids. Owing to the rich nutrients, seaweeds and seaweed extract can be used as functional ingredients by modifying the nutrients composition to reduce the proportion of available carbohydrates, delaying the gastric emptying time and the absorption rate of glucose by increasing the digesta viscosity, and attenuating the digesting rate by blocking the activity of digestive enzymes. This review presents the concept of using seaweed as unconventional ingredients that can function synergistically to reduce the glycemic potency of cereal products.
1. Introduction
The prevalence of type 2 diabetes (T2D) has escalated exponentially from 110 million adults in 1994 to 536.6 million in 2021, and it is estimated to reach 578 million by 2030 and 700 million by 2045 [1,2]. The low- and middle-income countries in total contribute to most adult diabetes cases (414.0 million) in 2021 [3]. When the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) emerged in the global pandemic [4], T2D came to be an important risk factor for severe morbidity and mortality related to COVID-19 infection (coronavirus disease-2019) [5,6,7,8,9]. The glycemic management is, therefore, one public priority of the population at a high risk of developing T2D, particularly in Asians who are predisposed to poorer insulin sensitivity and higher postprandial blood glucose excursions compared to Caucasians [10,11,12]. The glycemic index (GI) is a numerical value used to characterize the impact of carbohydrate foods to raise postprandial blood glucose concentration and guide people to choose foods that raise the glucose slowly [13]. The GI value is calculated as the percentage of the area under the 2-h blood glucose response curve (AUC) after consuming food containing 50 g of available carbohydrates in comparison to the AUC generated by 50 g glucose in the same participants [13]. If a food has a GI lower than 56 it is considered to have a low glycemic potency, where a GI between 56–69 is medium, and a GI higher than 69 is high [14]. Adhering to a diet of low glycemic potency (glycemic index (GI) < 56) lowers the acute impact of foods on postprandial glycemic response and is strongly associated with better long-term glycemic management to reduce T2D risk [14].
One challenge regarding glycemic management is that the staple food that accounts for 60% of total daily energy consumption in low- and medium-income countries, such as most of the Asian population, is high GI (GI ≥ 70) refined cereal foods [15,16]. For example, white rice, which has been associated with postprandial hyperglycemia and increased risk of T2D, is the main staple food in the Asian diet [17,18,19]. Although cereals with the lower glycemic potency (i.e., lower GI) is well perceived as a protective dietary factor of the T2D risk [20], people may favor refined cereals with preferred sensory characteristics (i.e., color, odor, texture, and flavor) to the healthy alternative since food habits, culture, and family tradition play a significant role in accepting cereal foods [21]. Replacing refined cereal foods with animal-based foods such as meat and meat products can even further increase the risk of T2D [22] in Asian populations that have already been experiencing a shift towards a modern meat dietary pattern [23,24]. Reducing the glycemic potency of cereal foods becomes an important and practical strategy.
Current strategies to reduce the glycemic potency of cereal foods are commonly achieved through the following mechanisms: (1) reducing the available carbohydrate, (2) reducing the post-consumption digestion rate, (3) reducing post-digestion glucose absorption rate, and (4) increasing postprandial blood glucose clearance rate [25] (Figure 1).
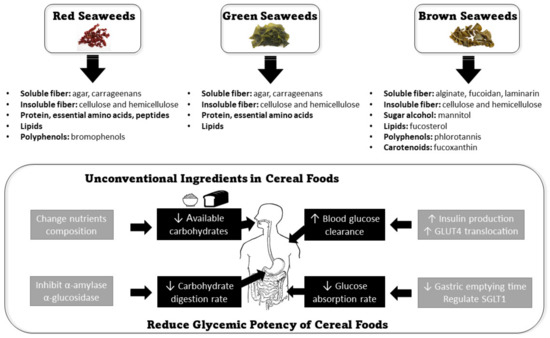
Figure 1.
The applications of seaweeds in reducing the glycemic potency of cereal foods.
Seaweeds, which have been used as food ingredients in some coastal regions [26], are a renewable source of high-value nutrients, including dietary fiber, protein, functional fatty acids, prebiotic polysaccharides, polyphenols, carotenoids, and minerals [27]. They can be re-invented as unconventional ingredients in lowering the glycemic potency of cereal foods by modifying the nutrient composition to reduce the energy contribution from available carbohydrates, attenuating the digesting rate by blocking the activity of digestive enzymes, and delaying the gastric emptying time and the glucose absorption rate by increasing the digesta viscosity. In fact, seaweeds have attracted attention from food industries as functional ingredients to fortify the nutritional composition and health-related beneficial properties in bakery, dairy, fish, meat, and vegetable-based food products [28]. According to the Seafood Source report, the number of new food products containing ingredients derived from seaweeds launched in the European market has increased by 147 per cent in four years (from 2011 to 2015) demonstrating Western countries’ growing interest in these substances [28,29].
The previous reviews and perspective articles reviewed the potential biological properties of various seaweeds as a nutritional supplement in the long-term prevention and treatment of diseases, such as anti-cancer, anti-inflammatory, anti-hypertensive, anti-microbial, anti-viral, anti-obesity, and neuroprotective functions [30]. Many studies have focused on the supplementary value of either individual seaweed extract with high-value bioactive nutrients or comparing the nutritional values between different types of seaweeds [30]. Nevertheless, to the authors’ best knowledge, no current article has collectively presented the strategies of using edible seaweeds as added functional ingredients to reduce carbohydrate-rich food’s glycemic potency or glycemic index. As the contents of bioactive compounds vary between seaweed species, reformulating the high GI or glycemic potency of cereal food products does not need to be limited to one seaweed or one nutrient. Instead, multiple seaweed-derived ingredients can work synergistically to combine the different peculiar compositions of various seaweed species. The present article presents the concept of using various seaweeds as innovative ingredients in cereal products, and that can function synergistically to target different mechanisms to reduce the glycemic potency.
In this review, we also summarize published clinical studies that investigated the effect of seaweeds and seaweed extracts on the acute glycemic response in healthy adults, and we review the potential mechanisms that could explain the beneficial role of seaweeds and seaweeds extract on glucose metabolism. The eligible studies include studies that recruited adults (≥18 years old) reported as healthy with a body mass index (BMI) between 18.5 and 40 kg/m2, parallel and crossover randomized controlled trials (RCTs) that investigated the acute effect of seaweed species and/or extracts on to up to 3-h postprandial blood or plasma glucose, using placebo as control.
Given that the present article is not a systematic review, we may not have identified some studies and publication bias should be acknowledged. However, both authors conducted the literature search independently using a combination of medical subject heading (MeSH) search terms and free text search terms. The search terms include combinations of “healthy”, “adult”, “normal weight and/or overweight and/or obese”, “seaweed and/or seaweed extract”, “placebo”, “blood glucose and/or plasma glucose and/or serum glucose”, “randomized controlled trials”, and “postprandial”. We searched the publications between January 2000 and January 2022.
2. Reducing the Glycemic Potency of Cereal Foods: A Diet Challenge
Cereal foods, which is a group of grains including rice, sorghum, maize, millet, barley, buckwheat, and the staple foods made of flour (e.g., pasta, bread, roti, noodles, etc.), remain a vital high-carbohydrate commodity in nourishing the still increasing world population [15]. Grain processing is a prerequisite for eating cereals, with milling fractionation resulting in the removal of bran fraction together with many compounds beneficial for human metabolism concentrating energy-dense parts into flour. The rapid urbanization in low- and middle-income countries brought dietary change toward an increasing amount of medium and high GI cereal foods [23,31], such as biscuits, cakes, bars, and cereal snacks, which are the carriers of high amounts of added sugar, lipids, and salt [32] (Table 1). GI values of most common cereal foods are in the high GI category (GI ≥ 70) and the medium GI category (GI 56–69) [14].

Table 1.
Glycemic index (GI) of common cereal products.
Incorporating low GI cereal foods in the daily diet should be an important component of glycemic management [14]. Livesey et al. [33] conducted a meta-analysis of 24-prospective cohorts of 7.5 million person-years of follow-up and reported a significant dose-dependent association between glycemic load and the risk of T2D. It is recommended that when the daily carbohydrate intake is between 200–250 g (median value in Western diets), the target GI should be below 40 [33]. When the daily carbohydrate intake exceeds 250 g per day, high GI foods (>70) should be limited to a lower frequency [33]. When the daily carbohydrate intake exceeds 300 g per day (over 60% of energy), an average GI of 33 should be recommended by introducing more low GI foods (GI < 50) [33].
Lowering the glycemic potency (i.e., GI) of cereal foods directly impacts the postprandial blood glucose excursion. The amount of glucose available in the bloodstream mainly depends on several components, including the available carbohydrate content in foods, carbohydrate digestion, hormonal regulation, glucose transport mechanisms and glycolysis [52]. The primary glucose available for the human body comes from the breaking down of the dietary carbohydrate (mostly from starch in cereal foods) by enzymes, including α-amylases and oligossacharidases (e.g., α-glucosidase and sucrase) to monosaccharides (mainly glucose) in mouth and duodenum [52]. In the small intestine, the active transport ensures a one-way flow of glucose D-isomers from the gut to the epithelial cells regardless of glucose gradient via activating sodium Na-glucose transporter 1 (SGLT1) [52]. The absorbed glucose within the capillaries is then transported via mesenteric circulation to target cells for harness energy [52].
The glucose metabolism is closely controlled by pancreatic hormones glucagon and insulin. While the glucagon handles the breakdown of stored liver glycogen and stimulation of gluconeogenesis and ketogenesis, insulin’s roles are to promote glucose uptake by cells, stimulate protein and fat synthesis, and inhibit glucagon activities [52]. Other hormones, including glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) produced by intestinal L-cell, are also responsible for glucose clearance in blood capillaries, slowing down gastric emptying, suppressing postprandial glucagon production, and increasing glucose-dependent insulin production. After uptake, glucoses undergo glycolytic flux to provide cellular energy and intermediates for energy supply for metabolism in humans [52]. In total, 23% of the glucose is disposed to the brain, 29% to splanchnic tissues (liver and gut), and 25% to insulin-dependent skeletal tissues, while the rest is used by other tissues such as the heart, adipose tissue, and the kidney.
Quality of cereal foods from a nutritional perspective became apparent as people became more aware of the impact of sedentary lifestyles, increased availability of foods with high energy density and high GI and changing eating patterns on health. The interplay between cereal foods quality (nutrition and glycemic potency) and the human digestive system brings about changes in physiology important for the maintenance of healthy homeostasis, particularly glucose homeostasis. Intending to improve the nutritional profile and lower the glycemic potency of cereal foods, research activities around the world emphasized studying physiological mechanisms and factors leading to lower glycemic potency and to the development of technologies suitable for rendering unconventional ingredients to improve the health benefit in cereal food products.
3. Seaweed: The Unconventional Potential of the Traditional Food Ingredients in Lowering the Glycemic Impact
The glycemic index can be attenuated by adjusting the nutrient composition to reduce the energy contribution from available carbohydrates, mitigating the digesting rate by blocking the activity of digestive enzymes, and delaying gastric emptying time and glucose absorption rate by increasing the digesta viscosity. Seaweeds, which provide a range of high-value nutrients, such as dietary fiber, protein, functional fatty acids, prebiotic polysaccharides, polyphenols, and carotenoids, can lower glycemic potency via interfering with the mechanisms. In this section, we review the use of seaweeds in modern days and the macro-and micro-nutrients of seaweed that may exhibit glycemic-lowering effects.
3.1. Seaweed: A Traditional Food Ingredients in Modern Days
Seaweeds or algae are autotrophic organisms that generate energy through photosynthesis [53]. They are the sea “vegetables” that provide nutrition and energy as feeds to other marine organisms [53]. The seaweeds are classified taxonomically into four main groups according to their pigmentations: red seaweed (Rhodophyta), green seaweed (Chlorophyta), brown seaweed (Phaeophyta), and blue-green seaweed (Cyanophyta) [53]. Red seaweed, such as Corallina officinalis, Tenarea tortuosa (formerly named Lithophyllum tortuosum), Asparagopsis armata, Palmaria palmata, and Mastocarpus stellatus, are the most abundant and primitive class. Brown seaweed (phylum Ochrophyta, class Phaeophyceae) is further divided into two subgroups: kelps mainly in the Western Cantabrians and Iberian Peninsula Atlantic coasts; (Laminaria hyperborean, Laminaria ochroleuca, and Saccorhiza polyschides), and fucales (Fucus vesiculosus, Fucus serratus, Himanthalia elongata, and Ascophyllum nodosum) [54,55]. Green seaweed, such as Ulva lactuca, is less abundant than the other groups [32]. Currently, only around 200 out of approximately 10,000 species are used as food ingredients [56,57]. Around 10 species are cultivated to provide 30 percent of the 120 million tones (measured in wet weight) of the world aquaculture production in 2019 [57]. In 2021, red seaweed and brown seaweed were ranked the second and the third-largest species groups in global aquaculture, respectively [57].
Red, brown, and green seaweeds have been used as direct food ingredients or processed foods in the coastal regions of Asian countries (e.g., China, Japan, and Korea), few European countries (e.g., Greenland and Iceland), and some South American countries (e.g., Mexico and Chile) [53,58,59]. The average adult in Japan and South Korea consumes around 14.3 g (wet weight) dietary seaweeds per day [60,61]. Amongst the seaweed foods, the most recognized are Porphyra/Pyropia spp. (Nori), Laminaria/Saccharina spp. (Kombu), and Undaria spp. (Wakame) [61]. Due to the global migration and cultural infiltration and the advancing technologies in food preservation and transportation, more regions have started to consume seaweed as direct food ingredients [53,62,63]. Further, due to the year-round availability, seaweeds have gained much attention as an inexpensive source for plant-derived extract. The seaweeds are commonly extracted to make food hydrocolloids such as agar, carrageenan, and alginates. The growth of consumers’ demands for healthy foods, sustainable food production, and plant-based foods has motivated the research for unconventional use of edible seaweeds beyond the traditional culinary use [64].
From a nutritional standpoint, the nutrients in seaweeds are excellent substances that can improve the carbohydrate quality and lower the glycemic potency of cereal foods. Similar to most terrestrial edible plants, seaweeds are excellent sources of dietary fiber, omega 3 fatty acids, polyphenols, and phytochemicals such as carotenoids [65]. These nutrients have been recognized and endorsed for health benefits and potential development of functional ingredients [57]. These seaweed-derived nutrients may have technological properties that make them viable to be incorporated in food products when the quantity is carefully evaluated to meet the required sensory quality [27,64,65].
Nevertheless, the availability of the nutrients and the nutrient composition of seaweeds growing in the wild are highly variable between seasons [66,67,68]. For example, the total seaweed carbohydrates content in wet weight reaches the highest in the maximum growth periods and in the summer months when photosynthetic activity is the highest [67]. The lipid content is correlated to the temperature change and reaches the highest concentration in winter and spring [69]. The protein content reaches the highest concentration in August [68]. The synthesis of the bioactive compounds generally responds to the intense irradiation and high oxygen concentration that typically happens in February to act against the free radicals and other oxidizing agents [68]. The moisture content of fresh seaweed is very high (up to 94% of the biomass, Table 2), which also varies between seasons in the wild [27]. The advancing seaweed cultivation technologies have solved the problems of nutrient variations in wild-harvested seaweeds [70,71]. For instance, by modifying the abiotic factors, the production of target nutrients can be well controlled [72,73].

Table 2.
Nutrient composition of some green algae (chlorophyta), red algae (Rhodophyta), and brown algae (phaeophyceae).
3.2. Carbohydrates: Dietary Fiber and Polysaccharides
Seaweeds have high amounts of total carbohydrates, ranging between 20–76% of dry weight (DW) depending on the species (Table 2) [27]. The highest total carbohydrate contents are found in species such as Fucus, Ascophyllum, Porphyra, and Palmaria [27]. Carbohydrates in seaweeds exist as cell wall structural, storage, and functional mucopolysaccharides [27]. In terms of regulating glycemic potency, seaweed is a good source of carbohydrates alternative, since the majority is available as dietary fibers that cannot be utilized and absorbed by the human body. In fact, seaweed has even higher levels of total dietary fiber than terrestrial plant foods [27,66]. Based on DW, seaweed’s average total dietary fiber content can vary from 36% to 60% (Table 2). The available carbohydrate, such as glucose, mannose, and galactose are in small quantity [27].
Seaweeds contain two main fractions of dietary fibers: around 25% water-soluble fibers [73] and 21.8% insoluble fibers [55]. Soluble fibers, such as agars, alginates, fucoidan, carrageenan, laminararan, and porphyran can form viscous gels in water in the human gastrointestinal tract to promote satiety, improve intestinal microbiota, and slow down the digestion and absorption of other nutrients [27]. Insoluble fibers, including cellulose, hemicelluloses (2–10% and 9% DW, respectively), mannans, and xylan are mainly from cell wall polysaccharides (i.e., the building blocks to support the thallus in water) [27]. They cannot form gels in water or fermented in the large intestine, but can keep water content in the structural matrix to facilitate large intestine transit and increase the fecal mass [27]. The ratio of soluble to insoluble fiber (S/I) in seaweeds varies depending on the type of seaweed and growing conditions, but is generally higher than the values found in terrestrial edible plants [55]. In this review, we are going to scoop the glucose-attenuating effect of the seaweed dietary fibers.
3.2.1. Alginate
Alginate was discovered as a major dietary fiber in brown algae (Phaeophyceae) from the genera Laminaria, Undaria, and Hizikia [27]. It is a glucose polymer that comprises a block-wise monomers α-l-guluronic acid (G residue segments) and β-d-mannuronic acids (M residue segments) to form alternating G-blocks and M-blocks or MG-blocks [103]. The viscosity and gelling forming ability of alginate is contributed by the content of G-residues [104], and play an important role in reducing the post-digestion nutrients absorption and gastrointestinal signaling to control satiation and satiety [105,106]. One unique property of alginate is its ability to form gel in both acid environments (pH < 3.5 at stomach) and form ionic gelation in the presence of divalent cations (such as sodium and calcium) [105]. Now, in both alginates forms, alginic acid (a linear polyuronic acid) and salt (sodium alginates (SA) and calcium alginate Ca-alginate), are widely applied as natural hydrocolloids to thicken aqueous solutions to form desired food formation and quality [107].
Alginate has been reported to decrease the concentration of serum cholesterol [108], prevent the absorption of toxic chemical substances in the gut [109,110], and prevent a postprandial increase of blood glucose and insulin [111]. Previous in vitro research showed that the predominant postprandial blood glucose-suppression effect of calcium alginate is by its ability to inhibit brush border enzyme α-glucosidase [112,113]. In addition, the positive correlation between the viscosity of the starch suspension and the decrease of starch digestion suggests the main suppression effect comes from the increase of viscosity by alginates, which act as a barrier to physically interfere with the contact between α-amylase and starch and between α-glucosidase and maltose [112].
The acute postprandial glycemic potency of alginate-fortified foods and beverages in healthy adults was investigated [114,115,116,117,118,119] (Table 3). The beverages fortified with sodium alginate reported a significant reduction in peak glucose concentration (Cmax) between 6–53.2% [114,115,116,119]. Fortification with higher sodium alginate (8.13 g) more significantly attenuated the Cmax and postprandial peak insulin than the same beverage with lower sodium alginate (4.06 g) [115]. Compared to a similar quantity of total dietary fiber, sodium alginate exhibited a significant glucose-lowering effect [119]. A pre-load of beverage fortified with sodium alginate extract (15 g) also attenuated the postprandial glucose excursion after a subsequent meal by 40% [116]. The significant reduction of Cmax and incremental area under the glucose excursion curve (iAUCgluose) was also reported for sodium alginate fortified solid food (crispy bar containing 50 g available carbohydrate) [117] (p < 0.05). Kato et al. [118] found that both 5% (3.2 g) and 8% (5.0 g) calcium alginate in mixed meals significantly reduced the Cmax and iAUCgluose (p < 0.05) in healthy subjects. The results from acute clinical trials strongly support the potential of using alginate salt (sodium alginate and calcium alginate) to lower the glycemic potency of cereal food.

Table 3.
Human trials of the efficacy of functional seaweed extract on acute postprandial glucose.
3.2.2. Fucoidan
Fucoidan is a complex sulphated polysaccharide found mainly in the cell walls of brown seaweeds [120]. It is primarily composed of a pentose sugar backbone comprising of repeating hexose deoxy sugar L-fucose residues that are linked by α-(1–3), α-(1–3)-α-(1–4), and in some minor cases by α-(1–3)-α-(1–2) linkages with sulphate groups presented in C-2, C-3, and C-4 positions [120]. It also contains some other neutral monosaccharides, including glucose, galactose, xylose, mannose, and rhamnose [121,122] and may contain acetate and uronic acids [123].
The monosaccharide composition, the degree of sulphation, and molecular weight of fucoidan highly differ between species and within species between different harvesting seasons [124,125] and is linked directly to its bioactivity [126,127]. The most bioactive fraction of fucoidan is that it contains mainly fucose, variable proportions of galactose, and a high-sulfate low-uronic acid proportion; whereas the low bioactive fraction contains other monosaccharides apart from fucose and a low-sulfate high-uronic acid proportion [128]. Fucoidan’s size varies from <10 kDa (low molecular weight fucoidan (LMWF)), 10–10,000 kDa (medium molecular weight fucoidan (MMWF)), to >10,000 kDa (high molecular weight fucoidan (HMWF)) [123,129]. High molecular weight may cause low cell membrane permeability, low bioavailability, and lower potential clinical efficacy [130]. For example, LMWF (4.58–6.5 kDa) exhibits potential anticoagulant effect, whilst a higher molecular weight >850 kDa lacked certain activity[131,132]. HMWF was reported to have no significant effect on long-term glycemic control in diabetic patents [133]. LMWF, on the other hand, has been reported to impact glycemic control via promoting insulin secretion [127], protecting pancreatic [23], and improving insulin resistance [131,134]. As a result, HMWF depolymerisation to synthesize oligomeric components of smaller molecular weight has been advocated to improve bioactivity and bioaccessibility [129].
In the past 10 years, the antidiabetic potential of fucoidan as an unconventional bio-compound has gained attention with the therapeutic effects at various stages of glucose metabolism. Fucoidan extract from Undaria pinnatifida, Fucus vesiculosus, and Ecklonia maxima has been reported to act as the inhibitor of α-glucosidase [132,135] and implicated as a potential antidiabetic agent [136,137,138]. Fucoidan from Ascophyllum nodosum and Turbinaria ornata has exhibited an inhibitory effect on α-amylase [136,139]. Fucoidan’s negative charge may help to regulate the SGLT1 to slow down the post-digestion glucose absorption in the intestinal lumen [125]. Fucoidan extracts from Cucumaria frondose [131] and Acaudina molpadioides [140] have been reported to increase insulin production by activating the PI3K/PKB pathway and increase the blood glucose uptake by muscle and fat tissue via activating glucose transporter 4 (GLUT4) translocation. It has been implicated as an inhibitor of dipeptidyl peptidase IV (DPP IV), which is an enzyme responsible for the rapid degradation of incretin hormones such as GLP-1 [141,142], to increase insulin production [143]. In addition to the acute effect, HMWF has been reported to improve the bowel movements (i.e., stool frequency) in T2D patients [96], which may assist the long-term T2D management. However, a pre-load of low dose (0.5 g) and high dose (2.0 g) fucoidan extract did not report a significant hypoglycemic effect when compared to a placebo (cellulose) in healthy adults [144]. Again, no significant effect on postprandial glycemia was found in adults who are overweight or obese otherwise healthy [145] (Table 3). The inconsistency could be a result of the inclusion of low bioactivity and bioaccessibility of fucoidan as an intervention. Since the inadequate data on molecular weight and degree of sulphation, it is difficult to associate the clinical efficacy to the structural and chemical composition.
3.2.3. Other Dietary Fibers and Polysaccharides
Besides the two soluble fibers, alginate and fucoidan, which stand out for the acute hypoglycemic effect, other dietary fibers are directly and indirectly associated with postprandial blood glucose management [55]. Laminarin, a class of low-molecular-weight (5 kDa) storage β-glucans found mainly in brown seaweeds Laminaria, Undaria pinnatifida, Fucus vesiculosus, and Saccharina species (up to 35% DW) has multiple (1,3)-β-D-glucan units comprising (1,3)-D-glucopyranose residues with 6-O-branching in the primary chain and (1,6)-links between chains [146]. Depending on the level of branching, it can be water-soluble or insoluble. Laminarin has many reported biofunctional activities, including blocking starch digestion, activating macrophages leading to immunostimulatory, anti-inflammatory, anti-tumor, and wound-healing activities [147]. Agar (100–30,000 kDa), which commonly presents in red seaweeds Gracilaria and Gelidium, is the first registered hydrocolloid in Europe (registration number E406) [148]. It contains around 40% of agaro-oligosaccharides, which has been reported to possess a potential activity against α-glucosidase [149], suppressing the generation of pro-inflammatory cytokines and the activity of the pro-oxidative enzyme that associated with producing nitric oxide [150]. Ulvan, a water-soluble sulphated polysaccharide from the green seaweed Ulva and Enteromorpha, comprises mainly rhamnose, uronic acid, and xylose [27]. A systematic review has discussed its function in anti-oxidant and anti-hyperlipidemic [151], which may assist the long-term glycemic management. Carrageenan (193–324 kDa), commonly obtained from red seaweeds of Eucheuma and Kappaphycus, are found in three major forms, κ- carrageenan and ι-carrageenan that have gelatinization properties, and a thickening substance λ-carrageenan [55]. Carrageenan gel has been reported to exhibit a range of pharmacological applications, including antiviral properties, by blocking the transmission of HIV [152] and STD viruses [153], anticoagulant activity by inhibiting platelet aggregation [154]. However, since carrageenan has been reported to cause reproducible inflammatory reactions and lead to glucose intolerance [155], it is not recommended as an unconventional food ingredient for people at higher risk of dysglycemia. Mannitol is a sugar alcohol corresponding to mannose found in brown seaweeds Laminaria and Ecklonia. It is commonly used to replace sucrose in sugar-free compound coating due to its sweet taste [27].
3.3. Protein
Total protein content varies significantly between species and harvest seasons. It is generally low in brown seaweed (5–24% DW) and high in red and green seaweeds (10–47% DW) [159]. (Table 2) The content of proteins, peptides, and amino acids of red and green seaweeds reaches the maximum in summer [75,159]. Total protein can represent up to 44% DW in Porphyra tenera (nori) and Ulva spp. [84], which is comparable to high-protein vegetables such as soy [110]. The maximum content in brown seaweeds is found in Undaria in spring, which is around 24% DW [160] followed by Laminaria and Sargassum (around 20% DW) [90] and Fucus (12.99% DW) [74]. Most seaweed species are a rich sources of all essential amino acids at the recommended level by WHO [27]. Although some amino acids, including threonine, lysine, tryptophan, sulphur amino acids (cysteine and methionine), and histidine are limited in algal proteins [66], the levels are still higher than those in terrestrial edible plants [27].
The functional amino acids present in seaweeds have shown key biological activities [66]. Peptides extracted from red seaweed Palmaria palmate exhibited significantly higher DPP IV inhibition capacity (IC50 > 200 µM) than the natural origin peptides from edible terrestrial plants, collagen, and milk protein [161]. The collected data suggest the potential for increasing insulin production to improve postprandial glucose clearance [161].
3.4. Lipids
Seaweeds contain a lower lipid quantity (1–4.5% DW) than other marine organisms [110]. (Table 2) The contribution of seaweed lipids as a food energy source appears to be low than terrestrial oil-rich edible plants such as nuts and soybeans [162]. However, the proportion of long-chain polyunsaturated fatty acids (PUFAs) in seaweeds is higher than in terrestrial edible plants [162]. Particularly, the eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) that belong to the omega-3 fatty acids family are linked to a range of biochemically and physiologically important functions [162]. Red seaweeds contain a higher quantity of EPA, palmitic acid, oleic acid, and arachidonic acid; brown seaweeds contain higher oleic acid, linoleic acid, and α-linolenic acid but low EPA; green seaweeds have a greater quantity of linoleic acid and α-linolenic, palmitic, oleic, and DHA [163]. Both red and brown algae are good sources of omega-3 and omega-6 fatty acids [164]. The amounts and concentrations of these PUFAs vary depending on environmental temperature, with lower temperatures favoring their production. The biological benefit of EPA and DHA on hyperlipidemia, blood pressure, cardiac function, vascular reactivity, endothelial function, and anti-inflammation have been extensively studied [162,165].
Furthermore, the prominent lipid in seaweed oil, phospholipids (62–78% phosphatidylcholine and 10–23% phosphatidylglycerol of the total lipid), has shown greater resistance to oxidation and a higher bioavailability [166]. Phospholipids in foods act as natural emulsifiers, aiding and easing the digestion and absorption of fatty acids, cholesterol, and other lipophilic nutrients [166]. Therefore, seaweed oils have many advantages over plant and fish oils as unconventional ingredients in that they are much more resistant to deterioration (rancidity) [166]. Fucosterol, another prominent sterol found in brown seaweed, has been shown to exert a high affinity of inhibitory effect on α-amylase [167] and α-glucosidase, and promote the postprandial glucose clearance by suppressing the expression of protein-tyrosine phosphatase 1B (PTP1B which inversely regulates insulin signalling) [168,169].
3.5. Polyphenols
Polyphenols are not abundant in seaweeds. Compared to red and green seaweed (only <1% DW) [82], brown seaweeds have relatively higher levels of polyphenols (up to 4 g GAE/100 g DW) [27]. One of the main polyphenols in brown seaweeds, phlorotannis, is derived from the polymerization of several phloroglucinol units (1,3,5-trihydroxybenzene), has been reported to inhibit α-glucosidase and α-amylase activities [170]. The inhibitory effect of phlorotannis can be noncompetitive (phlorofurofucoeckol-A, 7-phloroeckol, and dioxinodehydroeckol) and competitive (dieckol and eckol) [171]. Bromophenols, found in both red seaweed (Rhodymeniaceae) and brown seaweed (Leathesia nana) also exhibit the α-glucosidase inhibitory effect [172]. Furthermore, both phlorotannins and bromophenols can increase the insulin-mediated glucose uptake by skeletal muscles by inhibiting the activity of PTP1B, which regulates the leptin and insulin signaling pathways [172,173]. In vivo studies have reported Fucus vesiculosus and Ascophyllum nodosum attenuated postprandial blood glucose excursion and dropped insulin peak in rats with diet-induced steatosis [170]. Further studies observed the same hypoglycemic effect in mice with non-alcoholic steatohepatitis (NASH) [170] and rats with diet-induced steatosis [174] after being treated with phlorotannins extract.
3.6. Carotenoids
Carotenoids that are synthesized naturally by photosynthetic seaweeds give a range of pigmentations [175,176]. Fucoxanthin is one of the major xanthophyll found abundantly in brown seaweeds such as Fucus spiralis (171 mg/kg DW) and Ascophyllum nodosum (660 mg/kg DW) [177]. One of the anti-diabetic values of fucoxanthin is supported by its α-glucosidase inhibitory function, which delays the digestion and absorption of glucose [178,179]. Another anti-diabetic mechanism of fucoxanthin is via upregulating the GLUT4 expression in skeletal muscle via the induction of Peroxisome proliferator-activated receptor-γ (PPARγ) coactivator-1α and increasing Akt phosphorylation [172,173]. The animal models (C57BL/6J mice fed in high-fat diet) are reported to have normalized GLUT4 expression and increased uptake of glucose in skeletal muscle [180,181]. The hypoglycemic and hypoinsulinemic effect of fucoxanthin was also reported in diabetic/obese KK-Ay mice [178] and obese KK-Ay mice [179].
4. Proposed Strategies for Seaweeds-Derived Functional Ingredients in Glycemic Control
4.1. Reducing the Available Carbohydrate
Lowering available carbohydrate content can be achieved by replacing the available carbohydrates with other macronutrients derived from seaweeds (Figure 2). For example, mannitol from brown seaweeds Laminaria and Ecklonia can be used as a replacement for sucrose (i.e., sugar) in sugar-free compound coating without depriving the sweet taste [27]. The full amino acid profile in seaweed proteins [27], particularly the red and green seaweed, which contains up to 47% protein in dry mass [159], makes them perfect candidates to substitute flours in large quantities to reduce the available carbohydrates.
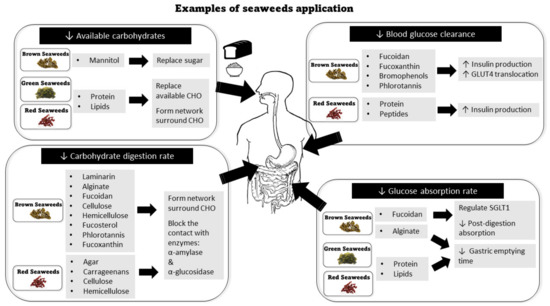
Figure 2.
Examples of seaweeds application in various strategies to lower glycemic impact in cereal foods.
The addition of exogenous proteins to the cereal products can limit the starch availability and digestibility by forming protein—starch interactions network surrounding starch granules, which builds the barrier to digestive enzymatic access [182,183,184]. Further treatment on seaweed protein including hydrolysis [185], low-temperature drying and denaturing [186] could further improve the bioactivity. Similarly, the addition of lipids to cereal products can affect glycemic potency by interacting with starch to form a network surrounding the starch that limits enzymatic accessibility and also starch granule swelling [187]. Under rising temperatures, the exogenous seaweed lipids and starch can form lipid-amylose complexes which have been demonstrated to resist starch digestion [188,189]. A previous study showed olive oil formed a complex with amylose in wheat bread and significantly reduced the postprandial glycemic response as compared to baked bread without oil [190]. Thus, we can expect that the combination of seaweed lipids with high-amylose rice, flour, or other cereal products may also significantly impact glycemic potency.
4.2. Reducing Post-Consumption Digestion Rate
Reduction of post-consumption digestion rate can be achieved by blocking the brush border enzyme α-amylase α-glucosidase. Dietary fibers in seaweed are one of the most common ingredients applied to cereal food products to limit starch digestion rate through this mechanism (Figure 2). With different solubility, molecular weight, ionic groups, viscosity, and gelling capacity, different seaweed dietary fibers can provide different functions required to govern the starch digestibility in cereals. Agar and carrageenans from red seaweeds and laminarin (β-glucans) from brown seaweeds (e.g., Undaria pinnatifida and Fucus vesiculosus) have been used as gelling and thickening agents in food ingredients [55,137,139]. They may form a continuous hydrated network surrounding the starch granules [191] and interact with the starch granule surface to create a barrier for the enzymatic access (α-amylase and α-glucosidase) to starch granules. Thus, the barriers restrict the amylose leaching during the gelatinization [140,186,187]. A similar blocking effect has been found in brown seaweed fibers alginate [112,113] and fucoidan [132,135]. The soluble fiber may further increase the viscosity of digesta [103] after ingestion to reduce the rate of glucose release from digestion in the intestinal phase [192,193]. The rich cellulose and hemicellulose content in all seaweeds may bind α-amylase to inhibit the depolarization of starch, as observed in wheat bran [194].
In addition to dietary fibers, other nutrients found in seaweeds have been identified as α-amylase and α-glucosidase inhibitors and do not affect the viscosity of food products. They include brown seaweed phospholipids, fucosterol [167], two polyphenols, phlorotannis (from brown seaweeds) [170], and bromophenols (from red and brown seaweeds) [172], and fucoxanthin (from brown seaweeds) [180,195]
4.3. Reduce the Rate of Post-Digestion Glucose Absorption
The rate of post-digestion glucose absorption can be attenuated by delaying the gastric emptying time. Dietary fibers derived from seaweeds can increase the viscosity of food, which leads to slowing down the gastric emptying rate to slow down the rate of glucose absorption and release into the bloodstream [107,189,191] (Figure 2). For example, during post-ingestion alginate can form solid intragastric gels in digesta to distend the stomach and delay gastric emptying and glucose absorption [115,196]. Fucoidan’s negative charge may help to regulate and manipulate the SGLT1 to slow down the post-digestion absorption of glucose in the intestinal lumen [125].
It is postulated that the addition of lipids and proteins in cereal foods can delay gastric emptying time [197]. Previous acute nutritional human trials demonstrated consuming pancakes with added unsaturated fatty acids (e.g., sunflower oil, olive oil, butter, and medium-chain triglyceride oil) significantly delayed the gastric emptying time and reduced postprandial glycemic response than consuming pancakes without added oil [197]. Co-ingestion of olive oil or grapeseed oil [198], and rapeseed oil [199] ameliorating the GI of cereal foods including rice porridge and bread. Particularly, when both proteins and lipids are co-ingested with the cereal foods, the glycemic impact was more significantly attenuated [199]. In fact, compared to lipids, protein has a more significant effect on slowing down gastric emptying rate [200] by forming protein clots in the acidic stomach [201].
4.4. Increase the Rate of Postprandial Glucose Clearance
The blood glucose update can be interfered with by several functional nutrients found in seaweeds (Figure 2). Fucoidan from brown algae Cucumaria frondose [131] and Acaudina molpadioides [140] may increase blood glucose clearance through two mechanisms: promoting insulin production by activating the PI3K/PKB pathway and inhibiting DPP IV [141,142]; activating GLUT4 translocation [140]. Consuming proteins with carbohydrates in a context meal may also promote insulin secretion [202,203]. The peptides extracted from red seaweed Palmaria palmate can significantly inhibit DPP-IV to increase insulin production [160]. Polyphenols, such as phlorotannins and bromophenols can increase the insulin-mediated glucose clearance by skeletal muscle by activating the PI3K/PKB pathway [172,173]. Fucoxanthin has also shown the potential of regulating GLUT4 to increase blood glucose clearance [204].
4.5. Future Perspective in Clinical Evidence and Application
The limited evidence from the acute postprandial glycemic study of seaweeds gave mixed results, which require cautious interpretation (Table 3). When consuming Undaria pinnatifida (Wakame) with 200 g rice, a significant reduction in postprandial glucose (after 30 min) and iAUC of postprandial glucose response was reported in people with prediabetes [157]. In another study, consuming 70 g Undaria pinnatifida (Wakame) with a meal did not significantly change the postprandial glucose excursion in healthy adults, however, a significant reduction in postprandial glucose at 30 min was reported after these healthy adults consumed 70 g Undaria pinnatifida sporophylls (Mekabu) with the same meal [158]. However, the preload studies failed to exhibit significant efficacy. The study compared two doses Fucus vesiculosus powder (0.5 g and 2.0 g) preload to cellulose as a placebo reported no significant improvement between treatments [144]. Therefore, comparing the soluble fiber rich Fucus vesiculosus powder with other fiber that also has attenuating effect on postprandial glycaemia could be the reason for lacking statistically significant difference. Similarly, the combination of Ascophyllum nodosum and Fucus vesiculosus (0.5 g) preload showed no significant reduction in postprandial glucose but a significant reduction in iAUC of postprandial insulin response [156]. Due to the small sample size and the possible carry-over effect in a cross-over design, it is difficult to interpret and draw conclusions from the limited number of acute clinical trials. Thus, more high-quality clinical trials are required to determine the effective seaweed and cereal foods combination and optimal dose. Moreover, the nutrients content, bioactivity, and bioaccessibility vary significant between seaweeds and within seaweeds between harvesting seasons. The administration should be carefully evaluated to maximize the efficacy of various seaweeds. For example, a combination of protein rich red seaweeds and fiber rich brown seaweeds could add value to the hypoglycemic property.
The common method of incorporating seaweed ingredients is by dispersing them in powder form or the form of extracts as part of the formulation of traditional cereal products [205]. However, a high quantity of soluble fiber may compete with the other ingredients for water absorption and result in a harder food texture [192], which may lower the sensory quality of the cereal products. Therefore, the type and the concentration of soluble fiber used in cereal products should be evaluated carefully to balance the required function without compromising consumer liking. Successful application of seaweeds powder in cereal-based products, such as bread, noodles, cake, cookies, biscuits, and extruded maize. For example, Himanthalia elongata was used as a source of dietary fiber to enhance the phytochemical content of breadsticks at doses of 5–15% [205]. To maintain the sensory qualities, the recommended concentration of seaweed-derived products should remain between 0.5–8% in wheat bread, 3–30% in wheat noodles, 5–20% in pasta, 2.5–20% in cakes, 5–60% in biscuits, 3–9% in cookies, and 3.5% in extrudes maize [205].
5. Conclusions
Many of the common cereal food in the Asian diet is in the moderate to high GI categories including rice grain, wheat flour, and bakery products that are often made with sugars and refined flour. There is a trend of increasing high GI cereal food consumption in low- and mid-income countries as urbanization accelerate. With the prevalence of diabetes surges, food industries and mindful consumers must incorporate sustainable and functional nutrients from seaweeds as unconventional ingredients into cereal foods to lower the glycemic potency. This review discussed the potential of seaweeds, which have never been a staple food in the Western diet and the diet in inland regions, despite their popularity in a few coastal regions, and the applications based on the synergetic approaches of adding unconventional ingredients.
Seaweeds are a high-quality and nutritious food source by virtue of the high functional dietary fiber and polysaccharides, high content of essential and functional amino acids, high-quality lipids (i.e., omega-3 fatty acids), polyphenols, and carotenoids. Traditionally, seaweeds are used by their technological properties (i.e., gelling, thickening, and stabilizing), such as agar, alginates, and carrageenan as hydrocolloids. Many studies have focused on the supplementary value of either individual seaweeds extract (dietary fiber, omega-3 fatty acids, protein, polyphenols, carotenoids, and minerals) or individual seaweed species in improving the glycemia in people at higher risk of diabetes. To ameliorate acute postprandial blood glucose, reformulate the high GI or glycemic potency of staple food, such as cereal food products, can be achieved by multiple strategies with the addition of multiple seaweed-derived ingredients. As the contents of bioactive and technological compounds vary between seaweed species, the modification does not need to be limited to one seaweed. Instead, multiple seaweed-derived ingredients can work synergistically (i.e., rich functional dietary fibers from brown seaweeds and protein content from red and green algae) to combine the different peculiar compositions of different seaweed species. The strategy’s effectiveness will ultimately depend on how well consumers accept the new textural and sensorial features of the food, whether glycemic impact can be concealed with minimal dietary habit change. The food industry should exploit seaweeds’ nutritional benefits by developing tasty ingredients, reformulated products, and appropriate marketing strategies to reach consumers.
Author Contributions
Conceptualization: L.W.L. and J.-H.C.; investigation: L.W.L.; resources: L.W.L. and J.-H.C.; writing—original draft preparation: L.W.L.; writing—review and editing: J.-H.C.; visualization: L.W.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research received New Zealand National Science Challenge High-Value-Nutrition (HVN) program, via the Ministry for Business, Innovation and Employment (MBIE, grant number 3719728) and National Natural Science Foundation of China (No. 31601472); Guangdong Province Zhujiang Talent Program (No. 2019ZT08H476); Shenzhen Science and Technology Program (No. KQTD20180412181334790).
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Zheng, Y.; Ley, S.H.; Hu, F.B. Global Aetiology and Epidemiology of Type 2 Diabetes Mellitus and Its Complications. Nat. Rev. Endocrinol. 2018, 14, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Saeedi, P.; Petersohn, I.; Salpea, P.; Malanda, B.; Karuranga, S.; Unwin, N.; Colagiuri, S.; Guariguata, L.; Motala, A.A.; Ogurtsova, K.; et al. Global and Regional Diabetes Prevalence Estimates for 2019 and Projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas, 9th Edition. Diabetes Res. Clin. Pract. 2019, 157, 107843. [Google Scholar] [CrossRef] [PubMed]
- International Diabetes Federation. IDF Atlas 10th Edition. 2021. Available online: https://idf.org/e-library/epidemiology-research/diabetes-atlas.html?id=171 (accessed on 8 February 2022).
- World Health Organization. WHO Coronavirus (COVID-19) Dashboard Overview of Current Cases. Available online: https://covid19.who.int/ (accessed on 23 January 2022).
- Holman, N.; Knighton, P.; Kar, P.; O’Keefe, J.; Curley, M.; Weaver, A.; Barron, E.; Bakhai, C.; Khunti, K.; Wareham, N.J.; et al. Risk Factors for COVID-19-Related Mortality in People with Type 1 and Type 2 Diabetes in England: A Population-Based Cohort Study. Lancet Diabetes Endocrinol. 2020, 8, 823–833. [Google Scholar] [CrossRef]
- Singh, A.K.; Gillies, C.L.; Singh, R.; Singh, A.; Chudasama, Y.; Coles, B.; Seidu, S.; Zaccardi, F.; Davies, M.J.; Khunti, K. Prevalence of Co-Morbidities and Their Association with Mortality in Patients with COVID-19: A Systematic Review and Meta-Analysis. Diabetes Obes. Metab. 2020, 22, 1915–1924. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Zhang, J.; Sun, X.; Wang, L.; Xu, Y.; Zhang, Y.; Liu, X.; Dong, C. Influence of Diabetes Mellitus on the Severity and Fatality of SARS-CoV-2 (COVID-19) Infection. Diabetes Obes. Metab. 2020, 22, 1907–1914. [Google Scholar] [CrossRef]
- Dennis, J.M.; Mateen, B.A.; Sonabend, R.; Thomas, N.J.; Patel, K.A.; Hattersley, A.T.; Denaxas, S.; McGovern, A.P.; Vollmer, S.J. Type 2 Diabetes and COVID-19-Related Mortality in the Critical Care Setting: A National Cohort Study in England, March-July 2020. Diabetes Care 2021, 44, 50–57. [Google Scholar] [CrossRef]
- Hayek, S.; Ben-Shlomo, Y.; Balicer, R.; Byrne, K.; Katz, M.; Kepten, E.; Raz, I.; Roitman, E.; Zychma, M.; Barda, N. Preinfection Glycaemic Control and Disease Severity among Patients with Type 2 Diabetes and COVID-19: A Retrospective, Cohort Study. Diabetes Obes. Metab. 2021, 23, 1995–2000. [Google Scholar] [CrossRef]
- Tan, W.S.K.; Tan, S.-Y.; Henry, C.J. Ethnic Variability in Glycemic Response to Sucrose and Isomaltulose. Nutrients 2017, 9, 347. [Google Scholar] [CrossRef]
- Kataoka, M.; Venn, B.J.; Williams, S.M.; Te Morenga, L.A.; Heemels, I.M.; Mann, J.I. Glycaemic Responses to Glucose and Rice in People of Chinese and European Ethnicity. Diabet. Med. 2013, 30, e101–e107. [Google Scholar] [CrossRef]
- Dickinson, S.; Colagiuri, S.; Faramus, E.; Petocz, P.; Brand-Miller, J.C. Postprandial Hyperglycemia and Insulin Sensitivity Differ among Lean Young Adults of Different Ethnicities. J. Nutr. 2002, 132, 2574–2579. [Google Scholar] [CrossRef]
- Jenkins, D.J.; Wolever, T.M.; Taylor, R.H.; Barker, H.; Fielden, H.; Baldwin, J.M.; Bowling, A.C.; Newman, H.C.; Jenkins, A.L.; Goff, D.V. Glycemic Index of Foods: A Physiological Basis for Carbohydrate Exchange. Am. J. Clin. Nutr. 1981, 34, 362–366. [Google Scholar] [CrossRef] [PubMed]
- Augustin, L.S.A.; Kendall, C.W.C.; Jenkins, D.J.A.; Willett, W.C.; Astrup, A.; Barclay, A.W.; Björck, I.; Brand-Miller, J.C.; Brighenti, F.; Buyken, A.E.; et al. Glycemic Index, Glycemic Load and Glycemic Response: An International Scientific Consensus Summit from the International Carbohydrate Quality Consortium (ICQC). Nutr. Metab. Cardiovasc. Dis. 2015, 25, 795–815. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations FAOSTAT Statistical Database. Available online: https://www.fao.org/faostat/en/#data/FBS (accessed on 30 January 2022).
- Cui, Z.; Dibley, M.J. Trends in Dietary Energy, Fat, Carbohydrate and Protein Intake in Chinese Children and Adolescents from 1991 to 2009. Br. J. Nutr. 2012, 108, 1292–1299. [Google Scholar] [CrossRef] [PubMed]
- Seah, J.Y.H.; Koh, W.-P.; Yuan, J.-M.; Van Dam, R.M. Rice Intake and Risk of Type 2 Diabetes: The Singapore Chinese Health Study. Eur. J. Nutr. 2019, 58, 3349–3360. [Google Scholar] [CrossRef] [PubMed]
- Zuñiga, Y.L.M.; Rebello, S.A.; Oi, P.L.; Zheng, H.; Lee, J.; Tai, E.S.; Van Dam, R.M. Rice and Noodle Consumption Is Associated with Insulin Resistance and Hyperglycaemia in an Asian Population. Br. J. Nutr. 2014, 111, 1118–1128. [Google Scholar] [CrossRef]
- Sun, Q.; Spiegelman, D.; Van Dam, R.M.; Holmes, M.D.; Malik, V.S.; Willett, W.C.; Hu, F.B. White Rice, Brown Rice, and Risk of Type 2 Diabetes in US Men and Women. Arch. Intern. Med. 2010, 170, 961–969. [Google Scholar] [CrossRef]
- Aune, D.; Norat, T.; Romundstad, P.; Vatten, L.J. Whole Grain and Refined Grain Consumption and the Risk of Type 2 Diabetes: A Systematic Review and Dose-Response Meta-Analysis of Cohort Studies. Eur. J. Epidemiol. 2013, 28, 845–858. [Google Scholar] [CrossRef]
- Lu, L.W.; Monro, J.; Lu, J.; Rush, E. The Effect of Cold Treatment of Parboiled Rice with Lowered Glycaemic Potency on Consumer Liking and Acceptability. Foods 2018, 7, 207. [Google Scholar] [CrossRef]
- Aune, D.; Ursin, G.; Veierød, M.B. Meat Consumption and the Risk of Type 2 Diabetes: A Systematic Review and Meta-Analysis of Cohort Studies. Diabetologia 2009, 52, 2277–2287. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, Z.; Du, W.; Huang, F.; Jiang, H.; Bai, J.; Zhang, X.; Zhang, B.; Wang, H. Twenty-Five-Year Trends in Dietary Patterns among Chinese Adults from 1991 to 2015. Nutrients 2021, 13, 1327. [Google Scholar] [CrossRef]
- Batis, C.; Sotres-Alvarez, D.; Gordon-Larsen, P.; Mendez, M.A.; Adair, L.; Popkin, B. Longitudinal Analysis of Dietary Patterns in Chinese Adults from 1991 to 2009. Br. J. Nutr. 2014, 111, 1441–1451. [Google Scholar] [CrossRef]
- Wee, M.S.M.; Henry, C.J. Reducing the Glycemic Impact of Carbohydrates on Foods and Meals: Strategies for the Food Industry and Consumers with Special Focus on Asia. Compr. Rev. Food Sci. Food Saf. 2020, 19, 670–702. [Google Scholar] [CrossRef] [PubMed]
- Rajapakse, N.; Kim, S.-K. Nutritional and Digestive Health Benefits of Seaweed. Adv. Food Nutr. Res. 2011, 64, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Holdt, S.L.; Kraan, S. Bioactive Compounds in Seaweed: Functional Food Applications and Legislation. J. Appl. Phycol. 2011, 23, 543–597. [Google Scholar] [CrossRef]
- Wells, M.L.; Potin, P.; Craigie, J.S.; Raven, J.A.; Merchant, S.S.; Helliwell, K.E.; Smith, A.G.; Camire, M.E.; Brawley, S.H. Algae as Nutritional and Functional Food Sources: Revisiting Our Understanding. J. Appl. Phycol. 2017, 29, 949–982. [Google Scholar] [CrossRef] [PubMed]
- Afonso, N.C.; Catarino, M.D.; Silva, A.; Cardoso, S.M. Brown Macroalgae as Valuable Food Ingredients. Antioxidants 2019, 8, 365. [Google Scholar] [CrossRef]
- Shannon, E.; Abu-Ghannam, N. Seaweeds as Nutraceuticals for Health and Nutrition. Phycologia 2019, 58, 563–577. [Google Scholar] [CrossRef]
- Tak, M.; Shankar, B.; Kadiyala, S. Dietary Transition in India: Temporal and Regional Trends, 1993 to 2012. Food Nutr. Bull. 2019, 40, 254–270. [Google Scholar] [CrossRef]
- Poutanen, K. Past and Future of Cereal Grains as Food for Health. Trends Food Sci. Technol. 2012, 25, 58–62. [Google Scholar] [CrossRef]
- Livesey, G.; Taylor, R.; Livesey, H.; Liu, S. Is There a Dose-Response Relation of Dietary Glycemic Load to Risk of Type 2 Diabetes? Meta-Analysis of Prospective Cohort Studies. Am. J. Clin. Nutr. 2013, 97, 584–596. [Google Scholar] [CrossRef]
- Sun, L.; Ranawana, D.V.; Leow, M.K.-S.; Henry, C.J. Effect of Chicken, Fat and Vegetable on Glycaemia and Insulinaemia to a White Rice-Based Meal in Healthy Adults. Eur. J. Nutr. 2014, 53, 1719–1726. [Google Scholar] [CrossRef]
- Kongkachuichai, R.; Charoensiri, R.; Meekhruerod, A.; Kettawan, A. Effect of Processing Conditions on Bioactive Compounds and Glycemic Index of the Selected Landrace Rice Variety in Pre-Diabetes. J. Cereal Sci. 2020, 94, 102994. [Google Scholar] [CrossRef]
- Se, C.-H.; Chuah, K.-A.; Mishra, A.; Wickneswari, R.; Karupaiah, T. Evaluating Crossbred Red Rice Variants for Postprandial Glucometabolic Responses: A Comparison with Commercial Varieties. Nutrients 2016, 8, 308. [Google Scholar] [CrossRef] [PubMed]
- Shobana, S.; Kokila, A.; Lakshmipriya, N.; Subhashini, S.; Ramya Bai, M.; Mohan, V.; Malleshi, N.G.; Anjana, R.M.; Henry, C.J.K.; Sudha, V. Glycaemic Index of Three Indian Rice Varieties. Int. J. Food Sci. Nutr. 2012, 63, 178–183. [Google Scholar] [CrossRef]
- Chen, Y.-J.; Sun, F.-H.; Wong, S.H.-S.; Huang, Y.-J. Glycemic Index and Glycemic Load of Selected Chinese Traditional Foods. World J. Gastroenterol. 2010, 16, 1512–1517. [Google Scholar] [CrossRef]
- Nicholas, D.; Hazila, K.K.; Chua, H.P.; Rosniyana, A. Nutritional Value and Glycemic Index of Bario Rice Varieties. J. Trop. Agric. Food Sci. 2014, 42, 1–8. [Google Scholar]
- Yang, C.-H.; Chang, C.-W.; Lin, J. White Rice Glycemic Index Measured in Venous and Capillary Blood Samples. Food Sci. Technol. Res. 2017, 23, 297–304. [Google Scholar] [CrossRef]
- Prasad, M.P.R.; Rao, B.D.; Kalpana, K.; Rao, M.V.; Patil, J.V. Glycaemic Index and Glycaemic Load of Sorghum Products. J. Sci. Food Agric. 2015, 95, 1626–1630. [Google Scholar] [CrossRef]
- RamyaBai, M.; Wedick, N.M.; Shanmugam, S.; Arumugam, K.; Nagarajan, L.; Vasudevan, K.; Gunasekaran, G.; Rajagopal, G.; Spiegelman, D.; Malik, V.; et al. Glycemic Index and Microstructure Evaluation of Four Cereal Grain Foods. J. Food Sci. 2019, 84, 3373–3382. [Google Scholar] [CrossRef]
- Ren, X.; Chen, J.; Molla, M.M.; Wang, C.; Diao, X.; Shen, Q. In Vitro Starch Digestibility and in Vivo Glycemic Response of Foxtail Millet and Its Products. Food Funct. 2016, 7, 372–379. [Google Scholar] [CrossRef]
- Kim, D.-Y.; Kim, Y.; Lim, H. Glycaemic Indices and Glycaemic Loads of Common Korean Carbohydrate-Rich Foods. Br. J. Nutr. 2019, 121, 416–425. [Google Scholar] [CrossRef] [PubMed]
- Lok, K.Y.; Chan, R.; Chan, D.; Li, L.; Leung, G.; Woo, J.; Lightowler, H.J.; Henry, C.J.K. Glycaemic Index and Glycaemic Load Values of a Selection of Popular Foods Consumed in Hong Kong. Br. J. Nutr. 2010, 103, 556–560. [Google Scholar] [CrossRef] [PubMed]
- Tan, W.S.K.; Tan, W.J.K.; Ponnalagu, S.D.O.; Koecher, K.; Menon, R.; Tan, S.-Y.; Henry, C.J. The Glycaemic Index and Insulinaemic Index of Commercially Available Breakfast and Snack Foods in an Asian Population. Br. J. Nutr. 2018, 119, 1151–1156. [Google Scholar] [CrossRef] [PubMed]
- Soong, Y.Y.; Quek, R.Y.C.; Henry, C.J. Glycemic Potency of Muffins Made with Wheat, Rice, Corn, Oat and Barley Flours: A Comparative Study between in Vivo and in Vitro. Eur. J. Nutr. 2015, 54, 1281–1285. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Wei Jie Tan, K.; Jeyakumar Henry, C. Co-Ingestion of Essence of Chicken to Moderate Glycaemic Response of Bread. Int. J. Food Sci. Nutr. 2015, 66, 931–935. [Google Scholar] [CrossRef]
- Hettiaratchi, U.P.K.; Ekanayake, S.; Welihinda, J. Glycaemic Indices of Three Sri Lankan Wheat Bread Varieties and a Bread-Lentil Meal. Int. J. Food Sci. Nutr. 2009, 60 (Suppl. S4), 21–30. [Google Scholar] [CrossRef]
- Santhi Sirisha, K.; Vijaya Lakshmi, V. Estimation of Glycemic Index of Ragi Recipes Incorporated with Curry Leaf Powder. Int. J. Recent. Adv. Multidiscip. Res. 2016, 3, 1936–1939. [Google Scholar]
- Nurjanah, N.; Mahani; Muchtadi, D.; Palupi, N.S.; Widowati, S. Chemical Characteristics and Glycemic Index of Processed Products from Corn Starch Modified with Green Tea Polyphenols. IOP Conf. Ser. Earth Environ. Sci. 2020, 443, 012029. [Google Scholar] [CrossRef]
- Molnar, C.; Gair, J. Concepts of Biology—1st Canadian Edition; BCcampus: Victoria, BC, Canada, 2015; ISBN 978-1-989623-99-2. [Google Scholar]
- Pereira, L. Edible Seaweeds of the World; CRC Press: Boca Raton, FL, USA, 2016; ISBN 978-1-4987-3050-1. [Google Scholar]
- Chan, K.; Chen, S.; Chen, P. Astaxanthin Attenuated Thrombotic Risk Factors in Type 2 Diabetic Patients. J. Funct. Foods 2019, 53, 22–27. [Google Scholar] [CrossRef]
- Peñalver, R.; Lorenzo, J.M.; Ros, G.; Amarowicz, R.; Pateiro, M.; Nieto, G. Seaweeds as a Functional Ingredient for a Healthy Diet. Mar. Drugs 2020, 18, 301. [Google Scholar] [CrossRef]
- Ferdouse, F.; Holdt, S.L.; Smith, R.; Murúa, P.; Yang, Z. The Global Status of Seaweed Production, Trade and Utilization. GLOBEFISH Res. Programme 2018, 124, 7. [Google Scholar]
- Cai, J.; Lovatelli, A.; Aguilar-Manjarrez, J.; Cornish, L.; Dabbadie, L.; Desrochers, A.; Diffey, S.; Garrido Gamarro, E.; Geehan, J.; Hurtado, A.; et al. Seaweeds and Microalgae: An Overview for Unlocking Their Potential in Global Aquaculture Development. FAO Fish. Aquac. Circ. 2021. [Google Scholar] [CrossRef]
- O’Connor, K. Seaweed: A Global History; Reaktion Books: London, UK, 2017; ISBN 978-1-78023-799-2. [Google Scholar]
- Pérez-Lloréns, J.L.; Mouritsen, O.G.; Rhatigan, P.; Cornish, M.L.; Critchley, A.T. Seaweeds in Mythology, Folklore, Poetry, and Life. J. Appl. Phycol. 2020, 32, 3157–3182. [Google Scholar] [CrossRef]
- Fukuda, S.; Saito, H.; Nakaji, S.; Yamada, M.; Ebine, N.; Tsushima, E.; Oka, E.; Kumeta, K.; Tsukamoto, T.; Tokunaga, S. Pattern of Dietary Fiber Intake among the Japanese General Population. Eur. J. Clin. Nutr. 2007, 61, 99–103. [Google Scholar] [CrossRef]
- Kim, E.; Ju, S.-Y. Asthma and Dietary Intake of Fish, Seaweeds, and Fatty Acids in Korean Adults. Nutrients 2019, 11, 2187. [Google Scholar] [CrossRef]
- Figueroa, V.; Farfán, M.; Aguilera, J.M. Seaweeds as Novel Foods and Source of Culinary Flavors. Food Rev. Int. 2021, 1–26. [Google Scholar] [CrossRef]
- Pérez-Lloréns, J.L. Seaweed Consumption in the Americas. Gastronomica 2019, 19, 49–59. [Google Scholar] [CrossRef]
- Granato, D.; Barba, F.J.; Bursać Kovačević, D.; Lorenzo, J.M.; Cruz, A.G.; Putnik, P. Functional Foods: Product Development, Technological Trends, Efficacy Testing, and Safety. Annu. Rev. Food Sci. Technol. 2020, 11, 93–118. [Google Scholar] [CrossRef]
- Mendis, E.; Kim, S.-K. Chapter 1—Present and Future Prospects of Seaweeds in Developing Functional Foods. In Advances in Food and Nutrition Research; Kim, S.-K., Ed.; Marine Medicinal Foods; Academic Press: Cambridge, MA, USA, 2011; Volume 64, pp. 1–15. [Google Scholar]
- Hayes, M. Chapter 14—Seaweeds: A Nutraceutical and Health Food. In Seaweed Sustainability; Tiwari, B.K., Troy, D.J., Eds.; Academic Press: San Diego, CA, USA, 2015; pp. 365–387. ISBN 978-0-12-418697-2. [Google Scholar]
- Vidal, A.; Fallarero, A.; De Andrade-Wartha, E.R.S.; De Oliveira e Silva, A.M.; De Lima, A.; Torres, R.P.; Vuorela, P.; Mancini-Filho, J. Composición química y actividad antioxidante del alga marina roja Bryothamnion triquetrum (S.G.Gmelin) Howe. Rev. Bras. Cienc. Farm. 2006, 42, 589–600. [Google Scholar] [CrossRef]
- Rajapakse, N.; Kim, S.-K. Chapter 2—Nutritional and Digestive Health Benefits of Seaweed. In Advances in Food and Nutrition Research; Kim, S.-K., Ed.; Marine Medicinal Foods; Academic Press: San Diego, CA, USA, 2011; Volume 64, pp. 17–28. [Google Scholar]
- Schiener, P.; Black, K.D.; Stanley, M.S.; Green, D.H. The Seasonal Variation in the Chemical Composition of the Kelp Species Laminaria Digitata, Laminaria Hyperborea, Saccharina Latissima and Alaria Esculenta. J. Appl. Phycol. 2015, 27, 363–373. [Google Scholar] [CrossRef]
- Praiboon, J.; Palakas, S.; Noiraksa, T.; Miyashita, K. Seasonal Variation in Nutritional Composition and Anti-Proliferative Activity of Brown Seaweed, Sargassum Oligocystum. J. Appl. Phycol. 2018, 30, 101–111. [Google Scholar] [CrossRef]
- Nelson, M.M.; Phleger, C.F.; Nichols, P.D. Seasonal Lipid Composition in Macroalgae of the Northeastern Pacific Ocean. Bot. Mar. 2002, 45, 58–65. [Google Scholar] [CrossRef]
- García-Poza, S.; Leandro, A.; Cotas, C.; Cotas, J.; Marques, J.C.; Pereira, L.; Gonçalves, A.M.M. The Evolution Road of Seaweed Aquaculture: Cultivation Technologies and the Industry 4.0. Int. J. Environ. Res. Public Health 2020, 17, 6528. [Google Scholar] [CrossRef] [PubMed]
- Pak, N.; Araya, H. Macroalgas Marinas Comestibles de Chile Como Fuente de Fibra Dietética: Efecto en la Digestibilidad Aparente de Proteínas, Fibra y Energía y Peso de Deposiciones en Ratas. Available online: http://www.alanrevista.org/ediciones/ediciones/1996/1/art-9/ (accessed on 4 February 2022).
- Lorenzo, J.M.; Agregán, R.; Munekata, P.E.S.; Franco, D.; Carballo, J.; Şahin, S.; Lacomba, R.; Barba, F.J. Proximate Composition and Nutritional Value of Three Macroalgae: Ascophyllum Nodosum, Fucus vesiculosus and Bifurcaria Bifurcata. Mar. Drugs 2017, 15, E360. [Google Scholar] [CrossRef] [PubMed]
- Holdt, S.L. Nutrient Reduction in Aquaculture Waste by Macroalgae Production; SDU Odense: Odense, Denmark, 2010; Available online: https://orbit.dtu.dk/en/publications/nutrient-reduction-in-aquaculture-waste-by-macroalgae-production (accessed on 2 February 2022).
- Agregán, R.; Munekata, P.E.; Domínguez, R.; Carballo, J.; Franco, D.; Lorenzo, J.M. Proximate Composition, Phenolic Content and in Vitro Antioxidant Activity of Aqueous Extracts of the Seaweeds Ascophyllum Nodosum, Bifurcaria Bifurcata and Fucus vesiculosus. Effect of Addition of the Extracts on the Oxidative Stability of Canola Oil under Accelerated Storage Conditions. Food Res. Int. 2017, 99, 986–994. [Google Scholar] [CrossRef] [PubMed]
- Alves, C.; Pinteus, S.; Simões, T.; Horta, A.; Silva, J.; Tecelão, C.; Pedrosa, R. Bifurcaria Bifurcata: A Key Macro-Alga as a Source of Bioactive Compounds and Functional Ingredients. Int. J. Food Sci. Technol. 2016, 51, 1638–1646. [Google Scholar] [CrossRef]
- Gómez-Ordóñez, E.; Jiménez-Escrig, A.; Rupérez, P. Dietary Fibre and Physicochemical Properties of Several Edible Seaweeds from the Northwestern Spanish Coast. Food Res. Int. 2010, 43, 2289–2294. [Google Scholar] [CrossRef]
- Ortiz, J.; Romero, N.; Robert, P.; Araya, J.; Lopez-Hernández, J.; Bozzo, C.; Navarrete, E.; Osorio, A.; Rios, A. Dietary Fiber, Amino Acid, Fatty Acid and Tocopherol Contents of the Edible Seaweeds Ulva Lactuca and Durvillaea Antarctica. Food Chem. 2006, 99, 98–104. [Google Scholar] [CrossRef]
- Herbreteau, F.; Coiffard, L.J.M.; Derrien, A.; Roeck-Holtzhauer, Y.D. The Fatty Acid Composition of Five Species of Macroalgae. Bot. Mar. 1997, 40, 25–28. [Google Scholar] [CrossRef]
- Díaz-Rubio, M.E.; Pérez-Jiménez, J.; Saura-Calixto, F. Dietary Fiber and Antioxidant Capacity in Fucus vesiculosus Products. Int. J. Food Sci. Nutr. 2009, 60, 23–34. [Google Scholar] [CrossRef]
- Dawczynski, C.; Schubert, R.; Jahreis, G. Amino Acids, Fatty Acids, and Dietary Fibre in Edible Seaweed Products. Food Chem. 2007, 103, 891–899. [Google Scholar] [CrossRef]
- Marsham, S.; Scott, G.W.; Tobin, M.L. Comparison of Nutritive Chemistry of a Range of Temperate Seaweeds. Food Chem. 2007, 100, 1331–1336. [Google Scholar] [CrossRef]
- Foti, M.C. Antioxidant Properties of Phenols. J. Pharma. Pharmacol. 2007, 59, 1673–1685. [Google Scholar] [CrossRef] [PubMed]
- Horn, S.J. Bioenergy from Brown Seaweeds; Fakultet for Naturvitenskap og Teknologi: Trondheim, Norway, 2000; ISBN 978-82-7984-134-0. [Google Scholar]
- Wen, X.; Peng, C.; Zhou, H.; Lin, Z.; Lin, G.; Chen, S.; Li, P. Nutritional Composition and Assessment of Gracilaria Lemaneiformis Bory. J. Int. Plant Biol. 2006, 48, 1047–1053. [Google Scholar] [CrossRef]
- Tibbetts, S.M.; Milley, J.E.; Lall, S.P. Nutritional Quality of Some Wild and Cultivated Seaweeds: Nutrient Composition, Total Phenolic Content and in Vitro Digestibility. J. Appl. Phycol. 2016, 28, 3575–3585. [Google Scholar] [CrossRef]
- Jiménez-Escrig, A.; Sánchez-Muniz, F.J. Dietary Fibre from Edible Seaweeds: Chemical Structure, Physicochemical Properties and Effects on Cholesterol Metabolism. Nutr. Res. 2000, 20, 585–598. [Google Scholar] [CrossRef]
- Rupérez, P.; Saura-Calixto, F. Dietary Fibre and Physicochemical Properties of Edible Spanish Seaweeds. Eur. Food Res. Technol. 2001, 212, 349–354. [Google Scholar] [CrossRef]
- Cofrades, S.; López-López, I.; Bravo, L.; Ruiz-Capillas, C.; Bastida, S.; Larrea, M.T.; Jiménez-Colmenero, F. Nutritional and Antioxidant Properties of Different Brown and Red Spanish Edible Seaweeds. Food Sci. Technol. Int. 2010, 16, 361–370. [Google Scholar] [CrossRef]
- Simpson, F.J.; Shacklock, P.F. The Cultivation of Chondrus Crispus. Effect of Temperature on Growth and Carrageenan Production. Bot. Mar. 1979, 22, 295–298. [Google Scholar] [CrossRef]
- Rupérez, P. Mineral Content of Edible Marine Seaweeds. Food Chem. 2002, 79, 23–26. [Google Scholar] [CrossRef]
- Denis, C.; Morançais, M.; Li, M.; Deniaud, E.; Gaudin, P.; Wielgosz-Collin, G.; Barnathan, G.; Jaouen, P.; Fleurence, J. Study of the Chemical Composition of Edible Red Macroalgae Grateloupia Turuturu from Brittany (France). Food Chem. 2010, 119, 913–917. [Google Scholar] [CrossRef]
- Khairy, H.M.; El-Shafay, S.M. Seasonal variations in the biochemical composition of some common seaweed species from the coast of Abu Qir Bay, Alexandria, Egypt. Oceanologia 2013, 55, 435–452. [Google Scholar] [CrossRef]
- Mohy El-Din, S.M.; El-Ahwany, A.M.D. Bioactivity and Phytochemical Constituents of Marine Red Seaweeds (Jania Rubens, Corallina Mediterranea and Pterocladia Capillacea). J. Taibah Univ. Sci. 2016, 10, 471–484. [Google Scholar] [CrossRef]
- Ortiz, J.; Uquiche, E.; Robert, P.; Romero, N.; Quitral, V.; Llantén, C. Functional and Nutritional Value of the Chilean Seaweeds Codium Fragile, Gracilaria Chilensis and Macrocystis Pyrifera. Eur. J. Lipid Sci. Technol. 2009, 111, 320–327. [Google Scholar] [CrossRef]
- Matanjun, P.; Mohamed, S.; Mustapha, N.M.; Muhammad, K. Nutrient Content of Tropical Edible Seaweeds, Eucheuma Cottonii, Caulerpa Lentillifera and Sargassum Polycystum. J. Appl. Phycol. 2009, 21, 75–80. [Google Scholar] [CrossRef]
- Nguyen, V.T.; Ueng, J.-P.; Tsai, G.-J. Proximate Composition, Total Phenolic Content, and Antioxidant Activity of Seagrape (Caulerpa Lentillifera). J. Food Sci. 2011, 76, C950–C958. [Google Scholar] [CrossRef]
- Peña-Rodríguez, A.; Mawhinney, T.P.; Ricque-Marie, D.; Cruz-Suárez, L.E. Chemical Composition of Cultivated Seaweed Ulva Clathrata (Roth) C. Agardh. Food Chem. 2011, 129, 491–498. [Google Scholar] [CrossRef]
- Yaich, H.; Garna, H.; Besbes, S.; Paquot, M.; Blecker, C.; Attia, H. Chemical Composition and Functional Properties of Ulva Lactuca Seaweed Collected in Tunisia. Food Chem. 2011, 128, 895–901. [Google Scholar] [CrossRef]
- Nwosu, F.; Morris, J.; Lund, V.A.; Stewart, D.; Ross, H.A.; McDougall, G.J. Anti-Proliferative and Potential Anti-Diabetic Effects of Phenolic-Rich Extracts from Edible Marine Algae. Food Chem. 2011, 126, 1006–1012. [Google Scholar] [CrossRef]
- Brownlee, I.A.; Allen, A.; Pearson, J.P.; Dettmar, P.W.; Havler, M.E.; Atherton, M.R.; Onsøyen, E. Alginate as a Source of Dietary Fiber. Crit. Rev. Food Sci. Nutr. 2005, 45, 497–510. [Google Scholar] [CrossRef]
- Draget, K.I.; Skjåk-Bræk, G.; Smidsrød, O. Alginate Based New Materials. Int. J. Biol. Macromol. 1997, 21, 47–55. [Google Scholar] [CrossRef]
- Hoad, C.L.; Rayment, P.; Spiller, R.C.; Marciani, L.; de Celis Alonso, B.; Traynor, C.; Mela, D.J.; Peters, H.P.F.; Gowland, P.A. In Vivo Imaging of Intragastric Gelation and Its Effect on Satiety in Humans. J. Nutr. 2004, 134, 2293–2300. [Google Scholar] [CrossRef] [PubMed]
- Solah, V.A.; Kerr, D.A.; Adikara, C.D.; Meng, X.; Binns, C.W.; Zhu, K.; Devine, A.; Prince, R.L. Differences in Satiety Effects of Alginate- and Whey Protein-Based Foods. Appetite 2010, 54, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Sim, S.Y.; Noor Aziah, A.A.; Cheng, L.H. Characteristics of Wheat Dough and Chinese Steamed Bread Added with Sodium Alginates or Konjac Glucomannan. Food Hydrocoll. 2011, 25, 951–957. [Google Scholar] [CrossRef]
- Nishide, E.; Uchida, N. Effects of Ulva Powder on the Ingestion and Excretion of Cholesterol in Rats. In Proceedings of the 17th International Seaweed Symposium, Cape Town, South Africa, 28 January–2 February 2001; Oxford University Press: Oxford, UK, 2003; pp. 165–168. [Google Scholar]
- Kim, H.; Lee, J.-H. Antimicrobial Activities against Methicillin-Resistant Staphylococcus Aureus from Macroalgae. J. Industrial Eng. Chem. 2008, 14, 568–572. [Google Scholar] [CrossRef]
- Murata, M.; Nakazoe, J. Production and Use of Marine AIgae in Japan. Jpn. Agric. Res. Q. 2001, 35, 281–290. [Google Scholar] [CrossRef]
- Torsdottir, I.; Alpsten, M.; Holm, G.; Sandberg, A.-S.; Tölli, J. A Small Dose of Soluble Alginate-Fiber Affects Postprandial Glycemia and Gastric Emptying in Humans with Diabetes. J. Nutr. 1991, 121, 795–799. [Google Scholar] [CrossRef] [PubMed]
- Idota, Y.; Kato, T.; Shiragami, K.; Koike, M.; Yokoyama, A.; Takahashi, H.; Yano, K.; Ogihara, T. Mechanism of Suppression of Blood Glucose Level by Calcium Alginate in Rats. Biol. Pharma. Bull. 2018. [Google Scholar] [CrossRef] [PubMed]
- Idota, Y.; Kogure, Y.; Kato, T.; Yano, K.; Arakawa, H.; Miyajima, C.; Kasahara, F.; Ogihara, T. Relationship between Physical Parameters of Various Metal Ions and Binding Affinity for Alginate. Biol. Pharm. Bull. 2016, 39, 1893–1896. [Google Scholar] [CrossRef]
- Huang, Z.; Wang, Y.; Shafer, R.; Winn, N.C.; Kanaley, J.A.; Vardhanabhuti, B. Glycemic Effects Following the Consumption of Mixed Soy Protein Isolate and Alginate Beverages in Healthy Adults. Food Funct. 2019, 10, 1718–1725. [Google Scholar] [CrossRef]
- El Khoury, D.; Goff, H.D.; Berengut, S.; Kubant, R.; Anderson, G.H. Effect of Sodium Alginate Addition to Chocolate Milk on Glycemia, Insulin, Appetite and Food Intake in Healthy Adult Men. Eur. J. Clin. Nutr. 2014, 68, 613–618. [Google Scholar] [CrossRef] [PubMed]
- Georg Jensen, M.; Kristensen, M.; Astrup, A. Effect of Alginate Supplementation on Weight Loss in Obese Subjects Completing a 12-Wk Energy-Restricted Diet: A Randomized Controlled Trial. Am. J. Clin. Nutr. 2012, 96, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.A.; Lai, C.-S.; Corwin, H.; Ma, Y.; Maki, K.C.; Garleb, K.A.; Wolf, B.W. Inclusion of Guar Gum and Alginate into a Crispy Bar Improves Postprandial Glycemia in Humans. J. Nutr. 2004, 134, 886–889. [Google Scholar] [CrossRef] [PubMed]
- Kato, T.; Idota, Y.; Shiragami, K.; Koike, M.; Nishibori, F.; Tomokane, M.; Seki, T.; Itabashi, K.; Hakoda, K.; Takahashi, H.; et al. Randomized, Double-Blind, Crossover Clinical Trial of the Effect of Calcium Alginate in Noodles on Postprandial Blood Glucose Level. Biol. Pharm. Bull. 2018, 41, 1367–1371. [Google Scholar] [CrossRef] [PubMed]
- Wolf, B.W.; Lai, C.-S.; Kipnes, M.S.; Ataya, D.G.; Wheeler, K.B.; Zinker, B.A.; Garleb, K.A.; Firkins, J.L. Glycemic and Insulinemic Responses of Nondiabetic Healthy Adult Subjects to an Experimental Acid-Induced Viscosity Complex Incorporated into a Glucose Beverage. Nutrition 2002, 18, 621–626. [Google Scholar] [CrossRef]
- Fitton, J.H.; Stringer, D.N.; Karpiniec, S.S. Therapies from Fucoidan: An Update. Mar. Drugs 2015, 13, 5920–5946. [Google Scholar] [CrossRef]
- Skriptsova, A.V.; Shevchenko, N.M.; Zvyagintseva, T.N.; Imbs, T.I. Monthly Changes in the Content and Monosaccharide Composition of Fucoidan from Undaria Pinnatifida (Laminariales, Phaeophyta). J. Appl. Phycol. 2010, 22, 79–86. [Google Scholar] [CrossRef]
- Ale, M.T.; Meyer, A.S. Fucoidans from Brown Seaweeds: An Update on Structures, Extraction Techniques and Use of Enzymes as Tools for Structural Elucidation. RSC Adv. 2013, 3, 8131–8141. [Google Scholar] [CrossRef]
- Senthilkumar, K.; Manivasagan, P.; Venkatesan, J.; Kim, S.-K. Brown Seaweed Fucoidan: Biological Activity and Apoptosis, Growth Signaling Mechanism in Cancer. Int. J. Biol. Macromol. 2013, 60, 366–374. [Google Scholar] [CrossRef]
- Fletcher, H.R.; Biller, P.; Ross, A.B.; Adams, J.M.M. The Seasonal Variation of Fucoidan within Three Species of Brown Macroalgae. Algal Res. 2017, 22, 79–86. [Google Scholar] [CrossRef]
- Mabate, B.; Daub, C.D.; Malgas, S.; Edkins, A.L.; Pletschke, B.I. Fucoidan Structure and Its Impact on Glucose Metabolism: Implications for Diabetes and Cancer Therapy. Mar. Drugs 2021, 19, 30. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.-T.; Rioux, L.-E.; Turgeon, S. Molecular Weight and Sulfate Content Modulate the Inhibition of α-Amylase by Fucoidan Relevant for Type 2 Diabetes Management. Pharma. Nutr. 2015, 3. [Google Scholar] [CrossRef]
- Shan, X.; Liu, X.; Hao, J.; Cai, C.; Fan, F.; Dun, Y.; Zhao, X.; Liu, X.; Li, C.; Yu, G. In Vitro and in Vivo Hypoglycemic Effects of Brown Algal Fucoidans. Int. J. Biol. Macromol. 2016, 82, 249–255. [Google Scholar] [CrossRef]
- Ponce, N.M.A.; Stortz, C.A. A Comprehensive and Comparative Analysis of the Fucoidan Compositional Data Across the Phaeophyceae. Front. Plant Sci. 2020, 11. [Google Scholar] [CrossRef]
- Wang, Y.; Xing, M.; Cao, Q.; Ji, A.; Liang, H.; Song, S. Biological Activities of Fucoidan and the Factors Mediating Its Therapeutic Effects: A Review of Recent Studies. Mar. Drugs 2019, 17, 183. [Google Scholar] [CrossRef]
- Suprunchuk, V.E. Low-Molecular-Weight Fucoidan: Chemical Modification, Synthesis of Its Oligomeric Fragments and Mimetics. Carbohydr. Res. 2019, 485, 107806. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, J.; Zhao, Y.; Hu, S.; Shi, D.; Xue, C. Fucoidan from Sea Cucumber Cucumaria Frondosa Exhibits Anti-Hyperglycemic Effects in Insulin Resistant Mice via Activating the PI3K/PKB Pathway and GLUT4. J. Biosci. Bioeng. 2016, 121, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Zheng, Y.; Wang, J.; Ma, S.; Yu, Y.; White, W.L.; Yang, S.; Yang, F.; Lu, J. Fucoidan Extracted from Undaria Pinnatifida: Source for Nutraceuticals/Functional Foods. Mar. Drugs 2018, 16, 321. [Google Scholar] [CrossRef] [PubMed]
- Sakai, C.; Abe, S.; Kouzuki, M.; Shimohiro, H.; Ota, Y.; Sakinada, H.; Takeuchi, T.; Okura, T.; Kasagi, T.; Hanaki, K. A Randomized Placebo-Controlled Trial of an Oral Preparation of High Molecular Weight Fucoidan in Patients with Type 2 Diabetes with Evaluation of Taste Sensitivity. Yonago Acta Med. 2019, 62, 14–23. [Google Scholar] [CrossRef]
- Kim, K.-J.; Yoon, K.-Y.; Lee, B.-Y. Fucoidan Regulate Blood Glucose Homeostasis in C57BL/KSJ M+/+db and C57BL/KSJ Db/Db Mice. Fitoterapia 2012, 83, 1105–1109. [Google Scholar] [CrossRef]
- Koh, H.S.A.; Lu, J.; Zhou, W. Structural Dependence of Sulfated Polysaccharide for Diabetes Management: Fucoidan From Undaria Pinnatifida Inhibiting α-Glucosidase More Strongly Than α-Amylase and Amyloglucosidase. Front. Pharm. 2020, 11, 831. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.-T.; Rioux, L.-E.; Turgeon, S.L. Alpha-Amylase and Alpha-Glucosidase Inhibition Is Differentially Modulated by Fucoidan Obtained from Fucus vesiculosus and Ascophyllum Nodosum. Phytochemistry 2014. [Google Scholar] [CrossRef] [PubMed]
- Daub, C.D.; Mabate, B.; Malgas, S.; Pletschke, B.I. Fucoidan from Ecklonia Maxima Is a Powerful Inhibitor of the Diabetes-Related Enzyme, α-Glucosidase. Int. J. Biol. Macromol. 2020, 151, 412–420. [Google Scholar] [CrossRef] [PubMed]
- January, G.G.; Naidoo, R.K.; Kirby-McCullough, B.; Bauer, R. Assessing Methodologies for Fucoidan Extraction from South African Brown Algae. Algal Res. 2019, 40, 101517. [Google Scholar] [CrossRef]
- Senthil, S.L.; Kumar, T.V.; Geetharamani, D.; Maruthupandi, T. Screening of Seaweeds Collected from Southeast Coastal Area of India for α-Amylase Inhibitory Activity, Antioxidant Activity and Biocompatibility. Int. J. Pharm. Pharm. Sci 2013, 5, 240–244. [Google Scholar]
- Hu, S.; Xia, G.; Wang, J.; Wang, Y.; Li, Z.; Xue, C. Fucoidan from Sea Cucumber Protects against High-Fat High-Sucrose Diet-Induced Hyperglycaemia and Insulin Resistance in Mice. J. Funct. Foods 2014, 10, 128–138. [Google Scholar] [CrossRef]
- Pozharitskaya, O.N.; Obluchinskaya, E.D.; Shikov, A.N. Mechanisms of Bioactivities of Fucoidan from the Brown Seaweed Fucus vesiculosus L. of the Barents Sea. Mar. Drugs 2020, 18, 275. [Google Scholar] [CrossRef]
- Unnikrishnan, P.S.; Suthindhiran, K.; Jayasri, M.A. Inhibitory Potential of Turbinaria Ornata against Key Metabolic Enzymes Linked to Diabetes. Biomed. Res. Int. 2014, 2014, 783895. [Google Scholar] [CrossRef]
- Havale, S.H.; Pal, M. Medicinal Chemistry Approaches to the Inhibition of Dipeptidyl Peptidase-4 for the Treatment of Type 2 Diabetes. Bioorg. Med. Chem. 2009, 17, 1783–1802. [Google Scholar] [CrossRef] [PubMed]
- Murray, M.; Dordevic, A.L.; Ryan, L.; Bonham, M.P. The Impact of a Single Dose of a Polyphenol-Rich Seaweed Extract on Postprandial Glycaemic Control in Healthy Adults: A Randomised Cross-Over Trial. Nutrients 2018, 10, 270. [Google Scholar] [CrossRef]
- Hernández-Corona, D.M.; Martínez-Abundis, E.; González-Ortiz, M. Effect of Fucoidan Administration on Insulin Secretion and Insulin Resistance in Overweight or Obese Adults. J. Med. Food 2014, 17, 830–832. [Google Scholar] [CrossRef] [PubMed]
- Kadam, S.U.; Tiwari, B.K.; O’Donnell, C.P. Extraction, Structure and Biofunctional Activities of Laminarin from Brown Algae. Int. J. Food Sci. Technol. 2015, 50, 24–31. [Google Scholar] [CrossRef]
- Lee, J.Y.; Kim, Y.-J.; Kim, H.J.; Kim, Y.-S.; Park, W. Immunostimulatory Effect of Laminarin on RAW 264.7 Mouse Macrophages. Molecules 2012, 17, 5404–5411. [Google Scholar] [CrossRef] [PubMed]
- Trigueiro, T.G.; Pereira, D.C.; Martins, A.P.; Colepicolo, P.; Marinho-Soriano, E. Cultivation of Three Color Strains of Gracilaria Domingensis in an Integrated Organic System. Int. Aquat. Res. 2017, 9, 225–233. [Google Scholar] [CrossRef][Green Version]
- Chen, H.-M.; Zheng, L.; Yan, X.-J. The Preparation and Bioactivity Research of Agaro-Oligosaccharides. Food Technol. Biotechnol. 2005, 43, 29–36. [Google Scholar]
- Enoki, T.; Sagawa, H.; Tominaga, T.; Nishiyama, E.; Koyama, N.; Sakai, T.; Yu, F.-G.; Ikai, K.; Kato, I. Drugs, Foods or Drinks with the Use of Algae-Derived Physiologically Active Substances 1999. Available online: https://patentscope.wipo.int/search/en/detail.jsf?docId=WO1999024447 (accessed on 23 January 2022).
- Kidgell, J.T.; Magnusson, M.; De Nys, R.; Glasson, C.R.K. Ulvan: A Systematic Review of Extraction, Composition and Function. Algal Res. 2019, 39, 101422. [Google Scholar] [CrossRef]
- Skoler-Karpoff, S.; Ramjee, G.; Ahmed, K.; Altini, L.; Plagianos, M.G.; Friedland, B.; Govender, S.; Kock, A.D.; Cassim, N.; Palanee, T.; et al. Efficacy of Carraguard for Prevention of HIV Infection in Women in South Africa: A Randomised, Double-Blind, Placebo-Controlled Trial. Lancet 2008, 372, 1977–1987. [Google Scholar] [CrossRef]
- Cáceres, P.J.; Carlucci, M.J.; Damonte, E.B.; Matsuhiro, B.; Zúñiga, E.A. Carrageenans from Chilean Samples of Stenogramme Interrupta (Phyllophoraceae): Structural Analysis and Biological Activity. Phytochemistry 2000, 53, 81–86. [Google Scholar] [CrossRef]
- Groult, H.; Cousin, R.; Chot-Plassot, C.; Maura, M.; Bridiau, N.; Piot, J.-M.; Maugard, T.; Fruitier-Arnaudin, I. λ-Carrageenan Oligosaccharides of Distinct Anti-Heparanase and Anticoagulant Activities Inhibit MDA-MB-231 Breast Cancer Cell Migration. Mar. Drugs 2019, 17, 140. [Google Scholar] [CrossRef]
- Bhattacharyya, S.; Feferman, L.; Unterman, T.; Tobacman, J.K. Exposure to Common Food Additive Carrageenan Alone Leads to Fasting Hyperglycemia and in Combination with High Fat Diet Exacerbates Glucose Intolerance and Hyperlipidemia without Effect on Weight. J. Diabetes Res. 2015, 2015, 513429. [Google Scholar] [CrossRef]
- Paradis, M.-E.; Couture, P.; Lamarche, B. A Randomised Crossover Placebo-Controlled Trial Investigating the Effect of Brown Seaweed (Ascophyllum Nodosum and Fucus vesiculosus) on Postchallenge Plasma Glucose and Insulin Levels in Men and Women. Appl. Physiol. Nutr. Metab. 2011, 36, 913–919. [Google Scholar] [CrossRef]
- Yoshinaga, K.; Mitamura, R. Effects of Undaria Pinnatifida (Wakame) on Postprandial Glycemia and Insulin Levels in Humans: A Randomized Crossover Trial. Plant Foods Hum. Nutr. 2019, 74, 461–467. [Google Scholar] [CrossRef]
- Tanemura, Y.; Yamanaka-Okumura, H.; Sakuma, M.; Nii, Y.; Taketani, Y.; Takeda, E. Effects of the Intake of Undaria Pinnatifida (Wakame) and Its Sporophylls (Mekabu) on Postprandial Glucose and Insulin Metabolism. J. Med. Investig. 2014, 61, 291–297. [Google Scholar] [CrossRef]
- Mohamed, S.; Hashim, S.N.; Rahman, H.A. Seaweeds: A Sustainable Functional Food for Complementary and Alternative Therapy. Trends Food Sci. Technol. 2012, 23, 83–96. [Google Scholar] [CrossRef]
- Plaza, M.; Cifuentes, A.; Ibáñez, E. In the Search of New Functional Food Ingredients from Algae. Trends Food Sci. Technol. 2008, 19, 31–39. [Google Scholar] [CrossRef]
- Xia, E.-Q.; Zhu, S.-S.; He, M.-J.; Luo, F.; Fu, C.-Z.; Zou, T.-B. Marine Peptides as Potential Agents for the Management of Type 2 Diabetes Mellitus—A Prospect. Mar. Drugs 2017, 15, 88. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Huertas, E. Health Effects of Oleic Acid and Long Chain Omega-3 Fatty Acids (EPA and DHA) Enriched Milks. A Review of Intervention Studies. Pharmacol. Res. 2010, 61, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Kumari, P.; Kumar, M.; Gupta, V.; Reddy, C.R.K.; Jha, B. Tropical Marine Macroalgae as Potential Sources of Nutritionally Important PUFAs. Food Chem. 2010, 120, 749–757. [Google Scholar] [CrossRef]
- Sánchez-Machado, D.I.; López-Cervantes, J.; López-Hernández, J.; Paseiro-Losada, P. Fatty Acids, Total Lipid, Protein and Ash Contents of Processed Edible Seaweeds. Food Chem. 2004, 85, 439–444. [Google Scholar] [CrossRef]
- Sirot, V.; Dumas, C.; Desquilbet, L.; Mariotti, F.; Legrand, P.; Catheline, D.; Leblanc, J.-C.; Margaritis, I. A Restricted Cubic Spline Approach to Assess the Association between High Fat Fish Intake and Red Blood Cell EPA + DHA Content. Nutr. Metab. Cardiovasc. Dis. 2012, 22, 318–326. [Google Scholar] [CrossRef]
- Lu, H.; Nielsen, N.; Baron, C.; Jacobsen, C. Marine Phospholipids: The Current Understanding of Their Oxidation Mechanisms and Potential Uses for Food Fortification. Crit. Rev. Food Sci. Nutr. 2015, 57. [Google Scholar] [CrossRef] [PubMed]
- Payghami, N.; Jamili, S.; Rustaiyan, A.; Saeidnia, S.; Nikan, M.; Gohari, A.R. Alpha-Amylase Inhibitory Activity and Sterol Composition of the Marine Algae, Sargassum Glaucescens. Pharma. Res. 2015, 7, 314. [Google Scholar]
- Jung, H.A.; Islam, M.N.; Lee, C.M.; Oh, S.H.; Lee, S.; Jung, J.H.; Choi, J.S. Kinetics and Molecular Docking Studies of an Anti-Diabetic Complication Inhibitor Fucosterol from Edible Brown Algae Eisenia Bicyclis and Ecklonia Stolonifera. Chem. Biol. Int. 2013, 206, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Yang, C.; Liu, B.; Lin, L.; Sarker, S.D.; Nahar, L.; Yu, H.; Cao, H.; Xiao, J. Bioactive Compounds from Marine Macroalgae and Their Hypoglycemic Benefits. Trends Food Sci. Technol. 2018, 72, 1–12. [Google Scholar] [CrossRef]
- Gabbia, D.; De Martin, S. Brown Seaweeds for the Management of Metabolic Syndrome and Associated Diseases. Molecules 2020, 25, 4182. [Google Scholar] [CrossRef] [PubMed]
- Lopes, G.; Barbosa, M.; Andrade, P.B.; Valentão, P. Phlorotannins from Fucales: Potential to Control Hyperglycemia and Diabetes-Related Vascular Complications. J. Appl. Phycol. 2019, 31, 3143–3152. [Google Scholar] [CrossRef]
- Liu, M.; Hansen, P.E.; Lin, X. Bromophenols in Marine Algae and Their Bioactivities. Mar. Drugs 2011, 9, 1273–1292. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-H.; Jeon, Y.-J. Anti-Diabetic Effects of Brown Algae Derived Phlorotannins, Marine Polyphenols through Diverse Mechanisms. Fitoterapia 2013, 86, 129–136. [Google Scholar] [CrossRef]
- Roy, M.-C.; Anguenot, R.; Fillion, C.; Beaulieu, M.; Bérubé, J.; Richard, D. Effect of a Commercially-Available Algal Phlorotannins Extract on Digestive Enzymes and Carbohydrate Absorption in Vivo. Food Res. Int. 2011, 44, 3026–3029. [Google Scholar] [CrossRef]
- Wright, S.W.; Jeffrey, S.W. Pigment Markers for Phytoplankton Production. In Marine Organic Matter: Biomarkers, Isotopes and DNA; Springer: Berlin, Germany, 2006; pp. 71–104. [Google Scholar]
- Zhang, Y.; Fang, H.; Xie, Q.; Sun, J.; Liu, R.; Hong, Z.; Yi, R.; Wu, H. Comparative Evaluation of the Radical-Scavenging Activities of Fucoxanthin and Its Stereoisomers. Molecules 2014, 19, 2100–2113. [Google Scholar] [CrossRef]
- Bae, M.; Kim, H. The Role of Vitamin C, Vitamin D, and Selenium in Immune System against COVID-19. Molecules 2020, 25, 5346. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H.; Hosokawa, M.; Sashima, T.; Miyashita, K. Dietary Combination of Fucoxanthin and Fish Oil Attenuates the Weight Gain of White Adipose Tissue and Decreases Blood Glucose in Obese/Diabetic KK-Ay Mice. J. Agric. Food Chem. 2007, 55, 7701–7706. [Google Scholar] [CrossRef] [PubMed]
- Hosokawa, M.; Miyashita, T.; Nishikawa, S.; Emi, S.; Tsukui, T.; Beppu, F.; Okada, T.; Miyashita, K. Fucoxanthin Regulates Adipocytokine MRNA Expression in White Adipose Tissue of Diabetic/Obese KK-Ay Mice. Arch. Biochem. Biophys. 2010, 504, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Zaharudin, N.; Staerk, D.; Dragsted, L.O. Inhibition o.of α-Glucosidase Activity by Selected Edible Seaweeds and Fucoxanthin. Food Chem. 2019, 270, 481–486. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H.; Hosokawa, M.; Sashima, T.; Murakami-Funayama, K.; Miyashita, K. Anti-Obesity and Anti-Diabetic Effects of Fucoxanthin on Diet-Induced Obesity Conditions in a Murine Model. Mol. Med. Rep. 2009, 2, 897–902. [Google Scholar] [CrossRef]
- Laleg, K.; Barron, C.; Santé-Lhoutellier, V.; Walrand, S.; Micard, V. Protein Enriched Pasta: Structure and Digestibility of Its Protein Network. Food Funct. 2016, 7, 1196–1207. [Google Scholar] [CrossRef]
- Lu, Z.-H.; Donner, E.; Yada, R.Y.; Liu, Q. Physicochemical Properties and in Vitro Starch Digestibility of Potato Starch/Protein Blends. Carbohydr. Polym. 2016, 154, 214–222. [Google Scholar] [CrossRef]
- Yang, C.; Zhong, F.; Douglas Goff, H.; Li, Y. Study on Starch-Protein Interactions and Their Effects on Physicochemical and Digestible Properties of the Blends. Food Chem. 2019, 280, 51–58. [Google Scholar] [CrossRef]
- Echave, J.; Fraga-Corral, M.; Garcia-Perez, P.; Popović-Djordjević, J.; Avdović, E.H.; Radulović, M.; Xiao, J.; Prieto, M.A.; Simal-Gandara, J. Seaweed Protein Hydrolysates and Bioactive Peptides: Extraction, Purification, and Applications. Mar. Drugs 2021, 19, 500. [Google Scholar] [CrossRef]
- Badmus, U.O.; Taggart, M.A.; Boyd, K.G. The Effect of Different Drying Methods on Certain Nutritionally Important Chemical Constituents in Edible Brown Seaweeds. J. Appl. Phycol. 2019, 31, 3883–3897. [Google Scholar] [CrossRef]
- Debet, M.R.; Gidley, M.J. Three Classes of Starch Granule Swelling: Influence of Surface Proteins and Lipids. Carbohydr. Polym. 2006, 64, 452–465. [Google Scholar] [CrossRef]
- Ai, Y.; Hasjim, J.; Jane, J. Effects of Lipids on Enzymatic Hydrolysis and Physical Properties of Starch. Carbohydr. Polym. 2013, 92, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Holm, J.; Björck, I.; Ostrowska, S.; Eliasson, A.-C.; Asp, N.-G.; Larsson, K.; Lundquist, I. Digestibility of Amylose-Lipid Complexes in-Vitro and in-Vivo. Starch 1983, 35, 294–297. [Google Scholar] [CrossRef]
- Lau, E.; Zhou, W.; Henry, C.J. Effect of Fat Type in Baked Bread on Amylose-Lipid Complex Formation and Glycaemic Response. Br. J. Nutr. 2016, 115, 2122–2129. [Google Scholar] [CrossRef] [PubMed]
- Koh, L.W.; Kasapis, S.; Lim, K.M.; Foo, C.W. Structural Enhancement Leading to Retardation of in Vitro Digestion of Rice Dough in the Presence of Alginate. Food Hydrocoll. 2009, 23, 1458–1464. [Google Scholar] [CrossRef]
- Marcano, J.; Hernando, I.; Fiszman, S. In Vitro Measurements of Intragastric Rheological Properties and Their Relationships with the Potential Satiating Capacity of Cheese Pies with Konjac Glucomannan. Food Hydrocoll. 2015, 51. [Google Scholar] [CrossRef]
- Regand, A.; Chowdhury, Z.; Tosh, S.M.; Wolever, T.M.S.; Wood, P. The Molecular Weight, Solubility and Viscosity of Oat Beta-Glucan Affect Human Glycemic Response by Modifying Starch Digestibility. Food Chem. 2011, 129, 297–304. [Google Scholar] [CrossRef]
- Dhital, S.; Gidley, M.J.; Warren, F.J. Inhibition of α-Amylase Activity by Cellulose: Kinetic Analysis and Nutritional Implications. Carbohydr. Polym. 2015, 123, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.A.; Islam, M.N.; Lee, C.M.; Jeong, H.O.; Chung, H.Y.; Woo, H.C.; Choi, J.S. Promising Antidiabetic Potential of Fucoxanthin Isolated from the Edible Brown Algae Eisenia Bicyclis and Undaria Pinnatifida. Fish. Sci. 2012, 78, 1321–1329. [Google Scholar] [CrossRef]
- Harden, C.J.; Craig Richardson, J.; Dettmar, P.W.; Corfe, B.M.; Paxman, J.R. An Ionic-Gelling Alginate Drink Attenuates Postprandial Glycaemia in Males. J. Funct. Foods 2012, 4, 122–128. [Google Scholar] [CrossRef]
- Clegg, M.E.; Pratt, M.; Markey, O.; Shafat, A.; Henry, C.J.K. Addition of Different Fats to a Carbohydrate Food: Impact on Gastric Emptying, Glycaemic and Satiety Responses and Comparison with in Vitro Digestion. J. Funct. Foods 2012, 48, 91–97. [Google Scholar] [CrossRef]
- Henry, C.J.K.; Lightowler, H.J.; Newens, K.J.; Pata, N. The Influence of Adding Fats of Varying Saturation on the Glycaemic Response of White Bread. Int. J. Food Sci. Nutr. 2008, 59, 61–69. [Google Scholar] [CrossRef]
- Hätönen, K.A.; Virtamo, J.; Eriksson, J.G.; Sinkko, H.K.; Sundvall, J.E.; Valsta, L.M. Protein and Fat Modify the Glycaemic and Insulinaemic Responses to a Mashed Potato-Based Meal. Br. J. Nutr. 2011, 106, 248–253. [Google Scholar] [CrossRef]
- Akhavan, T.; Luhovyy, B.L.; Panahi, S.; Kubant, R.; Brown, P.H.; Anderson, G.H. Mechanism of Action of Pre-Meal Consumption of Whey Protein on Glycemic Control in Young Adults. J. Nutr. Biochem. 2014, 25, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Ye, A.; Cui, J.; Dalgleish, D.; Singh, H. The Formation and Breakdown of Structured Clots from Whole Milk during Gastric Digestion. Food Funct. 2016, 7, 4259–4266. [Google Scholar] [CrossRef]
- Quek, R.; Bi, X.; Henry, C.J. Impact of Protein-Rich Meals on Glycaemic Response of Rice. Br. J. Nutr. 2016, 115, 1194–1201. [Google Scholar] [CrossRef]
- Sugiyama, M.; Tang, A.C.; Wakaki, Y.; Koyama, W. Glycemic Index of Single and Mixed Meal Foods among Common Japanese Foods with White Rice as a Reference Food. Eur. J. Clin. Nutr. 2003, 57, 743–752. [Google Scholar] [CrossRef]
- Desterke, C.; Turhan, A.G.; Bennaceur-Griscelli, A.; Griscelli, F. PPARγ Cistrome Repression during Activation of Lung Monocyte-Macrophages in Severe COVID-19. iScience 2020, 23, 101611. [Google Scholar] [CrossRef] [PubMed]
- Quitral, V.; Sepúlveda, M.; Gamero-Vega, G.; Jiménez, P. Seaweeds in Bakery and Farinaceous Foods: A Mini-Review. Int. J. Food Sci. 2021, 100403. [Google Scholar] [CrossRef]
- Cox, S.; Abu-Ghannam, N. Incorporation of Himanthalia Elongata Seaweed to Enhance the Phytochemical Content of Breadsticks Using Response Surface Methodology (RSM). Int. Food Res. J. 2013, 40, 1537–1545. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).