Characteristic Volatiles and Cultivar Classification in 35 Apple Varieties: A Case Study of Two Harvest Years
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Chemicals
2.3. HS-SPME
2.4. GC-MS Analysis
2.5. Compound Qualification and Quantification
2.6. Calculation of OAV
2.7. Statistical Analysis
3. Results and Discussion
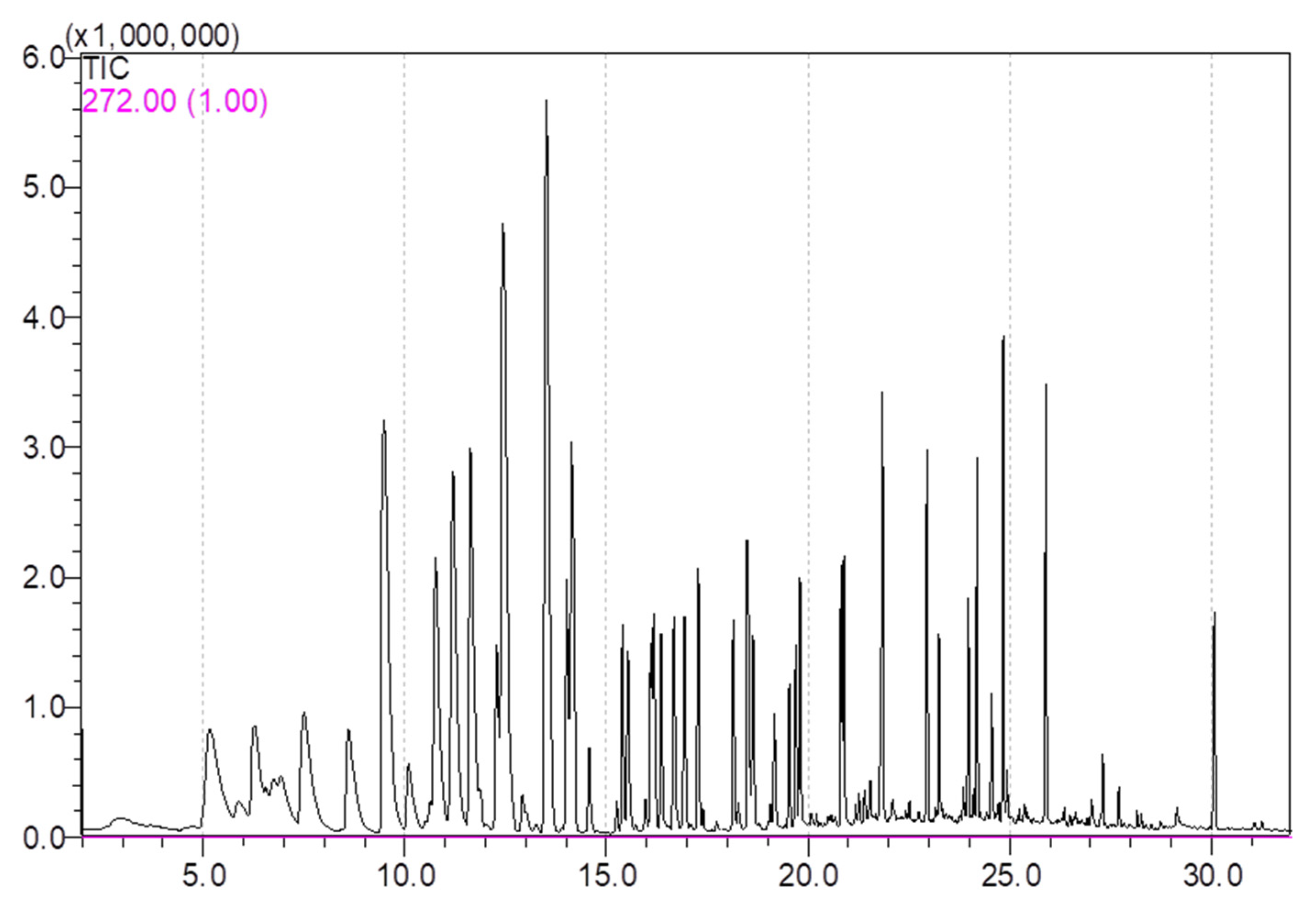
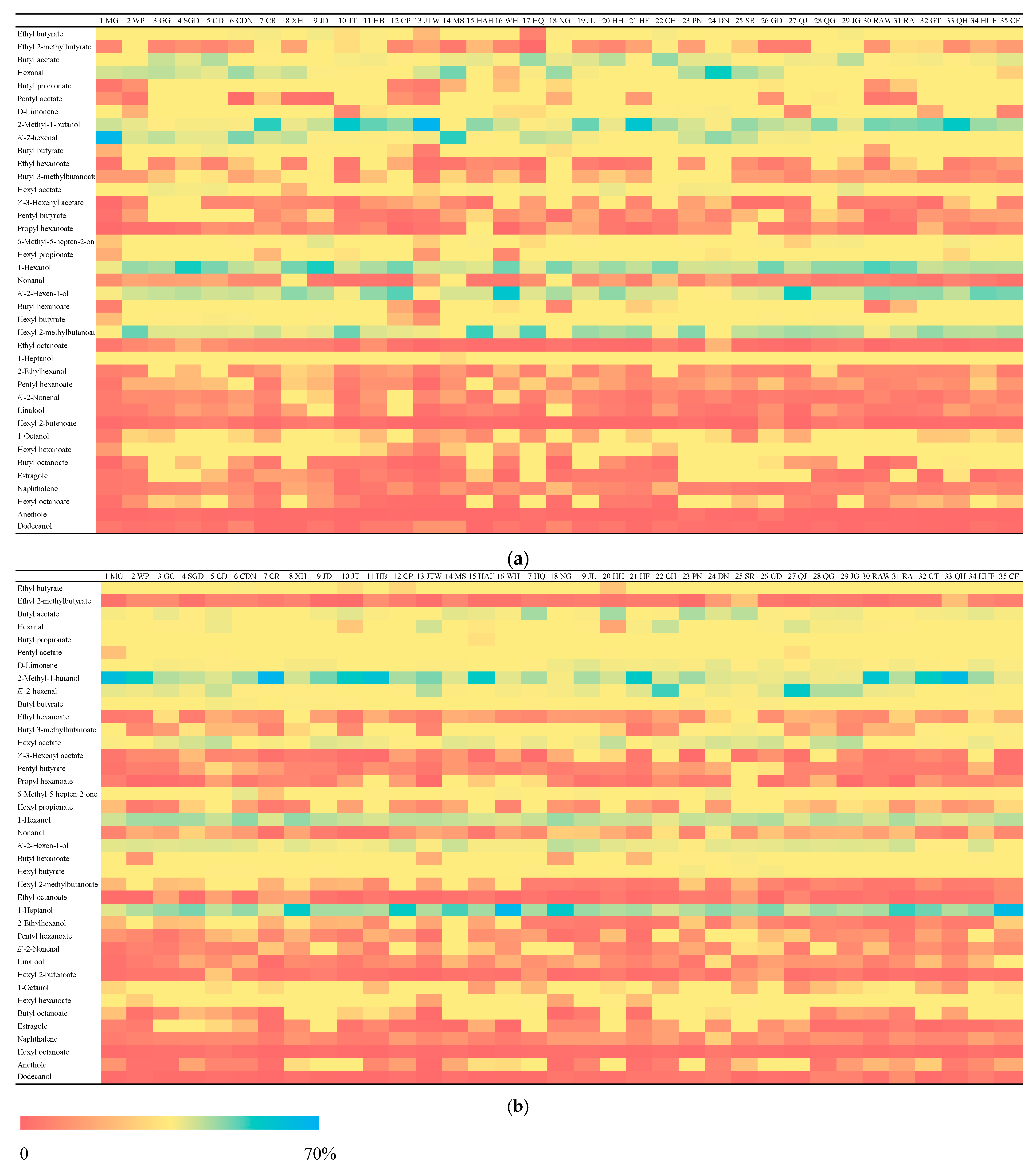
3.1. Volatile Compounds Analysis
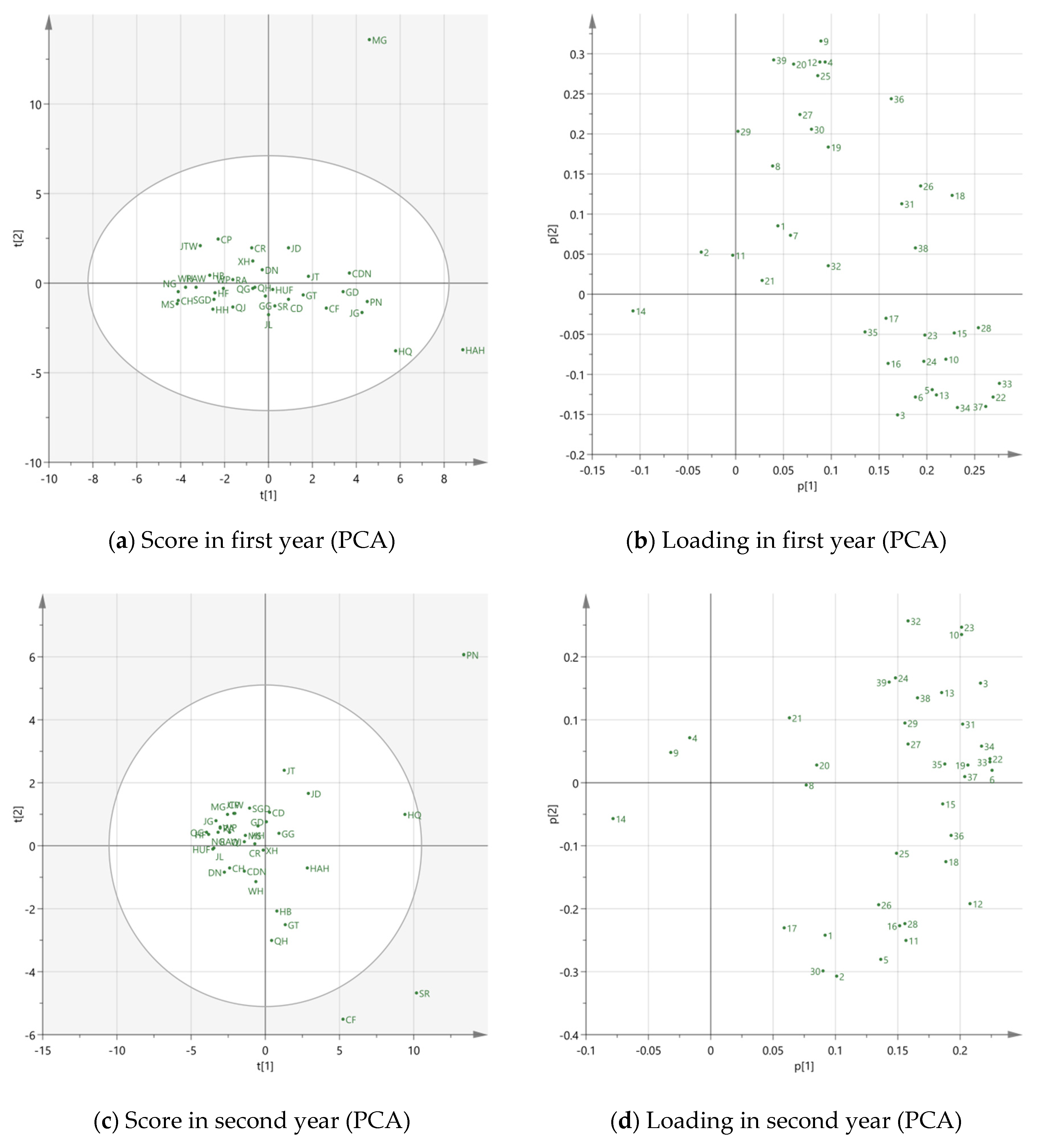
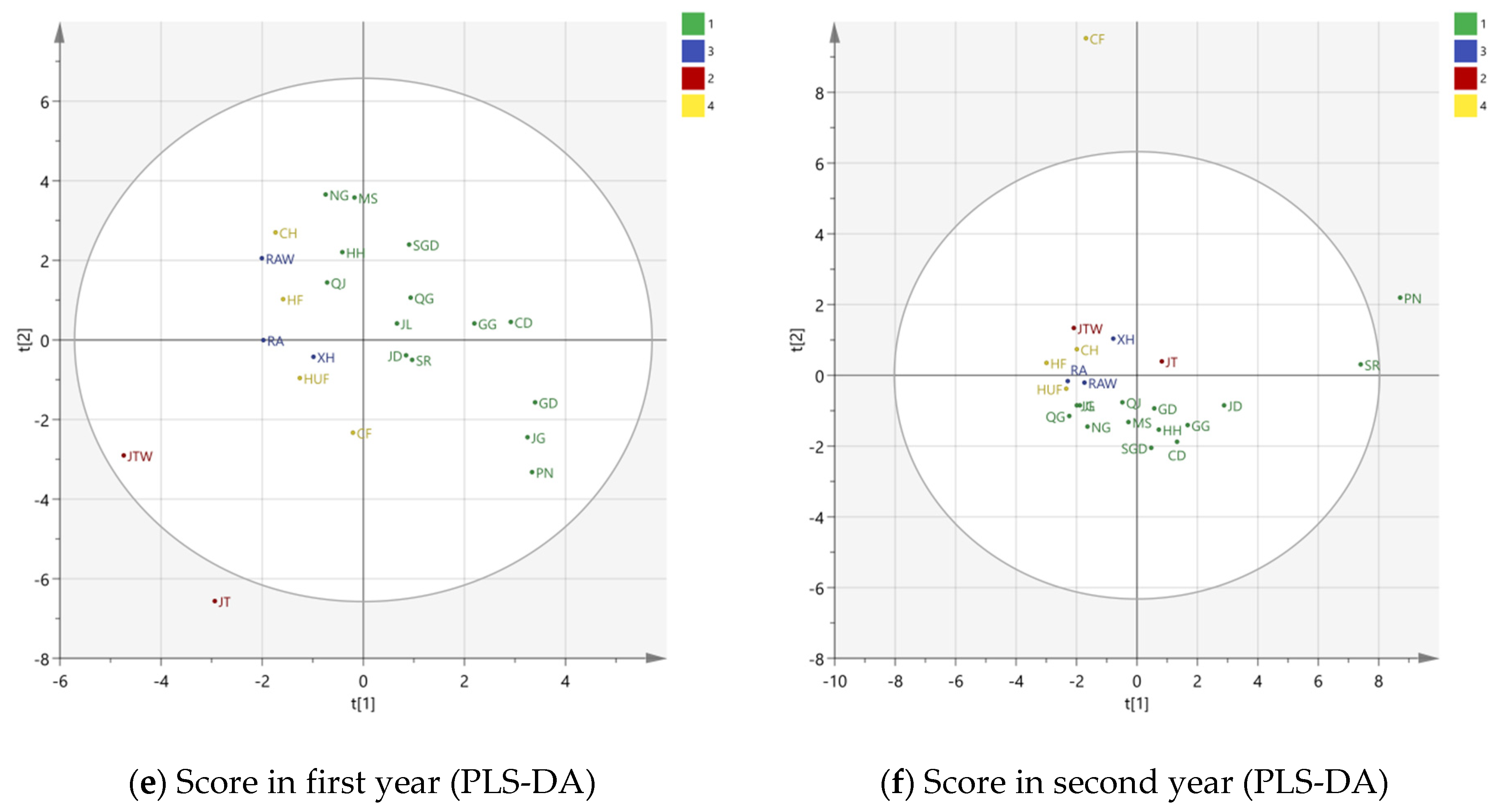
3.2. Multivariate Analysis
3.2.1. PCA
3.2.2. PLS-DA
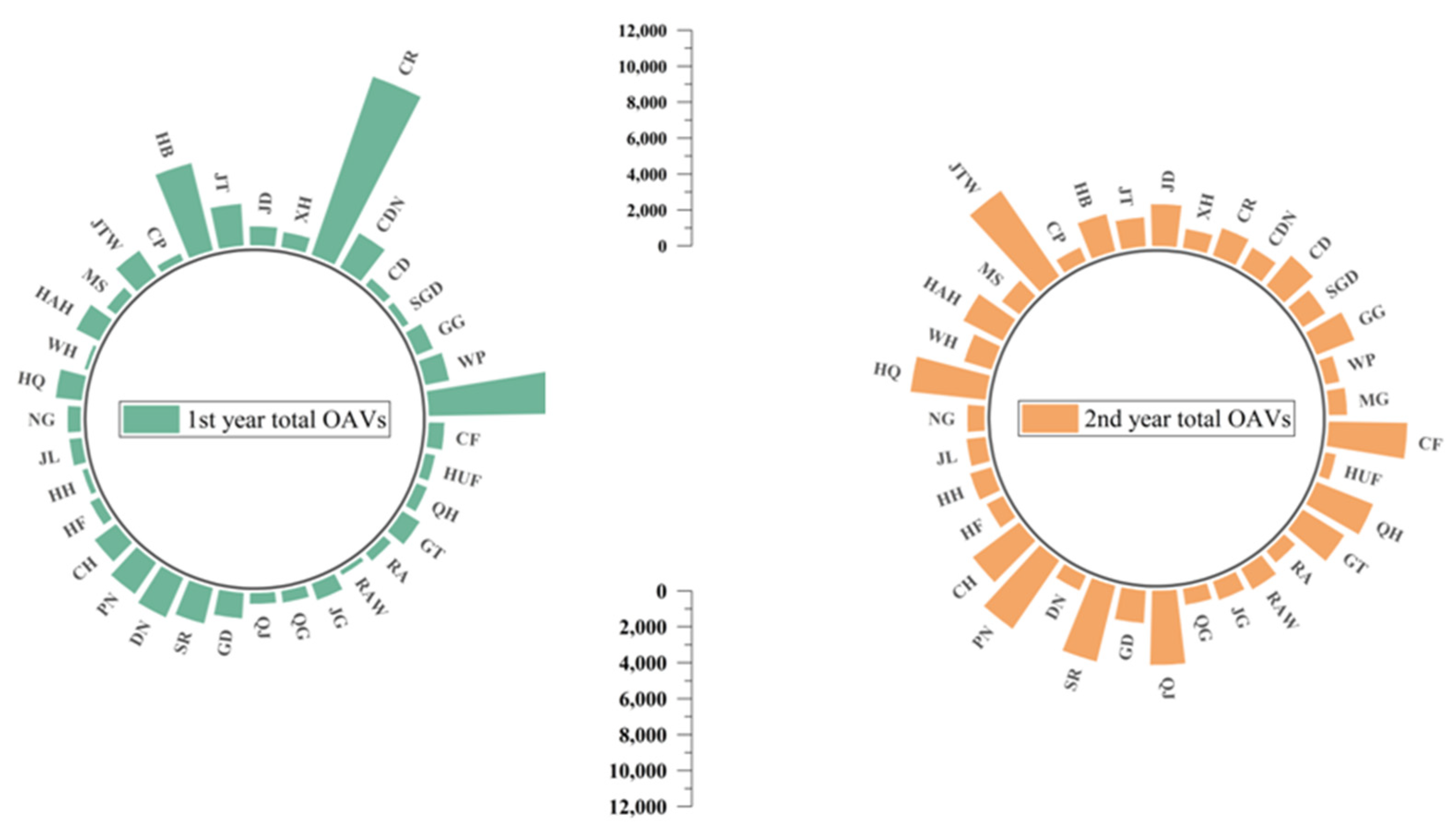
3.3. Odor-Active Compounds
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dimick, P.S.; Hoskin, J.C.; Acree, T.E. Review of apple flavor—State of the art. Crit. Rev. Food Sci. Nutr. 1983, 18, 387–409. [Google Scholar] [CrossRef] [PubMed]
- Dixon, J.; Hewett, E.W. Factors affecting apple aroma/flavour volatile concentration: A Review. N. Z. J. Crop Hortic. Sci. 2000, 28, 155–173. [Google Scholar] [CrossRef] [Green Version]
- Echeverria, G.; Fuentes, T.; Graell, J.; Lara, I.; López, M. Aroma volatile compounds of ‘Fuji’ apples in relation to harvest date and cold storage technology: A comparison of two seasons. Postharvest Biol. Technol. 2004, 32, 29–44. [Google Scholar] [CrossRef]
- Mehinagic, E.; Royer, G.; Symoneaux, R.; Jourjon, A.F.; Prost, C. Characterization of Odor-Active Volatiles in Apples: Influence of Cultivars and Maturity Stage. J. Agric. Food Chem. 2006, 54, 2678–2687. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, A.; Graell, J.; Lara, I. Volatile ester-synthesising capacity throughout on-tree maturation of ‘Golden Reinders’ apples. Sci. Hortic. 2011, 131, 6–14. [Google Scholar] [CrossRef]
- Fellman, J.; Mattison, D.; Fan, X.; Mattheis, J. Fuji’apple storage characteristics in relation to growing conditions and harvest maturity in Washington State. In Proceedings of the 7th International Controlled Atmosphere Research Conference, Davis, CA, USA, 13–18 July 1997; University of California: Davis, CA, USA, 1997; Volume 2, pp. 232–234. [Google Scholar]
- Ferreira, L.; Perestrelo, R.; Caldeira, M.; Câmara, J.S. Characterization of volatile substances in apples from rosaceae family by headspace solid-phase microextraction followed by GC-qMS. J. Sep. Sci. 2009, 32, 1875–1888. [Google Scholar] [CrossRef] [Green Version]
- Espino-Díaz, M.; Sepúlveda, D.R.; González-Aguilar, G.; Olivas, G.I. Biochemistry of Apple Aroma: A Review. Food Technol. Biotechnol. 2016, 54, 375–397. [Google Scholar] [CrossRef]
- Aprea, E.; Corollaro, M.L.; Betta, E.; Endrizzi, I.; Demattè, M.L.; Biasioli, F.; Gasperi, F. Sensory and instrumental profiling of 18 apple cultivars to investigate the relation between perceived quality and odour and flavour. Food Res. Int. 2012, 49, 677–686. [Google Scholar] [CrossRef]
- Xiaobo, Z.; Jiewen, Z. Comparative analyses of apple aroma by a tin-oxide gas sensor array device and GC/MS. Food Chem. 2008, 107, 120–128. [Google Scholar] [CrossRef]
- Vrhovsek, U.; Lotti, C.; Masuero, D.; Carlin, S.; Weingart, G.; Mattivi, F. Quantitative metabolic profiling of grape, apple and raspberry volatile compounds (VOCs) using a GC/MS/MS method. J. Chromatogr. B 2014, 966, 132–139. [Google Scholar] [CrossRef]
- Aprea, E.; Gika, H.; Carlin, S.; Theodoridis, G.; Vrhovsek, U.; Mattivi, F. Metabolite profiling on apple volatile content based on solid phase microextraction and gas-chromatography time of flight mass spectrometry. J. Chromatogr. A 2011, 1218, 4517–4524. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yang, C.; Li, S.; Yang, L.; Wang, Y.; Zhao, J.; Jiang, Q. Volatile characteristics of 50 peaches and nectarines evaluated by HP–SPME with GC–MS. Food Chem. 2009, 116, 356–364. [Google Scholar] [CrossRef]
- Zhu, J.; Wang, L.; Xiao, Z.; Niu, Y. Characterization of the key aroma compounds in mulberry fruits by application of gas chromatography–olfactometry (GC-O), odor activity value (OAV), gas chromatography-mass spectrometry (GC–MS) and flame photometric detection (FPD). Food Chem. 2018, 245, 775–785. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.-Y.; Zhang, Q.; Zhong, C.-H.; Guo, M.-Q. Volatile fingerprints and biomarkers of three representative kiwifruit cultivars obtained by headspace solid-phase microextraction gas chromatography mass spectrometry and chemometrics. Food Chem. 2019, 271, 211–215. [Google Scholar] [CrossRef]
- Zielinski, A.; Haminiuk, C.; Nunes, C.A.; Schnitzler, E.; Van Ruth, S.M.; Granato, D. Chemical Composition, Sensory Properties, Provenance, and Bioactivity of Fruit Juices as Assessed by Chemometrics: A Critical Review and Guideline. Compr. Rev. Food Sci. Food Saf. 2014, 13, 300–316. [Google Scholar] [CrossRef]
- Giannetti, V.; Mariani, M.B.; Mannino, P.; Marini, F. Volatile fraction analysis by HS-SPME/GC-MS and chemometric modeling for traceability of apples cultivated in the Northeast Italy. Food Control. 2017, 78, 215–221. [Google Scholar] [CrossRef]
- Cuevas, F.; Moreno-Rojas, J.; Arroyo, F.T.; Daza, A.; Ruiz-Moreno, M. Effect of management (organic vs conventional) on volatile profiles of six plum cultivars (Prunus salicina Lindl.). A chemometric approach for varietal classification and determination of potential markers. Food Chem. 2016, 199, 479–484. [Google Scholar] [CrossRef]
- Komthong, P.; Hayakawa, S.; Katoh, T.; Igura, N.; Shimoda, M. Determination of potent odorants in apple by headspace gas dilution analysis. LWT 2006, 39, 472–478. [Google Scholar] [CrossRef]
- El Hadi, M.A.M.; Zhang, F.-J.; Wu, F.-F.; Zhou, C.-H.; Tao, J. Advances in Fruit Aroma Volatile Research. Molecules 2013, 18, 8200–8229. [Google Scholar] [CrossRef]
- Noriega, F.; Mardones, C.; Fischer, S.; García-Viguera, C.; Moreno, D.A.; López, M.D. Seasonal changes in white strawberry: Effect on aroma, phenolic compounds and its biological activity. J. Berry Res. 2021, 11, 103–118. [Google Scholar] [CrossRef]
- Zarid, M.; Bueso, M.C.; Fernández-Trujillo, J.P. Seasonal effects on flesh volatile concentrations and texture at harvest in a near-isogenic line of melon with introgression in LG X. Sci. Hortic. 2020, 266, 109244. [Google Scholar] [CrossRef]
- Schwieterman, M.L.; Colquhoun, T.A.; Jaworski, E.; Bartoshuk, L.M.; Gilbert, J.L.; Tieman, D.M.; Odabasi, A.Z.; Moskowitz, H.R.; Folta, K.M.; Klee, H.J.; et al. Strawberry Flavor: Diverse Chemical Compositions, a Seasonal Influence, and Effects on Sensory Perception. PLoS ONE 2014, 9, e88446. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blanpied, G.D.; Silsby, K.J. Predicting Harvest Date Windows for Apples; Cornell University: Ithaca, NY, USA, 1992; pp. 6–7. [Google Scholar]
- Zacca, J.J.; Giudice, G.H.; Souza, M.P.; Caldas, L.N.; Vieira, M.L.; Machado, A.H. Development and validation of analytical method for identification of new psychoactive substances using linear retention indexes and gas chromatography-mass spectrometry. J. Chromatogr. A 2020, 1636, 461783. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Deng, J.; Bi, J.; Wu, X.; Zhang, B. Cultivar classification of cloudy apple juices from substandard fruits in China based on aroma profile analyzed by HS-SPME/GC-MS. LWT 2018, 102, 304–309. [Google Scholar] [CrossRef]
- Noiton, D.A.; Alspach, P.A. Founding Clones, Inbreeding, Coancestry, and Status Number of Modern Apple Cultivars. J. Am. Soc. Hortic. Sci. 1996, 121, 773–782. [Google Scholar] [CrossRef] [Green Version]
- Gao, Y.; Wang, K.; Wang, D.J.; Gong, X.; Liu, F.Z. Molecular id establishment of apple cultivars by tp-m13-ssr. Acta Hortic. Sin. 2016, 43, 25–37. [Google Scholar]
- Zhu, D.; Ren, X.; Wei, L.; Cao, X.; Ge, Y.; Liu, H.; Li, J. Collaborative analysis on difference of apple fruits flavour using electronic nose and electronic tongue. Sci. Hortic. 2020, 260, 108879. [Google Scholar] [CrossRef]
- Schumacher, K.; Asche, S.; Heil, M.; Mittelstädt, F.; Dietrich, H.; Mosandl, A. Methyl-Branched Flavor Compounds in Fresh and Processed Apples. J. Agric. Food Chem. 1998, 46, 4496–4500. [Google Scholar] [CrossRef]
- Nikfardjam, M.P.; Maier, D. Development of a headspace trap HRGC/MS method for the assessment of the relevance of certain aroma compounds on the sensorial characteristics of commercial apple juice. Food Chem. 2011, 126, 1926–1933. [Google Scholar] [CrossRef]
- Komthong, P.; Igura, N.; Shimoda, M. Effect of ascorbic acid on the odours of cloudy apple juice. Food Chem. 2007, 100, 1342–1349. [Google Scholar] [CrossRef]
- Rowan, D.D.; Lane, H.P.; Allen, J.M.; Fielder, S.; Hunt, M.B. Biosynthesis of 2-Methylbutyl, 2-Methyl-2-butenyl, and 2-Methylbutanoate Esters in Red Delicious and Granny Smith Apples Using Deuterium-Labeled Substrates. J. Agric. Food Chem. 1996, 44, 3276–3285. [Google Scholar] [CrossRef]
- Yahia, E.M.; Acree, T.E.; Liu, F.W. The evolution of some odour-active volatiles during the maturation and ripening of apples on the tree. Lebensm. Wiss. Technol. 1990, 23, 488–493. [Google Scholar]
- Contreras, C.; Tjellström, H.; Beaudry, R.M. Relationships between free and esterified fatty acids and LOX-derived volatiles during ripening in apple. Postharvest Biol. Technol. 2016, 112, 105–113. [Google Scholar] [CrossRef]
- Sousa, A.; Vareda, J.; Pereira, R.; Silva, C.; Câmara, J.S.; Perestrelo, R. Geographical differentiation of apple ciders based on volatile fingerprint. Food Res. Int. 2020, 137, 109550. [Google Scholar] [CrossRef] [PubMed]
- Pandit, S.S.; Chidley, H.G.; Kulkarni, R.S.; Pujari, K.H.; Giri, A.P.; Gupta, V.S. Cultivar relationships in mango based on fruit volatile profiles. Food Chem. 2009, 114, 363–372. [Google Scholar] [CrossRef]
- Qin, G.; Tao, S.; Cao, Y.; Wu, J.; Zhang, H.; Huang, W.; Zhang, S. Evaluation of the volatile profile of 33 Pyrus ussuriensis cultivars by HS-SPME with GC–MS. Food Chem. 2012, 134, 2367–2382. [Google Scholar] [CrossRef]
- Esparza, X.; Moyano, E.; Cosialls, J.; Galceran, M. Determination of naphthalene-derived compounds in apples by ultra-high performance liquid chromatography-tandem mass spectrometry. Anal. Chim. Acta 2013, 782, 28–36. [Google Scholar] [CrossRef]
- Irigoyen, A.; Ortigosa, M.; Juansaras, I.; Oneca, M.; Torre, P. Influence of an adjunct culture of Lactobacillus on the free amino acids and volatile compounds in a Roncal-type ewe’s-milk cheese. Food Chem. 2007, 100, 71–80. [Google Scholar] [CrossRef]
- Barron, L.J.R.; Redondo, Y.; Aramburu, M.; Gil, P.; Pérez-Elortondo, F.J.; Albisu, M.; Nájera, A.I.; de Renobales, M.; Fernández-García, E. Volatile composition and sensory properties of industrially produced Idiazabal cheese. Int. Dairy J. 2007, 17, 1401–1414. [Google Scholar] [CrossRef]
- Zepka, L.Q.; Garruti, D.S.; Sampaio, K.L.; Mercadante, A.Z.; Da Silva, M.A.A. Aroma compounds derived from the thermal degradation of carotenoids in a cashew apple juice model. Food Res. Int. 2014, 56, 108–114. [Google Scholar] [CrossRef] [Green Version]
- Van Gemert, L.J. Odour thresholds. In Compilations of Odour Threshold Values in Air, Water and Other Media, 2nd ed.; Oliemans Punter & Partner: Utrecht, The Netherlands, 2011. [Google Scholar]
- Lara, I.; Echeverría, G.; Graell, J.; López, M.L. Volatile Emission after Controlled Atmosphere Storage of Mondial Gala Apples (Malus domestica): Relationship to Some Involved Enzyme Activities. J. Agric. Food Chem. 2007, 55, 6087–6095. [Google Scholar] [CrossRef] [PubMed]
- Pino, J.A.; Febles, Y. Odour-active compounds in banana fruit cv. Giant Cavendish. Food Chem. 2013, 141, 795–801. [Google Scholar] [CrossRef] [PubMed]
- Czerny, M.; Christlbauer, M.; Christlbauer, M.; Fischer, A.; Granvogl, M.; Hammer, M.; Hartl, C.; Hernandez, N.M.; Schieberle, P. Re-investigation on odour thresholds of key food aroma compounds and development of an aroma language based on odour qualities of defined aqueous odorant solutions. Eur. Food Res. Technol. 2008, 228, 265–273. [Google Scholar] [CrossRef]
- Bowen, A.J.; Reynolds, A.G. Aroma compounds in Ontario Vidal and Riesling icewines. I. Effects of harvest date. Food Res. Int. 2015, 76, 540–549. [Google Scholar] [CrossRef]
- Zhu, J.; Xiao, Z. Characterization of the key aroma compounds in peach by gas chromatography–olfactometry, quantitative measurements and sensory analysis. Eur. Food Res. Technol. 2019, 245, 129–141. [Google Scholar] [CrossRef]
- Zhu, J.; Chen, F.; Wang, L.; Niu, Y.; Chen, H.; Wang, H.; Xiao, Z. Characterization of the Key Aroma Volatile Compounds in Cranberry (Vaccinium macrocarpon Ait.) Using Gas Chromatography–Olfactometry (GC-O) and Odor Activity Value (OAV). J. Agric. Food Chem. 2016, 64, 4990–4999. [Google Scholar] [CrossRef]
- Zheng, L.-Y.; Sun, G.-M.; Liu, Y.-G.; Lv, L.-L.; Yang, W.-X.; Zhao, W.-F.; Wei, C.-B. Aroma Volatile Compounds from Two Fresh Pineapple Varieties in China. Int. J. Mol. Sci. 2012, 13, 7383–7392. [Google Scholar] [CrossRef] [Green Version]
- Du, X.; Plotto, A.; Baldwin, E.; Rouseff, R. Evaluation of Volatiles from Two Subtropical Strawberry Cultivars Using GC–Olfactometry, GC-MS Odor Activity Values, and Sensory Analysis. J. Agric. Food Chem. 2011, 59, 12569–12577. [Google Scholar] [CrossRef]
| No. | Name | Code | Reported Parentages or Origin | Harvest Date | No. | Name | Code | Reported Parentages or Origin | Harvest Date |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Miguo | MG | unknown | 9.28 | 19 | Judeline | JL | Golden Delicious | 9.28 |
| 2 | White Pippin | WP | unknown | 9.28 | 20 | Huahong | HH | Golden Delicious | 9.28 |
| 3 | Grimes Golsen | GG | Golden Delicious | 9.28 | 21 | Hanfu | HF | Fuji | 9.28 |
| 4 | Stark Spur Golden Delicous | SGD | Golden Delicious | 9.28 | 22 | Changhong | CH | Fuji | 9.28 |
| 5 | Cloden | CD | Golden Delicious | 9.28 | 23 | Pinova | PN | Golden Delicious | 9.28 |
| 6 | Cardinal | CDN | unknown | 9.28 | 24 | Dounan | DN | unknown | 9.28 |
| 7 | Calville Rouge | CR | unknown | 9.28 | 25 | Starkrimson | SR | Golden Delicious | 9.28 |
| 8 | Xinhua | XH | Ralls | 9.28 | 26 | Golden Delicious | GD | Golden Delicious | 9.28 |
| 9 | Jonagold | JD | Golden Delicious/ Jonathan | 9.28 | 27 | Qiujin | QJ | Ralls/Golden Delicious | 9.28 |
| 10 | Jonathan | JT | Jonathan | 9.28 | 28 | Qinguan | QG | Golden Delicious | 10.9 |
| 11 | Hadi Bolaite | HB | unknown | 9.28 | 29 | Jiguan | JG | Golden Delicious | 10.9 |
| 12 | Clapp | CP | unknown | 9.28 | 30 | Ralls (weeping) | RAW | Ralls | 10.9 |
| 13 | Jonathan (New) | JTW | Jonathan | 9.28 | 31 | Ralls | RA | Ralls | 10.9 |
| 14 | Mutsu Spur | MS | Golden Delicious | 9.28 | 32 | Gongteng | GT | unknown | 10.9 |
| 15 | Hahong | HAH | unknown | 9.28 | 33 | Qihu 7 | QH | unknown | 10.9 |
| 16 | Wenhong | WH | unknown | 9.28 | 34 | Huafu | HUF | Fuji | 10.9 |
| 17 | Hongqiaowang | HQ | unknown | 9.28 | 35 | Changfu 2 | CF | Fuji | 10.9 |
| 18 | Ningguan | NG | Golden Delicious | 9.28 |
| No. | Name | Retention Time (min) | Retention Index | CAS | Quantitative m/z | Qualitative m/z | R2 | Odor Descriptions | Thresholds (μg/L, in Water) | First Year | Second Year | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| OAV | VIP | OAV | VIP | ||||||||||
| 1 | Ethyl butyrate | 5.889 | 1057 | 105-54-4 | 60 | 88/89 | 0.9993 | oxidized apple, sweet | 9 a | 0.70 | >1 | 0.90 | |
| 2 | Ethyl 2-methylbutyrate | 6.283 | 1073 | 7452-79-1 | 74 | 57/102 | 0.9993 | fruity | 0.13 b | >1 | 1.17 | >1 | 1.01 |
| 3 | Butyl acetate | 6.756 | 1094 | 123-86-4 | 61 | 43/56 | 0.9993 | fruity, apple | 66 b | 0.86 | >1 | 0.72 | |
| 4 | Hexanal | 6.922 | 1101 | 66-25-1 | 82 | 44/45 | 0.9916 | grass | 5 a | >1 | 0.98 | >1 | 1.42 |
| 5 | Butyl propionate | 8.603 | 1163 | 590-01-2 | 57 | 56/75 | 0.9982 | 25 c | 0.77 | >1 | 1.11 | ||
| 6 | Pentyl acetate | 9.484 | 1196 | 628-63-7 | 43 | 70/61 | 0.9968 | cooked apple, banana | 43 a | 0.92 | >1 | 0.72 | |
| 7 | D-Limonene | 10.095 | 1218 | 5989-27-5 | 68 | 67/93 | 0.9901 | citrus, mint | 34 c | 0.72 | >1 | 0.90 | |
| 8 | 2-Methyl-1-butanol | 10.492 | 1233 | 137-32-6 | 56 | 57/70 | 0.9922 | wine | 500 b | 1.73 | >1 | 1.83 | |
| 9 | E-2-hexenal | 10.628 | 1238 | 6728-26-3 | 98 | 80/83 | 0.9938 | green, apple like | 110 b | 0.93 | >1 | 0.96 | |
| 10 | Butyl butyrate | 10.76 | 1243 | 109-21-7 | 71 | 43/89 | 0.9962 | fruity, berry | 100 d | 0.95 | 0.76 | ||
| 11 | Ethyl hexanoate | 11.195 | 1259 | 123-66-0 | 88 | 70/99 | 0.9936 | fruit | 22 a | 0.61 | 1.07 | ||
| 12 | Butyl 3-methylbutanoate | 11.627 | 1274 | 109-19-3 | 85 | 56/103 | 0.9933 | ethereal-fruity | 17 d | 0.77 | 0.87 | ||
| 13 | Hexyl acetate | 12.278 | 1298 | 142-92-7 | 61 | 73/84 | 0.9962 | sweet, fruity, floral | 115 a | 0.97 | >1 | 1.21 | |
| 14 | Z-3-hexenyl acetate | 13.253 | 1334 | 3681-71-8 | 82 | 67/43 | 0.9990 | green | 13 a | 1.43 | 1.26 | ||
| 15 | Pentyl butyrate | 13.507 | 1344 | 540-18-1 | 71 | 70/89 | 0.9983 | banana | 210 a | 1.13 | 0.78 | ||
| 16 | Propyl hexanoate | 13.650 | 1349 | 626-77-7 | 99 | 61/117 | 0.9967 | fruit | / | / | 0.47 | / | 1.19 |
| 17 | 6-Methyl-5-hepten-2-one | 14.014 | 1363 | 110-93-0 | 108 | 93/111 | 0.9951 | mushroom, pepper | 68 a | 0.89 | >1 | 1.25 | |
| 18 | Hexyl propionate | 14.091 | 1366 | 2445-76-3 | 75 | 57/84 | 0.9974 | sweet | 8 a | 1.12 | >1 | 0.95 | |
| 19 | 1-Hexanol | 14.570 | 1384 | 111-27-3 | 56 | 43/55 | 0.9946 | floral, green | 500 b | >1 | 1.25 | >1 | 0.85 |
| 20 | Nonanal | 15.529 | 1421 | 124-19-6 | 98 | 81/82 | 0.9963 | citus-like, floral | 2.53 b | 1.12 | >1 | 0.89 | |
| 21 | E-2-hexen-1-ol | 15.967 | 1438 | 928-95-0 | 82 | 41/67 | 0.9996 | green, walnut | 2319 a | 1.12 | >1 | 1.63 | |
| 22 | Butyl hexanoate | 16.099 | 1443 | 626-82-4 | 99 | 87/117 | 0.9929 | herbaceous | 700 b | 0.83 | 0.70 | ||
| 23 | Hexyl butyrate | 16.167 | 1446 | 2639-63-6 | 89 | 69/84 | 0.9937 | fruity | 250 c | 0.97 | 0.79 | ||
| 24 | Hexyl 2-methylbutanoate | 16.467 | 1457 | 10032-15-2 | 103 | 74/87 | 0.9913 | fruity, pungent | 22 d | >1 | 1.15 | 0.87 | |
| 25 | Ethyl octanoate | 16.667 | 1465 | 106-32-1 | 88 | 57/101 | 0.9932 | fruit, fat | 19.3 a | 0.83 | 0.76 | ||
| 26 | 1-Heptanol | 17.273 | 1489 | 111-70-6 | 70 | 55/56 | 0.9933 | chemical, green | 425 a | 1.13 | >1 | 0.96 | |
| 27 | 2-Ethylhexanol | 18.146 | 1525 | 104-76-7 | 57 | 41/43 | 0.9939 | rose, green | 2548 a | 0.53 | 0.78 | ||
| 28 | Pentyl hexanoate | 18.636 | 1546 | 540-07-8 | 70 | 43/117 | 0.9979 | / | / | 0.94 | / | 1.18 | |
| 29 | E-2-nonenal | 19.161 | 1568 | 18829-56-6 | 55 | 43/70 | 0.9911 | fatty, green | 0.69 e | >1 | 1.13 | >1 | 0.95 |
| 30 | Linalool | 19.529 | 1584 | 78-70-6 | 93 | 71/121 | 0.9991 | flower, lavender | 0.17 e | >1 | 1.01 | >1 | 1.18 |
| 31 | Hexyl 2-butenoate | 19.689 | 1590 | 19089-92-0 | 87 | 41/69 | 0.9970 | / | / | 0.93 | / | 0.65 | |
| 32 | 1-Octanol | 19.794 | 1595 | 111-87-5 | 56 | 55/70 | 0.9971 | green | 110 f | 0.98 | 1.01 | ||
| 33 | Hexyl hexanoate | 20.819 | 1650 | 6378-65-0 | 117 | 84/99 | 0.9973 | green | 6400 b | 0.72 | 0.65 | ||
| 34 | Butyl octanoate | 20.881 | 1653 | 589-75-3 | 145 | 127/101 | 0.9984 | fruit | / | / | 0.90 | / | 0.64 |
| 35 | Estragole | 21.837 | 1708 | 140-67-0 | 148 | 117/121 | 0.9997 | licorice, anise | 16 e | 0.78 | 0.75 | ||
| 36 | Naphthalene | 22.943 | 1783 | 91-20-3 | 128 | 64/102 | 0.9973 | Tar, floral, fruity | 6 a | 1.38 | >1 | 0.79 | |
| 37 | Hexyl octanoate | 23.963 | 1861 | 1117-55-1 | 84 | 56/145 | 0.9969 | herb, green, oil | / | / | 0.79 | / | 0.79 |
| 38 | Anethole | 24.180 | 1878 | 104-46-1 | 148 | 117/147 | 0.9984 | 50 a | 0.73 | 0.69 | |||
| 39 | Dodecanol | 25.883 | 2027 | 112-53-8 | 55 | 69/70 | 0.9968 | fat, wax | 16 a | 1.40 | 1.20 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, X.; Bi, J.; Fauconnier, M.-L. Characteristic Volatiles and Cultivar Classification in 35 Apple Varieties: A Case Study of Two Harvest Years. Foods 2022, 11, 690. https://doi.org/10.3390/foods11050690
Wu X, Bi J, Fauconnier M-L. Characteristic Volatiles and Cultivar Classification in 35 Apple Varieties: A Case Study of Two Harvest Years. Foods. 2022; 11(5):690. https://doi.org/10.3390/foods11050690
Chicago/Turabian StyleWu, Xinye, Jinfeng Bi, and Marie-Laure Fauconnier. 2022. "Characteristic Volatiles and Cultivar Classification in 35 Apple Varieties: A Case Study of Two Harvest Years" Foods 11, no. 5: 690. https://doi.org/10.3390/foods11050690
APA StyleWu, X., Bi, J., & Fauconnier, M.-L. (2022). Characteristic Volatiles and Cultivar Classification in 35 Apple Varieties: A Case Study of Two Harvest Years. Foods, 11(5), 690. https://doi.org/10.3390/foods11050690







