Mineral Composition and Bioaccessibility in Rocket and Purslane after Zn Biofortification Process
Abstract
:1. Introduction
2. Materials and Methods
2.1. Production of Zn-Biofortified Purslane and Rocket
2.2. Mineral Profile of Rocket and Purslane
2.3. In Vitro Gastrointestinal Digestion Process
2.4. Analysis of Mineral Content in Digested Sample
2.5. Percentage of Recommended Daily Allowance and Hazard Quotient for Zn Intake
2.6. Statistical Analysis
3. Results and Discussion
3.1. Mineral Analysis
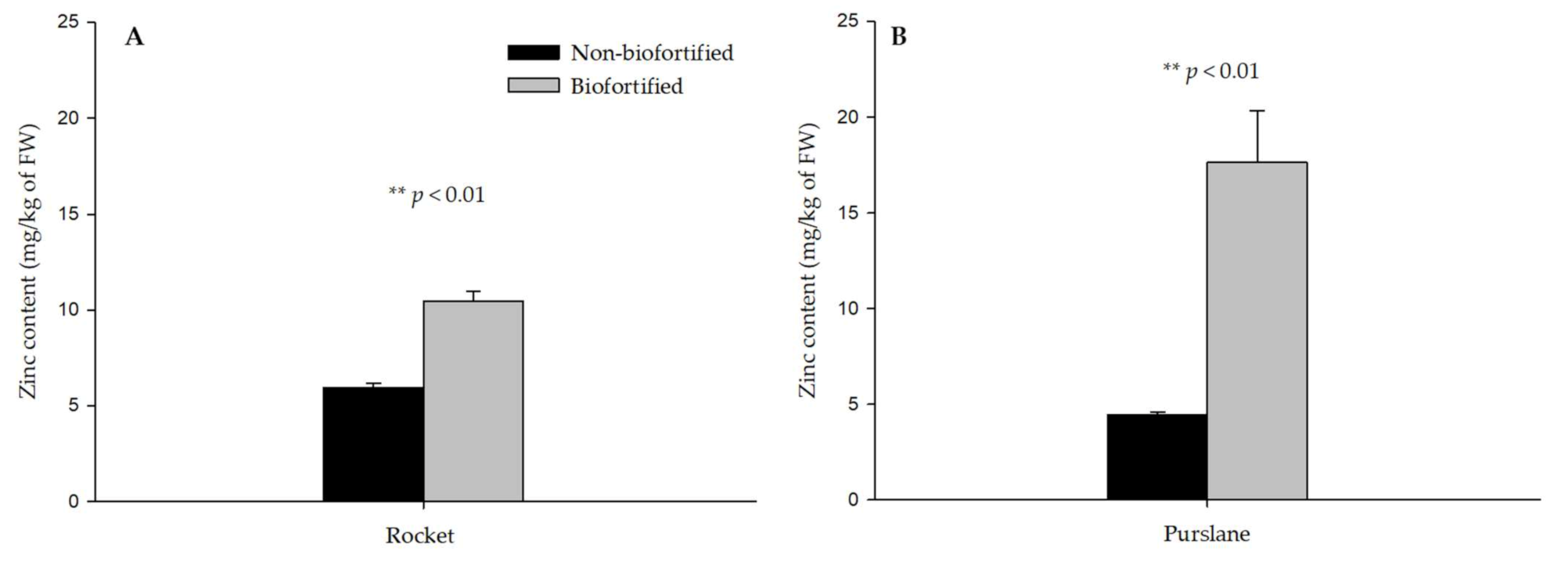
3.2. Mineral Profile of Biofortified and Non-Biofortified Rocket and Purslane
3.3. Mineral Bioaccessibility in Rocket and Purslane after the Biofortification Process
3.4. Daily Intake, Coverage of RDA-Zn (Male and Female), and Hazard Quotient
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Andreini, C.; Banci, L.; Bertini, I.; Rosato, A. Counting the zinc-proteins encoded in the human genome. J. Proteome Res. 2006, 5, 196–201. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Reddy, P.K.; Kumar, B. Trace Element Zinc, a Nature’s Gift to Fight Unprecedented Global Pandemic COVID-19. Biol. Trace Elem. Res. 2020, 199, 3213–3221. [Google Scholar] [CrossRef]
- Scherz, H.; Kirchhoff, E. Trace elements in foods: Zinc contents of raw foods—a comparison of data originating from different geographical regions of the world. J. Food Comp. Anal. 2006, 19, 420–433. [Google Scholar] [CrossRef]
- Nemzer, B.; Al-Taher, F.; Abshiru, N. Phytochemical composition and nutritional value of different plant parts in two cultivated and wild purslane (Portulaca oleracea L.) genotypes. Food Chem. 2020, 320, 126621. [Google Scholar] [CrossRef]
- De Almeida, H.J.; Vergara Carmona, V.M.; Ferreira Inocêncio, M.; Furtini Neto, A.E.; Cecílio Filho, A.B.; Mauad, M. Soil Type and Zinc Doses in Agronomic Biofortification of Lettuce Genotypes. Agronomy 2020, 10, 124. [Google Scholar] [CrossRef] [Green Version]
- Khoja, K.; Buckley, A.F.; Aslam, M.; Sharp, A.P.; Latunde-Dada, G.O. In vitro bioaccessibility and bioavailability of iron from mature and microgreen fenugreek, rocket and broccoli. Nutrients 2020, 12, 1057. [Google Scholar] [CrossRef] [Green Version]
- National Institute of Health (NIH). Available online: https://ods.od.nih.gov/factsheets/Zinc-HealthProfessional/#h2 (accessed on 10 November 2021).
- Wessells, K.R.; Brown, K.H. Estimating the global prevalence of zinc deficiency: Results based on zinc availability in national food supplies and the prevalence of stunting. PLoS ONE 2012, 7, e50568. [Google Scholar]
- Kumssa, D.B.; Joy, E.J.; Ander, E.L.; Watts, M.J.; Young, S.D.; Walker, S.; Broadley, M.R. Dietary calcium and zinc deficiency risks are decreasing but remain prevalent. Sci. Rep. 2015, 5, 10974. [Google Scholar] [CrossRef] [Green Version]
- Shah, D.; Sachdev, H.S.; Gera, T.; De-Regil, L.M.; Peña-Rosas, J.P. Fortification of staple foods with zinc for improving zinc status and other health outcomes in the general population. Cochrane Database Syst. Rev. 2016, 6, CD010697. [Google Scholar] [CrossRef]
- International Zinc Nutrition Consultative Group. BIZIFED2. Available online: https://www.izincg.org/ (accessed on 2 November 2021).
- VIRCA Plus. Available online: https://cassavaplus.org/ (accessed on 29 October 2021).
- Harvest Plus. Available online: https://www.harvestplus.org/ (accessed on 29 October 2021).
- Carvalho, S.M.; Vasconcelos, M.W. Producing more with less: Strategies and novel technologies for plant-based food biofortification. Food Res. Int 2013, 54, 961–971. [Google Scholar] [CrossRef]
- Van Der Straeten, D.; Bhullar, N.K.; De Steur, H.; Gruissem, W.; MacKenzie, D.; Pfeiffer, W.; Bouis, H. Multiplying the efficiency and impact of biofortification through metabolic engineering. Nat. Commun. 2020, 11, 5203. [Google Scholar] [CrossRef]
- Buturi, C.V.; Mauro, R.P.; Fogliano, V.; Leonardi, C.; Giuffrida, F. Mineral Biofortification of Vegetables as a Tool to Improve Human Diet. Foods 2021, 10, 223. [Google Scholar] [CrossRef]
- Montesano, F.F.; D’Imperio, M.; Parente, A.; Cardinali, A.; Renna, M.; Serio, F. Green bean biofortification for Si through soilless cultivation: Plant response and Si bioaccessibility in pods. Sci. Rep. 2016, 6, 31662. [Google Scholar] [CrossRef] [Green Version]
- Rouphael, Y.; Kyriacou, M.C. Enhancing quality of fresh vegetables through salinity eustress and biofortification applications facilitated by soilless cultivation. Front. Plant Sci. 2018, 9, 1254. [Google Scholar] [CrossRef] [PubMed]
- Gonnella, M.; Renna, M.; D’Imperio, M.; Santamaria, P.; Serio, F. Iodine biofortification of four brassica genotypes is effective already at low rates of potassium iodate. Nutrients 2019, 11, 451. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fanasca, S.; Colla, G.; Maiani, G.; Venneria, E.; Rouphael, Y.; Azzini, E.; Saccardo, F. Changes in antioxidant content of tomato fruits in response to cultivar and nutrient solution composition. J. Agric. Food Chem. 2006, 54, 4319–4325. [Google Scholar] [CrossRef] [PubMed]
- White, P.J.; Broadley, M.R. Biofortification of crops with seven mineral elements often lacking in human diets–iron, zinc, copper, calcium, magnesium, selenium and iodine. New Phytol. 2009, 182, 49–84. [Google Scholar] [CrossRef]
- Egea-Gilabert, C.; Ruiz-Hernández, M.V.; Parra, M.Á.; Fernández, J.A. Characterization of purslane (Portulaca oleracea L.) accessions: Suitability as ready-to-eat product. Sci. Hortic. 2014, 172, 73–81. [Google Scholar] [CrossRef]
- Santamaria, P.; Elia, A.; Serio, F.; Todaro, E. A survey of nitrate and oxalate content in fresh vegetables. J. Sci. Food Agric. 1999, 79, 1882–1888. [Google Scholar] [CrossRef]
- Cuervo, A.; Valdés, L.; Salazar, N.; de los Reyes-Gavilán, C.G.; Ruas-Madiedo, P.; Gueimonde, M.; González, S. Pilot study of diet and microbiota: Interactive associations of fibers and polyphenols with human intestinal bacteria. J. Agric. Food Chem. 2014, 62, 5330–5336. [Google Scholar] [CrossRef]
- Aires, A.; Marques, E.; Carvalho, R.; Rosa, E.A.; Saavedra, M.J. Evaluation of biological value and appraisal of polyphenols and glucosinolates from organic baby-leaf salads as antioxidants and antimicrobials against important human pathogenic bacteria. Molecules 2013, 18, 4651–4668. [Google Scholar] [CrossRef]
- Fernández-Navarro, T.; Salazar, N.; Gutiérrez-Díaz, I.; Sánchez, B.; Rúas-Madiedo, P.; Clara, G.; González, S. Bioactive compounds from regular diet and faecal microbial metabolites. Eur. J. Nutr. 2018, 57, 487–497. [Google Scholar] [CrossRef] [PubMed]
- Sandstroèm, B. Micronutrient interactions: Effects on absorption and bioavailability. Br. J. Nutr. 2001, 85, S181–S185. [Google Scholar] [CrossRef]
- Kyomugasho, C.; Gwala, S.; Christiaens, S.; Kermani, Z.J.; Van Loey, A.M.; Grauwet, T.; Hendrickx, M.E. Pectin nanostructure influences pectin-cation interactions and in vitro-bioaccessibility of Ca2+, Zn2+, Fe2+ and Mg2+-ions in model systems. Food Hydrocoll. 2017, 62, 299–310. [Google Scholar] [CrossRef]
- D'Antuono, I.; Garbetta, A.; Linsalata, V.; Minervini, F.; Cardinali, A. Polyphenols from artichoke heads (Cynara cardunculus (L.) subsp. scolymus Hayek): In vitro bio-accessibility, intestinal uptake and bioavailability. Food Funct. 2015, 6, 1268–1277. [Google Scholar] [CrossRef]
- Hemalatha, S.; Platel, K.; Srinivasan, K. Zinc and iron contents and their bioaccessibility in cereals and pulses consumed in India. Food Chem. 2007, 102, 1328–1336. [Google Scholar] [CrossRef]
- Camara, F.; Amaro, M.A.; Barbera, R.; Clemente, G. Bioaccessibility of minerals in school meals: Comparison between dialysis and solubility methods. Food Chem. 2005, 92, 481–489. [Google Scholar] [CrossRef]
- D’Imperio, M.; Renna, M.; Cardinali, A.; Buttaro, D.; Serio, F.; Santamaria, P. Calcium biofortification and bioaccessibility in soilless “baby leaf” vegetable production. Food Chem. 2016, 213, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Di Gioia, F.; Renna, M.; Santamaria, P. Sprouts, Microgreens and “Baby Leaf” Vegetables. In Minimally Processed Refrigerated Fruits and Vegetables; Springer: Boston, MA, USA, 2017; pp. 403–432. [Google Scholar]
- D’Imperio, M.; Montesano, F.F.; Montemurro, N.; Parente, A. Posidonia natural residues as growing substrate component: An ecofriendly method to improve nutritional profile of brassica microgreens. Front. Plant Sci. 2021, 12, 580596. [Google Scholar] [CrossRef]
- Ferruzzi, M.G.; Failla, M.L.; Schwartz, S.J. Assessment of degradation and intestinal cell uptake of carotenoids and chlorophyll derivatives from spinach puree using an in vitro digestion and Caco-2 human cell model. J. Agric. Food Chem. 2001, 49, 2082–2089. [Google Scholar] [CrossRef]
- Hamilton, E.M.; Barlow, T.S.; Gowing, C.J.B.; Watts, M.J. Bioaccessibility performance data for fifty-seven elements in guidance materials BGS 102. Microchem. J. 2015, 123, 131–138. [Google Scholar] [CrossRef] [Green Version]
- Integrated Risk Information System (Iris). Integrated Risk Information System—Database 2011; United States Environmental Protection Agency: Washington, DC, USA, 2011. [Google Scholar]
- Bączek-Kwinta, R.; Baran, A.; Simlat, M.; Lang, J.; Bieniek, M.; Florek, B. Enrichment of Different Plant Seeds with Zinc and Assessment of Health Risk of Zn-Fortified Sprouts Consumption. Agronomy 2020, 10, 937. [Google Scholar] [CrossRef]
- Noulas, C.; Tziouvalekas, M.; Karyotis, T. Zinc in soils, water and food crops. J. Trace Elem. Med. Biol. 2018, 49, 252–260. [Google Scholar] [CrossRef]
- Barrameda-Medina, Y.; Blasco, B.; Lentini, M.; Esposito, S.; Baenas, N.; Moreno, D.A.; Ruiz, J.M. Zinc biofortification improves phytochemicals and amino-acidic profile in Brassica oleracea cv. Bronco. Plant Sci. 2017, 258, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Barrameda-Medina, Y.; Lentini, M.; Esposito, S.; Ruiz, J.M.; Blasco, B. Zn-biofortification enhanced nitrogen metabolism and photorespiration process in green leafy vegetable Lactuca sativa L. J. Sci. Food Agric. 2017, 97, 1828–1836. [Google Scholar] [CrossRef] [Green Version]
- Zou, T.; Xu, N.; Hu, G.; Pang, J.; Xu, H. Biofortification of soybean sprouts with zinc and bioaccessibility of zinc in the sprouts. J. Sci. Food Agric. 2014, 94, 3053–3060. [Google Scholar] [CrossRef] [PubMed]
- Di Gioia, F.; Petropoulos, S.A.; Ozores-Hampton, M.; Morgan, K.; Rosskopf, E.N. Zinc and Iron Agronomic Biofortification of Brassicaceae Microgreens. Agronomy 2019, 9, 677. [Google Scholar] [CrossRef] [Green Version]
- Rietra, R.P.J.J.; Heinen, M.; Dimkpa, C.O.; Bindraban, P.S. Effects of nutrient antagonism and synergism on yield and fertilizer use efficiency. Commun. Soil Sci. Plant Anal. 2017, 48, 1895–1920. [Google Scholar] [CrossRef] [Green Version]
- D’Imperio, M.; Montesano, F.F.; Renna, M.; Parente, A.; Logrieco, A.F.; Serio, F. Hydroponic Production of Reduced-Potassium Swiss Chard and Spinach: A Feasible Agronomic Approach to Tailoring Vegetables for Chronic Kidney Disease Patients. Agronomy 2019, 9, 627. [Google Scholar] [CrossRef] [Green Version]
- Rousseau, S.; Kyomugasho, C.; Celus, M.; Hendrickx, M.E.; Grauwet, T. Barriers impairing mineral bioaccessibility and bioavailability in plant-based foods and the perspectives for food processing. Crit. Rev. Food Sci. Nutr. 2020, 60, 826–843. [Google Scholar] [CrossRef]
- D'Imperio, M.; Renna, M.; Cardinali, A.; Buttaro, D.; Santamaria, P.; Serio, F. Silicon biofortification of leafy vegetables and its bioaccessibility in the edible parts. J. Sci. Food Agric. 2016, 96, 751–756. [Google Scholar] [CrossRef] [PubMed]
- Fernández-García, E.; Carvajal-Lérida, I.; Pérez-Gálvez, A. In vitro bioaccessibility assessment as a prediction tool of nutritional efficiency. Nutr. Res. 2009, 29, 751–760. [Google Scholar] [CrossRef] [PubMed]
- Bertin, R.L.; Maltez, H.F.; de Gois, J.S.; Borges, D.L.; Borges, G.D.S.C.; Gonzaga, L.V.; Fett, R. Mineral composition and bioaccessibility in Sarcocornia ambigua using ICP-MS. J. Food Compost. Anal. 2016, 47, 45–51. [Google Scholar] [CrossRef]
- Khouzam, R.B.; Pohl, P.; Lobinski, R. Bioaccessibility of essential elements from white cheese, bread, fruit and vegetables. Talanta 2011, 86, 425–428. [Google Scholar] [CrossRef] [PubMed]
- Holmes, R.P.; Goodman, H.O.; Assimos, D.G. Dietary oxalate and its intestinal absorption. Scanning Microsc. 1995, 9, 16. [Google Scholar]
- Cabrera, W.E.; Schrooten, I.; De Broe, M.E.; d’Haese, P.C. Strontium and bone. J. Bone Miner. Res. 1999, 14, 661–668. [Google Scholar] [CrossRef] [PubMed]

| Element | LOD | LOQ | Found | Certified | Recovery |
|---|---|---|---|---|---|
| µg/L | mg/kg DW | (%) | |||
| Al | 0.8904 | 2.6982 | 624 ± 33.65 | 598 ± 7.1 | 104 |
| B | 0.0451 | 0.1365 | 29.7 ± 0.29 | 33.0 ± 0.42 | 90 |
| Ca | 0.0698 | 0.2116 | 49,437 ± 113.4 | 50,450 ± 550 | 98 |
| Fe | 0.2923 | 0.8853 | 358.3 ± 0.92 | 367 ± 4.3 | 98 |
| K | 0.7344 | 2.2255 | 30,443 ± 99 | 26,760 ± 480 | 113 |
| Mg | 0.1458 | 0.4420 | 11,649 ± 35.03 | 12,000 | 97 |
| Mn | 0.1898 | 0.5752 | 264.1 ± 1.24 | 246 ± 7.1 | 107 |
| Sr | 0.2068 | 0.6267 | 88.0 ± 0.401 | 85.0 | 104 |
| Zn | 0.1763 | 0.5343 | 30.7 ± 0.205 | 30.9 ± 0.55 | 99 |
| Element | BF | Residue | MB | Certified | BF% | Recovery |
|---|---|---|---|---|---|---|
| mg/kg | (%) | |||||
| Al | 25.0 ± 0.335 | 309 ± 18.78 | 334 ± 18.45 | 598 ± 7.1 | 4.19 ± 0.05 | 56 ± 3.08 |
| B | 21.8 ± 0.833 | 10.1 ± 0.099 | 32 ± 0.82 | 33 ± 0.42 | 66.1 ± 2.52 | 97 ± 2.49 |
| Ca | 31411 ± 149 | 16,310 ± 718 | 47,720 ± 867 | 50,450 ± 550 | 62.3 ± 0.29 | 95 ± 1.71 |
| Fe | 18.6 ± 0.03 | 228 ± 11.98 | 247 ± 12.03 | 367 ± 4.3 | 5.1 ± 0.009 | 67 ± 3.27 |
| K | 19,653 ± 161 | 589 ± 19.38 | 20,242 ± 182 | 26,760 ± 480 | 73.4 ± 0.61 | 76 ± 0.79 |
| Mg | 12,807 ± 82.3 | 545 ± 30.58 | 13,351 ± 113 | 12,000 | 107 ± 0.69 | 111 ± 0.94 |
| Mn | 213.6 ± 0.156 | 50.2 ± 2.103 | 264 ± 2.259 | 246 ± 7.1 | 86.8 ± 0.06 | 107 ± 0.92 |
| Sr | 58.9 ± 0.668 | 30.8 ± 1.408 | 90 ± 2.147 | 85.0 | 69.2 ± 0.79 | 105 ± 2.52 |
| Zn | 18.9 ± 0.664 | 10.2 ± 0.66 | 29 ± 1.332 | 30.9 ± 0.55 | 61.2 ± 2.15 | 94 ± 4.31 |
| Al | B | Ca | Fe | K | Mg | Mn | Sr | ||
|---|---|---|---|---|---|---|---|---|---|
| Species | Treatment | mg/kg of Fresh Weight | |||||||
| Rocket | Non-biofortified | 3.27 ± 0.13 | 2.66 ± 0.05 | 3155 ± 288 | 6.56 ± 0.28 | 7084 ± 545 | 482 ± 36.7 | 2.02 ± 0.02 | 6.23 ± 0.12 |
| Biofortified | 3.37 ± 0.25 | 2.72 ± 0.09 | 3572 ± 80.9 | 6.81 ± 0.15 | 7657 ± 627 | 556 ± 20.9 | 2.46 ± 0.16 | 6.10 ± 0.36 | |
| Significance | ns | ns | ns | ns | ns | ns | ns | ns | |
| Purslane | Non-biofortified | 0.89 ± 0.13 | 2.32 ± 0.06 | 875 ± 26.5 | 4.21 ± 0.17 | 4373 ± 199 | 818 ± 18.3 | 7.66 ± 0.34 | 2.54 ± 0.07 |
| Biofortified | 0.89 ± 0.07 | 2.41 ± 0.23 | 1004 ± 67.8 | 3.93 ± 0.31 | 4184 ± 212 | 894 ± 55.4 | 9.44 ± 0.91 | 3.00 ± 0.155 | |
| Significance | ns | ns | ns | ns | ns | ns | ns | ns | |
| Al | B | Ca | Fe | K | Mg | Mn | Sr | ||
|---|---|---|---|---|---|---|---|---|---|
| Species | Treatment | mg/kg of Fresh Weight | |||||||
| Rocket | Non-biofortified | 0.57 ± 0.02 | 2.28 ± 0.22 | 2352 ± 50.0 a | 1.24 ± 0.12 | 7428 ± 342 | 437 ± 13.4 | 1.56 ± 0.05 c | 6.15 ± 0.16 a |
| Biofortified | 0.49 ± 0.05 | 2.44 ± 0.22 | 2232 ± 1.47 b | 1.4 ± 0.07 | 7617 ± 254 | 466 ± 2.99 | 1.61 ± 0.06 c | 5.57 ± 0.04 b | |
| Purslane | Non-biofortified | 0.09 ± 0.01 | 1.77 ± 0.08 | 59.6 ± 2.39 c | 2.33 ± 0.08 | 4422 ± 75.2 | 818 ± 25.1 | 7.33 ± 0.40 b | 1.14 ± 0.02 c |
| Biofortified | 0.09 ± 0.01 | 1.96 ± 0.06 | 63.8 ± 3.96 c | 1.91 ± 0.27 | 4104 ± 41.8 | 880 ± 83.5 | 8.53 ± 0.29 a | 1.29 ± 0.06 c | |
| Significance | |||||||||
| Zn | ns | ns | ns | ns | ns | ns | ns | ns | |
| Species (S) | *** | ** | *** | *** | *** | *** | *** | *** | |
| Zn × S | ns | ns | * | ns | ns | ns | * | ** | |
| Daily Zn Intake (mg Zn/Day) | RDA-Zn Coverage (%) | HQ | |||
|---|---|---|---|---|---|
| Species | Treatment | Male | Female | ||
| Rocket | Non-biofortified | 0.43 ± 0.02 c | 3.88 ± 0.14 c | 5.34 ± 0.21 c | 0.278 ± 0.011 c |
| Biofortified | 0.74 ± 0.02 b | 6.79 ± 0.14 b | 9.29 ± 0.20 b | 0.534 ± 0.012 b | |
| Purslane | Non-biofortified | 0.32 ± 0.02 c | 2.94 ± 0.15 c | 4.05 ± 0.20 c | 0.233 ± 0.017 c |
| Biofortified | 1.69 ± 0.19 a | 15.4 ± 1.83 a | 21.14 ± 2.52 a | 1.086 ± 0.129 a | |
| Significance | |||||
| Zn | *** | *** | *** | *** | |
| Species (S) | *** | *** | *** | *** | |
| Zn × S | *** | *** | *** | *** | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
D’Imperio, M.; Montesano, F.F.; Serio, F.; Santovito, E.; Parente, A. Mineral Composition and Bioaccessibility in Rocket and Purslane after Zn Biofortification Process. Foods 2022, 11, 484. https://doi.org/10.3390/foods11030484
D’Imperio M, Montesano FF, Serio F, Santovito E, Parente A. Mineral Composition and Bioaccessibility in Rocket and Purslane after Zn Biofortification Process. Foods. 2022; 11(3):484. https://doi.org/10.3390/foods11030484
Chicago/Turabian StyleD’Imperio, Massimiliano, Francesco Fabiano Montesano, Francesco Serio, Elisa Santovito, and Angelo Parente. 2022. "Mineral Composition and Bioaccessibility in Rocket and Purslane after Zn Biofortification Process" Foods 11, no. 3: 484. https://doi.org/10.3390/foods11030484
APA StyleD’Imperio, M., Montesano, F. F., Serio, F., Santovito, E., & Parente, A. (2022). Mineral Composition and Bioaccessibility in Rocket and Purslane after Zn Biofortification Process. Foods, 11(3), 484. https://doi.org/10.3390/foods11030484










