Co-Encapsulation of Tannic Acid and Resveratrol in Zein/Pectin Nanoparticles: Stability, Antioxidant Activity, and Bioaccessibility
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Nutraceutical-Loaded Nanoparticles
2.3. Particle Size and Zeta Potential Measurements
2.4. Field Emission Scanning Electron Microscopy (SEM)
2.5. Nutraceutical Encapsulation
2.6. Nanoparticle Dispersion Stability
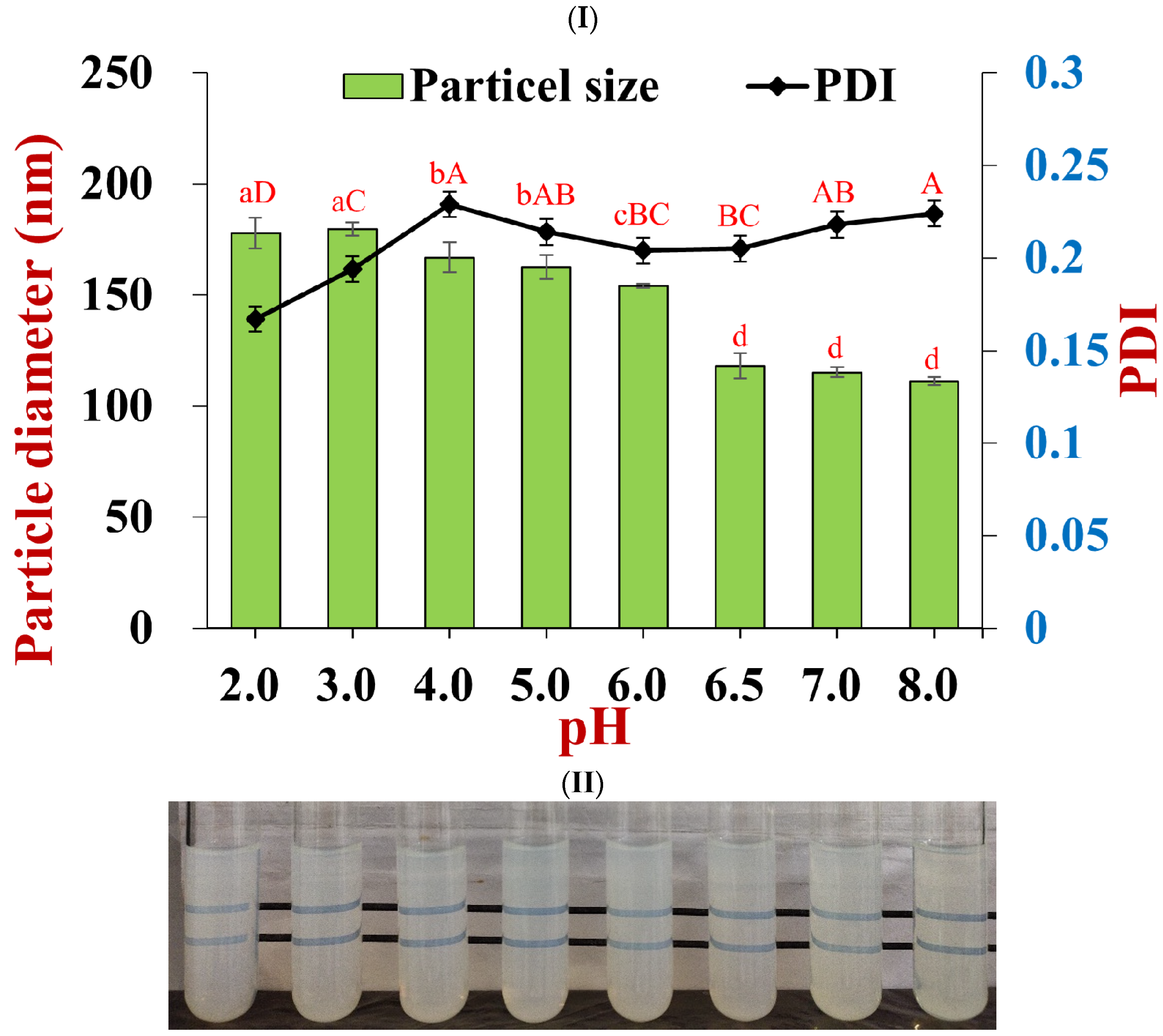
2.6.1. pH Stability
2.6.2. Salt Stability
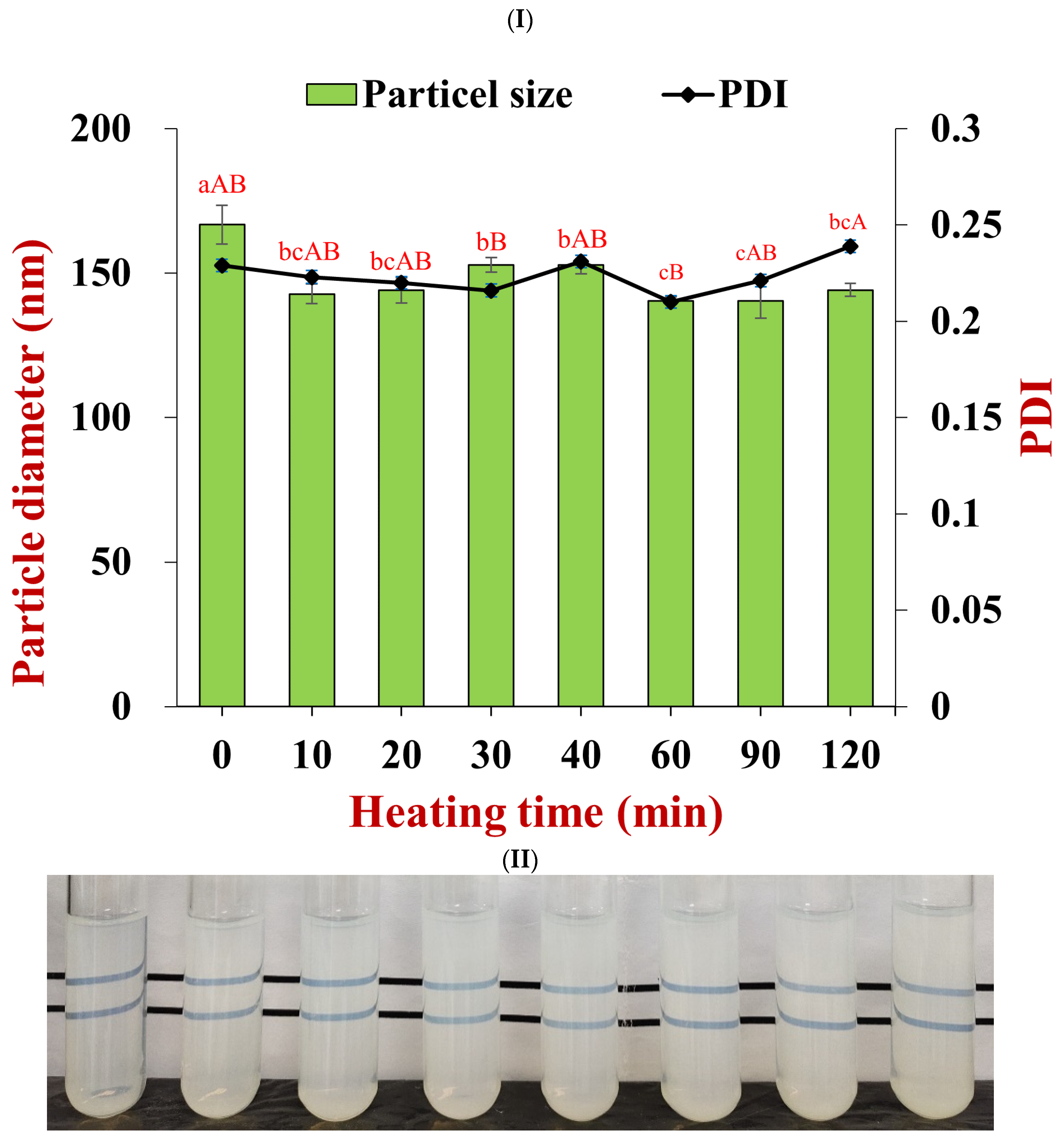
2.6.3. Heat Stability
2.6.4. Storage Stability
2.7. In Vitro Antioxidant Activity Determination of Co-Encapsulated Nanoparticles
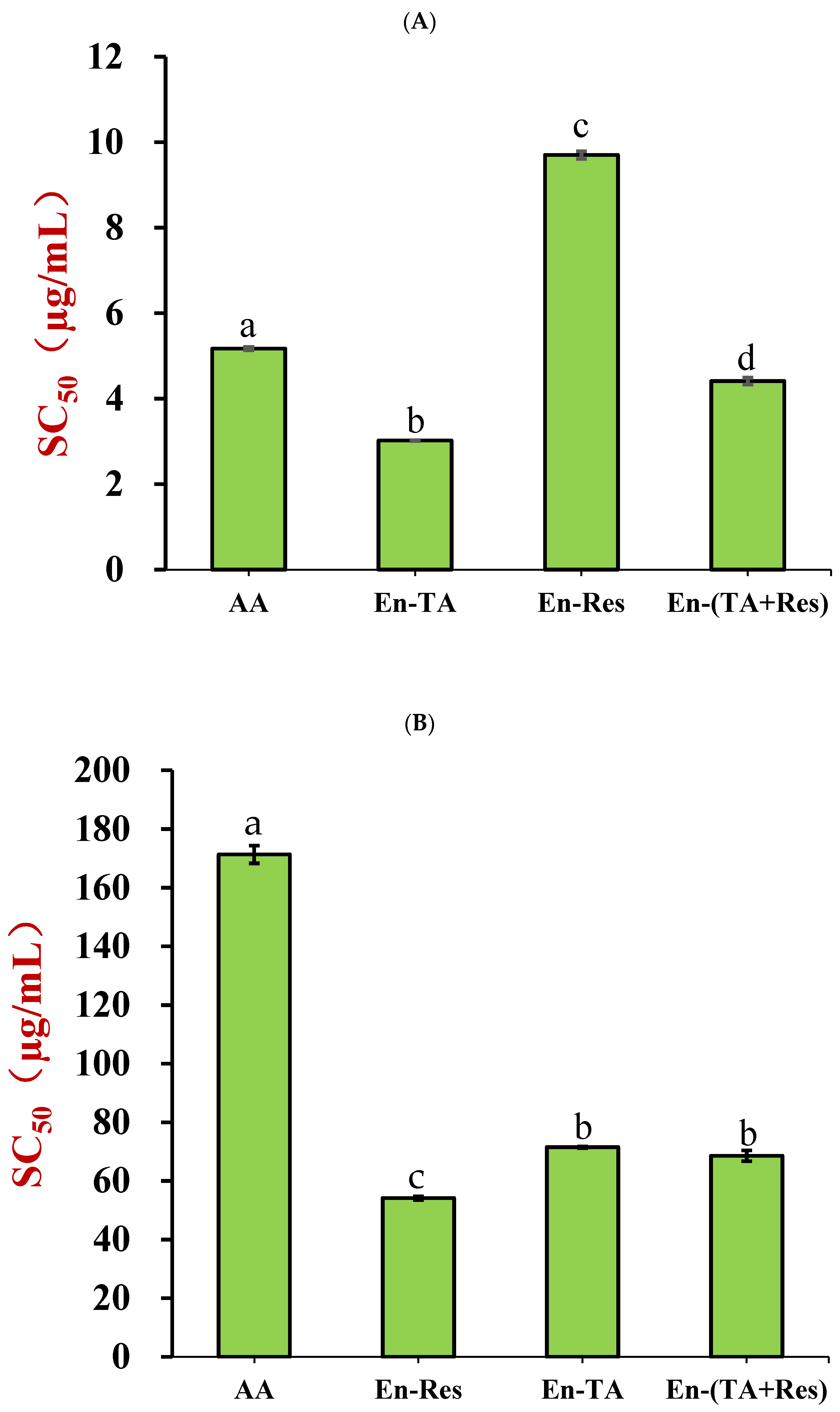
2.7.1. DPPH·Scavenging Activity
2.7.2. ABTS+· Scavenging Capacity
2.8. HepG2 Cell Culture and Cytotoxicity Assay
2.9. Determination of Oxidation Markers in Peroxide-Treated HepG2 Cells
2.10. Simulated Gastrointestinal Digestion
2.11. Statistical Analysis
3. Results and Discussion
3.1. Morphology and Loading Efficiency of Co-Encapsulated Nanoparticles
3.2. Particle Dispersion Stability under Environmental Stress
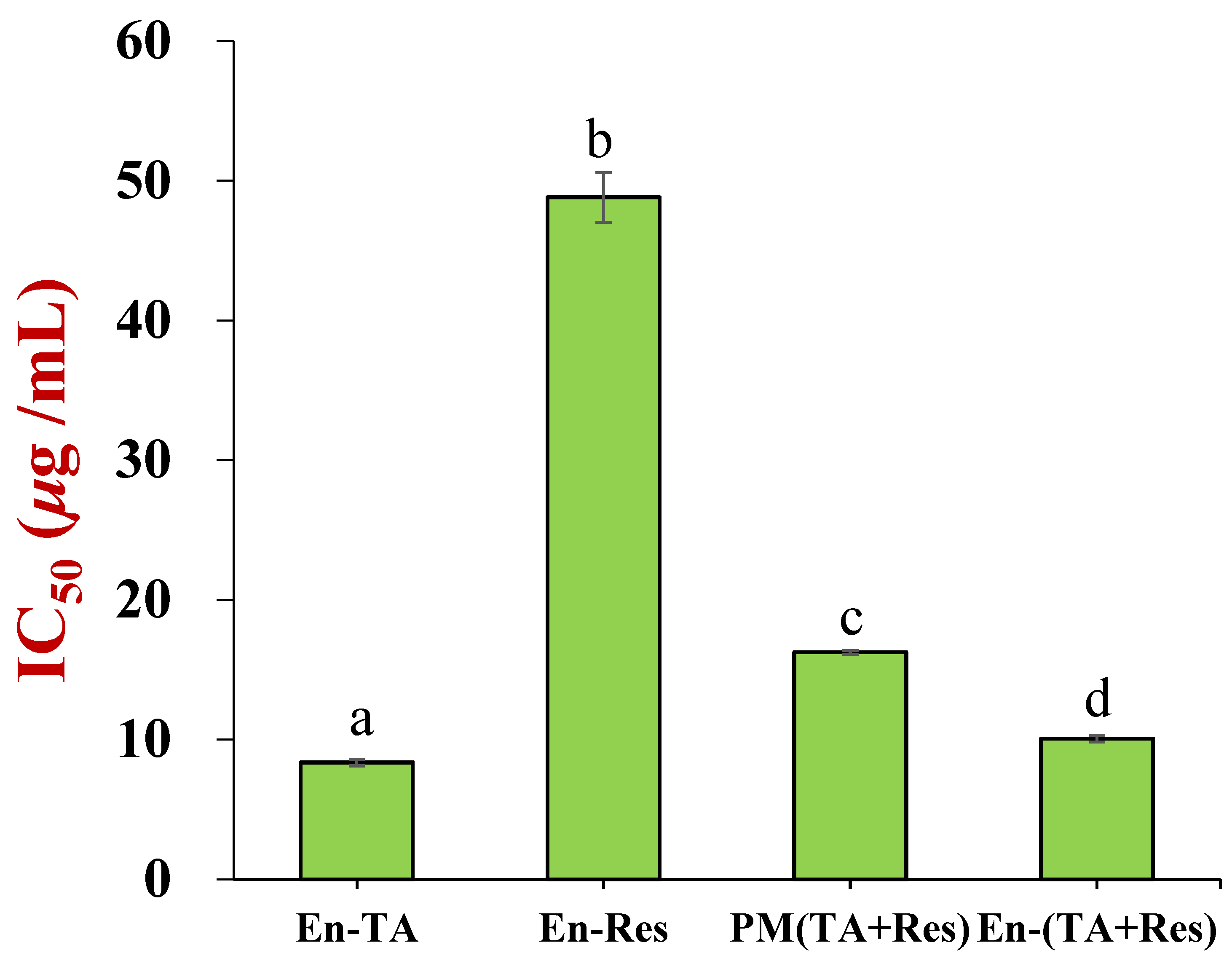
3.3. In Vitro Antioxidant Activities of Co-Encapsulated Nanoparticles
3.4. Effects of Co-Encapsulated Polyphenols on Peroxide-Induced Oxidative Stress in HepG2 Cells
3.4.1. HepG2 Cell Cytotoxicity
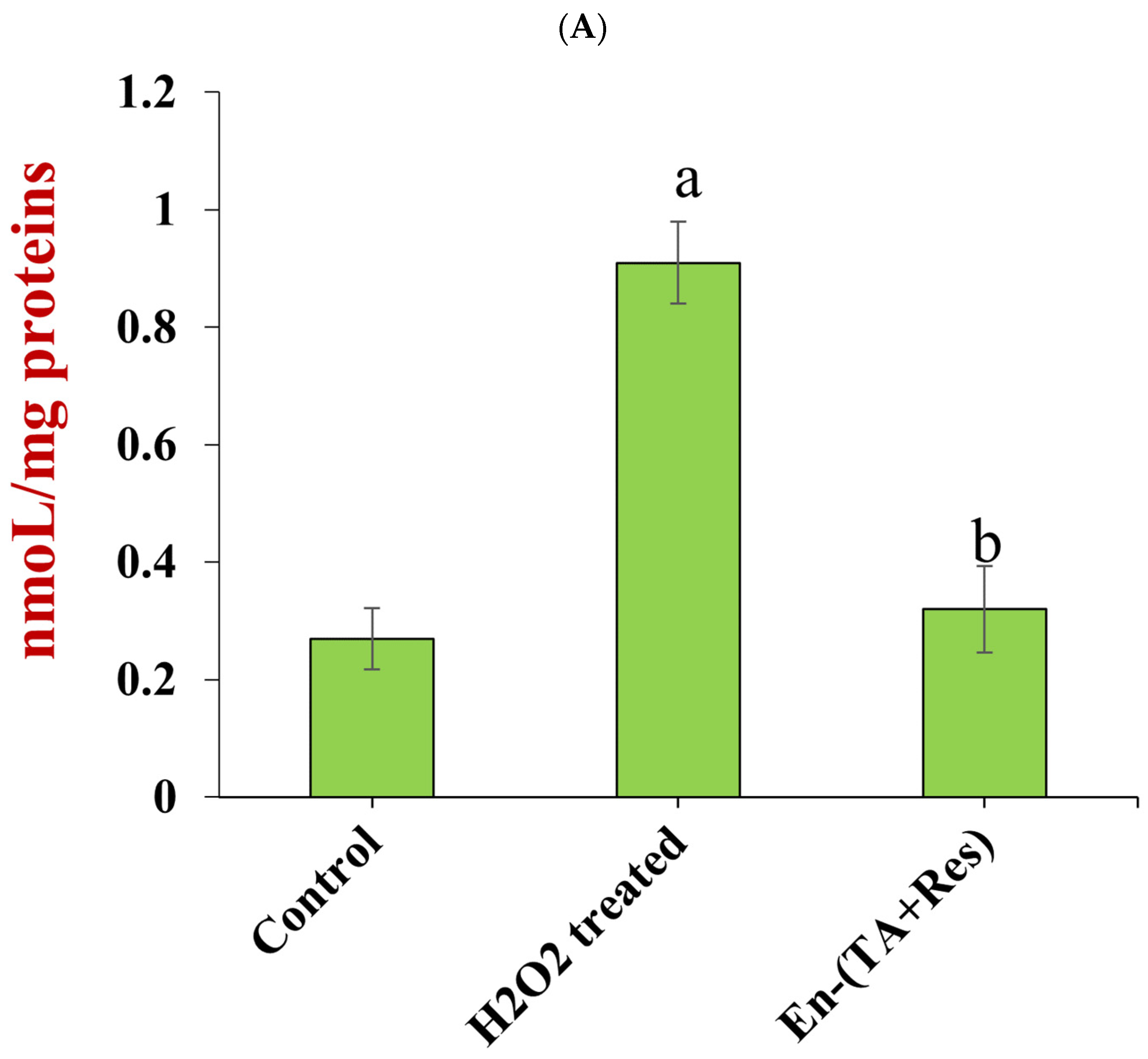
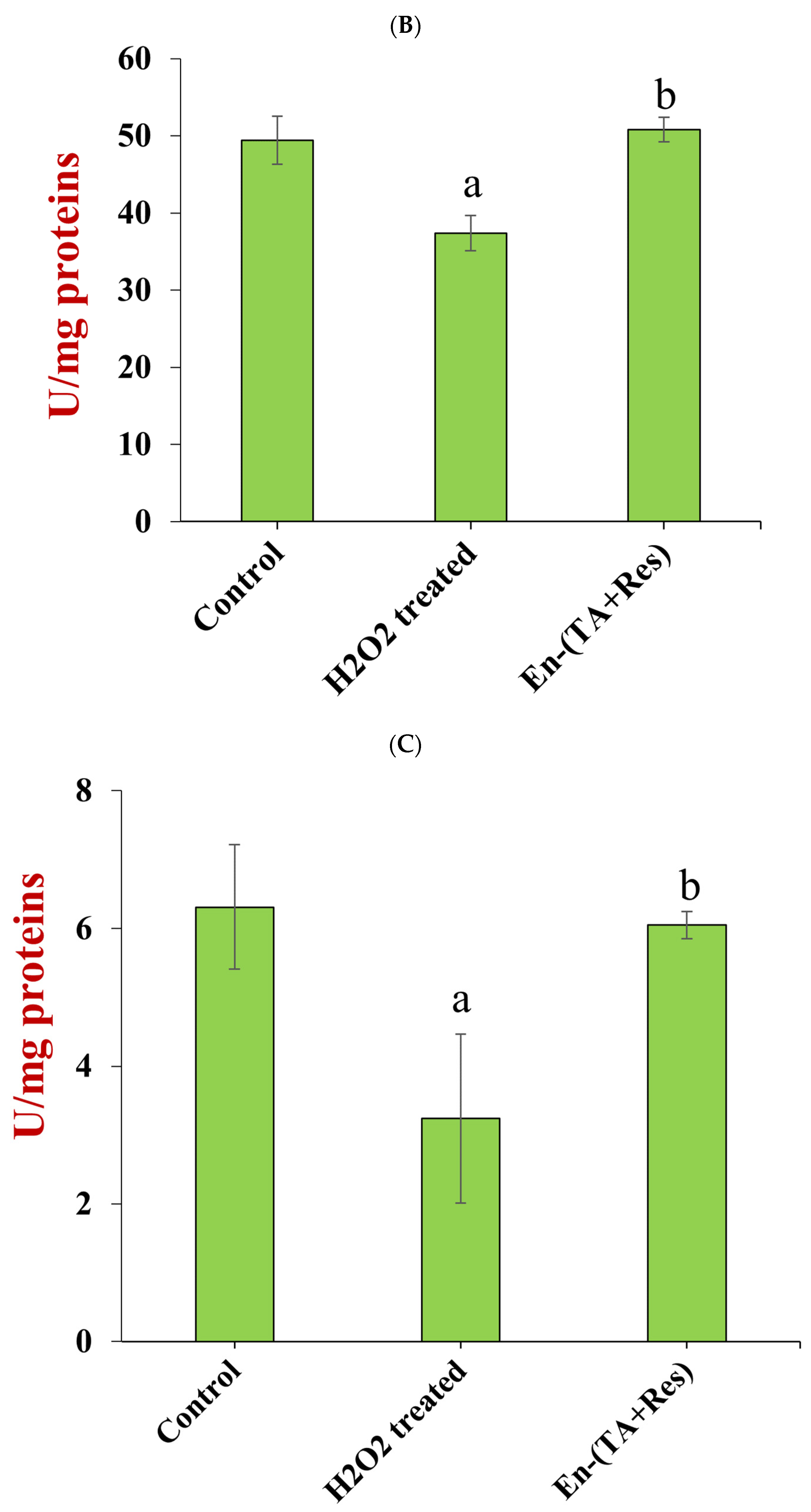
3.4.2. MDA Concentration and Antioxidant Enzyme Activity
3.5. Tannic Acid and Resveratrol Release under Simulated Gastrointestinal Conditions
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Maviah, M.B.J.; Farooq, M.A.; Mavlyanova, R.; Veroniaina, H.; Filli, M.S.; Aquib, M.; Kesse, S.; Boakye-Yiadom, K.O.; Wang, B. Food Protein-Based Nanodelivery Systems for Hydrophobic and Poorly Soluble Compounds. AAPS PharmSciTech 2020, 21, 101. [Google Scholar] [CrossRef]
- Patel, A.R.; Velikov, K.P. Colloidal delivery systems in foods: A general comparison with oral drug delivery. LWT-Food Sci. Technol. 2011, 44, 1958–1964. [Google Scholar] [CrossRef]
- Tan, Y.B.; McClements, D.J. Plant-Based Colloidal Delivery Systems for Bioactives. Molecules 2021, 26, 6895. [Google Scholar] [CrossRef] [PubMed]
- Hu, K.; McClements, D.J. Fabrication of biopolymer nanoparticles by antisolvent precipitation and electrostatic deposition: Zein-alginate core/shell nanoparticles. Food Hydrocoll. 2015, 44, 101–108. [Google Scholar] [CrossRef]
- Gagliardi, A.; Froiio, F.; Salvatici, M.C.; Paolino, D.; Cosco, D. Characterization and refinement of zein-based gels. Food Hydrocoll. 2019, 101, 105555. [Google Scholar] [CrossRef]
- Gomez-Estaca, J.; Balaguer, M.P.; Gavara, R.; Hernandez-Munoz, P. Formation of zein nanoparticles by electrohydrodynamic atomization: Effect of the main processing variables and suitability for encapsulating the food coloring and active ingredient curcumin. Food Hydrocoll. 2012, 28, 82–91. [Google Scholar] [CrossRef]
- Sun, X.; Pan, C.; Ying, Z.; Yu, D.; Ouyang, X.K. Stabilization of zein nanoparticles with k-carrageenan and tween 80 for encapsulation of curcumin. Int. J. Biol. Macromol. 2020, 146, 549–559. [Google Scholar] [CrossRef]
- Yuan, Y.; Ma, M.; Zhang, S.; Liu, C.; Xu, Y. Effect of sophorolipid on the curcumin-loaded ternary composite nanoparticles self-assembled from zein and chondroitin sulfate. Food Hydrocoll. 2021, 113, 106493. [Google Scholar] [CrossRef]
- Feng, Z.; Khan, M.A.; Hao, C.; Li, L. Co-encapsulation of α-tocopherol and resveratrol within zein nanoparticles: Impact on antioxidant activity and stability. J. Food Eng. 2018, 247, 9–18. [Google Scholar]
- Brotons-Canto, A.; Gonzalez-Navarro, C.J.; Gurrea, J.; Gonzalez-Ferrero, C.; Irache, J.M. Zein nanoparticles improve the oral bioavailability of resveratrol in humans. J. Drug Deliv. Sci. Technol. 2020, 57, 101704. [Google Scholar] [CrossRef]
- Hu, K.; McClements, D.J. Fabrication of surfactant-stabilized zein nanoparticles: A pH modulated antisolvent precipitation method. Food Res. Int. 2014, 64, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.; Hu, Y.; Tiwari, J.K.; Velikov, K.P. Synthesis and characterisation of zein–curcumin colloidal particles. Soft Matter 2010, 6, 6192–6199. [Google Scholar] [CrossRef]
- Chen, J.; Zheng, J.; McClements, D.J.; Xiao, H. Tangeretin-loaded protein nanoparticles fabricated from zein/beta-lactoglobulin: Preparation, characterization, and functional performance. Food Chem. 2014, 158, 466–472. [Google Scholar] [CrossRef]
- Hu, K.; Huang, X.; Gao, Y.; Huang, X.; Xiao, H.; McClements, D.J. Core-shell biopolymer nanoparticle delivery systems: Synthesis and characterization of curcumin fortified zein-pectin nanoparticles. Food Chem. 2015, 182, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Fu, Y.; Niu, F.; Zhang, H.; Li, X.; Li, X. Evaluation of the colloidal/chemical performance of core-shell nanoparticle formed by zein and gum Arabic. Colloids Surf. A Physicochem. Eng. Asp. 2019, 560, 130–135. [Google Scholar] [CrossRef]
- Ghobadi-Oghaz, N.; Asoodeh, A.; Mohammadi, M. Fabrication, characterization and in vitro cell exposure study of zein-chitosan nanoparticles for co-delivery of curcumin and berberine. Int. J. Biol. Macromol. 2022, 204, 576–586. [Google Scholar] [CrossRef]
- McClements, D.J. Nano-enabled personalized nutrition: Developing multicomponent-bioactive colloidal delivery systems. Adv. Colloid Interface Sci. 2020, 282, 102211. [Google Scholar] [CrossRef]
- Chen, S.; Li, Q.; McClements, D.J.; Han, Y.; Dai, L.; Mao, L.; Gao, Y. Co-delivery of curcumin and piperine in zein-carrageenan core-shell nanoparticles: Formation, structure, stability and in vitro gastrointestinal digestion. Food Hydrocoll. 2020, 99, 105334. [Google Scholar] [CrossRef]
- Carrasco-Sandoval, J.; Aranda-Bustos, M.; Henríquez-Aedo, K.; López-Rubio, A.; Fabra, M.J. Bioaccessibility of different types of phenolic compounds co-encapsulated in alginate/chitosan-coated zein nanoparticles. LWT 2021, 149, 112024. [Google Scholar] [CrossRef]
- Tchuenbou-Magaia, F.L.; Tolve, R.; Anyadike, U.; Giarola, M.; Favati, F. Co-encapsulation of vitamin D and rutin in chitosan-zein microparticles. J. Food Meas. Charact. 2022, 16, 2060–2070. [Google Scholar] [CrossRef]
- Chen, S.; Zhang, Y.; Han, Y.; McClements, D.J.; Liao, W.; Mao, L.; Yuan, F.; Gao, Y. Fabrication of multilayer structural microparticles for co-encapsulating coenzyme Q10 and piperine: Effect of the encapsulation location and interface thickness. Food Hydrocoll. 2020, 109, 106090. [Google Scholar] [CrossRef]
- Chen, S.; Han, Y.; Jian, L.; Liao, W.; Zhang, Y.; Gao, Y. Fabrication, characterization, physicochemical stability of zein-chitosan nanocomplex for co-encapsulating curcumin and resveratrol. Carbohydr. Polym. 2020, 236, 116090. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Zhang, Y.; Qing, J.; Han, Y.; McClements, D.J.; Gao, Y. Core-shell nanoparticles for co-encapsulation of coenzyme Q10 and piperine: Surface engineering of hydrogel shell around protein core. Food Hydrocoll. 2020, 103, 105651. [Google Scholar] [CrossRef]
- Baldwin, A.; Booth, B.W. Biomedical applications of tannic acid. J. Biomater. Appl. 2022, 36, 1503–1523. [Google Scholar] [CrossRef] [PubMed]
- Foegeding, E.A.; Plundrich, N.; Schneider, M.; Campbell, C.; Lila, M.A. Protein-polyphenol particles for delivering structural and health functionality. Food Hydrocoll. 2017, 72, 163–173. [Google Scholar] [CrossRef]
- Jakobek, L. Interactions of polyphenols with carbohydrates, lipids and proteins. Food Chem. 2015, 175, 556–567. [Google Scholar] [CrossRef] [PubMed]
- Gülçin, İ.; Huyut, Z.; Elmastaş, M.; Aboul-Enein, H.Y. Radical scavenging and antioxidant activity of tannic acid. Arab. J. Chem. 2010, 3, 43–53. [Google Scholar] [CrossRef]
- Liang, X.; Cao, K.; Li, W.; Li, X.; McClements, D.J.; Hu, K. Tannic acid-fortified zein-pectin nanoparticles: Stability, properties, antioxidant activity, and in vitro digestion. Food Res. Int. 2021, 145, 110425. [Google Scholar] [CrossRef]
- Jafari, H.; Ghaffari-Bohlouli, P.; Niknezhad, S.V.; Abedi, A.; Izadifar, Z.; Mohammadinejad, R.; Varma, R.S.; Shavandi, A. Tannic acid: A versatile polyphenol for design of biomedical hydrogels. J. Mater. Chem. B 2022, 10, 5873–5912. [Google Scholar] [CrossRef]
- Jing, Y.; Diao, Y.; Yu, X. Free radical-mediated conjugation of chitosan with tannic acid: Characterization and antioxidant capacity. React. Funct. Polym. 2019, 135, 16–22. [Google Scholar] [CrossRef]
- Liu, Y.; Liang, X.; Zou, Y.; Peng, Y.; McClements, D.J.; Hu, K. Resveratrol-loaded biopolymer core-shell nanoparticles: Bioavailability and anti-inflammatory effects. Food Funct. 2020, 11, 4014–4025. [Google Scholar] [CrossRef] [PubMed]
- Caddeo, C.; Manconi, M.; Fadda, A.M.; Lai, F.; Lampis, S.; Diez-Sales, O.; Sinico, C. Nanocarriers for antioxidant resveratrol: Formulation approach, vesicle self-assembly and stability evaluation. Colloids Surf. B Biointerfaces 2013, 111, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Caddeo, C.; Lucchesi, D.; Fernandez-Busquets, X.; Valenti, D.; Penno, G.; Fadda, A.M.; Pucci, L. Efficacy of a resveratrol nanoformulation based on a commercially available liposomal platform. Int. J. Pharm. 2021, 608, 121086. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Dai, Y.; Cai, J.; Zhong, N.; Xiao, H.; McClements, D.J.; Hu, K. Resveratrol encapsulation in core-shell biopolymer nanoparticles: Impact on antioxidant and anticancer activities. Food Hydrocoll. 2017, 64, 157–165. [Google Scholar] [CrossRef]
- Wu, W.; Kong, X.; Zhang, C.; Hua, Y.; Chen, Y.; Li, X. Fabrication and characterization of resveratrol-loaded gliadin nanoparticles stabilized by gum Arabic and chitosan hydrochloride. LWT 2020, 129, 109532. [Google Scholar] [CrossRef]
- Shehzad, Q.; Rehman, A.; Jafari, S.M.; Zuo, M.; Khan, M.A.; Ali, A.; Khan, S.; Karim, A.; Usman, M.; Hussain, A.; et al. Improving the oxidative stability of fish oil nanoemulsions by co-encapsulation with curcumin and resveratrol. Colloids Surf. B Biointerfaces 2021, 199, 111481. [Google Scholar] [CrossRef]
- Lu, Z.; Cheng, B.; Hu, Y.; Zhang, Y.; Zou, G. Complexation of resveratrol with cyclodextrins: Solubility and antioxidant activity. Food Chem. 2009, 113, 17–20. [Google Scholar] [CrossRef]
- Peng, Y.; Li, X.; Gu, P.; Cheng, W.; Zhang, R.; Hu, K. Curcumin-loaded zein/pectin nanoparticles: Caco-2 cellular uptake and the effects on cell cycle arrest and apoptosis of human hepatoma cells (HepG2). J. Drug Deliv. Sci. Technol. 2022, 74, 103497. [Google Scholar] [CrossRef]
- Huang, X.; Liu, Y.; Zou, Y.; Liang, X.; Peng, Y.; McClements, D.J.; Hu, K. Encapsulation of resveratrol in zein/pectin core-shell nanoparticles: Stability, bioaccessibility, and antioxidant capacity after simulated gastrointestinal digestion. Food Hydrocoll. 2019, 93, 261–269. [Google Scholar] [CrossRef]
- Zou, Y.; Lu, Y.; Wei, D. Antioxidant Activity of a Flavonoid-Rich Extract of Hypericum perforatum L. in Vitro. J. Agric. Food Chem. 2004, 52, 5032–5039. [Google Scholar] [CrossRef]
- Erel, O. A novel automated direct measurement method for total antioxidant capacity using a new generation, more stable ABTS radical cation. Clin. Biochem. 2004, 37, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Huang, X.; Gong, Y.; Xiao, H.; McClements, D.J.; Hu, K. Enhancement of curcumin water dispersibility and antioxidant activity using core-shell protein-polysaccharide nanoparticles. Food Res. Int. 2016, 87, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Yao, K.; Chen, W.; Song, F.; McClements, D.J.; Hu, K. Tailoring zein nanoparticle functionality using biopolymer coatings: Impact on curcumin bioaccessibility and antioxidant capacity under simulated gastrointestinal conditions. Food Hydrocoll. 2018, 79, 262–272. [Google Scholar] [CrossRef]
- de Souza, V.B.; Thomazini, M.; Echalar Barrientos, M.A.; Nalin, C.M.; Ferro-Furtado, R.; Genovese, M.I.; Favaro-Trindade, C.S. Functional properties and encapsulation of a proanthocyanidin-rich cinnamon extract (Cinnamomum zeylanicum) by complex coacervation using gelatin and different polysaccharides. Food Hydrocoll. 2018, 77, 297–306. [Google Scholar] [CrossRef]
- Leena, M.M.; Anukiruthika, T.; Moses, J.A.; Anandharamakrishnan, C. Co-delivery of curcumin and resveratrol through electrosprayed core-shell nanoparticles in 3D printed hydrogel. Food Hydrocoll. 2022, 124, 107200. [Google Scholar] [CrossRef]
- Shukla, R.; Cheryan, M. Zein: The industrial protein from corn. Ind. Crops Prod. 2001, 13, 171–192. [Google Scholar] [CrossRef]
- Osamudiamen, P.M.; Oluremi, B.B.; Oderinlo, O.O.; Aiyelaagbe, O.O. Trans-resveratrol, piceatannol and gallic acid: Potent polyphenols isolated from Mezoneuron benthamianum effective as anticaries, antioxidant and cytotoxic agents. Sci. Afr. 2020, 7, e00244. [Google Scholar] [CrossRef]
- Wechtersbach, L.; Poklar Ulrih, N.; Cigić, B. Liposomal stabilization of ascorbic acid in model systems and in food matrices. LWT-Food Sci. Technol. 2012, 45, 43–49. [Google Scholar] [CrossRef]
- Wang, W.; Zhang, L.; Chen, T.; Guo, W.; Bao, X.; Wang, D.; Ren, B.; Wang, H.; Li, Y.; Wang, Y.; et al. Anticancer Effects of Resveratrol-Loaded Solid Lipid Nanoparticles on Human Breast Cancer Cells. Molecules 2017, 22, 1814. [Google Scholar] [CrossRef]
- Yi, G.; Din, J.U.; Zhao, F.; Liu, X. Effect of soybean peptides against hydrogen peroxide induced oxidative stress in HepG2 cells via Nrf2 signaling. Food Funct. 2020, 11, 2725–2737. [Google Scholar] [CrossRef]
- Mateos, R.; Goya, L.; Bravo, L. Determination of malondialdehyde by liquid chromatography as the 2,4-dinitrophenylhydrazone derivative: A marker for oxidative stress in cell cultures of human hepatoma HepG2. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2004, 805, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Song, Q.; Han, X.; Zhang, Y.; Zhang, Y.; Zhang, X.; Chu, X.; Zhang, F.; Chu, L. Multi-targeted protection of acetaminophen-induced hepatotoxicity in mice by tannic acid. Int. Immunopharmacol. 2017, 47, 95–105. [Google Scholar] [CrossRef] [PubMed]




| Storage Days | Mean Size (nm) | PDI | ζ-Potential (mV) |
|---|---|---|---|
| 0 | 166.8 ± 6.75 a | 0.229 ± 0.006 a | −26.0 ± 1.72 a |
| 7 | 167.0 ± 6.96 a | 0.220 ± 0.016 a | −27.5 ± 0.29 a |
| 28 | 162.4 ± 8.68 a | 0.216 ± 0.007 a | −26.5 ± 0.60 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liang, X.; Cheng, W.; Liang, Z.; Zhan, Y.; McClements, D.J.; Hu, K. Co-Encapsulation of Tannic Acid and Resveratrol in Zein/Pectin Nanoparticles: Stability, Antioxidant Activity, and Bioaccessibility. Foods 2022, 11, 3478. https://doi.org/10.3390/foods11213478
Liang X, Cheng W, Liang Z, Zhan Y, McClements DJ, Hu K. Co-Encapsulation of Tannic Acid and Resveratrol in Zein/Pectin Nanoparticles: Stability, Antioxidant Activity, and Bioaccessibility. Foods. 2022; 11(21):3478. https://doi.org/10.3390/foods11213478
Chicago/Turabian StyleLiang, Xiao, Wanting Cheng, Zhanhong Liang, Yiling Zhan, David Julian McClements, and Kun Hu. 2022. "Co-Encapsulation of Tannic Acid and Resveratrol in Zein/Pectin Nanoparticles: Stability, Antioxidant Activity, and Bioaccessibility" Foods 11, no. 21: 3478. https://doi.org/10.3390/foods11213478
APA StyleLiang, X., Cheng, W., Liang, Z., Zhan, Y., McClements, D. J., & Hu, K. (2022). Co-Encapsulation of Tannic Acid and Resveratrol in Zein/Pectin Nanoparticles: Stability, Antioxidant Activity, and Bioaccessibility. Foods, 11(21), 3478. https://doi.org/10.3390/foods11213478








