Inhibitory Effect against Listeria monocytogenes of Carbon Nanoparticles Loaded with Copper as Precursors of Food Active Packaging
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Synthesis, Loading with Copper and Purification of CNPs
2.3. Physico-Chemical Characterization of CNPs
2.4. Evaluation of the Amount of Copper Loaded onto CNPs
2.5. CNP-Cu Microbiological Sterility Tests
2.6. Listeria Monocytogene Growth Trend Analysis
2.7. Preliminary Tests on a Suitable Volume of Growing Broth
2.8. MIC Protocol
2.9. Quantification of Spontaneous Copper Migration from CNP-Surface to BHI
2.10. Evaluation of Redox Activity of CNPs by Electronic Paramagnetic Resonance (EPR)
2.11. Evaluation of the Reduction of Copper through Bicinchoninic Acid (BCA) Assay
2.12. Incubation of SCNP and SCNP-Cu in Food Simulants
2.12.1. Aqueous Food Simulants
2.12.2. Food Simulant Olive Oil
2.13. Quantification of Copper Ions Spontaneously Released into Food Simulants
2.14. Statistical Analysis
3. Results and Discussion
3.1. Synthesis and Physical-Chemical Characterization of Carbon Nanoparticles
3.2. Antimicrobial Effect of Copper Loaded Nanoparticles
3.3. Mechanism of Action of Loaded CNPs
3.4. Assessment of the Release of Copper Ions in Food Simulants
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gourama, H. Foodborne Pathogens—FDA. In Food Engineering Series; Springer: Berlin/Heidelberg, Germany, 2020; pp. 25–49. [Google Scholar]
- EURL Lm Technical Guidance Document: On Challenge Tests and Durability Studies for Assessing Shelf-Life of Ready-To-Eat Foods Related to Listeria Monocytogenes. 2021, pp. 1–60. Available online: https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&ved=2ahUKEwjNop7Hpaj6AhXjX_EDHec_CV8QFnoECAkQAQ&url=https%3A%2F%2Ffood.ec.europa.eu%2Fsystem%2Ffiles%2F2021-07%2Fbiosafety_fh_mc_tech-guide-doc_listeria-in-rte-foods_en_0.pdf&usg=AOvVaw2h9eTZWDgMpPedG-MDD6od (accessed on 14 September 2022).
- Lanni, L.; Morena, V.; Marchese, A.S.; Destro, G.; Ferioli, M.; Catellani, P.; Giaccone, V. Challenge Test as Special Tool to Estimate the Dynamic of Listeria monocytogenes and Other Foodborne Pathogens. Foods 2021, 11, 32. [Google Scholar] [CrossRef] [PubMed]
- Von Nussbaum, F.; Brands, M.; Hinzen, B.; Weigand, S.; Häbich, D. Antibacterial Natural Products in Medicinal Chemistry—Exodus or Revival? Angew. Chem. Int. Ed. 2006, 45, 5072–5129. [Google Scholar] [CrossRef] [PubMed]
- Makvandi, P.; Wang, C.Y.; Zare, E.N.; Borzacchiello, A.; Niu, L.N.; Tay, F.R. Metal-Based Nanomaterials in Biomedical Applications: Antimicrobial Activity and Cytotoxicity Aspects. Adv. Funct. Mater. 2020, 30, 1910021. [Google Scholar] [CrossRef]
- Zhang, M.; Zhang, K.; De Gusseme, B.; Verstraete, W.; Field, R. The antibacterial and anti-biofouling performance of biogenic silver nanoparticles by Lactobacillus fermentum. Biofouling 2014, 30, 347–357. [Google Scholar] [CrossRef]
- Franci, G.; Falanga, A.; Galdiero, S.; Palomba, L.; Rai, M.; Morelli, G.; Galdiero, M. Silver Nanoparticles as Potential Antibacterial Agents. Molecules 2015, 20, 8856–8874. [Google Scholar] [CrossRef]
- Dizaj, S.M.; Lotfipour, F.; Barzegar-Jalali, M.; Zarrintan, M.H.; Adibkia, K. Antimicrobial activity of the metals and metal oxide nanoparticles. Mater. Sci. Eng. C 2014, 44, 278–284. [Google Scholar] [CrossRef]
- Ingle, A.P.; Durán, N.; Rai, M. Bioactivity, mechanism of action, and cytotoxicity of copper-based nanoparticles: A review. Appl. Microbiol. Biotechnol. 2013, 98, 1001–1009. [Google Scholar] [CrossRef]
- Mamusa, M.; Barbero, F.; Montis, C.; Cutillo, L.; Gonzalez-Paredes, A.; Berti, D. Inclusion of oligonucleotide antimicrobials in biocompatible cationic liposomes: A structural study. J. Colloid Interface Sci. 2017, 508, 476–487. [Google Scholar] [CrossRef]
- Tacu, I.; Kokalari, I.; Abollino, O.; Albrecht, C.; Malandrino, M.; Ferretti, A.M.; Schins, R.P.F.; Fenoglio, I. Mechanistic Insights into the Role of Iron, Copper, and Carbonaceous Component on the Oxidative Potential of Ultrafine Particulate Matter. Chem. Res. Toxicol. 2021, 34, 767–779. [Google Scholar] [CrossRef]
- Mohamed, N.A.; El-Ghany, N.A.A. Novel aminohydrazide cross-linked chitosan filled with multi-walled carbon nanotubes as antimicrobial agents. Int. J. Biol. Macromol. 2018, 115, 651–662. [Google Scholar] [CrossRef]
- Song, K.; Gao, A.; Cheng, X.; Xie, K. Preparation of the superhydrophobic nano-hybrid membrane containing carbon nanotube based on chitosan and its antibacterial activity. Carbohydr. Polym. 2015, 130, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Jamróz, E.; Kopel, P.; Tkaczewska, J.; Dordevic, D.; Jancikova, S.; Kulawik, P.; Milosavljevic, V.; Dolezelikova, K.; Smerkova, K.; Svec, P.; et al. Nanocomposite Furcellaran Films—the Influence of Nanofillers on Functional Properties of Furcellaran Films and Effect on Linseed Oil Preservation. Polymers 2019, 11, 2046. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, N.; Ostadhossein, F.; Simchi, A. Physicochemical and antibacterial properties of chitosan-polyvinylpyrrolidone films containing self-organized graphene oxide nanolayers. J. Appl. Polym. Sci. 2016, 133, 43194. [Google Scholar] [CrossRef]
- Pal, N.; Dubey, P.; Gopinath, P.; Pal, K. Combined effect of cellulose nanocrystal and reduced graphene oxide into poly-lactic acid matrix nanocomposite as a scaffold and its anti-bacterial activity. Int. J. Biol. Macromol. 2017, 95, 94–105. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, S.; Lan, W.; Qin, W. Fabrication of polylactic acid/carbon nanotubes/chitosan composite fibers by electrospinning for strawberry preservation. Int. J. Biol. Macromol. 2018, 121, 1329–1336. [Google Scholar] [CrossRef]
- Montes, S.; Etxeberria, A.; Mocholi, V.; Rekondo, A.; Grande, H.; Labidi, J. Effect of combining cellulose nanocrystals and graphene nanoplatelets on the properties of poly(lactic acid) based films. Express Polym. Lett. 2018, 12, 543–555. [Google Scholar] [CrossRef]
- Rivera-Briso, A.L.; Aachmann, F.L.; Moreno-Manzano, V.; Serrano-Aroca, Á. Graphene oxide nanosheets versus carbon nanofibers: Enhancement of physical and biological properties of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) films for biomedical applications. Int. J. Biol. Macromol. 2020, 143, 1000–1008. [Google Scholar] [CrossRef]
- Borkow, G.; Gabbay, J. Copper, An Ancient Remedy Returning to Fight Microbial, Fungal and Viral Infections. Curr. Chem. Biol. 2009, 3, 272–278. [Google Scholar] [CrossRef]
- Wilks, S.A.; Michels, H.T.; Keevil, C.W. Survival of Listeria monocytogenes Scott A on metal surfaces: Implications for cross-contamination. Int. J. Food Microbiol. 2006, 111, 93–98. [Google Scholar] [CrossRef]
- Giachino, A.; Waldron, K.J. Copper tolerance in bacteria requires the activation of multiple accessory pathways. Mol. Microbiol. 2020, 114, 377–390. [Google Scholar] [CrossRef]
- Latorre, M.; Quesille-Villalobos, A.M.; Maza, F.; Parra, A.; Reyes-Jara, A. Synergistic effect of copper and low temperature over Listeria monocytogenes. BioMetals 2015, 28, 1087–1092. [Google Scholar] [CrossRef]
- Lemire, J.A.; Harrison, J.J.; Turner, R.J. Antimicrobial activity of metals: Mechanisms, molecular targets and applications. Nat. Rev. Microbiol. 2013, 11, 371–384. [Google Scholar] [CrossRef]
- Shankar, S.; Rhim, J.-W. Effect of copper salts and reducing agents on characteristics and antimicrobial activity of copper nanoparticles. Mater. Lett. 2014, 132, 307–311. [Google Scholar] [CrossRef]
- Velsankar, K.; Suganya, S.; Muthumari, P.; Mohandoss, S.; Sudhahar, S. Ecofriendly green synthesis, characterization and biomedical applications of CuO nanoparticles synthesized using leaf extract of Capsicum frutescens. J. Environ. Chem. Eng. 2021, 9, 106299. [Google Scholar] [CrossRef]
- Zuily, L.; Lahrach, N.; Fassler, R.; Genest, O.; Faller, P.; Sénèque, O.; Denis, Y.; Castanié-Cornet, M.-P.; Genevaux, P.; Jakob, U.; et al. Copper Induces Protein Aggregation, a Toxic Process Compensated by Molecular Chaperones. mBio 2022, 13, e03251–21. [Google Scholar] [CrossRef] [PubMed]
- Ameh, T.; Sayes, C.M. The potential exposure and hazards of copper nanoparticles: A review. Environ. Toxicol. Pharmacol. 2019, 71, 103220. [Google Scholar] [CrossRef] [PubMed]
- Alayande, A.B.; Obaid, M.; Kim, I.S. Antimicrobial mechanism of reduced graphene oxide-copper oxide (rGO-CuO) nanocomposite films: The case of Pseudomonas aeruginosa PAO1. Mater. Sci. Eng. C 2019, 109, 110596. [Google Scholar] [CrossRef] [PubMed]
- Mitra, D.; Kang, E.-T.; Neoh, K.G. Antimicrobial Copper-Based Materials and Coatings: Potential Multifaceted Biomedical Applications. ACS Appl. Mater. Interfaces 2019, 12, 21159–21182. [Google Scholar] [CrossRef]
- Tamayo, L.; Zapata, P.; Vejar, N.; Azócar, M.; Gulppi, M.; Zhou, X.; Thompson, G.; Rabagliati, F.; Páez, M. Release of silver and copper nanoparticles from polyethylene nanocomposites and their penetration into Listeria monocytogenes. Mater. Sci. Eng. C 2014, 40, 24–31. [Google Scholar] [CrossRef]
- Azam, A.; Ahmed, A.S.; Oves, M.; Khan, M.; Memic, A. Size-dependent antimicrobial properties of CuO nanoparticles against Gram-positive and -negative bacterial strains. Int. J. Nanomed. 2012, 7, 3527–3535. [Google Scholar] [CrossRef]
- Yoon, K.-Y.; Byeon, J.H.; Park, J.-H.; Hwang, J. Susceptibility constants of Escherichia coli and Bacillus subtilis to silver and copper nanoparticles. Sci. Total Environ. 2007, 373, 572–575. [Google Scholar] [CrossRef]
- Shankar, S.; Wang, L.-F.; Rhim, J.-W. Preparation and properties of carbohydrate-based composite films incorporated with CuO nanoparticles. Carbohydr. Polym. 2017, 169, 264–271. [Google Scholar] [CrossRef]
- Dey, A.; Pandey, G.; Rawtani, D. Functionalized nanomaterials driven antimicrobial food packaging: A technological advancement in food science. Food Control 2021, 131, 108469. [Google Scholar] [CrossRef]
- European Commission. Commission Regulation (EU) No 10/2011 of 14 January 2011 on plastic materials and articles intended to come into contact with food. Off. J. Eur. Union 2011, L12, 1. [Google Scholar]
- Mohan, R.; Shanmugharaj, A.M.; Hun, R.S. An efficient growth of silver and copper nanoparticles on multiwalled carbon nanotube with enhanced antimicrobial activity. J. Biomed. Mater. Res. Part B: Appl. Biomater. 2010, 96B, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Falco, C.; Weber, J.; White, R.J.; Howe, J.Y.; Titirici, M.-M. Carbohydrate-Derived Hydrothermal Carbons: A Thorough Characterization Study. Langmuir 2012, 28, 12373–12383. [Google Scholar] [CrossRef]
- Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically; Approved Standard, 9th ed.; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2012; volume 32.
- Madeo, M.; Musumeci, R.; Careddu, A.M.L.; Amato, E.; Pontello, M.M.; Cocuzza, C.E. Antimicrobial susceptibility of Listeria monocytogenes isolates from human cases in northern Italy, 2008–2010: MIC determination according to EUCAST broth microdilution method. J. Chemother. 2015, 27, 201–206. [Google Scholar] [CrossRef]
- ISO 11290-2; Microbiology of the Food Chain—Horizontal Method for the Detection and Enumeration of Listeria Monocytogenes and of Listeria spp.—Part 2: Enumeration Method. ISO: Geneva, Switzerland, 2017.
- Kokalari, I.; Gassino, R.; Giovannozzi, A.M.; Croin, L.; Gazzano, E.; Bergamaschi, E.; Rossi, A.M.; Perrone, G.; Riganti, C.; Ponti, J.; et al. Pro- and anti-oxidant properties of near-infrared (NIR) light responsive carbon nanoparticles. Free Radic. Biol. Med. 2019, 134, 165–176. [Google Scholar] [CrossRef]
- ISO 7218—Amendment 2013; Microbiology of Food and Animal Feeding Stuffs—General Requirements and Guidance for Microbiological Examinations. ISO: Geneva, Switzerland, 2007.
- EFSA Scientific Committee; Hardy, A.; Benford, D.; Halldorsson, T.; Jeger, M.J.; Knutsen, H.K.; More, S.; Naegeli, H.; Noteborn, H.; Ockleford, C.; et al. Guidance on risk assessment of the application of nanoscience and nanotechnologies in the food and feed chain: Part 1, human and animal health. EFSA J. 2018, 16, e05327. [Google Scholar] [CrossRef]
- Biel, S.S.; Gelderblom, H.R. Diagnostic electron microscopy is still a timely and rewarding method. J. Clin. Virol. 1999, 13, 105–119. [Google Scholar] [CrossRef]
- ISO/IEC 17025; General Requirements for the Competence of Testing and Calibration Laboratories. ISO: Geneva, Switzerland, 2017.
- ISO 11133; Microbiology of Food, Animal Feed and Water—Preparation, Production, Storage and Performance Testing of Culture Media. ISO: Geneva, Switzerland, 2018.
- ISO 20976-1; Microbiology of the Food Chain—Requirements and Guidelines for Conducting Challenge Tests of Food and Feed Products—Part 1: Challenge Tests to Study Growth Potential, Lag Time and Maximum Growth Rate. ISO: Geneva, Switzerland, 2019.
- Atcc. Listeria Monocytogenes (Murray et al.) Pirie Product Sheet (19117). Available online: www.atcc.org (accessed on 22 June 2022).
- Atcc. ATCC Bacteriology Culture Guide. Available online: www.atcc.org (accessed on 12 April 2021).
- ISO 6887-1; Microbiology of the Food Chain—Preparation of Test Samples, Initial Suspension and Decimal Dilutions for Microbiological Examination—Part 1: General Rules for the Preparation of the Initial Suspension and Decimal Dilutions. ISO: Geneva, Switzerland, 2017.
- ISO 20776-1; Susceptibility Testing of Infectious Agents and Evaluation of Performance of Antimicrobial Susceptibility Test Devices—Part 1: Broth Micro-Dilution Reference Method for Testing the In Vitro Activity of Antimicrobial Agents against Rapidly Growing aerobic Bacteria Involved in Infectious Diseases. ISO: Geneva, Switzerland, 2019.
- Cassella, R.J.; Brum, D.M.; de Paula, C.E.R.; Lima, C.F. Extraction induced by emulsion breaking: A novel strategy for the trace metals determination in diesel oil samples by electrothermal atomic absorption spectrometry. J. Anal. At. Spectrom. 2010, 25, 1704–1711. [Google Scholar] [CrossRef]
- da Silva, A.F.; Papai, R.; Luz, M.S.; Gaubeur, I. Analytical extraction procedure combined with atomic and mass spectrometry for the determination of tin in edible oil samples, and the potential application to other chemical elements. J. Food Compos. Anal. 2021, 96, 103759. [Google Scholar] [CrossRef]
- Bakircioglu, D.; Kurtulus, Y.B.; Yurtsever, S. Comparison of extraction induced by emulsion breaking, ultrasonic extraction and wet digestion procedures for determination of metals in edible oil samples in Turkey using ICP-OES. Food Chem. 2013, 138, 770–775. [Google Scholar] [CrossRef]
- Wuyke, H.; Oropeza, T.; Feo, L. Extraction induced by emulsion breaking for the determination of As, Co, Cr, Mn, Mo and Pb in heavy and extra-heavy crude oil samples by ICP-MS. Anal. Methods 2017, 9, 1152–1160. [Google Scholar] [CrossRef]
- Soddu, L.; Trinh, D.N.; Dunne, E.; Kenny, D.; Bernardini, G.; Kokalari, I.; Marucco, A.; Monopoli, M.P.; Fenoglio, I. Identification of physicochemical properties that modulate nanoparticle aggregation in blood. Beilstein J. Nanotechnol. 2020, 11, 550–567. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Commission Recommendation of 10.6.2022 on the definition of nanomaterial. Off. J. Eur. Union 2022, C229, 1. [Google Scholar]
- Kralik, P.; Beran, V.; Pavlik, I. Enumeration of Mycobacterium avium subsp. paratuberculosis by quantitative real-time PCR, culture on solid media and optical densitometry. BMC Res. Notes 2012, 5, 114. [Google Scholar] [CrossRef]
- Reyes-Jara, A.; Cordero, N.; Aguirre, J.; Troncoso, M.; Figueroa, G. Antibacterial Effect of Copper on Microorganisms Isolated from Bovine Mastitis. Front. Microbiol. 2016, 7, 626. [Google Scholar] [CrossRef]
- Parsons, C.; Lee, S.; Kathariou, S. Heavy Metal Resistance Determinants of the Foodborne Pathogen Listeria monocytogenes. Genes 2018, 10, 11. [Google Scholar] [CrossRef]
- Ladomersky, E.; Petris, M.J. Copper tolerance and virulence in bacteria. Metallomics 2015, 7, 957–964. [Google Scholar] [CrossRef]
- European Commission. Commission Regulation (EC) No 2073/2005 of 15 November 2005 on microbiological criteria for foodstuffs. Off. J. Eur. Union 2005, L338, 1. [Google Scholar]
- Park, E.-J.; Yi, J.; Kim, Y.; Choi, K.; Park, K. Silver nanoparticles induce cytotoxicity by a Trojan-horse type mechanism. Toxicol. Vitr. 2010, 24, 872–878. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Zeng, T.H.; Hofmann, M.; Burcombe, E.; Wei, J.; Jiang, R.; Kong, J.; Chen, Y. Antibacterial Activity of Graphite, Graphite Oxide, Graphene Oxide, and Reduced Graphene Oxide: Membrane and Oxidative Stress. ACS Nano 2011, 5, 6971–6980. [Google Scholar] [CrossRef]
- Barzan, G.; Kokalari, I.; Gariglio, G.; Ghibaudi, E.; Devocelle, M.; Monopoli, M.P.; Sacco, A.; Greco, A.; Giovannozzi, A.M.; Rossi, A.M.; et al. Molecular Aspects of the Interaction with Gram-Negative and Gram-Positive Bacteria of Hydrothermal Carbon Nanoparticles Associated with Bac8c2,5LeuAntimicrobial Peptide. ACS Omega 2022, 7, 16402–16413. [Google Scholar] [CrossRef] [PubMed]
- Mackevica, A.; Olsson, M.E.; Hansen, S.F. Silver nanoparticle release from commercially available plastic food containers into food simulants. J. Nanoparticle Res. 2016, 18, 5. [Google Scholar] [CrossRef]
- Koo, Y.J.; Pack, E.C.; Lee, Y.J.; Kim, H.S.; Jang, D.Y.; Lee, S.H.; Kim, Y.S.; Lim, K.M.; Choi, D.W. Determination of toxic metal release from metallic kitchen utensils and their health risks. Food Chem. Toxicol. 2020, 145, 111651. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Hurt, R.H. Ion Release Kinetics and Particle Persistence in Aqueous Nano-Silver Colloids. Environ. Sci. Technol. 2010, 44, 2169–2175. [Google Scholar] [CrossRef]
- Phothisarattana, D.; Harnkarnsujarit, N. Migration, aggregations and thermal degradation behaviors of TiO2 and ZnO incorporated PBAT/TPS nanocomposite blown films. Food Packag. Shelf Life 2022, 33, 100901. [Google Scholar] [CrossRef]
- Jokar, M.; Correia, M.; Loeschner, K. Behavior of silver nanoparticles and ions in food simulants and low fat cow milk under migration conditions. Food Control 2018, 89, 77–85. [Google Scholar] [CrossRef]

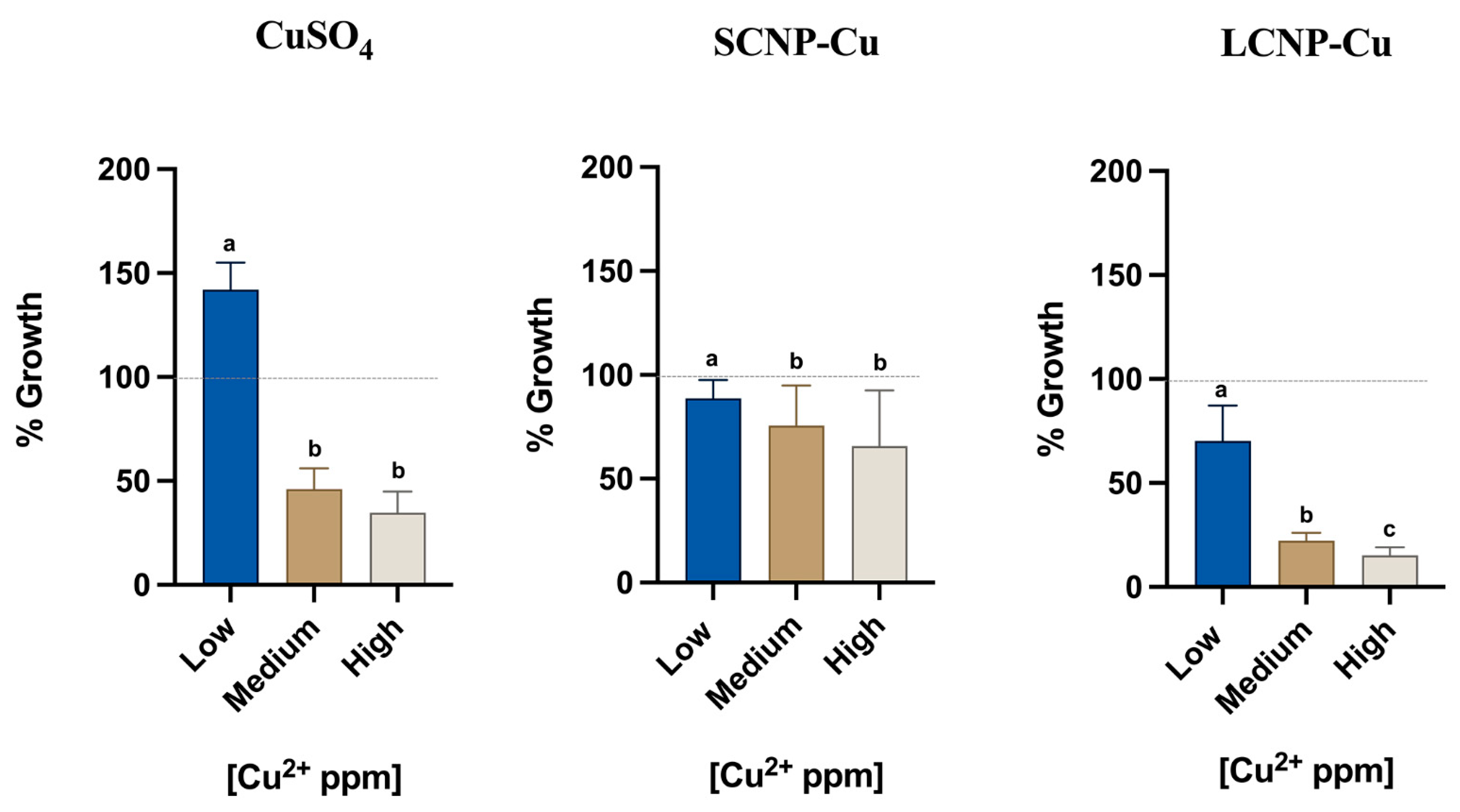
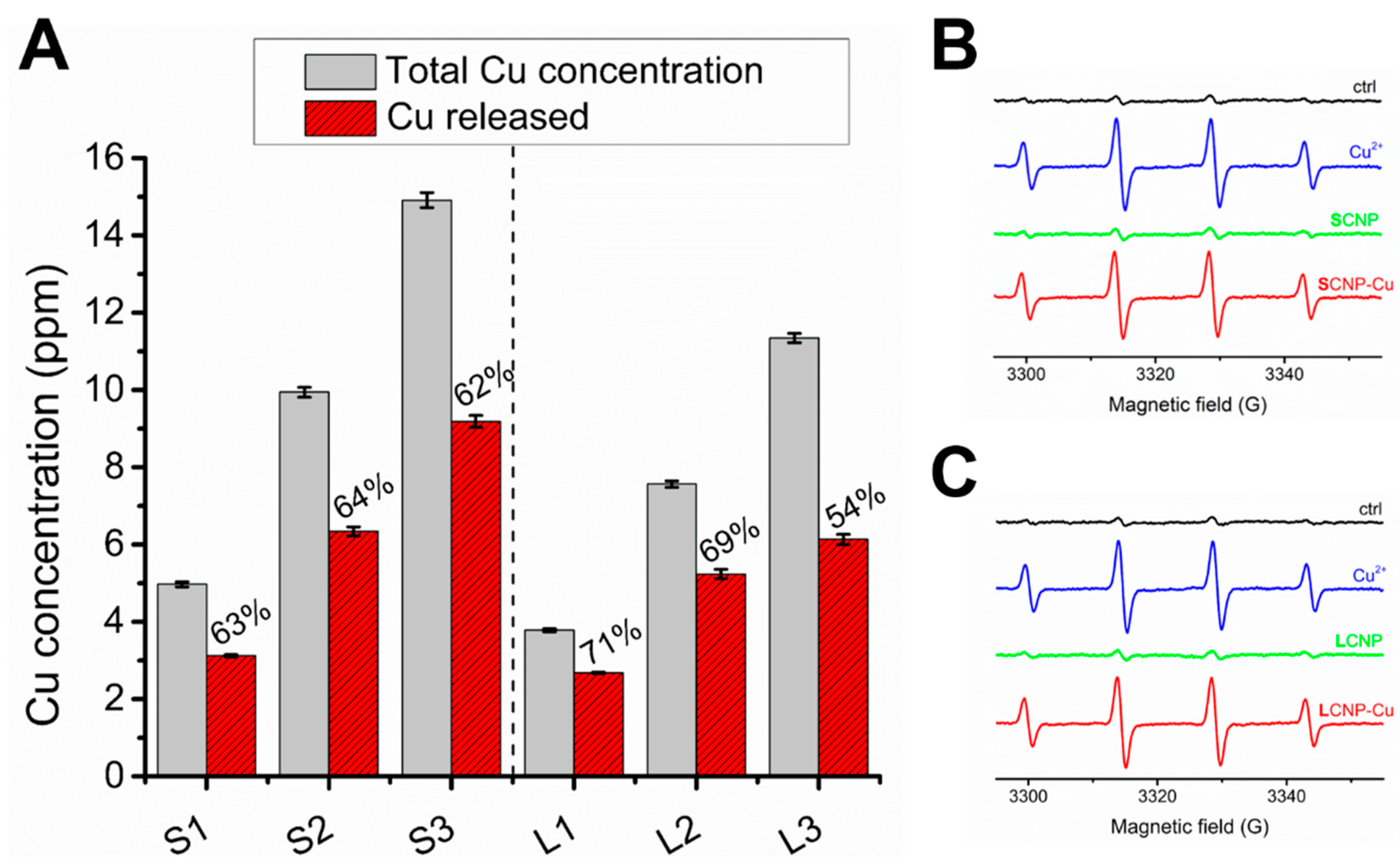
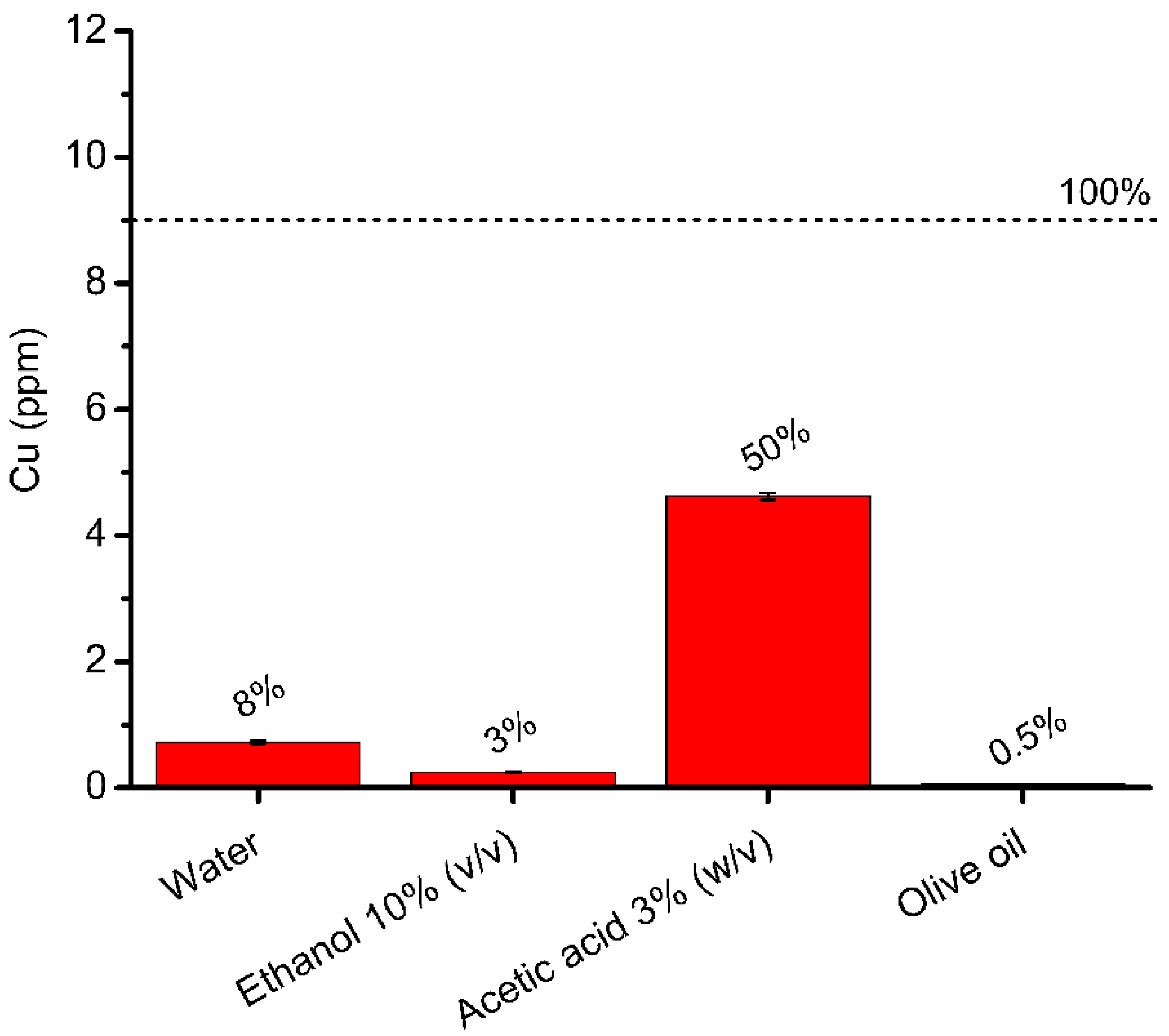
| Sample (Cu2+ ppm) | CuSO4 | SCNP-Cu | LCNP-Cu |
|---|---|---|---|
| Low | 5.0 | 5.0 | 3.9 |
| Medium | 11.1 | 9.9 | 7.8 |
| High | 19.0 | 14.9 | 11.6 |
| Hydrodynamic Diameter (Z-Average) nm ± SD * | PdI | ζ-Potential (mV) | Amount of Cu Loaded (µg Cu/mg CNP) | Amount of Cu Loaded (Ions Cu/nm2 of CNP Surface) ** | Concentration Cu in Suspension (ppm) | |
|---|---|---|---|---|---|---|
| SCNP | 181.0 ± 3.1 | 0.148 ± 0.006 | −42.4 ± 0.8 | - | - | - |
| SCNP-Cu | 185.0 ± 3.6 | 0.075 ± 0.034 | −46.0 ± 0.2 | 32.48 ± 0.42 | 7.60 ± 0.10 | 49.7 ± 0.6 |
| LCNP | 440.8 ± 13.6 | 0.036 ± 0.039 | −37.4 ± 0.7 | - | - | - |
| LCNP-Cu | 511.0 ± 8.1 | 0.086 ± 0.042 | −37.0 ± 1.4 | 1.13 ± 0.01 | 0.74 ± 0.01 | 37.9 ± 0.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scattareggia Marchese, A.; Destro, E.; Boselli, C.; Barbero, F.; Malandrino, M.; Cardeti, G.; Fenoglio, I.; Lanni, L. Inhibitory Effect against Listeria monocytogenes of Carbon Nanoparticles Loaded with Copper as Precursors of Food Active Packaging. Foods 2022, 11, 2941. https://doi.org/10.3390/foods11192941
Scattareggia Marchese A, Destro E, Boselli C, Barbero F, Malandrino M, Cardeti G, Fenoglio I, Lanni L. Inhibitory Effect against Listeria monocytogenes of Carbon Nanoparticles Loaded with Copper as Precursors of Food Active Packaging. Foods. 2022; 11(19):2941. https://doi.org/10.3390/foods11192941
Chicago/Turabian StyleScattareggia Marchese, Adriana, Elena Destro, Carlo Boselli, Francesco Barbero, Mery Malandrino, Giusy Cardeti, Ivana Fenoglio, and Luigi Lanni. 2022. "Inhibitory Effect against Listeria monocytogenes of Carbon Nanoparticles Loaded with Copper as Precursors of Food Active Packaging" Foods 11, no. 19: 2941. https://doi.org/10.3390/foods11192941
APA StyleScattareggia Marchese, A., Destro, E., Boselli, C., Barbero, F., Malandrino, M., Cardeti, G., Fenoglio, I., & Lanni, L. (2022). Inhibitory Effect against Listeria monocytogenes of Carbon Nanoparticles Loaded with Copper as Precursors of Food Active Packaging. Foods, 11(19), 2941. https://doi.org/10.3390/foods11192941






