The Glycemic Index and Human Health with an Emphasis on Potatoes
Abstract
:1. Introduction
2. GI Estimation
3. GL Estimation
4. Glycemic Index in Potatoes
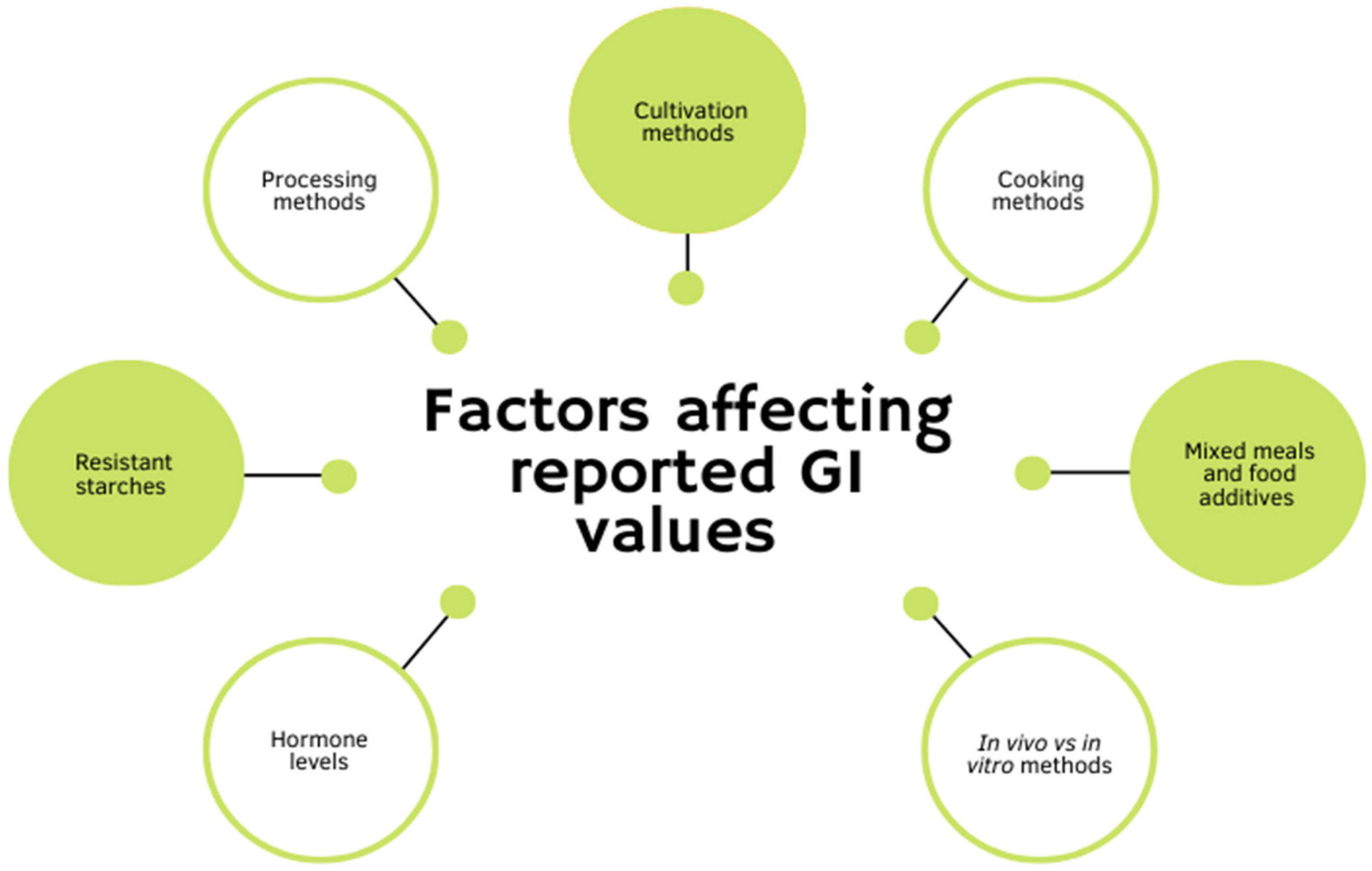
5. Factors Affecting the GI Values of Potatoes
5.1. Cooking Method
5.2. Method of Processing
5.3. Resistant Starches
5.4. Cultivation Methods
5.5. Mixed Meals and Food Additivies
5.6. Hormone Levels
5.7. In Vitro vs. In Vivo Measures
6. Potato Consumption Habits of the Next Generation and the Impact of GI on Human Health
7. Developing Low-GI Potatoes
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Higdon, J.; Drake, V.; Delage, B.; Liu, S. Glycemic Index and Glycemic Load. Available online: https://lpi.oregonstate.edu/mic/food-beverages/glycemic-index-glycemic-load (accessed on 22 April 2022).
- Augustin, L.S.A.; Kendall, C.W.C.; Jenkins, D.J.A.; Willett, W.C.; Astrup, A.; Barclay, A.W.; Björck, I.; Brand-Miller, J.C.; Brighenti, F.; Buyken, A.E.; et al. Glycemic Index, Glycemic Load and Glycemic Response: An International Scientific Consensus Summit from the International Carbohydrate Quality Consortium (ICQC). Nutr. Metab. Cardiovasc. Dis. 2015, 25, 795–815. [Google Scholar] [CrossRef] [Green Version]
- Li, Z.; Wang, D.; Ruiz-Narváez, E.A.; Peterson, K.E.; Campos, H.; Baylin, A. Starchy Vegetables and Metabolic Syndrome in Costa Rica. Nutrients 2021, 13, 1639. [Google Scholar] [CrossRef] [PubMed]
- Anderson, G.H.; Soeandy, C.D.; Smith, C.E. White Vegetables: Glycemia and Satiety. Adv. Nutr. 2013, 4, 356S–367S. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Xu, F.; Wu, Y.; Hu, H.; Dai, X. Progress of Potato Staple Food Research and Industry Development in China. J. Integr. Agric. 2017, 16, 2924–2932. [Google Scholar] [CrossRef]
- Tahvonen, R.; Hietanen, R.M.; Sihvonen, J.; Salminen, E. Influence of Different Processing Methods on the Glycemic Index of Potato (Nicola). J. Food Compos. Anal. 2006, 19, 372–378. [Google Scholar] [CrossRef]
- Lal, M.K.; Singh, B.; Sharma, S.; Singh, M.P.; Kumar, A. Glycemic Index of Starchy Crops and Factors Affecting Its Digestibility: A Review. Trends Food Sci. Technol. 2021, 111, 741–755. [Google Scholar] [CrossRef]
- Matthan, N.R.; Ausman, L.M.; Meng, H.; Tighiouart, H.; Lichtenstein, A.H. Estimating the Reliability of Glycemic Index Values and Potential Sources of Methodological and Biological Variability123. Am. J. Clin. Nutr. 2016, 104, 1004–1013. [Google Scholar] [CrossRef]
- Argyri, K.; Athanasatou, A.; Bouga, M.; Kapsokefalou, M. The Potential of an in Vitro Digestion Method for Predicting Glycemic Response of Foods and Meals. Nutrients 2016, 8, 209. [Google Scholar] [CrossRef] [Green Version]
- Magaletta, R.L.; DiCataldo, S.N.; Liu, D.; Li, H.L.; Borwankar, R.P.; Martini, M.C. In Vitro Method for Predicting Glycemic Index of Foods Using Simulated Digestion and an Artificial Neural Network. Cereal Chem. 2010, 87, 363–369. [Google Scholar] [CrossRef]
- Birt, D.F.; Boylston, T.; Hendrich, S.; Jane, J.-L.; Hollis, J.; Li, L.; McClelland, J.; Moore, S.; Phillips, G.J.; Rowling, M.; et al. Resistant Starch: Promise for Improving Human Health. Adv. Nutr. 2013, 4, 587–601. [Google Scholar] [CrossRef] [Green Version]
- Fernandes, G.; Velangi, A.; Wolever, T.M.S. Glycemic Index of Potatoes Commonly Consumed in North America. J. Am. Diet. Assoc. 2005, 105, 557–562. [Google Scholar] [CrossRef]
- Henry, C.J.K.; Lightowler, H.J.; Strik, C.M.; Storey, M. Glycaemic Index Values for Commercially Available Potatoes in Great Britain. Br. J. Nutr. 2005, 94, 917–921. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, F.S.; Foster-Powell, K.; Brand-Miller, J.C. International Tables of Glycemic Index and Glycemic Load Values: 2008. Diabetes Care 2008, 31, 2281–2283. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tian, J.; Chen, J.; Ye, X.; Chen, S. Health Benefits of the Potato Affected by Domestic Cooking: A Review. Food Chem. 2016, 202, 165–175. [Google Scholar] [CrossRef] [PubMed]
- Nayak, B.; Berrios, J.D.J.; Tang, J. Impact of Food Processing on the Glycemic Index (GI) of Potato Products. Food Res. Int. 2014, 56, 35–46. [Google Scholar] [CrossRef]
- Sajilata, M.G.; Singhal, R.S.; Kulkarni, P.R. Resistant Starch-A Review. Compr. Rev. Food Sci. Food Saf. 2006, 5, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Thed, S.T.; Phillips, R.D. Changes of Dietary Fiber and Starch Composition of Processed Potato Products during Domestic Cooking. Food Chem. 1995, 52, 301–304. [Google Scholar] [CrossRef]
- Haub, M.D.; Hubach, K.L.; Al-tamimi, E.K.; Ornelas, S.; Seib, P.A. Different Types of Resistant Starch Elicit Different Glucose Responses in Humans. J. Nutr. Metab. 2010, 2010, e230501. [Google Scholar] [CrossRef] [Green Version]
- Inan Eroglu, E.; Buyuktuncer, Z. The Effect of Various Cooking Methods on Resistant Starch Content of Foods. Nutr. Food Sci. 2017, 47, 522–533. [Google Scholar] [CrossRef]
- Jenkins, D.J.; Wolever, T.M.; Jenkins, A.L. Starchy Foods and Glycemic Index. Diabetes Care 1988, 11, 149–159. [Google Scholar] [CrossRef] [Green Version]
- Bahado-Singh, P.S.; Wheatley, A.O.; Ahmad, M.H.; Morrison, E.Y.S.A.; Asemota, H.N. Food Processing Methods Influence the Glycaemic Indices of Some Commonly Eaten West Indian Carbohydrate-Rich Foods. Br. J. Nutr. 2006, 96, 476–481. [Google Scholar] [PubMed]
- Jayanty, S.; Keller, C.; Thorne, J. Postharvest Research Report; San Luis Valley Research Center CSU: San Luis Valley, CO, USA, 2007. [Google Scholar]
- Ramaswwamyreddy, S.; Nagesh, M.A. Experimental Studies on Effect of Water and Soil Quality on Crop Yield. Aquat. Procedia 2015, 4, 1235–1242. [Google Scholar] [CrossRef]
- van Leeuwen, C.; Barbe, J.-C.; Darriet, P.; Geffroy, O.; Gomès, E.; Guillaumie, S.; Helwi, P.; Laboyrie, J.; Lytra, G.; Menn, N.L.; et al. Recent Advancements in Understanding the Terroir Effect on Aromas in Grapes and Wines. OENO One 2020, 54, 985–1006. [Google Scholar] [CrossRef]
- Bhat, R. Varying Amylose and Total Starch Content in Potato Tubers Derived from Finland and Sweden. Mater. Sci. 2015, 384. [Google Scholar]
- Lal, M.K.; Tiwari, R.K.; Kumar, R.; Naga, K.C.; Kumar, A.; Singh, B.; Raigond, P.; Dutt, S.; Chourasia, K.N.; Kumar, D.; et al. Effect of Potato Apical Leaf Curl Disease on Glycemic Index and Resistant Starch of Potato (Solanum tuberosum L.) Tubers. Food Chem. 2021, 359, 129939. [Google Scholar] [CrossRef]
- Kim, J.S.; Nam, K.; Chung, S.-J. Effect of Nutrient Composition in a Mixed Meal on the Postprandial Glycemic Response in Healthy People: A Preliminary Study. Nutr Res. Pract. 2019, 13, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Fajans, S.S.; Floyd, J.C.; Knopf, R.F.; Conn, F.W. Effect of Amino Acids and Proteins on Insulin Secretion in Man. In Schering Symposium on Endocrinology, Berlin, May 26 to 27, 1967; Elsevier: Amsterdam, The Netherlands, 1967; Volume 23, pp. 617–662. [Google Scholar] [CrossRef]
- Lal, M.K.; Tiwari, R.K.; Jaiswal, A.; Luthra, S.K.; Singh, B.; Kumar, S.; Gopalakrishnan, S.; Gaikwad, K.; Kumar, A.; Paul, V.; et al. Combinatorial Interactive Effect of Vegetable and Condiments with Potato on Starch Digestibility and Estimated in vitro Glycemic Response. Food Meas. 2022, 16, 2446–2458. [Google Scholar] [CrossRef]
- Bahrami, M.; Ataie-Jafari, A.; Hosseini, S.; Foruzanfar, M.H.; Rahmani, M.; Pajouhi, M. Effects of Natural Honey Consumption in Diabetic Patients: An 8-Week Randomized Clinical Trial. Int. J. Food Sci. Nutr. 2009, 60, 618–626. [Google Scholar] [CrossRef]
- Arcot, J.; Brand-Miller, J. A Preliminary Assessment of the Glycemic Index of Honey A Report for the Rural Industries Research and Development Corporation; Rural Industries Research and Development Corporation: Sydney, Australia, 2005; ISBN 978-1-74151-126-0. [Google Scholar]
- Ajibola, A.; Chamunorwa, J.P.; Erlwanger, K.H. Nutraceutical Values of Natural Honey and Its Contribution to Human Health and Wealth. Nutr. Metab. 2012, 9, 61. [Google Scholar] [CrossRef] [Green Version]
- Bobiş, O.; Dezmirean, D.S.; Moise, A.R. Honey and Diabetes: The Importance of Natural Simple Sugars in Diet for Preventing and Treating Different Type of Diabetes. Oxidative Med. Cell. Longev. 2018, 2018, 4757893. [Google Scholar] [CrossRef] [Green Version]
- Deibert, P.; König, D.; Kloock, B.; Groenefeld, M.; Berg, A. Glycaemic and Insulinaemic Properties of Some German Honey Varieties. Eur. J. Clin. Nutr. 2010, 64, 762–764. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Erejuwa, O.O.; Sulaiman, S.A.; Wahab, M.S.A.; Salam, S.K.N.; Salleh, M.S.M.; Gurtu, S. Comparison of Antioxidant Effects of Honey, Glibenclamide, Metformin, and Their Combinations in the Kidneys of Streptozotocin-Induced Diabetic Rats. Int. J. Mol. Sci. 2011, 12, 829–843. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Münstedt, K.; Böhme, M.; Hauenschild, A.; Hrgovic, I. Consumption of Rapeseed Honey Leads to Higher Serum Fructose Levels Compared with Analogue Glucose/Fructose Solutions. Eur. J. Clin. Nutr. 2011, 65, 77–80. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bantle, J.P. Dietary Fructose and Metabolic Syndrome and Diabetes. J. Nutr. 2009, 139, 1263S–1268S. [Google Scholar] [CrossRef] [Green Version]
- Gurr, M.I. Nutritional and Health Aspects of Sugars: Evaluation of New Findings; ILSI Europe: Brussels, Belgium, 1995. [Google Scholar]
- Foster-Powell, K.; Miller, J.B. International Tables of Glycemic Index. Am. J. Clin. Nutr. 1995, 62, 871S–890S. [Google Scholar] [CrossRef] [PubMed]
- Rajab, A.; Takruri, H.; Mishal, A.; Alkurd, R. Glycemic and Insulinemic Response of Different Types of Jordanian Honey in Healthy and Type 2 Diabetic Volunteers. Pak. J. Nutr. 2017, 16, 61–68. [Google Scholar] [CrossRef] [Green Version]
- Chepulis, L.; Francis, E. The Glycaemic Index of Manuka Honey. e-SPEN J. 2013, 8, e21–e24. [Google Scholar] [CrossRef]
- Atayoğlu, A.; Soylu, M.; Silici, S.; İnanç, N. Glycemic Index Values of Monofloral Turkish Honeys and the Effect Of their Consumption on Glucose Metabolism. Turk. J. Med. Sci. 2016, 46, 483–488. [Google Scholar] [CrossRef]
- Salvatore, T.; Nevola, R.; Pafundi, P.C.; Monaco, L.; Ricozzi, C.; Imbriani, S.; Rinaldi, L.; Sasso, F.C. Incretin Hormones: The Link between Glycemic Index and Cardiometabolic Diseases. Nutrients 2019, 11, 1878. [Google Scholar] [CrossRef] [Green Version]
- Shah, M.; Vella, A. Effects of GLP-1 on Appetite and Weight. Rev. Endocr. Metab. Disord. 2014, 15, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Radulescu, A.; Gannon, M.C.; Nuttall, F.Q. The Effect on Glucagon, Glucagon-Like Peptide-1, Total and Acyl-Ghrelin of Dietary Fats Ingested with and without Potato. J. Clin. Endocrinol. Metab. 2010, 95, 3385–3391. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brand-Miller, J.; Holt, S. Testing the Glycaemic Index of Foods: In Vivo, Not In Vitro. Eur. J. Clin. Nutr. 2004, 58, 700–701. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- We Are What We Eat: Healthy Eating Trends Around the World; Nielsen Company: New York, NY, USA, 2015.
- Wicks, M.; Wentzel-Viljoen, E. The Whole-Food Plant-Based Diet: What Does It Entail and What Lessons Can It Offer South African Dietitians? S. Afr. J. Clin. Nutr. 2021, 34, i–ii. [Google Scholar] [CrossRef]
- Hruby, A.; Hu, F.B. The Epidemiology of Obesity: A Big Picture. Pharmacoeconomics 2015, 33, 673–689. [Google Scholar] [CrossRef] [PubMed]
- Zafar, M.I.; Mills, K.E.; Zheng, J.; Regmi, A.; Hu, S.Q.; Gou, L.; Chen, L.-L. Low-Glycemic Index Diets as an Intervention for Diabetes: A Systematic Review and Meta-Analysis. Am. J. Clin. Nutr. 2019, 110, 891–902. [Google Scholar] [CrossRef] [PubMed]
- Navarre, D.A.; Brown, C.R.; Sathuvalli, V.R. Potato Vitamins, Minerals and Phytonutrients from a Plant Biology Perspective. Am. J. Potato Res. 2019, 96, 111–126. [Google Scholar] [CrossRef]
- Khlestkin, V.K.; Erst, T.V.; Rozanova, I.V.; Efimov, V.M.; Khlestkina, E.K. Genetic Loci Determining Potato Starch Yield and Granule Morphology Revealed by Genome-Wide Association Study (GWAS). PeerJ 2020, 8, e10286. [Google Scholar] [CrossRef]
- Stinard, P.; Robertson, D.; Schnable, P. Genetic Isolation, Cloning, and Analysis of a Mutator-Induced, Dominant Antimorph of the Maize Amylose Extender1 Locus. Plant Cell 1993, 5, 1555–1566. [Google Scholar] [CrossRef]
- Rivas, R.; Dratz, E.; Wagner, T.; Secor, G.; Leckband, A.; Sands, D.C. Rapid Screening Methods of Potato Cultivars for Low Glycemic Trait. bioRxiv 2021, bioRxiv:2021.08.09.455724. [Google Scholar]
| Type of Potato Product and Cooking Method | GI Value | Reference |
|---|---|---|
| ‘Maris Peer’ | 94 | [13] |
| ‘Maris Piper’ | 85 | [13] |
| ‘Desiree’ | 77 | [13] |
| ‘Estima’ | 66 | [13] |
| ‘Charlotte’ | 66 | [13] |
| ‘Marfona’ | 56 | [13] |
| ‘King Edward’ | 75 | [13] |
| ‘Nicola’ | 59 | [13] |
| Russet, baked | 111 | [1] |
| Potato, white, boiled (average) | 82 | [1] |
| California white potato, roasted | 72.3 ± 8.2 | [12] |
| Boiled red potato, hot | 89.4 ± 7.2 | [12] |
| Boiled red potato, cold | 56.2 ± 5.3 | [12] |
| French fries | 63.6 ± 5.5 | [12] |
| Chips/crisps | 56 ± 3 | [14] |
| Rice, white, boiled 1 | 66 | [1] |
| Rice, brown, boiled 1 | 50 | [1] |
| Cultivar | Potato Type | Total Starch (g/100 g) | Resistant Starch 1 (g/100 g Total Starch) | Non-Resistant Starch (g/100 g Total Starch) | |
|---|---|---|---|---|---|
| RS-Raw | RS-Baked | ||||
| Purple Majesty | Specialty | 70.46 (±0.2) | 13.43 (±1.8) | 3.24 (±1.2) | 96.86 (±12) |
| Yukon Gold | Yellows | 60.10 (±1.2) | 34.64 (±4.0) | 2.32 (±1.4) | 97.73 (±2.2) |
| Rio Grande Russet | Russets | 59.84 (±5.5) | 23.71 (±6.0) | 9.70 (±1.3) | 91.16 (±1.3) |
| Rio Colorado | Reds | 63.32 (±1.5) | 17.98 (±3.2) | 3.73 (±3.0) | 96.40 (±1.1) |
| Mountain Rose | Specialty | 62.16 (±1.8) | 12.15 (±1.4) | 6.71 (±2.9) | 93.72 (±2.3) |
| Lenape | Chip | 63.90 (±2.0) | 14.52 (±1.8) | 6.14 (±1.8) | 94.22 (±2.1) |
| CO94035-15RU | Russet | 68.49 (±2.1) | 20.27 (±4.0) | 5.33 (±1.11) | 94.94 (±1.1) |
| CO95051-7W | Chip | 71.81 (±1.1) | 32.86 (±5.4) | 5.72 (±2.3) | 94.59 (±2.2) |
| AC96052-1RU | Russet | 66.37 (±1.6) | 14.62 (±1.5) | 10.38 (±1.3) | 90.60 (±11) |
| CO97226-2R/R | Specialty | 60.48 (±2.8) | 9.81 (±1.1) | 8.77 (±2.8) | 91.93 (±0.9) |
| CO97232-1R/Y | Red | 66.00 (±6.0) | 23.78 (±3.2) | 5.49 (±3.2) | 94.80 (±1.1) |
| AC97521-1R/Y | Red | 61.02 (±3.2) | 23.76 (±1.2) | 7.07 (±1.9) | 93.39 (±1.8) |
| Honey Source | GI | Reference |
|---|---|---|
| Manuka | 54–59 | [42] |
| Citrus | 44.9 ± 15 | [43] |
| Thyme | 52.6 ± 20.1 | [43] |
| Lime | 55.3 ± 18.4 | [43] |
| Chestnut | 55.5 ± 20.2 | [43] |
| Pine | 58.8 ± 27 | [43] |
| Milk-Vetch | 69 ± 27.3 | [43] |
| Yellow Box | 31–39 | [43] |
| Stringybark | 40–48 | [32] |
| Red Gum | 43–49 | [32] |
| Iron Bark | 45–51 | [32] |
| Yapunya | 47–57 | [32] |
| S.No. | Variety | Type | % Amylose |
|---|---|---|---|
| 1 | Echo Russet | Russet | 39.9 |
| 2 | Gemstar | Russet | 35.5 |
| 3 | Russet Burbank | Russet | 42.4 |
| 4 | Classic Russet | Russet | 44.1 |
| 5 | Alturas | Russet | 36.7 |
| 6 | Clearwater Russet | Russet | 36.8 |
| 7 | Modoc | Red | 41.8 |
| 8 | Huckleberry Gold | Specialty | 41.4 |
| 9 | French Fingerling | Specialty | 37.2 |
| 10 | Waneta | Chip | 41.8 |
| 11 | Ruby Crescent | Specialty | 35.2 |
| 12 | Purple Fiesta | Specialty | 35.8 |
| 13 | Elfe | Specialty | 38.7 |
| 14 | Rainier Russet | Russet | 30.3 |
| 15 | Amarosa | Specialty | 41.6 |
| 16 | Ranger Russet | Russet | 54.2 |
| 18 | Red Rooster | Red | 46.3 |
| 19 | Shepody | Russet | 47.0 |
| 20 | Blushing Belle | Specialty | 46.8 |
| 22 | Russet Norkotah 296 | Russet | 32.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sagili, V.S.; Chakrabarti, P.; Jayanty, S.; Kardile, H.; Sathuvalli, V. The Glycemic Index and Human Health with an Emphasis on Potatoes. Foods 2022, 11, 2302. https://doi.org/10.3390/foods11152302
Sagili VS, Chakrabarti P, Jayanty S, Kardile H, Sathuvalli V. The Glycemic Index and Human Health with an Emphasis on Potatoes. Foods. 2022; 11(15):2302. https://doi.org/10.3390/foods11152302
Chicago/Turabian StyleSagili, Venkata Sai, Priyadarshini Chakrabarti, Sastry Jayanty, Hemant Kardile, and Vidyasagar Sathuvalli. 2022. "The Glycemic Index and Human Health with an Emphasis on Potatoes" Foods 11, no. 15: 2302. https://doi.org/10.3390/foods11152302
APA StyleSagili, V. S., Chakrabarti, P., Jayanty, S., Kardile, H., & Sathuvalli, V. (2022). The Glycemic Index and Human Health with an Emphasis on Potatoes. Foods, 11(15), 2302. https://doi.org/10.3390/foods11152302






