1. Introduction
Cow’s milk allergy (CMA) is a common allergy in the pediatric population with a prevalence of 2–3%, as shown in [
1]. Food allergy is a condition in which the immune system reacts unusually to specific foods and can be IgE-mediated, occurring within seconds or minutes after exposure, or non-IgE-mediated, which can take much longer to develop, sometimes up to several days [
2]. Food allergic reactions can occur in the skin, gastrointestinal tract, and respiratory tract or can even cause anaphylaxis. [
3]. IgE-mediated reactions occur after re-exposure to an allergen. At first exposure of the allergen, a T helper 2 (Th2) response results in the production of allergen-specific IgEs, which will bind to the high-affinity IgE receptors (FcεRI) on mast cells or basophils (sensitization phase). When re-exposed to the allergen, cross-linking of the surface-bound IgE with the allergen will result in degranulation of the cells and release of mediators such as histamine, leukotrienes, and inflammatory cytokines (effector phase). Important factors for an effective degranulation is the concentration of allergen-specific IgEs bound to the cells, the amount of the allergen, and the IgE affinity for the allergen [
4,
5].
Whey proteins represent about 20% of the total bovine milk proteins. The major whey allergens are beta-lactoglobulin (BLG) and alpha-lactalbumin (ALA). When whey proteins are enzymatically degraded, peptides showing biological functions can occur, and some seem to have beneficial immunomodulatory properties [
6,
7,
8,
9]. Hypoallergenic infant formulas are usually made by enzymatically degrading cow’s milk proteins, sometimes followed by further modifications such as heat treatment or ultrafiltration, and are used for the management of CMA in infants. Formula characteristics can be assessed by biochemical analysis, and reduced allergenicity can be analyzed by using in vitro immunological assays, in vivo animal allergy models, and in vivo in humans using the skin prick test (SPT), patch test, or oral challenge tests [
10].
New hypoallergenic compositions with improved treatment properties are needed, along with appropriate preclinical models, to evaluate these properties before introduction into humans. In the current study, the newly developed, extensive whey hydrolysate (eWH) was characterized on peptide level with molecular weight distribution (MWD) using gel permeation chromatography (GPC). Residual antigenicity was assessed using a BLG ELISA as well as gel electrophoresis, followed by immunoblotting with anti-BLG and anti-ALA antibodies. In vitro residual allergenicity was assessed by using RBL-2H3 cells, which express huFcεRIα and were sensitized with an oligoclonal pool of anti-BLG chuIgE antibodies. In vivo sensitizing capacity and allergenicity were investigated in a mouse model for CMA using female C3H/HeOuJ mice. After the in vitro and in vivo assessment of the eWH, a clinical study was conducted with the eWH, demonstrating that it did not evoke an allergic reaction in 90% (with 95% confidence) of infants or children with confirmed CMA [
11]. With this translational strategy from in vitro to in vivo and eventually confirmation in a clinical study, we demonstrate the clinical relevance of the preclinical results, which will improve predictability of (residual) allergenicity of the newly developed cow’s milk hydrolysates.
2. Methods
2.1. BLG Protein ELISA
Residual BLG proteins were determined by performing the BLG ELISA (ELISA Systems, Windsor, QLD, Australia) according to manufacturer instructions. This BLG ELISA has been fully validated for the use of cow’s milk hydrolysates by Merieux NutriSciences with a LOQ of 0.2 ppm. In short, one gram of hydrolysate and controls were diluted in 10 mL extraction buffer and thoroughly mixed, and pH was adjusted (pH between 6.8–7.4). The samples were warmed in a water bath of 60 °C for 15 min, with shaking for one minute every five minutes. Standards and samples were added in duplicate to the pre-coated wells and, after 10 s shaking, incubated for 15 min at room temperature (RT). Plates were washed, and conjugate was applied to the wells and, after 10 s shaking, incubated for 15 min at RT. Plates were washed, and substrate solution was added and, after shaking for 10 s, incubated for 10 min at RT. The reaction was stopped using stop solution, and absorbance was read within 30 min using a plate reader (PowerWave HT; BioTek, Santa Clara, CA, USA) at 450 nm (corrected for the absorbance at 650 nm).
2.2. Sodium Dodecylsulfate-Polyacrylamide Gel Electrophoresis (SDS-PAGE) and Immunoblotting
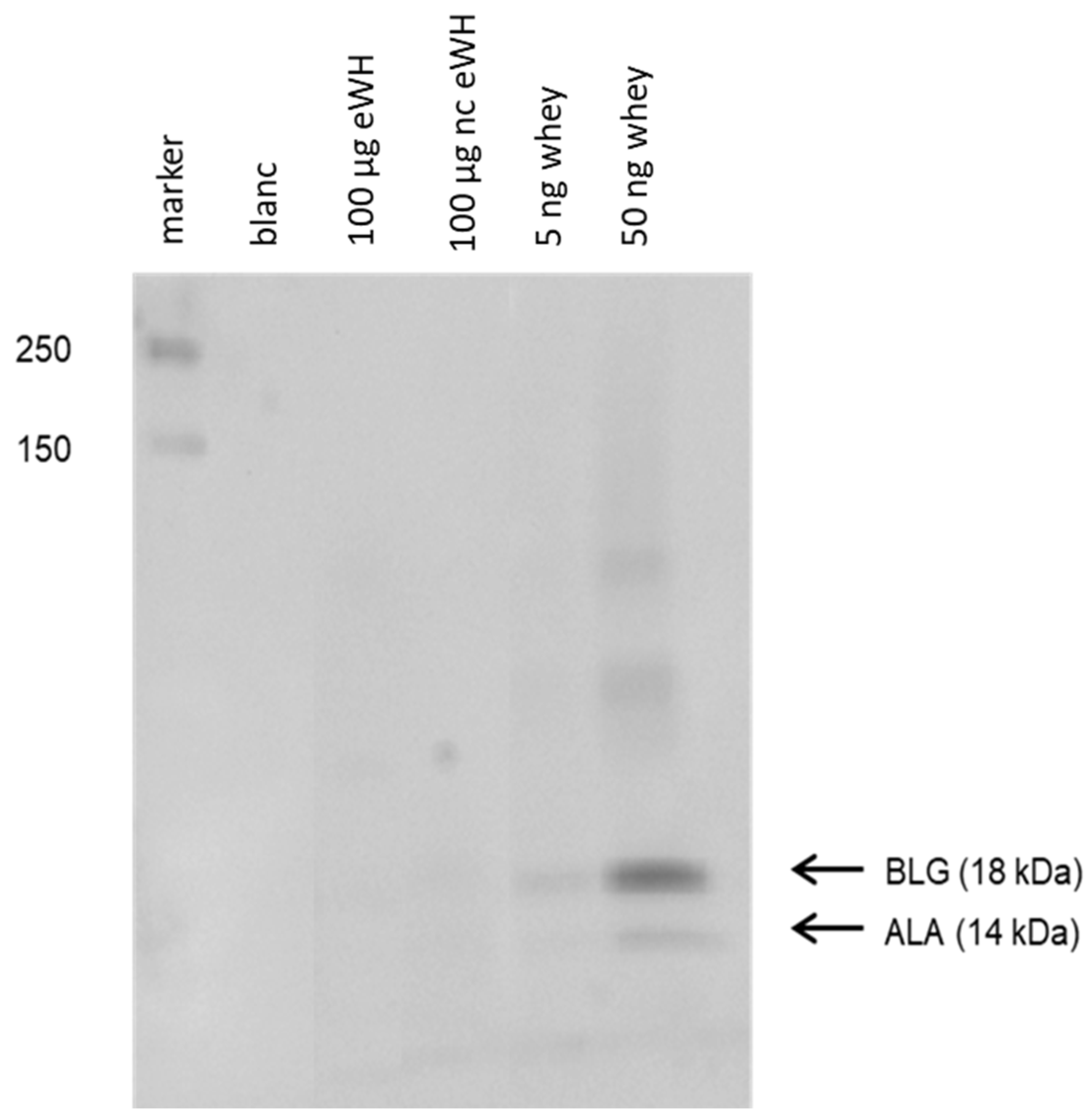
The residual proteins pattern of the hydrolysate was assessed with SDS-PAGE (Bio-Rad Mini-PROTEAN III system) using a 4–20% TGX™ gel (Bio-Rad, Hercules, CA, USA). Protein standards (Bio-Rad) were used to estimate the molecular weight of proteins of interest. The hydrolysates were diluted 1:5 with reducing sample buffer (6.05 g Tris, 8.0 g SDS, 3.2 g dithiothreitol, 20 mg bromophenol blue in 60 mL H2O, and 40 mL glycerol 87%, pH 6.8), and 100 µg of protein was added to the gel. Proteins were either stained using silver staining or blotted to a PVDF membrane (Trans Blot Turbo Mini, Bio-Rad, Hercules, CA, USA).
Immunoblotting was done by incubating the PVDF membranes for 2 h in TBST with 2% gelatin followed by incubation with an anti-ALA and an anti-BLG monoclonal antibody (1:30,000; Bethyl Laboratories, Uden, The Netherlands) in TBST with 1% gelatin for 1 h. The binding of antibody was visualized by using Lumi-light Plus Western blotting substrate (Roche Diagnostics, Rotkreuz, Switzerland), and the chemiluminescence signal was measured with the Chemidoc XRS (Bio-Rad).
2.3. MWD of Peptides by Gel Permeation Chromatography (GPC)
The MWD of the peptides after hydrolysis was characterized using GPC. Hydrolysate samples were dissolved in 5 mg/mL HPLC-eluent, and undissolved particles were removed by centrifugation (5 min at 13,000× g) if required, followed by filtration (0.45 μm cellulose acetate filter). Size exclusion chromatography was performed with a Superdex Peptide 10/300 column (GE Healthcare) at 30 °C, with a flow rate of 0.9 mL/minute using an injection of 20 μL of a 5 mg/mL HPLC-eluent (7.4 g trifluoro acetic acid and 1173 g acetonitrile in 3500 g H2O) solution. The column was calibrated with 10 peptide standards: cytochrome C (M = 12,327), aprotinin (M = 6500), adrenocorticotropic hormone from porcine pituitary gland (M = 4567), insulin A-chain oxidized ammonium salt from bovine pancreas (M = 2532), angiotensinogen 1–14 renin substrate porcine (M = 1759), bradykinin salt (M = 1060), bradykinin fragment 1–7 (M = 757), bradykinin fragment 1–5 (M = 573), Ala-Ala-Ala-Ala-Ala (M = 373), and Gly-Leu (M = 188), all of which were from Sigma Aldrich, St. Louis, MO, USA. The eluate was monitored at 200 nm.
2.4. Degranulation of RBL-huFcεRI Cells (RBL-hεIa-2B12 Cells)
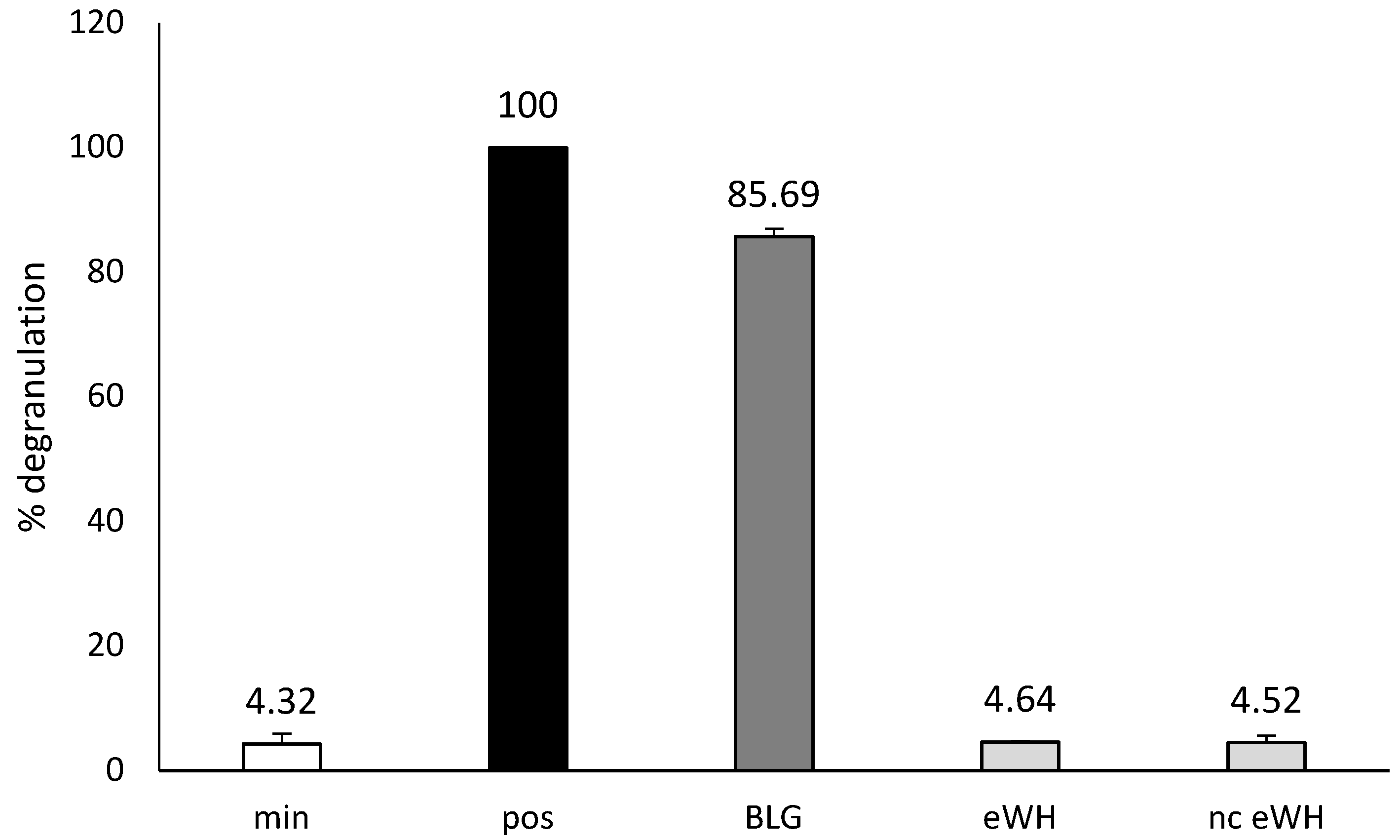
The cell-line RBL-hεIa-2B12, transfected with the α-chain of human (hu) FcεRI complex [
12,
13], was used for the RBL-huFcεRI degranulation assay. Degranulation of RBL-huFcεRI cells was performed as described previously [
12]. Confluent growing RBL-huFcεRI cells (1 × 10
5/well) in 96-well, flat-bottom culture plate were sensitized overnight with 3 μg/mL purified huIgE (Millipore) and stimulated with 10 μg/mL rabbit anti-huIgE antibodies (DakoCytomation) in Tyrode’s buffer for 1 h. huIgE/anti-huIgE served as a positive (pos) control (=100% degranulation). The sensitized cells with a pool of BLG-specific chimeric IgE monoclonal antibodies (Bioceros) were stimulated with anti-huIgE (10 µg/mL Tyrode’s buffer), BLG, or eWH (1 μg/mL in Tyrode’s buffer/HSA) for 1 h. The minimal degranulation (min) is the spontaneous degranulation of untreated cells and should always be below 20%. β-Hexosaminidase activity was measured by a fluorescence assay using 4-methylumbelliferyl-N-acetyl-α-D-glucosamine as a substrate [
14]. The β-hexosaminidase released into the medium was calculated against the positive control (100% degranulation).
2.5. Animals
Three-week-old female C3H/HeOuJ mice (Charles River Laboratories, Erkrath, Germany) where kept for a minimum of 2 generations on a cow’s-milk-protein-free diet (Research Diet Services, Wijk bij Duurstede, The Netherlands) in which casein and whey are replaced by soy proteins to avoid tolerance to cow’s milk proteins. After arrival (CKP, Wageningen University, Wageningen, The Netherlands), mice were randomly allocated to one of the 5 groups (
n = 8 per group): a negative control group (sensitized with PBS), a positive control group, an allergenicity assessment group with the newly developed eWH (both sensitized with whey), a sensitizing capacity group with the newly developed eWH, and a sensitizing capacity group with the control eHW. Mice were group housed (4/cage) in Makrolon type III cages per treatment, with 9 kGy irradiated sawdust bedding (Lignocel 9 s, J. Rettenmaier & Söhne GmbH, Rosenberg, Germany) and a mouse igloo with activation fast-trak (UNO) as environmental enrichment. Cow’s milk protein-free AIN93G [
15] chow and tap water were available ad libitum. Mice were kept on light/dark cycle of 12 h/12 h, temperature 19–21 °C, and 42–47% relative humidity, and all procedures were performed during the light cycle. The animal study was registered under the code DAN202, and all experimental procedures were approved by an external, independent Animal Experimental Committee (DEC consult, Soest, The Netherlands) on 30 September 2011 and complied with national legislation and the principles of good laboratory animal care following the EU directive for the protection of animals used for scientific purposes.
2.6. Oral Sensitization
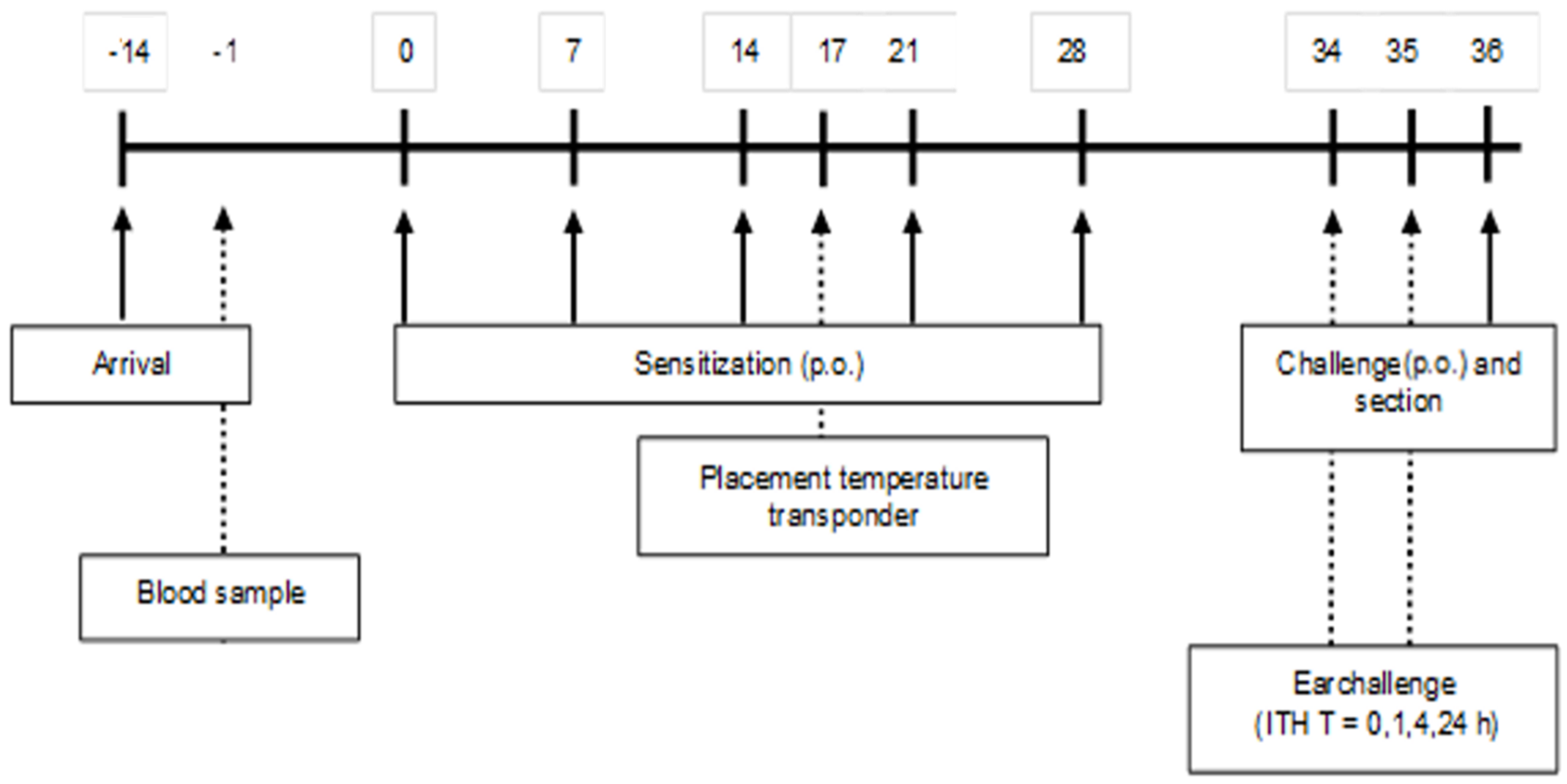
A schematic overview of the sensitization and challenge protocol is depicted in
Figure 1. After two weeks of acclimatization, (5-week-old) mice were sensitized weekly (first sensitization is day 0) for a period of 5 weeks with 0.5 mL of a homogenized mixture of 33.2% WPC80 and 66.8% demineralized WPC, the extensive whey hydrolysate (eWH), or a previous used negative control extensive whey hydrolysate (nc eWH) [
16] (40 mg/mL PBS) with cholera toxin (CT; 20 μg/mL PBS) as an adjuvant, per oral (p.o.) gavage (Gavage Feeding Needle (mouse 20–25 g), 20 G × 30 mm straight, 1.25 mm barrel tip (Fine Science Tools, Heidelberg, Germany). Control mice received CT and PBS. One week after the last sensitization, mice were sacrificed after oral challenge (100 mg/mL PBS) by cervical dislocation under isoflurane/O2 mixture anesthesia. Blood samples were collected by retro-orbital bleeding and centrifuged (15 min at 13,500 rpm) on the day prior to the first sensitization and on day 36. Sera were stored at −20 °C for measurement of whey-specific immunoglobulins, mouse mast cell protease-1 (mMCP-1), and cytokines.
2.7. Acute Allergen-Specific Skin Response
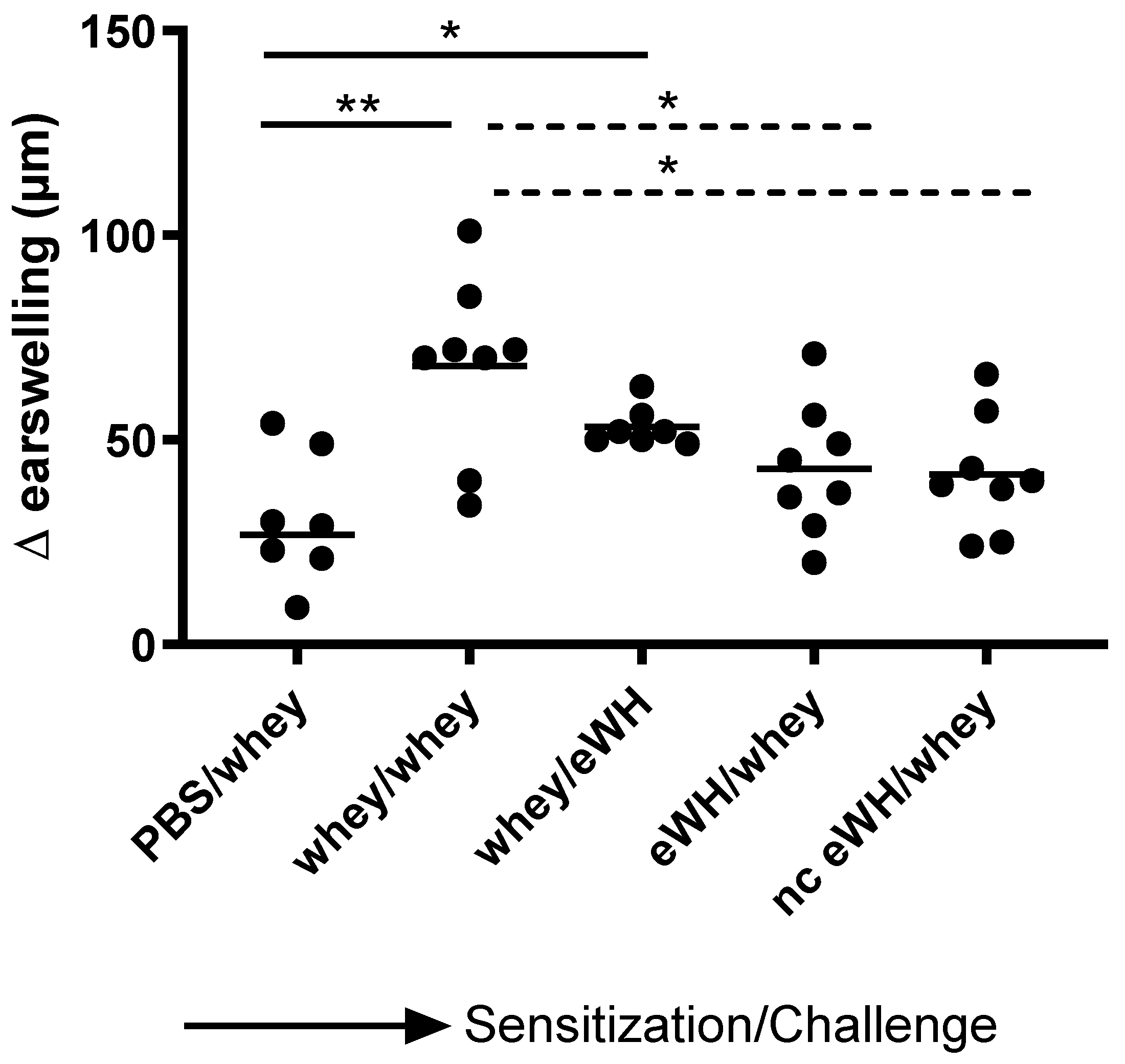
The acute allergen-specific skin response was measured after injection (30 G (yellow) needle (Terumo, Leuven, Belgium) for i.d. administration) of a mixture of 33.2% WPC80 and 66.8% demineralized whey protein concentrate or the eWH in the ear pinnae intradermally (i.d.). At day 36, mice sensitized orally with whey mixture, eWH, or nc eWH were injected i.d. in both ears with 20 μL whey mixture (1 mg/mL in PBS). On day 36, mice sensitized orally with WPC80 and demineralized whey protein concentrate mixture were injected i.d. in both ears with 20 μL eWH (1 mg/mL in PBS). Ear thickness was measured in duplicate using a digital micrometer (Mitutoyo, Veenendaal, The Netherlands). The allergen-specific net ear swelling was calculated by subtracting the basal thickness (0 h) from the thickness measured after 1, 4, and 24 h. The ear swelling is expressed as delta μm.
2.8. Shock Score
Degranulation of mast cells may cause an anaphylactic shock in severe allergic animals. To assess this anaphylactic shock severity, the animals were monitored closely, using a validated anaphylactic scoring (0, no symptoms; 1, scratching around nose and mouth; 2, swelling around the eyes and mouth, piloerection, reduced activity, higher breathing rate; 3, shortness of breath, blue rash around the mouth and tail, higher breathing rate; 4, no activity after stimulation, shivers, and muscle contractions [
17]). Mice were placed on heating mats when a shock score of 3 was reached with a decrease in body temperature. The humane endpoint was set at an anaphylaxis score 4. Body temperature was measured with a temperature transponder (Plexx), which was placed on day 17 (
Figure 1). The transponders were placed subcutaneously into the mice under isoflurane/O2 mixture anesthesia and were used to monitor the body temperature non-invasively at the timepoints t = 0, 15, 30, and 60 min.
2.9. Serum Mouse Mast Cell Protease-1 (mMCP-1)
Concentrations of serum mMCP-1 were analyzed with a mMCP-1 ELISA (mMCP-1 ELISA Ready-SET-Go!; eBiosciences, San Diego, CA, USA) according to manufacturers’ instructions.
2.10. Serum Whey-Specific Immunoglobulins
Levels of serum whey-specific IgE were analyzed by an in-house ELISA. Polystyrene high binding plates (Corning, Corning, NY, USA) were coated with 100 μL/well WPC80 (20 μg/mL) in coating buffer for 18 h at 4 °C. Plates were washed and blocked for 2 h with 0.5% HSA (Sigma Aldrich St. Louis, MO, USA). After washing, serum samples were applied in a 25× and a 75× dilution and incubated for 2 h at RT. Plates were washed and incubated with biotin-labelled rat anti-mouse IgE (BD Pharmingen, San Diego, CA, USA) at RT. Plates were washed and incubated with streptavidin-horseradish peroxidase 1:20,000 dilution (Sanquin, Amsterdam, The Netherlands) for 1 h. Plates were washed and developed with 1-Step Ultra TMB ELISA (Fisher Scientific, Waltham, MA, USA). The reaction was stopped with 4 M H2SO4 (Merck, Readington Township, NY, USA), and absorbance was measured at 450 nm on a plate reader (PowerWave HT; BioTek, Santa Clara, CA, USA).
2.11. Serum Th1/Th2 Cytokines
Serum cytokine levels of IL-2, IL-4, IL-5, IL-10 IL-12, IFN-γ, TNF-α, and GM-CSF were measured using a commercial Mouse cytokine Th1/Th2 magnetic bead immunoassay (BioRad, Hercules, CA, USA) on the Luminex Workstation Bio-Plex 200 System (BioRad, Hercules, CA, USA). The measurement was preformed according to the manufacturer’s instructions.
2.12. Statistical Analysis
The sample size calculation was based on the ear thickness as main parameter, using historical data, with a minimal effect size d = 0.25 and equal variation (0.17) for each experimental group. The mouse model data were analyzed using one-way ANOVA, followed by a Bonferroni’s multiple comparison post hoc test for selected comparisons. Data are represented as mean ± SD or as individual value plots. Statistical analyses were conducted using GraphPad Prism software version 8.0.0. Values of p < 0.05 were considered significant.
4. Discussion
The European Union introduced a regulation in 2007 on nutrition and health claims on foods to assure these claims are clear, accurate, and based on scientific evidence. For hydrolyzed formulas, it is key to affirm the absence of potential allergic epitopes. To assess the allergenic and sensitizing capacity of extensive whey hydrolysates, both in vitro methods and an in vivo mouse model of CMA were used in the current study.
After hydrolysis, the MWD of the peptides after hydrolysis showed that there were only peptides present <5000 Da, and SDS-PAGE showed no protein bands. There was no in vitro antigenicity (capacity to bind to antibodies) found by immunoblotting with anti-BLG/anti-ALA antibodies and in the BLG ELISA, indicating very low immunogenic properties of the eWH. This was further substantiated by the absence of in vitro allergenicity of the eWH in the RBL-huFcεRI degranulation assay, which has proven human relevance since the pool of chimeric antibodies cover the allergenic epitopes as assessed with epitope mapping with IgE of CMA infants and known from literature [
12]. It should be noted that the other allergen in whey, ALA, is only assessed with the immunoblotting when using anti-ALA antibodies, while both the ELISA and RBL-huFcεRI are specifically investigating BLG. In a previous study, however, we established that during the hydrolysis, BLG and ALA are broken down at the same rate [
18]
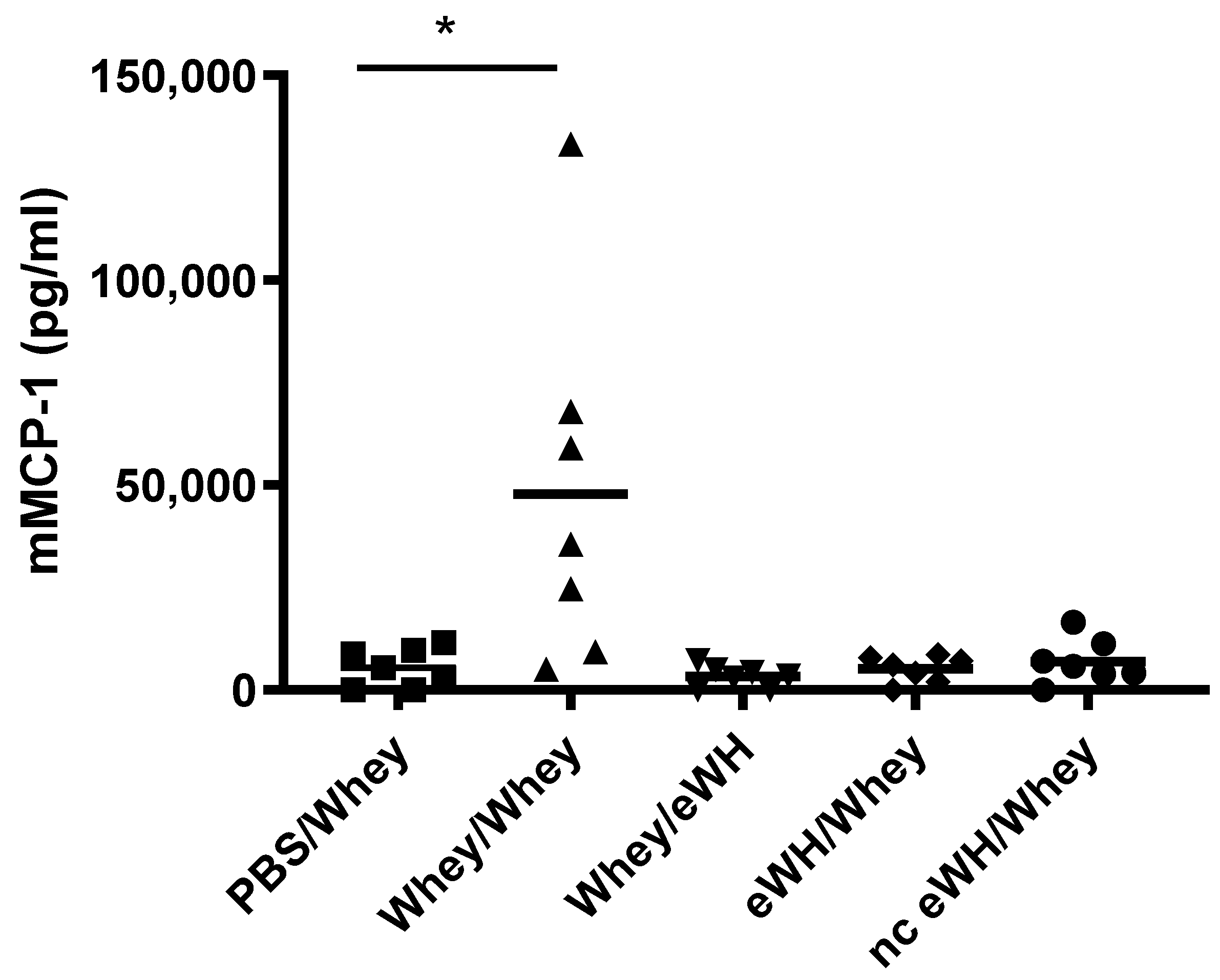
For the assessment of the hypoallergenic potential of the eWH in the CMA mouse model, mice were sensitized with whey and challenged with eWH. The positive control (i.e., both sensitization and challenge with whey) showed high levels of whey-specific IgE exemplified by a presence of allergic symptoms, acute allergic skin reactions, anaphylactic shock, decrease in body temperature, and elevated mMCP-1 concentrations. Mice sensitized with whey and challenged with eWH showed elevated levels of whey-specific IgE as expected. A reduction (non-significant) in ear swelling was observed compared to the positive control, but the ear swelling was significantly higher than found in non-sensitized mice, indicating a residual immune reaction to the eWH but not contributing to the allergic reaction since there were no anaphylactic symptoms and no decrease in body temperature or mMCP-1 observed with the eWH. Based on the overall assessment, the eWH is considered non-allergenic and thus safe for humans, including allergic individuals.
To assess the sensitizing potential of whey hydrolysates, mice were sensitized with either eWH or nc eWH and challenged with whey. Mice sensitized with either eWH or nc eWH showed no acute allergic skin reactions after challenge with whey, indicating that both hydrolysates were not able to sensitize to whey protein allergy. This was confirmed by the absence of whey-specific IgE and anaphylactic symptoms and decrease in body temperature and mMCP-1 after challenge with whey.
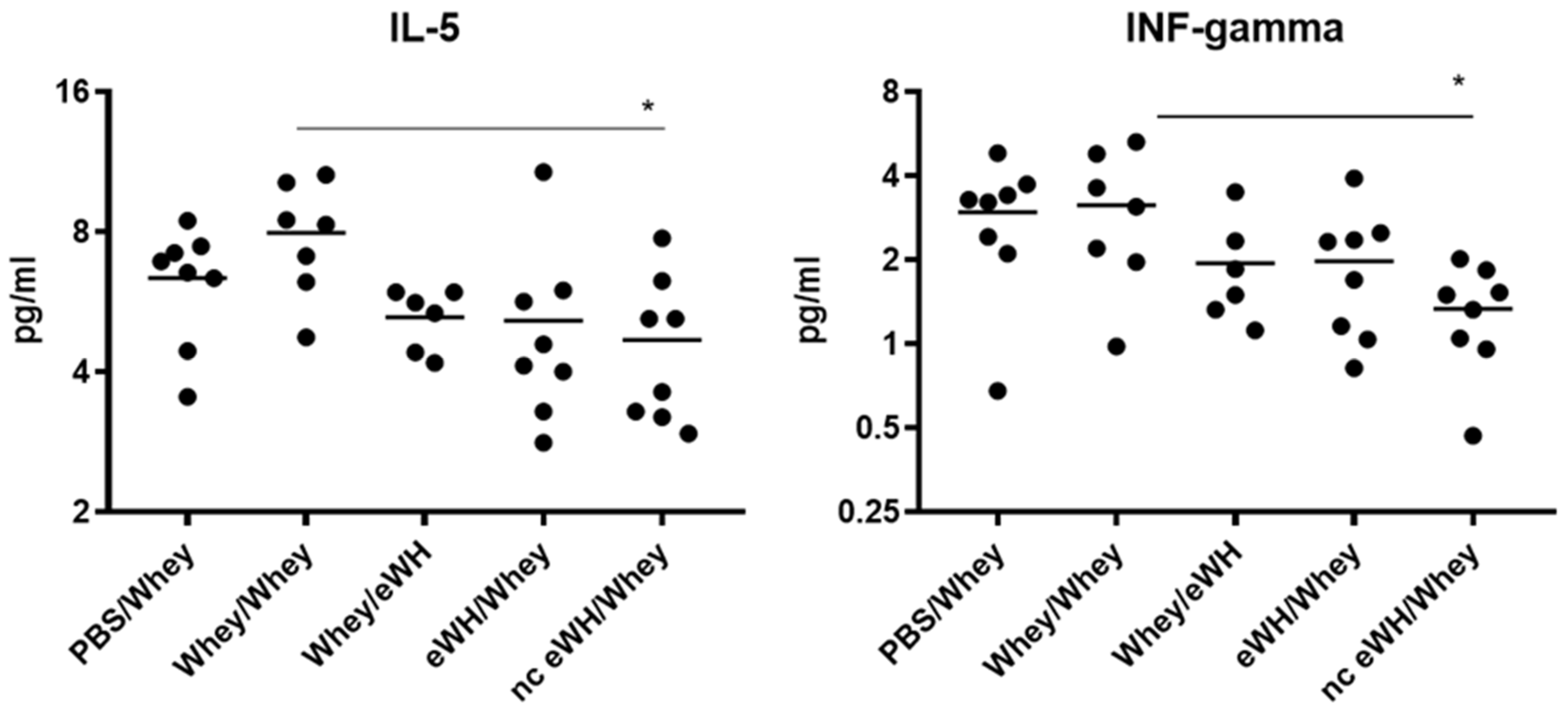
Allergic reactions are characterized by the activation of allergen-specific CD4+ T cells, which produces a specific set of cytokines, namely IL-4, IL-5, and IL-13. These cytokines trigger IgE class switching and the recruitment and activation of eosinophils and mast cells. IL-5 acts as an eosinophil stimulating factor [
19]. Only the nc eWH showed a significant decrease of IL-5, which is in line with the absence of an allergic response, whereas the eWH shows lower concentrations of IL-5, which were, however, not significant. Th1-type cytokines (IL-2, IL-10, IL-12, GM-CSF, IFN-gamma, TNF-alpha) initiate cell-mediated immune responses, which are necessary for the defense against viruses and bacteria. IFN-gamma is a cytokine that is critical for regulation of immune responses against viruses and bacteria and bridges the innate and specific immune response pathways, but it also inhibits Th2 cell differentiation. Decreased levels of IL-12 and IFN-g have been found in asthma, and this might diminish their ability to inhibit IgE synthesis [
20]. Even though the allergic response in the nc eWH was absent, together with decreased IL-5, the concentration of IFN-gamma is also significantly lower, which is an unexpected finding. The significance of these cytokines should be further explored in a time-course manner in the CMA mouse model.
Overall, using our in vitro and in vivo translational research approach for the assessment of sensitization capacity and residual allergenicity, our newly developed eWH can be considered safe for the use in CMA infants. In a subsequent clinical study that was recently published [
11], this was demonstrated as well: the eWH does not evoke an allergic reaction in 90% (with 95% confidence) of infants with confirmed CMA and can thus be regarded as safe.