Why Being ‘Stressed’ Is ‘Desserts’ in Reverse—The Effect of Acute Psychosocial Stress on Food Pleasure and Food Choice
Abstract
:1. Introduction
1.1. Physiological Mechanisms of the Endocrine System Affected by Stress
1.2. Stress-Induced Eating
1.3. Food Reward and Its Subcomponents
1.4. Purpose of the Current Study
- Investigate how explicit food liking, explicit food wanting, implicit food wanting, and relative food preference are affected by temporary acute stress vs. relaxation.
- Explore if actual snack food choice is affected by temporary acute stress vs. relaxation.
2. Materials and Methods
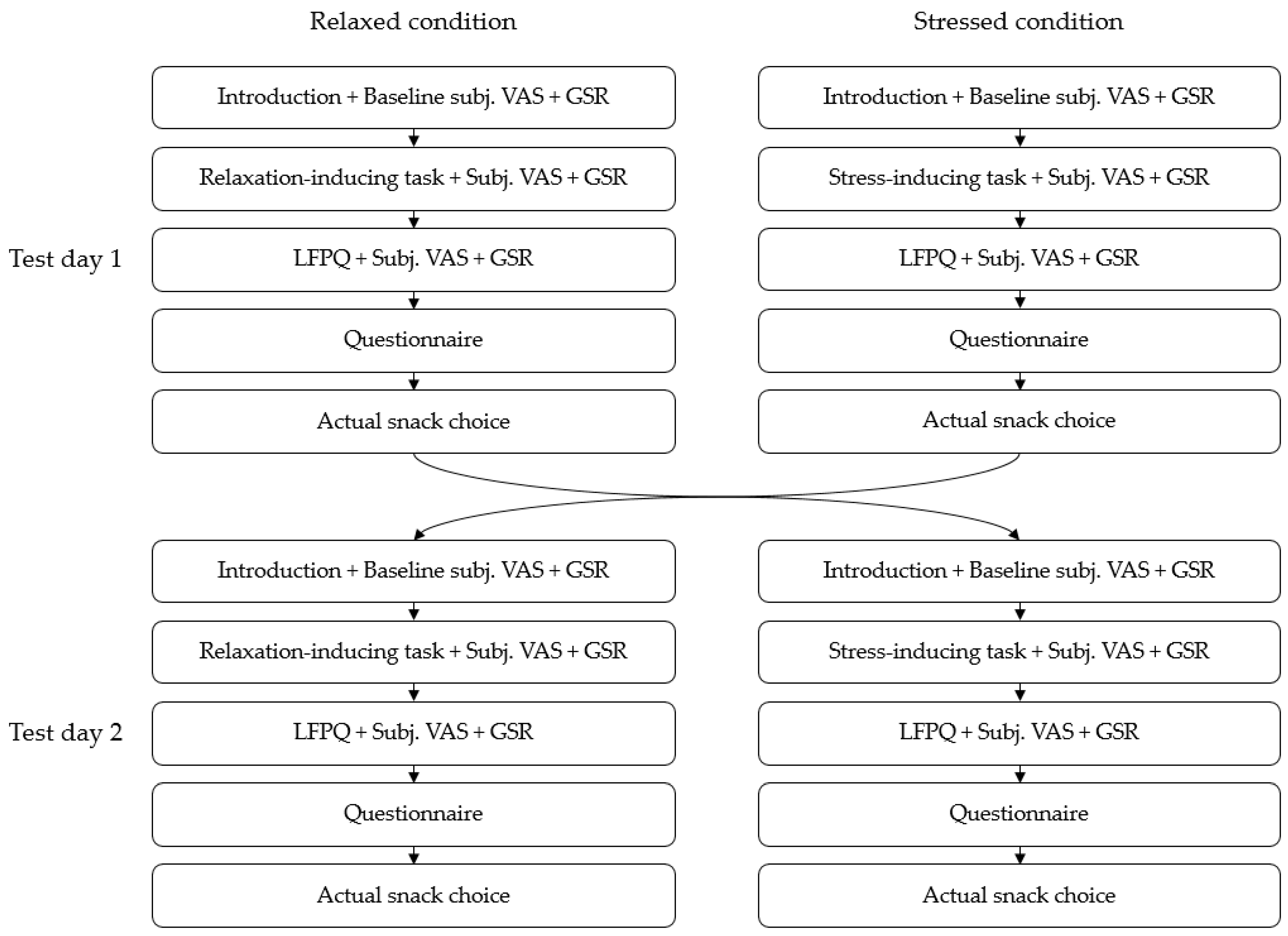
2.1. Study Design
2.2. Participants, Recruitment and Prescreening
Power Calculations
2.3. Study Procedure
2.4. Measure of Food Reward and Eating Behavior
2.4.1. Food Images in the LFPQ
2.4.2. Single Foods—Explicit Liking and Wanting in the LFPQ
2.4.3. Paired Foods—Food Choice and Implicit Wanting in the LFPQ
2.4.4. Actual Snack Choice
2.5. Cognitive Tasks
2.6. Subjective Sensations and Self-Report Questionnaires
2.7. Physiological Arousal
2.8. Data Analyses
3. Results
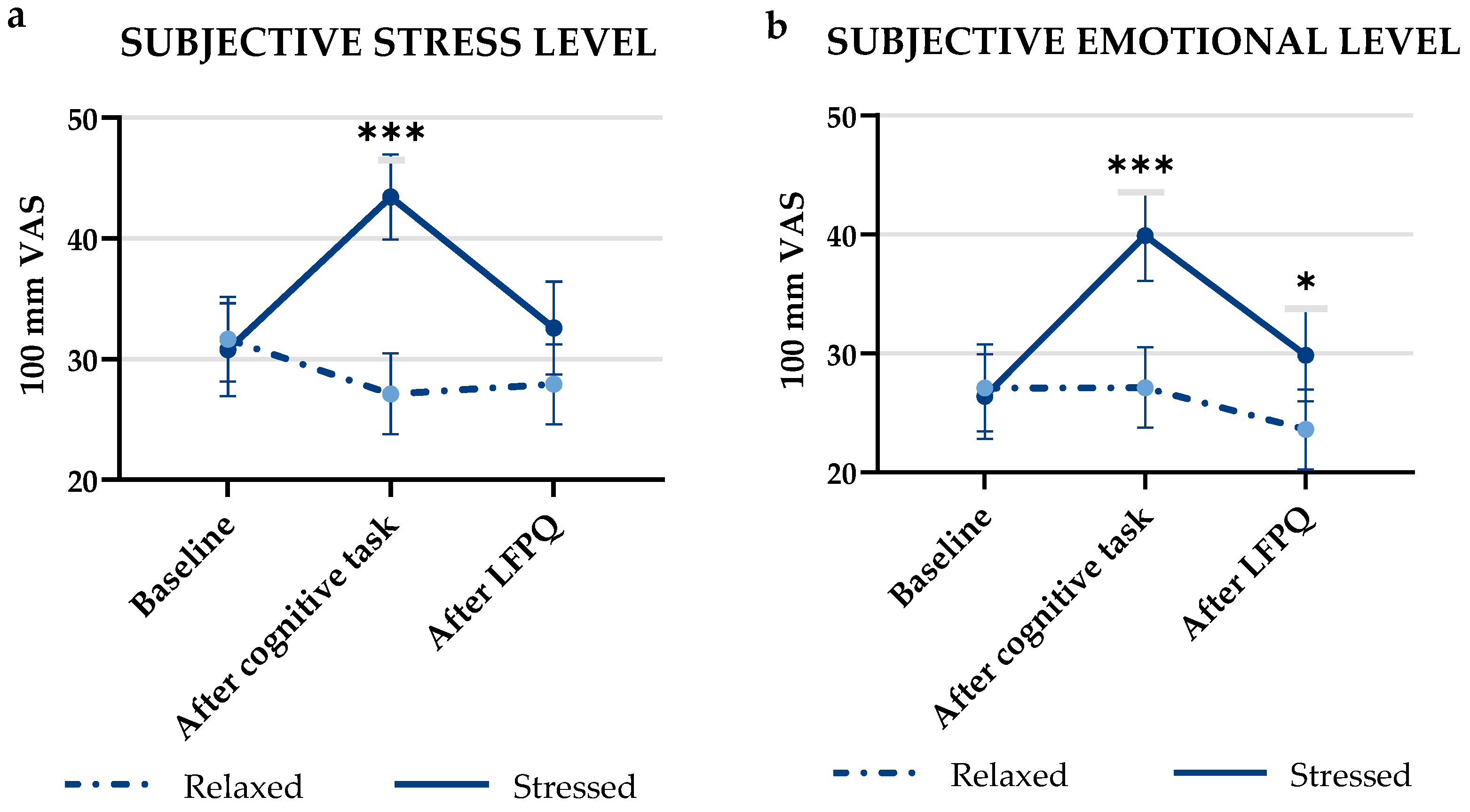
3.1. Manipulation Check on Stress and Emotional Level
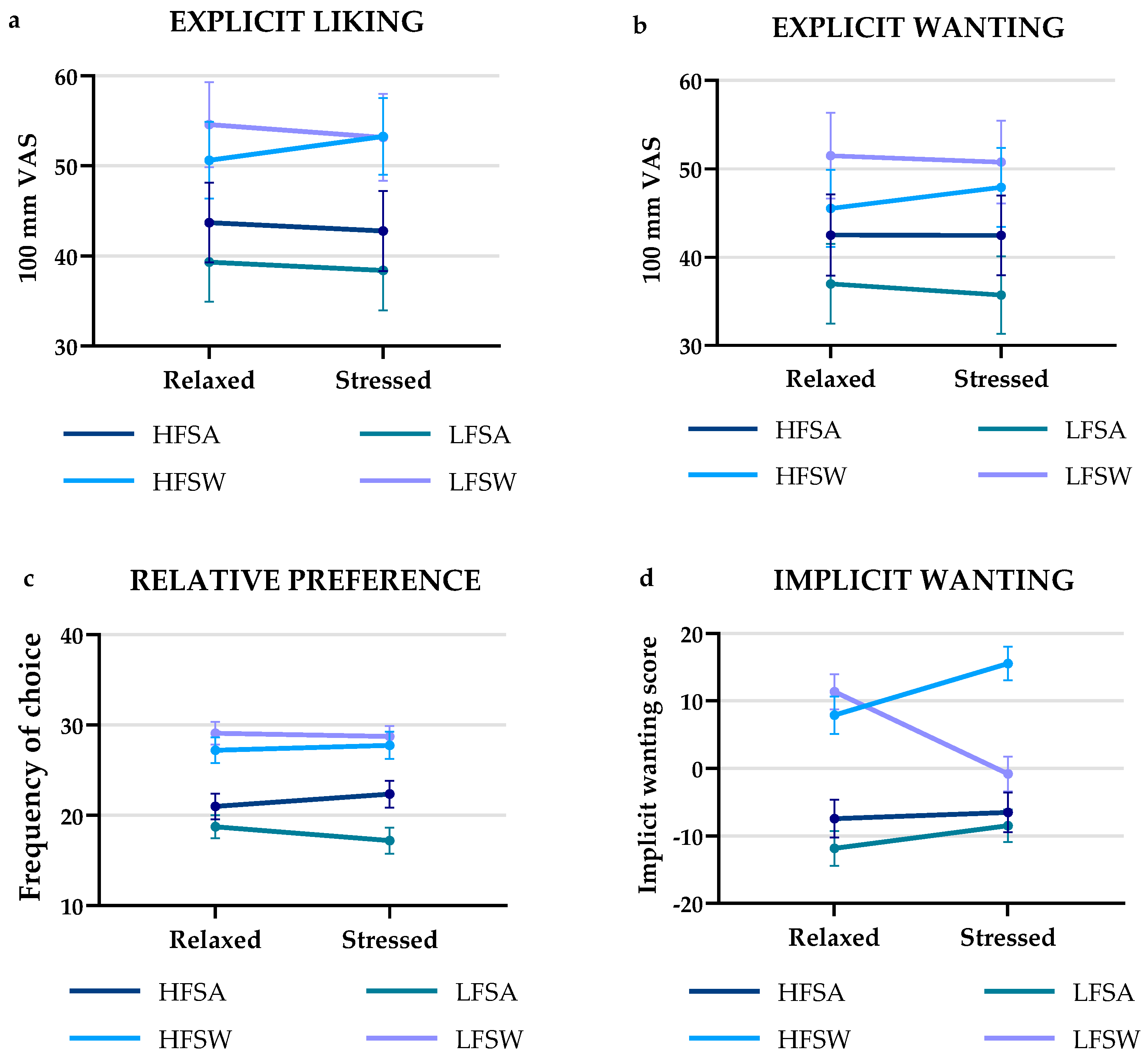
3.2. Eating Behaviour and Food Choice by Different Reward Measures
3.2.1. Explicit Liking and Wanting
3.2.2. Relative Preference
3.2.3. Implicit Wanting
3.2.4. Actual Snack Choice
4. Discussion
4.1. ‘Stressed’ Is ‘Desserts’ in Reverse for Some
4.2. Implications
4.3. Strengths and Limitations of the Study
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rosa, H. Fremmedgørelse og Acceleration, 1st ed.; Heinskou, M.B., Ed.; Hans Reitzel: Copenhagen, Denmark, 2014; ISBN 9788741257716. [Google Scholar]
- Brinkmann, S. Stress i arbejdslivet: Konstitueringen af en epidemi. In Nye Perspektiver på Stress; Andersen, M.F., Brinkmann, S., Eds.; Klim: Aarhus, Denmark, 2013; pp. 75–92. ISBN 9788771291674. [Google Scholar]
- Becker, D. One Nation under Stress: Social Uses of the Stress Concept; Oxford University Press: New York, NY, USA, 2013; ISBN 9780199742912. [Google Scholar]
- Kompier, M.A.J. New systems of work organization and workers’ health. Scand. J. Work Environ. Health 2006, 32, 421–430. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Epel, E.S.; Crosswell, A.D.; Mayer, S.E.; Prather, A.A.; Slavich, G.M.; Puterman, E.; Mendes, W.B. More than a feeling: A unified view of stress measurement for population science. Front. Neuroendocrinol. 2018, 49, 146–169. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.; Doyle, W.J.; Alper, C.M.; Janicki-Deverts, D.; Turner, R.B. Sleep habits and susceptibility to the common cold. Arch. Intern. Med. 2009, 169, 62–67. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cohen, S.; Gianaros, P.J.; Manuck, S.B. A stage model of stress and disease. Perspect. Psychol. Sci. 2016, 11, 456–463. [Google Scholar] [CrossRef]
- McEwen, B.S. Protective and damaging effects of stress mediators. N. Engl. J. Med. 1998, 338, 171–179. [Google Scholar] [CrossRef] [Green Version]
- Lupien, S.J.; McEwen, B.S.; Gunnar, M.R.; Heim, C. Effects of stress throughout the lifespan on the brain, behaviour and cognition. Nat. Rev. Neurosci. 2009, 10, 434–445. [Google Scholar] [CrossRef]
- Finch, L.E.; Cummings, J.R.; Tomiyama, A.J. Cookie or clementine? Psychophysiological stress reactivity and recovery after eating healthy and unhealthy comfort foods. Psychoneuroendocrinology 2019, 107, 26–36. [Google Scholar] [CrossRef]
- Dallman, M.F.; Pecoraro, N.; Akana, S.F.; La Fleur, S.E.; Gomez, F.; Houshyar, H.; Bell, M.E.; Bhatnagar, S.; Laugero, K.D.; Manalo, S. Chronic Stress and Obesity: A new view of “comfort food”. Proc. Natl. Acad. Sci. USA 2003, 100, 11696–11701. [Google Scholar] [CrossRef] [Green Version]
- Masih, T.; Dimmock, J.A.; Epel, E.S.; Guelfi, K.J. Stress-induced eating and the relaxation response as a potential antidote: A review and hypothesis. Appetite 2017, 118, 136–143. [Google Scholar] [CrossRef]
- Finch, L.E.; Tomiyama, A.J. Comfort eating, psychological stress, and depressive symptoms in young adult women. Appetite 2015, 95, 239–244. [Google Scholar] [CrossRef] [Green Version]
- Tomiyama, A.J. Stress and obesity. Annu. Rev. Psychol. 2019, 70, 703–718. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Herhaus, B.; Päßler, S.; Petrowski, K. Stress-related laboratory eating behavior in adults with obesity and healthy weight. Physiol. Behav. 2018, 196, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Sinha, R.; Jastreboff, A.M. Stress as a common risk factor for obesity and addiction. Biol. Psychiatry 2013, 73, 827–835. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Picard, M.; McEwen, B.S.; Epel, E.S.; Sandi, C. An energetic view of stress: Focus on mitochondria. Front. Neuroendocrinol. 2018, 49, 72–85. [Google Scholar] [CrossRef]
- Tomiyama, A.J.; Finch, L.E.; Cummings, J.R. Did that brownie do its job? Stress, eating, and the biobehavioral effects of comfort food. Emerg. Trends Soc. Behav. Sci. 2015, 1, 1–15. [Google Scholar] [CrossRef]
- Finch, L.E.; Tomiyama, A.J. Stress-induced eating dampens physiological and behavioral stress responses. In Nutrition in the Prevention and Treatment of Abdominal Obesity; Elsevier: Amsterdam, The Netherlands, 2014; pp. 189–195. ISBN 9780124078697. [Google Scholar]
- Adam, T.C.; Epel, E.S. Stress, eating and the reward system. Physiol. Behav. 2007, 91, 449–458. [Google Scholar] [CrossRef]
- Cohen, S.; Murphy, M.L.M.; Prather, A.A. Ten surprising facts about stressful life events and disease risk. Annu. Rev. Psychol. 2019, 70, 577–597. [Google Scholar] [CrossRef]
- Gunnar, M.R. Social buffering of stress in development: A career perspective. Perspect. Psychol. Sci. 2017, 12, 355–373. [Google Scholar] [CrossRef]
- Dickerson, S.S.; Gruenewald, T.L.; Kemeny, M.E. When the social self is threatened: Shame, physiology, and health. J. Pers. 2004, 72, 1191–1216. [Google Scholar] [CrossRef]
- Wallis, D.J.; Hetherington, M.M. Emotions and eating. Self-reported and experimentally induced changes in food intake under stress. Appetite 2009, 52, 355–362. [Google Scholar] [CrossRef]
- Finlayson, G.; King, N.; Blundell, J.E. Liking vs. wanting food: Importance for human appetite control and weight regulation. Neurosci. Biobehav. Rev. 2007, 31, 987–1002. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berridge, K.C.; Kringelbach, M.L. Pleasure systems in the brain. Neuron 2015, 86, 646–664. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berridge, K.C. Food reward: Brain substrates of wanting and liking. Neurosci. Biobehav. Rev. 1996, 20, 1–25. [Google Scholar] [CrossRef]
- Berridge, K.C.; Robinson, T.E. What is the role of dopamine in reward: Hedonic impact, reward learning, or incentive salience? Brain Res. Rev. 1998, 28, 309–369. [Google Scholar] [CrossRef]
- Pool, E.; Sennwald, V.; Delplanque, S.; Brosch, T.; Sander, D. Measuring wanting and liking from animals to humans: A systematic review. Neurosci. Biobehav. Rev. 2016, 63, 124–142. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pool, E.; Delplanque, S.; Coppin, G.; Sander, D. Is comfort food really comforting? Mechanisms underlying stress-induced eating. Food Res. Int. 2015, 76, 207–215. [Google Scholar] [CrossRef] [Green Version]
- Dalton, M.; Finlayson, G. Hedonics, satiation and satiety. In Satiation, Satiety and the Control of Food Intake; Elsevier: Amsterdam, The Netherlands, 2013; pp. 221–237. [Google Scholar]
- Kringelbach, M.L.; Stein, A.; van Hartevelt, T.J. The functional human neuroanatomy of food pleasure cycles. Physiol. Behav. 2012, 106, 307–316. [Google Scholar] [CrossRef]
- Finlayson, G.; King, N.; Blundell, J. The role of implicit wanting in relation to explicit liking and wanting for food: Implications for appetite control. Appetite 2008, 50, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Havermans, R.C. “You say it’s liking, i say it’s wanting …”. On the difficulty of disentangling food reward in man. Appetite 2011, 57, 286–294. [Google Scholar] [CrossRef]
- Berridge, K.C.; Ho, C.-Y.; Richard, J.M.; DiFeliceantonio, A.G. The tempted brain eats: Pleasure and Desire circuits in obesity and eating disorders. Brain Res. 2010, 1350, 43–64. [Google Scholar] [CrossRef] [Green Version]
- Berridge, K.C. ‘Liking’ and ‘wanting’ food rewards: Brain substrates and roles in eating disorders. Physiol. Behav. 2009, 97, 537–550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morris, M.J.; Beilharz, J.E.; Maniam, J.; Reichelt, A.C.; Westbrook, R.F. Why is obesity such a problem in the 21st century? The intersection of palatable food, cues and reward pathways, stress, and cognition. Neurosci. Biobehav. Rev. 2015, 58, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Thomsen, K.R.; Whybrow, P.C.; Kringelbach, M.L. Reconceptualizing anhedonia: Novel perspectives on balancing the pleasure networks in the human brain. Front. Behav. Neurosci. 2015, 9, 49. [Google Scholar] [CrossRef] [Green Version]
- Brewis, A.A. Stigma and the perpetuation of obesity. Soc. Sci. Med. 2014, 118, 152–158. [Google Scholar] [CrossRef]
- Oustric, P.; Thivel, D.; Dalton, M.; Beaulieu, K.; Gibbons, C.; Hopkins, M.; Blundell, J.; Finlayson, G. Measuring food preference and reward: Application and cross-cultural adaptation of the leeds food preference questionnaire in human experimental research. Food Qual. Prefer. 2020, 80, 103824. [Google Scholar] [CrossRef]
- The World Medical Association. WMA Declaration of Helsinki—Ethical Principles for Medical Research Involving Human Subjects. Available online: https://www.wma.net/policies-post/wma-declaration-of-helsinki-ethical-principles-for-medical-research-involving-human-subjects/ (accessed on 30 March 2022).
- Cohen, S.; Kamarck, T.; Mermelstein, R. A global measure of perceived stress. J. Health Soc. Behav. 1983, 24, 385–396. [Google Scholar] [CrossRef]
- Eskildsen, A.; Dalgaard, V.L.; Nielsen, K.J.; Andersen, J.H.; Zachariae, R.; Olsen, L.R.; Jørgensen, A.; Christiansen, D.H. Cross-cultural adaptation and validation of the Danish consensus version of the 10-item perceived stress scale. Scand. J. Work Environ. Health 2015, 41, 486–490. [Google Scholar] [CrossRef]
- Cauldron Science Gorilla Experiment Builder. Available online: www.gorilla.sc (accessed on 1 March 2022).
- Compusense Inc. Compusense20. Available online: www.compusense.com (accessed on 20 April 2022).
- Griffioen-Roose, S.; Finlayson, G.; Mars, M.; Blundell, J.E.; de Graaf, C. Measuring food reward and the transfer effect of sensory specific satiety. Appetite 2010, 55, 648–655. [Google Scholar] [CrossRef]
- Pedersen, H.; Beaulieu, K.; Finlayson, G.; Færch, K.; Jørgensen, M.E.; Lewis, J.I.; Lind, M.V.; Lauritzen, L.; Quist, J.S. Food reward after a traditional Inuit or a westernised diet in an Inuit population in Greenland. Nutrients 2022, 14, 561. [Google Scholar] [CrossRef]
- Pedersen, H.; Quist, J.S.; Jensen, M.M.; Clemmensen, K.K.B.; Vistisen, D.; Jørgensen, M.E.; Færch, K.; Finlayson, G. Investigation of eye tracking, electrodermal activity and facial expressions as biometric signatures of food reward and intake in normal weight adults. Food Qual. Prefer. 2021, 93, 104248. [Google Scholar] [CrossRef]
- Martins, C.; Aschehoug, I.; Ludviksen, M.; Holst, J.; Finlayson, G.; Wisloff, U.; Morgan, L.; King, N.; Bård, K. High-intensity interval training, appetite, and reward value of food in the obese. Med. Sci. Sports Exerc. 2017, 49, 1851–1858. [Google Scholar] [CrossRef] [PubMed]
- Peng-Li, D.; Andersen, T.; Finlayson, G.; Byrne, D.V.; Wang, Q.J. The impact of environmental sounds on food reward. Physiol. Behav. 2022, 245, 113689. [Google Scholar] [CrossRef] [PubMed]
- Polivy, J.; Herman, C.P. Distress and eating: Why do dieters overeat? Int. J. Eat. Disord. 1999, 26, 153–164. [Google Scholar] [CrossRef]
- Zellner, D.A.; Loaiza, S.; Gonzalez, Z.; Pita, J.; Morales, J.; Pecora, D.; Wolf, A. Food selection changes under stress. Physiol. Behav. 2006, 87, 789–793. [Google Scholar] [CrossRef] [PubMed]
- Starcke, K.; Agorku, J.D.; Brand, M. Exposure to unsolvable anagrams impairs performance on the Iowa gambling task. Front. Behav. Neurosci. 2017, 11, 114. [Google Scholar] [CrossRef]
- Eaton, J.; Tieber, C. The effects of coloring on anxiety, mood, and perseverance. Art Ther. 2017, 34, 42–46. [Google Scholar] [CrossRef]
- Holt, N.J.; Furbert, L.; Sweetingham, E. Cognitive and affective benefits of colouring: Two randomized controlled crossover studies. PsyArXiv 2018, 36, 200–208. [Google Scholar] [CrossRef]
- Lesage, F.-X.; Berjot, S.; Deschamps, F. Clinical stress assessment using a visual analogue scale. Occup. Med. 2012, 62, 600–605. [Google Scholar] [CrossRef] [Green Version]
- Bjelland, I.; Dahl, A.A.; Haug, T.T.; Neckelmann, D. The validity of the hospital anxiety and depression scale. J. Psychosom. Res. 2002, 52, 69–77. [Google Scholar] [CrossRef]
- BIOPAC Systems Inc. AcqKnowledge® 2022; BIOPAC Systems Inc.: Goleta, CA, USA, 2022. [Google Scholar]
- Rstudio Team RStudio: Integrated Development for R 2022; R.RStudio, PBC: Boston, MA, USA, 2022.
- Jensen, H.A.R.; Davidsen, M.; Møller, S.R.; Román, J.E.I.; Kragelund, K.; Christensen, A.I.; Ekholm, O. Danskernes Sundhed—Den Nationale Sundhedsprofil 2021; Sundhedsstyrelsen: Copenhagen, Denmark, 2022. [Google Scholar]
- World Health Organization (WHO). Depression and Other Common Mental Disorders: Global Health Estimates; WHO: Geneva, Switzerland, 2017. [Google Scholar]
- Biltoft-Jensen, A.P.; Gibbons, S.J.; Kørup, K.; Bestle, S.M.S.; Christenesen, B.J.; Trolle, E.; Lassen, A.D.; Matthiessen, J. Danskerne Er Verdensmestre i Slikindkøb. Available online: https://www.food.dtu.dk/nyheder/2021/03/danskerne-er-verdensmestre-i-slikindkoeb (accessed on 13 June 2022).
- Pedersen, A.N.; Christensen, T.; Matthiessen, J.; Knudsen, V.K.; Rosenlund-Sørensen, M.; Biltoft-Jensen, A.; Hinsch, H.-J.; Ygil, K.H.; Kørup, K.; Saxholt, E.; et al. Dietary Habits in Denmark 2011–2013. Main Results; National Food Institute, Technical University of Denmark: Søborg, Denmark, 2015; ISBN 978-87-93109-39-1. [Google Scholar]
- Epel, E.; Lapidus, R.; McEwen, B.; Brownell, K. Stress may add bite to appetite in women: A laboratory study of stress-induced cortisol and eating behavior. Psychoneuroendocrinology 2001, 26, 37–49. [Google Scholar] [CrossRef]
- Mela, D.J. Eating for pleasure or just wanting to eat? Reconsidering sensory hedonic responses as a driver of obesity. Appetite 2006, 47, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Richard, D.; Timofeeva, E. Energy balance regulation. In Obesity Prevention; Dubé, L., Bechara, A., Dagher, A., Drewnowski, A., Lebel, J., James, P., Yada, R.Y.B.T.-O.P., Eds.; Elsevier: San Diego, CA, USA, 2010; pp. 299–316. ISBN 978-0-12-374387-9. [Google Scholar]
- Oldham, M.; Tomiyama, A.J.; Robinson, E. The Psychosocial experience of feeling overweight promotes increased snack food consumption in women but not men. Appetite 2018, 128, 283–293. [Google Scholar] [CrossRef] [PubMed]
- Major, B.; Hunger, J.M.; Bunyan, D.P.; Miller, C.T. The ironic effects of weight stigma. J. Exp. Soc. Psychol. 2014, 51, 74–80. [Google Scholar] [CrossRef]

| Characteristics | |
|---|---|
| ntotal | 35 |
| Males/females (%) | 17/18 (49%/51%) |
| Age (years) * | 21.71 ± 2.04 (18–25) |
| Educational level | |
| 4 (11%) |
| 25 (71%) |
| 1 (3%) |
| 4 (11%) |
| 1 (3%) |
| Socioeconomic status | |
| 28 (80%) |
| 1 (3%) |
| 6 (17%) |
| BMI (kg/m2) 1,* | 22.14 ± 1.67 (19–25) |
| PSS-10 2,* | 16.11 ± 5.18 (7–27) |
| 12 (34%) |
| 22 (63%) |
| 1 (3%) |
| E% | E% | ||||||
|---|---|---|---|---|---|---|---|
| Pro | Carb | Fat | Pro | Carb | Fat | ||
| HFSA | LFSA | ||||||
| Potato chips | 5 | 36 | 55 | Salad | 18 | 50 | 19 |
| Cheese on cracker | 22 | 34 | 44 | Cucumber | 27 | 65 | 8 |
| Mixed nuts | 16 | 14 | 70 | Pretzels | 11 | 75 | 8 |
| Quiche | 15 | 24 | 59 | Turkey on crispbread | 31 | 63 | 6 |
| HFSW | LFSW | ||||||
| Donut | 5 | 41 | 50 | Mixed berry salad | 8 | 77 | 4 |
| Milk chocolate | 6 | 37 | 53 | Skittles | 0 | 84 | 9 |
| Blueberry muffin | 5 | 44 | 58 | Wine gums | 8 | 86 | 1 |
| Cinnamon roll | 5 | 38 | 54 | Banana | 5 | 84 | 4 |
| E% | ||||
|---|---|---|---|---|
| kJ/100 g | Pro | Carb | Fat | |
| HFSA: Potato chips | 2149 | 5 | 44 | 50 |
| HFSW: Chocolate bar | 2018 | 7 | 42 | 51 |
| LFSA: Wholegrain crackers | 1856 | 13 | 54 | 30 |
| LFSW: Banana | 396 | 5 | 90 | 2 |
| Explicit Liking (mm) | Explicit Wanting (mm) | |||||
| Relaxed | Stressed | p-value | Relaxed | Stressed | p-value | |
| HFSA | 43.70 (±26.18) | 42.76 (±26.25) | NS | 42.50 (±27.32) | 42.46 (±26.72) | NS |
| LFSA | 39.31 (±26.04) | 38.41 (±26.26) | NS | 36.97 (±26.65) | 35.71 (±25.90) | NS |
| HFSW | 50.63 (±25.11) | 53.26 (±25.26) | NS | 45.53 (±25.86) | 47.89 (±26.49) | NS |
| LFSW | 54.57 (±27.96) | 53.14 (±28.54) | NS | 51.49 (±28.77) | 50.76 (±27.76) | NS |
| Implicit wanting (range) | Frequency of choice (count) | |||||
| Relaxed | Stressed | p-value | Relaxed | Stressed | p-value | |
| HFSA | −38–36 | −25–41 | NS | 20.97 (±8.40) | 22.34 (±8.83) | NS |
| LFSA | −31–24 | −31–116 | NS | 18.74 (±7.61) | 17.20 (±8.49) | NS |
| HFSW | −28–34 | −14–57 | 0.006 | 27.20 (±8.52) | 27.74 (±8.88) | NS |
| LFSW | −18–46 | −13–71 | <0.001 | 29.09 (±7.42) | 28.71 (±6.93) | NS |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hyldelund, N.B.; Dalgaard, V.L.; Byrne, D.V.; Andersen, B.V. Why Being ‘Stressed’ Is ‘Desserts’ in Reverse—The Effect of Acute Psychosocial Stress on Food Pleasure and Food Choice. Foods 2022, 11, 1756. https://doi.org/10.3390/foods11121756
Hyldelund NB, Dalgaard VL, Byrne DV, Andersen BV. Why Being ‘Stressed’ Is ‘Desserts’ in Reverse—The Effect of Acute Psychosocial Stress on Food Pleasure and Food Choice. Foods. 2022; 11(12):1756. https://doi.org/10.3390/foods11121756
Chicago/Turabian StyleHyldelund, Nikoline Bach, Vita Ligaya Dalgaard, Derek Victor Byrne, and Barbara Vad Andersen. 2022. "Why Being ‘Stressed’ Is ‘Desserts’ in Reverse—The Effect of Acute Psychosocial Stress on Food Pleasure and Food Choice" Foods 11, no. 12: 1756. https://doi.org/10.3390/foods11121756
APA StyleHyldelund, N. B., Dalgaard, V. L., Byrne, D. V., & Andersen, B. V. (2022). Why Being ‘Stressed’ Is ‘Desserts’ in Reverse—The Effect of Acute Psychosocial Stress on Food Pleasure and Food Choice. Foods, 11(12), 1756. https://doi.org/10.3390/foods11121756









