Impact of Low-Temperature Storage on the Microstructure, Digestibility, and Absorption Capacity of Cooked Rice
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Rice Cooking
2.3. Low-Temperature Storage
2.4. Determination of Absorption Capacity
2.4.1. Oil Absorption Capacity
2.4.2. Cholesterol Absorption Capacity
2.4.3. Glucose Absorption Capacity
2.5. In Vitro Starch Digestibility
2.6. Estimation of Glycemic Index (eGI)
2.7. Microstructural Analysis
2.8. Statistical Analysis
3. Results and Discussion
3.1. Microstructure Changes after Storage
3.2. In Vitro Absorption Capacity
3.2.1. Oil Adsorption Capacity (OAC)
3.2.2. Cholesterol Absorption Capacity (CAC)
3.2.3. Glucose Adsorption Capacity (GAC)
3.3. Relationship between Absorption Capacity, Microstructural Characteristics, and Low-Temperature Storage Conditions
3.4. In Vitro Starch Digestibility
3.4.1. In Vitro Starch Digestion Profile and Kinetics
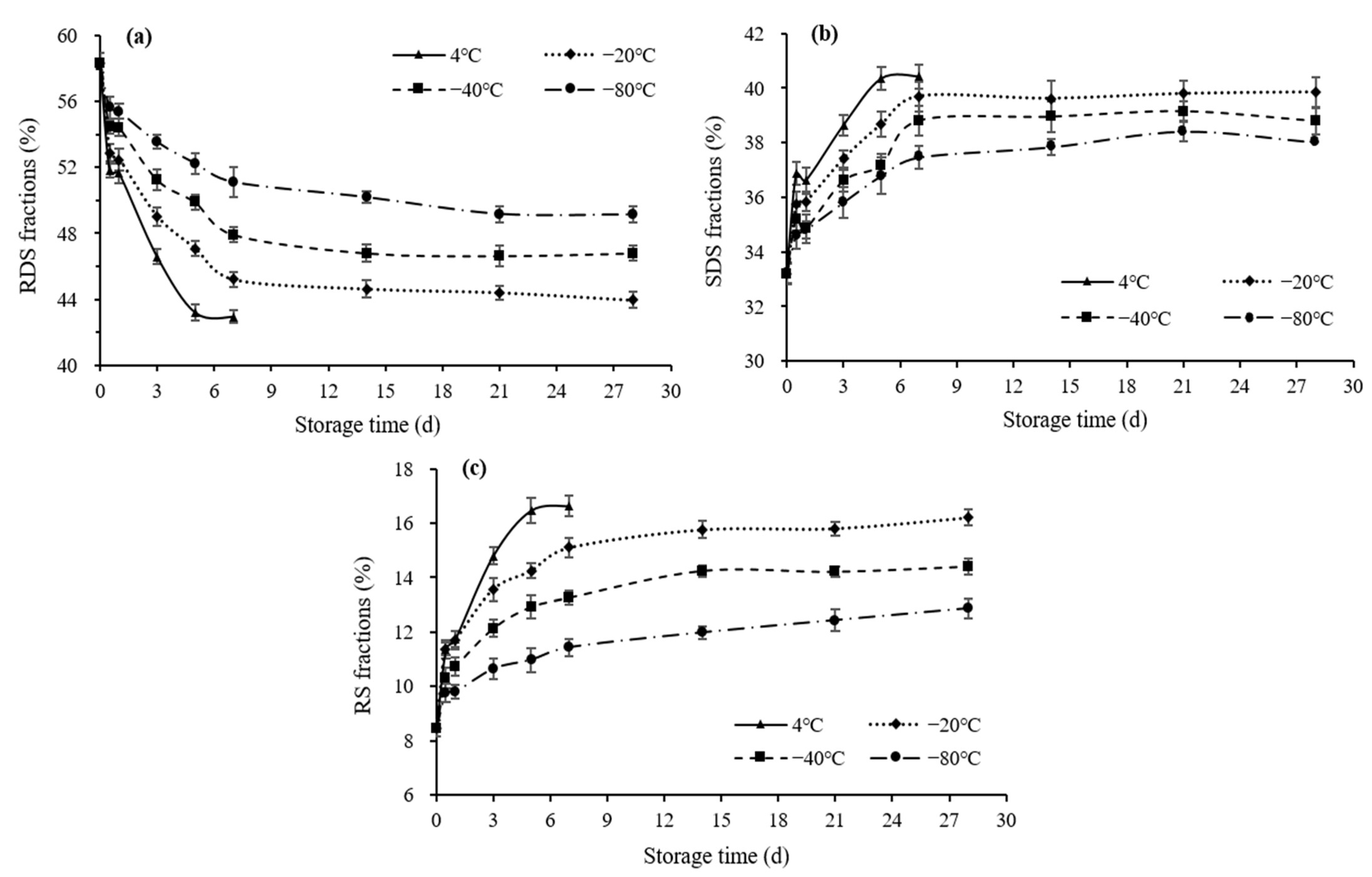
3.4.2. Starch Fractions
3.5. Estimated Glycemic Index
3.6. Pearson Correlation Analysis
3.7. Microstructural Changes during In Vitro Digestion
3.8. Relationship among Low-Temperature Storage Condition, Microstructure and Digestibility
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sultana, S.; Faruque, M.; Islam, M.R. Rice grain quality parameters and determination tools: A review on the current developments and future prospects. Int. J. Food Prop. 2022, 25, 1063–1078. [Google Scholar] [CrossRef]
- Prom-u-thai, C.; Rerkasem, B. Rice quality improvement. A review. Agron. Sustain. Dev. 2020, 40, 28. [Google Scholar] [CrossRef]
- Lu, L.W.; Venn, B.; Lu, J.; Monro, J.; Rush, E. Effect of cold storage and reheating of parboiled rice on postprandial glycaemic response, satiety, palatability and chewed particle size distribution. Nutrients 2017, 9, 475. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, L.W.; Monro, J.; Lu, J.; Rush, E. Effect of cold storage, reheating, and particle sizes on in vitro glucose release and starch digestibility among five rice products in Auckland, New Zealand. Rice Res. 2016, 4, 171. [Google Scholar]
- Larsen, H.N.; Rasmussen, O.W.; Rasmussen, P.H.; Alstrup, K.K.; Biswas, S.K.; Tetens, I.; Thilsted, S.H.; Hermansen, K. Glycaemic index of parboiled rice depends on the severity of processing: Study in type 2 diabetic subjects. Eur. J. Clin. Nutr. 2000, 54, 380–385. [Google Scholar] [CrossRef] [Green Version]
- Tian, J.; Cai, Y.; Qin, W.; Matsushita, Y.; Ye, X.; Ogawa, Y. Parboiling reduced the crystallinity and in vitro digestibility of non-waxy short grain rice. Food Chem. 2018, 257, 23–28. [Google Scholar] [CrossRef]
- Hung, P.V.; Chau, H.T.; Phi, N.T.L. In vitro digestibility and in vivo glucose response of native and physically modified rice starches varying amylose contents. Food Chem. 2016, 191, 74–80. [Google Scholar] [CrossRef]
- Azizi, R.; Capuano, E.; Nasirpour, A.; Pellegrini, N.; Golmakani, M.T.; Hosseini, S.M.H.; Farahnaky, A. Varietal differences in the effect of rice ageing on starch digestion. Food Hydrocolloid 2019, 95, 358–366. [Google Scholar] [CrossRef]
- Wang, S.; Li, P.; Zhang, T.; Wang, S.; Copeland, L. Trypsin and chymotrypsin are necessary for in vitro enzymatic digestion of rice starch. RSC Adv. 2017, 7, 3660–3666. [Google Scholar] [CrossRef] [Green Version]
- Gong, B.; Cheng, L.; Gilbert, R.G.; Li, C. Distribution of short to medium amylose chains are major controllers of in vitro digestion of retrograded rice starch. Food Hydrocolloid 2019, 96, 634–643. [Google Scholar] [CrossRef]
- Kong, F.; Oztop, M.H.; Singh, R.P.; McCarthy, M.J. Physical changes in white and brown rice during simulated gastric digestion. J. Food Sci. 2011, 76, 450–457. [Google Scholar] [CrossRef] [PubMed]
- Goebel, J.T.S.; Kaur, L.; Colussi, R.; Elias, M.C.; Singh, J. Microstructure of indica and japonica rice influences their starch digestibility: A study using a human digestion simulator. Food Hydrocolloid 2019, 94, 191–198. [Google Scholar] [CrossRef]
- Zhu, L.; Lin, Q.; Wilson, J.; Gu, M.; Shi, Y. Digestibility and physicochemical properties of rice (Oryza sativa) flours and starches differing in amylose content. Carbohyd. Polym. 2011, 86, 1751–1759. [Google Scholar] [CrossRef]
- Ye, J.; Hu, X.; Luo, S.; McClements, D.J.; Liang, L. Effect of endogenous proteins and lipids on starch digestibility in rice flour. Food Res. Int. 2018, 106, 404–409. [Google Scholar] [CrossRef] [PubMed]
- Hagenimana, A.; Ding, X.; Fang, T. Evaluation of rice flour modified by extrusion cooking. J. Cereal Sci. 2006, 43, 38–46. [Google Scholar] [CrossRef]
- Wang, S.; Sun, Y.; Wang, J.; Wang, S.; Copeland, L. Molecular disassembly of rice and lotus starches during thermal processing and its effect on starch digestibility. Food Funct. 2016, 7, 1188–1195. [Google Scholar] [CrossRef]
- Tamura, M.; Singh, J.; Kaur, L.; Ogawa, Y. Effect of post-cooking storage on texture and in vitro starch digestion of Japonica rice. J. Food Process Eng. 2019, 42, e12985. [Google Scholar] [CrossRef]
- Ren, X.; Chen, J.; Wang, C.; Molla, M.M.; Diao, X.; Shen, Q. In vitro starch digestibility, degree of gelatinization and estimated glycemic index of foxtail millet-derived products: Effect of freezing and frozen storage. J. Cereal Sci. 2016, 69, 166–173. [Google Scholar] [CrossRef]
- Puspitowati, S.; Driscoll, R.H. Effect of degree of gelatinisation on the rheology and rehydration kinetics of instant rice produced by freeze drying. Int. J. Food Prop. 2017, 10, 445–453. [Google Scholar] [CrossRef]
- Seetapan, N.; Limparyoon, N.; Gamonpilas, C.; Methacanon, P.; Fuongfuchat, A. Effect of cryogenic freezing on textural properties and microstructure of rice flour/tapioca starch blend gel. J. Food Eng. 2015, 151, 51–59. [Google Scholar] [CrossRef]
- Li, X.; Li, D.; Tian, H.; Park, K.H. Reducing retrogradation of gelatinized rice starch and rice meal under low temperature storage by addition of extremely thermostable maltogenic amylase during their cooking. Food Res. Int. 2014, 62, 1134–1140. [Google Scholar] [CrossRef]
- Wang, S.; Li, C.; Copeland, L.; Niu, Q.; Wang, S. Starch retrogradation: A comprehensive review. Compr. Rev. Food Sci. Food Saf. 2015, 14, 568–585. [Google Scholar] [CrossRef]
- Kwaśniewska-Karolak, I.; Mostowski, R. The influence of storage conditions on the staling rate and starch retrogradation in wheat rolls packaged in a modified atmosphere. J. Microb. Biotech. Food Sci. 2019, 8, 1188–1193. [Google Scholar] [CrossRef]
- Kono, S.; Kawamura, I.; Araki, T.; Sagara, Y. ANN modeling for optimum storage condition based on viscoelastic characteristics and sensory evaluation of frozen cooked rice. Int. J. Refrig. 2016, 65, 218–227. [Google Scholar] [CrossRef]
- Yu, S.; Ma, Y.; Sun, D.W. Effects of freezing rates on starch retrogradation and textural properties of cooked rice during storage. LWT-Food Sci. Technol. 2010, 43, 1138–1143. [Google Scholar] [CrossRef]
- Yu, S.; Ma, Y.; Liu, T.; Menager, L.; Sun, D.W. Impact of cooling rates on the staling behavior of cooked rice during storage. J. Food Eng. 2010, 96, 416–420. [Google Scholar] [CrossRef]
- Li, C.; Mense, A.L.; Brewer, L.R.; Lau, C.; Shi, Y. In vitro bile acid binding capacity of wheat bran with different particle sizes. Cereal Chem. 2017, 94, 654–658. [Google Scholar] [CrossRef]
- Drzikova, B.; Dongowski, G.; Gebhardt, E.; Habel, A. The composition of dietary fibre-rich extrudates from oat affects bile acid binding and fermentation in vitro. Food Chem. 2005, 90, 181–192. [Google Scholar] [CrossRef]
- Babu, A.S.; Mohan, R.J. Influence of prior pre-treatments on molecular structure and digestibility of succinylated foxtail millet starch. Food Chem. 2019, 295, 147–155. [Google Scholar] [CrossRef]
- Kahlon, T.S.; Berrios, J.d.J.; Smith, G.E.; Pan, J.L. In vitro bile acid binding capacity of milled wheat bran and milled extruded wheat bran at five specific mechanical energy levels. Cereal Chem. 2006, 83, 434–438. [Google Scholar] [CrossRef]
- AOAC. Official Method of Analysis, 2nd ed.; AOAC International: Gaithersburg, MD, USA, 2003. [Google Scholar]
- Lin, M.J.Y.; Humbert, E.S. Certain functional properties of sunflower meal products. J. Food Sci. 1974, 39, 368–370. [Google Scholar] [CrossRef]
- Ou, S.; Kwok, K.; Li, Y.; Fu, L. In vitro study of possible role of dietary fiber in lowering postprandial serum glucose. J. Agric. Food Chem. 2001, 49, 1026–1029. [Google Scholar] [CrossRef] [PubMed]
- Goñi, I.; Garcia-Alonso, A.; Saura-Calixto, F. A starch hydrolysis procedure to estimate glycemic index. Nutr. Res. 1997, 17, 427–437. [Google Scholar] [CrossRef]
- Englyst, H.N.; Kingman, S.M.; Cummings, J.H. Classification and measurement of nutritionally important starch fractions. Eur. J. Clin. Nutr. 1992, 46, 30–50. [Google Scholar]
- Chen, L.; Tian, Y.; Tong, Q.; Zhang, Z.; Jin, Z. Effect of pullulan on the water distribution, microstructure and textural properties of rice starch gels during cold storage. Food Chem. 2017, 214, 702–709. [Google Scholar] [CrossRef]
- Dun, H.; Liang, H.; Zhan, F.; Wei, X.; Chen, Y.; Wan, J.; Ren, Y.; Hu, L.; Li, B. Influence of O/W emulsion on gelatinization and retrogradation properties of rice starch. Food Hydrocolloid 2020, 103, 105652. [Google Scholar] [CrossRef]
- Luo, Y.; Niu, L.; Li, D.; Xiao, J. Synergistic effects of plant protein hydrolysates and xanthan gum on the short- and long-term retrogradation of rice starch. Int. J. Biol. Macromol. 2020, 144, 967–977. [Google Scholar] [CrossRef]
- Tian, J.; Chen, S.; Wu, C.; Chen, J.; Du, X.; Chen, J.; Liu, D.; Ye, X. Effects of preparation methods on potato microstructure and digestibility: An in vitro study. Food Chem. 2016, 211, 564–569. [Google Scholar] [CrossRef]
- Song, B.S.; Park, J.N.; Lee, J.W.; Kim, J.K.; Kim, J.H. Optimization of processing conditions to improve the rehydration and sensory properties of freeze-dried cooked rice. J. Food Process Pres. 2014, 38, 1244–1250. [Google Scholar] [CrossRef]
- Charoenrein, S.; Preechathammawong, N. Undercooling associated with slow freezing and its influence on the microstructure and properties of rice starch gels. J. Food Eng. 2010, 100, 310–314. [Google Scholar] [CrossRef]
- Cho, C.H.; Lee, H.G.; Jeong, S.; Yoo, S. Influence of packaging oxygen transmission rate on physical characteristics of frozen cooked rice under various freezing conditions. J. Food Sci. 2019, 84, 3483–3493. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Ma, Y.; Zheng, X.Q. Effects of low- and ultralow-temperature freezing on retrogradation and textural properties of rice starch gel during storage. J. Texture Stud. 2012, 43, 175–186. [Google Scholar] [CrossRef]
- Kono, S.; Kawamura, I.; Yamagami, S.; Araki, T.; Sagara, Y. Optimum storage temperature of frozen cooked rice predicted by ice crystal measurement, sensory evaluation and artificial neural network. Int. J. Refrig. 2015, 56, 165–172. [Google Scholar] [CrossRef]
- Kono, S.; Tobari, Y.; Araki, T.; Sagara, Y. Investigating the ice crystal morphology in frozen cooked rice based on size, fractal dimension and ANN modeling. Int. J. Refrig. 2017, 84, 210–219. [Google Scholar] [CrossRef]
- Sangokunle, O.O.; Sathe, S.K.; Singh, P. Purified starches from 18 pulses have markedly different morphology, oil absorption and water absorption capacities, swelling power, and turbidity. Starch-Stärke 2020, 72, 2000022. [Google Scholar] [CrossRef]
- Ahmad, S.; Nema, P.K.; Bashir, K. Effect of different drying techniques on physicochemical, thermal, and functional properties of seera. Dry Technol. 2018, 36, 1284–1291. [Google Scholar] [CrossRef]
- Benítez, V.; Rebollo-Hernanz, M.; Hernanz, S.; Chantres, S.; Aguilera, Y.; Martín-Cabrejas, M.A. Coffee parchment as a new dietary fiber ingredient: Functional and physiological characterization. Food Res. Int. 2019, 122, 105–113. [Google Scholar] [CrossRef]
- Singh, V.; Bedi, G.K.; Shri, R. In vitro and in vivo antidiabetic evaluation of selected culinary-medicinal mushrooms (Agaricomycetes). Int. J. Med. Mushrooms 2017, 19, 17–25. [Google Scholar] [CrossRef]
- Benítez, V.; Rebollo-Hernanz, M.; Aguilera, Y.; Bejerano, S.; Cañas, S.; Martín-Cabrejas, M.A. Extruded coffee parchment shows enhanced antioxidant, hypoglycaemic, and hypolipidemic properties by releasing phenolic compounds from the fibre matrix. Food Funct. 2021, 12, 1097–1110. [Google Scholar] [CrossRef]
- Demirkesen-Bicak, H.; Tacer-Caba, Z.; Nilufer-Erdil, D. Pullulanase treatments to increase resistant starch content of black chickpea (Cicer arietinum L.) starch and the effects on starch properties. Int. J. Biol. Macromol. 2018, 111, 505–513. [Google Scholar] [CrossRef]
- Huang, Y.L.; Ma, Y.S.; Tsai, Y.H.; Chang, S.K.C. In vitro hypoglycemic, cholesterol-lowering and fermentation capacities of fiber-rich orange pomace as affected by extrusion. Int. J. Biol. Macromol. 2019, 124, 796–801. [Google Scholar] [CrossRef] [PubMed]
- Rosin, P.M.; Lajolo, F.M.; Menezes, E.W. Measurement and characterization of dietary starches. J. Food Compos. Anal. 2002, 15, 367–377. [Google Scholar] [CrossRef] [Green Version]
- Sáyago-Ayerdi, S.G.; Tovar, J.; Osorio-Díaz, P.; Paredes-López, O.; Bello-Pérez, L.A. In vitro starch digestibility and predicted glycemic index of corn tortilla, black beans, and tortilla-bean mixture: Effect of cold storage. J. Agric. Food Chem. 2005, 53, 1281–1285. [Google Scholar] [CrossRef] [PubMed]
- Ayimbila, F.; Keawsompong, S. In vitro starch digestion and colonic fermentation of Thai jasmine rice. Starch-Stärke 2018, 70, 1800049. [Google Scholar] [CrossRef]
- Li, H.; Gui, Y.; Li, J.; Zhu, Y.; Cui, B.; Guo, L. Modification of rice starch using a combination of autoclaving and triple enzyme treatment: Structural, physicochemical and digestibility properties. Int. J. Biol. Macromol. 2020, 144, 500–508. [Google Scholar] [CrossRef]
- Bárcenas, M.E.; Rosell, C.M. Effect of frozen storage time on the bread crumb and aging of par-baked bread. Food Chem. 2006, 95, 438–445. [Google Scholar] [CrossRef]
- Tian, J.; Chen, S.; Chen, J.; Liu, D.; Ye, X. Cooking methods altered the microstructure and digestibility of the potato. Starch-Stärke 2018, 70, 1700241. [Google Scholar] [CrossRef]
- Tian, J.; Chen, S.; Shi, J.; Chen, J.; Liu, D.; Cai, Y.; Ogawa, Y.; Ye, X. Microstructure and digestibility of potato strips produced by conventional frying and air-frying: An in vitro study. Food Struct. 2017, 14, 30–35. [Google Scholar] [CrossRef]
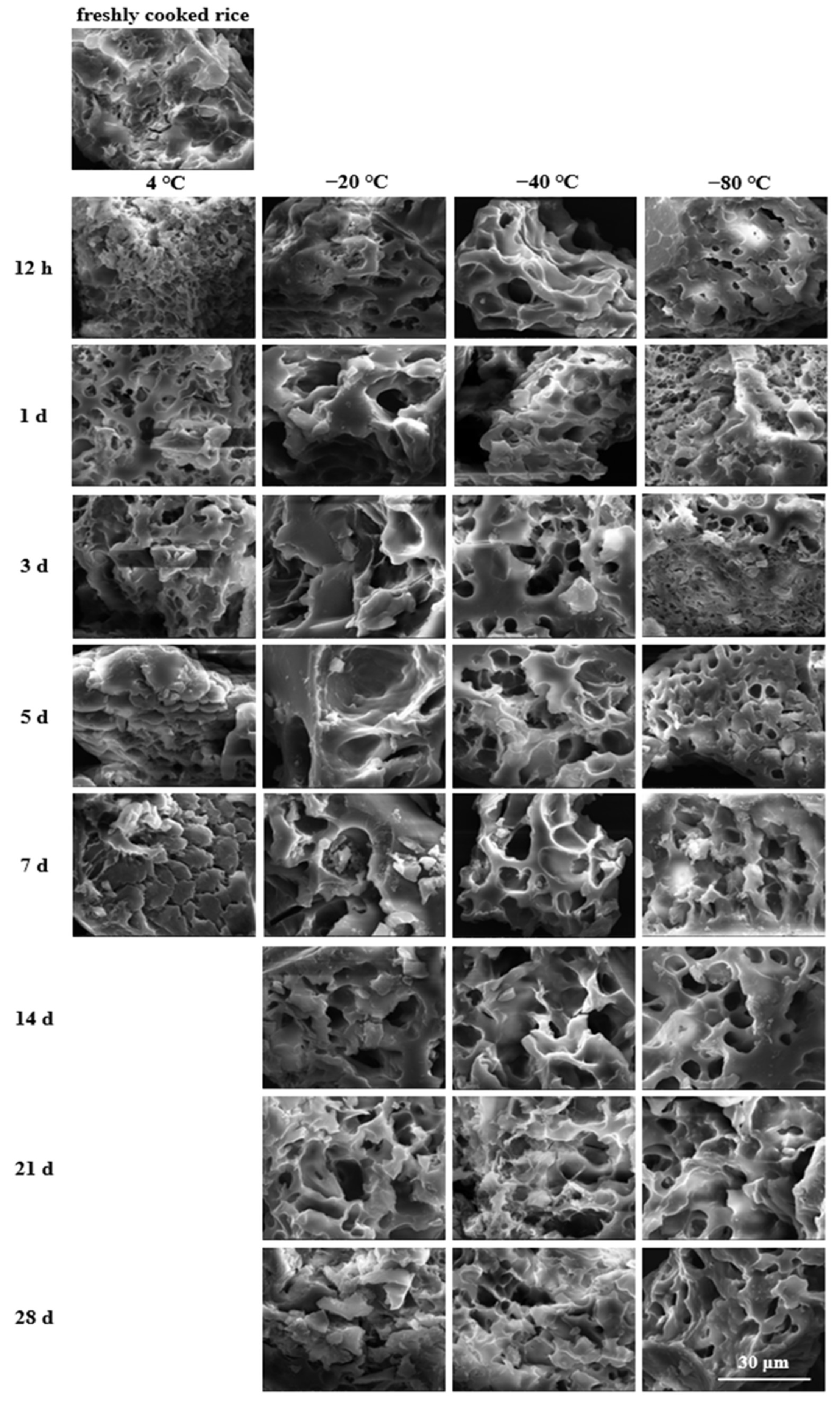
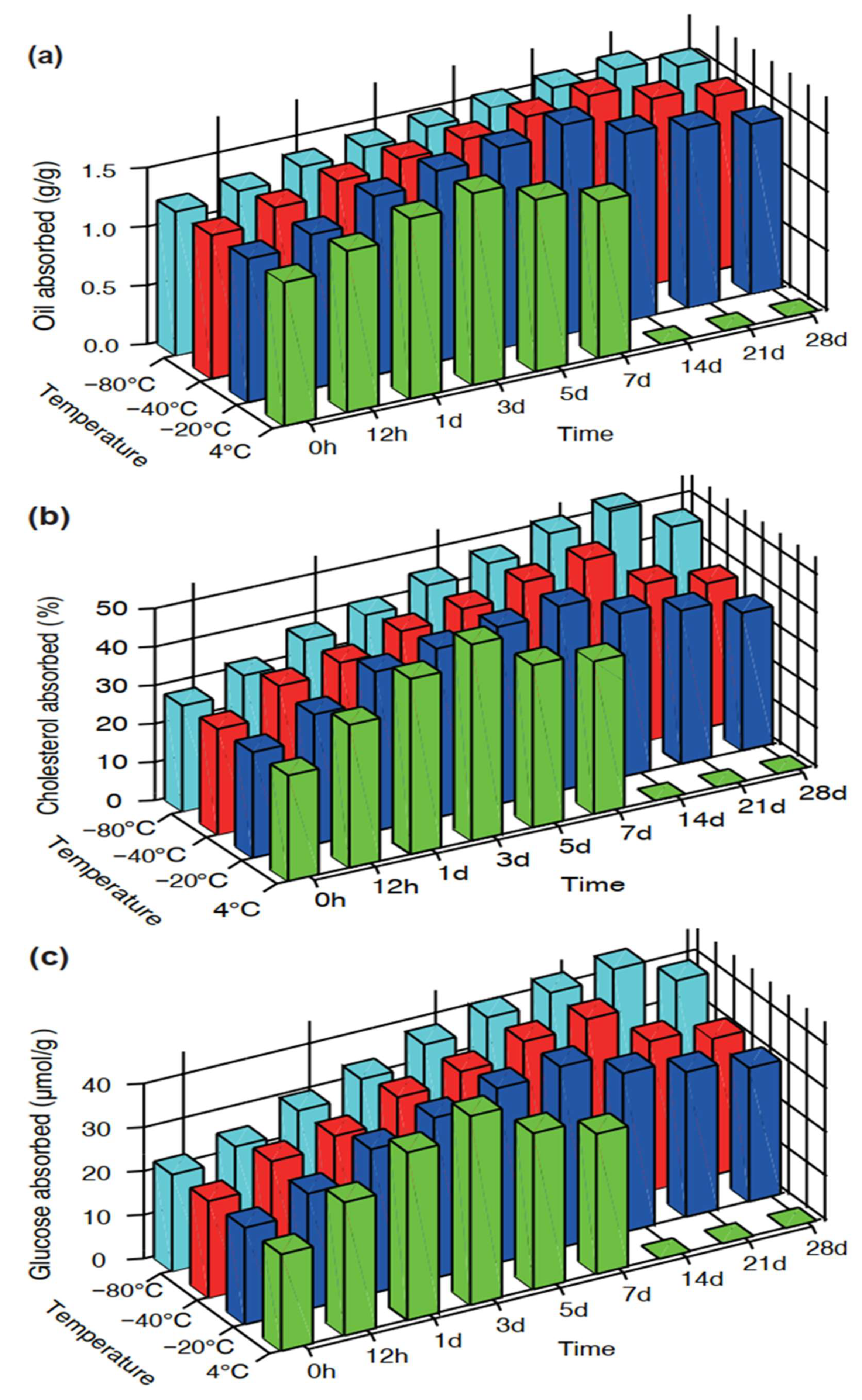
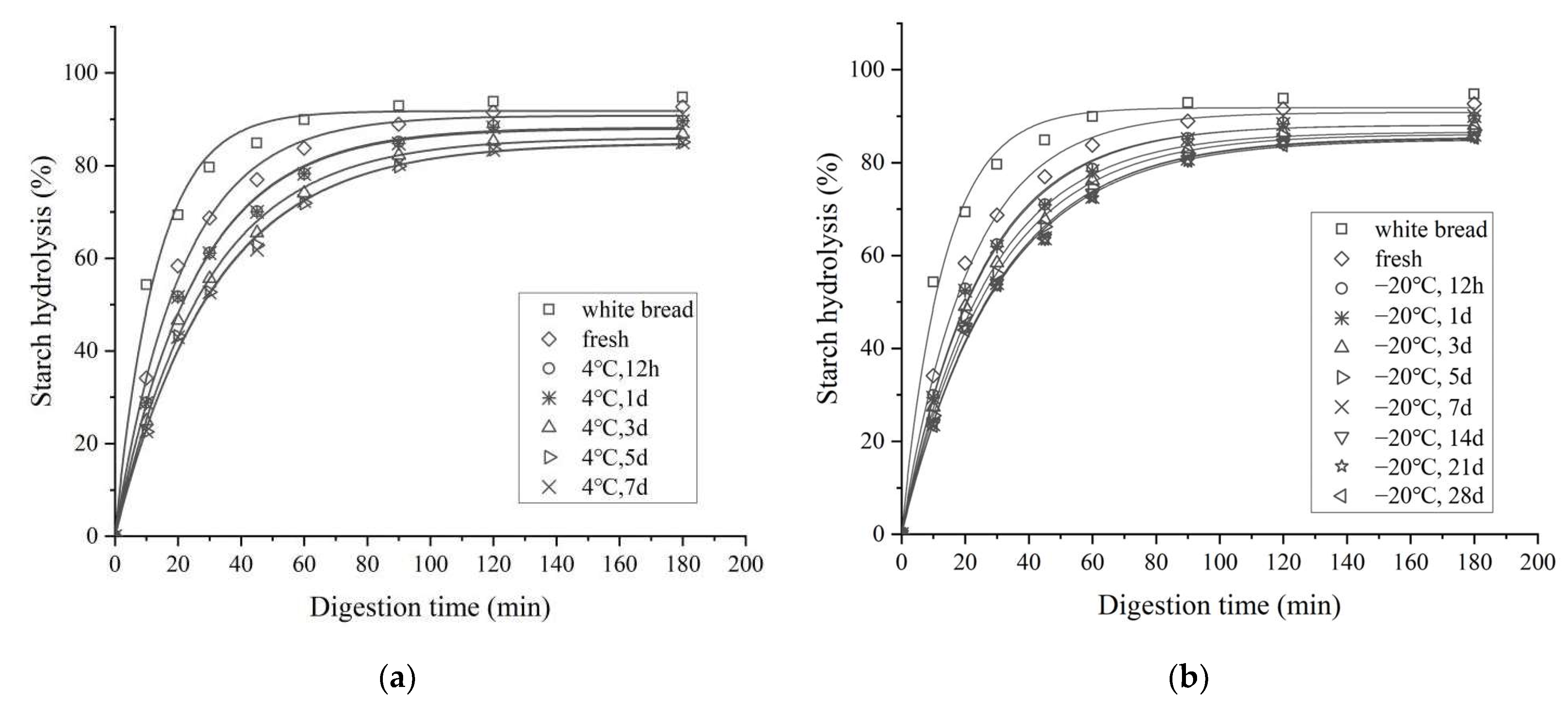
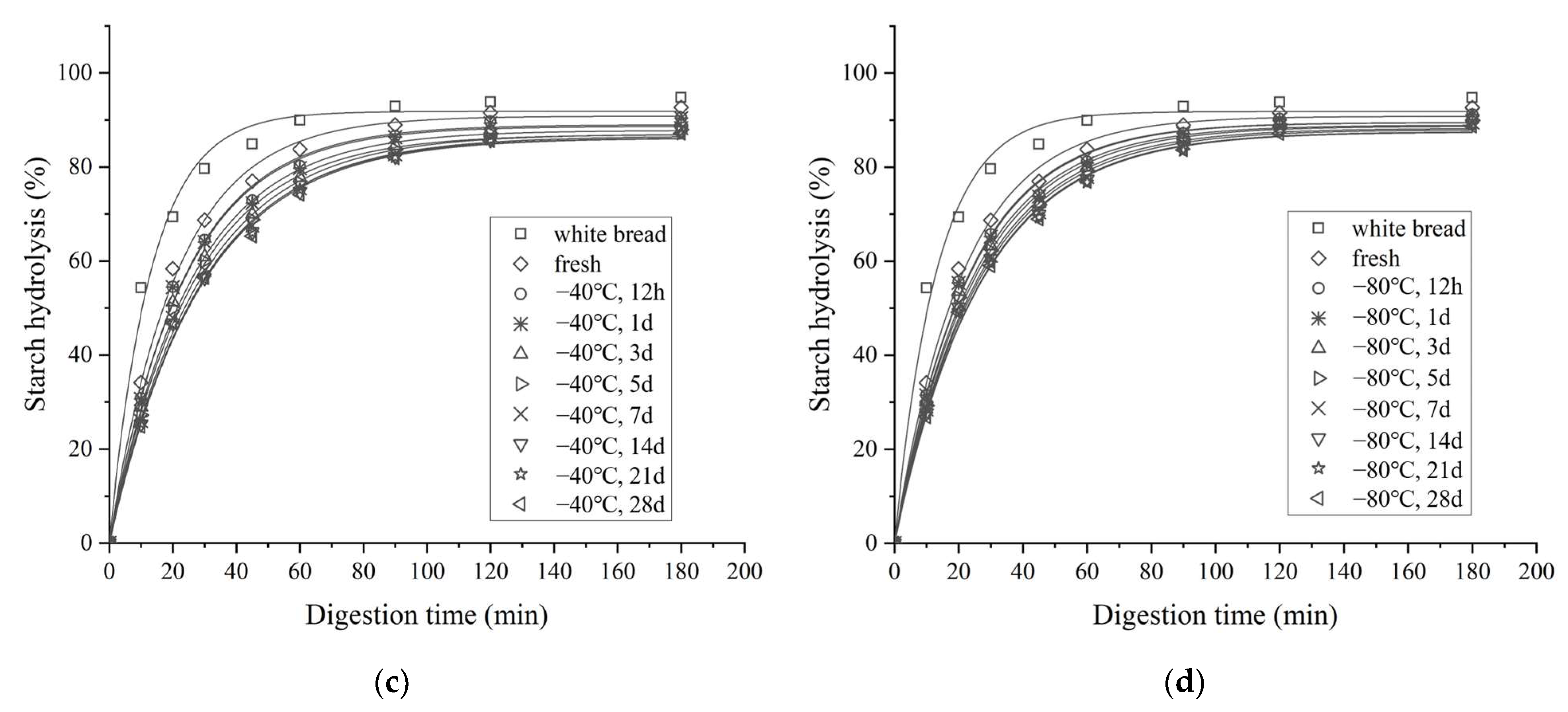




| Parameters | Storage Time (Days) | Storage Temperature | |||
|---|---|---|---|---|---|
| 4 °C | −20 °C | −40 °C | −80 °C | ||
| R2 | 0 | 0.9968 | 0.9968 | 0.9968 | 0.9968 |
| 0.5 | 0.9959 | 0.9958 | 0.9962 | 0.9963 | |
| 1 | 0.9960 | 0.9957 | 0.9956 | 0.9960 | |
| 3 | 0.9970 | 0.9965 | 0.9962 | 0.9963 | |
| 5 | 0.9979 | 0.9971 | 0.9967 | 0.9972 | |
| 7 | 0.9975 | 0.9966 | 0.9969 | 0.9967 | |
| 14 | 0.9976 | 0.9971 | 0.9971 | ||
| 21 | 0.9973 | 0.9975 | 0.9972 | ||
| 28 | 0.9980 | 0.9964 | 0.9974 | ||
| C∞ (%) | 0 | 92.69 ± 0.43 A,c | 92.69 ± 0.43 A,d | 92.69 ± 0.43 A,c | 92.69 ± 0.43 A,c |
| 0.5 | 89.67 ± 0.72 A,b | 89.68 ± 0.45 A,c | 90.49 ± 0.51 A,b | 91.17 ± 0.31 A,b | |
| 1 | 89.60 ± 0.67 A,b | 90.06 ± 0.56 A,c | 90.35 ± 0.46 A,b | 91.13 ± 0.33 A,b | |
| 3 | 86.82 ± 0.69 A,a | 88.03 ± 0.32 A,b | 89.53 ± 0.34 B,b | 90.67 ± 0.28 C,b | |
| 5 | 85.10 ± 1.06 A,a | 86.99 ± 0.69 AB,ab | 87.93 ± 0.31 B,a | 89.92 ± 0.53 C,ab | |
| 7 | 85.04 ± 0.91 A,a | 86.03 ± 0.58 A,a | 88.27 ± 0.28 B,a | 89.47 ± 0.30 C,a | |
| 14 | 85.80 ± 0.72 A,a | 87.76 ± 0.41 B,a | 89.24 ± 0.42 C,a | ||
| 21 | 85.47 ± 0.85 A,a | 86.92 ± 0.52 A,a | 88.99 ± 0.37 B,a | ||
| 28 | 85.40 ± 0.78 A,a | 87.15 ± 0.39 A,a | 88.70 ± 0.55 B,a | ||
| k × 10−2 (min−1) | 0 | 4.72 ± 0.08 A,c | 4.72 ± 0.08 A,d | 4.72 ± 0.08 A,d | 4.72 ± 0.08 A,d |
| 0.5 | 3.94 ± 0.12 A,b | 4.12 ± 0.07 AB,c | 4.28 ± 0.06 B,c | 4.40 ± 0.07 B,c | |
| 1 | 3.95 ± 0.07 A,b | 4.05 ± 0.04 A,c | 4.26 ± 0.08 B,c | 4.35 ± 0.05 B,c | |
| 3 | 3.47 ± 0.08 A,a | 3.75 ± 0.06 B,b | 3.94 ± 0.07 B,b | 4.15 ± 0.04 C,b | |
| 5 | 3.22 ± 0.06 A,a | 3.54 ± 0.05 B,ab | 3.85 ± 0.05 C,b | 4.04 ± 0.04 D,b | |
| 7 | 3.20 ± 0.11 A,a | 3.33 ± 0.09 A,a | 3.62 ± 0.06 B,a | 3.91 ± 0..02 C,a | |
| 14 | 3.35 ± 0.03 A,a | 3.50 ± 0.08 A,a | 3.84 ± 0.06 B,a | ||
| 21 | 3.31 ± 0.08 A,a | 3.54 ± 0.05 B,a | 3.76 ± 0.05 C,a | ||
| 28 | 3.32 ± 0.04 A,a | 3.49 ± 0.09 AB,a | 3.74 ± 0.09 B,a | ||
| Parameters | Storage Time (Days) | Storage Temperature | |||
|---|---|---|---|---|---|
| 4 °C | −20 °C | −40 °C | −80 °C | ||
| HI | 0 | 93.57 ± 0.21 A,d | 93.57 ± 0.21 A,d | 93.57 ± 0.21 A,e | 93.57 ± 0.21 A,d |
| 0.5 | 88.54 ± 0.17 A,c | 88.95 ± 0.28 A,c | 90.41 ± 0.19 B,d | 91.31 ± 0.19 C,c | |
| 1 | 88.30 ± 0.22 A,c | 88.71 ± 0.14 A,c | 90.04 ± 0.23 B,d | 91.14 ± 0.16 C,c | |
| 3 | 84.29 ± 0.19 A,b | 86.09 ± 0.44 B,b | 88.02 ± 0.16 C,c | 89.91 ± 0.40 D,b | |
| 5 | 81.83 ± 0.31 A,a | 84.71 ± 0.33 B,b | 86.76 ± 0.28 C,b | 89.30 ± 0.25 D,b | |
| 7 | 81.82 ± 0.26 A,a | 83.10 ± 0.25 B,a | 85.89 ± 0.19 C,ab | 88.38 ± 0.39 D,ab | |
| 14 | 82.93 ± 0.18 A,a | 84.90 ± 0.35 B,a | 87.78 ± 0.24 C,a | ||
| 21 | 82.51 ± 0.22 A,a | 84.79 ± 0.40 B,a | 87.06 ± 0.31 C,a | ||
| 28 | 82.47 ± 0.15 A,a | 84.50 ± 0.51 B,a | 86.84 ± 0.44 C,a | ||
| eGI | 0 | 91.08 ± 0.37 A,d | 91.08 ± 0.37 A,d | 91.08 ± 0.37 A,d | 91.08 ± 0.37 A,c |
| 0.5 | 88.32 ± 0.28 A,c | 88.54 ± 0.42 AB,c | 89.35 ± 0.22 B,c | 89.84 ± 0.18 B,b | |
| 1 | 88.19 ± 0.23 A,c | 88.41 ± 0.38 AB,c | 89.14 ± 0.12 B,c | 89.75 ± 0.36 B,b | |
| 3 | 85.99 ± 0.17 A,b | 86.97 ± 0.33 B,b | 88.03 ± 0.36 C,b | 89.07 ± 0.24 D,b | |
| 5 | 84.64 ± 0.42 A,a | 86.21 ± 0.25 B,b | 87.34 ± 0.29 C,b | 88.74 ± 0.39 D,ab | |
| 7 | 84.63 ± 0.35 A,a | 85.33 ± 0.18 A,a | 86.87 ± 0.34 B,ab | 88.23 ± 0.15 C,a | |
| 14 | 85.24 ± 0.15 A,a | 86.32 ± 0.19 B,a | 87.90 ± 0.32 C,a | ||
| 21 | 85.01 ± 0.21 A,a | 86.26 ± 0.28 B,a | 87.51 ± 0.24 C,a | ||
| 28 | 84.98 ± 0.26 A,a | 86.10 ± 0.32 B,a | 87.38 ± 0.33 C,a | ||
| Parameters | k | C∞ | HI | eGI | RDS | SDS |
|---|---|---|---|---|---|---|
| C∞ | 0.9745 | |||||
| HI | 0.9919 | 0.9927 | ||||
| eGI | 0.9919 | 0.9927 | 1.0000 | |||
| RDS | 0.9964 | 0.9841 | 0.9957 | 0.9957 | ||
| SDS | –0.9880 | –0.9491 | –0.9686 | –0.9686 | –0.9858 | |
| RS | –0.9809 | –0.9919 | –0.9965 | –0.9965 | –0.9894 | 0.9510 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, H.; Liu, B.; Bess, K.; Wang, Z.; Liang, M.; Zhang, Y.; Wu, Q.; Yang, L. Impact of Low-Temperature Storage on the Microstructure, Digestibility, and Absorption Capacity of Cooked Rice. Foods 2022, 11, 1642. https://doi.org/10.3390/foods11111642
Li H, Liu B, Bess K, Wang Z, Liang M, Zhang Y, Wu Q, Yang L. Impact of Low-Temperature Storage on the Microstructure, Digestibility, and Absorption Capacity of Cooked Rice. Foods. 2022; 11(11):1642. https://doi.org/10.3390/foods11111642
Chicago/Turabian StyleLi, Hui, Bingxiao Liu, Kezia Bess, Zhengxuan Wang, Mingcai Liang, Yan Zhang, Qiong Wu, and Lin Yang. 2022. "Impact of Low-Temperature Storage on the Microstructure, Digestibility, and Absorption Capacity of Cooked Rice" Foods 11, no. 11: 1642. https://doi.org/10.3390/foods11111642
APA StyleLi, H., Liu, B., Bess, K., Wang, Z., Liang, M., Zhang, Y., Wu, Q., & Yang, L. (2022). Impact of Low-Temperature Storage on the Microstructure, Digestibility, and Absorption Capacity of Cooked Rice. Foods, 11(11), 1642. https://doi.org/10.3390/foods11111642






