An Exploration of Listeria monocytogenes, Its Influence on the UK Food Industry and Future Public Health Strategies
Abstract
1. Introduction
2. Clinical Manifestations of Listeriosis and UK Outbreaks
3. Listeria in Food Manufacturing Environments
3.1. Occurrence of Listeria from Primary Food Production to Retail
3.2. Current Contamination Control Strategies
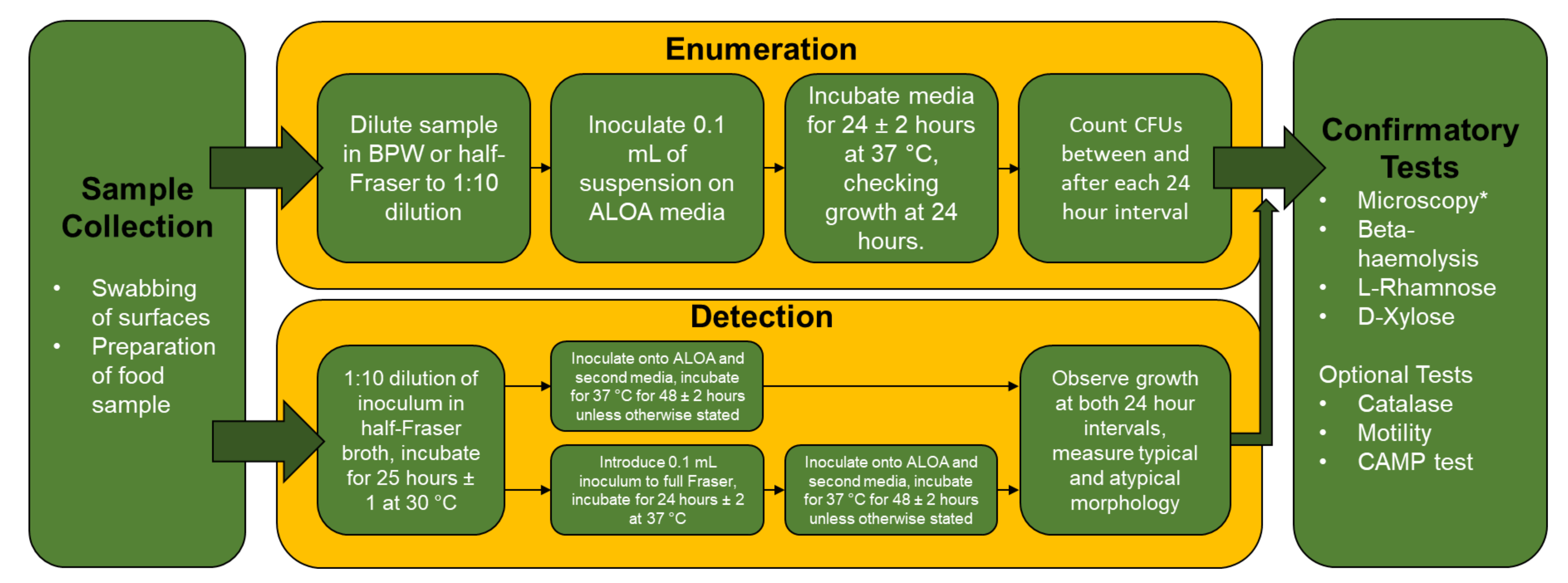
3.3. Microbiological Testing of Listeria spp. in the Food Industry
4. Stress Tolerances in Food Manufacturing Environments
4.1. Biocides in Food Manufacturing
4.2. Antibiotic Resistance and Biocide Cross-Resistance
5. Recommendations and Future Strategies
5.1. Research-Led Surveillance
5.2. Methodologies for Culturing Listeria spp.
5.3. Future Research
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Radoshevich, L.; Cossart, P. Listeria monocytogenes: Towards a complete picture of its physiology and pathogenesis. Nat. Rev. Microbiol. 2018, 16, 32–46. [Google Scholar] [CrossRef] [PubMed]
- Public Health England. Listeriosis in England and Wales: Summary for 2019. 2021. Available online: https://www.gov.uk/government/publications/listeria-monocytogenes-surveillance-reports/listeriosis-in-england-and-wales-summary-for-2019 (accessed on 25 April 2022).
- Public Health England. Listeriosis in England and Wales: Summary for Listeriosis in 2017. 2021. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/765214/listeriosis_in_england_and_wales_summary_for_2017.pdf (accessed on 25 April 2022).
- Public Health England. Listeriosis in England and Wales: Summary for Listeriosis in 2018. 2021. Available online: https://www.gov.uk/government/publications/listeria-monocytogenes-surveillance-reports/listeriosis-in-england-and-wales-summary-for-2018 (accessed on 25 April 2022).
- Chan, Y.C.; Hu, Y.; Chaturongakul, S.; Files, K.D.; Bowen, B.M.; Boor, K.J.; Wiedmann, M. Contributions of two-component regulatory systems, alternative sigma factors, and negative regulators to Listeria monocytogenes cold adaptation and cold growth. J. Food Prot. 2008, 71, 420–425. [Google Scholar] [CrossRef] [PubMed]
- Bucur, F.I.; Grigore-Gurgu, L.; Crauwels, P.; Riedel, C.U.; Nicolau, A.I. Resistance of Listeria monocytogenes to Stress Conditions Encountered in Food and Food Processing Environments. Front. Microbiol. 2018, 9, 2700. [Google Scholar] [CrossRef] [PubMed]
- Sleator, R.D.; Gahan, C.G.M.; Hill, C. A Postgenomic Appraisal of Osmotolerance in Listeria monocytogenes. Appl. Environ. Microbiol. 2003, 69, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Koutsoumanis, K.P.; Kendall, P.A.; Sofos, J.N. A comparative study on growth limits of Listeria monocytogenes as affected by temperature, pH and aw when grown in suspension or on a solid surface. Food Microbiol. 2004, 21, 415–422. [Google Scholar] [CrossRef]
- Mereghetti, L.; Quentin, R.; Marquet-Van Der Mee, N.; Audurier, A. Low Sensitivity of Listeria monocytogenes to Quaternary Ammonium Compounds. Appl. Environ. Microbiol. 2000, 66, 5083–5086. [Google Scholar] [CrossRef]
- Martínez-Suárez, J.V.; Ortiz, S.; López-Alonso, V. Potential Impact of the Resistance to Quaternary Ammonium Disinfectants on the Persistence of Listeria monocytogenes in Food Processing Environments. Front. Microbiol. 2016, 7, 638. [Google Scholar] [CrossRef]
- Gandhi, M.; Chikindas, M.L. Listeria: A foodborne pathogen that knows how to survive. Int. J. Food Microbiol. 2007, 113, 1–15. [Google Scholar] [CrossRef]
- Conficoni, D.; Losasso, C.; Cortini, E.; Di Cesare, A.; Cibin, V.; Giaccone, V.; Corno, G.; Ricci, A. Resistance to Biocides in Listeria monocytogenes Collected in Meat-Processing Environments. Front. Microbiol. 2016, 7, 1627. [Google Scholar] [CrossRef]
- Orsi, R.H.; den Bakker, H.C.; Wiedmann, M. Listeria monocytogenes lineages: Genomics, evolution, ecology, and phenotypic characteristics. Int. J. Med. Microbiol. 2011, 301, 79–96. [Google Scholar] [CrossRef]
- Lukinmaa, S.; Miettinen, M.; Nakari, U.; Korkeala, H.; Siitonen, A. Listeria monocytogenes Isolates from Invasive Infections: Variation of Sero- and Genotypes during an 11-Year Period in Finland. J. Clin. Microbiol. 2003, 41, 1694–1700. [Google Scholar] [CrossRef]
- McLauchlin, J.; Grant, K.A.; Amar, C.F.L. Human foodborne listeriosis in England and Wales, 1981 to 2015. Epidemiol. Infect. 2020, 148, e54. [Google Scholar] [CrossRef] [PubMed]
- Dreyer, M.; Aguilar-Bultet, L.; Rupp, S.; Guldimann, C.; Stephan, R.; Schock, A.; Otter, A.; Schüpbach, G.; Brisse, S.; Lecuit, M.; et al. Listeria monocytogenes sequence type 1 is predominant in ruminant rhombencephalitis. Sci. Rep. 2016, 16, 36419. [Google Scholar] [CrossRef] [PubMed]
- Salcedo, C.; Arreaza, L.; Alcalá, B.; de la Fuenta, L.; Vázquez, J.A. Development of a multilocus sequence typing method for analysis of Listeria monocytogenes clones. J. Clin. Microbiol. 2003, 41, 757–762. [Google Scholar] [CrossRef]
- Ragon, M.; Wirth, T.; Hollandt, F.; Lavenir, R.; Lecuit, M.; Le Monnier, A.; Brisse, S. A New Perspective on Listeria monocytogenes Evolution. PLoS Pathog. 2008, 4, e1000146. [Google Scholar] [CrossRef]
- Bergholz, T.M.; Shah, M.K.; Burall, L.S.; Rakic-Martinez, M.; Datta, A.R. Genomic and phenotypic diversity of Listeria monocytogenes clonal complexes associated with human listeriosis. Appl. Microbiol. Biotechnol. 2018, 102, 3475–3485. [Google Scholar] [CrossRef]
- Swaminathan, B.; Gerner-Smidt, P. The epidemiology of human listeriosis. Microbes Infect. 2007, 9, 1236–1243. [Google Scholar] [CrossRef]
- Drevets, D.A.; Bronze, M.S. Listeria monocytogenes: Epidemiology, human disease, and mechanisms of brain invasion. FEMS Immunol. Med. Microbiol. 2008, 53, 151–165. [Google Scholar] [CrossRef]
- Godshall, C.E.; Suh, G.; Lorber, V. Cutaneous Listeriosis. J. Clin. Microbiol. 2013, 51, 3591–3596. [Google Scholar] [CrossRef] [PubMed]
- Valckx, W.J.A.R.M.; Lutgens, S.P.M.; Haerkens-Arends, H.E.; Barneveld, P.C.; Beutler, J.J.; Hoogeveen, E.K. Listeria Endocarditis: A Diagnostic Challenge. J. Investig. Med. High Impact Case Rep. 2017, 5, 2324709617698995. [Google Scholar] [CrossRef]
- Cone, L.A.; Fitzmorris, A.O.; Hirschberg, J.M. Is Listeria monocytogenes an Important Pathogen for Prosthetic Joints? J. Clin. Rheumatol. 2001, 7, 34–37. [Google Scholar] [CrossRef] [PubMed]
- Bakardijev, A.I.; Theriot, J.A.; Portnoy, D.A. Listeria monocytogenes traffics from maternal organs to the placenta and back. PLoS Pathog. 2006, 2, e66. [Google Scholar]
- Imanishi, M.; Routh, J.A.; Klaber, M.; Gu, W.; Vanselow, M.S.; Jackson, K.A.; Sullivan-Chang, L.; Heinrichs, G.; Jain, N.; Albanese, B.; et al. Estimating the attack rate of pregnancy-associated listeriosis during a large outbreak. Infect. Dis. Obstet. Gynecol. 2015, 2015, 201479. [Google Scholar] [CrossRef] [PubMed]
- Pouillot, R.; Klontz, K.C.; Chen, Y.; Burall, L.S.; Macarisin, D.; Doyle, M.; Bally, K.M.; Strain, E.; Datta, A.R.; Hammack, T.S.; et al. Infectious Dose of Listeria monocytogenes in Outbreak Linked to Ice Cream, United States, 2015. Emerg. Infect. Dis. 2016, 22, 2113–2119. [Google Scholar] [CrossRef] [PubMed]
- Goulet, V.; King, L.A.; Vaillant, V.; de Valk, H. What is the incubation period for listeriosis? BMC Infect. Dis. 2013, 13, 11. [Google Scholar] [CrossRef]
- Angelo, K.M.; Jackson, K.A.; Wong, K.K.; Hoekstra, R.M.; Jackson, B.R. Assessment of the Incubation Period for Invasive Listeriosis. Clin. Infect. Dis. 2017, 63, 1487–1489. [Google Scholar] [CrossRef] [PubMed]
- British National Formulary. Central Nervous System Infections, Antibacterial Therapy. 2021. Available online: https://bnf.nice.org.uk/treatment-summary/central-nervous-system-infections-antibacterial-therapy.html (accessed on 25 April 2022).
- British National Formulary for Children. Central Nervous System Infections, Antibacterial Therapy. 2021. Available online: https://bnfc.nice.org.uk/treatment-summary/central-nervous-system-infections-antibacterial-therapy.html (accessed on 25 April 2022).
- Yu, P.A.; Tran, E.L.; Parker, C.M.; Kim, H.; Yee, E.L.; Smith, P.W.; Russell, Z.; Nelson, C.A.; Broussard, C.S.; Yu, Y.C.; et al. Safety of Antimicrobials During Pregnancy: A Systematic Review of Antimicrobials Considered for Treatment and Postexposure Prophylaxis of Plague. Clin. Infect. Dis. 2020, 70, 37–50. [Google Scholar] [CrossRef] [PubMed]
- UK Teratology Information Service. Use of Aminoglycoside Antibiotics in Pregnancy. 2017. Available online: https://www.medicinesinpregnancy.org/bumps/monographs/USE-OF-AMINOGLYCOSIDE-ANTIBIOTICS-IN-PREGNANCY/ (accessed on 25 April 2022).
- Bannister, B.A. Listeria monocytogenes meningitis associated with eating soft cheese. J. Infect. 1987, 15, 165–168. [Google Scholar] [CrossRef]
- Kerr, K.G.; Rotowa, N.A.; Hawkey, P.M.; Lacey, R.W. Incidence of Listeria spp. in Pre-cooked, Chilled Chicken Products as Determined by Culture and Enzyme-linked Immunoassay (ELISA). J. Food Prot. 1988, 53, 606–607. [Google Scholar] [CrossRef]
- McLauchlin, J. Distribution of serovars of Listeria monocytogenes isolated from different categories of patients with listeriosis. Eur. J. Clin. Microbiol. Dis. 1990, 9, 210–213. [Google Scholar] [CrossRef]
- McLauchlin, J.; Hall, S.M.; Velani, S.K.; Gilbert, R.J. Human listeriosis and paté: A possible association. BMJ 1991, 303, 773–775. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gilbert, R.J.; McLauchlin, J.; Velani, S.K. The contamination of paté by Listeria monocytogenes in England and Wales in 1989 and 1990. Epidemiol. Infect. 1993, 110, 543–551. [Google Scholar] [CrossRef] [PubMed]
- Morris, I.J.; Ribeiro, C.D. The occurrence of Listeria species in pâté: The Cardiff experience 1989. Epidemiol. Infect. 1991, 107, 111–117. [Google Scholar] [CrossRef]
- Advisory Committee on the Microbiological Safety of Food. Recent Trends in Listeriosis in the UK. 2003. Available online: https://acmsf.food.gov.uk/sites/default/files/mnt/drupal_data/sources/files/multimedia/pdfs/acm667.pdf (accessed on 25 April 2022).
- Graham, J.C.; Lanser, S.; Bignardi, G.; Pedler, S.; Hollyoak, V. Hospital-acquired listeriosis. J. Hosp. Infect. 2002, 51, 139. [Google Scholar] [CrossRef]
- Nolla-Salas, J.; Antó, J.M.; Almela, M.; Coll, P.; Gasser, I.; Plasencia, A. Incidence of listeriosis in Barcelona, Spain, in 1990. Eur. J. Clin. Microbiol. Infect. Dis. 1993, 12, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Büla, C.J.; Bille, J.; Glauser, M.P. An Epidemic of Food-Borne Listeriosis in Western Switzerland: Description of 57 Cases Involving Adults. Clin. Infect. Dis. 1995, 20, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Loncarevic, S.; Danielsson-Tham, M.; Tham, W. Occurrence of Listeria monocytogenes in soft and semi-soft cheeses in retail outlets in Sweden. Int. J. Food Microbiol. 1995, 26, 245–250. [Google Scholar] [CrossRef]
- Aureli, P.; Fiorucci, G.C.; Caroli, D.; Marchiaro, F.; Novara, O.; Leone, L.; Salmaso, S. An outbreak of febrile gastroenteritis associated with corn contaminated by Listeria monocytogenes. N. Eng. J. Med. 2000, 342, 1236–1241. [Google Scholar] [CrossRef] [PubMed]
- Public Health England. The Health Protection (Notification) Regulations 2010. 2010. Available online: https://www.legislation.gov.uk/uksi/2010/659/pdfs/uksi_20100659_en.pdf (accessed on 25 April 2022).
- McLauchlin, J.; Aird, H.; Amar, C.; Boyd, G.; Brindle, A.; Dallman, T.; Jalava, K.; Painset, A.; Simbo, A.; Swindlehurst, M. Listeriosis associated with pre-prepared sandwich consumption in hospital in England, 2017. Epidemiol. Infect. 2021, 149, e220. [Google Scholar] [CrossRef]
- Dawson, S.J.; Evans, M.R.; Willby, D.; Chamberlain, N.; Lewis, D.A. Listeria outbreak associated with sandwich consumption from a hospital retail shop, United Kingdom. Euro. Surveill. 2006, 11, 632. [Google Scholar] [CrossRef]
- Shetty, A.; McLauchlin, J.; Grant, K.; O’Brien, D.; Howard, T.; Davies, E.M. Outbreak of Listeria monocytogenes in an oncology unit associated with sandwiches consumed in hospital. J. Hosp. Infect. 2009, 71, 332–336. [Google Scholar] [CrossRef] [PubMed]
- Little, C.L.; Amar, C.F.L.; Awofisayo, A.; Grant, K.A. Hospital-acquired listeriosis associated with sandwiches in the UK: A cause for concern. J. Hosp. Infect. 2012, 82, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Public Health England. Investigation into an Outbreak of Listeria monocytogenes Infections Associated with Hospital-Provided Preprepared Sandwiches, UK May to July 2019. 2020. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/937907/2019-05-Listeria-CC8-Outbreak-Report.pdf (accessed on 25 April 2022).
- Lamden, K.H.; Fox, A.J.; Amar, C.F.L.; Little, C.L. A case of foodborne listeriosis linked to a contaminated food-production process. J. Med. Microbiol. 2013, 62, 1614–1616. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Awofisayo-Okuyelu, A.; Arunachalam, N.; Dallman, T.; Grant, K.A.; Aird, H.; McLauchlin, J.; Painset, A.; Amar, C. An Outbreak of Human Listeriosis in England between 2010 and 2012 Associated with the Consumption of Pork Pies. J. Food Prot. 2016, 79, 732–740. [Google Scholar] [CrossRef] [PubMed]
- Elson, R.; Awofisayo-Okuyelu, A.; Greener, T.; Swift, C.; Painset, A.; Amar, C.F.L.; Newton, A.; Aird, H.; Swindlehurst, M.; Elviss, N.; et al. Utility of Whole Genome Sequencing to Describe the Persistence and Evolution of Listeria monocytogenes Strains within Crabmeat Processing Environments Linked to Two Outbreaks of Listeriosis. J. Food Prot. 2019, 82, 30–38. [Google Scholar] [CrossRef] [PubMed]
- McLauchlin, J.; Aird, H.; Amar, C.; Barker, C.; Dallman, T.; Lai, S.; Painset, A.; Willis, C. An outbreak of human listeriosis associated with frozen sweet corn consumption: Investigations in the UK. Int. J. Food Microbiol. 2021, 338, 108994. [Google Scholar] [CrossRef]
- Legislation.gov. Commission Regulation (EC) No 2073/2005. 2020. Available online: https://www.legislation.gov.uk/eur/2005/2073/contents# (accessed on 25 April 2022).
- Mensah, L.D.; Julien, D. Implementation of food safety management systems in the UK. Food Control 2011, 22, 1216–1225. [Google Scholar] [CrossRef]
- Gerba, C.P. Quaternary ammonium biocides: Efficacy in application. Appl. Environ. Microbiol. 2015, 81, 464–469. [Google Scholar] [CrossRef]
- Piira, N.; Kosola, M.; Hellsten, C.; Fagerlund, A.; Lundén, J. Comparison of official food control results in Finland between food establishments with and without a certified food safety management system. Food Control 2021, 129, 108230. [Google Scholar] [CrossRef]
- Psomas, E.L.; Kafetzopoulos, D.P. HACCP effectiveness between ISO 22000 certified and non-certified dairy companies. Food Control 2015, 53, 134–139. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, J.; Chang, Z.; Liu, X.; Chen, W.; Yu, Y.; Wang, X.; Dong, Q.; Ye, Y.; Zhang, X. Listeria monocytogenes Contamination Characteristics in Two Ready-to-Eat Meat Plants From 2019 to 2020 in Shanghai. Front. Microbiol. 2021, 12, 729114. [Google Scholar] [CrossRef] [PubMed]
- Muhterem-Uyar, M.; Dalmasso, M.; Bolocan, A.S.; Hernandez, M.; Kapetanakou, A.E.; Kuchta, T.; Manios, S.G.; Melero, B.; Minarovičová, J.; Nicolau, A.I.; et al. Environmental sampling for Listeria monocytogenes control in food processing facilities reveals three contamination scenarios. Food Control 2015, 51, 94–107. [Google Scholar] [CrossRef]
- Voglauer, E.M.; Zwirzitz, B.; Thalguter, S.; Selberherr, E.; Wagner, M.; Rychli, K. Biofilms in Water Hoses of a Meat Processing Environment Harbor Complex Microbial Communities. Front. Microbiol. 2022, 13, 832213. [Google Scholar] [CrossRef] [PubMed]
- Leong, D.; NicAogáin, K.; Luque-Sastre, L.; McManamon, O.; Hunt, K.; Alvarez-Ordóñez, A.; Scollard, J.; Schmalenberger, A.; Fanning, S.; O’Bryne, C.; et al. A 3-year multi-food study of the presence and persistence of Listeria monocytogenes in 54 small food businesses in Ireland. Int. J. Food Microbiol. 2017, 249, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Leatherhead Food Research. Six Steps to Control Listeria. 2018. Available online: https://www.leatherheadfood.com/files/2018/08/Six-steps-to-control-Listeria-in-foods_3.pdf (accessed on 25 April 2022).
- Duze, S.T.; Marimani, M.; Patel, M. Tolerance of Listeria monocytogenes to biocides used in food processing environments. Food Microbiol. 2021, 97, 103758. [Google Scholar] [CrossRef]
- Ibusquiza, P.S.; Herrera, J.J.R.; Cabo, M.L. Resistance to benzalkonium chloride, peracetic acid and nisin during formation of mature biofilms by Listeria monocytogenes. Food Microbiol. 2011, 28, 418–425. [Google Scholar] [CrossRef]
- Bansal, M.; Nannapaneni, R.; Sharma, C.S.; Kiess, A. Listeria monocytogenes Response to Sublethal Chlorine Induced Oxidative Stress on Homologous and Heterologous Stress Adaptation. Front. Microbiol. 2018, 9, 2050. [Google Scholar] [CrossRef]
- Minarovičová, J.; Véghová, A.; Mikulášová, M.; Chovanová, R.; Šoltýs, K.; Drahovská, H.; Kaclíková, E. Benzalkonium chloride tolerance of Listeria monocytogenes strains isolated from a meat processing facility is related to presence of plasmid-borne bcrABC cassette. Antonie Van Leeuwenhoek 2018, 111, 1913–1923. [Google Scholar] [CrossRef]
- Ulusoy, B.H.; Chirkena, K. Two perspectives of Listeria monocytogenes hazards in dairy products: The prevalence and the antibiotic resistance. Food Qual. Saf. 2019, 3, 233–241. [Google Scholar] [CrossRef]
- Atuonwu, J.C.; Leadley, C.; Bosman, A.; Tassou, S.A. High-pressure processing, microwave, ohmic, and conventional thermal pasteurization: Quality aspects and energy economics. J. Food Process Eng. 2019, 43, e13328. [Google Scholar] [CrossRef]
- Lado, B.H.; Yousef, A.E. Selection and Identification of a Listeria monocytogenes Target Strain for Pulsed Electric Field Process Optimization. Appl. Environ. Microbiol. 2003, 69, 2223–2229. [Google Scholar] [CrossRef] [PubMed]
- Nicholas, R.; Dunton, P.; Tatham, A.; Fielding, L. The effect of ozone and open air factor on surface-attached and biofilm environmental Listeria monocytogenes. J. Appl. Microbiol. 2013, 115, 555–564. [Google Scholar] [CrossRef]
- Public Health England. Guidelines for Assessing the Microbiological Safety of Ready-to-Eat Foods Placed on the Market. 2009. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/363146/Guidelines_for_assessing_the_microbiological_safety_of_ready-to-eat_foods_on_the_market.pdf (accessed on 25 April 2022).
- Legislation.gov. Food Safety Act 1990. 2022. Available online: https://www.legislation.gov.uk/ukpga/1990/16/contents (accessed on 25 April 2022).
- 17728:2015; Microbiology of the Food Chain. Sampling Techniques for Microbiological Analysis of Food and Feed Samples. ISO: Geneva, Switzerland, 2015.
- 18593:2018; Microbiology of the Food Chain. Horizontal Methods for Surface Sampling. ISO: Geneva, Switzerland, 2018.
- 11290-1:2017; Microbiology of the food Chain—Horizontal Method for the Detection and Enumeration of Listeria monocytogenes and of Listeria spp.—Part 1: Detection Method. ISO: Geneva, Switzerland, 2017.
- 11290-2:2017; Microbiology of the food Chain—Horizontal Method for the Detection and Enumeration of Listeria monocytogenes and of Listeria spp.—Part 2: Enumeration Method. ISO: Geneva, Switzerland, 2017.
- Gasanov, U.; Hughers, D.; Hansbro, P.M. Methods for the isolation and identification of Listeria spp. and Listeria monocytogenes: A review. FEMS Microbiol. Rev. 2005, 29, 851–875. [Google Scholar] [CrossRef] [PubMed]
- Beumer, R.R.; te Giffel, M.C.; Anthonie, S.V.R.; Cox, L.J. The effect of acriflavine and nalidixic acid on the growth of Listeria spp. in enrichment media. Food Microbiol. 1996, 13, 137–148. [Google Scholar] [CrossRef]
- Bannenberg, J.W.; Tempelaars, M.H.; Zwietering, M.H.; Abee, T.; den Besten, H.M.W. Heterogeneity in single-cell outgrowth of Listeria monocytogenes in half Fraser enrichment broth is affected by strain variability and physiological state. Food. Res. Int. 2021, 150, 110783. [Google Scholar] [CrossRef]
- Ottaviani, F.; Ottaviani, M.; Agosti, M. Differential agar medium for Listeria monocytogenes. Ind Aliment. 1997, 36, 888–895. [Google Scholar]
- Stessl, B.; Luf, W.; Wagner, M.; Schoder, D. Performance testing of six chromogenic ALOA-type media for the detection of Listeria monocytogenes. J Appl. Microbiol. 2009, 106, 651–659. [Google Scholar] [CrossRef]
- Duffy, G.; Walsh, D.; Sheridan, J.J.; Logue, C.M.; Harrington, D.; Blair, I.S.; McDowell, D.A. Comparison of selective and non-selective enrichment media in the detection of Listeria monocytogenes from meat containing Listeria innocua. J. Appl. Microbiol. 2001, 90, 994–999. [Google Scholar] [CrossRef]
- Pinto, M.; Burri, S.; Mena, C.; Almeida, G.; Carneiro, L.; Teixeira, P.; Gibbs, P.A. Comparison of Oxford Agar, PALCAM and Listeria monocytogenes Blood Agar for the recovery of L. monocytogenes from foods and environmental samples. Food Control. 2001, 12, 511–514. [Google Scholar] [CrossRef]
- Greenwood, M.; Willis, C.; Doswell, P.; Allen, G.; Pathak, K. Evaluation of chromogenic media for the detection of Listeria species in food. J. Appl. Microbiol. 2005, 99, 1340–1345. [Google Scholar] [CrossRef]
- Park, S.; Chang, P.; Ryu, S.; Kang, D. Development of a novel selective and differential medium for the isolation of Listeria monocytogenes. Appl. Environ. Microbiol. 2014, 80, 1020–1025. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cummins, A.J.; Fielding, A.K.; Mclauchlin, J. Listeria ivanovii infection in a patient with AIDS. J. Infect. 1994, 28, 89–91. [Google Scholar] [CrossRef]
- Bolocan, A.S.; Pennone, V.; O’Connor, P.M.; Coffey, A.; Nicolau, A.I.; McAuliffe, O.; Jordan, K. Inhibition of Listeria monocytogenes biofilms by bacteriocin-producing bacteria isolated from mushroom substrate. J. Appl. Microbiol. 2016, 122, 279–293. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, V.B.; Destro, M.T. Listeria monocytogenes Serotype 1/2b and 4b Isolates from Human Clinical Cases and Foods Show Differences in Tolerance to Refrigeration and Salt Stress. J. Food Prot. 2014, 77, 1519–1526. [Google Scholar] [CrossRef]
- Maury, M.M.; Tsai, Y.; Charlier, C.; Touchon, M.; Chenal-Francisque, V.; Leclercq, A.; Criscuolo, A.; Gaultier, C.; Roussel, S.; Brisabois, A.; et al. Uncovering Listeria monocytogenes hypervirulence by harnessing its biodiversity. Nat. Genet. 2016, 48, 308–313. [Google Scholar] [CrossRef]
- Hingston, P.; Chen, J.; Dhillon, B.K.; Laing, C.; Bertelli, C.; Gannon, V.; Tasara, T.; Allen, K.; Brinkman, F.S.L.; Hansen, L.T.; et al. Genotypes Associated with Listeria monocytogenes Isolates Displaying Impaired or Enhanced Tolerances to Cold, Salt, Acid, or Desiccation Stress. Front. Microbiol. 2017, 8, 369. [Google Scholar] [CrossRef]
- Holah, J.T.; Taylor, J.H.; Dawson, D.J.; Hall, K.E. Biocide use in the food industry and the disinfectant resistance of persistent strains of Listeria monocytogenes and Escherichia coli. J. Appl. Microbiol. 2002, 92, 111S–120S. [Google Scholar] [CrossRef]
- Møretrø, T.; Schirmer, B.C.T.; Heir, E.; Fagerlund, A.; Hjemli, P.; Langsrud, S. Tolerance to quaternary ammonium compound disinfectants may enhance growth of Listeria monocytogenes in the food industry. Int. J. Food Microbiol. 2017, 241, 215–224. [Google Scholar] [CrossRef]
- Roedel, A.; Dieckmann, R.; Brendeback, H.; Hammerl, J.A.; Kleta, S.; Noll, M.; Al Dahouk, S.; Vincze, S. Biocide-Tolerant Listeria monocytogenes Isolates from German Food Production Plants Do Not Show Cross-Resistance to Clinically Relevant Antibiotics. Appl. Environ. Microbiol. 2019, 85, e01253. [Google Scholar] [CrossRef]
- Rodríguez-López, P.; Rodríguez-Herrera, J.J.; Vázquez-Sánchez, D.; Cabo, M.L. Current Knowledge on Listeria monocytogenes Biofilms in Food-Related Environments: Incidence, Resistance to Biocides, Ecology and Biocontrol. Foods 2018, 7, 85. [Google Scholar] [CrossRef]
- Dutta, V.; Elhanafi, D.; Kathariou, S. Conservation and Distribution of the Benzalkonium Chloride Resistance Cassette bcrABC in Listeria monocytogenes. Appl. Environ. Microbiol. 2013, 79, 6067–6074. [Google Scholar] [CrossRef]
- Parsons, C.; Lee, S.; Jayeola, V.; Kathariou, S. Novel Cadmium Resistance Determinant in Listeria monocytogenes. Appl. Environ. Microbiol. 2017, 83, e02580-16. [Google Scholar] [CrossRef] [PubMed]
- Ratani, S.S.; Siletzky, R.M.; Dutta, V.; Yildirim, S.; Osborne, J.A.; Lin, W.; Hitchins, A.D.; Ward, T.J.; Kathariou, S. Heavy metal and disinfectant resistance of Listeria monocytogenes from foods and food processing plants. Appl. Environ. Microbiol. 2012, 78, 6938–6945. [Google Scholar] [CrossRef]
- To, M.S.; Favrin, S.; Romanova, N.; Griffiths, M.W. Postadaptational resistance to benzalkonium chloride and subsequent physicochemical modifications of Listeria monocytogenes. Appl. Environ. Microbiol. 2002, 68, 5258–5264. [Google Scholar] [CrossRef] [PubMed]
- Romanova, N.A.; Wolffs, P.F.G.; Brovko, L.Y.; Griffiths, M.W. Role of Efflux Pumps in Adaptation and Resistance of Listeria monocytogenes to Benzalkonium Chloride. Appl. Environ. Microbiol. 2006, 72, 3498–3503. [Google Scholar] [CrossRef] [PubMed]
- Robbins, J.B.; Fisher, C.W.; Moltz, A.G.; Martin, S.E. Elimination of Listeria monocytogenes biofilms by ozone, chlorine, and hydrogen peroxide. J. Food Prot. 2005, 68, 494–498. [Google Scholar] [CrossRef]
- Andrade, J.C.; João, A.L.; Alonso, C.S.; Barreto, A.S.; Henriques, A.R. Genetic Subtyping, Biofilm-Forming Ability and Biocide Susceptibility of Listeria monocytogenes Strains Isolated from a Ready-to-Eat Food Industry. Antibiotics 2020, 9, 416. [Google Scholar] [CrossRef]
- Ramamurthy, T.; Ghosh, A.; Pazhani, G.P.; Shinoda, S. Current Perspectives on Viable but Non-Culturable (VBNC) Pathogenic Bacteria. Front. Public Health 2014, 2, 103. [Google Scholar] [CrossRef]
- Ferro, S.; Amorico, T.; Deo, P. Role of food sanitising treatments in inducing the ‘viable but nonculturable’ state of microorganisms. Food Control. 2018, 91, 321–329. [Google Scholar] [CrossRef]
- Noll, M.; Trunzer, K.; Vondran, A.; Vincze, S.; Dieckmann, R.; Al Dahouk, S.; Gold, C. Benzalkonium Chloride Induces a VBNC State in Listeria monocytogenes. Microorganisms 2020, 8, 184. [Google Scholar] [CrossRef]
- Zolfaghari, M.; Rezaei, M.; Mobarez, A.M.; Moghaddam, M.F.; Hosseini, H.; Khezri, M. Virulence genes expression in viable but non-culturable state of Listeria monocytogenes in fish meat. Food Sci. Technol. Int. 2020, 26, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Lindbäck, T.; Rottenberg, M.E.; Roche, S.M.; Liv, M.R. The ability to enter into an avirulent viable but non-culturable (VBNC) form is widespread among Listeria monocytogenes isolates from salmon, patients and environment. Vet. Res. 2010, 41, 8. [Google Scholar] [CrossRef] [PubMed]
- Cappelier, J.M.; Besnard, V.; Roche, S.; Garrec, N.; Zundel, E.; Velge, P.; Federighi, M. Avirulence of viable but non-culturable Listeria monocytogenes cells demonstrated by in vitro and in vivo models. Vet. Res. 2005, 36, 589–599. [Google Scholar] [CrossRef]
- Montanari, C.; Tabanelli, G.; Barbieri, F.; Mora, D.; Duncan, R.; Gardini, F.; Arioli, S. Listeria monocytogenes sensitivity to antimicrobial treatments depends on cell origin. Sci. Rep. 2022, 12, 3676. [Google Scholar] [CrossRef] [PubMed]
- Troxler, R.; Von Graevenitz, A.; Funke, F.; Wiedemann, B.; Stock, I. Natural antibiotic susceptibility of Listeria species: L. grayi, L. innocua, L. ivanovii, L. monocytogenes, L. seeligeri and L. welshimeri strains. Clin. Microbiol. Infect. 2000, 6, 525–535. [Google Scholar] [CrossRef]
- Noll, M.; Kleta, S.; Al Dahouk, S. Antibiotic susceptibility of 259 Listeria monocytogenes strains isolated from food, food-processing plants and human samples in Germany. J. Infect. Public Health 2018, 11, 572–577. [Google Scholar] [CrossRef]
- Andriyanov, P.A.; Zhurilov, P.A.; Liskova, E.A.; Karpova, T.I.; Sokolova, E.V.; Yushina, Y.K.; Zaiko, E.V.; Bataeva, D.S.; Voronina, O.L.; Psareva, E.K.; et al. Antimicrobial Resistance of Listeria monocytogenes Strains Isolated from Humans, Animals, and Food Products in Russia in 1950–1980, 2000–2005, and 2018–2021. Antibiotics 2021, 10, 1206. [Google Scholar] [CrossRef]
- Panera-Martínez, S.; Rodríguez-Melcón, C.; Serrano-Galán, V.; Alonso-Calleja, C.; Capita, R. Prevalence, quantification and antibiotic resistance of Listeria monocytogenes in poultry preparations. Food Control. 2022, 135, 108608. [Google Scholar] [CrossRef]
- Walsh, D.; Duffy, G.; Sheridan, J.J.; Blair, I.S.; McDowell, D.A. Antibiotic resistance among Listeria, including Listeria monocytogenes, in retail foods. J. Appl. Microbiol. 2001, 90, 517–522. [Google Scholar] [CrossRef]
- Olaimat, A.N.; Al-Holy, M.A.; Shahbaz, H.M.; Al-Nabulsi, A.A.; Ghoush, M.H.A.; Osaili, T.M.; Ayyash, M.M.; Holley, R.A. Emergence of Antibiotic Resistance in Listeria monocytogenes Isolated from Food Products: A Comprehensive Review. Compr. Rev. Food Sci. Food Saf. 2018, 17, 1277–1292. [Google Scholar] [CrossRef]
- Baquero, F.; Lanza, V.F.; Duval, M.; Coque, T.M. Ecogenetics of antibiotic resistance in Listeria monocytogenes. Mol. Microbiol. 2020, 113, 570–579. [Google Scholar] [CrossRef]
- Godreuil, S.; Galimand, M.; Gerbaud, G.; Jacquet, C.; Courvalin, P. Efflux Pump Lde Is Associated with Fluoroquinolone Resistance in Listeria monocytogenes. Antimicrob. Agents Chemother. 2003, 47, 704–708. [Google Scholar] [CrossRef]
- Jiang, X.; Yu, T.; Xu, P.; Xu, X.; Ji, S.; Gao, W.; Shi, L. Role of Efflux Pumps in the in vitro Development of Ciprofloxacin Resistance in Listeria monocytogenes. Front. Microbiol. 2018, 9, 2350. [Google Scholar] [CrossRef] [PubMed]
- Rakic-Martinez, M.; Drevets, D.A.; Dutta, V.; Katic, V.; Kathariou, S. Listeria monocytogenes strains selected on ciprofloxacin or the disinfectant benzalkonium chloride exhibit reduced susceptibility to ciprofloxacin, gentamicin, benzalkonium chloride, and other toxic compounds. Appl. Environ. Microbiol. 2011, 77, 8714–8721. [Google Scholar] [CrossRef] [PubMed]
- Kremer, P.H.C.; Lees, J.A.; Koopmans, M.M.; Ferwerda, B.; Arends, A.W.M.; Feller, M.M.; Schipper, K.; Seron, M.V.; van der Ende, A.; Brouwer, M.C.; et al. Benzalkonium tolerance genes and outcome in Listeria monocytogenes meningitis. Clin. Microbiol. Infect. 2017, 23, 265.e1–265.e7. [Google Scholar] [CrossRef]
- Law, J.W.; Mutalib, N.A.; Chan, K.; Lee, L. An insight into the isolation, enumeration, and molecular detection of Listeria monocytogenes in food. Front. Microbiol. 2015, 6, 1227. [Google Scholar] [CrossRef] [PubMed]
- Zarei, M.; Maktabi, S.; Ghorbanpour, M. Prevalence of Listeria monocytogenes, Vibrio parahaemolyticus, Staphylococcus aureus, and Salmonella spp. in seafood products using multiplex polymerase chain reaction. Foodborne Pathog. Dis. 2012, 9, 108–112. [Google Scholar] [CrossRef]
- Ryu, J.; Park, S.H.; Yeom, Y.S.; Shrivastav, A.; Lee, S.; Kim, Y.; Kim, H. Simultaneous detection of Listeria species isolated from meat processed foods using multiplex PCR. Food Control 2013, 32, 659–664. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, L.; Jin, L.; Jin, M.; Shen, Z.; An, S.; Chao, F.; Li, J. Development and application of an oligonucleotide microarray for the detection of food-borne bacterial pathogens. Appl. Microbiol. Biotechnol. 2007, 76, 225–233. [Google Scholar] [CrossRef]
- Kim, S.; Kim, J.; Han, W.; Jung, B.; Chuong, P.; Joo, H.; Ba, H.; Son, W.; Jee, Y.; Yoon, B.; et al. Development and Evaluation of an Immunochromatographic Assay for Screening Listeria spp. in Pork and Milk. Food Sci. Biotechnol. 2007, 16, 515–519. [Google Scholar]
- Wideman, N.E.; Oliver, J.D.; Crandall, P.G.; Jarvis, N.A. Detection and Potential Virulence of Viable but Non-Culturable (VBNC) Listeria monocytogenes: A Review. Microorganisms 2021, 9, 194. [Google Scholar] [CrossRef] [PubMed]
- Desneux, J.; Biscuit, A.; Picard, S.; Pourcher, A. Fate of Viable but Non-culturable Listeria monocytogenes in Pig Manure Microcosms. Front. Microbiol. 2016, 7, 245. [Google Scholar] [CrossRef] [PubMed]
- Løvdal, T.; Hovda, M.B.; Björkblom, B.; Møller, S.G. Propidium monoazide combined with real-time quantitative PCR underestimates heat-killed Listeria innocua. J. Mirobiol. Methods 2011, 85, 164–169. [Google Scholar] [CrossRef] [PubMed]
- Morey, A.; McKee, S.R.; Dickson, J.S.; Singh, M. Efficacy of ultraviolet light exposure against survival of Listeria monocytogenes on conveyor belts. Foodborne Pathog. Dis. 2010, 7, 737–740. [Google Scholar] [CrossRef] [PubMed]
- Kawacka, I.; Olejnik-Schmidt, A.; Schmidt, M.; Sip, A. Effectiveness of Phage-Based Inhibition of Listeria monocytogenes in Food Products and Food Processing Environments. Microorganisms 2020, 8, 1764. [Google Scholar] [CrossRef] [PubMed]
- Vongkamjan, K.; Roof, S.; Stasiewicz, M.J.; Wiedmann, M. Persistent Listeria monocytogenes subtypes isolated from a smoked fish processing facility included both phage susceptible and resistant isolates. Food Microbiol. 2013, 35, 38–48. [Google Scholar] [CrossRef]
- Panebianco, F.; Rubiola, S.; Chiesa, F.; Civera, T.; Ciccio, P.A.D. Effect of Gaseous Ozone on Listeria monocytogenes Planktonic Cells and Biofilm: An In Vitro Study. Foods 2021, 10, 1484. [Google Scholar] [CrossRef]
- Shu, X.; Singh, M.; Karampudi, N.B.R.; Bridges, D.F.; Kitazumi, A.; Wu, V.C.H.; De Los Reyes, B.G. Responses of Escherichia coli and Listeria monocytogenes to ozone treatment on non-host tomato: Efficacy of intervention and evidence of induced acclimation. PLoS ONE 2021, 16, e0256324. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Macleod, J.; Beeton, M.L.; Blaxland, J. An Exploration of Listeria monocytogenes, Its Influence on the UK Food Industry and Future Public Health Strategies. Foods 2022, 11, 1456. https://doi.org/10.3390/foods11101456
Macleod J, Beeton ML, Blaxland J. An Exploration of Listeria monocytogenes, Its Influence on the UK Food Industry and Future Public Health Strategies. Foods. 2022; 11(10):1456. https://doi.org/10.3390/foods11101456
Chicago/Turabian StyleMacleod, Joshua, Michael L. Beeton, and James Blaxland. 2022. "An Exploration of Listeria monocytogenes, Its Influence on the UK Food Industry and Future Public Health Strategies" Foods 11, no. 10: 1456. https://doi.org/10.3390/foods11101456
APA StyleMacleod, J., Beeton, M. L., & Blaxland, J. (2022). An Exploration of Listeria monocytogenes, Its Influence on the UK Food Industry and Future Public Health Strategies. Foods, 11(10), 1456. https://doi.org/10.3390/foods11101456





