Nutritional Viewpoints on Eggs and Cholesterol
Abstract
1. Introduction
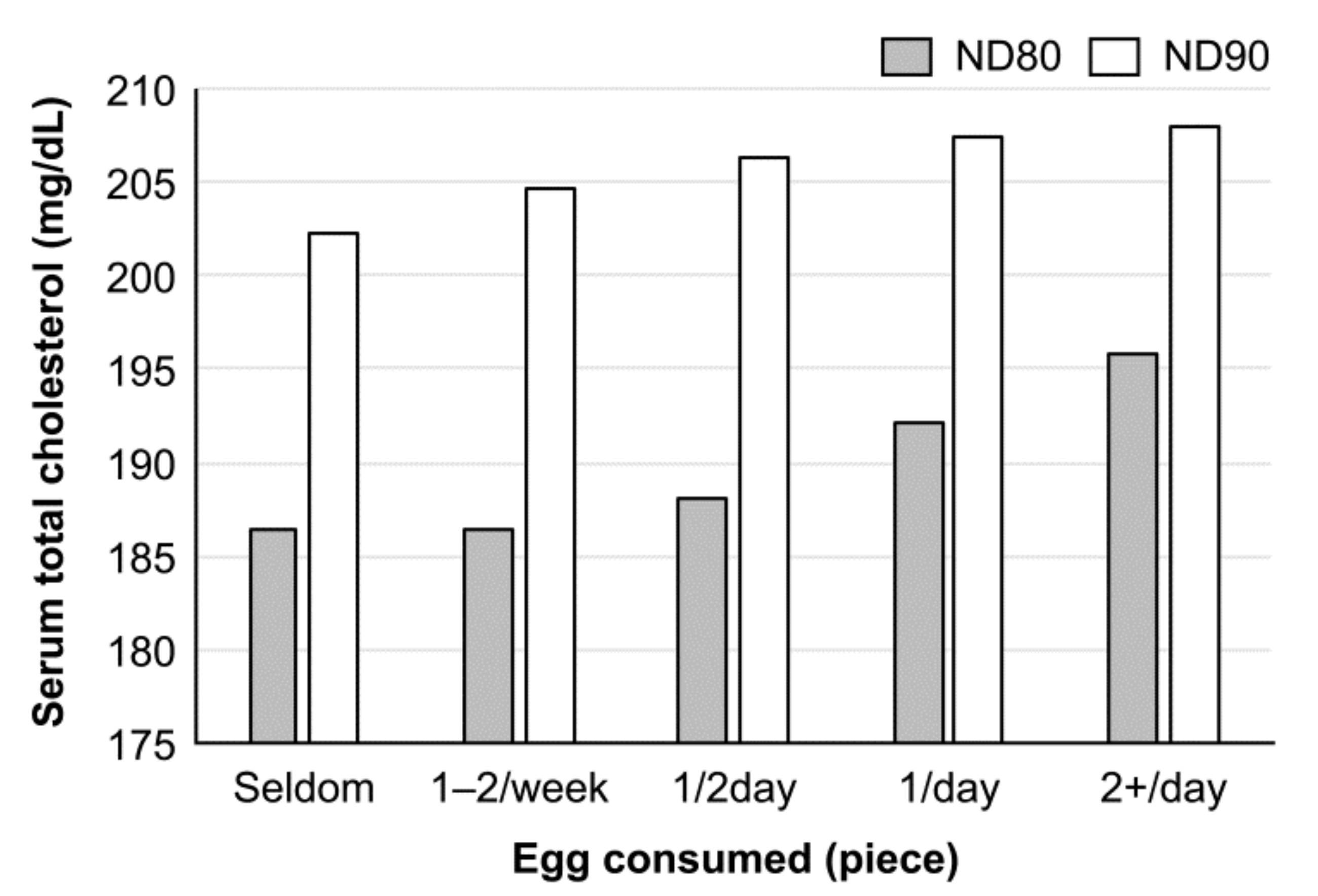
2. Egg Consumption and Response of Blood Cholesterol in Japanese
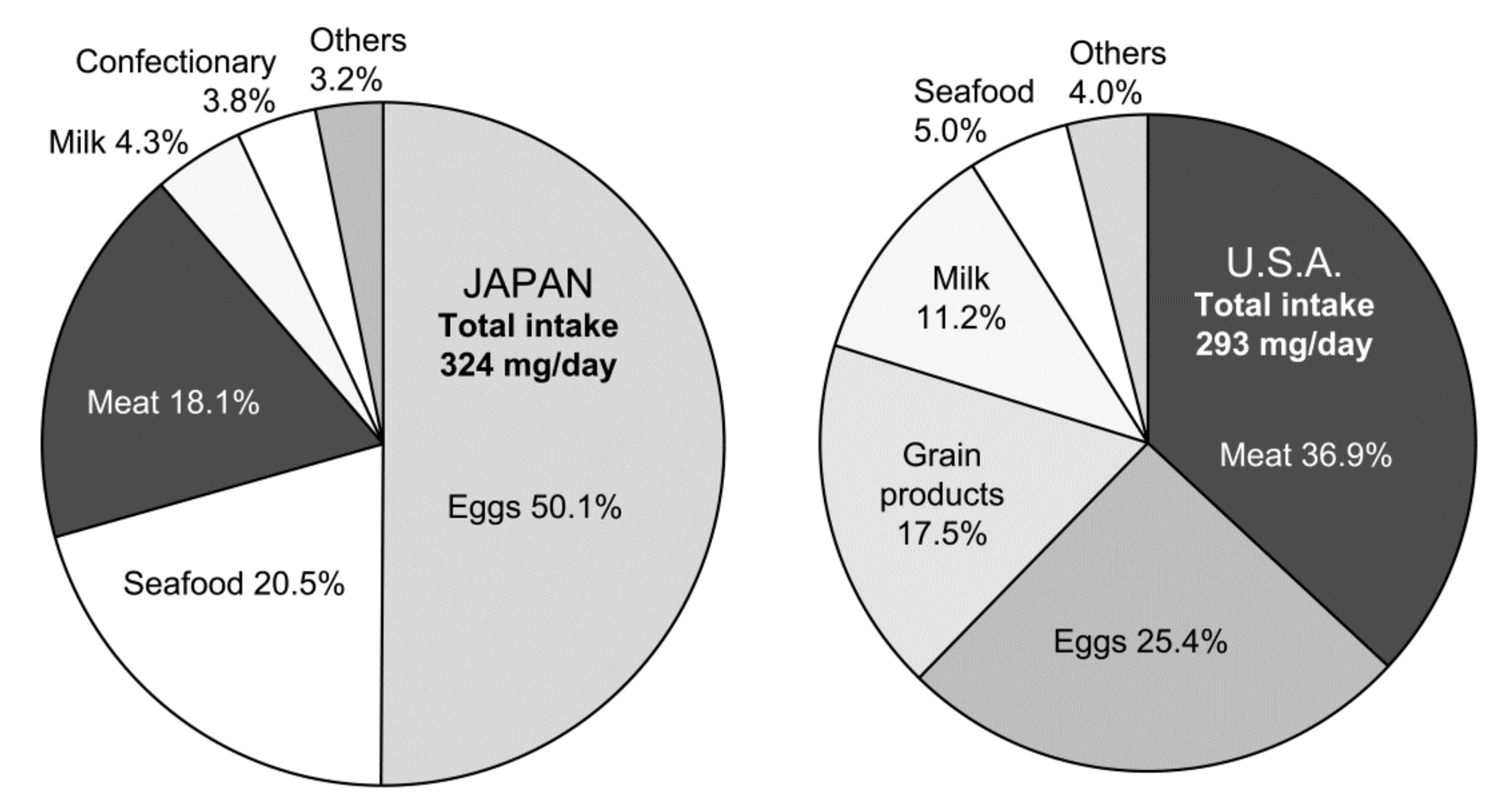
3. Egg and Health: Comparison between Japan and the US
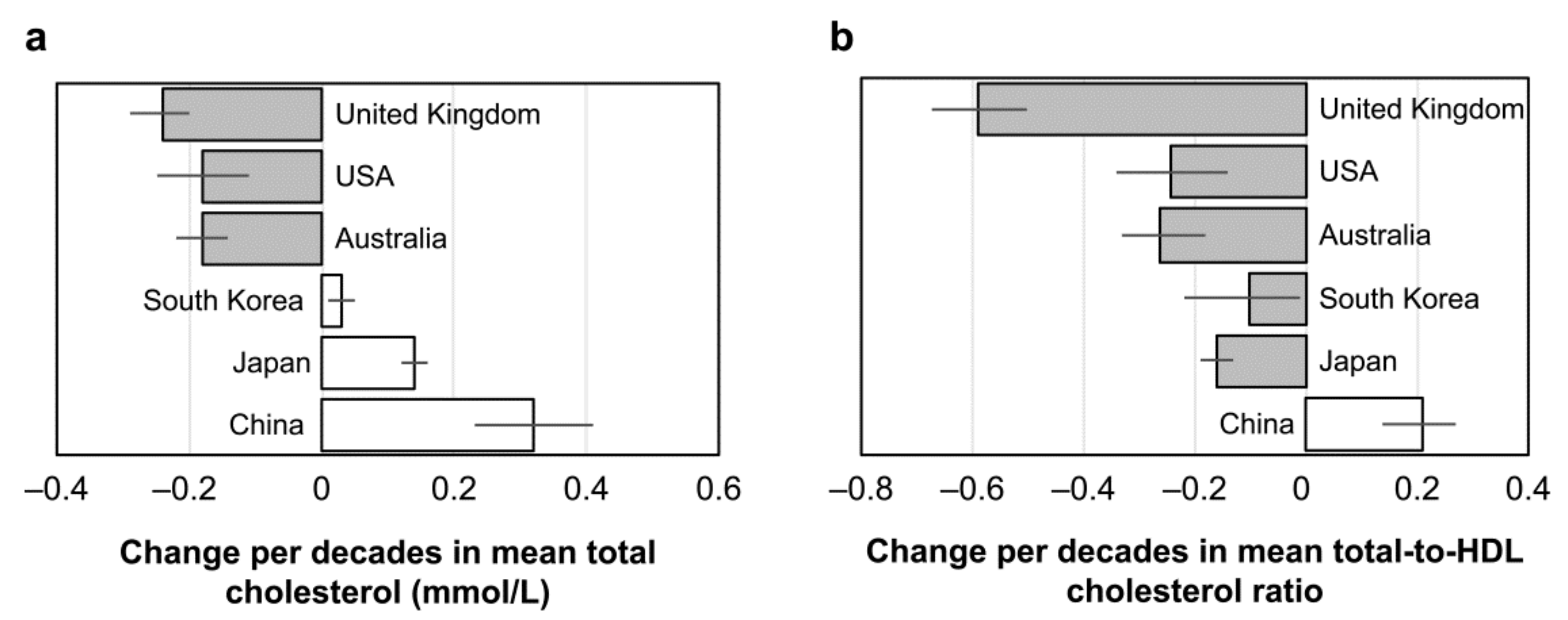
4. Egg Consumption and Health: Global View
4.1. Cardiovascular Diseases
4.2. Stroke and Hypertension
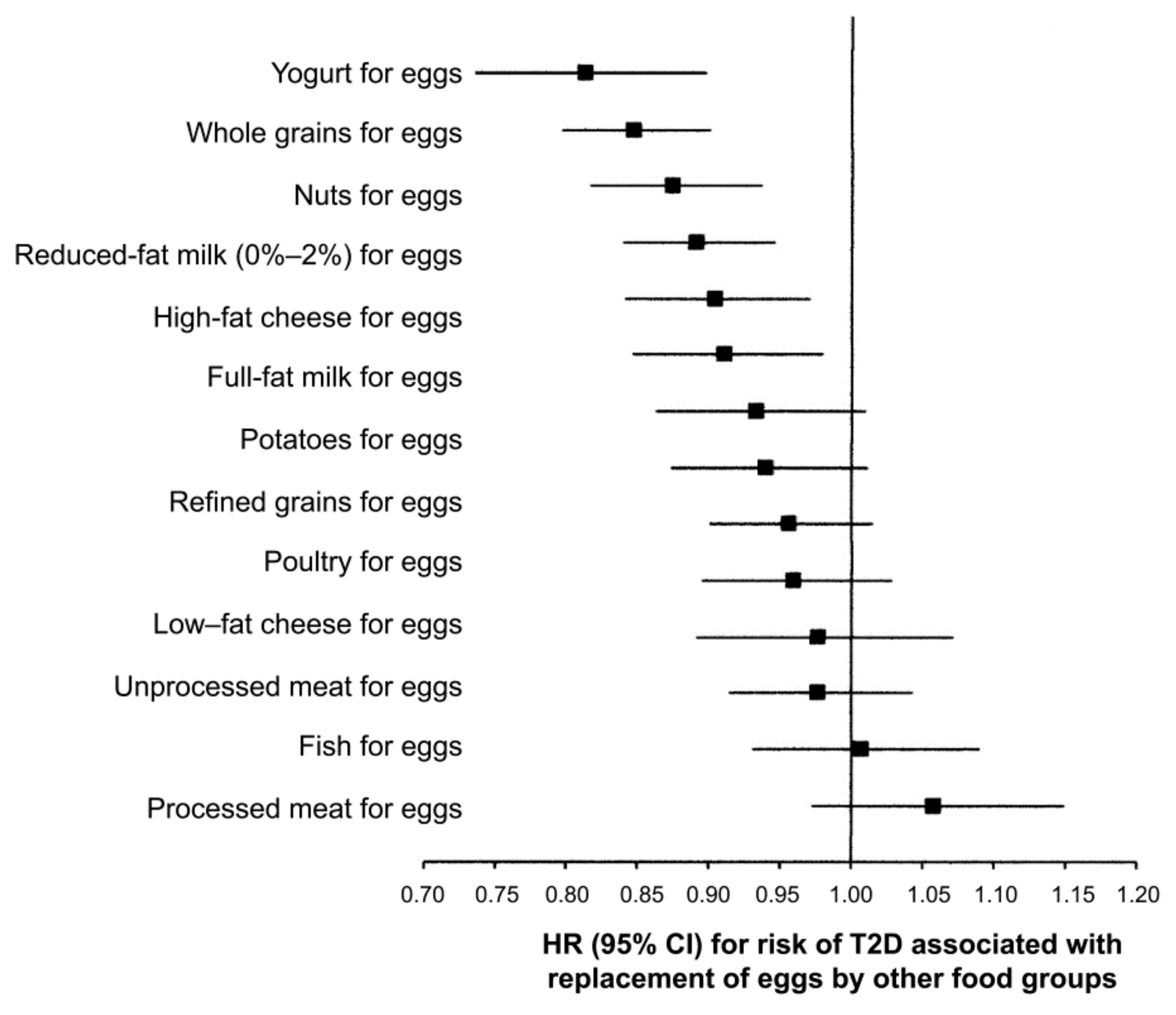
5. Egg and Type 2 Diabetes Mellitus
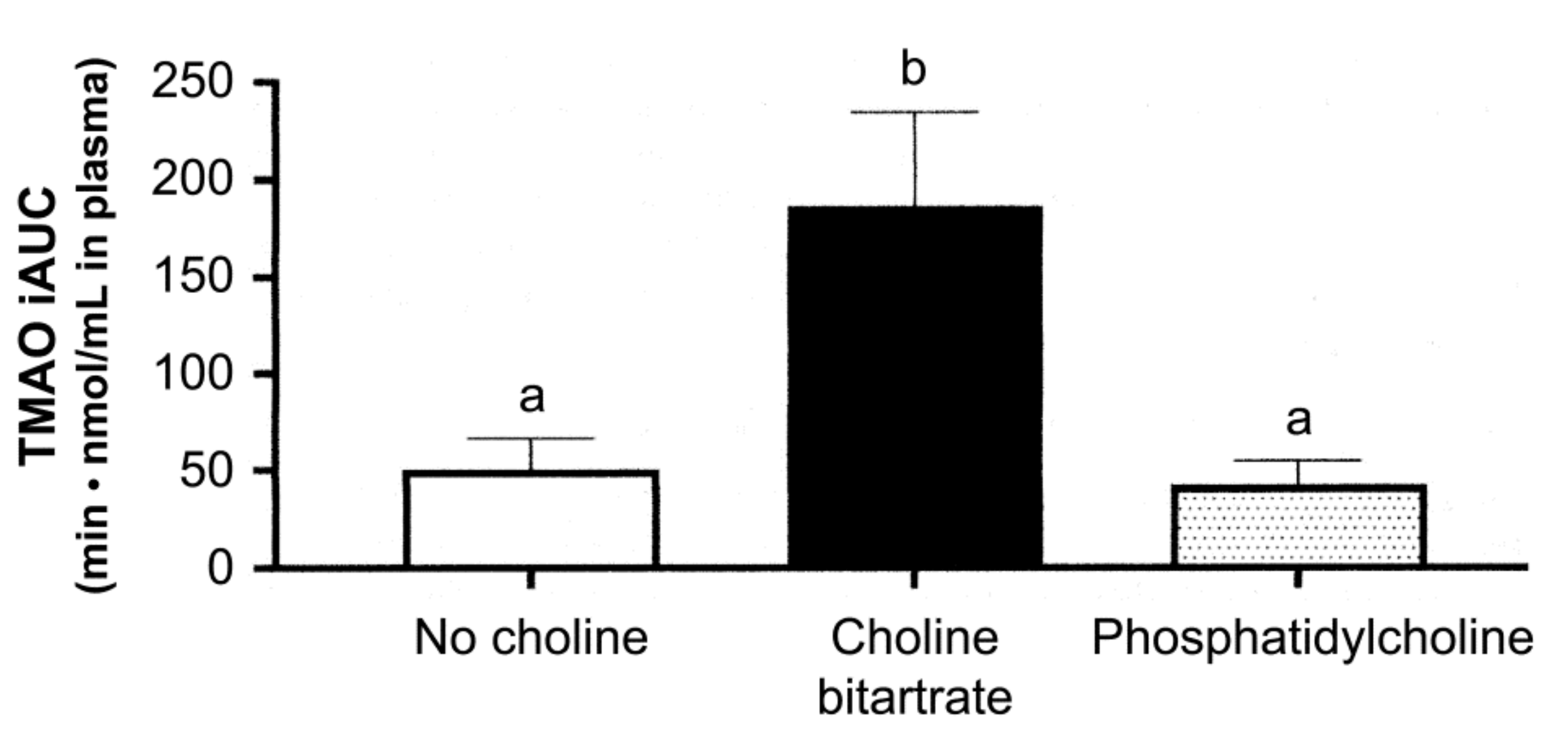
6. Debates around Trimethylamine Oxide (TMAO)
7. Cholesterol-lowering Components in Egg
8. Rebuttal to “Innocent Theory” of Egg Consumption
9. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- U.S. Department of Agriculture; U.S. Department of Health and Human Services. Dietary Guidelines for Americans 2020–2025, 9 ed.; USDA Publication: Washington, DC, USA, 2020. Available online: https://www.dietaryguidelines.gov/sites/default/files/2020-12/Dietary_Guidelines_for _Americans_2020-2025.pdf (accessed on 24 February 2021).
- Dietary Reference Intakes for Japanese; The Ministry of Health, Labour and Welfare: Tokyo, Japan, 2020. Available online: https://www.mlw.go.jp/content/10904750/000586553.pdf (accessed on 24 February 2021).
- Sabaté, J.; Burkholder-Cooley, N.M.; Segovia-Siapco, G.; Oda, K.; Wells, B.; Orlich, M.J.; Fraser, G.F. Unscrambling the relations of egg and meat consumption with type 2 diabetes risk. Am. J. Clin. Nutr. 2018, 108, 1121–1128. [Google Scholar] [CrossRef]
- Drouin-Chartier, J.-P.; Schwab, A.L.; Chen, S.; Li, Y.; Sacks, F.M.; Bernard, R.; Manson, J.E.; Willet, W.C.; Stampfer, M.J.; Hu, F.B.; et al. Egg consumption and risk of type 2 diabetes: Findings from 3 large US cohort studies of men and women and a systematic review and meta-analysis of prospective cohort studies. Am. J. Clin. Nutr. 2020, 112, 619–630. [Google Scholar] [CrossRef] [PubMed]
- Matsuoka, R.; Shirouchi, B.; Kawamura, S.; Baba, S.; Shiratake, S.; Nagata, K.; Imaizumi, K.; Sato, M. Dietary egg white protein inhibits lymphatic lipid transport in thoracic lymph duct-cannulated rats. J. Agric. Food Chem. 2014, 62, 10694–10700. [Google Scholar] [CrossRef] [PubMed]
- Matsuoka, R.; Usuda, M.; Masuda, Y.; Kunou, M.; Utsunomiya, K. Lactic-fermented egg white reduced serum cholesterol concentrations in mildly hypercholesterolemic Japanese men: A double-blind parallel-arm design. Lipids Health Dis. 2017, 16, 101. [Google Scholar] [CrossRef]
- Lee, Y.; Han, C.Y.; Bae, M.; Park, Y.K.; Lee, J.-Y. Egg phospholipids exert an inhibitory effect on intestinal cholesterol absorption in mice. Nutr. Res. Pract. 2019, 13, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Ferraro, R.A.; Fischer, N.M.; Xun, F.H.; Michos, E.D. Nutrition and physical activity recommendations from the United States and European cardiovascular guidelines: A comparative review. Curr. Opin. Cardiol. 2020, 35, 508–516. [Google Scholar] [CrossRef] [PubMed]
- Geiker, N.R.W.; Larsen, M.L.; Dyerberg, J.; Stender, S.; Astrup, A. Egg consumption, cardiovascular diseases and type 2 diabetes. Eur. J. Clin. Nutr. 2018, 72, 44–56. [Google Scholar] [CrossRef] [PubMed]
- McNamara, D.J. The fifty year rehabilitation of the egg. Nutrients 2015, 7, 8716–8722. [Google Scholar] [CrossRef]
- Eckel, R.H. Eggs and beyond: Is dietary cholesterol no longer important? Am. J. Clin. Nutr. 2015, 102, 235–236. [Google Scholar] [CrossRef] [PubMed]
- Griffin, B.A. Eggs: Good or bad? Proc. Nutr. Soc. 2016, 75, 259–264. [Google Scholar] [CrossRef]
- Xu, Z.; McClure, S.T.; Appel, L.J. Dietary cholesterol intake and sources among U.S. adults: Results from National Health and Nutrition Examination Surveys (NHANES), 2001-2014. Nutrients 2018, 10, 771. [Google Scholar] [CrossRef] [PubMed]
- Sugaiyama, D.; Turin, T.C.; Yeasmin, F.; Rumana, N.; Watanabe, M.; Higashiyama, A.; Takegami, M.; Kokubo, Y.; Okamura, T.; Miyamoto, Y. Hypercholesterolemia and lifetime risk of coronary heart disease in the general Japanese population: Results from the Suita cohort study. J. Atheroscler. Thromb. 2020, 27, 60–70. [Google Scholar] [CrossRef] [PubMed]
- NCD Risk Factor Collaboration (NCD-RisC). Repositioning of the global epicentre of non-optimal cholesterol. Nature 2020, 582, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Broad Leib, E.M.; Shapiro, M.; Chan, A.; Negoweitti, N.; Borzi, L.; Etessami, S.; Hartmann, T.; Hoover, A.; Jeong, J.; Loucks, S.; et al. Doctoring our Diet, Policy Tools to Include Nutrition in U.S. Medical Training. In Food Law and Policy Clinic; Harvard Law School: Boston, MA, USA, 2019; pp. 1–35. [Google Scholar]
- Crowley, J.; Ball, L.; Hiddink, G.J. Nutrition in medical education: A systematic review. Lancet Planet Health 2019, 3, e379–e389. [Google Scholar] [CrossRef]
- Macaninch, E.; Bucker, L.; Amin, P.; Broadley, I.; Crocombe, D.; Herath, D.; Jaffee, A.; Carter, H.; Golubric, R.; Rajput-Ray, M.; et al. Time for nutrition in medical education. BMJ Nutr. Prev. Health 2020, 3, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.; Okamura, T.; Tamaki, S.; Kadowaki, T.; Hayakawa, T.; Kita, Y.; Okayama, A.; Ueshima, H. NIPPON DATA80 Research Group. Egg consumption, serum cholesterol, and cause-specific and all-cause mortality: The National Integrated Project for Prospective Observation of Non-communicable Disease and Its Trends in the Aged, 1980 (NIPPON DATA80). Am. J. Clin. Nutr. 2004, 80, 58–63. [Google Scholar] [CrossRef]
- Nakamura, Y.; Okamura, T.; Kita, Y.; Okuda, N.; Kadota, A.; Miura, K.; Okayama, A.; Ueshima, H. NIPPON DATA90 Research Group. Re-evaluation of the associations of egg intake with serum total cholesterol and cause-specific and total mortality in Japanese women. Eur. J. Clin. Nutr. 2018, 72, 841–847. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y. NIPPON DATA90-How do you read a successive change? Lipid 2011, 22, 20–25. (in Japanese). [Google Scholar]
- Okami, Y.; Ueshima, H.; Nakamura, Y.; Kondo, K.; Kadota, A.; Okuda, N.; Okamura, T.; Miura, K. NIPPON DATA80/90 and NIPPON DATA2010 Research Group. Time-related changes in relationships between the Keys score, dietary lipids, and serum total cholesterol in Japan-NIPPON DATA80/90/2010. Circ. J. 2018, 83, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.; Iso, H.; Kita, Y.; Ueshima, H.; Okada, K.; Konishi, M.; Inoue, M.; Tsugane, S. Egg consumption, serum total cholesterol concentrations and coronary heart disease incidence: Japan Public Health Center-based prospective study. Br. J. Nutr. 2006, 96, 921–928. [Google Scholar] [CrossRef] [PubMed]
- Okami, Y.; Ueshima, H.; Nakamura, Y.; Okuda, N.; Nakagawa, H.; Sakata, K.; Saitoh, S.; Okayama, A.; Yoshita, K.; Choudhury, S.R.; et al. The relationship of dietary cholesterol with serum low-density lipoprotein cholesterol and confounding by reverse causality: The INTERLIPID Study. J. Atheroscler. Thromb. 2019, 26, 170–182. [Google Scholar] [CrossRef]
- Homma, Y.; Kobayashi, T.; Yamaguchi, H.; Ozawa, H.; Homma, K.; Ishikawa, K. Apolipoprotein-E phenotype and basal activity of low-density lipoprotein receptor are independent of changes in plasma lipoprotein subfractions after cholesterol ingestion in Japanese subjects. Nutrition 2001, 17, 310–314. [Google Scholar] [CrossRef]
- Blesso, C.N.; Fernandez, M.L. Dietary cholesterol, serum lipids, and heart disease: Are eggs working for or against you? Nutrients 2018, 10, 426. [Google Scholar] [CrossRef] [PubMed]
- Kishimoto, Y.; Taguchi, C.; Suzuki-Sugihara, N.; Saita, E.; Usuda, M.; Wang, W.; Masuda, Y.; Kondo, K. The effect of the consumption of egg on serum lipids and antioxidant status in healthy subjects. J. Nutr. Sci. Vitaminol. 2016, 62, 361–365. [Google Scholar] [CrossRef] [PubMed]
- Taguchi, C.; Kishimoto, Y.; Suzuki-Sugihara, N.; Saita, E.; Usuda, M.; Wang, W.; Masuda, Y.; Kondo, K. Regular egg consumption at breakfast by Japanese women university students improves daily nutrient intakes: Open-labeled observations. Asia Pac. J. Clin. Nutr. 2018, 27, 359–365. [Google Scholar] [CrossRef] [PubMed]
- Kishimoto, Y.; Taguchi, C.; Saita, E.; Suzuki-Sugihara, N.; Nishiyama, H.; Wang, W.; Masuda, Y.; Kondo, K. Additional consumption of one egg per day increases serum lutein plus zeaxanthin concentration and lowers oxidized low-density lipoprotein in moderately hypercholesterolemic males. Food Res. Int. 2017, 99, 944–949. [Google Scholar] [CrossRef]
- NCD Risk Factor Collaboration (NCD-RisC). National trends in total cholesterol obscure heterogeneous changes in HDL and non-HDL cholesterol and total-to-HDL cholesterol ratio: A pooled analysis of 458 population-based studies in Asian and Western countries. Int. J. Epidemiol. 2020, 49, 173–192. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.-J.; Lee, H.S.; Chang, H.-J.; Koh, S.-B.; Lee, J.-W. Association of dietary lipid intake with low-density lipoprotein cholesterol levels: Analysis of two independent population-based studies. Eur. J. Nutr. 2020, 59, 2557–2567. [Google Scholar] [CrossRef]
- Saito, I.; Yamagishi, K.; Kokubo, Y.; Yatsuya, H.; Iso, H.; Sawada, N.; Inoue, M.; Tsugane, S. Non-high-density lipoprotein cholesterol and risk of stroke subtypes and coronary heart disease: The Japan Public Health Center-based prospective (JPHC) study. J. Atheroscler. Thromb. 2020, 27, 363–374. [Google Scholar] [CrossRef]
- Terry, A.L.; Herrick, K.A.; Afful, J.; Ahluwalia, N. Seafood Consumption in the United States, 2013–2016; US Department of Health & Human Services: Washington, DC, USA, 2018; pp. 1–8.
- Thompson, M.; Hein, N.; Hanson, C.; Smith, L.M.; Anderson-Berry, A.; Richter, C.K.; Bisselou, K.S.; Appiah, A.K.; Kris-Etherton, P.; Skulas-Ray, A.C.; et al. Omega-3 fatty acid intake by age, gender, and pregnancy status in the United States: National Health and Nutrition Examination Survey 2003–2014. Nutrients 2019, 11, 177. [Google Scholar] [CrossRef]
- Stark, K.D.; Van Elswyk, M.E.; Higgins, M.R.; Weatherford, C.A.; Salem, N., Jr. Global survey of the omega-3 fatty acids, docosahexaenoic acid and eicosapentaenoic acid in the blood stream of healthy adults. Prog. Lipid Res. 2016, 63, 132–152. [Google Scholar] [CrossRef] [PubMed]
- Jackson, K.H.; Polreis, J.M.; Tintle, N.L.; Kris-Etherton, P.M.; Harris, W.S. Association of reported fish intake and supplementation status with the omega-3 index. Prostagl. Leukot. Essent. Fatty Acids 2019, 142, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Liao, L.M.; Weinstein, S.J.; Sinha, R.; Graubard, B.I.; Albanes, D. Association between plant and animal protein intake and overall and cause-specific mortality. JAMA Internal Med. 2020, 180, 1173–1184. [Google Scholar] [CrossRef] [PubMed]
- Crimarco, A.; Springfield, S.; Petlura, C.; Streaty, T.; Cunanan, K.; Lee, J.; Fielding-Singh, P.; Carter, M.M.; Topf, M.A.; Wastyk, H.C.; et al. A randomized crossover trial on the effect of plant-based compared with animal-based meat on trimethylamine-N-oxide and cardiovascular disease risk factors in generally healthy adults: Study with Appetizing Plantfood-Meat Eating Alternative Trail (SWAP-MEAT). Am. J. Clin. Nutr. 2020, 112, 1188–1199. [Google Scholar] [CrossRef] [PubMed]
- Kurihara, A.; Okamura, T.; Sugiyama, D.; Higashiyama, A.; Watanabe, M.; Okuda, N.; Kadota, A.; Miyagawa, N.; Fujiyoshi, A.; Yoshida, K.; et al. Vegetable protein intake was inversely associated with cardiovascular mortality in a 15-year follow-up study of the general Japanese population. J. Atheroscler. Thromb. 2019, 26, 198–206. [Google Scholar] [CrossRef]
- Tong, T.Y.N.; Appleby, P.N.; Key, T.J.; Dahm, C.C.; Overvad, K.; Olsen, A.; Tjønneland, A.; Katzke, V.; Kühn, T.; Boeing, H.; et al. The associations of major foods and fibre with risks of ischaemic and haemorrhagic stroke: A prospective study of 418 329 participants in the EPIC cohort across nine European countries. Eur. Heart J. 2020, 41, 2632–2640. [Google Scholar] [CrossRef]
- Zhong, V.W.; Horn, L.V.; Cornelis, M.C.; Wilkins, J.T.; Ning, H.; Carnethon, M.R.; Greenland, P.; Mentz, R.J.; Tucker, K.L.; Zhao, L.; et al. Associations of dietary cholesterol or egg consumption with incident cardiovascular disease and mortality. JAMA 2019, 321, 1081–1095. [Google Scholar] [CrossRef]
- Carson, J.A.S.; Lichtenstein, A.H.; Anderson, C.A.M.; Appel, L.J.; Kris-Etherton, P.M.; Meyer, K.A.; Peterson, K.; Polonsky, T.; Van Horn, L. Dietary cholesterol and cardiovascular risk. A science advisory from the American Heart Association. Circulation 2020, 141, e39–e53. [Google Scholar] [CrossRef] [PubMed]
- Deng, C.; Lu, Q.; Gong, B.; Li, L.; Chang, L.; Fu, L.; Zhao, Y. Stroke and food groups: An overview of systematic reviews and meta-analysis. Public Health Nutr. 2018, 21, 766–776. [Google Scholar] [CrossRef] [PubMed]
- Bechthold, A.; Boeing, H.; Schwedhelm, C.; Hoffmann, G.; Knüppel, A.; Iqbal, K.; De Henauw, S.; Michels, N.; Devleesschauwer, B.; Schlesinger, S.; et al. Food groups and risk of coronary heart disease, stroke and heart failure: A systematic review and dose-response meta-analysis of prospective studies. Crit. Rev. Food Sci. Nutr. 2019, 59, 1071–1090. [Google Scholar] [CrossRef]
- Sacks, F.M.; Lichtenstein, A.H.; Wu, J.H.Y.; Appel, L.J.; Creager, M.A.; Kris-Etherton, P.M.; Miller, M.; Rimm, E.B.; Rudel, L.L.; Robinson, J.G.; et al. Dietary fats and cardiovascular disease. A presidential advisory from the American Heart Association. Circulation 2017, 136, e1–e23. [Google Scholar] [CrossRef] [PubMed]
- Kris-Etherton, P.M.; Krauss, R.K. Public health guidelines should recommend reducing saturated fat consumption as much as possible: YES. Am. J. Clin. Nutr. 2020, 112, 13–18. [Google Scholar] [CrossRef]
- Hooper, L.; Martin, N.; Jimoh, O.F.; Kirk, C.; Foster, E.; Abdelhamid, A.S. Reduction in saturated fat intake for cardiovascular disease (review). Cochrane Database Syst. Rev. 2020, 8, CD011737. [Google Scholar] [CrossRef] [PubMed]
- Heileson, J.L. Dietary saturated fat and heart disease: A narrative review. Nutr. Rev. 2020, 78, 474–485. [Google Scholar] [CrossRef]
- Astrup, A.; Magkos, F.; Bier, D.M.; Brenna, J.T.; de Oliveira Otto, M.C.; Hill, J.O.; King, J.C.; Mente, A.; Ordovas, J.M.; Volek, J.S.; et al. Saturated fats and health: A reassessment and proposal for food-based recommendations: JACC State-of-the-Art review. J. Am. Coll. Cardiol. 2020, 76, 844–857. [Google Scholar] [CrossRef]
- Duarte, C.; Boccardi, V.; Andrade, P.A.; Souza Lopes, A.C.; Jacques, P.F. Diary versus other saturated fats source and cardiometabolic risk markers: Systematic review of randomized controlled trials. Crit. Rev. Food Sci. Nutr. 2021, 61, 450–461. [Google Scholar] [CrossRef]
- Kim, J.E.; Campbell, W.W. Dietary cholesterol contained in whole eggs is not well absorbed and dose not acutely affect plasma total cholesterol concentration in men and women: Results from 2 randomized controlled crossover studies. Nutrients 2018, 10, 1272. [Google Scholar] [CrossRef] [PubMed]
- Lemos, B.S.; Medina-Vera, I.; Blesso, C.N.; Fernandez, M.L. Intake of 3 eggs per day when compared to a choline bitartrate supplement, downregulates cholesterol synthesis without changing the LDL/HDL ratio. Nutrients 2018, 10, 258. [Google Scholar] [CrossRef]
- Kuang, H.; Yang, F.; Zhang, Y.; Wang, T.; Chen, G. The impact of egg nutrient composition and its consumption on cholesterol homeostasis. Cholesterol 2018, 6303810. [Google Scholar] [CrossRef]
- Lopez-Jimenez, F. Egg: Are They Good or Bad for My Cholesterol? Mayo Clinic Health Letter, 9 January 2020; Available online: https:/www.mayoclinic.org/diseases-conditions/high-bloood-choleserol/expert-answers/cholesterol/faq-20058468?p=1. Cited from MedlinePlus Twitter Update, Cholesterol Also Called: Hypercholesterolemia, Hyperlipidemia, Hyperlipoproteinemia. Available online: https://medlineplus.gov/cholesterol.html. (accessed on 24 February 2021). [Google Scholar]
- Emamat, H.; Totmaj, A.S.; Tangestani, H.; Hekmadoost, A. The effect of egg and its derivatives on vascular function: A systematic review of interventional studies. Clin. Nutr. ESPEN 2020, 39, 15–21. [Google Scholar] [CrossRef]
- Lv, Y.; Kraus, V.B.; Gao, X.; Yin, Z.; Zhou, J.; Mao, C.; Duan, J.; Zeng, Y.; Brasher, M.S.; Shi, W.; et al. Higher dietary diversity scores and protein-rich food consumption were associated with lower risk of all-cause mortality in the oldest old. Clin. Biochem. 2020, 39, 2246–2254. [Google Scholar] [CrossRef]
- Liu, R.; Li, D.; Ding, L.; Liu, Y.; Sun, X.; Xue, F. Egg intake in extremely undernourished Chinese women during reproductive age and subsequent nonfatal perimenopausal coronary events. Asia Pac. J. Clin. Nutr. 2019, 28, 584–592. [Google Scholar] [CrossRef]
- Papanikolaou, Y.; Fulgoni III, V.L. Eggs are cost-efficient in delivering several shortfall nutrients in the American diet: A cost-analysis in children and adults. Nutrients 2020, 12, 2406. [Google Scholar] [CrossRef] [PubMed]
- Dehghan, M.; Mente, A.; Rangarajan, S.; Mohan, V.; Lear, S.; Swaminathan, S.; Wielgosz, A.; Pamela Seron, P.; Avezum, A.; Lopez-Jaramillo, P.; et al. Association of egg intake with blood lipids, cardiovascular disease, and mortality in 177,000 people in 50 countries. Am. J. Clin. Nutr. 2020, 111, 795–803. [Google Scholar] [CrossRef]
- Djoussé, L.; Ho, Y.L.; Nguyen, Y.-M.T.; Quaden, R.M.; Gagnon, D.R.; Gaziano, J.M.; Cho, K. Egg consumption and risk of coronary artery disease in the Million Veteran Program. Clin. Nutr. 2020, 39, 2842–2847. [Google Scholar] [CrossRef] [PubMed]
- Duyuler, S.; Özbek, K.; Balci, K.G.; Çelebi, Ö.Ö.; Duyuler, P.T. Egg consumption and cardiovascular health: How many eggs a day keep the doctor away. Angiology 2020, 71, 667. [Google Scholar] [CrossRef] [PubMed]
- Mazidi, M.; Katsiki, N.; Mikhailidis, D.P.; Pencina, M.J.; Banach, M. Egg consumption and risk of total and cause-specific mortality: An individual-based cohort study and pooling prospective studies on behalf of the Lipid and Blood Pressure Meta-analysis Collaboration (LBPMC) Group. J. Am. Coll. Nutr. 2019, 38, 552–563. [Google Scholar] [CrossRef]
- Mah, E.; Oliver Chen, C.-Y.; Liska, D.J. The effect of egg consumption on cardiometabolic health outcomes: And umbrella review. Public Health Nutr. 2020, 23, 935–955. [Google Scholar] [CrossRef] [PubMed]
- Aljohi, H.; Dopler-Nelson, M.; Cifuentes, M.; Wilson, T.A. The consumption of 12 eggs per week for 1 year does not alter fasting serum markers of cardiovascular disease in older adults with early macular degeneration. J. Nutr Intermed. Metab. 2019, 15, 35–41. [Google Scholar] [CrossRef]
- Rouhani, M.H.; Rashidi-Pourfard, N.; Salehi-Abargouei, A.; Karimi, M.; Haghighatdoost, F. Effect of egg consumption on blood lipids: A systematic review and meta-analysis of randomized clinical trials. J. Am. Coll. Nutr. 2018, 37, 99–110. [Google Scholar] [CrossRef]
- Krittanawong, C.; Narashimhan, B.; Wang, Z.; Hassan Virk, H.U.; Farrell, A.M.; Zhang, H.J.; Tang, W.H.W. Association between egg consumption and risk of cardiovascular outcomes: A systematic review and meta-analysis. Am. J. Med. 2021, 134, 76–82. [Google Scholar] [CrossRef]
- Marventano, S.; Godos, J.; Tieri, M.; Ghelfi, F.; Titta, L.; Lafranconi, A.; Gambera, A.; Alonzo, E.; Sciacca, S.; Buscemi, S.; et al. Egg consumption and human health: An umbrella review of observational studies. Int. J. Food Sci. Nutr. 2020, 71, 325–331. [Google Scholar] [CrossRef]
- Abdollahi, A.M.; Virtanen, H.E.K.; Voutilainen, S.; Kurl, S.; Tuomainen, T.-P.; Salonen, J.T.; Virtanen, J.K. Egg consumption, cholesterol intake, and risk of incident stroke in men: The Kuopio Ishaemic Heart Disease Risk Factor Study. Am. J. Clin. Nutr. 2019, 110, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.X.; Wong, C.H.; Kim, J.E. Impact of whole egg intake on blood pressure, lipids and lipoproteins in middle-aged and older population: A systematic review and meta-analysis of randomized controlled trials. Nutr. Metab. Cardiovasc. Dis. 2019, 29, 653–664. [Google Scholar] [CrossRef]
- Song, J.; Jiang, X.; Cao, Y.; Juan, J.; Wu, T.; Hu, Y. Interaction between ATP-binding cassette A1 (ABCA1) variant and egg consumption for the risk of ischemic stroke and carotid atherosclerosis: A family-based study in the Chinese population. J. Athreroscler. Thromb. 2019, 26, 835–845. [Google Scholar] [CrossRef] [PubMed]
- Yu, G.; Fu, H.; Huang, W.; Zhang, N.; Deng, D.; Li, G.; Lei, H. A dietary patter of higher fish, egg, milk, nut, vegetable and fruit, and lower salt intake correlates with the prevalence and control of hypertension. Am. J. Hypertens. 2018, 31, 679–686. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, D.-Z. Red meat, poultry, and egg consumption with the risk of hypertension: A meta-analysis of prospective cohort studies. J. Hum. Hypertens. 2018, 32, 507–517. [Google Scholar] [CrossRef]
- Margerison, C.; Riddell, L.J.; McNaughton, S.A.; Nowson, C.A. Associations between dietary patterns and blood pressure in a sample of Australian adults. Nutr. J. 2020, 19, 5. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, C.-J.; Madika, A.-L.; Bonnet, F.; Fagherazzi, G.; Lajous, M.; Boutron-Ruault, M.-C. Cholesterol and egg intakes, and risk of hypertension in large prospective cohort of French women. Nutrients 2020, 12, 1350. [Google Scholar] [CrossRef] [PubMed]
- Li, M.-Y.; Chen, J.-H.; Chen, C.; Kang, Y.-N. Association between egg consumption and cholesterol concentration: A systematic review and meta-analysis of randomized controlled trials. Nutrients 2020, 12, 1995. [Google Scholar] [CrossRef] [PubMed]
- MINERVA. Eggs and risk of heart diseases and other stories. BMJ 2018, 363, k4600. [Google Scholar] [CrossRef]
- Soliman, G.A. Dietary cholesterol and the lack of evidence in cardiovascular disease. Nutrients 2018, 10, 780. [Google Scholar] [CrossRef] [PubMed]
- Vazqiez-Rutz, Z.; De la Funente-Arrillaga, C.; Bes-Rastrollo, M.; Zazpe, I.; Santiago, S.; Razquin, C.; Toledo, E.; Martinez-Gonzalez, M.Á. Egg consumption and dyslipidemia in a Mediterranean cohort. Nutr. Hosp. 2018, 35, 153–161. [Google Scholar] [CrossRef]
- Guo, J.; Hobbs, D.A.; Cockroft, J.R.; Elwood, P.C.; Pickering, J.E.; Lovegrove, J.A.; Givens, D.I. Association between egg consumption and cardiovascular disease events, diabetes and all-cause mortality. Eur. J. Nutr. 2018, 57, 2943–2952. [Google Scholar] [CrossRef]
- Melough, M.M.; Chung, S.-J.; Fernandez, M.L.; Chun, O.K. Association of eggs with dietary nutrient adequacy and cardiovascular risk factors in US adults. Public Health Nutr. 2019, 22, 2033–2042. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Lam, T.H.; Jiang, C.Q.; Zhang, W.S.; Zhu, F.; Jin, Y.L.; Woo, J.; Cheng, K.K.; Thomas, G.N. Egg consumption and the risk of cardiovascular diseases and all-cause mortality: Guangshou Biobank Cohort Study and meta-analysis. Eur. J. Nutr. 2019, 58, 785–796. [Google Scholar] [CrossRef]
- Kalogeropoulos, A.P.; Papanastasiou, C.A. Egg consumption: To eat or not to eat? J. Thrac. Dis. 2019, 11, 2185–2187. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, P.; Jiao, J.; Wu, F.; Mao, L.; Zhang, Y. Egg and egg-sourced cholesterol consumption in relation to mortality: Findings from population-based nationwide cohort. Clin. Nutr. 2020, 39, 3520–3527. [Google Scholar] [CrossRef] [PubMed]
- Xia, P.-F.; Pan, X.-F.; Chen, C.; Wang, Y.; Ye, Y.; Pan, A. Dietary intakes of eggs and cholesterol in relation to all-cause and heart disease mortality: A prospective cohort study. J. Am. Heart Assoc. 2020, 9, e015743. [Google Scholar] [CrossRef]
- Godos, J.; Micek, A.; Brzostek, T.; Toledo, E.; Iacoviello, L.; Attrup, A.; Franco, O.H.; Galvano, F.; Martinez-Gonzalez, M.A.; Grosso, G. Egg consumption and cardiovascular risk: A dose-response meta-analysis of prospective cohort studies. Eur. J. Nutr. 2020. [Google Scholar] [CrossRef] [PubMed]
- Chrysant, S.G.; Chrysant, G.S. The debate over egg consumption and incident cardiovascular disease. Cardiol. Rev. 2020, 18. [Google Scholar] [CrossRef] [PubMed]
- Takagi, H.; Hari, Y.; Nakashima, K.; Kuno, T.; Ando, T. Egg consumption and coronary artery disease: A nice knockdown argument. Angiology 2020, 71, 589–601. [Google Scholar] [CrossRef]
- Drouin-Chartier, J.-P.; Chen, S.; Li, Y.; Schwab, A.L.; Stampfer, M.J.; Sacks, F.M.; Rosner, B.; Willett, W.C.; Hu, F.B.; Bhupathiraju, S.N. Egg consumption and risk of cardiovascular disease: Three large prospective US cohort studies, systematic review, and updated meta-analysis. BMJ 2020, 368, m513. [Google Scholar] [CrossRef]
- Qin, C.; Lv, J.; Guo, Y.; Bian, Z.; Si, J.; Yang, L.; Chen, Y.; Zhou, Y.; Zhang, H.; Liu, J.; et al. Associations of egg consumption with cardiovascular disease in a cohort study of 0.5 million Chinese adults. Heart 2018, 104, 1756–1763. [Google Scholar] [CrossRef]
- DiMarco, D.M.J.; Fernandez, M.L. Differences in response to egg-derived dietary cholesterol result in distinct lipoprotein profiles while plasma concentrations of carotenoids and choline are not affected in a young healthy population. J. Agr. Food Res. 2019, 1, 100014. [Google Scholar] [CrossRef]
- Tang, H.; Cao, X.; Yang, X.; Zhang, Y. Egg consumption and stroke risk: A systematic review and dose-response meta-analysis of prospective studies. Front. Nutr. 2020, 7, 153. [Google Scholar] [CrossRef]
- Chen, G.-C.; Chen, L.-H.; Mossavar-Rahmani, Y.; Kamensky, V.; Shadvyab, A.H.; Haring, B.; Wild, R.A.; Silver, B.; Kuller, L.H.; Sun, Y.; et al. Dietary cholesterol and egg intake in relation to incident cardiovascular disease and all-cause and cause-specific mortality in postmenopausal women. Am. J. Clin. Nutr. 2020, npaa353. [Google Scholar] [CrossRef] [PubMed]
- Yamagishi, K.; Iso, H.; Kokubo, Y.; Saito, I.; Yatsuya, H.; Ishihara, J.; Inoue, M.; Tsugane, S. JPHC Study Group. Dietary intake of saturated fatty acids and incident stroke and coronary heart disease in Japanese communities: The JPHC Study. Eur. Heart J. 2013, 34, 1225–1232. [Google Scholar] [CrossRef] [PubMed]
- Muto, M.; Ezaki, O. High dietary saturated fat is associated with a low risk of intracerebral hemorrhage and ischemic stroke in Japanese but not in Non-Japanese: A review and meta-analysis of prospective cohort studies. J. Atheroscler. Thromb. 2018, 25, 375–392. [Google Scholar] [CrossRef]
- Yamagishi, K.; Iso, H.; Tsugane, S. Saturated fat intake and cardiovascular disease in Japanese population. J. Atheroscler. Thromb. 2015, 22, 435–439. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Kim, J. Egg consumption is associated with a lower risk of type 2 diabetes in middle -aged and older men. Nutr. Res. Pract. 2018, 12, 396–405. [Google Scholar] [CrossRef]
- Djoussé, L.; Petrone, A.B.; Hickson, D.A.; Talegawkar, S.A.; Dubbert, P.M.; Taylor, H.; Tucker, K.L. Egg consumption and risk of type 2 diabetes among African Americans: The Jackson Heart Study. Clin. Nutr. 2016, 35, 679–684. [Google Scholar] [CrossRef] [PubMed]
- Tamez, M.; Vitranen, J.K.; Lajous, M. Egg consumption and risk of incident type 2 diabetes: A dose-response meta-analysis of prospective cohort studies. Br. J. Nutr. 2016, 115, 2212–2218. [Google Scholar] [CrossRef]
- Richard, C.; Cristall, L.; Fleming, E.; Lewis, E.D.; Ricupero, M.; Jacobs, R.L.; Field, C.J. Impact of egg consumption on cardiovascular risk factors in individuals with type 2 diabetes and at risk for developing diabetes: A systematic review of randomized nutritional intervention studies. Can. J. Diabetes 2017, 41, 453–463. [Google Scholar] [CrossRef]
- Fuller, N.R.; Sainsbury, A.; Caterson, I.D.; Denyer, G.; Fong, M.; Gerofi, J.; Leung, C.; Lau, N.S.; Williams, K.H.; Januszewski, A.S.; et al. Effect of a high-egg diet on cardiometabolic risk factors in people with type 2 diabetes: The Diabetes and Egg (DIABEGG) Study-randomized weight-loss and follow-up phase. Am. J. Clin. Nutr. 2018, 107, 921–931. [Google Scholar] [CrossRef] [PubMed]
- Ni, L.-P.; Du, L.-Y.; Huang, Y.-Q.; Zhou, J.-Y. Egg consumption and risk of type 2 diabetes mellitus in middle and elderly Chinese population. An observational study. Medicine 2020, 99, e19752. [Google Scholar] [CrossRef]
- Kurotani, K.; Nanri, A.; Goto, A.; Mizoue, T.; Noda, M.; Oba, S.; Sawada, N.; Tsugane, S. Japan Public Health Center-based Prospective Study Group. Cholesterol and egg intakes and the risk of type 2 diabetes: The Japan Public Health Center-based Prospective Study. Br. J. Nutr. 2014, 112, 1636–1643. [Google Scholar] [CrossRef] [PubMed]
- Pourafshar, S.; Akhavan, N.S.; George, K.S.; Foley, E.M.; Jonson, S.A.; Keshavarz, B.; Navaei, N.; Davoudi, A.; Clark, E.A.; Arjmandi, B.H. Egg consumption may improve factors associated with glycemic control and insulin sensitivity in adults with pre- and type II diabetes. Food Funct. 2018, 9, 4469–4479. [Google Scholar] [CrossRef]
- Noerman, S.; Kärkkäinen, O.; Mattsson, A.; Paananen, J.; Lehtonen, M.; Nurmi, T.; Tuomainen, T.-P.; Voutilainen, S.; Hanhineva, K.; Virtanen, J.K. Metabolic profiling of high egg consumption and the associated lower risk of type 2 diabetes in middle-aged Finnish men. Mol. Nutr. Food Res. 2019, 63, e1800605. [Google Scholar] [CrossRef]
- Wang, X.; Son, M.; Meram, C.; Wu, J. Mechanism and potential of egg consumption and egg bioactive components on Type 2 diabetes. Nutrients 2019, 11, 357. [Google Scholar] [CrossRef] [PubMed]
- Petry, C.J.; Ong, K.K.; Hughes, I.A.; Acerini, C.L.; Dunger, D.B. Temporal trends in maternal food intake frequencies and associations with gestational diabetes: The Cambridge Baby Growth Study. Nutrients 2019, 11, 2822. [Google Scholar] [CrossRef] [PubMed]
- Maki, K.C.; Palacios, O.M.; Kramer, M.W.; Trivedi, R.; Dicklin, M.R.; Wilcox, M.L.; Maki, C.E. Effects of substituting eggs for high-carbohydrate breakfast foods on the cardiometabolic risk-factor profile in adults at risk for type 2 diabetes mellitus. Eur. J. Clin. Nutr. 2020, 74, 784–795. [Google Scholar] [CrossRef]
- Schulze, M.B.; Martínez-González, M.A.; Fung, T.T.; Lichtenstein, A.H.; Forouhi, N.G. Food based dietary patterns and chronic disease prevention. BMJ 2018, 361, k2396. [Google Scholar] [CrossRef]
- Jang, J.; Shin, M.-J.; Kim, O.Y.; Park, K. Longitudinal association between egg consumption and the risk of cardiovascular disease: Interaction with type 2 diabetes mellitus. Nutr. Diabetes 2018, 8, 20. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, M.; Shi, Z. Higher egg consumption associated with increased risk of diabetes in Chinese adults—China Health and Nutrition survey. Br. J. Nutr. 2020, 8, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Zheng, R.; Xu, Y.; Niu, J.; Wu, S.; Li, M.; Lu, J.; Wang, S.; Xu, Y.; Wang, W.; Bi, Y.; et al. Type 2 diabetes RCTs in mainland China: Insights from a systematic review. Lancet Diabetes Endocrinol. 2020. [Google Scholar] [CrossRef]
- Lee, J.L.; Brett, N.R.; de Zepetnek, J.O.T.; Bellissimo, N. Effects of white potatoes consumed with eggs on satiety, food intake, and glycemic response in children and adolescents. J. Am. Coll. Nutr. 2020, 39, 147–154. [Google Scholar] [CrossRef]
- NCD Risk Factor Collaboration (NCD-RisC)-Americans Working Group. Trends in cardiometabolic risk factors in the Americas between 1980 and 2014: A pooled analysis of population-based surveys. Lancet Glob. Health 2020, 8, e123–e133. [Google Scholar] [CrossRef]
- Würtz, A.M.L.; Jakobsen, M.U.; Bertoia, M.L.; Hou, T.; Schmidt, E.B.; Willett, W.C.; Overvad, K.; Sun, Q.; Manson, J.E.; Hu, F.B.; et al. Replacing the consumption of red meat with other major dietary protein sources and risk of type 2 diabetes mellitus: A prospective cohort study. Am. J. Clin. Nutr. 2020, npaa284. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Ley, S.H.; Hu, F.B. Global Aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat. Rev. Endocrinol. 2018, 14, 88–98. [Google Scholar] [CrossRef]
- Organization for Economic Co-operation, and Development; OECD Obesity Update, 2017. Available online: https://www.oecd.org/health/obesity-update.htm (accessed on 24 February 2021).
- Bastin, M.; Andreelli, F. The gut microbiota and diabetic cardiomyopathy in humans. Diabetes Metab. 2020, 46, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Croyal, M.; Saulnier, P.-J.; Aguesse, A.; Gand, E.; Ragot, S.; Roussel, R.; Halimi, J.M.; Ducrocq, G.; Cariou, B.; Montaigne, D.; et al. Plasma trimethylamine N-oxide and risk of cardiovascular events in patients with type 2 diabetes. J. Clin. Endocrinol. Metab. 2020, 105, dgaa188. [Google Scholar] [CrossRef]
- Shama, S.; Liu, W. Omega-3 fatty acids and gut microbiota: A reciprocal interaction in nonalcoholic fatty liver disease. Dig. Dis. Sci. 2020, 65, 906–910. [Google Scholar] [CrossRef]
- Yang, S.; Li, X.; Yang, F.; Zhao, R.; Pan, X.; Liang, J.; Tian, L.; Li, X.; Liu, L.; Xing, Y.; et al. Gut microbiota-dependent marker TMAO in promoting cardiovascular disease: Inflammation mechanism, clinical prognostic, and potential as a therapeutic target. Front. Pharmacol. 2019, 10, 1360. [Google Scholar] [CrossRef] [PubMed]
- Naghipour, S.; Cox, A.J.; Peart, J.N.; Du Toit, E.F.; Headrick, J.P. Trimethylamine-N-oxide: Heart of the microbiota-CVD nexus? Nutr. Res. Rev. 2020, Jul 28, 1–22. [Google Scholar] [CrossRef]
- Heianza, Y.; Ma, W.; DiDonato, J.A.; Sun, Q.; Rimm, E.B.; Hu, F.B.; Rexrode, K.M.; Manson, J.E.; Qi, L. Long-term changes in gut microbial metabolite trimethylamine N-oxide and coronary heart disease risk. J. Am. Coll. Cardiol. 2020, 75, 763–772. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, Y.; Ke, B.; Du, J. TMAO: How gut microbiota contributes to heart failure. Transl. Res. 2020, 228, 109–125. [Google Scholar] [CrossRef] [PubMed]
- Arias, N.; Arboleya, S.; Allisson, J.; Kaliszewska, A.; Higarza, S.G.; Gueimonde, M.; Arias, J.L. The relationship between choline bioavailability from diet, intestinal microbiota composition, and its modulation of human diseases. Nutrients 2020, 12, 2340. [Google Scholar] [CrossRef]
- Farhangi, M.A. Gut microbiota-dependent trimethylamine N-oxide and all-cause mortality: Findings from an updated systematic review and meta-analysis. Nutrition 2020, 78, 110856. [Google Scholar] [CrossRef] [PubMed]
- Koay, Y.C.; Chen, Y.-C.; Wali, J.A.; Luk, A.W.S.; Li, M.; Doma, H.; Reimark, R.; Zaldivia, M.T.K.; Habtom, H.T.; Frank, A.E.; et al. Plasma levels of TMAO can be increased with ‘healthy’ and ‘unhealthy’ diets and do not correlate with the extent of atherosclerosis but with plaque instability. Cardiovac. Res. 2020, cvaa094. [Google Scholar] [CrossRef]
- Spence, J.D. Trimethylamine N-Oxide: Not just red meat-egg yolk and renal function are also important. Eur. Heart J. 2019, 40, 3498. [Google Scholar] [CrossRef] [PubMed]
- Hamaya, R.; Ivey, K.L.; Lee, D.H.; Wang, M.; Li, J.; Franke, A.; Sun, Q.; Rimm, E.B. Association of diet with circulating trimethylamine-N-oxide concentration. Am. J. Clin. Nutr. 2020, 112, 1448–1455. [Google Scholar] [CrossRef]
- Miller, C.A.; Corbin, K.D.; da Costa, K.-A.; Zhang, S.; Zhao, X.; Galanko, J.A.; Blevins, T.; Bennett, B.J.; O’Connor, A.; Zeisel, S.H. Effect of egg ingestion on trimethylamine-N-oxide production in humans: A randomized, controlled, dose-response study. Am. J. Clin. Nutr. 2014, 100, 778–786. [Google Scholar] [CrossRef]
- Blesso, C.N. Egg phospholipids and cardiovascular health. Nutrients 2015, 7, 2731–2747. [Google Scholar] [CrossRef]
- Sugano, M. An aspect of physiological functions of choline: Trimethylamine-N-oxide. Oleoscience 2017, 17, 217–222. (in Japanese). [Google Scholar] [CrossRef]
- Zhu, C.; Sawrey-Kubicek, L.; Bardagjy, A.S.; Houts, H.; Tang, X.; Sacchi, R.; Randolph, J.M.; Steinberg, F.M.; Zivkovic, A.M. Whole egg consumption increases plasma choline and betaine without affecting TMAO levels or gut microbiome in overweight postmenopausal women. Nutr. Res. 2020, 78, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Nagata, C.; Wada, K.; Tamura, T.; Konishi, K.; Kawachi, T.; Tsuji, M.; Nakamura, K. Choline and betaine intakes are not associated with cardiovascular disease mortality risk in Japanese men and women. J. Nutr. 2015, 145, 1787–1792. [Google Scholar] [CrossRef]
- Shirouchi, B.; Yamanaka, R.; Tanaka, S.; Kawatou, F.; Hayashi, T.; Takayama, A.; Nakao, A.; Goromaru, R.; Iwamoto, M.; Sato, M. Quantities of phospholipid molecular classes in Japanese meals and prediction of their sources by multiple regression analysis. J. Nutr. Sci. Vitaminol. 2018, 64, 215–221. [Google Scholar] [CrossRef]
- Wallace, T.C.; Fulgoni, V.L., III. Assessment of total choline intakes in the United States. J. Am. Coll. Nutr. 2016, 35, 108–112. [Google Scholar] [CrossRef] [PubMed]
- DiMarco, D.M.; Missimer, A.; Murillo, A.G.; Lemos, B.S.; Malysheva, O.V.; Caudill, M.A.; Blesso, C.N.; Fernandez, M.L. Intake of up to 3 eggs/day increases HDL cholesterol and plasma choline while plasma trimethylamine N-oxide is unchanged in healthy population. Lipids 2017, 52, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Hagen, I.V.; Helland, A.; Bratlie, M.; Midttun, Ø.; McCann, A.; Sveir, H.; Rosenlund, G.; Mellgren, G.; Ueland, P.M.; Gudbrandson, O.A. TMAO, creatine and 1-methylhistidine in serum and urine are potential biomarkers of cod and salmon intake: A randomized clinical trial in adults with overweight or obesity. Eur. J. Nutr. 2020, 59, 2249–2259. [Google Scholar] [CrossRef]
- Yu, D.; Shu, X.O.; Rivera, E.S.; Zhang, X.; Cai, Q.; Calcutt, M.W.; Xiang, Y.-B.; Li, H.; Gao, Y.-T.; Wang, T.J.; et al. Urinary levels of trimethylamine-N-Oxide and incident coronary heart disease: A prospective investigation among urban Chinese adults. J. Am. Heart Assoc. 2019, 8, e010606. [Google Scholar] [CrossRef] [PubMed]
- Kühn, T.; Rohrmann, S.; Sookthai, D.; Johnson, T.; Katzke, V.; Kaaks, R.; von Eckardstein, A.; Müller, D. Intra-individual variation of plasma trimethylamine-N-oxide (TMAO), betaine and choline over 1 year. Clin. Chem. Lab. Med. 2017, 55, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Cho, C.E.; Aardema, N.D.J.; Bunnell, M.L.; Larson, D.P.; Aguilar, S.S.; Bergeson, J.R.; Malysheva, O.V.; Caudill, M.A.; Lefevre, M. Effect of choline forms and gut microbiota composition on trimethylamine-N-oxide response in healthy men. Nutrients 2020, 12, 2220. [Google Scholar] [CrossRef] [PubMed]
- Dannenberg, L.; Zikeli, D.; Benkhoff, M.; Ahlbrecht, S.; Kelm, M.; Levkau, B.; Polzin, A. Targeting the human microbiome and its metabolite TMAO in cardiovascular prevention and therapy. Pharmacol. Ther. 2020, 213, 107584. [Google Scholar] [CrossRef] [PubMed]
- Simó, C.; Gracia-Cañas, V. Dietary bioactive ingredients to modulate the gut metabolite-derived metabolite TMAO. New opportunities for functional food development. Food Funct. 2020, 11, 6745–6776. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Li, Q.; Jlang, H. Gut microbiota in atherosclerosis: Focus on trimethylamine N-oxide. APMIS 2020, 128, 353–366. [Google Scholar] [CrossRef]
- Yin, X.; Gibbons, H.; Rundle, M.; Frost, G.; McNulty, B.A.; Nugent, A.P.; Walton, J.; Flynn, A.; Berennan, L. The relationship between fish intake and urinary trimethylamine-N-oxide. Mol. Nutr. Food Res. 2020, 64, e1900799. [Google Scholar] [CrossRef] [PubMed]
- Papandreou, C.; Moré, M.; Bellamine, A. Trimethylamine N-oxide in relation to cardiometabolic health-cause or effect? Nutrients 2020, 12, 1330. [Google Scholar] [CrossRef] [PubMed]
- Asato, L.; Wang, M.F.; Chan, Y.C.; Yeh, S.H.; Chung, H.M.; Chung, S.Y.; Chida, S.; Uezato, T.; Suzuki, I.; Yamagata, N.; et al. Effect of egg white on serum cholesterol concentration in young women. J. Nutr. Sci. Vitaminol. 1996, 42, 87–96. [Google Scholar] [CrossRef]
- Oda, H. Functions of sulfur-containing amino acids in lipid metabolism. J. Nutr. 2006, 136, 1666S–1669S. [Google Scholar] [CrossRef]
- Matsuoka, R.; Takahashi, Y.; Kimura, M.; Masuda, Y.; Kunou, M. Heating has no effect on the net protein utilisation from egg whites in rats. Sci. World J. 2017, 2017, 6817196. [Google Scholar] [CrossRef]
- Nagaoka, S.; Masaoka, M.; Zhang, Q.; Hasegawa, M.; Watanabe, K. Egg ovomucin attenuates hypercholesterolemia in rats and inhibits cholesterol absorption in Caco-2 cells. Lipids 2002, 37, 267–272. [Google Scholar] [CrossRef]
- Matsuoka, R.; Kimura, M.; Muto, A.; Masuda, Y.; Sato, M.; Imaizumi, K. Mechanism for the cholesterol-lowering action of egg white protein in rats. Biosci. Biotechnol. Biochem. 2008, 72, 1506–1512. [Google Scholar] [CrossRef]
- Arimitsu, K.; Takashi, K.; Matsuoka, R.; Narita, K.; Idei, A. The good function of lactic fermented egg white “Lacty Egg”. Jpn. J. Food Eng. 2015, 16, 79–82. (in Japanese). [Google Scholar] [CrossRef]
- Matsuoka, R.; Takahashi, Y.; Muto, A.; Kimura, M. Heated egg white has no effect, but lactic fermented and unheated egg white reduces abdominal fat in rats. Lipids Health Dis. 2019, 18, 187. [Google Scholar] [CrossRef]
- Ikeda, I.; Matsuoka, R.; Hamada, T.; Mitsui, K.; Imabayashi, S.; Uchino, A.; Sato, M.; Kuwano, E.; Itamura, T.; Yamada, K.; et al. Cholesterol esterase accelerates intestinal cholesterol absorption. Biochim. Biophys. Acta 2002, 1571, 34–44. [Google Scholar] [CrossRef]
- Spence, J.D.; Jenkins, D.J.A.; Davignon, J. Dietary cholesterol and egg yolks: Not for patients at risk of vascular disease. Can. J. Cardiol. 2010, 26, e336–e339. [Google Scholar] [CrossRef]
- Spence, J.D. Diet for stroke prevention. Stroke Vasc. Neurol. 2018, 3, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Spence, J.D.; Jenkins, D. Cardiovascular benefit of egg consumption is most unlikely. Heart 2018, 104, 1805–1806. [Google Scholar] [CrossRef]
- Mirannda, J.M.; Anton, X.; Redondo-Valbbuena, C.; Roca-Saavedra, P.; Rodriguez, J.A.; Lamas, A.; Franco, C.M.; Cepeda, A. Egg and egg-derived foods: Effects on human health and use as functional foods. Nutrients 2015, 7, 706–729. [Google Scholar] [CrossRef] [PubMed]
- Lui, X.; Zhao, H.L.; Thiessen, S.; House, J.D.; Jones, P.J.H. Effect of plant sterol-enriched diets on plasma and egg yolk cholesterol concentrations and cholesterol metabolism in laying hens. Poult. Sci. 2010, 89, 270–275. [Google Scholar] [CrossRef]
- Laudadio, V.; Ceci, E.; Lastelia, N.M.B.; Tufarelli, V. Dietary high-polyphenols extra-virgin olive oil is effective in reducing cholesterol content in eggs. Lipids Health Dis. 2015, 14. [Google Scholar] [CrossRef] [PubMed]
- Li, X.L.; He, W.L.; Wang, Z.B.; Xu, T.S. Effects of Chinese herbal mixture on performance, egg quality and blood biochemical parameters of lying hens. J. Anim. Physiol. Anim. Nutr. 2016, 100, 1041–1049. [Google Scholar] [CrossRef]
- Morris, S.S.; Beesabathuni, K.; Headey, D. An egg for everyone: Pathways to universal access to one of nature’s most nutritious foods. Matern. Child Nutr. 2018, 14, e12679. [Google Scholar] [CrossRef]
- Zamora-Ros, R.; Cayssials, V.; Cleries, R.; Redondo, M.L.; Sanchez, M.J.; Rodriguez-Barranco, M.; Sanchez-Cruz, J.J.; Mokoroa, O.; Gil, L.; Amiano, P.; et al. Moderate egg consumption and all^-cause and specific-cause mortality in the Spanish European Prospective into Cancer and Nutrition (EPIC) study. Eur. J. Nutr. 2019, 58, 2003–2201. [Google Scholar] [CrossRef]
- Zhuang, P.; Wu, E.; Mao, L.; Zhu, F.; Zhang, Y.; Chen, X.; Jiao, J.; Zhang, Y. Egg and cholesterol consumption and mortality from cardiovascular and different causes in the United States: A population based cohort study. PLOS Med. 2021, 18, e1003508. [Google Scholar] [CrossRef] [PubMed]
| Energy | Protein g (E%) | Lipids g (E%) | Carbohydrate | SFA | MUFA | PUFA g (E%) | Chol | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| (kcal) | Total | Animal | Total | Animal | g (E%) | g (E%) | g (E%) | n-6 | n-3 | mg | |
| Japan | 1930 | 71.8 | 39.3 | 61.0 | 31.8 | 254.0 | 17.59 | 22.80 | 10.75 | 2.51 | 340 |
| (14.9) | (28.4) | (52.6) | (8.20) | (10.6) | (5.01) | (1.17) | |||||
| USA | 2155 | 81.9 | 52.4 | 88.2 | 49.9 | 248 | 28.7 | 30.2 | 18.6 | 2.2 | 307 |
| (15.2) | (36.8) | 46.0 | (12.0) | (12.6) | (8.69) | (0.92) | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sugano, M.; Matsuoka, R. Nutritional Viewpoints on Eggs and Cholesterol. Foods 2021, 10, 494. https://doi.org/10.3390/foods10030494
Sugano M, Matsuoka R. Nutritional Viewpoints on Eggs and Cholesterol. Foods. 2021; 10(3):494. https://doi.org/10.3390/foods10030494
Chicago/Turabian StyleSugano, Michihiro, and Ryosuke Matsuoka. 2021. "Nutritional Viewpoints on Eggs and Cholesterol" Foods 10, no. 3: 494. https://doi.org/10.3390/foods10030494
APA StyleSugano, M., & Matsuoka, R. (2021). Nutritional Viewpoints on Eggs and Cholesterol. Foods, 10(3), 494. https://doi.org/10.3390/foods10030494






