Food–Drug Interactions with Fruit Juices
Abstract
1. Introduction
2. Absorption, Distribution, Metabolism and Elimination
3. Importance of Cytochrome P450 Enzymes and Drug Transporters in Drug Interactions
4. Fruit Juices
“Fruit juice is the fermentable but unfermented product obtained from the edible part of the fruit which is sound, ripe and fresh or preserved by chilling or freezing of one or more kinds mixed having the characteristic color, flavor, and taste typical of the juice of the fruit from which it comes.”
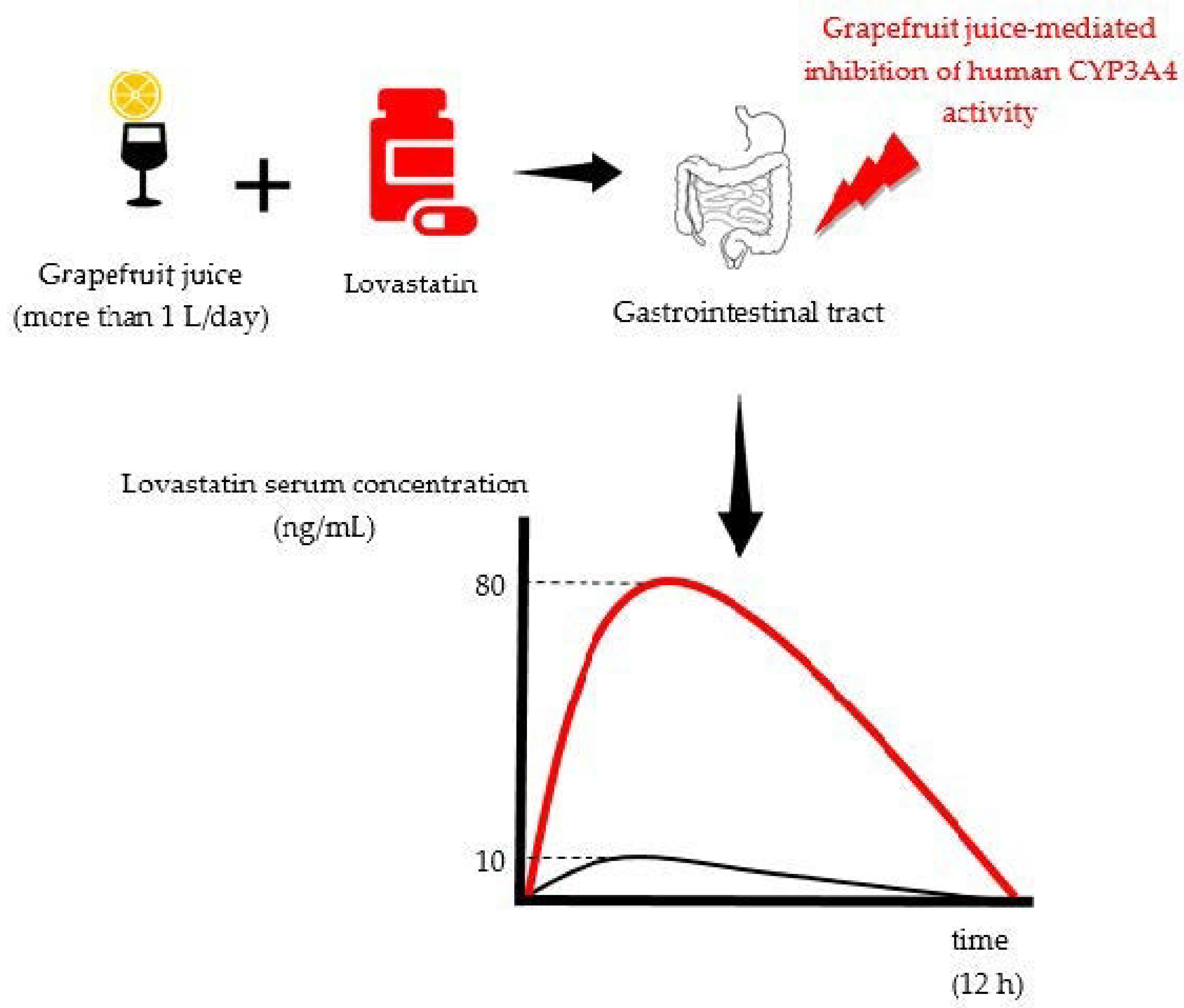
5. Drug Interactions with Common Fruit Juices
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Thanacoody, R. Drug interactions. In Clinical Pharmacy and Therapeutics; Whittlesea, C., Hodson, K., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 53–65. [Google Scholar]
- Amadi, C.N.; Mgbahurike, A.A. Selected food/herb–drug interactions. Am. J. Ther. 2018, 25, 423–433. [Google Scholar] [CrossRef]
- Mouly, S.; Lloret-Linares, C.; Sellier, P.-O.; Sene, D.; Bergmann, J.F. Is the clinical relevance of drug-food and drug-herb interactions limited to grapefruit juice and saint-john’s wort? Pharmacol. Res. 2017, 118, 82–92. [Google Scholar] [CrossRef]
- Bursać Kovačević, D.; Brdar, D.; Fabečić, P.; Barba, F.J.; Lorenzo, J.M.; Putnik, P. Strategies to achieve a healthy and balanced diet: Fruits and vegetables as a natural source of bioactive compounds. In Agri-Food Industry Strategies for Healthy Diets and Sustainability; Barba, F.J., Putnik, P., Bursać Kovačević, D., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 51–88. [Google Scholar]
- Putnik, P.; Pavlić, B.; Šojić, B.; Zavadlav, S.; Žuntar, I.; Kao, L.; Kitonić, D.; Kovačević, D.B. Innovative hurdle technologies for the preservation of functional fruit juices. Foods 2020, 9, 699. [Google Scholar] [CrossRef] [PubMed]
- Zawiah, M.; Yousef, A.-M.; Khan, A.H.; Al-Ashwal, F.Y.; Matar, A.; Alkhawaldeh, B.; Nassar, R.; Abduljabbar, R.; Abdo Ahmed, A.A. Food-drug interactions: Knowledge among pharmacists in jordan. PLoS ONE 2020, 15, e0234779. [Google Scholar] [CrossRef] [PubMed]
- Tozer, T.N.; Rowland, M. Essentials of Pharmacokinetics and Pharmacodynamics; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2016. [Google Scholar]
- Tozer, W.K.; Shargel, L.; Yu, A.B.C. Applied Biopharmaceutics & Pharmacokinetics; Lippincott Williams & Wilkins Publishers: Philadelphia, PA, USA, 2016. [Google Scholar]
- Beringer, P. Winter’s Basic Clinical Pharmacokinetics; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2018. [Google Scholar]
- Persky, A.M.; Pollack, G.M. Foundations in Pharmacokinetics; UNC Press: Chapel Hill, NC, USA, 2017. [Google Scholar]
- Derendorf, H.; Schmidt, S. Rowland and Tozer’s Clinical Pharmacokinetics and Pharmacodynamics: Concepts and Applications, 5th ed.; LWW Lippincott Williams and Wilkins: Philadelphia, PA, USA, 2019. [Google Scholar]
- Anderson, K.E. Effects of specific foods and dietary components on drug metabolism. In Handbook of Drug-Nutrient Interactions; Humana Press: Totowa, NJ, USA, 2009; pp. 243–265. [Google Scholar]
- Rendic, S.; Medic Saric, M. Metabolism of Drugs and Selected Xenobiotics; Medicinska Naklada: Zagreb, Croatia, 2013; pp. 154–157. [Google Scholar]
- Liang, Y.; Li, S.; Chen, L. The physiological role of drug transporters. Protein Cell 2015, 6, 334–350. [Google Scholar] [CrossRef] [PubMed]
- Mao, Q.; Lai, Y.; Wang, J. Drug transporters in xenobiotic disposition and pharmacokinetic prediction. Drug Metab. Dispos. 2018, 46, 561–566. [Google Scholar] [CrossRef]
- McLean, C.; Wilson, A.; Kim, R.B. Impact of transporter polymorphisms on drug development: Is it clinically significant? J. Clin. Pharmacol. 2016, 56, S40–S58. [Google Scholar] [CrossRef]
- Jargin, S. Grapefruit: Perspectives in nutrition and pharmacology. J. Intercult. Ethnopharmacol. 2017, 6, 339. [Google Scholar] [CrossRef]
- Margină, D.; Ilie, M.; Grădinaru, D.; Androutsopoulos, V.P.; Kouretas, D.; Tsatsakis, A.M. Natural products—Friends or foes? Toxicol. Lett. 2015, 236, 154–167. [Google Scholar] [CrossRef]
- Choi, J.H.; Ko, C.M. Food and drug interactions. J. Lifestyle Med. 2017, 7, 1–9. [Google Scholar] [CrossRef]
- Hanley, M.J.; Masse, G.; Harmatz, J.S.; Court, M.H.; Greenblatt, D.J. Pomegranate juice and pomegranate extract do not impair oral clearance of flurbiprofen in human volunteers: Divergence from in vitro results. Clin. Pharmacol. Ther. 2012, 92, 651–657. [Google Scholar] [CrossRef] [PubMed]
- Farkas, D.; Greenblatt, D.J. Influence of fruit juices on drug disposition: Discrepancies betweenin vitroand clinical studies. Expert Opin. Drug Metab. Toxicol. 2008, 4, 381–393. [Google Scholar] [CrossRef] [PubMed]
- Yalcin, H.; Çapar, T.D. Bioactive compounds of fruits and vegetables. In Minimally Processed Refrigerated Fruits and Vegetables; Springer: Berlin, Germany, 2017; pp. 723–745. [Google Scholar]
- Koubaa, M.; Barba, F.J.; Bursać Kovačević, D.; Putnik, P.; Santos, M.D.; Queirós, R.P.; Moreira, S.A.; Inácio, R.S.; Fidalgo, L.G.; Saraiva, J.A. Pulsed electric field processing of fruit juices. In Fruit Juices: Extraction, Composition, Quality and Analysis; Rajauria, G., Tiwari, B., Eds.; Academic Press: Oxford, UK, 2018; pp. 437–449. [Google Scholar]
- The Council of the European Union. Council of the European Union. Council directive 2001/112/ec of 20 december 2001 relating to fruit juices and certain similar products intended for human consumption. In 2001/112/EC; European Union, Ed.; Official Journal of the European Communities: Brussels, Belgium, 2001; pp. L 10/58–L 10/66. [Google Scholar]
- Ashurst, P.R. Introduction. In Chemistry and Technology of Soft Drinks and Fruit Juices; Wiley-Blackwell: Hoboken, NJ, USA, 2016; pp. 1–14. [Google Scholar]
- Benmeziane, F.; Abdourhamane, A.; Guedaoura, A. Nutritional quality and bioactive compounds of some fruit juices. Adv. Environ. Biol. 2016, 10, 242–249. [Google Scholar]
- Mallhi, T.H.; Sarriff, A.; Adnan, A.S.; Khan, Y.H.; Qadir, M.I.; Hamzah, A.A.; Khan, A.H. Effect of fruit/vegetable-drug interactions on cyp450, oatp and p-glycoprotein: A systematic review. Trop. J. Pharm. Res. 2015, 14, 1927–1935. [Google Scholar] [CrossRef]
- Rodríguez-Fragoso, L.; Martínez-Arismendi, J.L.; Orozco-Bustos, D.; Reyes-Esparza, J.; Torres, E.; Burchiel, S.W. Potential risks resulting from fruit/vegetable-drug interactions: Effects on drug-metabolizing enzymes and drug transporters. J. Food Sci. 2011, 76, R112–R124. [Google Scholar] [CrossRef]
- Kantola, T.; Kivistö, K.T.; Neuvonen, P.J. Grapefruit juice greatly increases serum concentrations of lovastatin and lovastatin acid. Clin. Pharmacol. Ther. 1998, 63, 397–402. [Google Scholar] [CrossRef]
- Bailey, D.G.; Malcolm, J.; Arnold, O.; David Spence, J. Grapefruit juice–drug interactions. Br. J. Clin. Pharmacol. 2002, 46, 101–110. [Google Scholar] [CrossRef]
- Bailey, D.G. Predicting clinical relevance of grapefruit-drug interactions: A complicated process. J. Clin. Pharm. Ther. 2017, 42, 125–127. [Google Scholar] [CrossRef]
- Manson, J.M.; Freyssinges, C.; Ducrocq, M.B.; Stephenson, W.P. Postmarketing surveillance of lovastatin and simvastatin exposure during pregnancy. Reprod. Toxicol. 1996, 10, 439–446. [Google Scholar] [CrossRef]
- ALTOPREV®. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/021316s029lbl.pdf (accessed on 1 December 2020).
- Hanley, M.J.; Cancalon, P.; Widmer, W.W.; Greenblatt, D.J. The effect of grapefruit juice on drug disposition. Expert Opin. Drug Metab. Toxicol. 2011, 7, 267–286. [Google Scholar] [CrossRef]
- Mertens-Talcott, S.U.; Zadezensky, I.; De Castro, W.V.; Derendorf, H.; Butterweck, V. Grapefruit-drug interactions: Can interactions with drugs be avoided? J. Clin. Pharmacol. 2006, 46, 1390–1416. [Google Scholar] [CrossRef] [PubMed]
- Paine, M.F.; Widmer, W.W.; Hart, H.L.; Pusek, S.N.; Beavers, K.L.; Criss, A.B.; Brown, S.S.; Thomas, B.F.; Watkins, P.B. A furanocoumarin-free grapefruit juice establishes furanocoumarins as the mediators of the grapefruit juice–felodipine interaction. Am. J. Clin. Nutr. 2006, 83, 1097–1105. [Google Scholar] [CrossRef] [PubMed]
- Bailey, D.G.; Dresser, G.K.; Leake, B.F.; Kim, R.B. Naringin is a major and selective clinical inhibitor of organic anion-transporting polypeptide 1a2 (oatp1a2) in grapefruit juice. Clin. Pharmacol. Ther. 2007, 81, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, K.; Guo, L.; Ohashi, N.; Yoshikawa, M.; Yamazoe, Y. Amounts and variation in grapefruit juice of the main components causing grapefruit–drug interaction. J. Chromatogr. B 2000, 741, 195–203. [Google Scholar] [CrossRef]
- Fukatsu, S.; Fukudo, M.; Masuda, S.; Yano, I.; Katsura, T.; Ogura, Y.; Oike, F.; Takada, Y.; Inui, K.-i. Delayed effect of grapefruit juice on pharmacokinetics and pharmacodynamics of tacrolimus in a living-donor liver transplant recipient. Drug Metab. Pharmacokinet. 2006, 21, 122–125. [Google Scholar] [CrossRef]
- Dreier, J.P.; Endres, M. Statin-associated rhabdomyolysis triggered by grapefruit consumption. Neurology 2004, 62, 670. [Google Scholar] [CrossRef]
- Koziolek, M.; Alcaro, S.; Augustijns, P.; Basit, A.W.; Grimm, M.; Hens, B.; Hoad, C.L.; Jedamzik, P.; Madla, C.M.; Maliepaard, M.; et al. The mechanisms of pharmacokinetic food-drug interactions—A perspective from the ungap group. Eur. J. Pharm. Sci. 2019, 134, 31–59. [Google Scholar] [CrossRef]
- Grenier, J.; Fradette, C.; Morelli, G.; Merritt, G.; Vranderick, M.; Ducharme, M. Pomelo juice, but not cranberry juice, affects the pharmacokinetics of cyclosporine in humans. Clin. Pharmacol. Ther. 2006, 79, 255–262. [Google Scholar] [CrossRef]
- Sermsappasuk, P.; Chaiyakunapruk, N.; Lohitnavy, M.; Kitiyakara, K. Assessing clinical evidence of drug interactions betweencitrus juices and cyclosporine. Asian Biomed. 2013, 7, 477–489. [Google Scholar]
- Gertz, B.J.; Holland, S.D.; Kline, W.F.; Matuszewski, B.K.; Freeman, A.; Quan, H.; Lasseter, K.C.; Mucklow, J.C.; Porras, A.G. Studies of the oral bioavailability of alendronate. Clin. Pharmacol. Ther. 1995, 58, 288–298. [Google Scholar] [CrossRef]
- Gertz, B.J.; Holland, S.D.; Kline, W.F.; Matuszewski, B.K.; Porras, A.G. Clinical pharmacology of alendronate sodium. Osteoporos. Int. 1993, 3, 13–16. [Google Scholar] [CrossRef] [PubMed]
- Malhotra, S. Seville orange juice-felodipine interaction: Comparison with dilute grapefruit juice and involvement of furocoumarins. Clin. Pharmacol. Ther. 2001, 69, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Edwards, D.; Fitzsimmons, M.; Schuetz, E.; Yasuda, K.; Ducharme, M.; Warbasse, L.; Woster, P.; Schuetz, J.; Watkins, P. 6′,7′-dihydroxybergamottin in grapefruit juice and seville orange juice: Effects on cyclosporine disposition, enterocyte cyp3a4, and p-glycoprotein. Clin. Pharmacol. Ther. 1999, 65, 237–244. [Google Scholar] [CrossRef]
- Abdelkawy, K.S.; Donia, A.M.; Turner, R.B.; Elbarbry, F. Effects of lemon and seville orange juices on the pharmacokinetic properties of sildenafil in healthy subjects. Drugs R D 2016, 16, 271–278. [Google Scholar] [CrossRef][Green Version]
- Al-Ghazawi, M.A.; Tutunji, M.S.; AbuRuz, S.M. The effects of pummelo juice on pharmacokinetics of sildenafil in healthy adult male jordanian volunteers. Eur. J. Clin. Pharmacol. 2009, 66, 159–163. [Google Scholar] [CrossRef]
- Žuntar, I.; Krivohlavek, A.; Kosić-Vukšić, J.; Granato, D.; Bursać Kovačević, D.; Putnik, P. Pharmacological and toxicological health risk of food (herbal) supplements adulterated with erectile dysfunction medications. Curr. Opin. Food Sci. 2018, 24, 9–15. [Google Scholar] [CrossRef]
- Senthilkumaran, S.; Suresh, P.; Thirumalaikolundusubramanian, P.; Balamurugan, N. Priapism, pomegranate juice, and sildenafil: Is there a connection? Urol. Ann. 2012, 4, 108. [Google Scholar] [CrossRef]
- Dresser, G. Fruit juices inhibit organic anion transporting polypeptide–mediated drug uptake to decrease the oral availability of fexofenadine. Clin. Pharmacol. Ther. 2002, 71, 11–20. [Google Scholar] [CrossRef]
- Jeon, H.; Jang, I.-J.; Lee, S.; Ohashi, K.; Kotegawa, T.; Ieiri, I.; Cho, J.-Y.; Yoon, S.H.; Shin, S.-G.; Yu, K.-S.; et al. Apple juice greatly reduces systemic exposure to atenolol. Br. J. Clin. Pharmacol. 2013, 75, 172–179. [Google Scholar] [CrossRef]
- Tapaninen, T.; Neuvonen, P.J.; Niemi, M. Orange and apple juice greatly reduce the plasma concentrations of the oatp2b1 substrate aliskiren. Br. J. Clin. Pharmacol. 2011, 71, 718–726. [Google Scholar] [CrossRef]
- Lilja, J. Orange juice substantially reduces the bioavailability of the β-adrenergic–blocking agent celiprolol. Clin. Pharmacol. Ther. 2004, 75, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Mougey, E.B.; Lang, J.E.; Wen, X.; Lima, J.J. Effect of citrus juice and slco2b1 genotype on the pharmacokinetics of montelukast. J. Clin. Pharmacol. 2011, 51, 751–760. [Google Scholar] [CrossRef] [PubMed]
- Wallace, A.W.; Victory, J.M.; Amsden, G.W. Lack of bioequivalence when levofloxacin and calcium-fortified orange juice are coadministered to healthy volunteers. J. Clin. Pharmacol. 2003, 43, 539–544. [Google Scholar] [CrossRef] [PubMed]
- Neuhofel, A.L.; Wilton, J.H.; Victory, J.M.; Hejmanowski, L.G.; Amsden, G.W. Lack of bioequivalence of ciprofloxacin when administered with calcium-fortified orange juice: A new twist on an old interaction. J. Clin. Pharmacol. 2002, 42, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Fairweather-Tait, S.; Hickson, K.; McGaw, B.; Reid, M. Orange juice enhances aluminium absorption from antacid preparation. Eur. J. Clin. Nutr. 1994, 48, 71–73. [Google Scholar] [PubMed]
- Balay, K.S.; Hawthorne, K.M.; Hicks, P.D.; Griffin, I.J.; Chen, Z.; Westerman, M.; Abrams, S.A. Orange but not apple juice enhances ferrous fumarate absorption in small children. J. Pediatr. Gastroenterol. Nutr. 2010, 50, 545–550. [Google Scholar] [CrossRef]
- Aronson, J.K. Meyler’s Side Effects of Drugs: The International Encyclopedia of Adverse Drug Reactions and Interactions; Aronson, J.K., Ed.; Elsevier Science: Amsterdam, The Netherlands, 2016; pp. 1030–1053. [Google Scholar]
- Weaver, C.M. Potassium and health. Adv. Nutr. 2013, 4, 368S–377S. [Google Scholar] [CrossRef]
- St-Jules, D.E.; Goldfarb, D.S.; Sevick, M.A. Nutrient non-equivalence: Does restricting high-potassium plant foods help to prevent hyperkalemia in hemodialysis patients? J. Renal Nutr. 2016, 26, 282–287. [Google Scholar] [CrossRef]
- Stacewicz-Sapuntzakis, M.; Bowen, P.E.; Hussain, E.A.; Damayanti-Wood, B.I.; Farnsworth, N.R. Chemical composition and potential health effects of prunes: A functional food? Crit. Rev. Food Sci. Nutr. 2001, 41, 251–286. [Google Scholar] [CrossRef]
- Wallace, T. Dried plums, prunes and bone health: A comprehensive review. Nutrients 2017, 9, 401. [Google Scholar] [CrossRef]
- Sánchez-Pérez, S.; Comas-Basté, O.; Rabell-González, J.; Veciana-Nogués, M.; Latorre-Moratalla, M.; Vidal-Carou, M. Biogenic amines in plant-origin foods: Are they frequently underestimated in low-histamine diets? Foods 2018, 7, 205. [Google Scholar] [CrossRef]
- Bushra, R.; Aslam, N.; Khan, A. Food drug interactions. Oman Med. J. 2011, 26, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Garcia, E.; Santos, C. Monoamine Oxidase Inhibitor Toxicity. Available online: https://www.ncbi.nlm.nih.gov/books/NBK459386/ (accessed on 24 October 2020).
- Chen, J.J. Pharmacologic safety concerns in parkinson’s disease: Facts and insights. Int. J. Neurosci. 2011, 121, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Aston, J.L.; Lodolce, A.E.; Shapiro, N.L. Interaction between warfarin and cranberry juice. Pharmacotherapy 2006, 26, 1314–1319. [Google Scholar] [CrossRef] [PubMed]
- Mergenhagen, K.A.; Sherman, O. Elevated international normalized ratio after concurrent ingestion of cranberry sauce and warfarin. Am. J. Health-Syst. Pharm. 2008, 65, 2113–2116. [Google Scholar] [CrossRef] [PubMed]
- Medicines and Healthcare Products Regulatory Agency/Committee on Safety of Medicines. Interaction between warfarin and cranberry juice: New advice. Curr. Probl. Pharmacovigil. 2004, 30, 10. [Google Scholar]
- Suvarna, R. Possible interaction between warfarin and cranberry juice. BMJ 2003, 327, 1454. [Google Scholar] [CrossRef][Green Version]
- Ansell, J.; McDonough, M.; Zhao, Y.; Harmatz, J.S.; Greenblatt, D.J. The absence of an interaction between warfarin and cranberry juice: A randomized, double-blind trial. J. Clin. Pharmacol. 2009, 49, 824–830. [Google Scholar] [CrossRef]
- Johnson, J.A.; Cavallari, L.H. Warfarin pharmacogenetics. Trends Cardiovasc. Med. 2015, 25, 33–41. [Google Scholar] [CrossRef]
- Zikria, J.; Goldman, R.; Ansell, J. Cranberry juice and warfarin: When bad publicity trumps science. Am. J. Med. 2010, 123, 384–392. [Google Scholar] [CrossRef]
- Backman, J. Lack of correlation between in vitro and in vivo studies on the effects of tangeretin and tangerine juice on midazolam hydroxylation. Clin. Pharmacol. Ther. 2000, 67, 382–390. [Google Scholar] [CrossRef] [PubMed]
- Putnik, P.; Barba, F.J.; Lorenzo, J.M.; Gabrić, D.; Shpigelman, A.; Cravotto, G.; Bursać Kovačević, D. An integrated approach to mandarin processing: Food safety and nutritional quality, consumer preference, and nutrient bioaccessibility. Compr. Rev. Food Sci. Food Saf. 2017, 16, 1345–1358. [Google Scholar] [CrossRef]
- Obermeier, M.T.; White, R.E.; Yang, C.S. Effects of bioflavonoids on hepatic p450 activities. Xenobiotica 2009, 25, 575–584. [Google Scholar] [CrossRef]
- Kim, H.; Yoon, Y.-J.; Shon, J.-H.; Cha, I.-J.; Shin, J.-G.; Liu, K.-H. Inhibitory effects of fruit juices on cyp3a activity. Drug Metab. Dispos. 2006, 34, 521–523. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Ma, Z.; Luo, X.; Li, X. Effects of mulberry fruit (morus alba l.) consumption on health outcomes: A mini-review. Antioxidants 2018, 7, 69. [Google Scholar] [CrossRef]
- Rodriguez-Fragoso, L.; Reyes-Esparz, J. Fruit/vegetable-drug interactions: Effects on drug metabolizing enzymes and drug transporters. In Drug Discovery; IntechOpen: London, UK, 2013; pp. 1–33. [Google Scholar]
- Piver, B.; Berthou, F.; Dreano, Y.; Lucas, D. Inhibition of cyp3a, cyp1a and cyp2e1 activities by resveratrol and other non volatile red wine components. Toxicol. Lett. 2001, 125, 83–91. [Google Scholar] [CrossRef]
- Hidaka, M.; Fujita, K.-i.; Ogikubo, T.; Yamasaki, K.; Iwakiri, T.; Okumura, M.; Kodama, H.; Arimori, K. Potent inhibition by star fruit of human cytochrome p450 3a (cyp3a) activity. Drug Metab. Dispos. 2004, 32, 581–583. [Google Scholar] [CrossRef]
- Eskra, J.N.; Schlicht, M.J.; Bosland, M.C. Effects of black raspberries and their ellagic acid and anthocyanin constituents on taxane chemotherapy of castration-resistant prostate cancer cells. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef]
- Gerber, W.; Steyn, J.; Kotzé, A.; Hamman, J. Beneficial pharmacokinetic drug interactions: A tool to improve the bioavailability of poorly permeable drugs. Pharmaceutics 2018, 10, 106. [Google Scholar] [CrossRef]
- Taniguchi, S. Treatment of psoriasis by cyclosporine and grapefruit juice. Arch. Dermatol. 1996, 132. [Google Scholar] [CrossRef]
- van Agtmael, M.A.; Gupta, V.; van der Wösten, T.H.; Rutten, J.P.B.; van Boxtel, C.J. Grapefruit juice increases the bioavailability of artemether. Eur. J. Clin. Pharmacol. 1999, 55, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Adegoke, S.A.; Oyelami, O.A.; Olatunya, O.S.; Adeyemi, L.A. Effects of lime juice on malaria parasite clearance. Phytother. Res. 2011, 25, 1547–1550. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.; Wang, Y.; Guo, J.; Chu, H.; Gao, Y.; Pang, L. Blueberry improves the therapeutic effect of etanercept on patients with juvenile idiopathic arthritis: Phase iii study. Tohoku J. Exp. Med. 2015, 237, 183–191. [Google Scholar] [CrossRef][Green Version]
- Blanco-Rojo, R.; Pérez-Granados, A.M.; Toxqui, L.; González-Vizcayno, C.; Delgado, M.A.; Vaquero, M.P. Efficacy of a microencapsulated iron pyrophosphate-fortified fruit juice: A randomised, double-blind, placebo-controlled study in spanish iron-deficient women. Br. J. Nutr. 2011, 105, 1652–1659. [Google Scholar] [CrossRef] [PubMed]
| Fruit Juice Type | Examples of Drugs | Suggested Mechanism of an Interaction | Reference |
|---|---|---|---|
| Grapefruit | CYP3A4 substrates HMG-CoA reductase inhibitors (simvastatin, lovastatin) Immunosuppressives (cyclosporine) Antiarrhythmics (amiodarone) Anticonvulsants (carbamazepine) | via CYP3A4, or/and P-gp | [34,35] |
| PDE5 inhibitor (sildenafil) | [50,51] | ||
| Antimalarial agent (artemether) | [88] | ||
| Orange | Bisphosphonates (alendronate) | physicochemical interaction | [45] |
| Antihistamines (fexofenadine) | OATP transporters, or/and P-gp | [52] | |
| Beta-blocker (atenolol) | [55] | ||
| Anti-asthmatic agent (montelukast) | [56] | ||
| Calcium-fortified orange juice | Aluminum and iron supplements | physicochemical interaction | [59,60] |
| Antibiotics (fluoroquinolones) | [57,58] | ||
| Seville orange | PDE5 inhibitor (sildenafil) | via CYP3A4 | [48] |
| Pomegranate | PDE5 inhibitor (sildenafil) | via CYP3A4 | [50,51] |
| Pomelo | PDE5 inhibitor (sildenafil) | physicochemical interaction | [49] |
| Immunosuppressives (cyclosporine) | P-gp | [42,43] | |
| Apple | Antihistamines (fexofenadine) | OATP transporters, or/and P-gp | [52] |
| Beta-blocker (atenolol) | [53] | ||
| Anti-asthmatic agent (montelukast) | [54] | ||
| Antihypertensive agent (aliskiren) | [54] | ||
| Antihistamines (fexofenadine) | [52] | ||
| Blueberry | TNF-α inhibitor (etanercept) | Beneficial interaction suggested to be due to anti-oxidant/anti-inflammatory properties of blueberries | [90] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petric, Z.; Žuntar, I.; Putnik, P.; Bursać Kovačević, D. Food–Drug Interactions with Fruit Juices. Foods 2021, 10, 33. https://doi.org/10.3390/foods10010033
Petric Z, Žuntar I, Putnik P, Bursać Kovačević D. Food–Drug Interactions with Fruit Juices. Foods. 2021; 10(1):33. https://doi.org/10.3390/foods10010033
Chicago/Turabian StylePetric, Zvonimir, Irena Žuntar, Predrag Putnik, and Danijela Bursać Kovačević. 2021. "Food–Drug Interactions with Fruit Juices" Foods 10, no. 1: 33. https://doi.org/10.3390/foods10010033
APA StylePetric, Z., Žuntar, I., Putnik, P., & Bursać Kovačević, D. (2021). Food–Drug Interactions with Fruit Juices. Foods, 10(1), 33. https://doi.org/10.3390/foods10010033







