Remineralization of Demineralized Enamel and Dentine Using 3 Dentifrices—An InVitro Study
Abstract
:1. Introduction
- To monitor electrical resistance in demineralized enamel and dentine using ECM after exposure to fluoridated dentifrices;
- Using microradiography, to quantify mineral loss and the response of demineralized enamel and dentine to fluoridated dentifrices;
- To assess the association between electrical resistance measurements of treated enamel and dentine (with placebo and fluoride dentifrices and saliva storage solution) with microradiographic data.
2. Materials and Methods
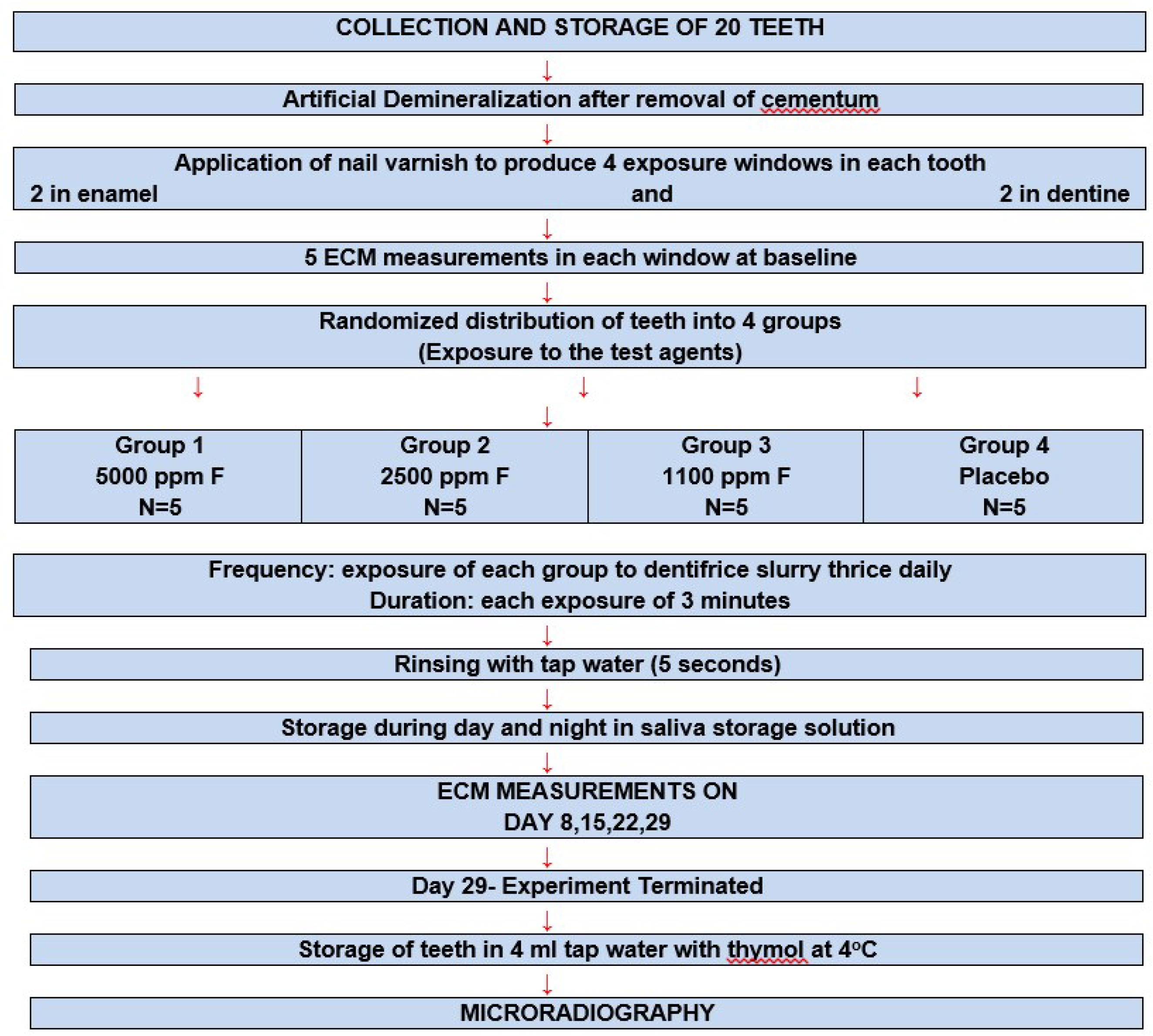
2.1. Collection and Storage of Teeth
2.2. Artificial Demineralization
2.3. Exposure to the Test Dentifrices
2.4. Remineralization
2.5. Measurement of Tooth Resistance Using ECM
2.6. Operating Principle
2.7. Microradiography
2.8. Repeatability of the Experiment (ECM Measurements)
2.9. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hargreaves, J.A.; Chester, C.G. Clinical trial among Scottish children of an anticaries dentifrice containing 2% sodium monofluorophosphate. Community Dent. Oral Epidemiol 1973, 1, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Hargreaves, J.A.; Thompson, G.W.; Wagg, B.J. Changes in caries prevalence of Isle of Lewis children between 1971 and 1981. Caries Res. 1983, 17, 554–559. [Google Scholar] [CrossRef] [PubMed]
- Marthaler, T.M. Explanations for changing patterns of disease in the Western world in Guggenheim. In Cariology Today; Karger Publishers: Basel, Switzerland, 1984; pp. 13–23. [Google Scholar]
- American Dental Association Council on Scientific Affairs. Profesionally applied topical fluoride: Evidence based clinical recommendations. J. Am. Dent. Assoc. 2006, 137, 1151–1159. [Google Scholar] [CrossRef] [PubMed]
- Marinho, V.C.C.; Higgins, J.P.T.; Logan, S.; Sheiham, A. Fluoride gels for preventing dental caries in children and aldolescents. Cochrane Database Syst. Rev. (Interv. Rev.) 2002. [Google Scholar] [CrossRef]
- Hardman, M.C.; Davies, G.M.; Duxbury, J.T.; Davies, R.M. A cluster randomized controlled trial to evaluate the effectiveness of fluoride varnish as a public health measure to reduce caries in children. Caries Res. 2007, 41, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Ekstrand, K.; Martignon, S.; Holm-Pedersen, P. Development and evaluation of two root caries controlling programmes for home based frail people older than 75 years. Gerodontology 2008, 25, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Lu, K.H.; Ruhlman, C.D.; Chung, K.L.; Sturzenberger, O.P.; Lehnhoff, R.W. A 3 year clinical comparison of a sodium monofluorophosphate dentifrice with sodium fluoride dentifrices on dental caries in children. ASDC J. Dent. Child 1987, 54, 241–244. [Google Scholar]
- Hagen, A.R. The state of fluoride in dentifrice systems. ActaOdontol. Scand. 1972, 30, 167–186. [Google Scholar]
- Featherstone, J.D.B.; Zero, D.T. Laboratory and human studies to elucidate the mechanism of action of fluoride containing dentifrices. In Clinical and Biological Aspects of Dentifrice’s; Embery, G., Rolla, G., Eds.; Oxford University Press: Oxford, UK, 1992; pp. 41–50. [Google Scholar]
- Marinho, V.C.; Higgins, J.P.; Sheiham, A.; Logan, S. Fluoride toothpastes for preventing dental caries in children and adolescents. Cochrane Database Syst. Rev. 2003, CD002278. [Google Scholar] [CrossRef]
- Cruz, R.; Ogaard, B.; Rolla, G. Acquisition of alkali soluble fluoride by enamel through treatment with NaF—Containing toothpaste in-vitro. Eur. J. Oral Sci. 1992, 100, 81–87. [Google Scholar] [CrossRef]
- Petersen, P.E.; Ogawa, H. Prevention of dental caries through the use of fluoride–The WHO approach. Community Dent Health 2016, 33, 66–68. [Google Scholar] [PubMed]
- Legeros, R.Z.; Legeros, J.P. Phosphate minerals in human tissues. In Phosphate Minerals; Nriagu, J.O., Moore, P.B., Eds.; Springer: Berlin/Heidelberg, Germany, 1984; pp. 351–385. [Google Scholar]
- Teruel Jde, D.; Alcolea, A.; Hernández, A.; Ruiz, A.J. Comparison of chemical composition of enamel and dentine in human, bovine, porcine and bovine teeth. Arch. Oral Biol. 2015, 60, 768–775. [Google Scholar] [CrossRef] [PubMed]
- Kang, D.; Amarasiriwardena, D.; Goodman, A.H. Application of laser ablation–inductively coupled plasma–mass spectrometry (LA–ICP–MS) to investigate trace metal spatial distributions in human tooth enamel and dentine growth layers and pulp. Anal. Bioanal. Chem. 2004, 378, 1608–1615. [Google Scholar] [CrossRef] [PubMed]
- He, L.H.; Swain, M.V. Understanding the mechanical behavior of human enamel from its structural and compositional characteristics. J. Mech. Behav. Biomed. 2008, 1, 18–29. [Google Scholar] [CrossRef] [PubMed]
- Schofield, R.M.S.; Nesson, M.H.; Richardson, K.A. Tooth hardness increases with zinc-content in mandibles of young adult leaf-cutter ants. Naturwissenschaften 2002, 89, 579–583. [Google Scholar] [PubMed]
- Ten Cate, J.M.; Nyvad, B.; Van De Plassche-Simons, Y.M.; Fejerskov, O. A quantitative analysis of mineral loss and shrinkage of in-vitro demineralized human root surfaces. J. Dent. Res. 1991, 70, 1371–1374. [Google Scholar] [CrossRef] [PubMed]
- Baysan, A.; Lynch, E.; Ellwood, R.; Davies, R.; Petersson, L.; Borsboom, P. Reversal of primary root caries using dentifrices containing 5000 and 1100 ppm fluoride. Caries Res. 2001, 35, 41–46. [Google Scholar] [PubMed]
- Tavss, E.A.; Mellberg, J.R.; Joziak, M.; Gambogi, R.J.; Fisher, S.W. Relationship between dentifrice fluoride concentration and clinical caries reduction. Am. J. Dent. 2003, 16, 369–374. [Google Scholar] [PubMed]
- Walsh, T.; Worthington, H.; Glenny, A.M.; Marinho, V.C.; Jeroncic, A. Fluoride toothpastes of different concentrations for preventing dental caries. Cochrane Database Syst. Rev. 2019, 3, CD007868. [Google Scholar] [CrossRef]
- Pretty, I.A. High fluoride concentration toothpastes for children and adolescents. Caries Res. 2016, 50 (Suppl. S1), 9–14. [Google Scholar] [CrossRef] [PubMed]
- Ricketts, D.; Kidd, E.A.M.; Smith, B.G.N.; Wilson, R.F. Clinical and radiographic diagnosis of occlusal caries: A study invitro. J. Oral Rehab. 1995, 22, 15–20. [Google Scholar] [CrossRef]
- Lussi, A.; Jaeggi-Scharer, T. Prediction of the erosive potential of some beverages. Caries Res. 1995, 29, 349–354. [Google Scholar] [CrossRef] [PubMed]
- Ashley, P. The Use of Electrical Impedance Measurements for the Diagnosis of Posterior Occlusal Caries in Clinical Trial. Ph.D. Thesis, University of London, London, UK, 1997. [Google Scholar]
- Ruben, J.; Arends, J. Shrinkage prevention of invitro demineralized human dentine in transversal microradiography. Caries Res. 1993, 27, 262–265. [Google Scholar] [CrossRef] [PubMed]
- De Jong, E.D.J.; ten Bosch, J.J.; Noordmans, J. Optimised microcomputer guided quantitative microradiography on dental mineralized tissue slices. Phys. Med. Biol. 1987, 32, 1209–1220. [Google Scholar]
- Ogaard, B.; Rolla, G.; Arends, J. Invivo progress of enamel and root surface lesions under plaque as a function of time. Caries Res. 1988, 22, 302–305. [Google Scholar] [CrossRef] [PubMed]
- Kusano, S.C.; Tenuta, L.M.; Cury, A.A.; Cury, J.A. Timing of fluoride toothpaste use and enamel-dentine demineralization. Braz. Oral Res. 2011, 25, 383–387. [Google Scholar] [CrossRef] [PubMed]
- Dunipace, A.J.; Zhang, W.; Beiswanger, A.J.; Stookey, G.K. An invitro model for studying the efficacy of fluoride dentifrices in preventing root caries. Caries Res. 1994, 28, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Von der Fehr, F.R.; Loe, H.; Theilade, E. Experimental caries in man. Caries Res. 1970, 4, 131–148. [Google Scholar] [CrossRef]
- Arends, J.; ten Bosch, J.J. Demineralization and remineralization techniques. J. Dent. Res. 1992, 71, 924–928. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Cao, S.; Wang, H.; Li, Y.; Kishen, A.; Deng, X.; Yang, X.; Wang, Y.; Cong, C.; Wang, H.; et al. Biomimetic remineralization of demineralized dentine using scaffold of CMC/ACP nanocomplexes in an in vitro tooth model of deep caries. PLoS ONE 2015, 10, e0116553. [Google Scholar] [CrossRef] [PubMed]
- Klont, B.; ten Cate, J.M. Remineralization of bovine incisor root lesions in vitro: The role of the collagenous matrix. Caries Res. 1991, 25, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Klont, B.; ten Cate, J.M. Susceptibility of the collagenous matrix from bovine incisor roots to proteolysis after in vitro lesion formation. Caries Res. 1991, 25, 46–50. [Google Scholar] [CrossRef] [PubMed]
- ten Cate, J.M. Remineralization of caries lesions extend into dentine. J. Dent. Res. 2001, 80, 1407–1411. [Google Scholar] [CrossRef] [PubMed]
- ten Cate, J.M. Remineralization of deep enamel dentine caries lesions. Aust. Dent. J. 2008, 53, 281–285. [Google Scholar] [CrossRef] [PubMed]
- Preston, K.P.; Smith, P.W.; Higham, S.M. The influence of varying fluoride concentrations on in vitro remineralisation of artificial dentinal lesions with differing lesion morphologies. Arch. Oral Biol. 2008, 53, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Arends, J.; Christoffersen, J.; Ruben, J.; Jongebloed, W.L. Remineralization of Bovine dentine invitro. Caries Res. 1989, 23, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Phankosol, P.; Ettinger, R.L.; Hick, M.J.; Wefel, J.S. Depth of penetration of invitro root surface lesions. J. Dent. Res. 1985, 64, 897–899. [Google Scholar] [CrossRef]
- Menaker, L.; Morhart, R.E.; Navia, J. The Biologic Basis of Dental Caries: An Oral Biology Textbook; Harper & Row: London, UK, 1980; p. 532. [Google Scholar]
- Jones, S.J.; Boyde, A. Dentine mineralization, demineralization and microhardness: Recent studies using scanning microscopies. In Dentine and Dentine Reactions in the Oral Cavity; Thylstrup, A., Leach, S.A., Qvist, V., Eds.; IRL Press: Oxford, UK, 1987; pp. 33–35. [Google Scholar]
- Borzabadi-Farahani, A.; Eslamipour, F.; Asgari, I. Association between orthodontic treatment need and caries experience. Acta Odontol. Scand. 2011, 69, 2–11. [Google Scholar] [CrossRef]
- Walsh, L.J.; Healey, D.L. Prevention and caries risk management in teenage and orthodontic patients. Aust. Dent. J. 2019, 64 (Suppl. S1), S37–S45. [Google Scholar] [CrossRef]
| Test Dentifrices | Tissue Enamel | Tissue Dentine | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| DAYS OF EXPERIMENT | ||||||||||
| 0 | 8 | 15 | 22 | 29 | 0 | 8 | 15 | 22 | 29 | |
| Placebo Log10 Int value. | 4.07 ± 1.53 | 3.92 ± 1.16 | 4.06 ± 1.16 | 3.56 ± 1.18 | 3.87 ± 0.90 | 2.13 ± 0.89 | 1.82 ± 0.98 | 2.15 ± 0.88 | 2.60 ± 0.60 | 3.06 ± 0.87 |
| Log10 End value. | 1.28 ± 0.62 | 1.33 ± 0.30 | 1.37 ± 0.27 | 1.23 ± 0.28 | 1.33 ± 0.23 | 0.69 ± 0.34 | 0.47 ± 0.63 | 0.71 ± 0.34 | 0.93 ± 0.24 | 1.07 ± 0.31 |
| Colgate Winter Fresh gel Log10 Int value. | 4.11 ± 1.86 | 5.79 ± 1.21 | 5.01 ± 1.15 | 4.64 ± 1.48 | 4.64 ± 1.43 | 1.87 ± 0.63 | 4.00 ± 2.02 | 2.19 ± 1.90 | 2.64 ± 1.73 | 2.88 ± 1.32 |
| Log10 End value. | 1.24 ± 0.69 | 1.73 ± 0.24 | 1.59 ± 0.23 | 1.47 ± 0.40 | 1.49 ± 0.32 | 0.56 ± 0.39 | 1.20 ± 0.72 | 0.44 ± 0.97 | 0.78 ± 0.63 | 0.98 ± 0.41 |
| Fluocaril Bi-Fluoré Log10 Int value. | 4.81 ± 0.9 | 5.26 ± 1.72 | 3.82 ± 1.40 | 4.17 ± 1.31 | 4.21 ± 1.20 | 2.47 ± 1.20 | 3.87 ± 2.31 | 1.33 ± 0.72 | 2.00 ± 0.79 | 1.65 ± 0.60 |
| Log10 End value. | 1.55 ± 0.20 | 1.59 ± 0.42 | 1.29 ± 0.32 | 1.39 ± 0.31 | 1.40 ± 0.29 | 0.80 ± 0.50 | 1.00 ± 1.09 | 0.10 ± 0.79 | 0.53 ± 0.66 | 0.40 ± 0.57 |
| Colgate Prevident Log10 Int value. | 4.60 ± 0.88 | 5.50 ± 0.76 | 5.16 ± 1.40 | 4.02 ± 1.20 | 3.76 ± 0.90 | 2.16 ± 0.00 | 4.93 ± 0.55 | 2.92 ± 1.49 | 2.50 ± 1.20 | 2.34 ± 1.07 |
| Log10 End value. | 1.50 ± 0.20 | 1.70 ± 0.14 | 1.60 ± 1.00 | 1.34 ± 0.35 | 1.30 ± 0.24 | 0.73 ± 0.37 | 1.59 ± 0.11 | 0.93 ± 0.60 | 0.82 ± 0.42 | 0.76 ± 0.42 |
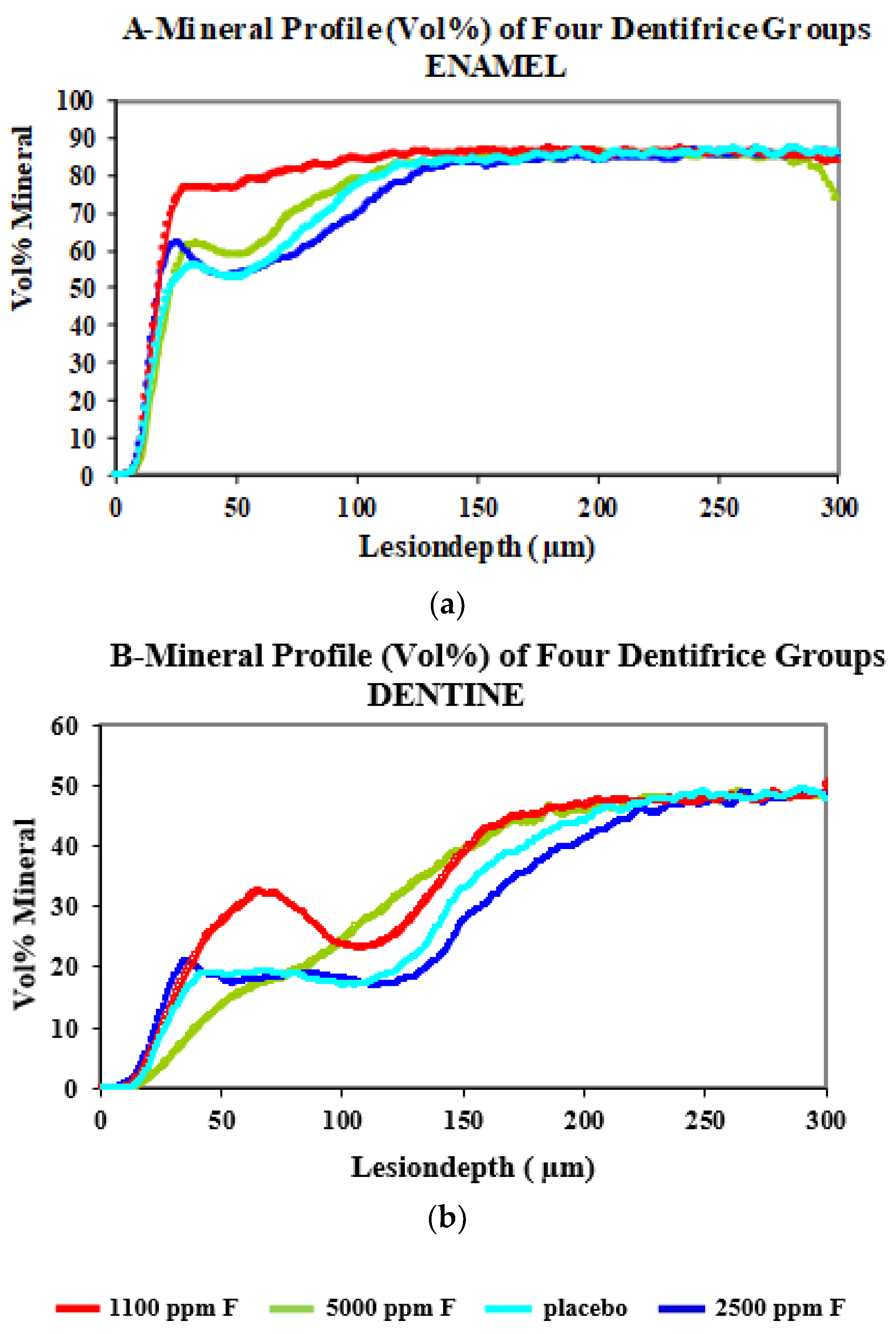
| Test Dentifrice’s | Lesion Depth (Ld, µm) Mean ± SD | Mineral Loss (Kg/m2) Mean ± SD | Mineral Loss Value (∆Z) Vol%. µm | Ratio of Mineral Loss ∆Z/Ld |
|---|---|---|---|---|
| Colgate PreviDent (5000 ppm F) | 97.1 ± 25.7 | 0.10 ± 0.02 | 3062 | 32.8 |
| Fluocaril (2500 ppm F) | 110.2 ± 35.4 | 0.11 ± 0.05 | 3619 | 33.5 |
| Colgate Winterfresh Gel (1100 ppm F) | 50.6 ± 35.8 | 0.05 ± 0.02 | 1613 | 41 |
| Placebo (Non-Fluoridated) | 100.1 ± 20.3 | 0.11 ± 0.02 | 3414 | 34.4 |
| Test Dentifrice’s | Lesion Depth (Ld, µm) Mean ± SD | Mineral Loss (Kg/m2) Mean ± SD | Mineral Loss Value (∆Z) Vol%. µm | Ratio of Mineral Loss ∆Z/Ld |
|---|---|---|---|---|
| Colgate PreviDent (5000 ppm F) | 153.5 ± 22.2 | 0.14 ± 0.03 | 4420 | 28.7 |
| Fluocaril (2500 ppm F) | 183.7 ± 32.2 | 0.16 ± 0.03 | 5152 | 28.1 |
| Colgate Winterfresh Gel (1100 ppm F) | 154.8 ± 22.6 | 0.12 ± 0.03 | 3696 | 23.7 |
| Placebo (Non-Fluoridated) | 169.7 ± 25.9 | 0.15 ± 0.03 | 4867 | 28.5 |
| Variables | Lesion Depth | Minerals Loss Value | Volume of Mineral Loss | ECM |
|---|---|---|---|---|
| Lesion | 1.00 | 0.99 | 0.99 | −0.73 |
| Mineral | 0.99 | 1.00 | 1.00 | −0.72 |
| Volume | 0.99 | 1.00 | 1.00 | −0.72 |
| ECM | −0.73 | −0.72 | −0.72 | 1.00 |
| Variables | Lesion Depth | Mineral Loss | Volume of Mineral Loss | ECM |
|---|---|---|---|---|
| Lesion | 1.00 | 0.85 | 0.85 | −0.54 |
| Mineral | 0.85 | 1.00 | 1.00 | −0.49 |
| Volume | 0.85 | 1.00 | 1.00 | −0.49 |
| ECM | −0.54 | −0.49 | −0.49 | 1.00 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Talwar, M.; Borzabadi-Farahani, A.; Lynch, E.; Borsboom, P.; Ruben, J. Remineralization of Demineralized Enamel and Dentine Using 3 Dentifrices—An InVitro Study. Dent. J. 2019, 7, 91. https://doi.org/10.3390/dj7030091
Talwar M, Borzabadi-Farahani A, Lynch E, Borsboom P, Ruben J. Remineralization of Demineralized Enamel and Dentine Using 3 Dentifrices—An InVitro Study. Dentistry Journal. 2019; 7(3):91. https://doi.org/10.3390/dj7030091
Chicago/Turabian StyleTalwar, Manjit, Ali Borzabadi-Farahani, Edward Lynch, Peter Borsboom, and Jan Ruben. 2019. "Remineralization of Demineralized Enamel and Dentine Using 3 Dentifrices—An InVitro Study" Dentistry Journal 7, no. 3: 91. https://doi.org/10.3390/dj7030091
APA StyleTalwar, M., Borzabadi-Farahani, A., Lynch, E., Borsboom, P., & Ruben, J. (2019). Remineralization of Demineralized Enamel and Dentine Using 3 Dentifrices—An InVitro Study. Dentistry Journal, 7(3), 91. https://doi.org/10.3390/dj7030091






