Dysregulated Phosphate Metabolism, Periodontal Disease, and Cancer: Possible Global Health Implications
Abstract
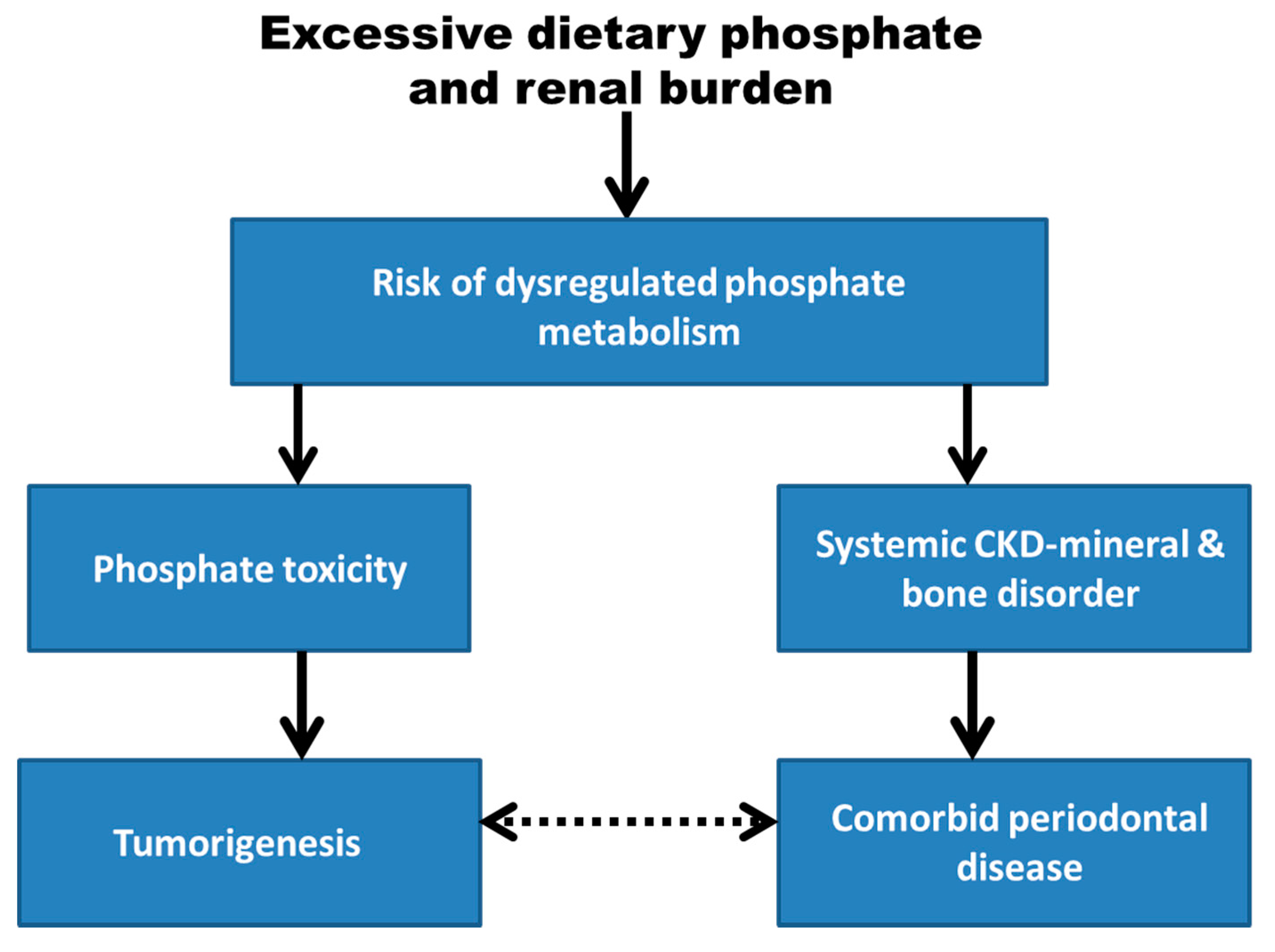
:1. Introduction
2. Phosphate Toxicity as a Global Health Burden
3. Dysregulated Phosphate Metabolism and Cancer
4. Periodontal Disease
5. Chronic Kidney Disease-Mineral and Bone Disorder
6. Conclusions
Funding
Conflicts of Interest
References
- Tonetti, M.S.; Jepsen, S.; Jin, L.; Otomo-Corgel, J. Impact of the global burden of periodontal diseases on health, nutrition and wellbeing of mankind: A call for global action. J. Clin. Periodontol. 2017, 44, 456–462. [Google Scholar] [CrossRef] [PubMed]
- Corbella, S.; Veronesi, P.; Galimberti, V.; Weinstein, R.; Del Fabbro, M.; Francetti, L. Is periodontitis a risk indicator for cancer? A meta-analysis. PLoS ONE 2018, 13, e0195683. [Google Scholar] [CrossRef] [PubMed]
- Michaud, D.S.; Fu, Z.; Shi, J.; Chung, M. Periodontal disease, tooth loss, and cancer risk. Epidemiol. Rev. 2017, 39, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Mai, X.; LaMonte, M.J.; Hovey, K.M.; Freudenheim, J.L.; Andrews, C.A.; Genco, R.J.; Wactawski-Wende, J. Periodontal disease severity and cancer risk in postmenopausal women: The Buffalo OsteoPerio Study. Cancer Causes Control 2016, 27, 217–228. [Google Scholar] [CrossRef] [PubMed]
- Nwizu, N.N.; Marshall, J.R.; Moysich, K.; Genco, R.J.; Hovey, K.M.; Mai, X.; LaMonte, M.J.; Freudenheim, J.L.; Wactawski-Wende, J. Periodontal Disease and Incident Cancer Risk among Postmenopausal Women: Results from the Women’s Health Initiative Observational Cohort. Cancer Epidemiol. Prev. Biomark. 2017, 26, 1255–1265. [Google Scholar] [CrossRef] [PubMed]
- Michaud, D.; Kelsey, K.; Papathanasiou, E.; Genco, C.; Giovannucci, E. Periodontal disease and risk of all cancers among male never smokers: An updated analysis of the Health Professionals Follow-up Study. Ann. Oncol. 2016, 27, 941–947. [Google Scholar] [CrossRef] [PubMed]
- Freudenheim, J.L.; Genco, R.J.; LaMonte, M.J.; Millen, A.E.; Hovey, K.M.; Mai, X.; Nwizu, N.; Andrews, C.A.; Wactawski-Wende, J. Periodontal disease and breast cancer: Prospective cohort study of postmenopausal women. Cancer Epidemiol. Prev. Biomark. 2016, 25, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Shi, T.; Min, M.; Sun, C.; Zhang, Y.; Liang, M.; Sun, Y. Periodontal disease and susceptibility to breast cancer: A meta-analysis of observational studies. J. Clin. Periodontol. 2018, 45, 1025–1033. [Google Scholar] [CrossRef] [PubMed]
- Michaud, D.S.; Izard, J. Microbiota, oral microbiome, and pancreatic cancer. Cancer J. 2014, 20, 203. [Google Scholar] [CrossRef] [PubMed]
- Maisonneuve, P.; Amar, S.; Lowenfels, A.B. Periodontal disease, edentulism, and pancreatic cancer: A meta-analysis. Ann. Oncol. 2017, 28, 985–995. [Google Scholar] [CrossRef] [PubMed]
- Javed, F.; Warnakulasuriya, S. Is there a relationship between periodontal disease and oral cancer? A systematic review of currently available evidence. Crit. Rev. Oncol./Hematol. 2016, 97, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Yao, Q.-W.; Zhou, D.-S.; Peng, H.-J.; Ji, P.; Liu, D.-S. Association of periodontal disease with oral cancer: A meta-analysis. Tumor Biol. 2014, 35, 7073–7077. [Google Scholar] [CrossRef] [PubMed]
- Momen-Heravi, F.; Babic, A.; Tworoger, S.S.; Zhang, L.; Wu, K.; Smith-Warner, S.A.; Ogino, S.; Chan, A.T.; Meyerhardt, J.; Giovannucci, E. Periodontal disease, tooth loss and colorectal cancer risk: Results from the Nurses’ Health Study. Int. J. Cancer 2017, 140, 646–652. [Google Scholar] [CrossRef] [PubMed]
- Barton, M.K. Evidence accumulates indicating periodontal disease as a risk factor for colorectal cancer or lymphoma. CA: Cancer J. Clin. 2017, 67, 173–174. [Google Scholar] [CrossRef] [PubMed]
- Bertrand, K.A.; Shingala, J.; Evens, A.; Birmann, B.M.; Giovannucci, E.; Michaud, D.S. Periodontal disease and risk of non-Hodgkin lymphoma in the Health Professionals Follow-Up Study. Int. J. Cancer 2017, 140, 1020–1026. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.-T.; Deng, A.-P.; Li, C.; Xia, L.-Y.; Niu, Y.-M.; Leng, W.-D. Periodontal disease and risk of head and neck cancer: A meta-analysis of observational studies. PLoS ONE 2013, 8, e79017. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.T.; Xia, L.Y.; Zhang, Y.G.; Li, S.; Leng, W.D.; Kwong, J.S. Periodontal Disease and Incident Lung Cancer Risk: A Meta-Analysis of Cohort Studies. J. Periodontol. 2016, 87, 1158–1164. [Google Scholar] [CrossRef] [PubMed]
- Chrysanthakopoulos, N. Correlation between periodontal disease indices and lung cancer in Greek adults: A case—control study. Exp. Oncol. 2016, 38, 49–53. [Google Scholar] [CrossRef]
- Lee, J.-H.; Kweon, H.H.-I.; Choi, J.-K.; Kim, Y.-T.; Choi, S.-H. Association between periodontal disease and prostate cancer: Results of a 12-year longitudinal cohort study in South Korea. J. Cancer 2017, 8, 2959. [Google Scholar] [CrossRef] [PubMed]
- Chrysanthakopoulos, N.A.; Oikonomou, A.A. A case-control study of the periodontal condition in gastric cancer patients. Stomatol. Dis. Sci. 2017, 1, 55–61. [Google Scholar] [CrossRef]
- Brown, R.B.; Razzaque, M.S. Phosphate toxicity: A stealth biochemical stress factor? Med. Mol. Morphol. 2016, 49, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Erem, S.; Razzaque, M.S. Dietary phosphate toxicity: An emerging global health concern. Histochem. Cell Biol. 2018, 150, 711–719. [Google Scholar] [CrossRef] [PubMed]
- Osuka, S.; Razzaque, M.S. Can features of phosphate toxicity appear in normophosphatemia? J. Bone Miner. Metab. 2012, 30, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Ohnishi, M.; Razzaque, M.S. Dietary and genetic evidence for phosphate toxicity accelerating mammalian aging. FASEB J. 2010, 24, 3562–3571. [Google Scholar] [CrossRef] [PubMed]
- Razzaque, M.S. Phosphate toxicity: New insights into an old problem. Clin. Sci. 2011, 120, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.B.; Razzaque, M.S. Phosphate toxicity and tumorigenesis. Biochimica et Biophysica Acta (BBA)-Rev. Cancer 2018, 1869, 303–309. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.B.; Razzaque, M.S. Dysregulation of phosphate metabolism and conditions associated with phosphate toxicity. Bonekey Rep. 2015, 4, 705. [Google Scholar] [CrossRef] [PubMed]
- D’Arcangelo, M.; Brustugun, O.; Xiao, Y.; Choi, Y.; Behrens, C.; Solis, L.; Wang, Y.; Firestein, R.; Boyle, T.; Lund-Iversen, M. 194 Prevalence and prognostic significance of sodium-dependent phosphate transporter 2B (NaPi2B) protein expression in non-small cell lung cancer (NSCLC). Ann. Oncol. 2014, 25, iv66–iv67. [Google Scholar] [CrossRef]
- Jacquemet, G.; Hamidi, H.; Ivaska, J. Filopodia in cell adhesion, 3D migration and cancer cell invasion. Curr. Opin. Cell Biol. 2015, 36, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Kuang, Y.; Nagy, J.D.; Elser, J.J. Biological stoichiometry of tumor dynamics: Mathematical models and analysis. Discret. Contin. Dyn. Syst. Ser. B 2004, 4, 221–240. [Google Scholar]
- Ward, D.; Griffin, A. Phosphorus incorporation into nucleic acids and proteins of liver nuclei of normal and azo dye-fed rats. Cancer Res. 1955, 15, 456–461. [Google Scholar] [PubMed]
- Wang, H.; Peng, R.; Wang, J.; Qin, Z.; Xue, L. Circulating microRNAs as potential cancer biomarkers: The advantage and disadvantage. Clin. Epigenetics 2018, 10, 59. [Google Scholar] [CrossRef] [PubMed]
- Papaloucas, C.D.; Papaloucas, M.D.; Kouloulias, V.; Neanidis, K.; Pistevou-Gompaki, K.; Kouvaris, J.; Zygogianni, A.; Mystakidou, K.; Papaloucas, A.C. Measurement of blood phosphorus: A quick and inexpensive method for detection of the existence of cancer in the body. Too good to be true, or forgotten knowledge of the past? Med. Hypotheses 2014, 82, 24–25. [Google Scholar] [CrossRef] [PubMed]
- Wilson, K.M.; Shui, I.M.; Mucci, L.A.; Giovannucci, E. Calcium and phosphorus intake and prostate cancer risk: A 24-y follow-up study. Am. J. Clin. Nutr. 2015, 101, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Camalier, C.E.; Young, M.R.; Bobe, G.; Perella, C.M.; Colburn, N.H.; Beck, G.R. Elevated phosphate activates N-ras and promotes cell transformation and skin tumorigenesis. Cancer Prev. Res. 2010, 3, 359–370. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.; Xu, C.-X.; Lim, H.-T.; Park, S.-J.; Shin, J.-Y.; Chung, Y.-S.; Park, S.-C.; Chang, S.-H.; Youn, H.-J.; Lee, K.-H. High dietary inorganic phosphate increases lung tumorigenesis and alters Akt signaling. Am. J. Respir. Crit. Care Med. 2009, 179, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Z.; Hu, J.; King, J.; Jay, G.; Campbell, T. Inhibition of hepatocellular carcinoma development in hepatitis B virus transfected mice by low dietary casein. Hepatology 1997, 26, 1351–1354. [Google Scholar] [PubMed]
- Reeves, P.G.; Nielsen, F.H.; Fahey, G.C., Jr. AIN-93 purified diets for laboratory rodents: Final report of the American Institute of Nutrition ad hoc writing committee on the reformulation of the AIN-76A rodent diet. J. Nutr. 1993, 123, 1939–1951. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; McKinnon, K.E.; Ha, S.W.; Beck, G.R. Inorganic phosphate induces cancer cell mediated angiogenesis dependent on forkhead box protein C2 (FOXC2) regulated osteopontin expression. Mol. Carcinog. 2015, 54, 926–934. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Cheng, H.; Roberts, T.M.; Zhao, J.J. Targeting the phosphoinositide 3-kinase (PI3K) pathway in cancer, Nature reviews. Drug Discov. 2009, 8, 627. [Google Scholar] [CrossRef] [PubMed]
- Chudek, J.; Nagy, A.; Kokot, F.; Podwinski, A.; Wiecek, A.; Ritz, E.; Kovacs, G. Phosphatemia is related to chromosomal aberrations of parathyroid glands in patients with hyperparathyroidism. J. Nephrol. 2007, 20, 164–172. [Google Scholar] [PubMed]
- Bobko, A.A.; Eubank, T.D.; Driesschaert, B.; Dhimitruka, I.; Evans, J.; Mohammad, R.; Tchekneva, E.E.; Dikov, M.M.; Khramtsov, V.V. Interstitial Inorganic Phosphate as a Tumor Microenvironment Marker for Tumor Progression. Sci. Rep. 2017, 7, 41233. [Google Scholar] [CrossRef] [PubMed]
- Yamada, S.; Tokumoto, M.; Tatsumoto, N.; Taniguchi, M.; Noguchi, H.; Nakano, T.; Masutani, K.; Ooboshi, H.; Tsuruya, K.; Kitazono, T. Phosphate overload directly induces systemic inflammation and malnutrition as well as vascular calcification in uremia. Am. J. Physiol.-Renal Physiol. 2014, 306, F1418–F1428. [Google Scholar] [CrossRef] [PubMed]
- NIDCR. Periodontal (Gum) Disease. 2018. Available online: https://www.nidcr.nih.gov/research/data-statistics/periodontal-disease (accessed on 28 December 2018).
- Pendyala, G.; Joshi, S.; Chaudhari, S.; Gandhage, D. Links demystified: Periodontitis and cancer. Dent. Res. J. 2013, 10, 704. [Google Scholar]
- Akcalı, A.; Lang, N.P. Dental calculus: The calcified biofilm and its role in disease development. Periodontology 2000 2018, 76, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Mendes, L.; Azevedo, N.F.; Felino, A.; Pinto, M.G. Relationship between invasion of the periodontium by periodontal pathogens and periodontal disease: A systematic review. Virulence 2015, 6, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Askar, A.M. Hyperphosphatemia: The hidden killer in chronic kidney disease. Saudi Med. J. 2015, 36, 13. [Google Scholar] [CrossRef] [PubMed]
- Iff, S.; Craig, J.C.; Turner, R.; Chapman, J.R.; Wang, J.J.; Mitchell, P.; Wong, G. Reduced Estimated GFR and Cancer Mortality. Am. J. Kidney Dis. 2014, 63, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Linden, G.J.; Lyons, A.; Scannapieco, F.A. Periodontal systemic associations: Review of the evidence. J. Periodontol. 2013, 84, S8–S19. [Google Scholar] [CrossRef] [PubMed]
- Grubbs, V.; Vittinghoff, E.; Taylor, G.; Kritz-Silverstein, D.; Powe, N.; Bibbins-Domingo, K.; Ishani, A.; Cummings, S.R. The association of periodontal disease with kidney function decline: A longitudinal retrospective analysis of the MrOS dental study. Nephrol. Dial. Transpl. 2015, 31, 466–472. [Google Scholar] [CrossRef] [PubMed]
- Cholewa, M.; Madziarska, K.; Radwan-Oczko, M. The association between periodontal conditions, inflammation, nutritional status and calcium-phosphate metabolism disorders in hemodialysis patients. J. Appl. Oral Sci. 2018, 26, e20170495. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.C.; Lu, C.L.; Lu, K.C. Mineral bone disorders in chronic kidney disease. Nephrology 2018, 23, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Kanjevac, T.; Bijelic, B.; Brajkovic, D.; Vasovic, M.; Stolic, R. Impact of Chronic Kidney Disease Mineral and Bone Disorder on Jaw and Alveolar Bone Metabolism: A Narrative Review. Oral Health Prev. Dent. 2018, 16, 79–85. [Google Scholar] [PubMed]
- Ausavarungnirun, R.; Wisetsin, S.; Rongkiettechakorn, N.; Chaichalermsak, S.; Udompol, U.; Rattanasompattikul, M. Association of dental and periodontal disease with chronic kidney disease in patients of a single, tertiary care centre in Thailand. BMJ Open 2016, 6, e011836. [Google Scholar] [CrossRef] [PubMed]
- Zitt, E.; Lamina, C.; Sturm, G.; Knoll, F.; Lins, F.; Freistätter, O.; Kronenberg, F.; Lhotta, K.; Neyer, U. Interaction of time-varying albumin and phosphorus on mortality in incident dialysis patients. Clin. J. Am. Soc. Nephrol. 2011, 6, 2650–2656. [Google Scholar] [CrossRef] [PubMed]
- Lieverse, A.R. Diet and the aetiology of dental calculus. Int. J. Osteoarchaeol. 1999, 9, 219–232. [Google Scholar] [CrossRef]
- Tawfig, A. Dental Calculus Formation among Recurrent Renal Calculi Formers. Int. J. Dent. Oral Heal 2017, 3, 1–7. [Google Scholar] [CrossRef]
- Lim, E.; Hyun, S.; Lee, J.M.; Kim, S.; Lee, M.-J.; Lee, S.-M.; Oh, Y.-S.; Park, I.; Shin, G.-T.; Kim, H. Effects of education on low-phosphate diet and phosphate binder intake to control serum phosphate among maintenance hemodialysis patients: A randomized controlled trial. Kidney Res. Clin. Pract. 2018, 37, 69. [Google Scholar] [CrossRef] [PubMed]
- Martins, C.; Siqueira, W.L.; Oliveira, E.; Nicolau, J.; Primo, L.G. Dental calculus formation in children and adolescents undergoing hemodialysis. Pediatr. Nephrol. 2012, 27, 1961–1966. [Google Scholar] [CrossRef] [PubMed]
- Fiyaz, M.; Ramesh, A.; Ramalingam, K.; Thomas, B.; Shetty, S.; Prakash, P. Association of salivary calcium, phosphate, pH and flow rate on oral health: A study on 90 subjects. J. Indian Soc. Periodontol. 2013, 17, 454. [Google Scholar] [CrossRef] [PubMed]
- Panta, P.; Venna, V.R. Salivary RNA signatures in oral cancer detection. Anal. Cell. Pathol. 2014, 2014, 450629. [Google Scholar] [CrossRef] [PubMed]
- Kamak, G.; Yildirim, E.; Rencber, E. Evaluation of the relationship between periodontal risk and carotid artery calcifications on panoramic radiographs. Eur. J. Dent. 2015, 9, 483. [Google Scholar] [PubMed]
- Sen, S.; Chung, M.; Duda, V.; Giamberardino, L.; Hinderliter, A.; Offenbacher, S. Periodontal disease associated with aortic arch atheroma in patients with stroke or transient ischemic attack. J. Stroke Cerebrovasc. Dis. 2017, 26, 2137–2144. [Google Scholar] [CrossRef] [PubMed]
- Mashalkar, V.N.; Suragimath, G.; Zope, S.A.; Varma, S.A. A Cross-Sectional Study to Assess and Correlate Osteoporosis and Periodontitis among Postmenopausal Women: A Dual Energy X-Ray Absorptiometry Study. J. Mid-Life Health 2018, 9, 2. [Google Scholar]
- Ayed, M.S.; Shafiq, S.S.; Diab, H.M.; Alahmari, A.D.; Divakar, D.D. Assessing periapical dental radiographs as a screening parameter for early indications of osteoporosis in postmenopausal periodontal patients and root surface evaluation using spectrochemical analysis. Saudi Med. J. 2018, 39, 719. [Google Scholar] [CrossRef] [PubMed]
- Albandar, J.M.; Susin, C.; Hughes, F.J. Manifestations of systemic diseases and conditions that affect the periodontal attachment apparatus: Case definitions and diagnostic considerations. J. Clin. Periodontol. 2018, 45, S171–S189. [Google Scholar] [CrossRef] [PubMed]
- Penoni, D.C.; Torres, S.R.; Vettore, M.V.; Leão, A.T.T. Osteoporosis, tooth loss and functional dentition in elderly women: A cross-sectional study. Revista Científica do CRO-RJ (Rio de Janeiro Dent. J.) 2018, 3, 26–31. [Google Scholar]
- Bernal, M.; Elenkova, M.; Evensky, J.; Stein, S.H. Periodontal Disease and Osteoporosis-Shared Risk Factors and Potentiation of Pathogenic Mechanisms. Curr. Oral Health Rep. 2018, 5, 26–32. [Google Scholar] [CrossRef]
- Anand, P.; Sukul, S.; Kamra, P. Periodontal Diseases and Systemic Conditions: A Comprehensive Review. J. Adv. Med. Dent. Sci. Res. 2018, 6, 73–75. [Google Scholar]
- Lee, Y.-H.; Choi, J.-O.; Nam, S.-H. Relationship between periodontal diseases and bone diseases in some bone disease patients. Biomed. Res. 2018, 29, 2818–2824. [Google Scholar]
- Savić Pavičin, I.; Dumančić, J.; Jukić, T.; Badel, T. The relationship between periodontal disease, tooth loss and decreased skeletal bone mineral density in ageing women. Gerodontology 2017, 34, 441–445. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.B.; Razzaque, M.S. Chapter 31—Endocrine Regulation of Phosphate Homeostasis. In Textbook of Nephro-Endocrinology, 2nd ed.; Singh, A.K., Williams, G.H., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 539–548. [Google Scholar]
- Cavalier, E. Bone markers and chronic kidney diseases. J. Lab. Precis. Med. 2018, 3, 62. [Google Scholar] [CrossRef]
- Chen, Y.-C.; Sosnoski, D.M.; Mastro, A.M. Breast cancer metastasis to the bone: Mechanisms of bone loss. Breast Cancer Res. 2010, 12, 215. [Google Scholar] [CrossRef] [PubMed]
- Virchow, R. Kalk-metastasen. In Archiv für Pathologische Anatomie und Physiologie und für Klinische Medicin; Springer: Berlin, Germany, 1855; Volume 8, pp. 103–113. [Google Scholar]
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brown, R.B. Dysregulated Phosphate Metabolism, Periodontal Disease, and Cancer: Possible Global Health Implications. Dent. J. 2019, 7, 18. https://doi.org/10.3390/dj7010018
Brown RB. Dysregulated Phosphate Metabolism, Periodontal Disease, and Cancer: Possible Global Health Implications. Dentistry Journal. 2019; 7(1):18. https://doi.org/10.3390/dj7010018
Chicago/Turabian StyleBrown, Ronald B. 2019. "Dysregulated Phosphate Metabolism, Periodontal Disease, and Cancer: Possible Global Health Implications" Dentistry Journal 7, no. 1: 18. https://doi.org/10.3390/dj7010018
APA StyleBrown, R. B. (2019). Dysregulated Phosphate Metabolism, Periodontal Disease, and Cancer: Possible Global Health Implications. Dentistry Journal, 7(1), 18. https://doi.org/10.3390/dj7010018




