Comparison between Two Assessment Tests for Oral Hygiene: Adenosine Triphosphate + Adenosine Monophosphate Swab Test and Bacteria Number Counting by Dielectrophoretic Impedance Measurement
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
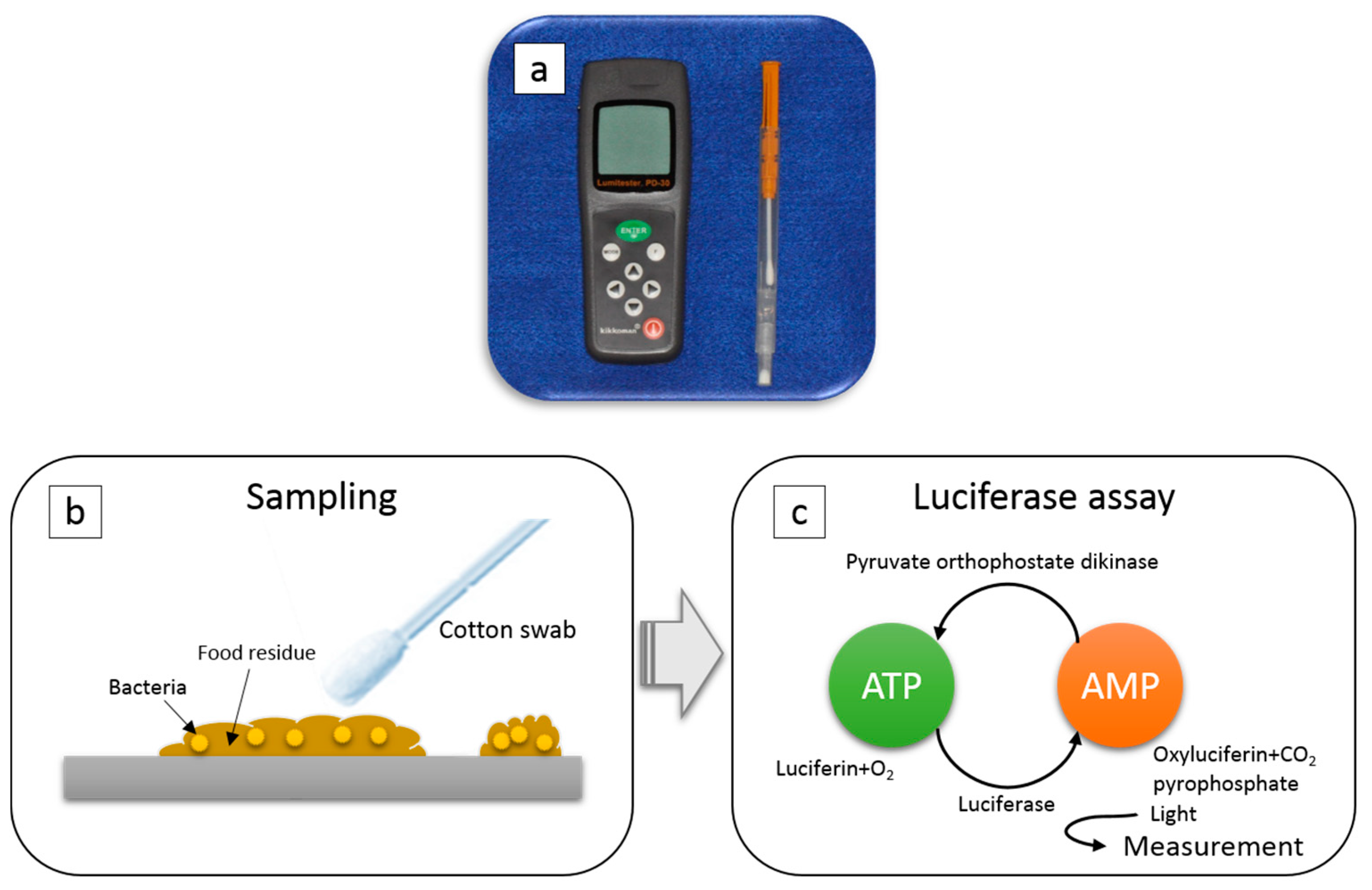
2.2. ATP + AMP Swab Test
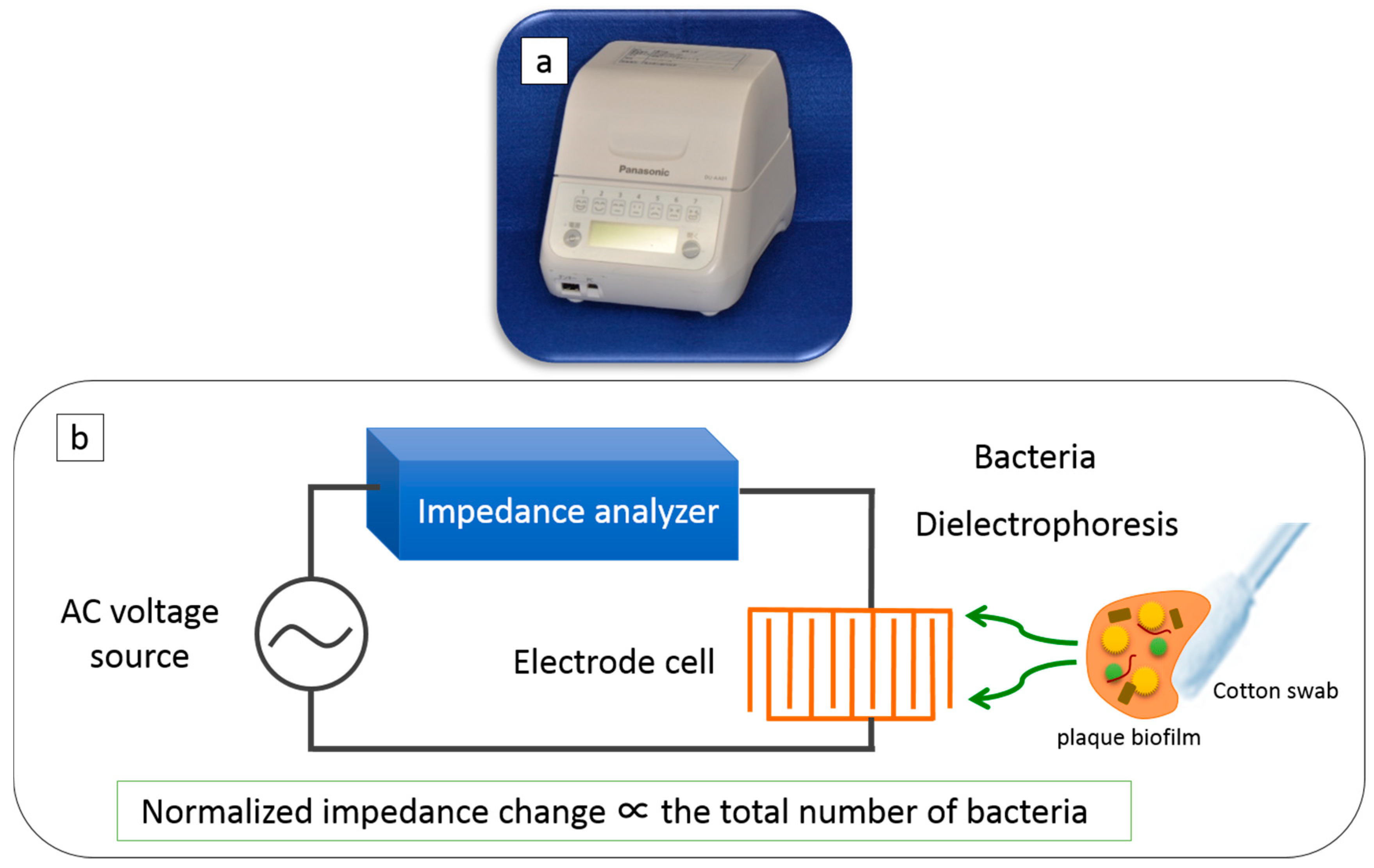
2.3. Counting Bacterial Number Using DEPIM
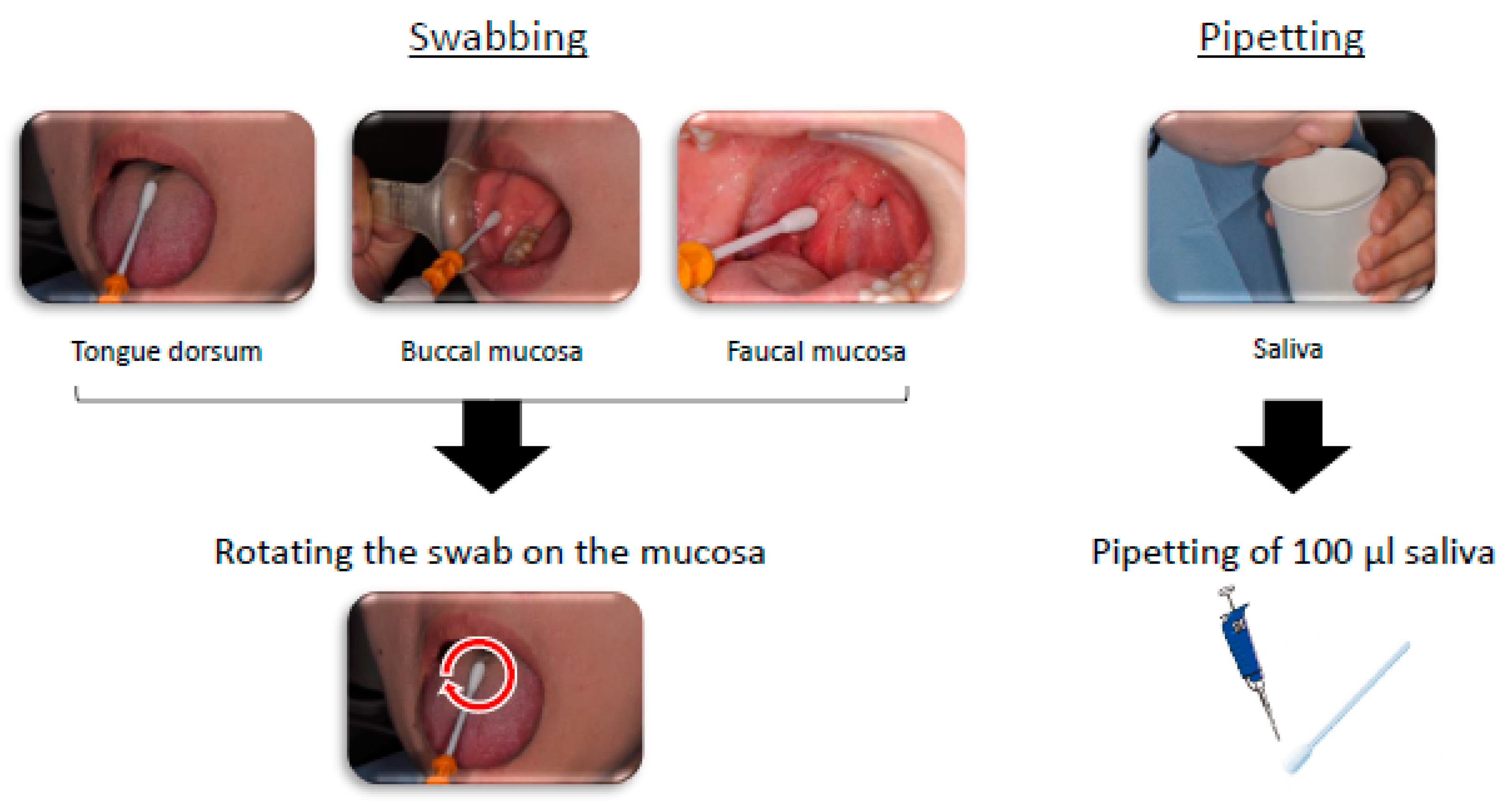
2.4. Sampling of Oral Bacteria
2.5. Subjective Assessment of Oral Hygiene
2.6. Statistical Analysis
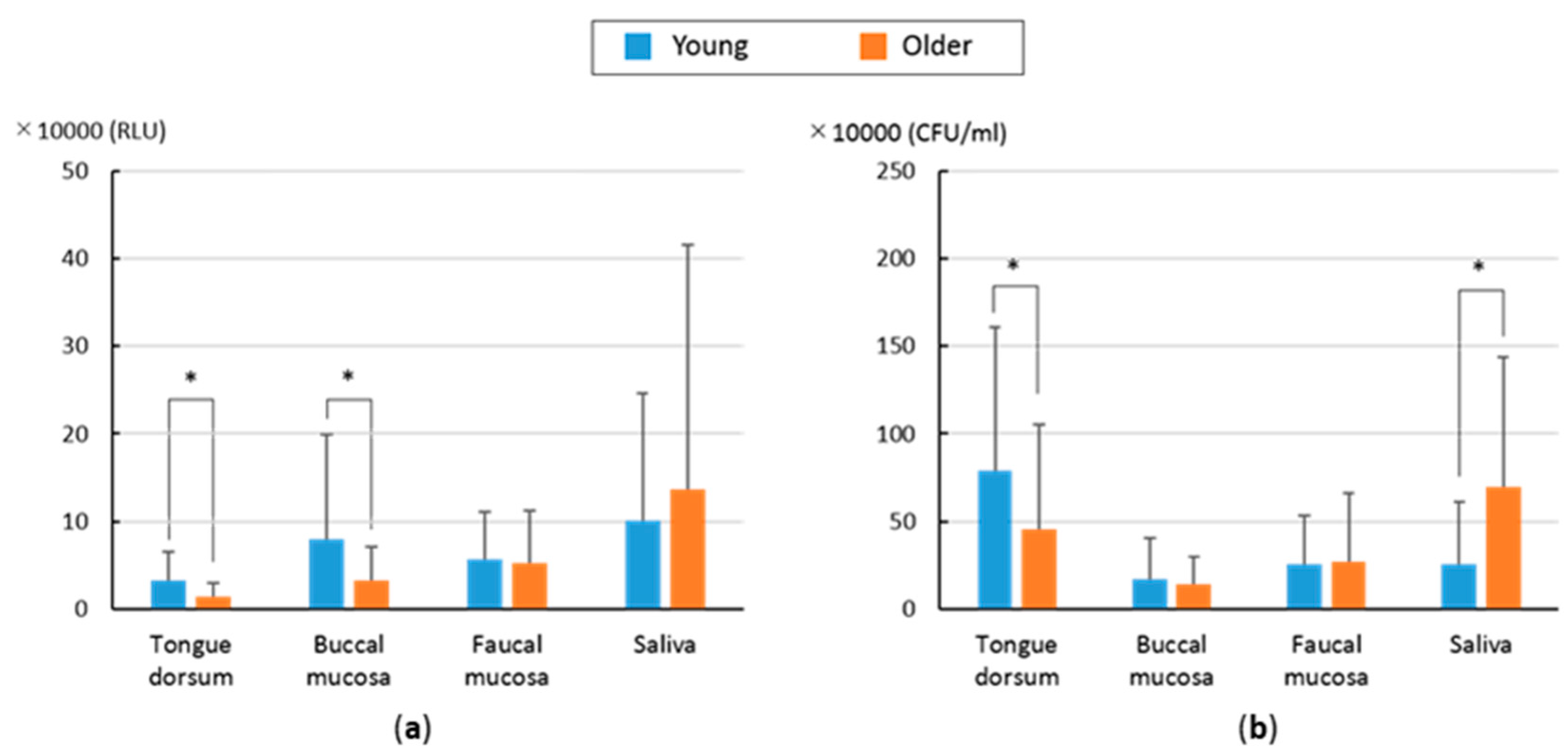
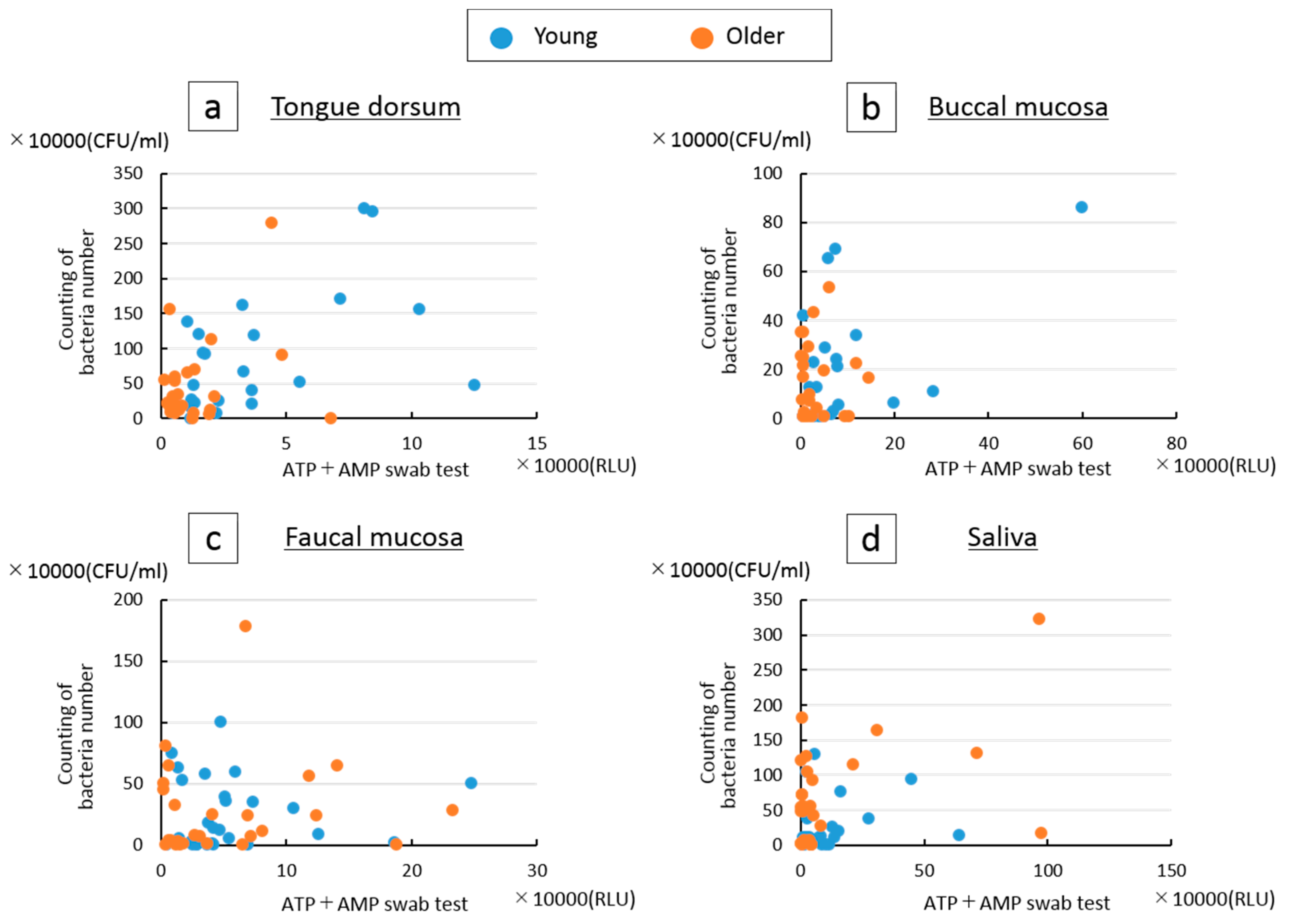
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Beaudette, J.R.; Fritz, P.C.; Sullivan, P.J.; Ward, W.E. Oral Health, Nutritional Choices, and Dental Fear and Anxiety. Dent. J. 2017, 5, 8. [Google Scholar] [CrossRef] [PubMed]
- Meurman, J.H.; Hämäläinen, P. Oral health and morbidity—Implications of oral infections on the elderly. Gerodontology 2006, 1, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Müller, F. Oral Hygiene Reduces the Mortality from Aspiration Pneumonia in Frail Elders. J. Dent. Res. 2015, 94, 14S–16S. [Google Scholar] [CrossRef] [PubMed]
- van der Maarel-Wierink, C.D.; Vanobbergen, J.N.; Bronkhorst, E.M.; Schols, J.M.; de Baat, C. Oral health care and aspiration pneumonia in frail older people: A systematic literature review. Gerodontology 2013, 1, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, P.U.; Larsen, P.; Håkonsen, S.J. The effectiveness of systematic perioperative oral hygiene in reduction of postoperativerespiratory tract infections after elective thoracic surgery in adults: A systematic review. JBI Database Syst. Rev. Implement. Rep. 2016, 14, 140–173. [Google Scholar] [CrossRef] [PubMed]
- Shigeishi, H.; Ohta, K.; Fujimoto, S.; Nakagawa, T.; Mizuta, K.; Ono, S.; Shimasue, H.; Ninomiya, Y.; Higashikawa, K.; Tada, M.; et al. Preoperative oral health care reduces postoperative inflammation and complications in oralcancer patients. Exp. Ther. Med. 2016, 12, 1922–1928. [Google Scholar] [CrossRef] [PubMed]
- Abe, S.; Ishihara, K.; Adachi, M.; Sasaki, H.; Tanaka, K.; Okuda, K. Professional oral care reduces influenza infection in elderly. Arch. Gerontol. Geriatr. 2006, 43, 157–164. [Google Scholar] [CrossRef]
- Shimizu, T.; Ueda, T.; Sakurai, K. New method for evaluation of tongue-coating status. J. Oral Rehabil. 2007, 34, 442–447. [Google Scholar] [CrossRef]
- Abe, S.; Ishihara, K.; Adachi, M.; Okuda, K. Oral hygiene evaluation for effective oral care in preventing pneumonia in dentate elderly. Arch. Gerontol. Geriatr. 2006, 43, 53–64. [Google Scholar] [CrossRef]
- Kleinfelder, J.W.; Müller, R.F.; Lange, D.E. Antibiotic susceptibility of putative periodontal pathogens in advanced periodontitis patients. J. Clin. Periodontol. 1999, 26, 347–351. [Google Scholar] [CrossRef]
- Funahara, M.; Hayashida, S.; Sakamoto, Y.; Yanamoto, S.; Kosai, K.; Yanagihara, K.; Umeda, M. Efficacy of topical antibiotic administration on the inhibition of perioperative oral bacterial growth in oral cancer patients: A preliminary study. Int. J. Oral Maxillofac. Surg. 2015, 44, 1225–1230. [Google Scholar] [CrossRef] [PubMed]
- Hirota, K.; Inagaki, S.; Hamada, R.; Ishihara, K.; Miyake, Y. Evaluation of a rapid oral bacteria quantification system using dielectrophoresis and the impedance measurement. Biocontrol Sci. 2014, 19, 45–49. [Google Scholar] [CrossRef] [PubMed]
- Suehiro, J.; Yatsunami, R.; Hamada, R.; Hara, M. Quantitative estimation of biological cell concentration suspended in aqueous medium by using dielectrophoretic impedance measurement method. J. Phys. D Appl. Phys. 1999, 32, 2814–2820. [Google Scholar] [CrossRef]
- Fazilat, S.; Sauerwein, R.; McLeod, J.; Finlayson, T.; Adam, E.; Engle, J.; Gagneja, P.; Maier, T.; Machida, C.A. Application of adenosine triphosphate-driven bioluminescence for quantification of plaque bacteria and assessment of oral hygiene in children. Pediatr. Dent. 2010, 32, 195–204. [Google Scholar] [PubMed]
- Kikutani, T.; Tamura, F.; Takahashi, Y.; Konishi, K.; Hamada, R. A novel rapid oral bacteria detection apparatus for effective oral care to prevent pneumonia. Gerodontology 2012, 29, e560–e565. [Google Scholar] [CrossRef] [PubMed]
- Hamada, R.; Suehiro, J.; Nakano, M.; Kikutani, T.; Konishi, K. Development of rapid oral bacteria detection apparatus based on dielectrophoretic impedance measurement method. IET Nanobiotechnol. 2011, 5, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, M.C.; Llama-Palacios, A.; Marín, M.J.; Figuero, E.; León, R.; Blanc, V.; Herrera, D.; Sanz, M. Validation of ATP bioluminescence as a tool to assess antimicrobial effects of mouthrinses in an in vitro subgingival-biofilm model. Med. Oral Patol. Oral Cir. Bucal 2013, 18, e86–e92. [Google Scholar] [CrossRef] [PubMed]
- Champiat, D.; Matas, N.; Monfort, B.; Fraass, H. Applications of biochemiluminescence to HACCP. Luminescence 2001, 16, 193–198. [Google Scholar] [CrossRef]
- Löe, H. The gingival index, the plaque index and the retention index systems. J. Periodontol. 1967, 38, 610–616. [Google Scholar] [CrossRef]
- Kikutani, T.; Tamura, F.; Tashiro, H.; Yoshida, M.; Konishi, K.; Hamada, R. Relationship between oral bacteria count and pneumonia onset in elderly nursing home residents. Geriatr. Gerontol. Int. 2015, 15, 417–421. [Google Scholar] [CrossRef]
- Tajima, S.; Ryu, M.; Ogami, K.; Ueda, T.; Sakurai, K. Time-dependent effects of tongue cleaning with mouthwash or mouth moisturising gel on the number of microbes on the tongue surface of elders with care needs. Gerodontology 2017, 34, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Endo, Y.; Ooka, T.; Hironaka, S.; Sugiyama, T.; Matsuhashi, K.; Abe, Y.; Tatsuno, M.; Mukai, Y.; Inoue, M. Oral pathogens in children with respiratory disease. Pediatr. Dent. J. 2014, 24, 159–166. [Google Scholar] [CrossRef]
- Masuku, S.M.; Babu, D.; Martin, E.M.; Koo, O.K.; O’Bryan, C.A.; Crandall, P.G.; Ricke, S.C. Cleaning and decontamination efficacy of wiping cloths and silver dihydrogen citrate on food contact surfaces. J. Appl. Microbiol. 2012, 113, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Kouta, M.; Ogasawara, T.; Shinotsuka, K.; Iwasaki, H.; Matsumura, K.; Okada, Y.; Haishima, H.; Fujita, M.; Sumi, Y. The effect og gel-type moisturizer on the number of bacteria in saliva after tooth-brushing. J. Jpn. Soc. Disabil. Oral Health 2016, 37, 16–21. [Google Scholar]
- Gibbons, R.J.; van Houte, J. Part 4 Microbiology. In Textbook of Oral Biology; W.B. Saunders Company: Philadelphia, PA, USA, 1978; pp. 684–705. [Google Scholar]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iwawaki, Y.; Muraoka, Y.; Higashiyama, H.; Kishimoto, T.; Liu, L.; Goto, T.; Ichikawa, T. Comparison between Two Assessment Tests for Oral Hygiene: Adenosine Triphosphate + Adenosine Monophosphate Swab Test and Bacteria Number Counting by Dielectrophoretic Impedance Measurement. Dent. J. 2019, 7, 10. https://doi.org/10.3390/dj7010010
Iwawaki Y, Muraoka Y, Higashiyama H, Kishimoto T, Liu L, Goto T, Ichikawa T. Comparison between Two Assessment Tests for Oral Hygiene: Adenosine Triphosphate + Adenosine Monophosphate Swab Test and Bacteria Number Counting by Dielectrophoretic Impedance Measurement. Dentistry Journal. 2019; 7(1):10. https://doi.org/10.3390/dj7010010
Chicago/Turabian StyleIwawaki, Yuki, Yuki Muraoka, Hiroaki Higashiyama, Takahiro Kishimoto, Lipei Liu, Takaharu Goto, and Tetsuo Ichikawa. 2019. "Comparison between Two Assessment Tests for Oral Hygiene: Adenosine Triphosphate + Adenosine Monophosphate Swab Test and Bacteria Number Counting by Dielectrophoretic Impedance Measurement" Dentistry Journal 7, no. 1: 10. https://doi.org/10.3390/dj7010010
APA StyleIwawaki, Y., Muraoka, Y., Higashiyama, H., Kishimoto, T., Liu, L., Goto, T., & Ichikawa, T. (2019). Comparison between Two Assessment Tests for Oral Hygiene: Adenosine Triphosphate + Adenosine Monophosphate Swab Test and Bacteria Number Counting by Dielectrophoretic Impedance Measurement. Dentistry Journal, 7(1), 10. https://doi.org/10.3390/dj7010010




