Phosphophoryn and Dentin Sialoprotein Effects on Dental Pulp Cell Migration, Proliferation, and Differentiation
Abstract
:1. Introduction
2. Materials and Methods
2.1. PP Effect on Rat Dental Pulp Cell MRPC-1
2.1.1. Cell Culture Preparation
2.1.2. Preparation of PP and Agarose Gel Beads Containing PP
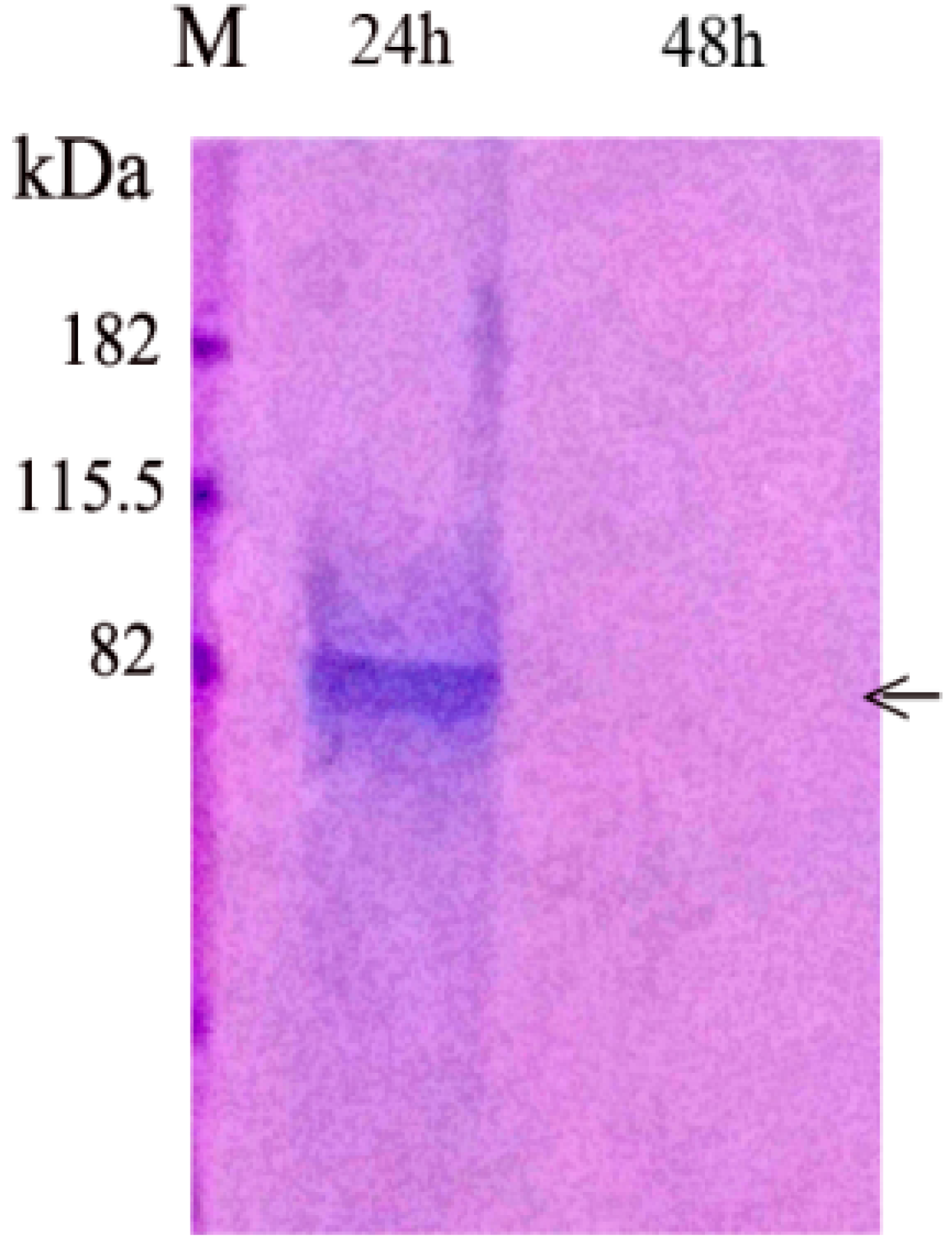
2.1.3. Release of Native PP from Agarose-1 μg PP Beads
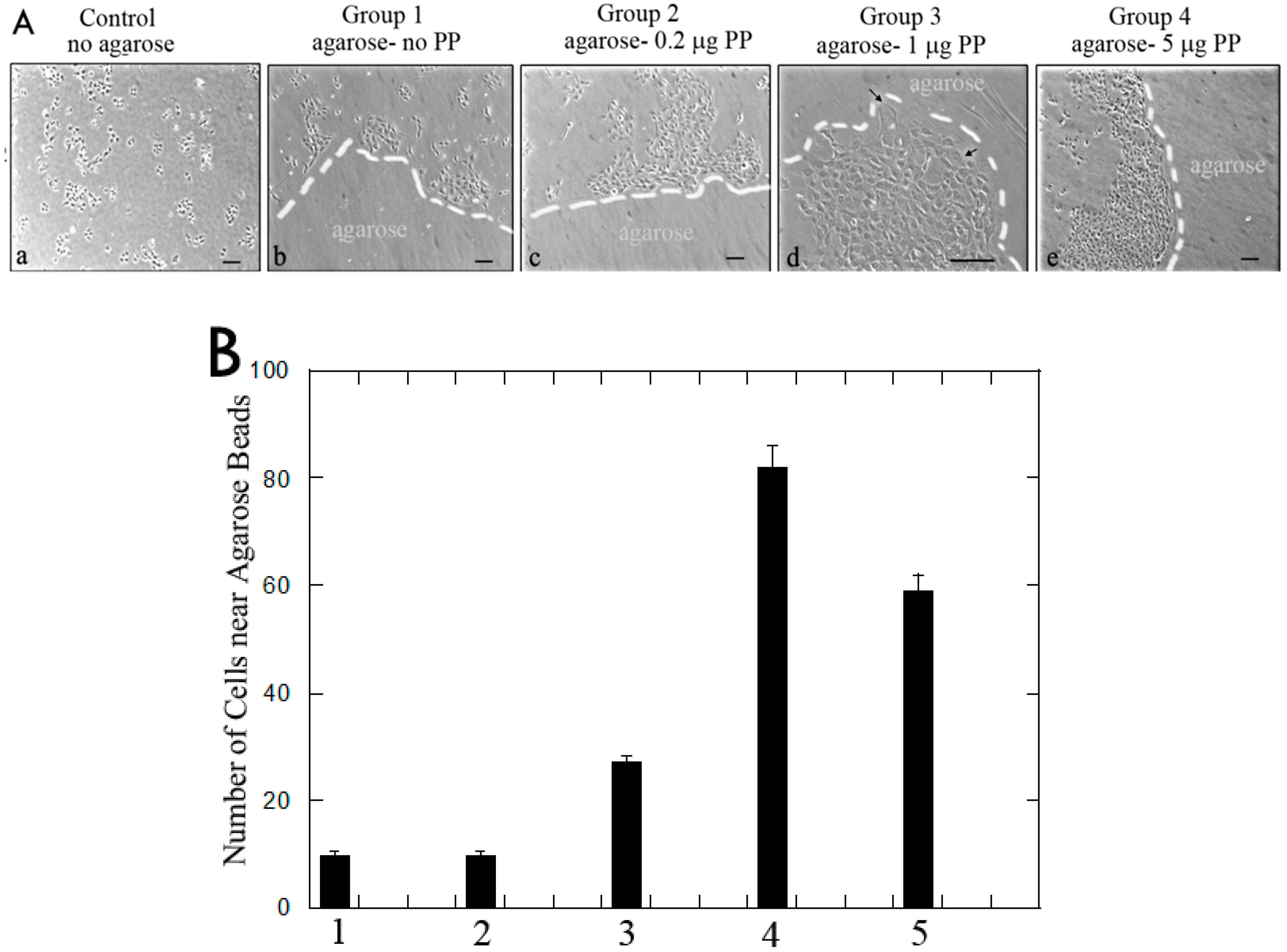
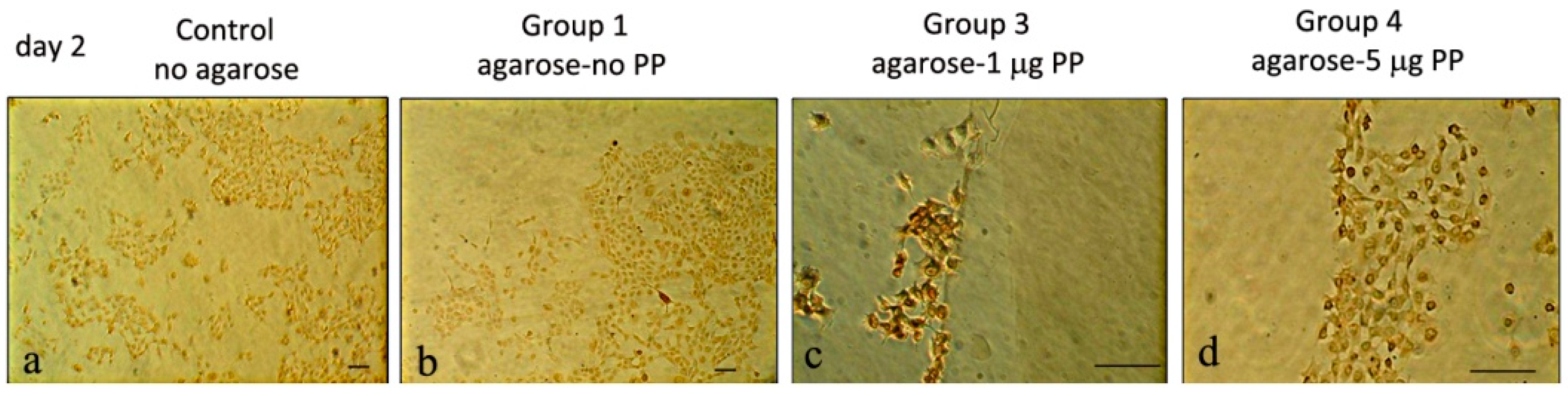
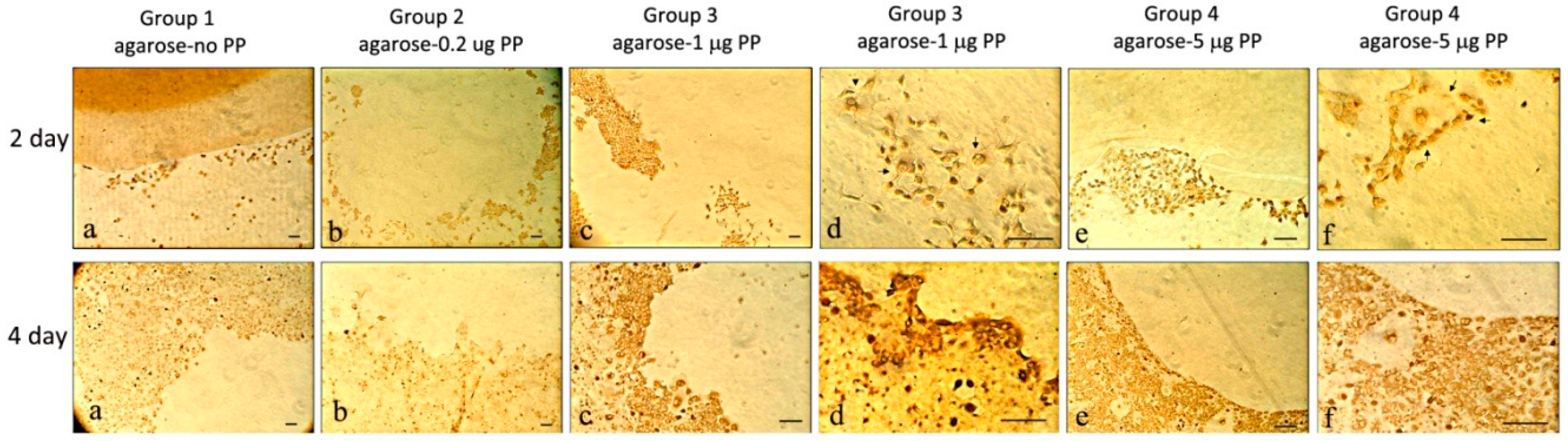
2.1.4. Cell Migration Analyses and Immunohistochemistry Analyses on MRPC-1 Cells
2.2. Effect of Recombinant DSP307 Protein and DSP/PP240 Protein Mixture on Rat M2H4 Dental Pulp Cells
2.2.1. Recombinant Protein Preparation
2.2.2. Effect of Recombinant DSP/PP on Rat Dental Pulp M2H4 Cell Proliferation
2.2.3. Detection of DSP-PP mRNA expression in rat dental pulp M2H4 cells cultured with DSP/PP240
2.2.4. M2H4 Cell Morphology Changes under Different Culture Medium
2.3. Statistical Analysis
3. Results
3.1. PP Effects on Rat Dental Pulp MRPC-1 Cells
3.1.1. PP Release from Agarose Beads
3.1.2. Rat Dental Pulp MRPC-1 Cell Migration Analysis
3.1.3. Col I and PP Expression in Rat Dental Pulp MRPC-1 Cells
3.2. Recombinant DSP/PP240 Protein Effects on M2H4 Cells
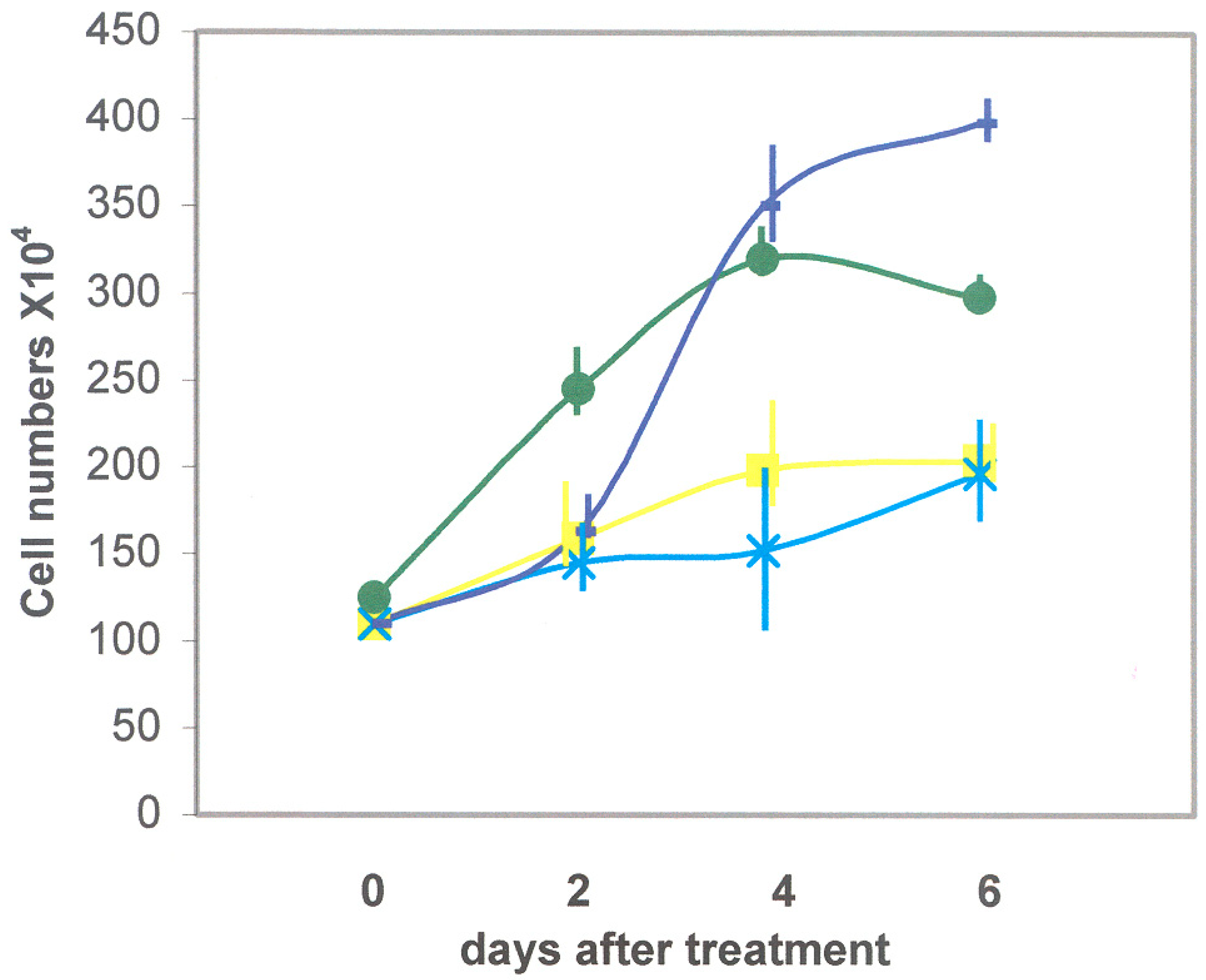
3.2.1. Recombinant DSP/PP240 Protein Effect on M2H4 Cell Proliferation
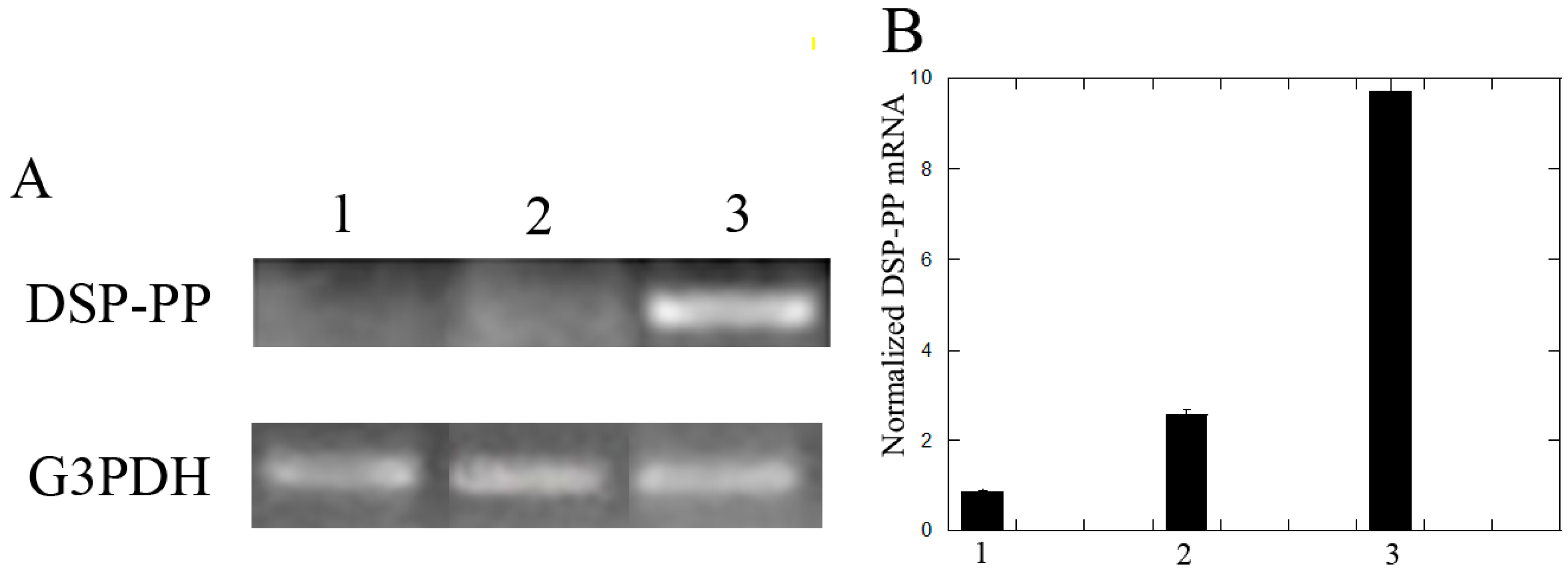
3.2.2. Recombinant DSP/PP240 Protein Mixture Plus Ascorbic Acid Up-Regulated DSP-PP mRNA Expression in M2H4 Cell Cultures
3.2.3. M2H4 Cell Morphology Changes That Occurred When Cultured in Various Medium
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Godovikova, V.; Ritchie, H.H. Dynamic processing of recombinant dentin sialoprotein-phosphophoryn protein. J. Biol. Chem. 2007, 282, 31341–31348. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, H.H.; Yee, C.T.; Tang, X.N.; Dong, Z.H.; Fuller, R.S. DSP-PP Precursor Protein Cleavage by Tolloid-Related-1 Protein and by Bone Morphogenetic Protein-1. PLoS ONE 2012, 7, e41110. [Google Scholar] [CrossRef] [PubMed]
- von Marschall, Z.; Fisher, L.W. Dentin sialophosphoprotein (DSPP) is cleaved into its two natural dentin matrix 2 products by three isoforms of bone morphogenetic protein-1 (BMP1). Matrix Biol. 2010, 29, 295–303. [Google Scholar] [CrossRef]
- Linde, A.; Lussi, A. Mineral induction by polyanionic dentin and bone proteins at physiological ionic conditions. Connect. Tissue Res. 1989, 21, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Linde, A.; Lussi, A.; Crenshaw, M.A. Mineral induction by immobilized polyanionic proteins. Calcif. Tissue Int. 1989, 44, 286–295. [Google Scholar] [CrossRef] [PubMed]
- Marsh, M.E. Self-association of calcium and magnesium complexes of dentin phosphophoryn. Biochemistry 1989, 28, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Marsh, M.E. Binding of Calcium and Phosphate Ions to Dentin Phosphophoryn. Biochemistry 1989, 28, 346–352. [Google Scholar] [CrossRef] [PubMed]
- Traub, W.; Jodaikin, A.; Arad, T.; Veis, A.; Sabsay, B. Dentin phosphophoryn binding to collagen fibrils. Matrix 1992, 12, 197–201. [Google Scholar] [CrossRef]
- Bronckers, A.L.J.J.; D’Souza, R.N.; Lyaruu, D.M.; Gay, S.; Woltgens, J.H.M. Dentin sialoprotein: Biosynthesis and developmental appearance in rat tooth germs in comparison with amelogenins, osteocalcin and collagen type-I. Cell Tissue Res. 1993, 272, 237–247. [Google Scholar] [CrossRef]
- Ritchie, H.H.; Berry, J.E.; Somerman, M.J.; Hanks, C.T.; Bronckers, A.L.J.J.; Hotton, D.; Papagerakis, P.; Berdal, A.; Butler, W.T. Dentin sialoprotein (DSP) transcripts: Developmentally sustained expression in odontoblasts and transient expression in pre-ameloblasts. Eur. J. Oral Sci. 1997, 105, 405–413. [Google Scholar] [CrossRef]
- Franceschi, R.T.; Iyer, B.S. Relationship between collagen synthesis and expression of the osteoblast phenotype in MC3T3-E1 cells. J. Bone Miner. Res. 1992, 7, 235–246. [Google Scholar] [CrossRef]
- Lundquist, P.; Ritchie, H.H.; Moore, K.; Lundgren, T.; Linde, A. Phosphate and calcium uptake by rat odontoblast-like MRPC-1 concomitant with mineralization. J. Bone Miner. Res. 2002, 17, 1801–1813. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, H.H.; Wang, L.-H. The presence of multiple rat DSP-PP transcripts. Biochim. Biophys. Acta 2000, 1493, 27–32. [Google Scholar] [CrossRef]
- Ritchie, H.H.; Wang, L.H.; Knudtson, K. A Novel Rat 523 Amino Acid Phosphophoryn: Nucleotide Sequence and Genomic Organization. Biochim. Biophys. Acta 2001, 1520, 212–222. [Google Scholar] [CrossRef]
- Ritchie, H.H.; Li, X.-R. A novel rat dentin mRNA coding only for dentin sialoprotein. Eur. J. Oral Sci. 2001, 109, 342–347. [Google Scholar] [CrossRef]
- Ritchie, H.H.; Liu, J.; Kasugai, S.; Moller, P. A mineralizing rat dental pulp cell subline expressing collagen type I and dentin sialoprotein-phosphophoryn transcripts. In Vitro Cell. Dev. Biol. 2002, 38, 25–29. [Google Scholar] [CrossRef]
- Yokota, M.; Nagata, T.; Ishida, H.; Wakano, Y. Clonal dental pulp cells (RDP 4-1, RPC-C2A) synthesize and secret osteopontin (SPP1, 2ar). Biochem. Biophys. Res. Commun. 1992, 189, 892–898. [Google Scholar] [CrossRef]
- Butler, W.T.; Finch, J.E.J.; Desteno, C.V. Chemical character of proteins in rat incisors. Biochim. Biophys. Acta 1972, 257, 167–171. [Google Scholar] [CrossRef]
- Stetler-Stevenson, W.G.; Veis, A. Type I collagen shows a specific binding affinity for bovine dentin phosphophoryn. Calcif. Tissue Int. 1986, 38, 135–141. [Google Scholar] [CrossRef]
- Bleicher, F.; Couble, M.; Farges, J.; Couble, P.; Maglorie, H. Sequential expression of matrix protein genes in developing rat teeth. Matrix Biol. 1999, 18, 133–143. [Google Scholar] [CrossRef]
- Franceschi, R.T. The role of ascorbic acid in mesenchymal differentiation. Nutr. Rev. 1992, 50, 65–70. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chuang, S.-F.; Chen, Y.-H.; Ma, P.; Ritchie, H.H. Phosphophoryn and Dentin Sialoprotein Effects on Dental Pulp Cell Migration, Proliferation, and Differentiation. Dent. J. 2018, 6, 70. https://doi.org/10.3390/dj6040070
Chuang S-F, Chen Y-H, Ma P, Ritchie HH. Phosphophoryn and Dentin Sialoprotein Effects on Dental Pulp Cell Migration, Proliferation, and Differentiation. Dentistry Journal. 2018; 6(4):70. https://doi.org/10.3390/dj6040070
Chicago/Turabian StyleChuang, Shu-Feng, Yu-Hsuan Chen, Peter Ma, and Helena H. Ritchie. 2018. "Phosphophoryn and Dentin Sialoprotein Effects on Dental Pulp Cell Migration, Proliferation, and Differentiation" Dentistry Journal 6, no. 4: 70. https://doi.org/10.3390/dj6040070
APA StyleChuang, S.-F., Chen, Y.-H., Ma, P., & Ritchie, H. H. (2018). Phosphophoryn and Dentin Sialoprotein Effects on Dental Pulp Cell Migration, Proliferation, and Differentiation. Dentistry Journal, 6(4), 70. https://doi.org/10.3390/dj6040070





