Twist1 Suppresses Cementoblast Differentiation
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture
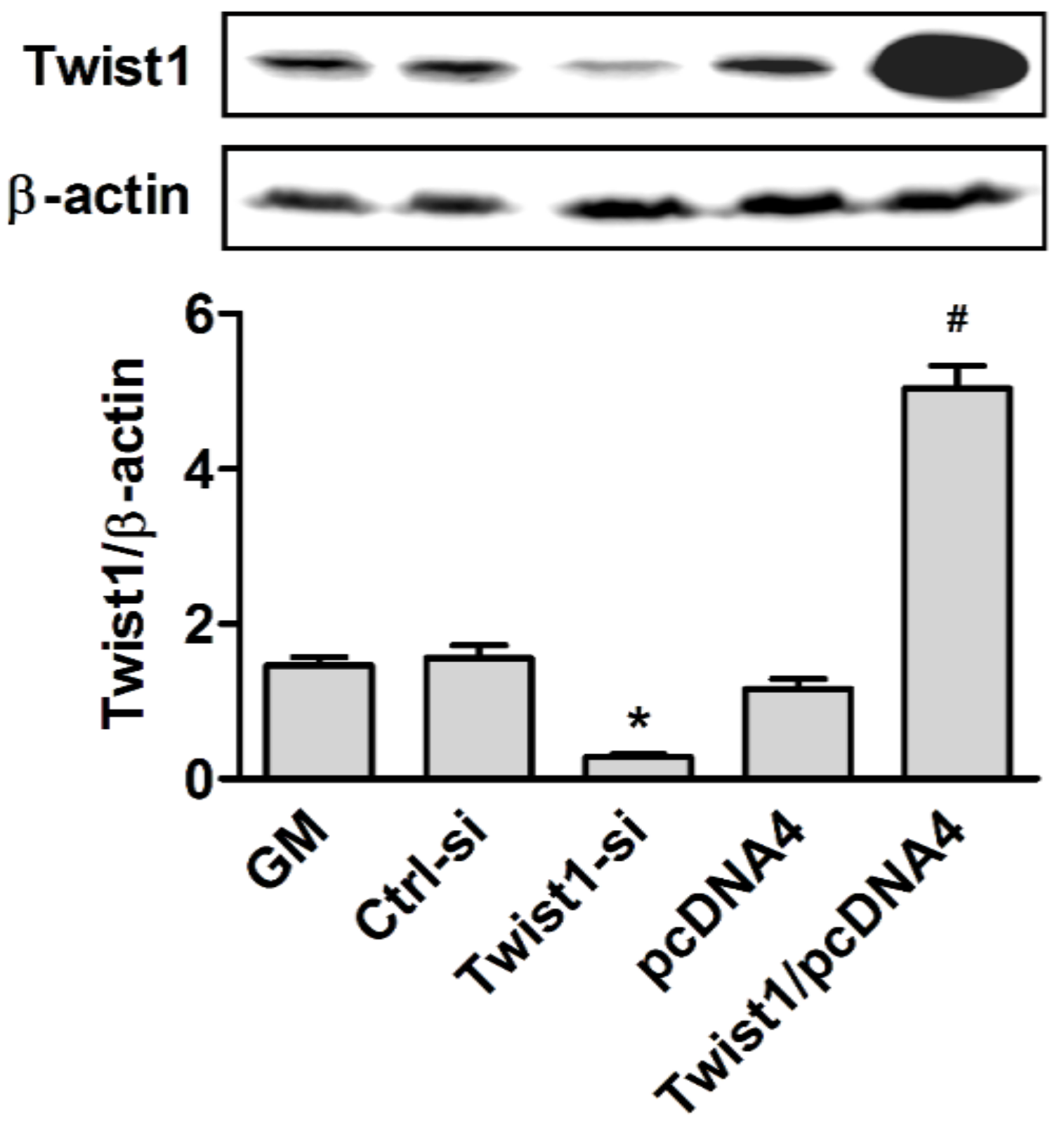
2.2. Forced Transient Overexpression or Knock-Down of Twist1
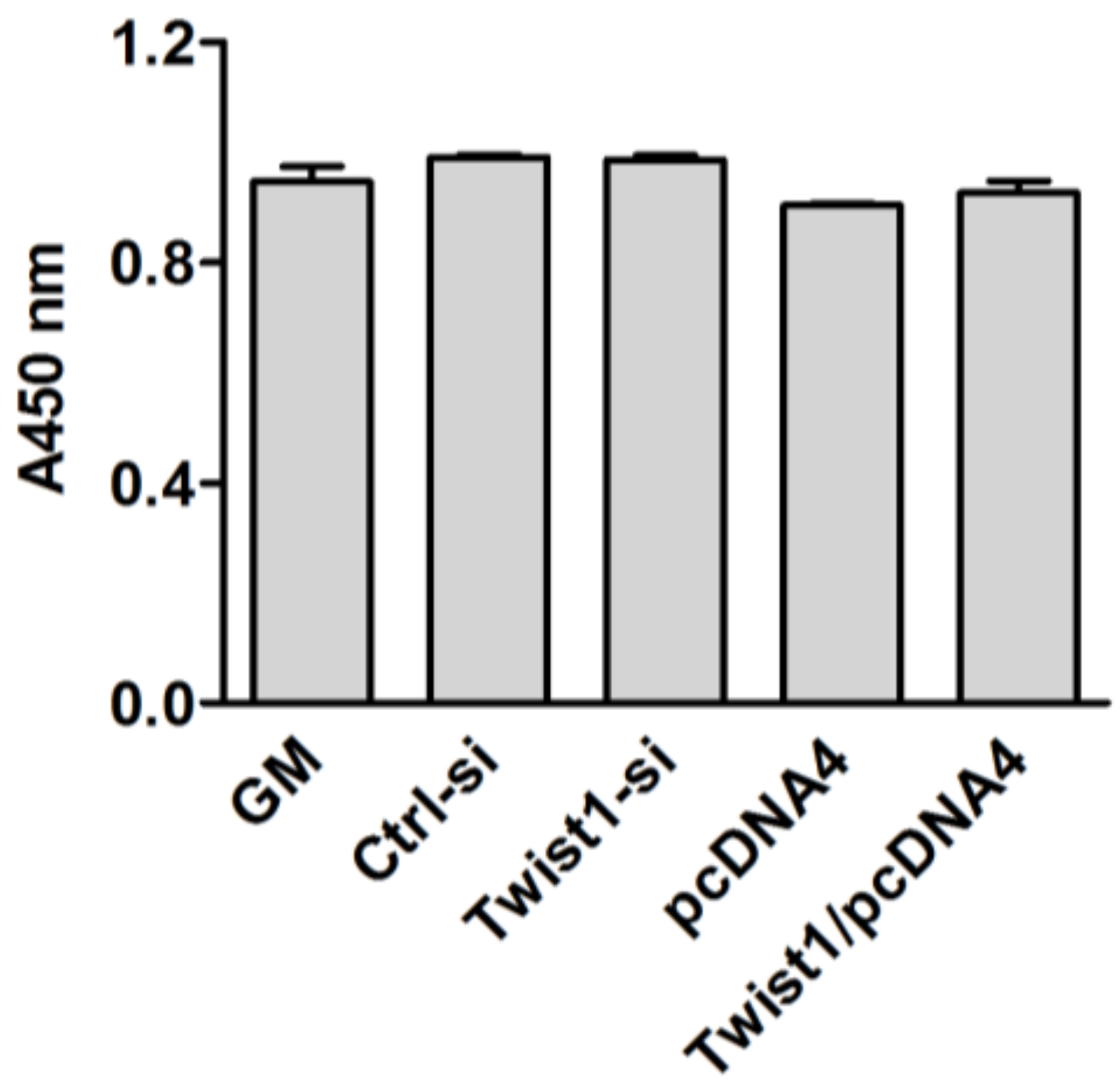
2.3. Cell Viability Assays
2.4. Western Blot Analysis
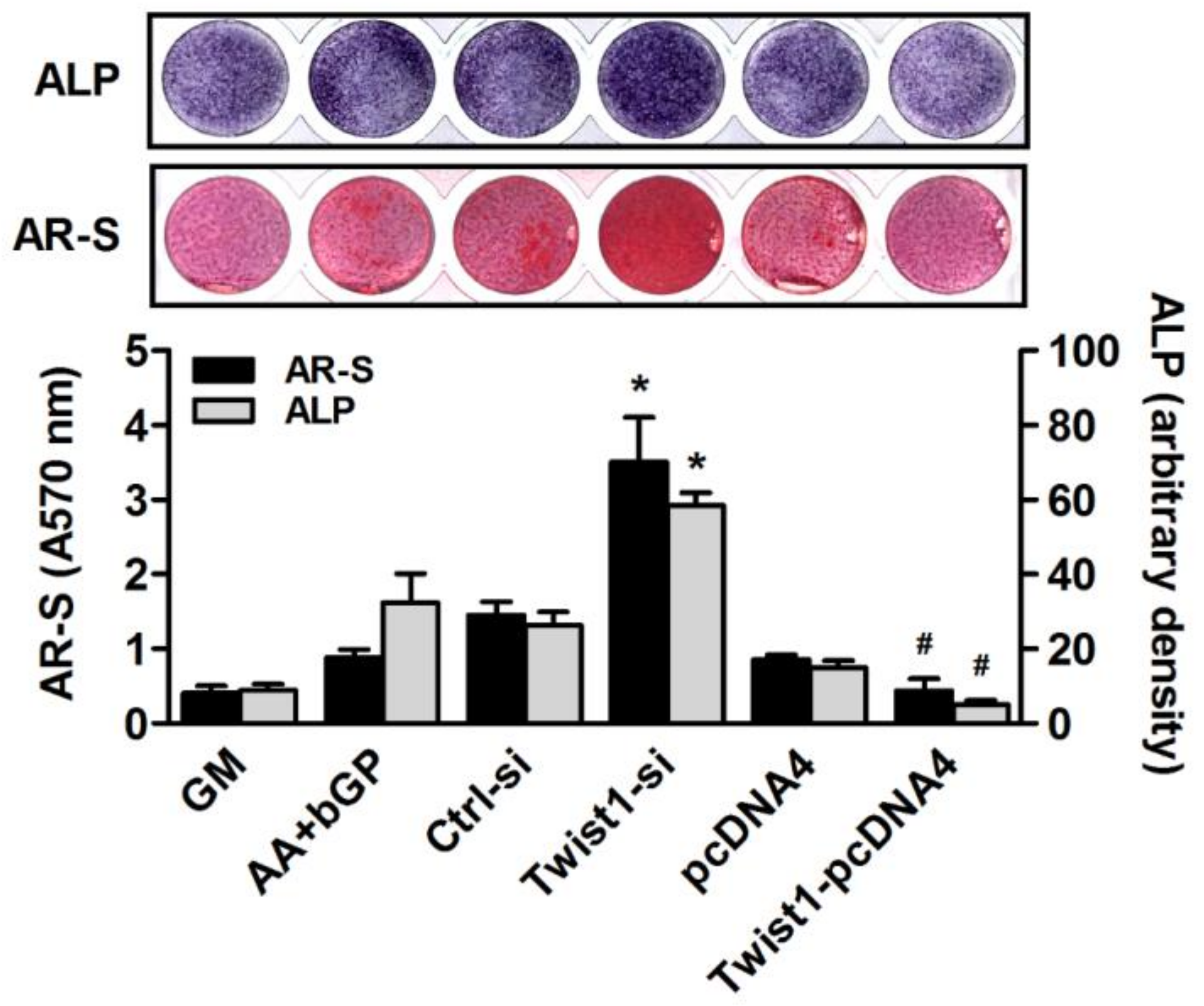
2.5. ALP Staining
2.6. Alizarin Red S Staining
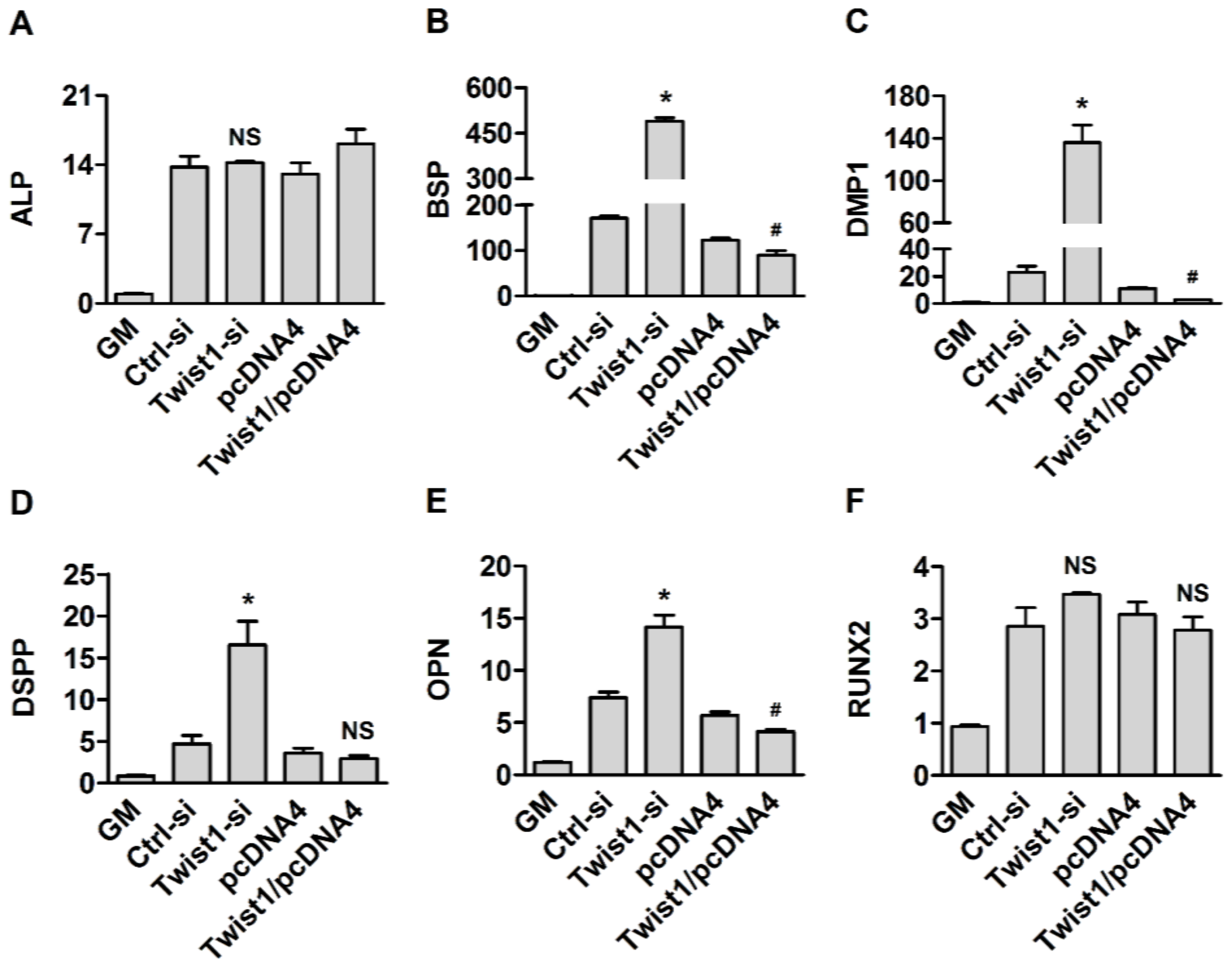
2.7. Quantitative Real Time Reverse Transcription Polymerase Chain Reaction (RT)-PCR
2.8. Statistical Analysis
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Saygin, N.E.; Giannobile, W.V.; Somerman, M.J. Molecular and cell biology of cementum. Periodontology 2000, 24, 73–98. [Google Scholar] [CrossRef]
- Bosshardt, D.D. Are cementoblasts a subpopulation of osteoblasts or a unique phenotype? J. Dent. Res. 2005, 84, 390–406. [Google Scholar] [CrossRef] [PubMed]
- D’Errico, J.A.; MacNeil, R.L.; Takata, T.; Berry, J.; Strayhorn, C.; Somerman, M.J. Expression of bone associated markers by tooth root lining cells, in situ and in vitro. Bone 1997, 20, 117–126. [Google Scholar] [CrossRef]
- D’Errico, J.A.; Berry, J.E.; Ouyang, H.; Strayhorn, C.L.; Windle, J.J.; Somerman, M.J. Employing a transgenic animal model to obtain cementoblasts in vitro. J. Periodontol. 2000, 71, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Zhang, Y.K.; Harris, S.; Ahuja, S.S.; Bonewald, L.F. MLO-Y4 osteocyte-like cells support osteoclast formation and activation. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 2002, 17, 2068–2079. [Google Scholar] [CrossRef] [PubMed]
- Kitagawa, M.; Tahara, H.; Kitagawa, S.; Oka, H.; Kudo, Y.; Sato, S.; Ogawa, I.; Miyaichi, M.; Takata, T. Characterization of established cementoblast-like cell lines from human cementum-lining cells in vitro and in vivo. Bone 2006, 39, 1035–1042. [Google Scholar] [CrossRef] [PubMed]
- Murray, S.S.; Glackin, C.A.; Winters, K.A.; Gazit, D.; Kahn, A.J.; Murray, E.J. Expression of helix-loop-helix regulatory genes during differentiation of mouse osteoblastic cells. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 1992, 7, 1131–1138. [Google Scholar] [CrossRef] [PubMed]
- Funato, N.; Ohtani, K.; Ohyama, K.; Kuroda, T.; Nakamura, M. Common regulation of growth arrest and differentiation of osteoblasts by helix-loop-helix factors. Mol. Cell. Boil. 2001, 21, 7416–7428. [Google Scholar] [CrossRef] [PubMed]
- Oshima, A.; Tanabe, H.; Yan, T.; Lowe, G.N.; Glackin, C.A.; Kudo, A. A novel mechanism for the regulation of osteoblast differentiation: Transcription of periostin, a member of the fasciclin I family, is regulated by the bHLH transcription factor, twist. J. Cell. Biochem. 2002, 86, 792–804. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.S.; Lowe, G.N.; Strong, D.D.; Wergedal, J.E.; Glackin, C.A. TWIST, a basic helix-loop-helix transcription factor, can regulate the human osteogenic lineage. J. Cell. Biochem. 1999, 75, 566–577. [Google Scholar] [CrossRef]
- Lee, M.S.; Lowe, G.; Flanagan, S.; Kuchler, K.; Glackin, C.A. Human Dermo-1 has attributes similar to twist in early bone development. Bone 2000, 27, 591–602. [Google Scholar] [CrossRef]
- Rice, R.; Thesleff, I.; Rice, D.P. Regulation of Twist, Snail, and Id1 is conserved between the developing murine palate and tooth. Dev. Dyn. Off. Publ. Am. Assoc. Anat. 2005, 234, 28–35. [Google Scholar]
- Prasad, M.; Butler, W.T.; Qin, C. Dentin sialophosphoprotein in biomineralization. Connect. Tissue Res. 2010, 51, 404–417. [Google Scholar] [CrossRef] [PubMed]
- Qin, C.; Brunn, J.C.; Cadena, E.; Ridall, A.; Tsujigiwa, H.; Nagatsuka, H.; Nagai, N.; Butler, W.T. The expression of dentin sialophosphoprotein gene in bone. J. Dent. Res. 2002, 81, 392–394. [Google Scholar] [CrossRef] [PubMed]
- Baba, O.; Qin, C.; Brunn, J.C.; Jones, J.E.; Wygant, J.N.; McIntyre, B.W.; Butler, W.T. Detection of dentin sialoprotein in rat periodontium. Eur. J. Oral Sci. 2004, 112, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Gibson, M.P.; Zhu, Q.; Liu, Q.; D’Souza, R.N.; Feng, J.Q.; Qin, C. Loss of dentin sialophosphoprotein leads to periodontal diseases in mice. J. Period. Res. 2013, 48, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Sun, H.; Song, F.; Wu, Y.; Wang, J. Yes-associated protein 1 promotes the differentiation and mineralization of cementoblast. J. Cell. Physiol. 2018, 233, 2213–2224. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T.; Phylactou, L.A.; Uney, J.B.; Ishikawa, I.; Eto, K.; Iseki, S. Twist is required for establishment of the mouse coronal suture. J. Anat. 2005, 206, 437–444. [Google Scholar] [CrossRef] [PubMed]
- Guenou, H.; Kaabeche, K.; Mee, S.L.; Marie, P.J. A role for fibroblast growth factor receptor-2 in the altered osteoblast phenotype induced by Twist haploinsufficiency in the Saethre-Chotzen syndrome. Hum. Mol. Genet. 2005, 14, 1429–1439. [Google Scholar] [CrossRef] [PubMed]
- Goho, C. Dental findings in Saethre-Chotzen syndrome: Report of case. ASDC J. Dent. Child 1998, 65, 136–137. [Google Scholar] [PubMed]
- Sosic, D.; Richardson, J.A.; Yu, K.; Ornitz, D.M.; Olson, E.N. Twist regulates cytokine gene expression through a negative feedback loop that represses NF-kappaB activity. Cell 2003, 112, 169–180. [Google Scholar] [CrossRef]
- El Ghouzzi, V.; Le Merrer, M.; Perrin-Schmitt, F.; Lajeunie, E.; Benit, P.; Renier, D.; Bourgeois, P.; Bolcato-Bellemin, A.L.; Munnich, A.; Bonaventure, J. Mutations of the TWIST gene in the Saethre-Chotzen syndrome. Nat. Genet. 1997, 15, 42–46. [Google Scholar] [CrossRef] [PubMed]
- Howard, T.D.; Paznekas, W.A.; Green, E.D.; Chiang, L.C.; Ma, N.; Ortiz de Luna, R.I.; Garcia Delgado, C.; Gonzalez-Ramos, M.; Kline, A.D.; Jabs, E.W. Mutations in TWIST, a basic helix-loop-helix transcription factor, in Saethre-Chotzen syndrome. Nat. Genet. 1997, 15, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Cakouros, D.; Raices, R.M.; Gronthos, S.; Glackin, C.A. Twist-ing cell fate: Mechanistic insights into the role of twist in lineage specification/differentiation and tumorigenesis. J. Cell. Biochem. 2010, 110, 1288–1298. [Google Scholar] [CrossRef] [PubMed]
- Qin, Q.; Xu, Y.; He, T.; Qin, C.; Xu, J. Normal and disease-related biological functions of Twist1 and underlying molecular mechanisms. Cell Res. 2012, 22, 90–106. [Google Scholar] [CrossRef] [PubMed]
- Bialek, P.; Kern, B.; Yang, X.; Schrock, M.; Sosic, D.; Hong, N.; Wu, H.; Yu, K.; Ornitz, D.M.; Olson, E.N.; et al. A twist code determines the onset of osteoblast differentiation. Dev. Cell 2004, 6, 423–435. [Google Scholar] [CrossRef]
- Yang, D.C.; Yang, M.H.; Tsai, C.C.; Huang, T.F.; Chen, Y.H.; Hung, S.C. Hypoxia inhibits osteogenesis in human mesenchymal stem cells through direct regulation of RUNX2 by TWIST. PLoS ONE 2011, 6, e23965. [Google Scholar] [CrossRef] [PubMed]
- Yousfi, M.; Lasmoles, F.; Marie, P.J. TWIST inactivation reduces CBFA1/RUNX2 expression and DNA binding to the osteocalcin promoter in osteoblasts. Biochem. Biophys. Res. Commun. 2002, 297, 641–644. [Google Scholar] [CrossRef]
- Isenmann, S.; Arthur, A.; Zannettino, A.C.; Turner, J.L.; Shi, S.; Glackin, C.A.; Gronthos, S. TWIST family of basic helix-loop-helix transcription factors mediate human mesenchymal stem cell growth and commitment. Stem Cells 2009, 27, 2457–2468. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Hassan, M.Q.; Li, Z.Y.; Stein, J.L.; Lian, J.B.; Van Wijnen, A.J.; Stein, G.S. Intricate gene regulatory networks of helix-loop-helix (HLH) proteins support regulation of bone-tissue related genes during osteoblast differentiation. J. Cell. Biochem. 2008, 105, 487–496. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Lu, Y.; Maciejewska, I.; Galler, K.M.; Cavender, A.; D’Souza, R.N. TWIST1 promotes the odontoblast-like differentiation of dental stem cells. Adv. Dent. Res. 2011, 23, 280–284. [Google Scholar] [CrossRef] [PubMed]
- Meng, T.; Huang, Y.; Wang, S.; Zhang, H.; Dechow, P.C.; Wang, X.; Qin, C.; Shi, B.; D’Souza, R.N.; Lu, Y. Twist1 is essential for tooth morphogenesis and odontoblast differentiation. J. Biol. Chem. 2015, 290, 29593–29602. [Google Scholar] [CrossRef] [PubMed]
- Arzate, H.; Olson, S.W.; Page, R.C.; Gown, A.M.; Narayanan, A.S. Production of a monoclonal antibody to an attachment protein derived from human cementum. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Boil. 1992, 6, 2990–2995. [Google Scholar] [CrossRef]
- Komaki, M.; Iwasaki, K.; Arzate, H.; Narayanan, A.S.; Izumi, Y.; Morita, I. Cementum protein 1 (CEMP1) induces a cementoblastic phenotype and reduces osteoblastic differentiation in periodontal ligament cells. J. Cell. Physiol. 2012, 227, 649–657. [Google Scholar] [CrossRef] [PubMed]
- Gay, I.C.; Chen, S.; MacDougall, M. Isolation and characterization of multipotent human periodontal ligament stem cells. Orthod. Craniofac. Res. 2007, 10, 149–160. [Google Scholar] [CrossRef] [PubMed]
- Flores, M.G.; Yashiro, R.; Washio, K.; Yamato, M.; Okano, T.; Ishikawa, I. Periodontal ligament cell sheet promotes periodontal regeneration in athymic rats. J. Clin. Periodontol. 2008, 35, 1066–1072. [Google Scholar] [CrossRef] [PubMed]
- Zeichner-David, M.; Oishi, K.; Su, Z.; Zakartchenko, V.; Chen, L.S.; Arzate, H.; Bringas, P., Jr. Role of Hertwig’s epithelial root sheath cells in tooth root development. Dev. Dyn. Off. Publ. Am. Assoc. Anat. 2003, 228, 651–663. [Google Scholar] [CrossRef] [PubMed]
| Gene | Primer Sequence | Size (bp) | GenBank Accession No. |
|---|---|---|---|
| ALP | F 5′-TATGGTAACGGGCCTGGCTAC-3′ R 5′-TGCTCATGGACGCCGTGAAGCA-3′ | 187 | NM_007431.2 |
| BSP | F 5′-TGAACAGACTCCGGCGCTAC-3′ R 5′-AGGGCAGCACAGGTCCTAA-3′ | 127 | NM_008318.3 |
| DMP1 | F 5′-AGAGGGACAGGCAAATAGTGAC-3′ R 5′-CATTGTCCTCATCGCCAAAGG-3′ | 176 | NM_016779.2 |
| DSPP | F 5′-GGAAGAGCCAAGATCAGGGAA-3′ R 5′-GCCTTGAGGCTTGTCAGACT-3′ | 173 | NM_010080.2 |
| OPN | F 5′-GCCGAGGTGATAGCTTGGCT-3′ R 5′-TGATCAGAGGGCATGCTCAG-3′ | 177 | NM_001204201.1 |
| RUNX2 | F 5′-CCAGGCAGGTGCTTCAGAACTG-3′ R 5′-ACATGCCGAGGGACATGCCTGA-3′ | 157 | NM_009820.5 |
| β-actin | F 5′-GATCTGGCACCACACCTTCT-3′ R 5′-GGGGTGTTGAAGGTCTCAAA-3′ | 138 | NM_007393.3 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moon, J.-S.; Kim, S.-D.; Ko, H.-M.; Kim, Y.-J.; Kim, S.-H.; Kim, M.-S. Twist1 Suppresses Cementoblast Differentiation. Dent. J. 2018, 6, 57. https://doi.org/10.3390/dj6040057
Moon J-S, Kim S-D, Ko H-M, Kim Y-J, Kim S-H, Kim M-S. Twist1 Suppresses Cementoblast Differentiation. Dentistry Journal. 2018; 6(4):57. https://doi.org/10.3390/dj6040057
Chicago/Turabian StyleMoon, Jung-Sun, Seong-Duk Kim, Hyun-Mi Ko, Young-Jun Kim, Sun-Hun Kim, and Min-Seok Kim. 2018. "Twist1 Suppresses Cementoblast Differentiation" Dentistry Journal 6, no. 4: 57. https://doi.org/10.3390/dj6040057
APA StyleMoon, J.-S., Kim, S.-D., Ko, H.-M., Kim, Y.-J., Kim, S.-H., & Kim, M.-S. (2018). Twist1 Suppresses Cementoblast Differentiation. Dentistry Journal, 6(4), 57. https://doi.org/10.3390/dj6040057





