Meth Mouth—A Growing Epidemic in Dentistry?
Abstract
:1. Introduction
2. “Crystal Meth”
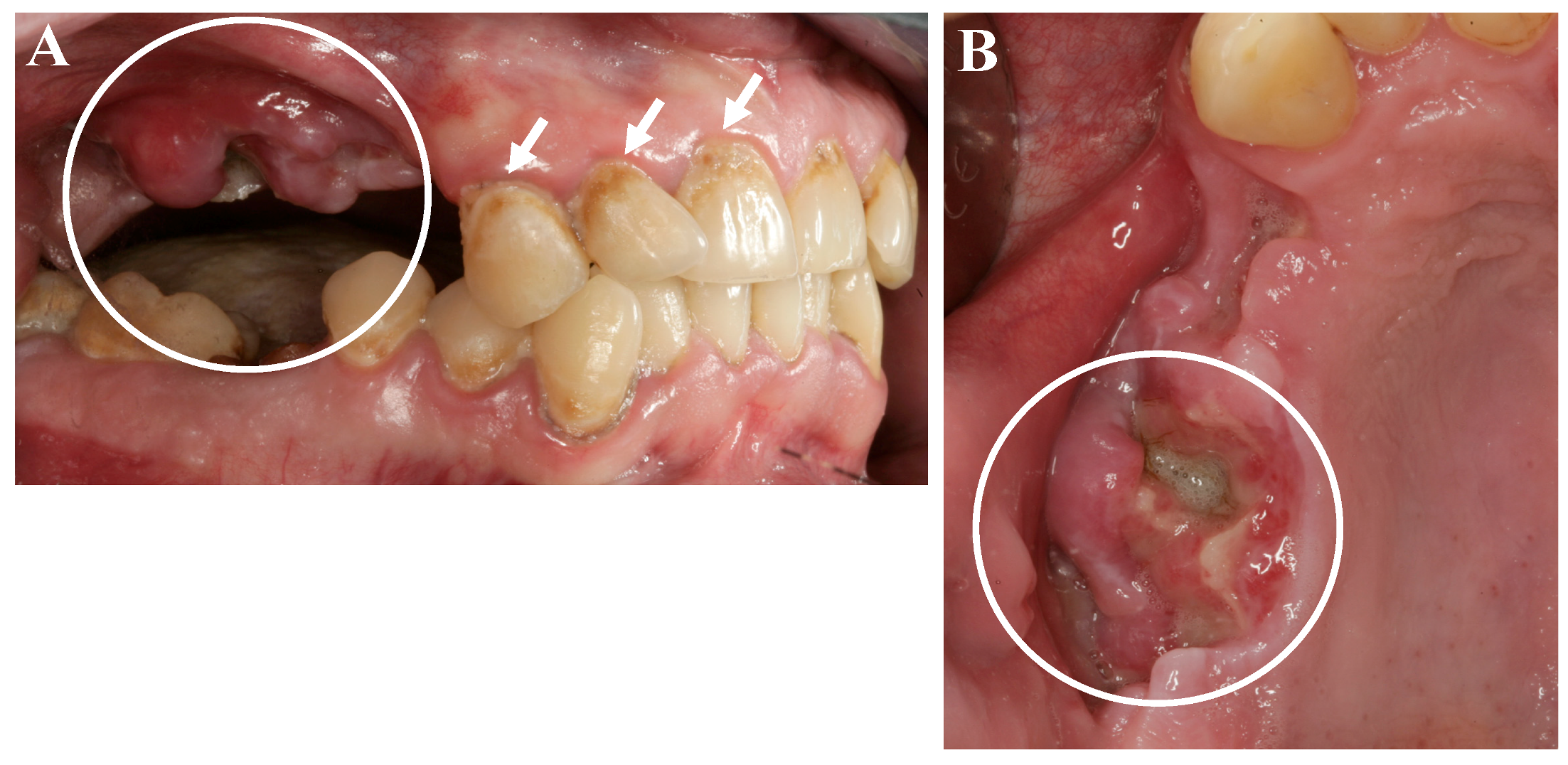
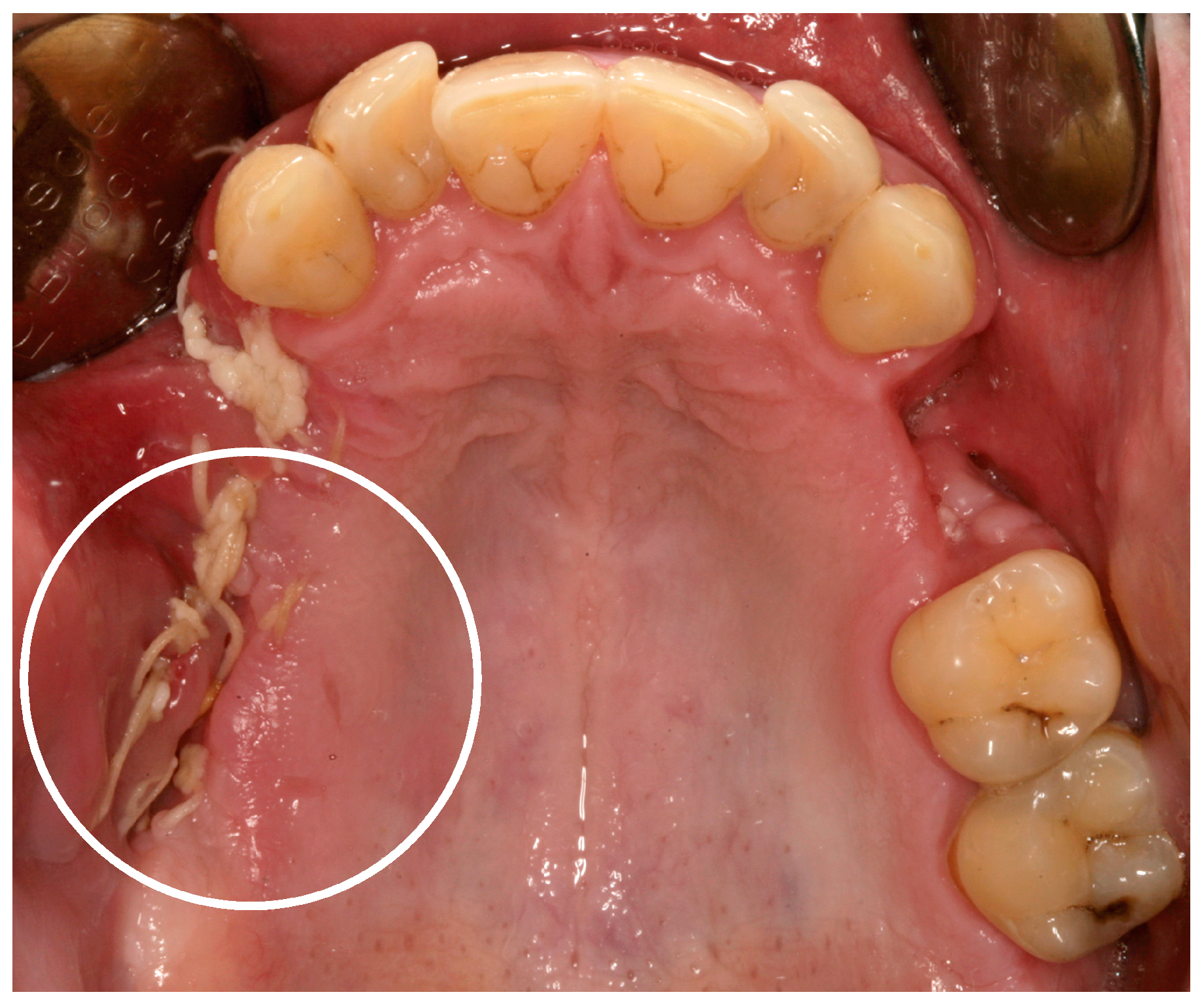
3. “Meth Mouth” and Jaw Necrosis
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- De-Carolis, C.; Boyd, G.A.; Mancinelli, L.; Pagano, S.; Eramo, S. Methamphetamine abuse and “meth mouth” in Europe. Med. Oral Patol. Oral Cir. Bucal 2015, 20, 205–210. [Google Scholar] [CrossRef]
- Rommel, N.; Rohleder, N.H.; Koerdt, S.; Wagenpfeil, S.; Härtel-Petri, R.; Wolff, K.D.; Kesting, M.R. Sympathomimetic effects of chronic methamphetamine abuse on oral health: A cross-sectional study. BMC Oral Health 2016, 16, 59. [Google Scholar] [CrossRef] [PubMed]
- Calipari, E.S.; Ferris, M.J. Amphetamine Mechanisms and Actions at the Dopamine Terminal Revisited. J. Neurosci. 2013, 33, 8923–8925. [Google Scholar] [CrossRef] [PubMed]
- Mattson, M.E. Emergency Department Visits Involving Methamphetamine: 2007 to 2011; The CBHSQ Report; Substance Abuse and Mental Health Services Administration (US): Rockville, MD, USA, 2014. [Google Scholar]
- Basin, E.M.; Medvedev, Y.A.; Serova, N.S.; Satueva, D.B.; Korshunova, A.V.; Babkova, A.A.; Kureshova, D.V. “KROKODIL”—Associated jaw osteonecrosis as new era of phossy jaw. In Proceedings of the XXII Congress of the European Association for Cranio-Maxillo-Facial Surgery (EACMFS), Prague, Czech Republic, 23–26 September 2014. [Google Scholar]
- Rhodus, N.L.; Little, J.W. Methamphetamine abuse and “meth mouth”. Northwest Dent. 2005, 84, 29–31. [Google Scholar] [PubMed]
- Poghosyan, Y.M.; Hakobyan, K.A.; Poghosyan, A.Y.; Avetisyan, E.K. Surgical treatment of jaw osteonecrosis in “Krokodil” drug addicted patients. J. Cranio-Maxillofac. Surg. 2014, 42, 1639–1643. [Google Scholar] [CrossRef] [PubMed]
- Hakobyan, K.; Poghosyan, Y. Spontaneous bone formation after mandible segmental resection in “krokodil” drug-related jaw osteonecrosis patient: Case report. Oral Maxillofac. Surg. 2017, 21, 267–270. [Google Scholar] [CrossRef] [PubMed]
- Pabst, A.M.; Werkmeister, R. Amphetamin-induzierte Osteonekrose des Oberkiefers. Zahnärztliche Mitteilungen 2016, 2016, 56–58. [Google Scholar]
- Van Poznak, C. Osteonecrosis of the jaw and bevacizumab therapy. Breast Cancer Res. Treat. 2010, 122, 189–191. [Google Scholar] [CrossRef] [PubMed]
- Koch, F.P.; Walter, C.; Hansen, T.; Jäger, E.; Wagner, W. Osteonecrosis of the jaw related to sunitinib. Oral Maxillofac. Surg. 2011, 15, 63–66. [Google Scholar] [CrossRef] [PubMed]
- Pichardo, S.E.; Kuypers, S.C.; van Merkesteyn, J.P. Denosumab osteonecrosis of the mandible: A new entity? A case report. J. Cranio-Maxillofac. Surg. 2013, 41, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Alsalleeh, F.; Keippel, J.; Adams, L.; Bavitz, B. Bisphosphonate-associated osteonecrosis of jaw reoccurrence after methotrexate therapy: A case report. J. Endod. 2014, 40, 1505–1507. [Google Scholar] [CrossRef] [PubMed]
- Assaf, A.T.; Zrnc, T.A.; Remus, C.C.; Adam, G.; Zustin, J.; Heiland, M.; Friedrich, R.E.; Derlin, T. Intraindividual comparison of preoperative 99mTc-MDP SPECT/CT and intraoperative and histopathological findings in patients with bisphosphonate- or denosumab- related osteonecrosis of the jaw. J. Cranio-Maxillofac. Surg. 2015, 43, 1461–1469. [Google Scholar] [CrossRef] [PubMed]
- Baur, D.A.; Altay, M.A.; Teich, S.; Schmitt Oswald, M.; Quereshy, F.A. Osteonecrosis of the jaw in a patient on raloxifene: A case report. Quintessence Int. 2015, 46, 423–428. [Google Scholar] [PubMed]
- Owosho, A.A.; Blanchard, A.; Levi, L.; Kadempour, A.; Rosenberg, H.; Yom, S.K.; Farooki, A.; Fornier, M.; Huryn, J.M.; Estilo, C.L. Osteonecrosis of the jaw in patients treated with denosumab for metastatic tumors to the bone: A series of thirteen patients. J. Cranio-Maxillofac. Surg. 2016, 44, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Pabst, A.M.; Krüger, M.; Ziebart, T.; Jacobs, C.; Sagheb, K.; Walter, C. The influence of geranylgeraniol on human oral keratinocytes after bisphosphonate treatment: An in vitro study. J. Cranio-Maxillofac. Surg. 2015, 43, 688–695. [Google Scholar] [CrossRef] [PubMed]
- Hoefert, S.; Yuan, A.; Munz, A.; Grimm, M.; Elayouti, A.; Reinert, S. Clinical course and therapeutic outcomes of operatively and non-operatively managed patientswith denosumab-related osteonecrosis of the jaw (DRONJ). J. Cranio-Maxillofac. Surg. 2017, 45, 570–578. [Google Scholar] [CrossRef] [PubMed]
- Ravenel, M.C.; Salinas, C.F.; Marlow, N.M.; Slate, E.H.; Evans, Z.P.; Miller, P.M. Methamphetamine abuse and oral health: A pilot study of “meth mouth”. Quintessence Int. 2012, 43, 229–237. [Google Scholar] [PubMed]
- Walter, C.; Klein, M.O.; Pabst, A.; Al-Nawas, B.; Duschner, H.; Ziebart, T. Influence of bisphosphonates on endothelial cells, fibroblasts, and osteogenic cells. Clin. Oral Investig. 2010, 14, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Walter, C.; Pabst, A.; Ziebart, T.; Klein, M.O.; Al-Nawas, B. Bisphosphonates affect migration ability and cell viability of HUVEC, fibroblasts and osteoblasts in-vitro. Oral Dis. 2011, 17, 194–199. [Google Scholar] [CrossRef] [PubMed]
- Pabst, A.M.; Ziebart, T.; Koch, F.P.; Taylor, K.Y.; Al-Nawas, B.; Walter, C. The influence of bisphosphonates on viability, migration, and apoptosis of human oral keratinocytes-in vitro study. Clin. Oral Investig. 2012, 16, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Pabst, A.M.; Ziebart, T.; Ackermann, M.; Konerding, M.A.; Walter, C. Bisphosphonates’ antiangiogenic potency in the development of bisphosphonate-associated osteonecrosis of the jaws: Influence on microvessel sprouting in an in vivo 3D Matrigel assay. Clin. Oral Investig. 2014, 18, 1015–1022. [Google Scholar] [CrossRef] [PubMed]
- Haskin, A.; Kim, N.; Aguh, C. A new drug with a nasty bite: A case of krokodil-induced skin necrosis in an intravenous drug user. JAAD Case Rep. 2016, 2, 174–176. [Google Scholar] [CrossRef] [PubMed]
- Escribano, A.B.; Negre, M.T.B.; Orenga, G.C.; Monfort, S.C.; Peiró, F.A.; Zapatero, S.M.; Cortés, G.H. Orally ingestion of krokodil in Spain: Report of a case. Adicciones 2016, 28, 242–245. [Google Scholar]
- Rustemeyer, J.; Melenberg, A.; Junker, K.; Sari-Rieger, A. Osteonecrosis of the maxilla related to long-standing methamphetamine abuse: A possible new aspect in the etiology of osteonecrosis of the jaw. Oral Maxillofac. Surg. 2014, 18, 237–241. [Google Scholar] [CrossRef] [PubMed]
- Marx, R.E. Bisphosphonat-induzierte Osteonekrose der Kiefer. In Ätiologie, Prävention, Behandlung; Quintessenz Verlags-GmbH: Berlin, Germany, 2013; pp. 6–7. [Google Scholar]
- Rommel, N.; Rohleder, N.H.; Wagenpfeil, S.; Haertel-Petri, R.; Kesting, M.R. Evaluation of methamphetamine-associated socioeconomic status and addictive behaviors, and their impact on oral health. Addict. Behav. 2015, 50, 182–187. [Google Scholar] [CrossRef] [PubMed]
- Rommel, N.; Rohleder, N.H.; Wagenpfeil, S.; Härtel-Petri, R.; Jacob, F.; Wolff, K.D.; Kesting, M.R. The impact of the new scene drug “crystal meth” on oral health: A case-control study. Clin. Oral Investig. 2016, 20, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Hamamoto, D.T.; Rhodus, N.L. Methamphetamine abuse and dentistry. Oral Dis. 2009, 15, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Chen, X.; Zheng, L.; Guo, L.; Li, X.; Shen, S. Comprehensive dental treatment for “meth mouth”: A case report and literature review. J. Formos. Med. Assoc. 2014, 113, 867–871. [Google Scholar] [CrossRef] [PubMed]
- Smit, D.A.; Naidoo, S. Oral health effects, brushing habits and management of methamphetamine users for the general dental practitioner. Br. Dent. J. 2015, 218, 531–536. [Google Scholar] [CrossRef] [PubMed]
- Ristow, O.; Otto, S.; Troeltzsch, M.; Hohlweg-Majert, B.; Pautke, C. Treament perspectives for medication-related osteonecrosis of the jaw (MR-ONJ). J. Cranio-Maxillofac. Surg. 2015, 43, 290–293. [Google Scholar] [CrossRef] [PubMed]
- Faucett, E.A.; Marsh, K.M.; Farshad, K.; Erman, A.B.; Chiu, A.G. Maxillary sinus manifestations of methamphetamine abuse. Allergy Rhinol. 2015, 6, 76–79. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pabst, A.; Castillo-Duque, J.C.; Mayer, A.; Klinghuber, M.; Werkmeister, R. Meth Mouth—A Growing Epidemic in Dentistry? Dent. J. 2017, 5, 29. https://doi.org/10.3390/dj5040029
Pabst A, Castillo-Duque JC, Mayer A, Klinghuber M, Werkmeister R. Meth Mouth—A Growing Epidemic in Dentistry? Dentistry Journal. 2017; 5(4):29. https://doi.org/10.3390/dj5040029
Chicago/Turabian StylePabst, Andreas, Juan Carlos Castillo-Duque, Axel Mayer, Marcus Klinghuber, and Richard Werkmeister. 2017. "Meth Mouth—A Growing Epidemic in Dentistry?" Dentistry Journal 5, no. 4: 29. https://doi.org/10.3390/dj5040029
APA StylePabst, A., Castillo-Duque, J. C., Mayer, A., Klinghuber, M., & Werkmeister, R. (2017). Meth Mouth—A Growing Epidemic in Dentistry? Dentistry Journal, 5(4), 29. https://doi.org/10.3390/dj5040029



